Chronic Stress and Cardiovascular Disease among Individuals Exposed to Lead: A Pilot Study
Abstract
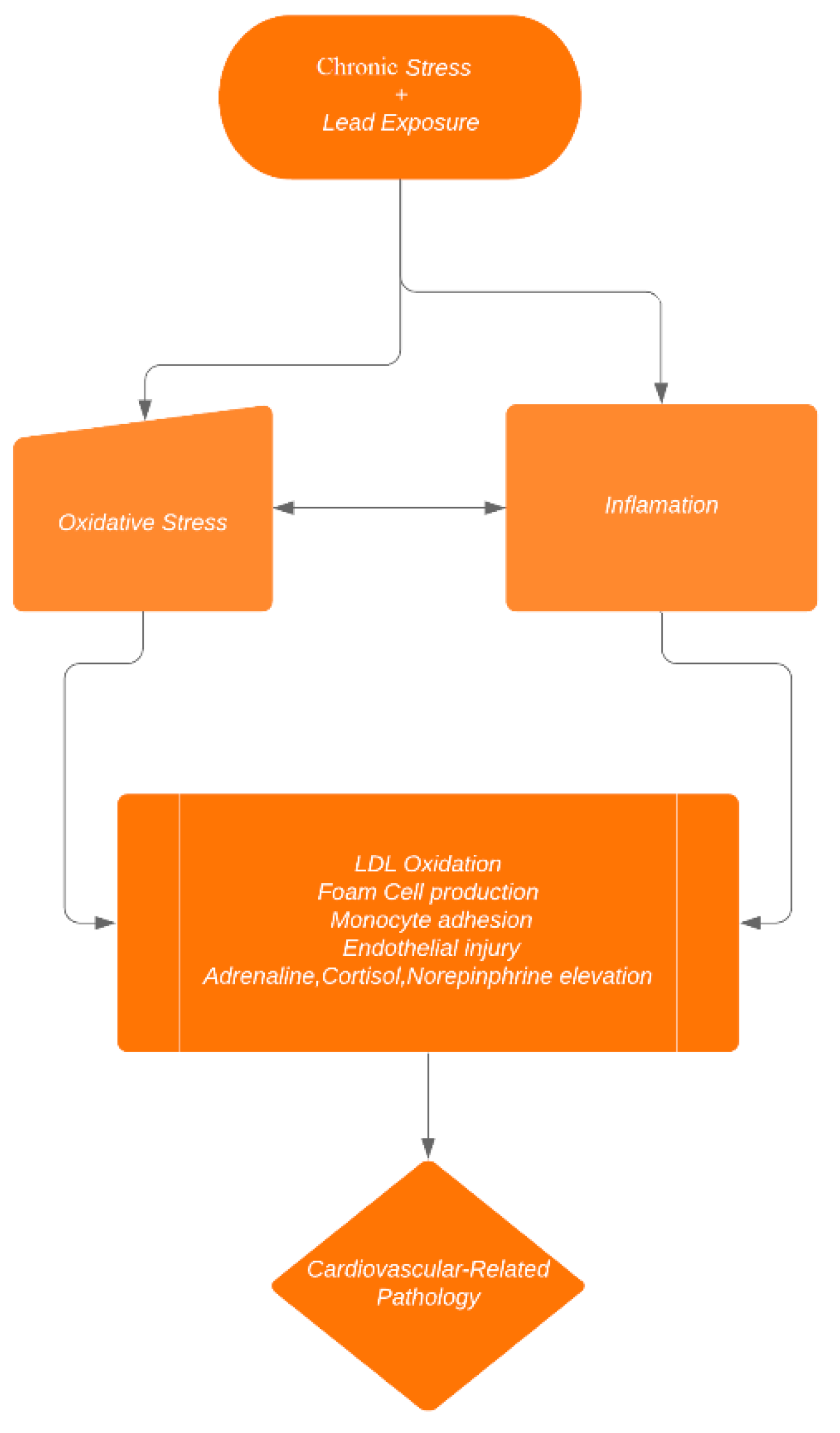
1. Introduction
2. Materials and Methods
2.1. Hypothesis
2.2. Research Design
2.2.1. Operationalising Allostatic Load
2.2.2. Data collection in NHANES
2.3. Data Analysis
3. Results
3.1. Study Variables Among Lead-Exposed and Less Lead-Exposed Participant
3.2. Geometric Mean GGT and Non-HDL-C in Lead-Exposed Individuals with High/Low Allostatic Load
3.3. Likelihood of Elevated Clinical Markers at AL Binary at 4 in Individuals Exposed to Lead
3.4. Association of AL with Markers of Interest in Lead-Exposed Individuals
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Meyer, P.A.; Brown, M.J.; Falk, H. Global approach to reducing lead exposure and poisoning. Mutat. Res. Rev. Mutat. Res. 2008, 659, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Gyasi, E. Sources of lead exposure in various countries. Rev. Environ. Health 2019, 34, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Téllez-Rojo, M.M.; Bellinger, D.; Smith, D.; Ettinger, A.S.; Lamadrid-Figueroa, H.; Schwartz, J.; Schnaas, L.; Mercado-García, A.; Hernández-Avila, M. Fetal lead exposure at each stage of pregnancy as a predictor of infant mental development. Environ. Health Perspect. 2006, 114, 1730–1735. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Gyasi, E. Lead Exposure and Cardiovascular Disease among Young and Middle-Aged Adults. Med Sci. 2019, 7, 103. [Google Scholar] [CrossRef]
- Loghman-Adham, M. Renal effects of environmental and occupational lead exposure. Environ. Health Perspect. 1997, 105, 928–939. [Google Scholar] [CrossRef]
- Reuben, A.; Schaefer, J.D.; Moffitt, T.E.; Broadbent, J.; Harrington, H.; Houts, R.M.; Ramrakha, S.; Poulton, R.; Caspi, A. Association of childhood lead exposure with adult personality traits and lifelong mental health. JAMA Psychiatry 2019, 76, 418–425. [Google Scholar] [CrossRef]
- Telišman, S.; Čolak, B.; Pizent, A.; Jurasović, J.; Cvitković, P. Reproductive toxicity of low-Level lead exposure in men. Environ. Res. 2007, 105, 256–266. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Guallar, E.; Silbergeld, E.K.; Rothenberg, S.J. Lead exposure and cardiovascular disease: A systematic review. Environ. Health Perspect. 2007, 472–482. [Google Scholar] [CrossRef]
- Patil, A.J.; Bhagwat, V.R.; Patil, J.A.; Dongre, N.N.; Ambekar, J.G.; Das, K.K. Occupational lead exposure in battery manufacturing workers, silver jewelry workers, and spray painters in western Maharashtra (India): Effect on liver and kidney function. J. Basic Clin. Physiol. Pharmacol. 2007, 18, 87–100. [Google Scholar] [CrossRef]
