A New Hybrid Sensor Design Based on a Patch Antenna with an Enhanced Sensitivity Using Frequency-Selective Surfaces (FSS) in the Microwave Region for Non-Invasive Glucose Concentration Level Monitoring
Abstract
1. Introduction
- This work proposes a novel, low-cost, and simple technique for microwave region identification using a traditional antenna and an FSS.
- In this study, the FSS structure in the hybrid sensor structure was designed to increase the sensitivity rather than the antenna performance, as is customary.
- The relatively high sensitivity of the hybrid sensor structure compared to that reported in the literature, its compact size, and the thin and flexible nature of the base material used makes this sensor structure a strong alternative for wearable sensor applications.
2. Materials and Methods
2.1. Dielectric Properties of Glucose–Distilled Water Solution
| 2.0542 × 10−14 | −1.8774 × 10−10 | 5.0497 × 10−7 | 6.1540 × 10−6 | 5.4444 | |
| − | −3.3059 × 10−11 | 3.0253 × 10−7 | −1.4017 × 10−3 | 74.0419 | |
| − | 7.7826 × 10−13 | −1.0155 × 10−9 | 1.5363 × 10−4 | 8.7785 |
2.2. Design and Fabrication of Hybrid Sensors
3. Simulation and Experiment Setup
4. Results
4.1. Dielectric Properties
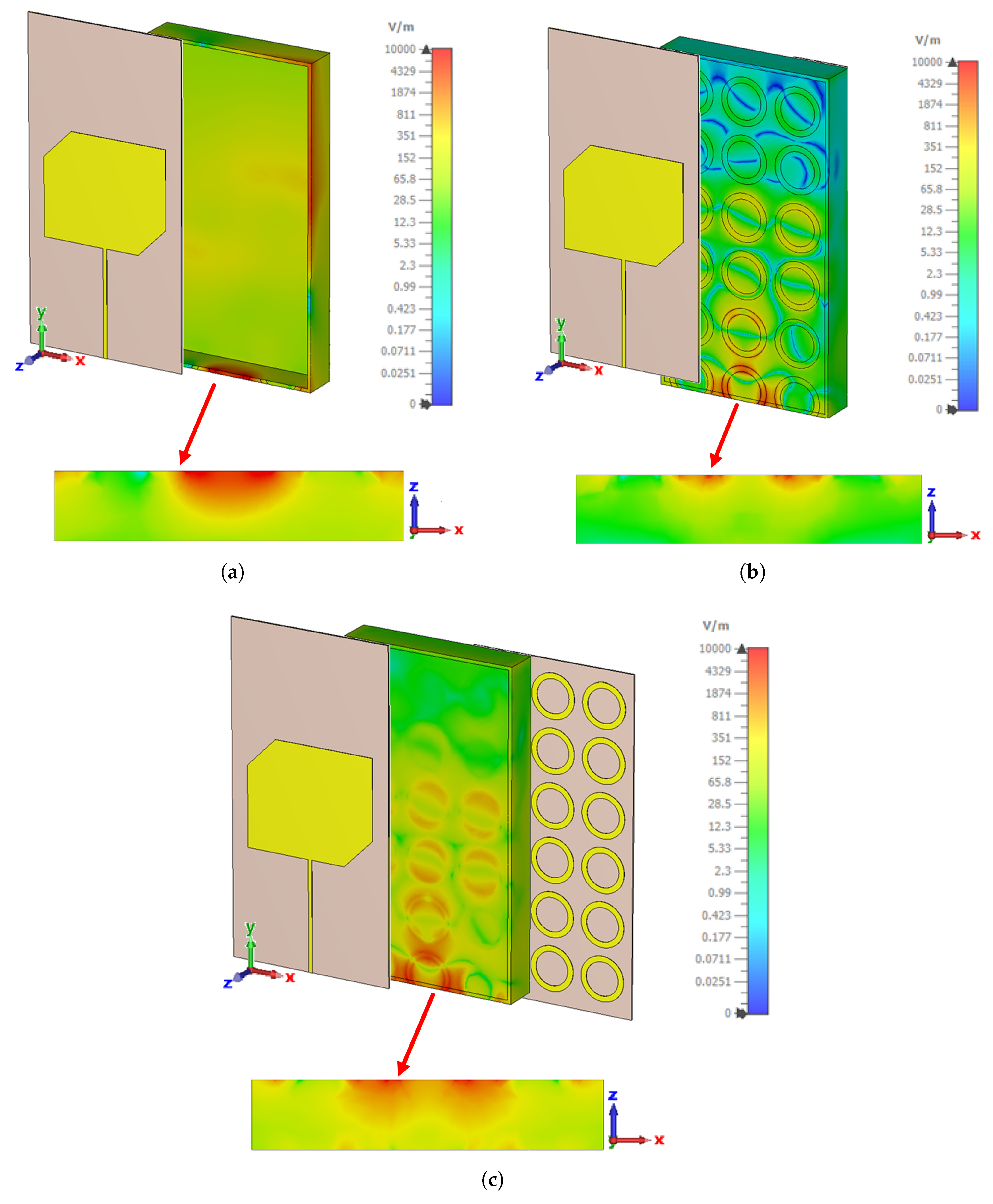
4.2. Simulation Results and Experimental Verification
4.3. Regression Model Performance
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| STPA | Square-Truncated Patch Antenna |
| FSS | Frequency-Selective Surface |
| Loss Tangent | |
| Relative Permittivity | |
| SUT | Sample Under Test |
| FOM | Figure of Merit |
| Resonance Frequency of the Antenna | |
| C | Glucose Concentration |
| Coefficient of Determination | |
| RF | Radio-Frequency |
| RR | Ring Resonator |
| HSSTPA | Hybrid Sensor with a Square-Truncated Patch Antenna |
| PLA | Polylactic Acid |
| SOL | Sort–Open–Load |
| Reflection Coefficient | |
| Real Part of Complex Permittivity | |
| Imaginary Part of Complex Permittivity | |
| SSTPA | Sensor with a Square-Truncated Patch Antenna |
| S | Sensitivity |
| Absolute Frequency Shift | |
| Change in Glucose–Distilled Water Solutions | |
| Q | Quality Factor |
