Association of HCV Infection with C-Reactive Protein: National Health and Nutrition Examination Survey (NHANES), 2009–2010
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Measurements
2.2.1. C-Reactive Protein (CRP)
2.2.2. Hepatitis-C Virus (HCV)
2.2.3. Covariates
2.2.4. Statistical Analyses
3. Results
3.1. Sample Characteristics
3.2. Bivariate Analyses
3.2.1. Mean CRP Levels among Participants
3.2.2. CRP Levels with Risk Factors
3.3. CRP levels by HCV-RNA
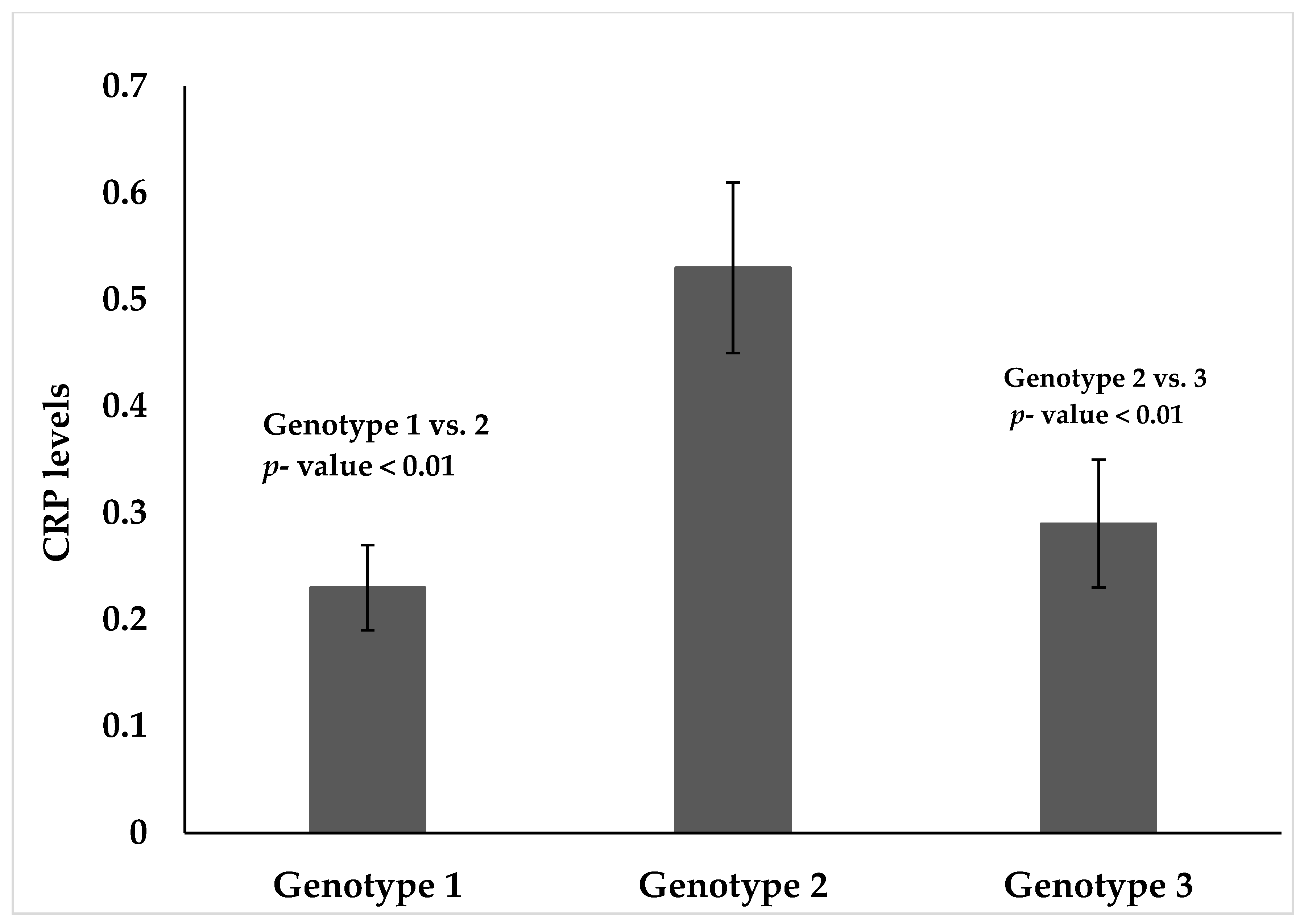
3.4. CRP Levels by HCV Genotype
4. Discussion
5. Strengths
6. Limitations
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Edlin, B.R.; Eckhardt, B.J.; Shu, M.A.; Holmberg, S.D.; Swan, T. Toward a more accurate estimate of the prevalence of hepatitis C in the United States. Hepatology 2015, 62, 1353–1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Europe’s Hepatitis Challenge. Available online: https://www.who.int/bulletin/volumes/96/12/18-021218/en/ (accessed on 12 December 2018).