- Obeng-Gyasi, E. Lead Exposure and Oxidative Stress-A Life Course Approach in US Adults. Toxics 2018, 6, 42. [Google Scholar] [CrossRef]
- Reuben, A.; Caspi, A.; Belsky, D.W.; Broadbent, J.; Harrington, H.; Sugden, K.; Houts, R.M.; Ramrakha, S.; Poulton, R.; Moffitt, T.E. Association of childhood blood lead levels with cognitive function and socioeconomic status at age 38 years and with IQ change and socioeconomic mobility between childhood and adulthood. JAMA 2017, 317, 1244–1251. [Google Scholar] [CrossRef] [PubMed]
- Joshu, C.E.; Boehmer, T.; Brownson, R.; Ewing, R. Personal, neighbourhood and urban factors associated with obesity in the United States. J. Epidemiol. Community Health 2008, 62, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.L.; Kubzansky, L.; McNeely, E.; Schwartz, J.; Spiro III, A.; Sparrow, D.; Wright, R.O.; Nie, H.; Hu, H. Stress as a potential modifier of the impact of lead levels on blood pressure: The normative aging study. Environ. Health Perspect. 2007, 115, 1154–1159. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Protective and damaging effects of stress mediators. New Engl. J. Med. 1998, 338, 171–179. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Protection and damage from acute and chronic stress: Allostasis and allostatic overload and relevance to the pathophysiology of psychiatric disorders. Ann. N. Y. Acad. Sci. 2004, 1032, 1–7. [Google Scholar] [CrossRef]
- McEwen, B.S. The neurobiology of stress: From serendipity to clinical relevance. Brain Res. 2000, 886, 172–189. [Google Scholar] [CrossRef]
- Ganzel, B.L.; Morris, P.A.; Wethington, E. Allostasis and the human brain: Integrating models of stress from the social and life sciences. Psychol. Rev. 2010, 117, 134. [Google Scholar] [CrossRef]
- Juster, R.-P.; McEwen, B.S.; Lupien, S.J. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci. Biobehav. Rev. 2010, 35, 2–16. [Google Scholar] [CrossRef]
- McEwen, B.S. Protective and damaging effects of stress mediators: Central role of the brain. Dialogues Clin. Neurosci. 2006, 8, 367. [Google Scholar]
- Organization, W.H. 2016.
- Hu, H.; Aro, A.; Payton, M.; Korrick, S.; Sparrow, D.; Weiss, S.T.; Rotnitzky, A. The relationship of bone and blood lead to hypertension: The Normative Aging Study. JAMA 1996, 275, 1171–1176. [Google Scholar] [CrossRef]
