PAOT-Liquid® Technology: An Easy Electrochemical Method for Evaluating Antioxidant Capacity of Wines
Abstract
1. Introduction
2. Material and Methods
2.1. Total Polyphenols Content (TPC)
2.2. DPPH Assay
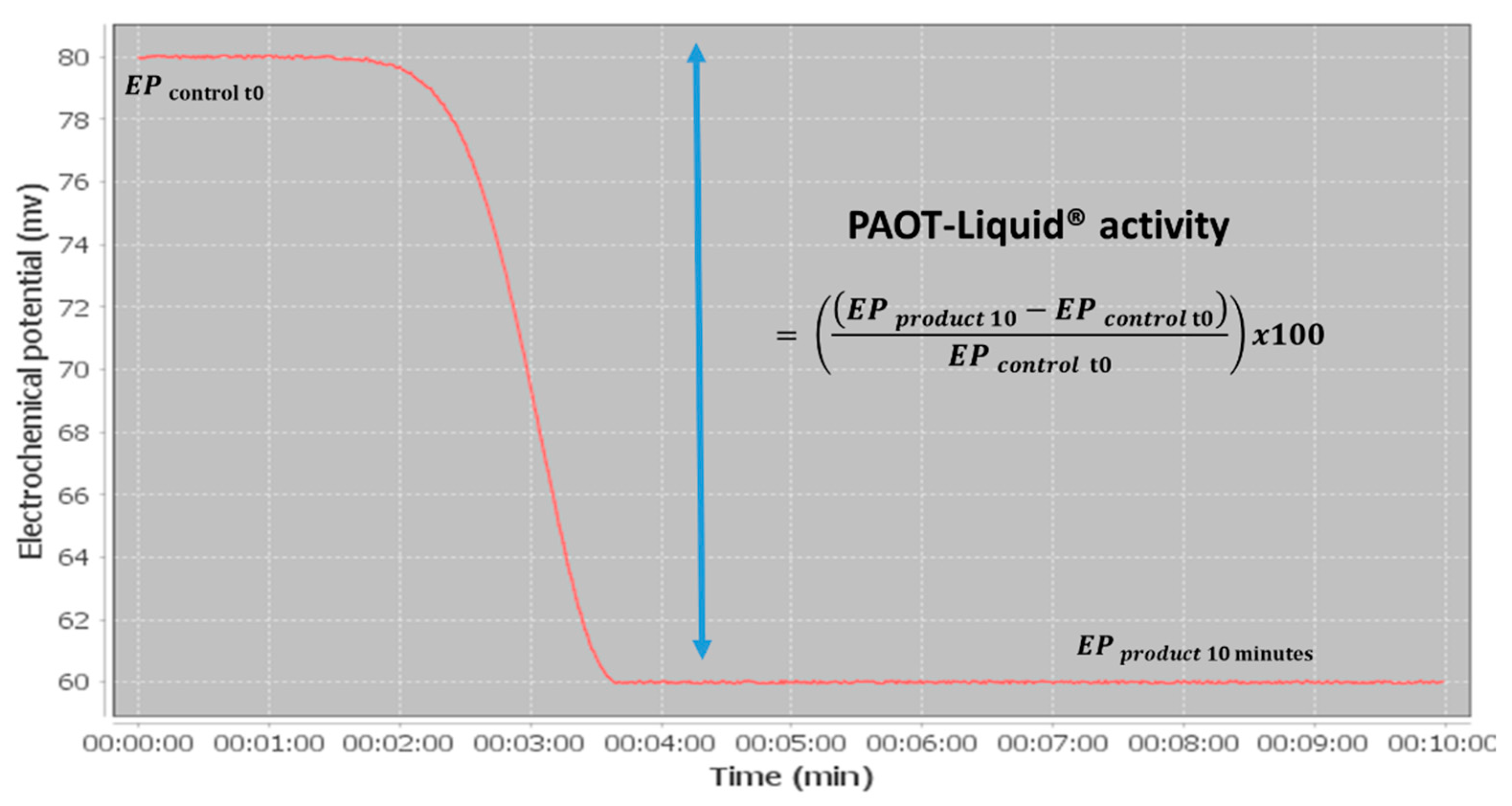
2.3. PAOT-Liquid® Assay
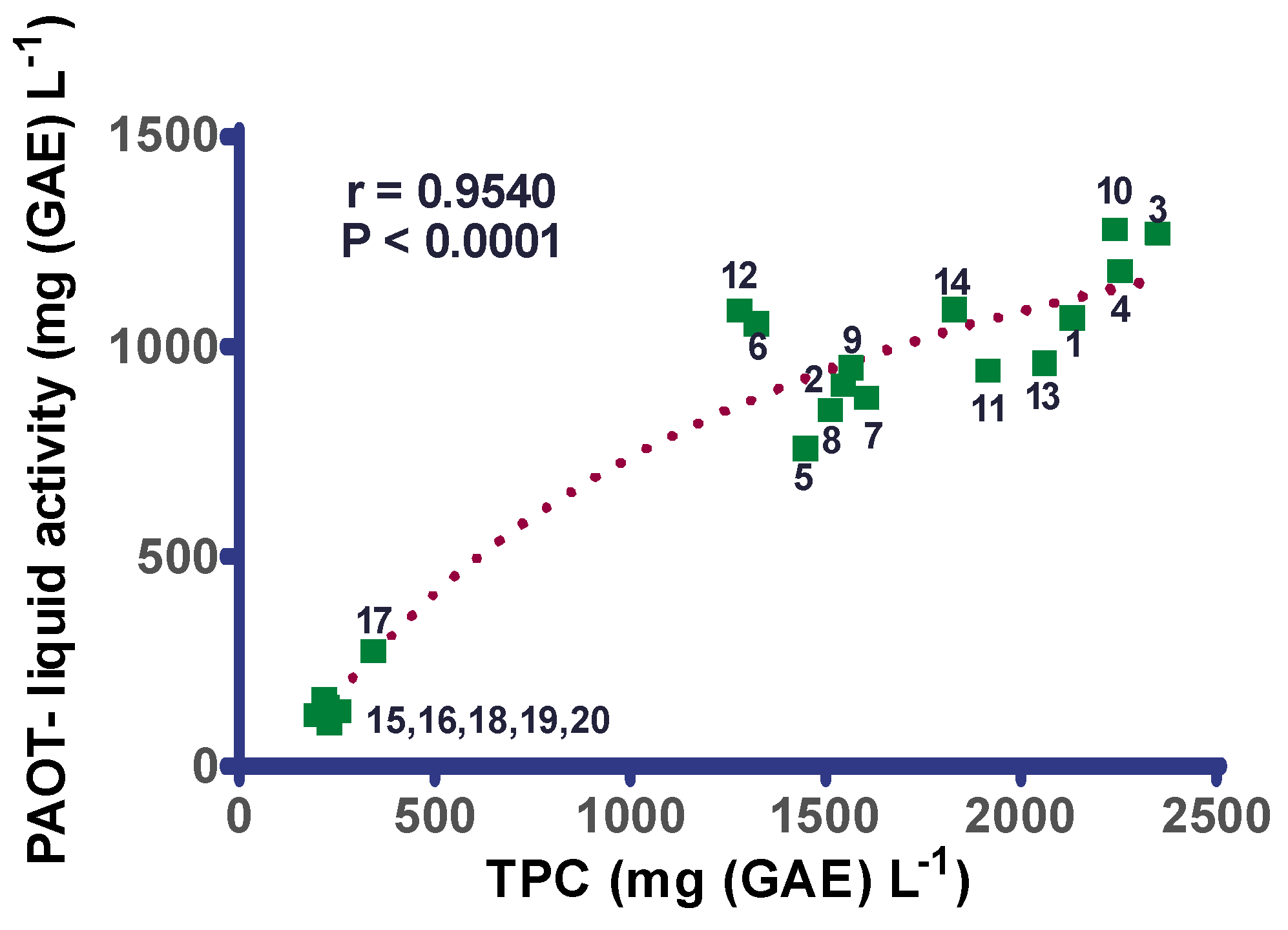
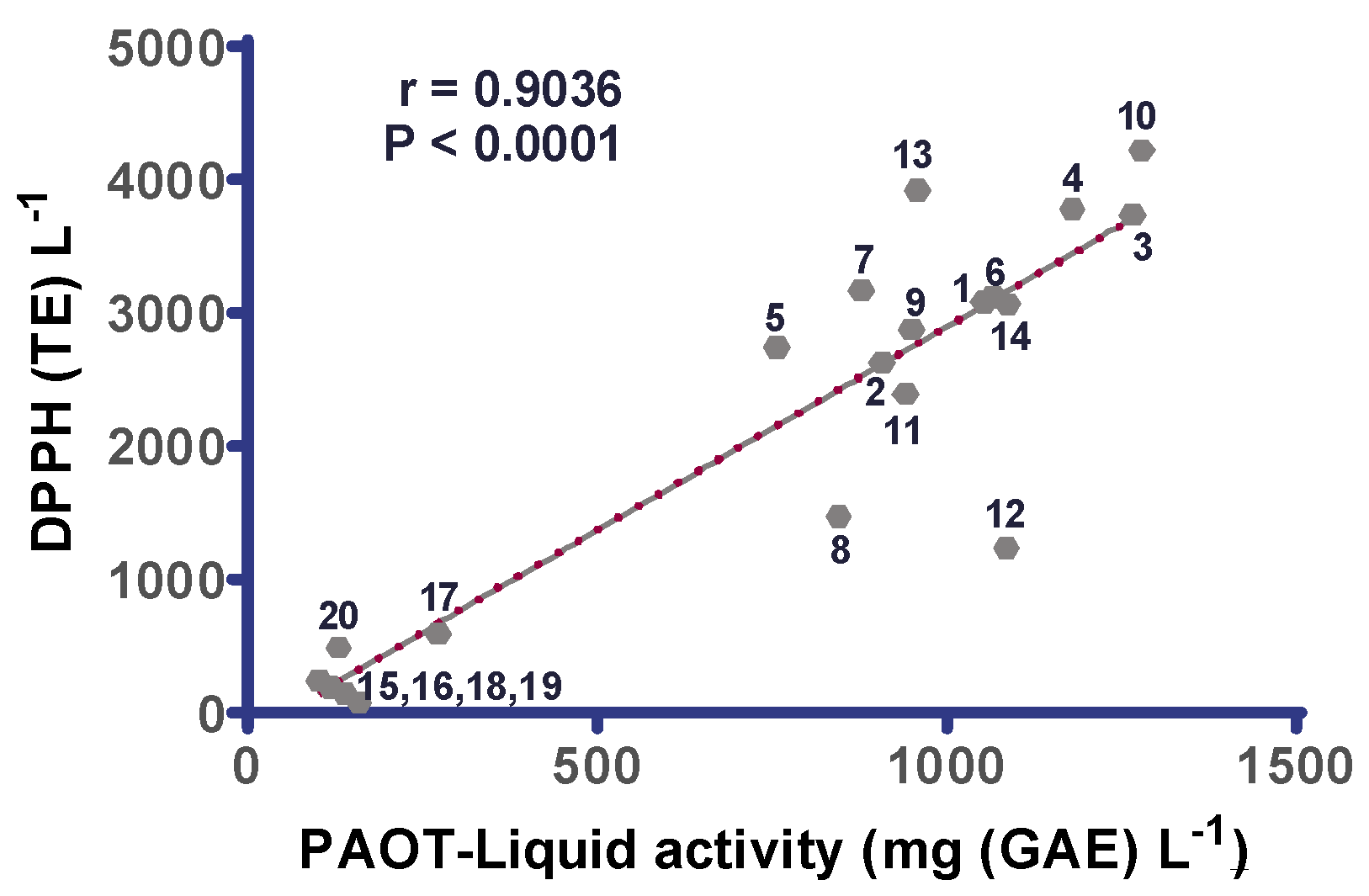
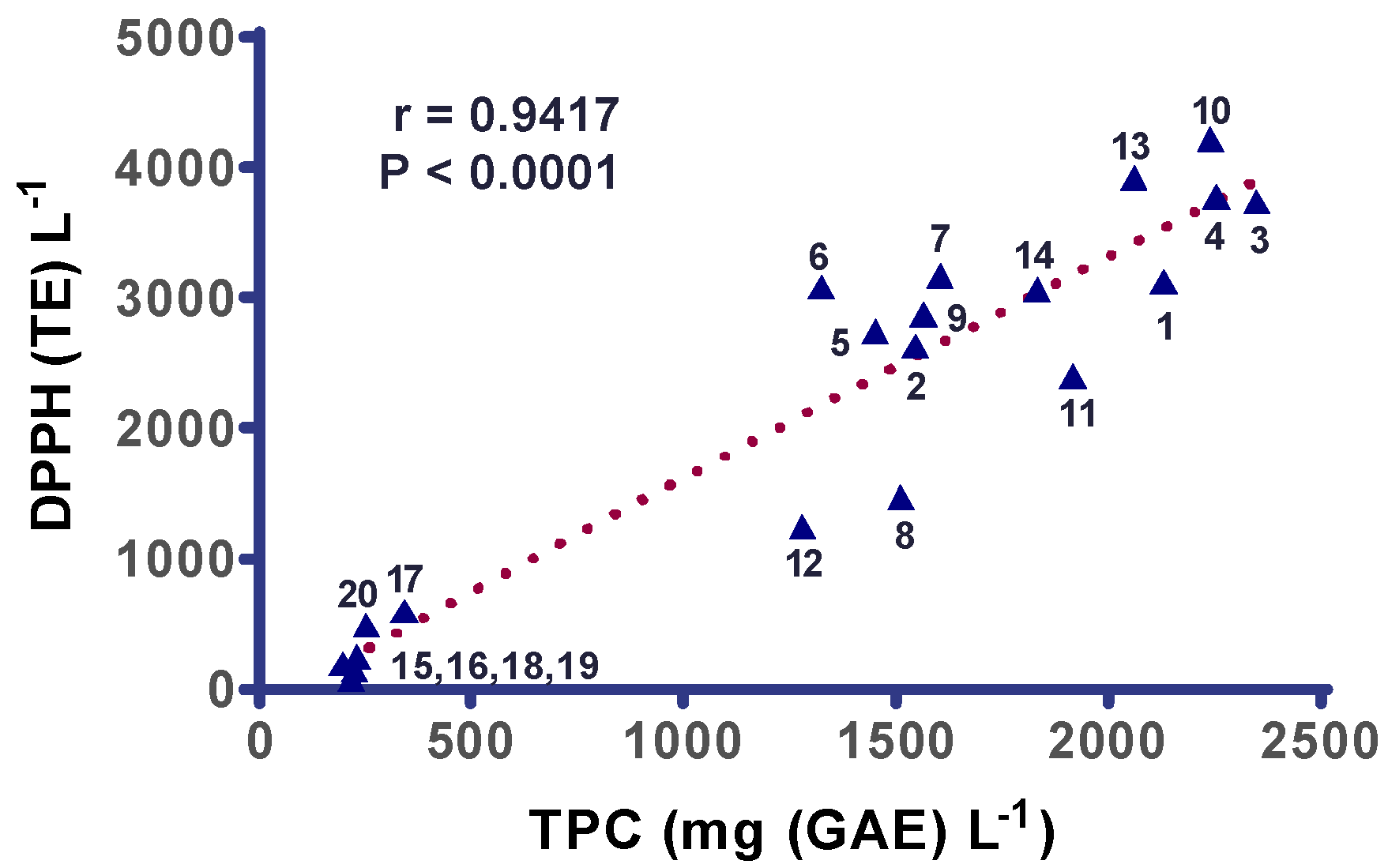
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Giustarini, G.; Dalle-Donne, I.; Tsikas, D.; Rossi, R. Oxidative stress and human diseases: Origin, link, measurement, mechanisms, and biomarkers. Crit. Rev. Clin. Lab. Sci. 2009, 45, 241–281. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Redefining oxidative stress. Antioxid. Redox Signal. 2006, 8, 1865–1879. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Dietary antioxidant flavonoids and risk of coronary heart disease: The zutphen elderly study. Lancet 1993, 342, 1007–1011. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Salas-Salvadó, J.; Estruch, R.; Corella, D.; Fitó, M.; Ros, E. Benefits of the Mediterranean diet: Insights from the PREDIMED Study. Prog. Cardiovasc. Dis. 2015, 58, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Sinatra, D.; Blanco, I.; Mulè, S.; La Verde, M.; Marranzano, M. Association between dietary phenolic acids and hypertension in a Mediterranean cohort. Nutrients 2017, 9, 1069. [Google Scholar] [CrossRef]
- Oak, M.-H.; Auger, C.; Belcastro, E.; Park, S.-H.; Lee, H.-H.; Schini-Kerth, V.B. Potential mechanisms underlying cardiovascular protection by polyphenols: Role of the endothelium. Free Radic. Biol. Med. 2018, 122, 161–170. [Google Scholar] [CrossRef]
