Clinical Perspective on Internet of Things Applications for Care of the Elderly
Abstract
1. Introduction
2. Research Background
3. Materials and Methods
- Phase 1: Identification of clinically required data for common diseases in the elderly population.
- Phase 2: Cross-case analysis of the data types identified in Phase 1 with the available IoT applications.
3.1. Phase 1: Identification of Clinically Required Data for Common Diseases in the Elderly Population
3.2. Phase 2: Cross-Case Analysis of Data Types Identified in Phase 1 with the Available IoT Applications
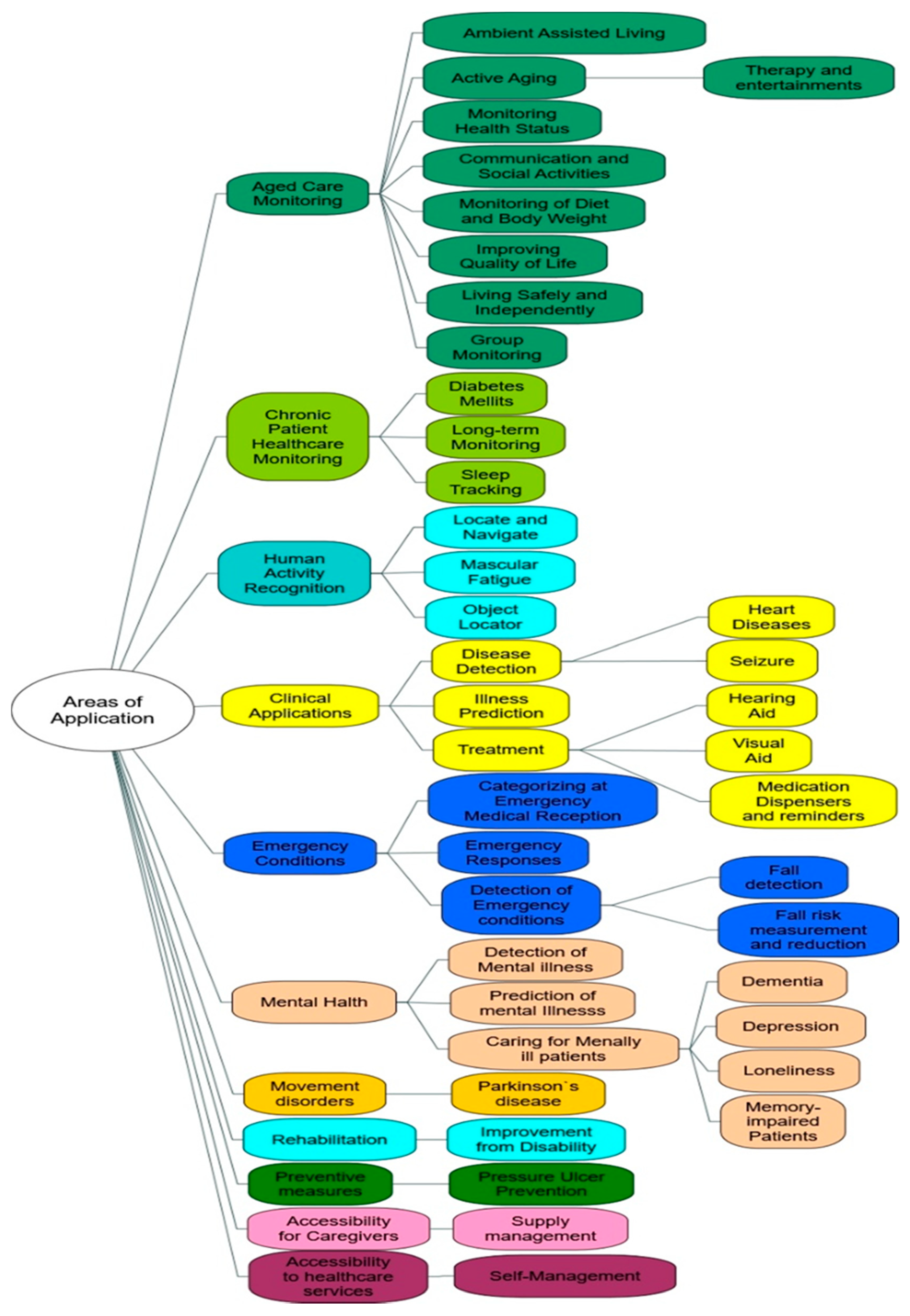
4. Results and Discussions
4.1. Cardiovascular Diseases (CVDs)
4.1.1. Vital Signs
4.1.2. Pulse Rhythms
4.1.3. Coronary Heart Disease
4.1.4. Stroke
4.1.5. Dyslipidemia
4.2. Diabetes Mellitus
4.3. Chronic Kidney Disease
4.4. Parkinson’s Disease
4.5. Sleep Tracking
4.6. Monitoring of Treatment and Rehabilitation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Disease | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Which data helps or supports clinicians for early diagnosis? | Which data helps to measure that the patient is responding well to treatment? | What types of data are needed to access in a follow up? | |
| Coronary Heart Disease |
|
|
|
| Stroke |
|
|
|
| Diabetes Mellitus |
|
|
|
| Hypertension |
|
|
|
| Chronic Renal Disease |
|
|
|
| Delirium |
|
|
|
| Parkinson’s Disease |
|
|
|
| Falls |
|
|
| Disease | Do We Have Any IoT to Support in Getting the Data? | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|---|
| Coronary Heart Disease | Yes |
|
|
|
| No |
|
|
| |
| Stroke | Yes |
|
|
|
| No |
|
|
| |
| Diabetes Mellitus | Yes |
|
|
|
| No |
|
|
| |
| Hypertension | Yes |
|
|
|
| No |
|
|
| |
| Chronic Renal Disease | Yes |
|
|
|
| No |
|
|
| |
| Delirium | Yes |
|
|
|
| No |
|
|
| |
| Parkinson’s Disease | Yes |
|
| |
| No |
|
|
| |
| Falls | Yes |
|
| |
| No |
|
|
References
- Cho, J. Current Status and Prospects of Health-Related Sensing Technology in Wearable Devices. J. Health Eng. 2019, 2019, 3924508. [Google Scholar] [CrossRef] [PubMed]
- Brüssow, H. What is health? Microb. Biotechnol. 2013, 6, 341–348. [Google Scholar] [CrossRef] [PubMed]
- United Nations. World Population Ageing 2019: Highlights; Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2019. [Google Scholar]
- Super Seniors. Our Aging Population. Available online: http://www.superseniors.msd.govt.nz/about-superseniors/media/key-statistics.html (accessed on 11 January 2020).