| FWHM | Full Width at Half Maximum |
References
- Ekmekci, E.; Kose, U.; Cinar, A.; Ertan, O.; Ekmekci, Z. The use of metamaterial type double-sided resonator structures in humidity and concentration sensing applications. Sens. Actuators A Phys. 2019, 297, 111559. [Google Scholar] [CrossRef]
- Abduljabar, A.A.; Clark, N.; Lees, J.; Porch, A. Dual Mode Microwave Microfluidic Sensor for Temperature Variant Liquid Characterization. IEEE Trans. Microw. Theory Tech. 2017, 65, 2572–2582. [Google Scholar] [CrossRef]
- Niksan, O.; Jain, M.C.; Shah, A.; Zarifi, M.H. A Nonintrusive Flow Rate Sensor Based on Microwave Split-Ring Resonators and Thermal Modulation. IEEE Trans. Microw. Theory Tech. 2022, 70, 1954–1963. [Google Scholar] [CrossRef]
- Nath, U.; Banerjee, S.; Santini, C.; Citroni, R.; Mangini, F.; Frezza, F. Simple and Cost-Effective Design of a THz-Metamaterial-Based Hybrid Sensor on a Single Substrate. Sensors 2025, 25, 3660. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, A.; Withayachumnankul, W.; Al-Sarawi, S.F.; Abbott, D. Metamaterial-Inspired Rotation Sensor with Wide Dynamic Range. IEEE Sens. J. 2014, 14, 2609–2614. [Google Scholar] [CrossRef]
- Wu, W.; Ren, M.; Pi, B.; Cai, W.; Xu, J. Displacement sensor based on plasmonic slot metamaterials. Appl. Phys. Lett. 2016, 108, 073106. [Google Scholar] [CrossRef]
- Kose, U.; Kartal, M. A Low-Cost Microwave Hybrid Biosensor for Glucose Concentration Sensing. In Proceedings of the 2024 International Conference on Applied and Theoretical Electricity (ICATE), Craiova, Romania, 24–26 October 2024; IEEE: New York, NY, USA, 2024; pp. 1–5. [Google Scholar] [CrossRef]
- Kose, U.; Kartal, M. Non-Invasive Microwave Glucose Sensor by Using a Hybrid Sensor Composed of a Frequency Selective Surface and Microstrip Patch Antenna. In Proceedings of the 2023 Photonics & Electromagnetics Research Symposium (PIERS), Prague, Czech Republic, 3–6 July 2023; IEEE: New York, NY, USA, 2023; pp. 1361–1367. [Google Scholar] [CrossRef]
- Kandwal, A.; Sharma, Y.D.; Jasrotia, R.; Kit, C.C.; Lakshmaiya, N.; Sillanpää, M.; Liu, L.W.; Igbe, T.; Kumari, A.; Sharma, R.; et al. A comprehensive review on electromagnetic wave based non-invasive glucose monitoring in microwave frequencies. Heliyon 2024, 10, e37825. [Google Scholar] [CrossRef]