- Birringer, M. Hormetics: Dietary triggers of an adaptive stress response. Pharm. Res. 2011, 28, 2680–2694. [Google Scholar] [CrossRef]
- Guilford, E.E.; Pezzuto, J.M. Wine and health: A review. Am. J. Enol. Vitucult. 2011, 62, 471–486. [Google Scholar] [CrossRef]
- Snopek, L.; Mlcek, J.; Sochorova, L.; Baron, M.; Hlavacova, I.; Jurikova, T.; Kizek, R.; Sedlackova, E.; Sochor, J. Contribution of red wine consumption to human health protection. Molecules 2018, 23, 1684. [Google Scholar] [CrossRef]
- Baiano, A.; Terracone, C.; Gambacorta, G.; La Notte, E. Phenolic content and antioxidant activity of primitivo wine: Comparison among winemaking technologies. J. Food Sci. 2009, 74, C258–C267. [Google Scholar] [CrossRef]
- Tabart, J.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Antioxidant capacity of black currant varies with organ, season, and cultivar. J. Agric. Food Chem. 2006, 54, 6271–6276. [Google Scholar] [CrossRef] [PubMed]
- Tadolini, B.; Juliano, C.; Piu, L.; Franconi, F.; Cabrini, L. Resveratrol inhibition of lipid peroxidation. Free Radic. Res. 2000, 33, 105–114. [Google Scholar] [CrossRef]
- Tabart, J.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Comparative antioxidant capacities of phenolic compounds measured by various tests. Food Chem. 2009, 113, 1226–1233. [Google Scholar] [CrossRef]