- De Meijer, C.; Wouterse, B.; Polder, J.; Koopmanschap, M. The effect of population aging on health expenditure growth: A critical review. Eur. J. Ageing 2013, 10, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; Dohan, M.; Veldandi, H. Digital Transformation Strategies for Healthcare Providers: Perspectives from Senior Leadership. In Proceedings of the 24th Americas Conference on Information Systems, New Orleans, LA, USA, 16 August 2018. [Google Scholar]
- Tun, S.Y.Y.; Madanian, S.; Mirza, F. Internet of things (IoT) applications for elderly care: A reflective review. Aging Clin. Exp. Res. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nikou, S.; Agahari, W.; Keijzer-Broers, W.; De Reuver, M. Digital healthcare technology adoption by elderly people: A capability approach model. Telemat. Inform. 2020, 53, 101315. [Google Scholar] [CrossRef]
- Al-Khafajiy, M.; Baker, T.; Chalmers, C.; Asim, M.; Kolivand, H.; Fahim, M.; Waraich, A. Remote health monitoring of elderly through wearable sensors. Multimed. Tools Appl. 2019, 78, 24681–24706. [Google Scholar] [CrossRef]
- Pinto, S.; Cabral, J.; Gomes, T. We-care: An IoT-based health care system for elderly people. In Proceedings of the 2017 IEEE International Conference on Industrial Technology (ICIT), Toronto, ON, Canada, 22–25 March 2017; pp. 1378–1383. [Google Scholar]
- Islam, S.M.R.; Kwak, D.; Kabir, H.; Hossain, M.; Kwak, K.-S. The Internet of Things for Health Care: A Comprehensive Survey. IEEE Access 2015, 3, 678–708. [Google Scholar] [CrossRef]
- Reddy, S. Can tech speed up emergency room care. Wall Street J. 2017. Available online: https://www.wsj.com/articles/can-tech-speed-up-emergency-room-care-1490629118 (accessed on 23 August 2017).
- Maeng, D.D.; Starr, A.E.; Tomcavage, J.F.; Sciandra, J.; Salek, D.; Griffith, D. Can telemonitoring reduce hospitalization and cost of care? A health plan’s experience in managing patients with heart failure. Popul. Health Manag. 2014, 17, 340–344. [Google Scholar] [CrossRef]
- Wagner, F.; Basran, J.; Bello-Haas, V.D. A Review of Monitoring Technology for Use with Older Adults. J. Geriatr. Phys. Ther. 2012, 35, 28–34. [Google Scholar] [CrossRef]
- Madanian, S.; Nguyen, H.H.; Mirza, F. Wearable Technology. In Encyclopedia of Gerontology and Population Aging; Gu, D., Dupre, M.E., Eds.; Springer International Publishing: Cham, Germany, 2019; pp. 1–8. [Google Scholar] [CrossRef]
- Habibzadeh, H.; Dinesh, K.; Shishvan, O.R.; Boggio-Dandry, A.; Sharma, G.; Soyata, T. A Survey of Healthcare Internet of Things (HIoT): A Clinical Perspective. IEEE Internet Things J. 2019, 7, 53–71. [Google Scholar] [CrossRef]
- Savvy. The Potentials and Pitfalls of the Artificial Pancreas. Available online: https://blogs.lt.vt.edu/savvy/potentials-pitfalls-artificial-pancreas/ (accessed on 25 September 2019).
