Role of Handheld In Vivo Reflectance Confocal Microscopy for the Diagnosis of Fabry Disease: A Case Report
Abstract
:1. Introduction
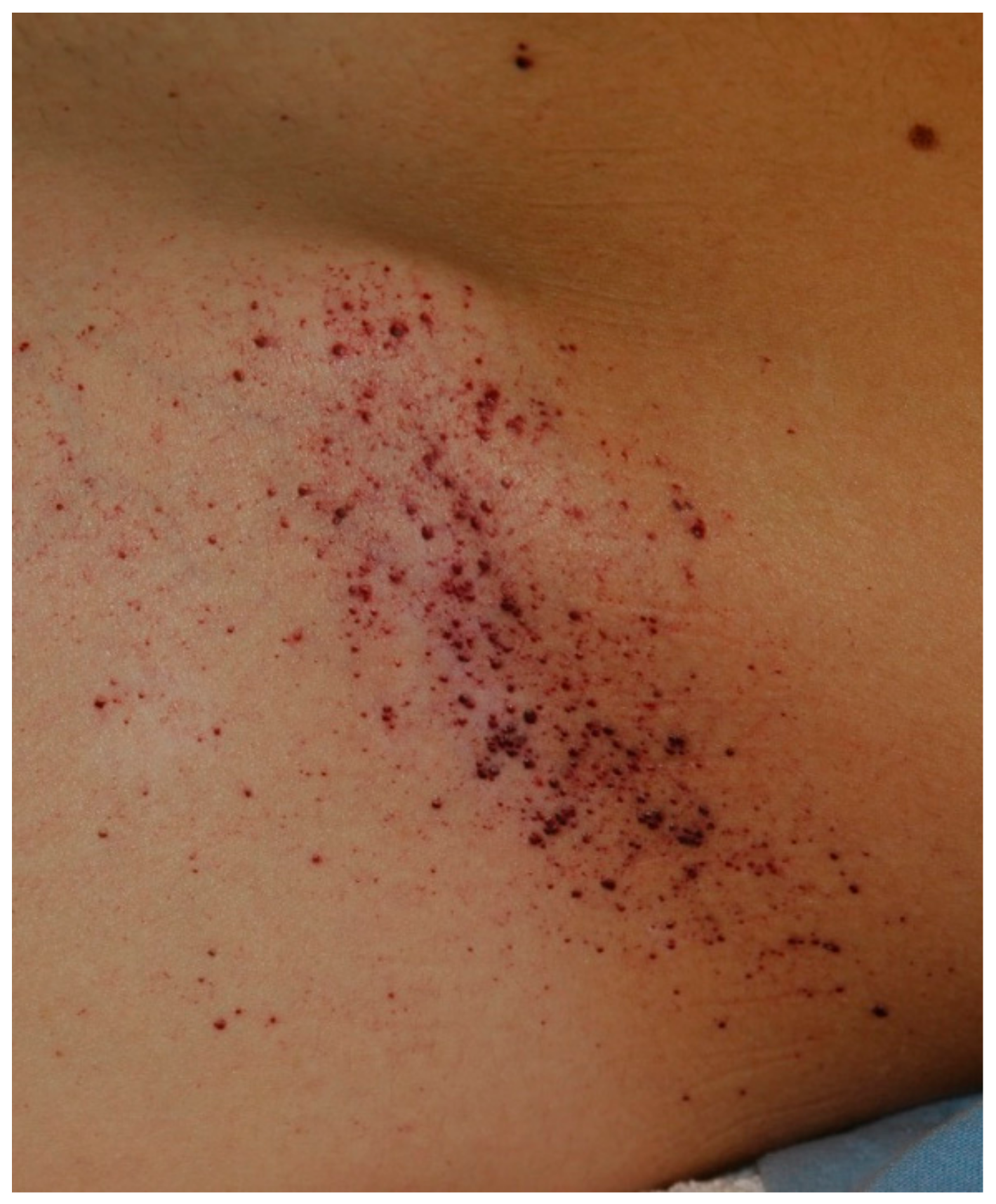
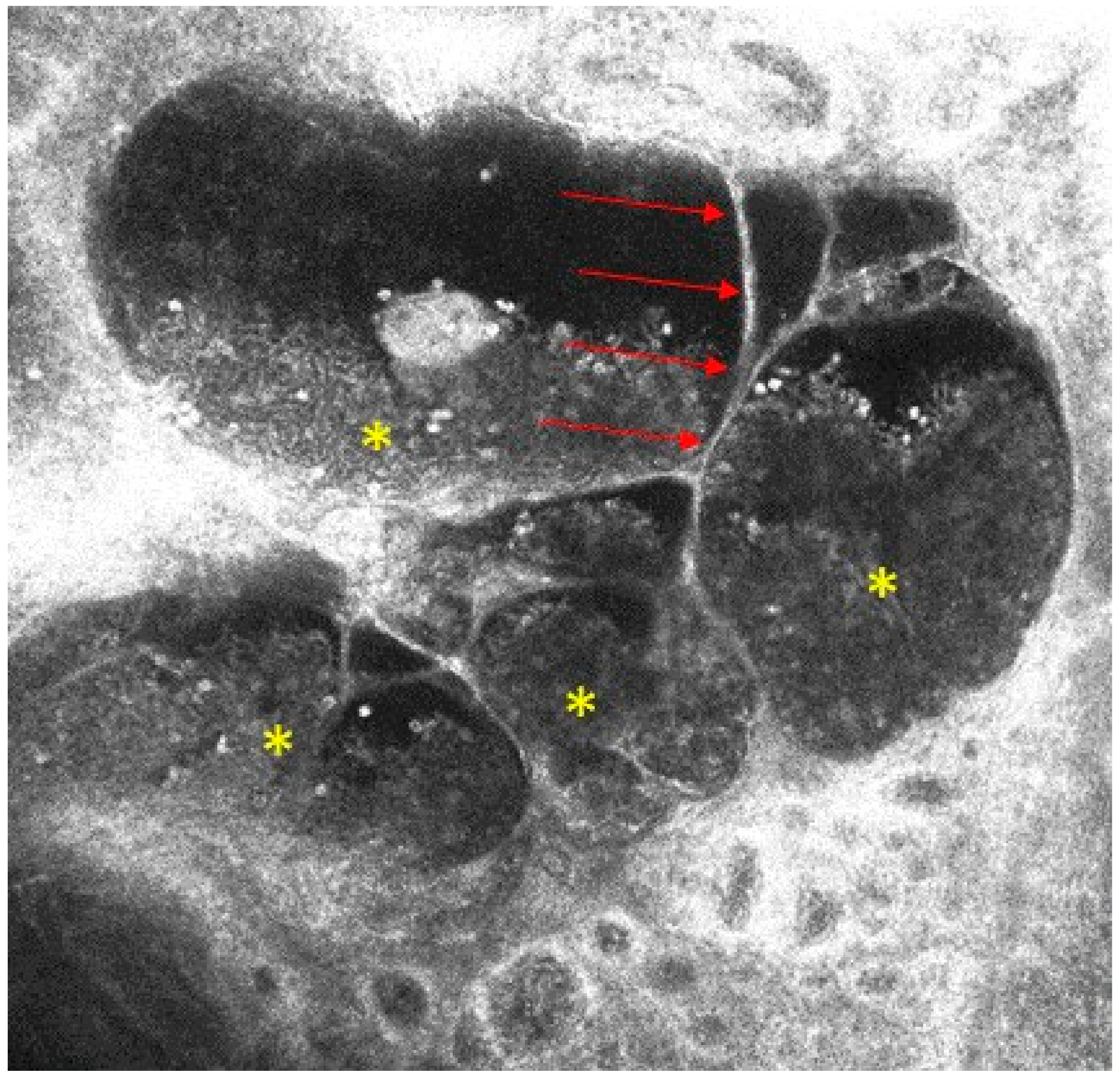
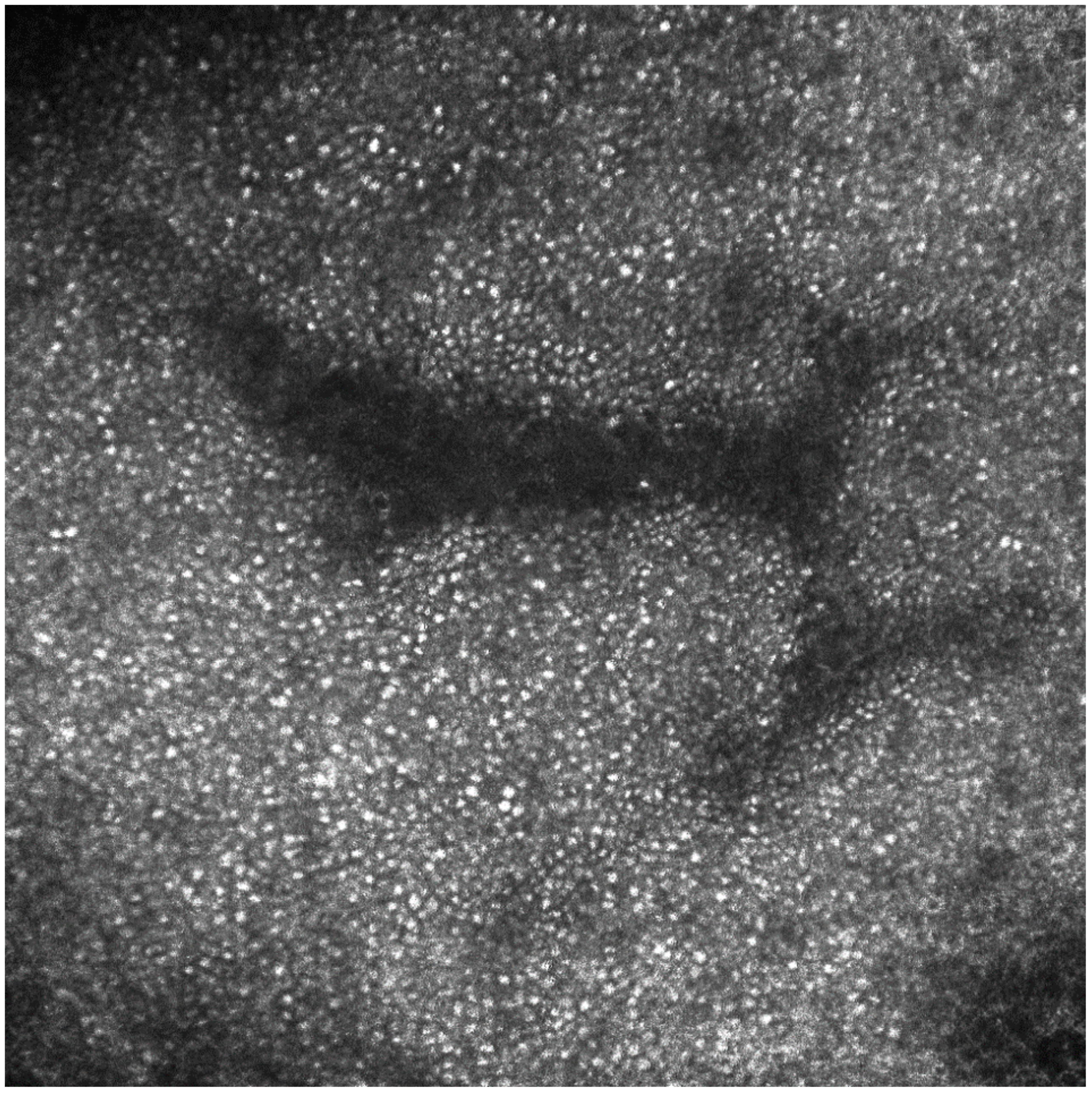
2. Case Report
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, B.; Adam, D.N. A Review of Fabry Disease. Skin Ther. Lett. 2018, 23, 4–6. [Google Scholar]
- Hoffmann, B.; Mayapetek, E. Fabry Disease—Often Seen, Rarely Diagnosed. Dtsch. Arztebl. Int. 2009, 106, 440–447. [Google Scholar] [PubMed]
- Wasielica-Poslednik, J.; Pfeiffer, N.; Reinke, J.; Pitz, S. Confocal laser-scanning microscopy allows differentiation between Fabry disease and amiodarone-induced keratopathy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 249, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Falke, K.; Büttner, A.; Schittkowski, M.; Stachs, O.; Kraak, R.; Zhivov, A.; Rolfs, A.; Guthoff, R. The microstructure of cornea verticillata in Fabry disease and amiodarone-induced keratopathy: A confocal laser-scanning microscopy study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2009, 247, 523–534. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cinotti, E.; Provvidenziale, L.; Fimiani, M.; Perrot, J.L.; Cambazard, F.; Rubegni, P. Role of Handheld In Vivo Reflectance Confocal Microscopy for the Diagnosis of Fabry Disease: A Case Report. Diseases 2018, 6, 55. https://doi.org/10.3390/diseases6030055
Cinotti E, Provvidenziale L, Fimiani M, Perrot JL, Cambazard F, Rubegni P. Role of Handheld In Vivo Reflectance Confocal Microscopy for the Diagnosis of Fabry Disease: A Case Report. Diseases. 2018; 6(3):55. https://doi.org/10.3390/diseases6030055
Chicago/Turabian StyleCinotti, Elisa, Luca Provvidenziale, Michele Fimiani, Jean Luc Perrot, Frederic Cambazard, and Pietro Rubegni. 2018. "Role of Handheld In Vivo Reflectance Confocal Microscopy for the Diagnosis of Fabry Disease: A Case Report" Diseases 6, no. 3: 55. https://doi.org/10.3390/diseases6030055
APA StyleCinotti, E., Provvidenziale, L., Fimiani, M., Perrot, J. L., Cambazard, F., & Rubegni, P. (2018). Role of Handheld In Vivo Reflectance Confocal Microscopy for the Diagnosis of Fabry Disease: A Case Report. Diseases, 6(3), 55. https://doi.org/10.3390/diseases6030055