- Nash, D.; Magder, L.; Lustberg, M.; Sherwin, R.W.; Rubin, R.J.; Kaufmann, R.B.; Silbergeld, E.K. Blood lead, blood pressure, and hypertension in perimenopausal and postmenopausal women. JAMA 2003, 289, 1523–1532. [Google Scholar] [CrossRef]
- Hertz-Picciotto, I.; Croft, J. Review of the relation between blood lead and blood pressure. Epidemiol. Rev. 1993, 15, 352–373. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, T.; Thijs, L.; Den Hond, E.; Roels, H.; Staessen, J.A. An epidemiological re-Appraisal of the association between blood pressure and blood lead: A meta-Analysis. J. Hum. Hypertens. 2002, 16, 123. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Gyasi, E.; Armijos, R.X.; Weigel, M.M.; Filippelli, G.M.; Sayegh, M.A. Cardiovascular-Related Outcomes in US Adults Exposed to Lead. Int. J. Environ. Res. Public Health 2018, 15, 759. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Resnick, H.E.; Jablonski, K.A.; Jones, K.L.; Jain, A.K.; Howard, W.J.; Robbins, D.C.; Howard, B.V. Non-HDL cholesterol as a predictor of cardiovascular disease in type 2 diabetes: The strong heart study. Diabetes Care 2003, 26, 16–23. [Google Scholar] [CrossRef]
- Blaha, M.J.; Blumenthal, R.S.; Brinton, E.A.; Jacobson, T.A.; National Lipid Association Taskforce on Non-HDL Cholesterol. The importance of non–HDL cholesterol reporting in lipid management. J. Clin. Lipidol. 2008, 2, 267–273. [Google Scholar] [CrossRef]
- Nantsupawat, N.; Booncharoen, A.; Wisetborisut, A.; Jiraporncharoen, W.; Pinyopornpanish, K.; Chutarattanakul, L.; Angkurawaranon, C. Appropriate Total cholesterol cut-Offs for detection of abnormal LDL cholesterol and non-HDL cholesterol among low cardiovascular risk population. Lipids Health Dis. 2019, 18, 28. [Google Scholar] [CrossRef]
- Lee, D.-H.; Blomhoff, R.; Jacobs, D.R. Review is serum gamma glutamyltransferase a marker of oxidative stress? Free Radic. Res. 2004, 38, 535–539. [Google Scholar] [CrossRef]
- Lim, J.-S.; Yang, J.-H.; Chun, B.-Y.; Kam, S.; Jacobs Jr, D.R.; Lee, D.-H. Is serum γ-Glutamyltransferase inversely associated with serum antioxidants as a marker of oxidative stress? Free Radic. Biol. Med. 2004, 37, 1018–1023. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014. [Google Scholar] [CrossRef]
- Steptoe, A.; Kivimäki, M. Stress and cardiovascular disease. Nat. Rev. Cardiol. 2012, 9, 360. [Google Scholar] [CrossRef] [PubMed]
- Reference Blood Lead Level for Adults. Available online: https://www.cdc.gov/niosh/topics/ables/description.html (accessed on 14 March 2020).
