Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases
Abstract
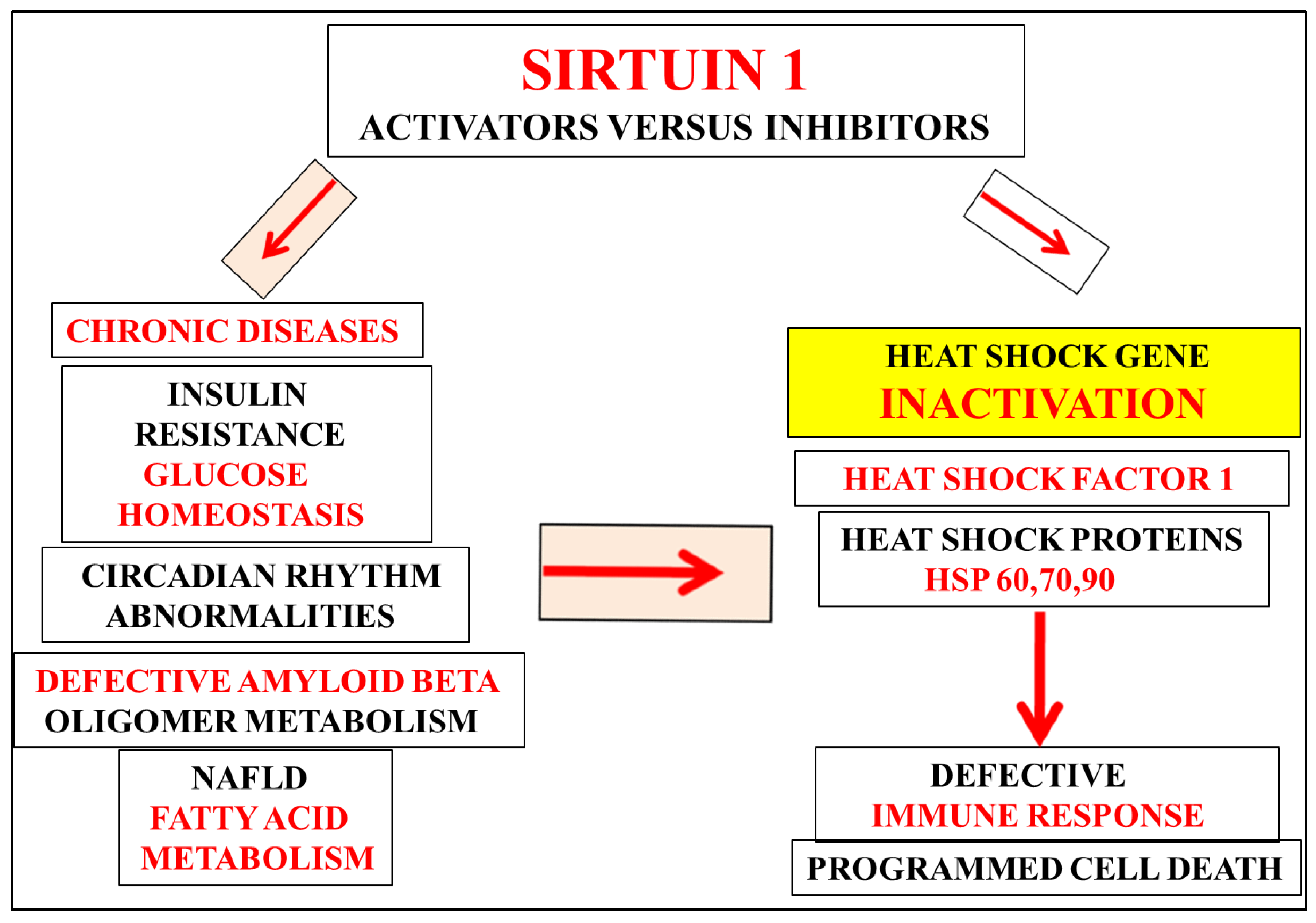
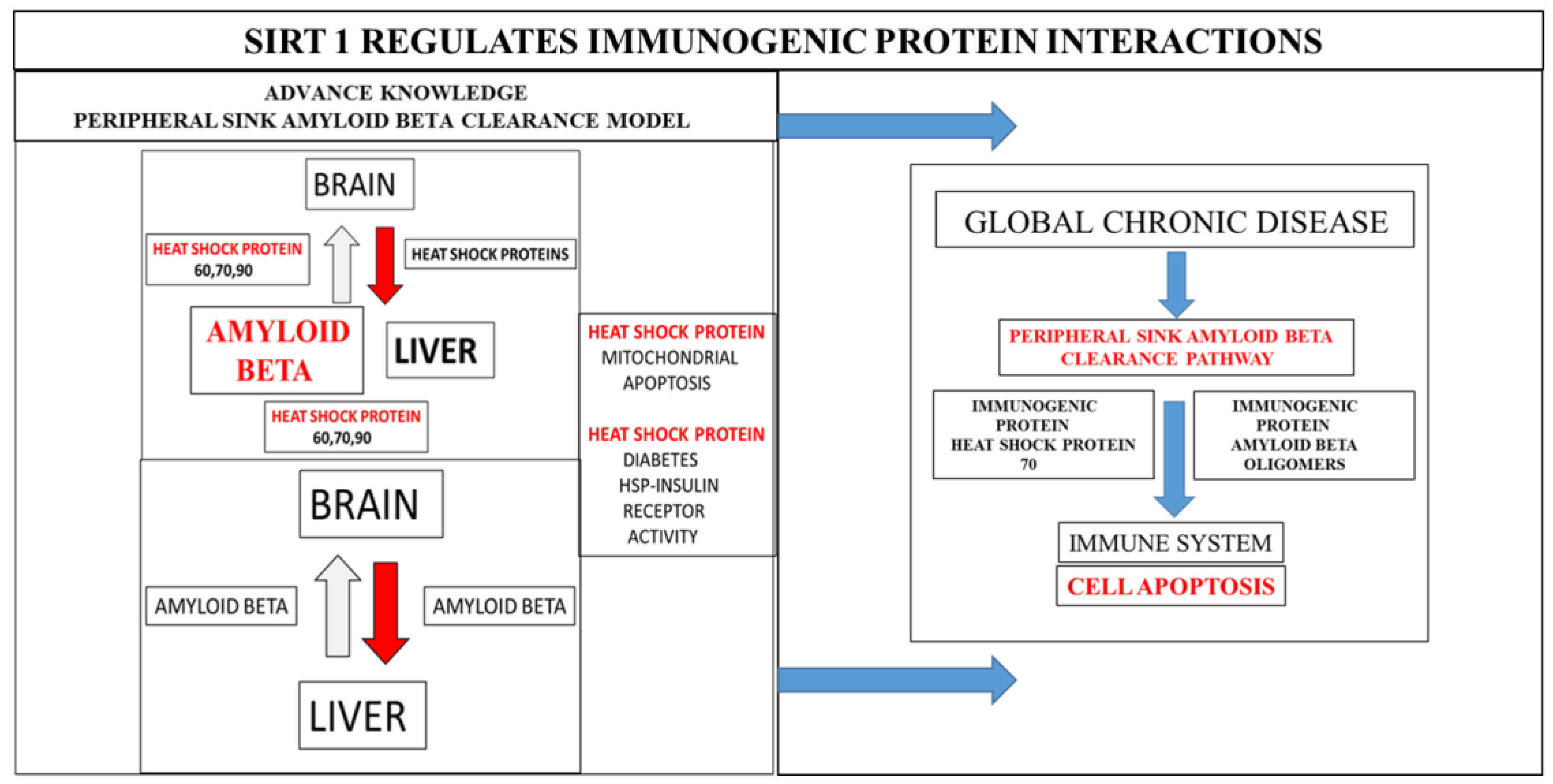
:Commentary
Funding
Acknowledgments
Conflicts of Interest
References
- Kakkar, V.; Meister-Broekema, M.; Minoia, M.; Carra, S.; Kampinga, H.H. Barcoding heat shock proteins to human diseases: Looking beyond the heat shock response. Dis. Models Mech. 2014, 7, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Vabulas, R.M.; Raychaudhuri, S.; Hayer-Hartl, M.; Ulrich, F. Protein Folding in the Cytoplasm and the Heat Shock Response. Cold Spring Harb. Perspect. Biol. 2010, 2, a004390. [Google Scholar] [CrossRef] [PubMed]
- Cantó, C. The heat shock factor HSF1 juggles protein quality control and metabolic regulation. J. Cell Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Pastor, R.; Burchfiel, E.T.; Thiele, D.J. Regulation of heat shock transcription factors and their roles in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 4–19. [Google Scholar] [CrossRef] [PubMed]
- Dalgediene, I.; Lasickiene, R.; Budvytyte, R.; Valincius, G.; Morkuniene, R.; Borutaite, V.; Zvirbliene, A. Immunogenic properties of amyloid beta oligomers. J. Biomed. Sci. 2013, 20, 10. [Google Scholar] [CrossRef] [PubMed]