- Wang, Z.; Yang, Z.; Dong, T. A Review of Wearable Technologies for Elderly Care that Can Accurately Track Indoor Position, Recognize Physical Activities and Monitor Vital Signs in Real Time. Sensors 2017, 17, 341. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, S.C. Wearable Sensors for Human Activity Monitoring: A Review. IEEE Sens. J. 2015, 15, 1321–1330. [Google Scholar] [CrossRef]
- Azimi, I.; Rahmani, A.; Liljeberg, P.; Tenhunen, H. Internet of things for remote elderly monitoring: A study from user-centered perspective. J. Ambient. Intell. Humaniz. Comput. 2016, 8, 273–289. [Google Scholar] [CrossRef]
- Pal, D.; Funilkul, S.; Charoenkitkarn, N.; Kanthamanon, P. Internet-of-Things and Smart Homes for Elderly Healthcare: An End User Perspective. IEEE Access 2018, 6, 10483–10496. [Google Scholar] [CrossRef]
- Foster, K.R.; Torous, J. The Opportunity and Obstacles for Smartwatches and Wearable Sensors. IEEE Pulse 2019, 10, 22–25. [Google Scholar] [CrossRef]
- Erdmier, C.; Hatcher, J.; Lee, M. Wearable device implications in the healthcare industry. J. Med. Eng. Technol. 2016, 40, 141–148. [Google Scholar] [CrossRef]
- Horwitz, L. Line between Consumer Wellness and Traditional Medicine Blurs Further. Available online: https://www.iotworldtoday.com/2020/01/28/line-between-consumer-wellness-and-traditional-medicine-blurs-further/ (accessed on 1 November 2020).
- Cruzes, D.S.; Dybå, T.; Runeson, P.; Höst, M. Case studies synthesis: A thematic, cross-case, and narrative synthesis worked example. Empir. Softw. Eng. 2015, 20, 1634–1665. [Google Scholar] [CrossRef]
- Khan, S.; Van Wynsberghe, R. Cultivating the under-mined: Cross-case analysis as knowledge mobilization. Forum Qual. Soz. Forum Qual. Soc. Res. 2008, 9. [Google Scholar] [CrossRef]
- Ralston, S.H.; Penman, I.D.; Strachan, M.W.; Hobson, R. Davidson’s Principles and Practice of Medicine, 23rd ed.; Elsevier Health Sciences: Edinburgh, UK, 2018. [Google Scholar]
- Mizokami, F.; Koide, Y.; Noro, T.; Furuta, K. Polypharmacy with Common Diseases in Hospitalized Elderly Patients. Am. J. Geriatr. Pharmacother. 2012, 10, 123–128. [Google Scholar] [CrossRef]
- Lee, P.; Cigolle, C.; Blaum, C. The Co-Occurrence of Chronic Diseases and Geriatric Syndromes: The Health and Retirement Study. J. Am. Geriatr. Soc. 2009, 57, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, I.B.; Raine, T.; Wiles, K.; Goodhart, A.; Hall, C.; O’Neill, H. Oxford Handbook of Clinical Medicine; Oxford University Press: Tonbridge, UK, 2017. [Google Scholar]
- Wockel, A.; Albert, U.-S.; Janni, W.; Scharl, A.; Kreienberg, R.; Stüber, T. The Screening, Diagnosis, Treatment, and Follow-Up of Breast Cancer. Dtsch. Aerzteblatt Online 2018, 115, 316–323. [Google Scholar] [CrossRef]
- Wöckel, A.; Festl, J.; Stüber, T.; Brust, K.; Stangl, S.; Heuschmann, P.U.; Albert, U.-S.; Budach, W.; Follmann, M.; Janni, W.; et al. Interdisciplinary Screening, Diagnosis, Therapy and Follow-up of Breast Cancer. Guideline of the DGGG and the DKG (S3-Level, AWMF Registry Number 032/045OL, December 2017)—Part 1 with Recommendations for the Screening, Diagnosis and Therapy of Breast Cancer. Geburtshilfe Frauenheilkd. 2018, 78, 927–948. [Google Scholar] [CrossRef]
- McDonagh, T.; Damy, T.; Doehner, W.; Lam, C.S.; Sindone, A.; Van Der Meer, P.; Cohen-Solal, A.; Kindermann, I.; Manito, N.; Pfister, O.; et al. Screening, diagnosis and treatment of iron deficiency in chronic heart failure: Putting the 2016 European Society of Cardiology heart failure guidelines into clinical practice. Eur. J. Hear. Fail. 2018, 20, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Pickering, D.; Marsden, J. How to measure blood glucose. Community Eye Heal. 2014, 27, 56–57. [Google Scholar]
- Kunzmann, K. FDA Approves First Cuffless Blood Pressure Monitoring Devices. Available online: https://www.mdmag.com/medical-news/fda-cuffless-blood-pressure-monitoring-devices (accessed on 31 January 2020).
- Lovett, L. FDA Clears Respiration Rate Measurement for Masimo’s Home Finger Pulse Oximeter. Available online: https://www.mobihealthnews.com/content/fda-clears-respiration-rate-measurement-masimos-home-finger-pulse-oximeter (accessed on 31 January 2020).