- Sabbah, W.; Watt, R.; Sheiham, A.; Tsakos, G. Effects of allostatic load on the social gradient in ischaemic heart disease and periodontal disease: Evidence from the Third National Health and Nutrition Examination Survey. J. Epidemiol. Community Health 2008, 62, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Goldbourt, U.; Yaari, S.; Medalie, J.H. Isolated low HDL cholesterol as a risk factor for coronary heart disease mortality: A 21-Year follow-Up of 8000 men. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Mahon, N.G.; Blackstone, E.H.; Francis, G.S.; Starling, R.C.; Young, J.B.; Lauer, M.S. The prognostic value of estimated creatinine clearance alongside functional capacity in ambulatory patients with chronic congestive heart failure. J. Am. Coll. Cardiol. 2002, 40, 1106–1113. [Google Scholar] [CrossRef]
- Horwich, T.B.; Kalantar-Zadeh, K.; MacLellan, R.W.; Fonarow, G.C. Albumin levels predict survival in patients with systolic heart failure. Am. Heart J. 2008, 155, 883–889. [Google Scholar] [CrossRef]
- Ackermann, R.T.; Cheng, Y.J.; Williamson, D.F.; Gregg, E.W. Identifying adults at high risk for diabetes and cardiovascular disease using hemoglobin A1c: National Health and Nutrition Examination Survey 2005–2006. Am. J. Prev. Med. 2011, 40, 11–17. [Google Scholar] [CrossRef]
- Isomaa, B.; Almgren, P.; Tuomi, T.; Forsén, B.; Lahti, K.; Nissen, M.; Taskinen, M.-R.; Groop, L. Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 2001, 24, 683–689. [Google Scholar] [CrossRef]
- Weverling-Rijnsburger, A.W.; Blauw, G.J.; Lagaay, A.M.; Knock, D.L.; Meinders, A.E.; Westendorp, R.G. Total cholesterol and risk of mortality in the oldest old. Lancet 1997, 350, 1119–1123. [Google Scholar] [CrossRef]
- Selevan, S.G.; Rice, D.C.; Hogan, K.A.; Euling, S.Y.; Pfahles-Hutchens, A.; Bethel, J. Blood lead concentration and delayed puberty in girls. New Engl. J. Med. 2003, 348, 1527–1536. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, E.-A.; Koh, D.-H.; Byun, K.; Ryu, H.-W.; Lee, S.-G. Blood lead levels of Korean lead workers in 2003–2011. Ann. Occup. Environ. Med. 2014, 26, 30. [Google Scholar] [CrossRef]
- Canfield, R.L.; Kreher, D.A.; Cornwell, C.; Henderson, C.R. Low-Level lead exposure, executive functioning, and learning in early childhood. Child Neuropsychol. 2003, 9, 35–53. [Google Scholar] [CrossRef] [PubMed]
- Lanphear, B.P.; Rauch, S.; Auinger, P.; Allen, R.W.; Hornung, R.W. Low-Level lead exposure and mortality in US adults: A population-Based cohort study. Lancet Public Health 2018, 3, e177–e184. [Google Scholar] [CrossRef]