- Maier, M.; Seabrook, T.J.; Lazo, N.D.; Jiang, L.; Das, P.; Janus, C.; Lemere, C.A. Short amyloid-beta (Abeta) immunogens reduce cerebral Abeta load and learning deficits in an Alzheimer’s disease mouse model in the absence of an Abeta-specific cellular immune response. J. Neurosci. 2006, 26, 4717–4728. [Google Scholar] [CrossRef] [PubMed]
- Kaul, G.; Thippeswamy, H. Role of Heat Shock Proteins in Diseases and Their Therapeutic Potential. Indian J. Microbiol. 2011, 51, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Jindal, S. Heat shock proteins: Applications in health and disease. Trends Biotechnol. 1996, 14, 17–20. [Google Scholar] [CrossRef]
- Martins, I.J. Heat shock gene Sirtuin 1 regulates post-prandial lipid metabolism with relevance to nutrition and appetite regulation in diabetes. Int J. Diabetes Clin. Diagn. 2016, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. Type 3 diabetes with links to NAFLD and Other Chronic Diseases in the Western World. Int. J. Diabetes 2016, 1, 1–5. [Google Scholar]
- Martins, I.J. Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases. Int. J. Mol. Sci. 2015, 16, 29554–29573. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. Nutritional Diets Accelerate Amyloid beta Metabolism and Prevent the Induction of Chronic Diseases and Alzheimer’s Disease. Available online: https://www.researchgate.net/publication/279533471_Nutritional_diets_accelerate_amyloid_beta_metabolism_and_prevent_the_induction_of_chronic_diseases_and_Alzheimer’s_disease (accessed on 13 May 2018).