- Isakadze, N.; Martin, S.S. How useful is the smartwatch ECG? Trends Cardiovasc. Med. 2020, 30, 442–448. [Google Scholar] [CrossRef]
- Sadek, I.; Demarasse, A.; Mokhtari, M. Internet of things for sleep tracking: Wearables vs. nonwearables. Heal. Technol. 2019, 10, 333–340. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration. Non-Contact Infrared Thermometers. Available online: https://www.fda.gov/medical-devices/general-hospital-devices-and-supplies/non-contact-infrared-thermometers (accessed on 6 October 2020).
- Moran, D.S.; Mendal, L. Core Temperature Measurement: Methods and Current Insights. Sports Med. 2002, 32, 879–885. [Google Scholar] [CrossRef]
- Goldman, A.; Azran, H.; Stern, T.; Grinstein, M.; Wilner, D. A Novel Electronic Device for Measuring Urine Flow Rate: A Clinical Investigation. Clin. Med. Insights Trauma Intensiv. Med. 2017, 8, 8. [Google Scholar] [CrossRef]
- Arshad, R. Smart IoTs Based Urine Measurement System. Master’s Thesis, Bournemouth University, Poole, UK, 2018. [Google Scholar]
- Muneer, A.; Fati, S.M.; Fuddah, S. Smart health monitoring system using IoT based smart fitness mirror. TELKOMNIKA Telecommun. Comput. Electron. Control. 2020, 18, 317. [Google Scholar] [CrossRef]
- Ando, B.; Baglio, S.; Marletta, V.; Pistorio, A.; Dibilio, V.; Mostile, G.; Nicoletti, A.; Zappia, M. A Wearable Device to Support the Pull Test for Postural Instability Assessment in Parkinson’s Disease. IEEE Trans. Instrum. Meas. 2017, 67, 218–228. [Google Scholar] [CrossRef]
- Rovini, E.; Maremmani, C.; Cavallo, F. How Wearable Sensors Can Support Parkinson’s Disease Diagnosis and Treatment: A Systematic Review. Front. Neurosci. 2017, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Preece, J. Best Fall Detection Sensors. Available online: https://www.toptenreviews.com/best-fall-detection-sensors (accessed on 6 October 2020).
- Laport, F.; Dapena, A.; Castro, P.M.; Vazquez-Araujo, F.J.; Iglesia, D. A Prototype of EEG System for IoT. Int. J. Neural Syst. 2020, 30, 2050018. [Google Scholar] [CrossRef] [PubMed]
- Charara, S. This Tremor Tracking Wearable Lets Parkinson’s and ET Patients Take Charge. Available online: https://www.wareable.com/health-and-wellbeing/trequant-tremor-tracking-wearable-parkinsons (accessed on 31 January 2020).
- Nova Biomedical. StatSensor® and StatSensor Xpress™ Creatinine and eGFR Meters. Available online: https://www.novabio.us/statstrip-creatinine/ (accessed on 7 February 2020).
- Gia, T.N.; Sarker, V.K.; Tcarenko, I.; Rahmani, A.; Westerlund, T.; Liljeberg, P.; Tenhunen, H. Energy efficient wearable sensor node for IoT-based fall detection systems. Microprocess. Microsyst. 2018, 56, 34–46. [Google Scholar] [CrossRef]
- Majumder, A.J.A.; Elsaadany, Y.; Elsaadany, M.; Ucci, D.R.; Rahman, F. A wireless IoT system towards gait detection in stroke patients. In Proceedings of the 2017 IEEE International Conference on Pervasive Computing and Communications Workshops (PerCom Workshops), Kona, HI, USA, 13–17 March 2017; pp. 449–454. [Google Scholar]
- Achanta, S.D.M.; Karthikeyan, T. A wireless IOT system towards gait detection technique using FSR sensor and wearable IOT devices. Int. J. Intell. Unmanned Syst. 2019, 8, 43–54. [Google Scholar] [CrossRef]
- Campbell, E.; Gaffney, A.J.; Allen, B.E.; Robinson, C.T. LipidPlus: A Device to Monitor Lipid Profile and Blood Glucose. US Pharm 2015, 6, 23. [Google Scholar]
- World Health Organization. Cardiovascular Diseases. Available online: https://www.who.int/health-topics/cardiovascular-diseases/#tab=tab_1 (accessed on 5 February 2020).
- Johns Hopkins Medicine. Vital Signs (Body Temperature, Pulse Rate, Respiration Rate, Blood Pressure). Available online: https://www.hopkinsmedicine.org/health/conditions-and-diseases/vital-signs-body-temperature-pulse-rate-respiration-rate-blood-pressure (accessed on 31 January 2020).
- Cleveland Clinic. Pulse & Heart Rate. Available online: https://my.clevelandclinic.org/health/diagnostics/17402-pulse--heart-rate (accessed on 31 October 2020).
- Masimo. MightySat® Rx Fingertip Pulse Oximeter. Available online: https://www.masimo.com/products/monitors/spot-check/mightysatrx/ (accessed on 31 October 2020).