- Poutaraud, A.; Guilloteau, L.; Gros, C.; Lobstein, A.; Meziane, S.; Steyer, D.; Moisan, M.P.; Foury, A.; Lansade, L. Lavender essential oil decreases stress response of horses. Environ. Chem. Lett. 2018, 16, 539–544. [Google Scholar] [CrossRef]
- Kaci, M.; Belhaffef, A.; Meziane, S.; Dostert, G.; Menu, P.; Velot, E.; Desobry, S.; Arab-Tehranv, E. Nanoemulsions and topical creams for the safe and effective delivery of lipophilic antioxidant coenzyme Q10. J. Food Process Technol. 2016, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Perez-Jimenez, J.; Fezeu, L.; Touvier, M.; Arnault, N.; Manach, C.; Hercberg, S.; Galan, P.; Scalbert, A. Dietary intake of 337 polyphenols in French adults. Am. J. Clin. Nutr. 2011, 93, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Chabert, P.; Auger, C.; Pincemail, J.; Schini-Kerth, V. Overview of plant-derived antioxidants. In Systems Biology of Free Radicals and Antioxidants; Läher, I., Ed.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 4005–4020. [Google Scholar]
- Gambella, M.; Campuzano, A.; Reviejo, J.; Pingardon, J.M. Electrochemical estimation of the polyphenol wine index in wines using a laccase biosensor. J. Agric. Food Chem. 2006, 54, 7960–7967. [Google Scholar] [CrossRef]
- Paixao, N.; Perestrelo, R.; Marques, J.C.; Camara, J.S. Relationship between antioxidant capacity and total phenolic content of red, rosé and white wines. Food Chem. 2007, 105, 204–214. [Google Scholar] [CrossRef]
- Pigani, L.; Rioli, C.; Foca, G.; Ulrici, A.; Seeber, R.; Terzi, F.; Zanardi, C. Determination of polyphenols content and colour index in wines trough PEDOT-modified electrodes. Anal. Bioanal. Chem. 2016, 408, 7329–7338. [Google Scholar] [CrossRef]