- Raj, S.; Tripathi, S.; Upadhyay, G.; Tripathi, S.S.; Tripathi, V.S. An Electromagnetic Band Gap-Based Complementary Split Ring Resonator Loaded Patch Antenna for Glucose Level Measurement. IEEE Sens. J. 2021, 21, 22679–22687. [Google Scholar] [CrossRef]
- Saleh, G.; Ateeq, I.S.; Al-Naib, I. Glucose Level Sensing Using Single Asymmetric Split Ring Resonator. Sensors 2021, 21, 2945. [Google Scholar] [CrossRef]
- Villena Gonzales, W.; Mobashsher, A.; Abbosh, A. The Progress of Glucose Monitoring—A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors. Sensors 2019, 19, 800. [Google Scholar] [CrossRef]
- Mahnashi, Y.; Qureshi, K.K.; Al-Shehri, A.A.; Attia, H. Design and Experimental Validation of a Noninvasive Glucose Monitoring System Using RF Antenna-Based Biosensor. IEEE Sens. J. 2023, 23, 2856–2864. [Google Scholar] [CrossRef]
- Harnsoongnoen, S. A Non-Contact Method for Detecting and Distinguishing Chloride and Carbonate Salts Based on Dielectric Properties Using a Microstrip Patch Sensor. Chemosensors 2023, 11, 158. [Google Scholar] [CrossRef]
- Di Filippo, D.; Sunstrum, F.N.; Khan, J.U.; Welsh, A.W. Non-Invasive Glucose Sensing Technologies and Products: A Comprehensive Review for Researchers and Clinicians. Sensors 2023, 23, 9130. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, T.; Foster, R.; Hao, Y. Radio-Frequency and Microwave Techniques for Non-Invasive Measurement of Blood Glucose Levels. Diagnostics 2019, 9, 6. [Google Scholar] [CrossRef]
- Jeong, J.M.; Bien, F.; Lee, J.G. Design of Orientation-Independent Non-Invasive Glucose Sensor Based on Meta-Structured Antenna. Electronics 2025, 14, 2295. [Google Scholar] [CrossRef]
- Xie, W.Q.; Gong, Y.X.; Yu, K.X. Rapid quantitative detection of glucose content in glucose injection by reaction headspace gas chromatography. J. Chromatogr. A 2017, 1520, 143–146. [Google Scholar] [CrossRef]
- Armoogum, V.; Boodhoo, K. Full optimization and validation of an HPLC method for the quantitative analysis of total sugars in a soft drink. Bull. Chem. Soc. Ethiop. 2020, 34, 419–426. [Google Scholar] [CrossRef]