- Masimo. MightySat® Fingertip Pulse Oximeter with Bluetooth LE, RRp, & PVi. Available online: https://www.masimopersonalhealth.com/products/mightysat-fingertip-pulse-oximeter-with-bluetooth-le-rrp-pvi (accessed on 1 November 2020).
- Biobeat. Medical Smart Monitoring. Available online: https://www.bio-beat.com/ (accessed on 31 October 2020).
- Research2Guidance. How the First FDA-Cleared Cuffless Blood Pressure Solution is Paving the Way to New Remote Monitoring Technologies?—Interview with Dr. Eisenkraft, Chief Medical Officer of Biobeat. Available online: https://research2guidance.com/how-the-first-fda-cleared-cuffless-blood-pressure-solution-is-paving-the-way-to-new-remote-monitoring-technologies-interview-with-dr-eisenkraft-chief-medical-officer-of-biobeat/#:~:text=Eisenkraft%3A%20Biobeat%20devices%20enables%20cost,this%20price%20will%20be%20reduced (accessed on 1 November 2020).
- American Heart Association. New Scientific Statement on Blood Pressure Measurement in People: American Heart Association Scientific Statement. Available online: https://newsroom.heart.org/news/new-scientific-statement-on-blood-pressure-measurement-in-people (accessed on 31 October 2020).
- Georgiou, K.; Larentzakis, A.V.; Khamis, N.N.; Alsuhaibani, G.I.; Alaska, Y.A.; Giallafos, E.J. Can Wearable Devices Accurately Measure Heart Rate Variability? A Systematic Review. Folia Med. 2018, 60, 7–20. [Google Scholar] [CrossRef]
- Samsung. What Does the Heart Rate Sensor Measure? Available online: https://www.samsung.com/us/heartratesensor/ (accessed on 28 December 2019).
- Bumgarner, J.M.; Lambert, C.T.; Hussein, A.A.; Cantillon, D.J.; Baranowski, B.; Wolski, K.; Lindsay, B.D.; Wazni, O.M.; Tarakji, K.G. Smartwatch Algorithm for Automated Detection of Atrial Fibrillation. J. Am. Coll. Cardiol. 2018, 71, 2381–2388. [Google Scholar] [CrossRef]
- Apple. Taking an ECG with the ECG App on Apple Watch Series 4, Series 5, or Series 6. Available online: https://support.apple.com/en-us/HT208955 (accessed on 31 October 2020).
- January, C.T.; Wann, L.S.; Calkins, H.; Chen, L.Y.; Cigarroa, J.E.; Cleveland, J.C.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; Furie, K.L.; et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society in Collaboration With the Society of Thoracic Surgeons. Circulation 2019, 140, e125–e151. [Google Scholar] [CrossRef]
- AliveCor. This Is Your Heart × 6. Available online: https://www.alivecor.com/kardiamobile6l/ (accessed on 31 October 2020).
- Albert, D. A six-lead heart monitor on your smartphone: An interview with David Albert. Future Cardiol. 2020, 16, 9–11. [Google Scholar] [CrossRef]
- AliveCor. KardiaMobile6L. Available online: https://store.alivecor.com/products/kardiamobile6l (accessed on 1 November 2020).
- MDsave. Electrocardiogram (EKG/ECG). Available online: https://www.mdsave.com/procedures/electrocardiogram-ekg-ecg/d182ff#:~:text=On%20Mdsave%2C%20the%20cost%20of,ranges%20from%20%2476%20to%20%24240 (accessed on 1 November 2020).
- Peace, K.; Wilensky, E.M.; Frangos, S.; MacMurtrie, E.; Shields, E.; Hujcs, M.; Levine, J.; Kofke, A.; Yang, W.; Le Roux, P.D. The Use of a Portable Head CT Scanner in the Intensive Care Unit. J. Neurosci. Nurs. 2010, 42, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Bennett, M. Aging and Atherosclerosis: Mechanisms, functional consequences, and potential therapeutics for cellular senescence. Circ. Res. 2012, 111, 245–259. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.M.; Duarte, J.S.; Von Hafe, P.; Gil, V.; De Oliveira, J.N.; De Sousa, G. Standardization of laboratory and lipid profile evaluation: A call for action with a special focus in 2016 ESC/EAS dyslipidaemia guidelines—Full report. Atheroscler. Suppl. 2018, 31, e1–e12. [Google Scholar] [CrossRef]
- Hall, A. The 7 Best Cholesterol Test Kits of 2020. Available online: https://www.verywellhealth.com/best-cholesterol-test-kits-4171705 (accessed on 1 November 2020).
- International Diabetes Federation. Diabetes Facts & Figures. Available online: https://www.idf.org/aboutdiabetes/what-is-diabetes/facts-figures.html (accessed on 5 February 2020).
- World Health Organization. Diabetes. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 31 January 2020).
- Healthline. New Diabetes Test Could Be More Accurate. Available online: https://www.healthline.com/health-news/new-diabetes-test-more-accurate (accessed on 6 October 2020).