- U.S. Department of Health & Human Services. Office of Population Affairs. Hepatitis C: Quick Facts. Available online: https://www.hhs.gov/opa/reproductive-health/fact-sheets/sexually-transmitted-diseases/hepatitis-c/index.html (accessed on 12 December 2018).
- Mitra, A.K. Hepatitis C-related hepatocellular carcinoma: Prevalence around the world, factors interacting, and role of genotypes. Epidemiol. Rev. 1999, 21, 180–187. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Health and Human Services. National Viral Hepatitis Action Plan 2017–2020; United States Department of Health and Human Services: Washington, DC, USA, 2017.
- Centers for Disease Control and Prevention (CDC). Viral Hepatitis. Hepatitis C Questions and Answers for Health Professionals; CDC, 2018. Available online: https://www.cdc.gov/hepatitis/hcv/hcvfaq.htm (accessed on 20 December 2018).
- Centers for Disease Control and Prevention (CDC). Hepatitis C. Know More Hepatitis. Why People Born 1945-1965 Should Get Tested; CDC, 2018. Available online: https://www.cdc.gov/knowmorehepatitis/index.htm (accessed on 20 December 2018).
- Ross, R. Atherosclerosis—An inflammatory disease. New Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O., 3rd; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Inflammation, hypertension, and the metabolic syndrome. JAMA 2003, 290, 3000–3002. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, A.R.; Srinivasan, S.R.; Chen, W.; Azevedo, M.J.; Berenson, G.S. Influence of low birth weight on C-reactive protein in asymptomatic younger adults: The Bogalusa heart study. BMC Res. Notes 2011, 4, 71. [Google Scholar] [CrossRef] [PubMed]
- Wong, N.D.; Pio, J.; Valencia, R.; Thakal, G. Distribution of C-reactive protein and its relation to risk factors and coronary heart disease risk estimation in the National Health and Nutrition Examination Survey (NHANES) III. J. Prev. Cardiol. 2001, 4, 109–114. [Google Scholar] [CrossRef]
- Freeman, D.J.; Norrie, J.; Caslake, M.J.; Gaw, A.; Ford, I.; Lowe, G.D.; O’Reilly, D.S.; Packard, C.J.; Sattar, N.; West of Scotland Coronary Prevention Study. C-reactive protein is an independent predictor of risk for the development of diabetes in the West of Scotland Coronary Prevention Study. Diabetes 2002, 51, 1596–1600. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A.D.; Manson, J.E.; Rifai, N.; Buring, J.E.; Ridker, P.M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA 2001, 286, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Stenvinkel, P. Inflammation in end-stage renal disease: The hidden enemy. Nephrology 2006, 11, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Dupuy, A.M.; Terrier, N.; Sénécal, L.; Morena, M.; Leray, H.; Canaud, B.; Cristol, J.P. Is C-reactive protein a marker of inflammation? Nephrology 2003, 24, 337–341. [Google Scholar]
- Adinolfi, L.E.; Restivo, L.; Zampino, R.; Guerrera, B.; Lonardo, A.; Ruggiero, L.; Riello, F.; Loria, P.; Florio, A. Chronic HCV infection is a risk of atherosclerosis. Role of HCV and HCV-related steatosis. Atherosclerosis 2012, 221, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Abaete De los Santos, C.; Antonello, V.; Antonello, I. An Assay: Hepatitis C does not incite increased C-reactive protein level in hemodialysis patients unless another inflammatory condition is associated. J. Gastroenterol. Hepatol. Res. 2017, 6, 2416–2418. [Google Scholar] [CrossRef]