- Mehta, G.; Macdonald, S.; Cronberg, A.; Rosselli, M.; Khera-Butler, T.; Sumpter, C.; Al-Khatib, S.; Jain, A.; Maurice, J.; Charalambous, C. Short-Term abstinence from alcohol and changes in cardiovascular risk factors, liver function tests and cancer-related growth factors: A prospective observational study. BMJ Open 2018, 8, e020673. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; De Ferranti, S.; Després, J.P.; Fullerton, H.J. Heart disease and stroke statistics-2016 update a report from the American Heart Association. Circulation 2016, 133, e38–e48. [Google Scholar] [PubMed]
- McCrindle, B.W. Cardiovascular consequences of childhood obesity. Can. J. Cardiol. 2015, 31, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Safford, M.M.; Brown, T.M.; Muntner, P.M.; Durant, R.W.; Glasser, S.; Halanych, J.H.; Shikany, J.M.; Prineas, R.J.; Samdarshi, T.; Bittner, V.A. Association of race and sex with risk of incident acute coronary heart disease events. JAMA 2012, 308, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Geronimus, A.T.; Hicken, M.; Keene, D.; Bound, J. “Weathering” and age patterns of allostatic load scores among blacks and whites in the United States. Am. J. Public Health 2006, 96, 826–833. [Google Scholar] [CrossRef]
- Crimmins, E.M.; Johnston, M.; Hayward, M.; Seeman, T. Age differences in allostatic load: An index of physiological dysregulation. Exp. Gerontol. 2003, 38, 731–734. [Google Scholar] [CrossRef]
- Kaestner, R.; Pearson, J.A.; Keene, D.; Geronimus, A.T. Stress, allostatic load, and health of Mexican immigrants. Soc. Sci. Q. 2009, 90, 1089–1111. [Google Scholar] [CrossRef]
- Lai, Z.; Bonilla, G.; Diaz, I.; Nery, J.G.; Sujaoti, K.; Amat, M.A.; Kokkoli, E.; Terasaki, O.; Thompson, R.W.; Tsapatsis, M.; et al. Microstructural Optimization of a Zeolite Membrane for Organic Vapor Separation. Science 2003, 300, 456–460. [Google Scholar] [CrossRef]
- Seeman, T.; Merkin, S.S.; Crimmins, E.; Koretz, B.; Charette, S.; Karlamangla, A. Education, income and ethnic differences in cumulative biological risk profiles in a national sample of US adults: NHANES III (1988–1994). Soc. Sci. Med. 2008, 66, 72–87. [Google Scholar] [CrossRef] [PubMed]
- Geronimus, A.T. The weathering hypothesis and the health of African-American women and infants: Evidence and speculations. Ethn. Dis. 1992, 2, 207–221. [Google Scholar] [PubMed]
- McEwen, B.S.; Seeman, T. Protective and damaging effects of mediators of stress: Elaborating and testing the concepts of allostasis and allostatic load. Ann. N. Y. Acad. Sci. 1999, 896, 30–47. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, K.N.; Masten, V.L.; Tiburzi, M.J.; Ford, D.P.; Bleecker, M.L. The factor structure of the Profile of Mood States (POMS) and its relationship to occupational lead exposure. J. Occup. Environ. Med. 1999, 41, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, D.; Spiro III, A.; Aro, A.; Hu, H. Relationship of bone and blood lead levels to psychiatric symptoms: The normative aging study. J. Occup. Environ. Med. 2003, 45, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Seeman, T.E.; Singer, B.H.; Rowe, J.W.; Horwitz, R.I.; McEwen, B.S. Price of adaptation—Allostatic load and its health consequences: MacArthur studies of successful aging. Arch. Intern. Med. 1997, 157, 2259–2268. [Google Scholar] [CrossRef]