- Regulation of Body Temperature and NAFLD in Global Population. Feb 2018. Asian Hospital and Healthcare Management e-Newsletter. Available online: https://www.asianhhm.com/.../regulation-of-body-temperature-and-nafld-in-global-po (accessed on 28 February 2018).
- Martins, I.J. Defective Interplay between Adipose Tissue and Immune System Induces Non Alcoholic Fatty Liver Disease. Updates Nutr. Disorders Ther. 2017, 1, 3. [Google Scholar]
- Martins, I.J. Appetite Control and Biotherapy in the Management of Autoimmune Induced Global Chronic Diseases. J. Clin. Immunol. Res. 2018, 2, 1–4. [Google Scholar]
- Martins, I.J. Diet and Nutrition reverse Type 3 Diabetes and Accelerated Aging linked to Global chronic diseases. J. Diabetes Res. Ther. 2016, 2, 1–6. [Google Scholar]
- Martins, I.J. Calorie Sensitive Anti-Aging Gene Regulates Hepatic Amyloid Beta Clearance in Diabetes and Neurodegenerative Diseases. EC Nutr. 2017, 1, 30–32. [Google Scholar]
- Martins, I.J. Unhealthy Diets Determine Benign or Toxic Amyloid Beta States and Promote Brain Amyloid Beta Aggregation. Austin J. Clin. Neurol. 2015, 2, 1060–1066. [Google Scholar]
- Martins, I.J. The Future of Genomic Medicine Involves the Maintenance of Sirtuin 1 in Global Populations. Int. J. Mol. Biol. 2017, 2, 00013. [Google Scholar]
- Martins, I.J. Biotherapy and the Immune System in Ageing Science. Acta Sci. Nutr. Health 2018, 2, 29–31. [Google Scholar]
- Labrecque, N.; Cermakian, N. Circadian Clocks in the Immune System. J. Biol. Rhythms. 2015, 30, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Habbal, O.A.; Al-Jabri, A.A. Circadian rhythm and the immune response: A review. Int. Rev. Immunol. 2009, 28, 93–108. [Google Scholar] [CrossRef] [PubMed]
- Scheiermann, C.; Kunisaki, Y.; Frenette, P.S. Circadian control of the immune system. Nat. Rev. Immunol. 2013, 13, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. Early diagnosis of neuron mitochondrial dysfunction may reverse global metabolic and neurodegenerative disease. GJMR 2016, 2, 1–8. [Google Scholar]
- Martins, I.J. Nutrition Therapy Regulates Caffeine Metabolism with Relevance to NAFLD and Induction of Type 3 Diabetes. J. Diabetes Metab. Disorders 2017, 4, 019. [Google Scholar] [CrossRef]
- Martins, I.J. Sirtuin 1, a Diagnostic Protein Marker and its Relevance to Chronic Disease and Therapeutic Drug Interventions. EC Pharmacol. Toxicol. 2018, 6, 209–215. [Google Scholar]
- Martins, I.J. Evaluation of diagnostic tests in human health and disease. J. Clin. Pathol. Lab. Med. 2018, 2, 13–15. [Google Scholar]
- Martins, I.J. The Limitations of Food Intake and Biomarkers in the Prevention of Chronic Diseases. Nov. Technol. Nutr. Food Sci. 2017, 1, 1–3. [Google Scholar]
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, I.J. Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases. Diseases 2018, 6, 39. https://doi.org/10.3390/diseases6020039
Martins IJ. Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases. Diseases. 2018; 6(2):39. https://doi.org/10.3390/diseases6020039
Chicago/Turabian StyleMartins, Ian James. 2018. "Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases" Diseases 6, no. 2: 39. https://doi.org/10.3390/diseases6020039
APA StyleMartins, I. J. (2018). Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases. Diseases, 6(2), 39. https://doi.org/10.3390/diseases6020039