- Che, W.; Zhang, B.; Liu, W.; Wei, Y.; Xu, Y.; Hu, D. Association between high-sensitivity C-reactive protein and N-Terminal Pro-B-Type Natriuretic Peptide in patients with hepatitis C virus infection. Mediat. Inflamm. 2012, 2012, 730923. [Google Scholar] [CrossRef] [PubMed]
- Tuncer, O.; Selahattin, A.; Hasan, A. Correlation of high sensitive C-reactive protein and hepatitis C virus RNA in anti-HCV-positive sera. Dicle Med. J. 2011, 38, 134–136. [Google Scholar]
- Shah, S.; Ma, Y.; Scherzer, R.; Huhn, G.; French, A.L.; Plankey, M.; Peters, M.G.; Grunfeld, C.; Tien, P.C. Association of HIV, hepatitis C virus and liver fibrosis severity with interleukin-6 and C-reactive protein levels. AIDS 2015, 29, 1325–1333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popovic, L.; Maja, J.; Miodrag, V.; Ljiljana, K.; Velimir, K.; Ivan, D. High sensitivity C-reactive protein as prediction factor of disease progression in patients with chronic hepatitis C and mild liver steatosis. Acta Med. 2010, 49, 14–18. [Google Scholar]
- Xie, Y.; Garza, G.; Dong, J. Hepatitis C virus genotype and subtype distribution in patient specimens tested at the University of Texas Medical Branch, Galveston, between January 2011 and November 2014. Lab. Med. 2016, 47, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). National Health and Nutrition Examination Survey; CDC: Atlanta, GA, USA, 2019.
- Goff, D.C.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 ACC/AHA Guideline on the assessment of cardiovascular risk. Circulation 2014, 129, S49–S73. [Google Scholar] [CrossRef] [PubMed]
- Ford, D.E.; Erlinger, T.P. Depression and C-reactive protein in US adults: Data from the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2004, 164, 1010–1014. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.K.; Alvarez, J.O.; Wahed, M.A.; Fuchs, G.J.; Stephensen, C.B. Predictors of serum retinol in children with shigellosis. Am. J. Clin. Nutr. 1998, 68, 1088–1094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Giudice, M.; Gangestad, S.W. Rethinking IL-6 and CRP: Why they are more than inflammatory biomarkers, and why it matters. Brain Behav. Immun. 2018, 70, 61–75. [Google Scholar] [CrossRef] [PubMed]
- Salter, M.L.; Lau, B.; Mehta, S.H.; Go, V.F.; Leng, S.; Kirk, G.D. Correlates of elevated interleukin-6 and C-reactive protein in persons with or at high risk for HCV and HIV infections. J. Acquir. Immune Defic. Syndr. 2013, 64, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Gale, M., Jr.; Foy, E.M. Evasion of intracellular host defence by hepatitis C virus. Nature 2005, 436, 939–945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reingold, J.; Wanke, C.; Kotler, D.; Lewis, C.; Tracy, R.; Heymsfield, S.; Tien, P.; Bacchetti, P.; Scherzer, R.; Grunfeld, C.; et al. Association of HIV infection and HIV/HCV coinfection with C-reactive protein levels: The fat redistribution and metabolic change in HIV infection (FRAM) study. J. Acquir. Immune Defic. Syndr. 2008, 48, 142–148. [Google Scholar] [CrossRef] [PubMed]
| Variables | Categories | Frequency | Weighted Frequency | Weighted Percent |
|---|---|---|---|---|
| Age (years) | ||||
| < 30 | 1241 | 46,010,147 | 22.42 | |