- National Institute for Health and Care Excellence. Type 2 Diabetes in Adults: Management. Available online: https://www.nice.org.uk/guidance/ng28/chapter/1-Recommendations#drug-treatment-2 (accessed on 5 February 2020).
- Diabetes.co.uk. HbA1c Test Kits: Home A1c Testing Kits. Available online: https://www.diabetes.co.uk/hba1c-meters/ (accessed on 5 February 2020).
- Zhang, C.G.; Chang, S.J.; Settu, K.; Chen, C.-J.; Liu, J.-T. High-sensitivity glycated hemoglobin (HbA1c) aptasensor in rapid-prototyping surface plasmon resonance. Sens. Actuators B Chem. 2019, 279, 267–273. [Google Scholar] [CrossRef]
- Diabetes.co.uk. Artificial Pancreas. Available online: https://www.diabetes.co.uk/artificial-pancreas.html (accessed on 25 September 2019).
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.-M.; Yang, C.-W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- National Kidney Foundation. Global Facts: About Kidney Disease. Available online: https://www.kidney.org/kidneydisease/global-facts-about-kidney-disease (accessed on 6 February 2020).
- Stevens, L.A.; Viswanathan, G.; Weiner, D.E. Chronic Kidney Disease and End-Stage Renal Disease in the Elderly Population: Current Prevalence, Future Projections, and Clinical Significance. Adv. Chronic Kidney Dis. 2010, 17, 293–301. [Google Scholar] [CrossRef]
- De Nicola, L.; Zoccali, C. Chronic kidney disease prevalence in the general population: Heterogeneity and concerns. Nephrol. Dial. Transpl. 2016, 31, 331–335. [Google Scholar] [CrossRef]
- Scanlon, B.K.; Levin, B.E.; Nation, D.A.; Katzen, H.L.; Guevara-Salcedo, A.; Singer, C.; Papapetropoulos, S. An accelerometry-based study of lower and upper limb tremor in Parkinson’s disease. J. Clin. Neurosci. 2013, 20, 827–830. [Google Scholar] [CrossRef]
- Chen, T.-Z.; Xu, G.-J.; Zhou, G.-A.; Wang, J.-R.; Chan, P.; Du, Y.-F. Postural sway in idiopathic rapid eye movement sleep behavior disorder: A potential marker of prodromal Parkinson’s disease. Brain Res. 2014, 1559, 26–32. [Google Scholar] [CrossRef]
- Romero, L.E.; Chatterjee, P.; Armentano, R.L. An IoT approach for integration of computational intelligence and wearable sensors for Parkinson’s disease diagnosis and monitoring. Health Technol. 2016, 6, 167–172. [Google Scholar] [CrossRef]
- Gulia, K.K.; Kumar, V.M. Sleep disorders in the elderly: A growing challenge. Psychogeriatrics 2018, 18, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Shelgikar, A.V.; Anderson, P.F.; Stephens, M.R. Sleep Tracking, Wearable Technology, and Opportunities for Research and Clinical Care. Chest 2016, 150, 732–743. [Google Scholar] [CrossRef] [PubMed]
- Gomes, D.; Placido, A.I.; Mó, R.; Simões, J.L.; Amaral, O.; Fernandes, I.; Lima, F.; Morgado, M.; Figueiras, A.; Herdeiro, M.T. Daily medication management and ddherence in the polymedicated elderly: A cross-sectional study in Portugal. Int. J. Environ. Res. Public Health 2020, 17, 200. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.-H.; Chen, T.-Y.; Yu, C.-R.; Shih, C.-S.; Liu, J.W.S. Smart Medication Dispenser: Design, Architecture and Implementation. IEEE Syst. J. 2010, 5, 99–110. [Google Scholar] [CrossRef]
- Varshney, U. Smart medication management system and multiple interventions for medication adherence. Decis. Support Syst. 2013, 55, 538–551. [Google Scholar] [CrossRef]
- Airehrour, D.; Madanian, S.; Abraham, A.M. Designing a memory-aid and reminder system for dementia patients and older adults. In Proceedings of the 17th International Conference on INFORMATICS in ECONOMY (IE 2018), Iasi, Romania, 17–20 May 2018; pp. 75–81. [Google Scholar]
- DLife. Advantages and Disadvantages of the Insulin Pump. Available online: https://dlife.com/advantages-and-disadvantages-of-the-insulin-pump/ (accessed on 25 September 2019).