- Arribas, A.S.; Martinez-Fernandez, M.; Moreno, M.; Bermejo, E.; Zapardiel, A.; Chicharro, M. Analysis of total polyphenols in wines by FIA with highly stable amperometric detection using carbon nanotube-modified electrodes. Food Chem. 2013, 136, 1183–1192. [Google Scholar] [CrossRef]
- Van Leeuw, R.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Antioxidant capacity and phenolic composition of red wines from various grape varieties: Specificity of pinot noir. J. Food Compos. Anal. 2014, 36, 40–50. [Google Scholar] [CrossRef]
- Mitić, M.N.; Obradović, M.V.; Grahovac, Z.B.; Pavlović, A.N. Antioxidant capacities and phenolic levels of different varieties of Serbian white wines. Molecules 2010, 22, 2016–2027. [Google Scholar] [CrossRef] [PubMed]
- Kevers, C.; Sipel, A.; Pincemail, J.; Dommes, J. Antioxidant capacity of hydrophilic food matrices: Optimization and validation of ORAC assay. Food Anal. Methods 2014, 7, 403–416. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Arranz, S.; Tabernero, M.; Díaz-Rubio, M.; Serrano, J.; Goñi, I.; Saura-Calixto, F. Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: Extraction, measurement and expression of results. Food Res. Int. 2008, 41, 274–285. [Google Scholar] [CrossRef]
- Available online: http://ie-antioxydants.com/fr/paot/paot-scan-deux-methodes/ (accessed on 20 January 2019).

| Number | Color | Region/Country | Name | Vintage | Year |
|---|---|---|---|---|---|