- Bartosiak, M.; Giersz, J.; Jankowski, K. Analytical monitoring of selenium nanoparticles green synthesis using photochemical vapor generation coupled with MIP-OES and UV–Vis spectrophotometry. Microchem. J. 2019, 145, 1169–1175. [Google Scholar] [CrossRef]
- Chang, J.H.; Lo, Y.L.; Cai, Z.Y.; Chang, C.M.; Wang, B.Y. Extraction of optical rotation angle in a turbid media using polarimetry system with single rotating polarizer. Measurement 2022, 199, 111508. [Google Scholar] [CrossRef]
- Wu, H.; Yan, Y.; Huang, Q.; Liang, G.; Qiu, F.; Ye, Z.; Liu, D. A simple, cost-effective and selective analysis of glucose via electrochemical impedance sensing based on copper and nitrogen co-doped carbon quantum dots. New J. Chem. 2020, 44, 12723–12728. [Google Scholar] [CrossRef]
- Ebrahimi, A.; Scott, J.; Ghorbani, K. Microwave reflective biosensor for glucose level detection in aqueous solutions. Sens. Actuators A Phys. 2020, 301, 111662. [Google Scholar] [CrossRef]
- Lyandres, O.; Yuen, J.M.; Shah, N.C.; VanDuyne, R.P.; Walsh, J.T.; Glucksberg, M.R. Progress Toward an In Vivo Surface-Enhanced Raman Spectroscopy Glucose Sensor. Diabetes Technol. Ther. 2008, 10, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Tang, X.; Wang, C.; Wu, Q. Integrated System for High-Sensitivity Differential Microwave Glucose Sensing. IEEE Sens. J. 2024, 24, 2975–2984. [Google Scholar] [CrossRef]
- Juan, C.G.; Potelon, B.; Quendo, C.; Garcia-Martinez, H.; Avila-Navarro, E.; Bronchalo, E.; Sabater-Navarro, J.M. Study of Qu-Based Resonant Microwave Sensors and Design of 3-D-Printed Devices Dedicated to Glucose Monitoring. IEEE Trans. Instrum. Meas. 2021, 70, 8005716. [Google Scholar] [CrossRef]
- Dhakal, R.; Kim, E.; Jo, Y.H.; Kim, S.S.; Kim, N.Y. Characterization of micro-resonator based on enhanced metal insulator semiconductor capacitor for glucose recognition. Med. Eng. Phys. 2017, 41, 55–62. [Google Scholar] [CrossRef]
- Tiwari, N.K.; Singh, S.P.; Mondal, D.; Akhtar, M.J. Flexible biomedical RF sensors to quantify the purity of medical grade glycerol and glucose concentrations. Int. J. Microw. Wirel. Technol. 2020, 12, 120–130. [Google Scholar] [CrossRef]