- Bahadori, S.; Immins, T.; Wainwright, T.W. A review of wearable motion tracking systems used in rehabilitation following hip and knee replacement. J. Rehabil. Assist. Technol. Eng. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Antón, D.; Nelson, M.; Russell, T.; Goñi, A.; Illarramendi, A. Validation of a Kinect-based telerehabilitation system with total hip replacement patients. J. Telemed. Telecare 2015, 22, 192–197. [Google Scholar] [CrossRef]
- Grant, S.; Blom, A.W.; Whitehouse, M.R.; Craddock, I.; Judge, A.; Tonkin, E.L.; Gooberman-Hill, R. Using home sensing technology to assess outcome and recovery after hip and knee replacement in the UK: The HEmiSPHERE study protocol. BMJ Open 2018, 8, e021862. [Google Scholar] [CrossRef]
- Eichler, S.; Rabe, S.; Salzwedel, A.; Mueller, S.; Stoll, J.; Tilgner, N.; John, M.; Wegscheider, K.; Mayer, F.; Völler, H. Effectiveness of an interactive telerehabilitation system with home-based exercise training in patients after total hip or knee replacement: Study protocol for a multicenter, superiority, no-blinded randomized controlled trial. Trials 2017, 18, 438. [Google Scholar] [CrossRef]
- Chen, Y.; Abel, K.T.; Janecek, J.T.; Chen, Y.; Zheng, K.; Cramer, S.C. Home-based technologies for stroke rehabilitation: A systematic review. Int. J. Med. Inform. 2019, 123, 11–22. [Google Scholar] [CrossRef]
- Ortiz-Piña, M.; Salas-Fariña, Z.; Mora-Traverso, M.; Martín-Martín, L.; Galiano-Castillo, N.; García-Montes, I.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Arroyo-Morales, M.; Mesa-Ruiz, A.; et al. A home-based tele-rehabilitation protocol for patients with hip fracture called @ctivehip. Res. Nurs. Heal. 2018, 42, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, M.A.; Galea, O.A.; O’Leary, S.P.; Hill, A.J.; Russell, T.G. Real-time telerehabilitation for the treatment of musculoskeletal conditions is effective and comparable to standard practice: A systematic review and meta-analysis. Clin. Rehabil. 2016, 31, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Alaba, F.A.; Othman, M.; Hashem, I.A.T.; Alotaibi, F. Internet of Things security: A survey. J. Netw. Comput. Appl. 2017, 88, 10–28. [Google Scholar] [CrossRef]
- Jaleel, A.; Mahmood, T.; Hassan, M.A.; Bano, G.; Khurshid, S.K. Towards Medical Data Interoperability Through Collaboration of Healthcare Devices. IEEE Access 2020, 8, 132302–132319. [Google Scholar] [CrossRef]
- Conti, M.; Dehghantanha, A.; Franke, K.; Watson, S. Internet of Things security and forensics: Challenges and opportunities. Future Gener. Comput. Syst. 2018, 78, 544–546. [Google Scholar] [CrossRef]
- Mahmoud, R.; Yousuf, T.; Aloul, F.; Zualkernan, I. Internet of things (IoT) security: Current status, challenges and prospective measures. In Proceedings of the 2015 10th International Conference for Internet Technology and Secured Transactions (ICITST), London, UK, 14–16 December 2015; pp. 336–341. [Google Scholar]
- Alkhatib, S.; Waycott, J.; Buchanan, G.; Bosua, R. Privacy and the Internet of Things (IoT) Monitoring Solutions for Older Adults: A Review. Stud. Heal. Technol. Inform. 2018, 252, 8–14. [Google Scholar]
- Hussain, I. Internet of Things: Challenges and research opportunities. CSI Trans. ICT 2017, 5, 87–95. [Google Scholar] [CrossRef]
- Kiourtis, A.; Mavrogiorgou, A.; Menychtas, A.; Maglogiannis, I.; Kyriazis, D. Structurally Mapping Healthcare Data to HL7 FHIR through Ontology Alignment. J. Med. Syst. 2019, 43, 62. [Google Scholar] [CrossRef]
- Lamprinakos, G.; Mousas, A.; Boufis, A.; Karmiris, P.; Mantzouratos, S.; Kapsalis, A.; Kaklamani, D.; Venieris, I. Using FHIR to develop a healthcare mobile application. In Proceedings of the 4th International Conference on Wireless Mobile Communication and Healthcare—“Transforming healthcare through innovations in mobile and wireless technologies”, Athens, Greece, 3–5 November 2014; pp. 132–135. [Google Scholar]
- HL7 FHIR. FHIR Overview. Available online: https://www.hl7.org/fhir/overview.html (accessed on 4 October 2019).