| 1 | red | Beaujolais/France | Moulin à vent | Gamay | 2015 |
| 2 | red | Cachapoal Valley/Chili | La Capitana | Merlot | 2014 |
| 3 | red | Bordeaux/France | Château Tuilerie Pages | Cabernet Franc, Merlot, Cabernet Sauvignon | 2014 |
| 4 | red | Bordeaux/France | Château la Tuilerie Graves | Merlot, Cabernet Sauvignon | 2016 |
| 5 | red | Corbières/France | Château Prat de Cest | Syrah, Grenache, Mourvedre | 2015 |
| 6 | red | Barossa Valley/South Australia | Lindeman’s Bin 50 | Shiraz | 2017 |
| 7 | red | Mendoza/Argentina | Trivento | Malbec | 2017 |
| 8 | red | Bardolino/Italy | Giovanni Righetti | Corvina, Rondinella, Molinari | 2017 |
| 9 | red | Saint-Chinian/France | Valdorb rouge | Syrah, Grenache, Carignan | 2017 |
| 10 | red | Colchagua Valley/Chili | Koyle Reserva | Cabernet Sauvignon | 2014 |
| 11 | red | Western Cape/South Africa | Baie Cap | Pinotage | 2017 |
| 12 | red | Bourgogne/France | La chance du Roy | Gamay, Pinot Noir | 2015 |
| 13 | red | Minervois, France | L’aigle de Minerve | Carignan, Syrah, Grenache, Mourvedre | 2016 |
| 14 | red | Côtes du Rhône Villages/France | Côtes du Rhône villages | Grenache/Syrah | 2016 |
| 15 | rosé | Pays d’Oc/France | Syrah Rosé | Syrah rosé | 2016 |
| 16 | rosé | Pays d’Oc/France | Vin Gris | Cinsault, Syrah, Carignan, Grenache | 2017 |
| 17 | rosé | Corse/France | La Petite Paillote | Niellucciu, Sciaccarellu | 2017 |
| 18 | white | Pays d’Oc/France | Vent Marin | Chardonnay | 2016 |
| 19 | white | Val de Loire/France | Sauvignon de Touraine | Sauvignon Blanc | 2017 |