- Turgul, V.; Kale, I. Permittivity extraction of glucose solutions through artificial neural networks and non-invasive microwave glucose sensing. Sens. Actuators A Phys. 2018, 277, 65–72. [Google Scholar] [CrossRef]
- Yi, Z.; Wang, C. Noninvasive Glucose Sensors Using Defective-Ground-Structure Coplanar Waveguide. IEEE Sens. J. 2023, 23, 195–201. [Google Scholar] [CrossRef]
- Govind, G.; Akhtar, M.J. Metamaterial-Inspired Microwave Microfluidic Sensor for Glucose Monitoring in Aqueous Solutions. IEEE Sens. J. 2019, 19, 11900–11907. [Google Scholar] [CrossRef]
- Gharbi, M.E.; Fernández-García, R.; Gil, I. Textile Antenna-Sensor for In Vitro Diagnostics of Diabetes. Electronics 2021, 10, 1570. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, X.; Ren, M.; Mao, S.; Dhakal, R.; Kim, N.Y.; Li, Y.; Yao, Z. Microfluidic microwave biosensor based on biomimetic materials for the quantitative detection of glucose. Sci. Rep. 2022, 12, 15961. [Google Scholar] [CrossRef]
- El Gharbi, M.; Fernández-García, R.; Ahyoud, S.; Gil, I. A review of flexiblewearable antenna sensors: Design, fabrication methods, and applications. Materials 2020, 13, 3781. [Google Scholar] [CrossRef] [PubMed]
- Kose, U.; Kartal, M. Impedance Matching of Microstrip Patch Antennas by Using Frequency Selective Surfaces (FSS). In Proceedings of the 2022 30th Signal Processing and Communications Applications Conference (SIU), Safranbolu, Turkey, 15–18 May 2022; IEEE: New York, NY, USA, 2022; pp. 1–4. [Google Scholar] [CrossRef]
- Anwar, R.; Mao, L.; Ning, H. Frequency Selective Surfaces: A Review. Appl. Sci. 2018, 8, 1689. [Google Scholar] [CrossRef]
- Gangwar, D.; Das, S.; Yadava, R.L.; Kanaujia, B.K. Frequency Selective Surface as Superstrate on Wideband Dielectric Resonator Antenna for Circular Polarization and Gain Enhancement. Wirel. Pers. Commun. 2017, 97, 3149–3163. [Google Scholar] [CrossRef]
- Cole, K.S.; Cole, R.H. Dispersion and absorption in dielectrics: II. Direct current characteristics. J. Chem. Phys. 1942, 10, 98–105. [Google Scholar] [CrossRef]