| Variable | Lead-Exposed (BLL ≥ 5 µg/dL) | Less Lead-Exposed (BLL < 5 µg/dL) | p-Value |
|---|---|---|---|
| N | 255 | 9526 | |
| Mean Age (95% CI) | 49.04 (46.95–51.15) | 38.27 (37.70–39.05) | <0.001 |
| Allostatic Load by Ethnicity (95% CI) | |||
| Non-Hispanic Black | 2.94 (2.66–3.23) | 2.24 (2.14–2.34) | <0.001 |
| Non-Hispanic White | 2.67 (2.42–2.92) | 2.43 (2.35–2.52) | 0.046 |
| Allostatic Load by Gender (95% CI) | |||
| Male | 2.68 (2.42–2.93) | 2.39 (2.33–2.46) | 0.032 |
| Female | 2.31 (1.64–2.98) | 2.35 (2.27–2.44) | 0.885 |
| Non-HDL-C (95% CI) | 152.12 (143.36–160.88) | 139.31 (138.08–140.54) | 0.006 |
| GGT (95% CI) | 39.34 (31.22–47.46) | 26.12 (25.24–27.01) | <0.001 |
| Smoking every day percent (95% CI) | 52.09 (45.79–58.33) | 38.88 (35.89–41.96) | 0.001 |
| Alcohol percent (95% CI) | 84.96 (77.90–90.05) | 75.81 (73.57–77.93) | 0.007 |
| Variable | Lead-Exposed (BLL ≥ 5 µg/dL) | Less Lead-Exposed (BLL < 5 µg/dL) |
|---|---|---|
| Non-HDL-C (95% CI) (High AL) | 167.48 (155.53–179.43) | 163.71 (161.78–164.64) |
| Non-HDL-C (95% CI) (Low AL) | 144. 77 (135.34–154.20) | 131.82 (130.63–133.02) |
| GGT (95% CI) (High AL) | 49.51 (30.73–68.30) | 33.33 (31.36–35.31) |
| GGT (95% CI) (Low AL) | 34.77 (27.12–42.43) | 23.92 (23.02–24.84) |
| Variables | lnAL Adjusted (95% CI) + | p-Value |
|---|---|---|
| GGT (LE) | 0.30 (−0.12, 0.72) | 0.148 |
| nonHDL-C (LE) | 0.79 (−0.13–1.70) | 0.088 |
| GGT (LLE) | 0.10 (0.03, 0.17) | 0.009 |
| nonHDL-C (LLE) | 0.60 (0.50–0.71) | <0.001 |
| Variables | lnAL Adjusted (95% CI) + | p-Value |
|---|---|---|
| GGT (LE) | 0.13 (0.01, 0.25) | 0.036 |
| nonHDL-C (LE) | 0.73 (0.48–0.97) | <0.001 |
| GGT (LLE) | 0.10 (0.02, 0.18) | 0.013 |
| nonHDL-C (LLE) | 0.596 (0.48–0.71) | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obeng-Gyasi, E.; Obeng-Gyasi, B. Chronic Stress and Cardiovascular Disease among Individuals Exposed to Lead: A Pilot Study. Diseases 2020, 8, 7. https://doi.org/10.3390/diseases8010007
Obeng-Gyasi E, Obeng-Gyasi B. Chronic Stress and Cardiovascular Disease among Individuals Exposed to Lead: A Pilot Study. Diseases. 2020; 8(1):7. https://doi.org/10.3390/diseases8010007
Chicago/Turabian StyleObeng-Gyasi, Emmanuel, and Barnabas Obeng-Gyasi. 2020. "Chronic Stress and Cardiovascular Disease among Individuals Exposed to Lead: A Pilot Study" Diseases 8, no. 1: 7. https://doi.org/10.3390/diseases8010007
APA StyleObeng-Gyasi, E., & Obeng-Gyasi, B. (2020). Chronic Stress and Cardiovascular Disease among Individuals Exposed to Lead: A Pilot Study. Diseases, 8(1), 7. https://doi.org/10.3390/diseases8010007