| 20 | white | Corse/France | La petite Paillote | Vermentino | 2017 |
| PAOT-Liquid® Assay mg (GAE) L−1 | |
|---|---|
| Flavano-3-ol Family | |
| Catechin | 504.56 ± 45.58 |
| Epicatechin (EC) | 730.2 ± 93.73 |
| Gallocatechin (GC) | 431.05 ± 35.61 |
| Epigallocatechin (EGC) | 545.58 ± 45.87 |
| Epigallocatechin gallate (EGCG) | 613.11 ± 0.57 |
| Flavonol Family | |
| Kaempferol | 404.56 ± 55.27 |
| Quercetin | 560.4 ± 0.85 |
| Myricetin | 677.78 ± 7.41 |
| Flavanone Family | |
| Hesperdin methyl chalcone | 51.85 ± 0.57 |
| Naringin | 53.28 ± 0.28 |
| Anthocyanidins Family | |
| Pelargonidin Chloride | 284.33 ± 3.42 |
| Delphinidin Chloride | 340.74 ± 69.23 |
| Cyanidin Chloride | 512.54 ± 5.13 |
| Other | |
| Trolox | 544.16 ± 16.81 |
| Number | Region/Country | TPC mg (GAE) L−1 | DPPH Assay µM (TE) L−1 | PAOT-Liquid® Assay mg (GAE) mg L−1 |
|---|---|---|---|---|
| Red wines | ||||
| 1 | Beaujolais/France | 2129 ± 17.9 | 3119 ± 47.7 | 1067.5 ± 17.86 |
| 2 | Cachapoal Valley/Chili | 1545 ± 40.1 | 2628 ± 24.9 | 908.02 ± 39.13 |
| 3 | Bordeaux/France | 2349 ± 18.2 | 3732 ± 32.6 | 1267.39 ± 30.2 |
| 4 | Bordeaux/France | 2253 ± 9.7 | 3773 ± 72.9 | 1180.21 ± 2.98 |
| 5 | Corbières/France | 1450 ± 20.3 | 2738 ± 65.3 | 757.03 ± 11.91 |
| 6 | Barossa Valley/South Australia | 1323 ± 12.8 | 3082 ± 51.3 | 1054.74 ± 17.86 |
| 7 | Mendoza/Argentina | 1603 ± 14.68 | 3168 ± 32.7 | 878.24 ± 26.79 |
| 8 | Bardolino/Italy | 1511 ± 11.8 | 1474 ± 11.0 | 846.35 ± 17.86 |
| 9 | Saint-Chinian/France | 1563 ± 24.9 | 2874 ± 44.8 | 950.55 ± 26.79 |