- Karacolak, T.; Moreland, E.C.; Topsakal, E. Cole-cole model for glucose-dependent dielectric properties of blood plasma for continuous glucose monitoring. Microw. Opt. Technol. Lett. 2013, 55, 1160–1164. [Google Scholar] [CrossRef]
- Cinar, A.; Basaran, S.C. 3D-printed sensor design based on metamaterial absorber for characterization of solid and liquid materials. Sens. Actuators A Phys. 2024, 368, 115166. [Google Scholar] [CrossRef]
- CST STUDIO SUITE®. version 2025. Software for Electromagnetic Simulation. Dassault Systèmes, the 3DEXPERIENCE® Company: Darmstadt, Germany, 2025. [Google Scholar]
- Wang, S.; Wang, W.; Zheng, Y. Reconfigurable Multimode Microwave Sensor with Resonance and Transmission Sensing Capabilities for Noninvasive Glucose Monitoring. IEEE Trans. Microw. Theory Tech. 2024, 72, 3102–3117. [Google Scholar] [CrossRef]
- Cen, C.; Yi, Z.; Zhang, G.; Zhang, Y.; Liang, C.; Chen, X.; Tang, Y.; Ye, X.; Yi, Y.; Wang, J.; et al. Theoretical design of a triple-band perfect metamaterial absorber in the THz frequency range. Results Phys. 2019, 14, 102463. [Google Scholar] [CrossRef]
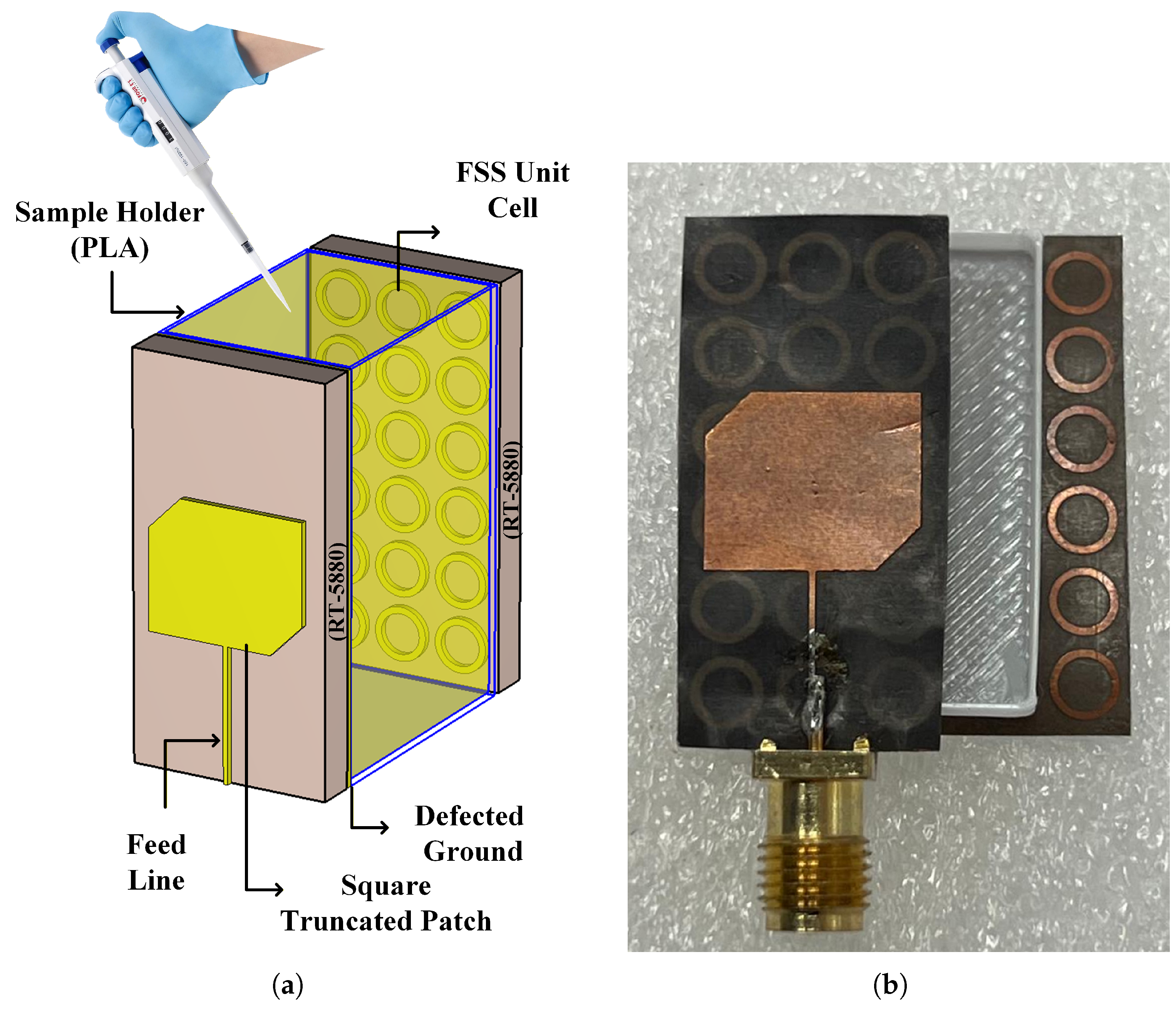





| Parts of Hybrid Sensor | Antenna | Defected Ground of Antenna | FSS | |||
|---|---|---|---|---|---|---|
| Design Parameters | Pxa | 15.00 | rg | 2.00 | Pxf | 15.00 |
| Pya | 30.00 | wg | 0.50 | Pyf | 30.00 | |
| Lxa | 12.00 | sgx | 0.75 | rf | 2.00 | |
| Lya | 10.00 | sgy | 1.125 | wf | 0.50 | |
| wfe | 0.355 | ugx | 0.75 | sfx | 0.75 | |
| u | 4.50 | ugy | 0.75 | sfy | 1.125 | |
| − | − | − | − | ufx | 0.75 | |
| − | − | − | − | ufy | 0.75 | |
| Sample (mg/dL) | Freq. (GHz) | Debye Model | Measurement | ||
|---|---|---|---|---|---|
| 0 | 6.010 | 72.156 | 22.114 | 72.074 | 22.092 |
| 72 | 6.050 | 71.964 | 22.225 | 71.864 | 22.303 |
| 216 | 6.060 | 71.758 | 22.212 | 71.829 | 22.339 |
| 300 | 6.090 | 71.597 | 22.323 | 71.687 | 22.461 |
| 500 | 6.140 | 71.292 | 22.458 | 71.388 | 22.517 |
| 600 | 6.150 | 71.188 | 22.484 | 71.226 | 22.778 |
| 1000 | 6.170 | 70.892 | 22.543 | 70.815 | 22.575 |
| 1500 | 6.200 | 70.617 | 22.657 | 70.616 | 22.801 |
| 3000 | 6.260 | 69.890 | 23.029 | 69.495 | 23.136 |