| Ready to be Used in the Clinical Setting | Not Ready to be Used in the Clinical Setting |
|---|---|
| Pulse Rate [35,36] | ECG [37] |
| Blood Pressure [35] | REM Sleep Disorders [38] |
| Temperature [39,40] | Reversal of Sleep–Awake Cycle [38] |
| Oxygen Saturation [35,36] | Urine Output [41,42] |
| Blood Glucose [34] | Fluid Intake and Output [41,42] |
| Body Weight and Height [43] | Postural Instability [44,45] |
| Early Detection of Falls [46] | EEG [47] |
| Tremor [48] | Urinary Retention and Incontinence [7] |
| Creatinine for Glomerular Filtration Rate [49] | |
| Balance [50] | |
| Gait [51,52] Lipid Profile [53] |
| Laboratory Test | Imaging Technology | Others |
|---|---|---|
| Cardiac Enzymes | CT/CT Angiogram | Rigidity/Increased Tone |
| Urea and Electrolytes | MRI/MR Angiogram | Bradykinesia/Hypokinesia |
| HBA1C | X-rays | Poor Executive Functioning |
| Full Blood Count | Echocardiogram | Depression |
| Hemoglobin | Ultrasound | Dementia |
| Liver Function Test | Cataract | Psychosis |
| Arterial Blood Gas | Disordered Thinking | |
| Septic Screen | Euphoric, Fearful, Depressed, or | |
| Lumber Punctured | Angry | |
| Thyroid Function Test | Language Impaired | |
| Urine Albumin | Illusion/Delusion/Hallucinations | |
| ESR | Inattention | |
| Malaria | Unaware/Disorientated | |
| Memory Deficits | ||
| Vision or Cognitive Function | ||
| Conscious Level | ||
| Aspiration | ||
| Constipation and Bowel Incontinence | ||
| Freezing while Walking | ||
| Posture (Proper Positioning) |
| IoT for Data Collection | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Yes | ECG [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Blood Glucose [34] Lipid Profile [53] | ECG (Continuous) [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Blood Glucose [34] Lipid Profile [53] | ECG [37] Blood Pressure [35] Pulse Rate [35,36] Oxygen Saturation [35,36] Blood Glucose [34] Lipid Profile [53] |
| No | Pulse Rhythms Cardiac Enzymes Chest X-ray Echocardiogram Urea and Electrolytes | Pulse Rhythms Cardiac Enzymes Chest X-ray Echocardiogram Urea and Electrolytes | Pulse Rhythms Chest X-ray Echocardiogram Urea and Electrolytes |
| IoT for Data Collection | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Yes | ECG [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Blood Glucose [34] Lipid Profile [53] | ECG (Continuous) [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Gait [51,52] | ECG [37] Blood Pressure [35] Pulse Rate [35,36] Oxygen Saturation [35,36] Pressure Ulcers [7] Gait [51,52] |
| No | CT/CT Angiogram MRI/MR Angiogram Echocardiogram Full Blood Count ESR Pulse Rhythms | Conscious Level Pulse Rhythms Aspiration Urinary Retention and Incontinence Constipation and Bowel Incontinence Posture | Conscious Level Pulse Rhythms Aspiration Urinary Retention and Incontinence Constipation and Bowel Incontinence Posture (Proper Positioning) |
| IoT for Data Collection | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Yes | Blood Glucose [34] | Blood Glucose [34] ECG [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Lipid Profile [53] | Blood Glucose [34] ECG [37] Blood Pressure [35] Pulse Rate [35,36] Temperature [39] Oxygen Saturation [35,36] Body Weight [43] Urine Output for Nephropathy [41,42] Lipid Profile [53] |
| No | HBA1C | HBA1c Chest X-ray Urea and Electrolytes | HBA1c Urea and Electrolytes Peripheral Pulses for Ischemia (DM Foot) Cataract |
| IoT for Data Collection | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Yes | Urine Output [41,42] Creatinine for Glomerular Filtration Rate (GFR) [49] Lipid Profile [53] | Blood Pressure [35] Pulse Rate [35,36] ECG [37] Blood Glucose [34] Fluid Intake and Output [41,42] Lipid Profile [53] | ECG [37] Blood Pressure [35] Pulse Rate [35,36] Oxygen Saturation [35,36] Blood Glucose [34] Lipid Profile [53] |
| No | Pulse Rhythms Cardiac Enzymes Chest X-ray Echocardiogram Urea and Electrolytes | Pulse Rhythms Cardiac Enzymes Chest X-ray Echocardiogram Urea and Electrolytes | Pulse Rhythms Cardiac Enzymes Chest X-ray Echocardiogram Urea and Electrolytes |
| IoT for Data Collection | Screening and Diagnosis | Monitoring | Follow Up |
|---|---|---|---|
| Yes | Tremors [48] | Postural Instability [44,45] REM Sleep Disorders [38] | |
| No | Rigidity or Increased Tone Bradykinesia or Hypokinesia | Poor Executive Functioning Freezing while Walking | Depression Dementia Psychosis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tun, S.Y.Y.; Madanian, S.; Parry, D. Clinical Perspective on Internet of Things Applications for Care of the Elderly. Electronics 2020, 9, 1925. https://doi.org/10.3390/electronics9111925
Tun SYY, Madanian S, Parry D. Clinical Perspective on Internet of Things Applications for Care of the Elderly. Electronics. 2020; 9(11):1925. https://doi.org/10.3390/electronics9111925
Chicago/Turabian StyleTun, Soe Ye Yint, Samaneh Madanian, and Dave Parry. 2020. "Clinical Perspective on Internet of Things Applications for Care of the Elderly" Electronics 9, no. 11: 1925. https://doi.org/10.3390/electronics9111925
APA StyleTun, S. Y. Y., Madanian, S., & Parry, D. (2020). Clinical Perspective on Internet of Things Applications for Care of the Elderly. Electronics, 9(11), 1925. https://doi.org/10.3390/electronics9111925