| 10 | Colchagua Valley/Chili | 2239 ± 20.8 | 4219 ± 64.6 | 1280.15 ± 6.34 |
| 11 | Western Cape/South Africa | 1915 ± 17.5 | 2395 ± 20.1 | 942.04 ± 2.98 |
| 12 | Bourgogne/France | 1278 ± 41.5 | 1240 ± 4.5 | 1086.64 ± 8.93 |
| 13 | Minervois, France | 2060 ± 8.8 | 3912 ± 63.5 | 959.05 ± 32.75 |
| 14 | Côtes du Rhône Villages/France | 1831 ± 37.8 | 3065 ± 57.2 | 1088.77 ± 17.86 |
| mean | 1789 | 2958 | 1016.47 | |
| SD | 367 | 854 | 153.11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joël, P.; Mouna-Messaouda, K.; Claire, K.; Jessica, T.; Raymond, E.E.; Smail, M. PAOT-Liquid® Technology: An Easy Electrochemical Method for Evaluating Antioxidant Capacity of Wines. Diseases 2019, 7, 10. https://doi.org/10.3390/diseases7010010
Joël P, Mouna-Messaouda K, Claire K, Jessica T, Raymond EE, Smail M. PAOT-Liquid® Technology: An Easy Electrochemical Method for Evaluating Antioxidant Capacity of Wines. Diseases. 2019; 7(1):10. https://doi.org/10.3390/diseases7010010
Chicago/Turabian StyleJoël, Pincemail, Kaci Mouna-Messaouda, Kevers Claire, Tabart Jessica, Ebabe Elle Raymond, and Meziane Smail. 2019. "PAOT-Liquid® Technology: An Easy Electrochemical Method for Evaluating Antioxidant Capacity of Wines" Diseases 7, no. 1: 10. https://doi.org/10.3390/diseases7010010
APA StyleJoël, P., Mouna-Messaouda, K., Claire, K., Jessica, T., Raymond, E. E., & Smail, M. (2019). PAOT-Liquid® Technology: An Easy Electrochemical Method for Evaluating Antioxidant Capacity of Wines. Diseases, 7(1), 10. https://doi.org/10.3390/diseases7010010