| ≥ 30 | 4370 | 159,146,706 | 77.57 | |
| Sex | ||||
| Female | 2726 | 100,219,509 | 51.14 | |
| Male | 2885 | 104,937,345 | 48.85 | |
| Race | ||||
| Hispanics | 1703 | 29,185,508 | 14.22 | |
| Whites | 2575 | 138,328,013 | 67.42 | |
| African American | 1013 | 23,061,139 | 11.24 | |
| Other races | 320 | 14,582,193 | 7.10 | |
| Received Blood Transfusion | ||||
| Yes | 590 | 19,569,140 | 9.65 | |
| No | 4956 | 183,052,596 | 90.34 | |
| Injection Drug Use | ||||
| Yes | 79 | 3,113,755 | 1.85 | |
| No | 4287 | 164,724,273 | 93.14 | |
| MSM | ||||
| Yes | 100 | 3,716,176 | 4.38 | |
| No | 2079 | 80,966,474 | 95.61 | |
| Smoking | ||||
| Yes | 1013 | 35,834,131 | 40.26 | |
| No | 1459 | 53,160,492 | 59.73 | |
| HIV | ||||
| Yes | 21 | 641,006 | 0.39 | |
| No | 4021 | 160,114,320 | 99.60 | |
| Hepatitis C Virus | ||||
| Yes | 106 | 3,322,600 | 1.61 | |
| No | 5505 | 201,834,254 | 98.4 | |
| C-Reactive Protein | ||||
| ≤ 0.22mg/dL | 2552 | 85,411,403 | 41.63 | |
| > 0.22mg/dL | 3059 | 119,745,451 | 58.36 |
| Categories | Mean ± SD * (mg/dL) | Selected Percentiles | ||||
|---|---|---|---|---|---|---|
| 10th | 25th | Median | 75th | 90th | ||
| HCV Positive | 0.12 ± 0.08 | 0.01 | 0.06 | 0.12 | 0.28 | 0.81 |
| HCV Negative | 0.24 ± 0.02 | 0.02 | 0.06 | 0.16 | 0.39 | 0.88 |
| Risk Factor Variables | Means ± SD (mg/dL) | p-Value |
|---|---|---|
| Race | <0.001 | |
| Hispanic | 0.39 ± 0.03 | |
| African American | 0.50 ± 0.04 | |
| White | 0.35 ± 0.02 | |
| Other | 0.20 ± 0.02 | |
| Sex | <0.001 | |
| Male | 0.30 ± 0.01 | |
| Female | 0.42 ± 0.01 | |
| Age | <0.05 | |
| <30 | 0.32 ± 0.02 | |
| ≥30 | 0.38 ± 0.02 | |
| HIV | 0.82 | |
| No | 0.35 ± 0.02 | |
| Yes | 0.36 ± 0.07 | |
| Drug use | 0.73 | |
| No | 0.36 ± 0.02 | |
| Yes | 0.38 ± 0.06 | |
| Smoking | 0.15 | |
| No | 0.38 ± 0.02 | |
| Yes | 0.43 ±0.03 | |
| MSM | <0.02 | |
| No | 0.30 ± 0.02 | |
| Yes | 0.22 ± 0.04 | |
| Blood Transfusion | <0.02 | |
| Received | 0.48 ± 0.05 | |
| Did not receive | 0.35 ± 0.01 |
| Categories | Mean ± SD * (mg/dL) | Selected Percentiles | ||||
|---|---|---|---|---|---|---|
| 10th | 25th | Median | 75th | 90th | ||
| HCV RNA positive | 0.56 ± 0.03 | 0.01 | 0.05 | 0.12 | 0.28 | 0.61 |
| HCV RNA Negative | 0.48 ± 0.05 | 0.02 | 0.07 | 0.19 | 0.43 | 1.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhuiyan, A.R.; Mitra, A.K.; Ogungbe, O.; Kabir, N. Association of HCV Infection with C-Reactive Protein: National Health and Nutrition Examination Survey (NHANES), 2009–2010. Diseases 2019, 7, 25. https://doi.org/10.3390/diseases7010025
Bhuiyan AR, Mitra AK, Ogungbe O, Kabir N. Association of HCV Infection with C-Reactive Protein: National Health and Nutrition Examination Survey (NHANES), 2009–2010. Diseases. 2019; 7(1):25. https://doi.org/10.3390/diseases7010025
Chicago/Turabian StyleBhuiyan, Azad R., Amal K. Mitra, Oluwabunmi Ogungbe, and Nusrat Kabir. 2019. "Association of HCV Infection with C-Reactive Protein: National Health and Nutrition Examination Survey (NHANES), 2009–2010" Diseases 7, no. 1: 25. https://doi.org/10.3390/diseases7010025
APA StyleBhuiyan, A. R., Mitra, A. K., Ogungbe, O., & Kabir, N. (2019). Association of HCV Infection with C-Reactive Protein: National Health and Nutrition Examination Survey (NHANES), 2009–2010. Diseases, 7(1), 25. https://doi.org/10.3390/diseases7010025