| 4000 | 6.270 | 69.327 | 23.224 | 69.200 | 23.373 |
| 5000 | 6.290 | 69.038 | 23.404 | 68.606 | 23.565 |
| Sample (mg/dL) | SSTPA Sim. | HSSTPA Sim. | HSSTPA Meas. | |||
|---|---|---|---|---|---|---|
| (GHz) | (dB) | (GHz) | (dB) | (GHz) | (dB) | |
| 0 | 5.664 | −11.540 | 6.008 | −18.624 | 6.013 | −7.997 |
| 72 | 5.664 | −11.499 | 6.048 | −19.564 | 6.050 | −16.852 |
| 216 | 5.660 | −11.546 | 6.052 | −20.775 | 6.058 | −20.544 |
| 300 | 5.672 | −11.484 | 6.096 | −19.818 | 6.091 | −24.343 |
| 500 | 5.676 | −11.489 | 6.136 | −19.539 | 6.145 | −13.382 |
| 600 | 5.688 | −11.454 | 6.144 | −20.181 | 6.155 | −14.053 |
| 1000 | 5.700 | −11.534 | 6.156 | −19.552 | 6.170 | −14.028 |
| 1500 | 5.700 | −11.491 | 6.188 | −21.517 | 6.205 | −27.541 |
| 3000 | 5.724 | −11.570 | 6.252 | −22.403 | 6.262 | −14.384 |
| 4000 | 5.732 | −11.545 | 6.264 | −22.312 | 6.273 | −16.394 |
| 5000 | 5.748 | −11.555 | 6.276 | −22.615 | 6.290 | −17.245 |
| , | 0.084 | 0.015 | 0.268 | 3.991 | 0.277 | 9.248 |
| S (kHz/mgdL−1), (dB/mgdL−1) | 16.800 | 0.030 | 53.600 | 7.981 | 55.436 | 18.497 |
| Ref. | Area (mm2) | Freq. Range (GHz) | Sensor | Concentration (mg/dL) | Sensing Parameters | Substrate | Sensitivity (S) (kHz/(mg/dL)) |
|---|---|---|---|---|---|---|---|
| [30] | 22 × 12 | 4.46–4.48 | coplanar waveguide | 0–1800 | RT-5880 | 3.88 | |
| [23] | N.A. | 2.4–2.6 | complementary split-ring resonator | 5–80 | RO-4350 | 5 | |
| [29] | 34 × 16 | 3.4–3.6 | microwave resonator integrated coplanar waveguide | 0–2000 | RO-3006 | 11 | |
| [28] | N.A. | 0.6–0.8 | interdigitated capacitor resonator-etched coplanar waveguide | 0–8000 | PET polyester | 20 | |
| [31] | 50 × 20 | 1.25–1.50 | improved split-ring resonator | 0–5000 | RT-6006 | 26 | |
| [25] | 42 × 40 | 1.8–2.5 | differential microwave resonator with defected ground structure | 0–360 | N.A. | 56.3 | |
| [42] | 100 × 35 | 1.47–3.43 | reconfigurable multi-mode sensing integrated with spoof surface plasmon polariton and spoof-localized surface plasmon | 0–800 | RO-4003 | 98.6 | |
| [33] | 25 × 25 | 3.6–4.0 | triple-ring microstrip patch antenna with defective ground structures | 50–500 | Teflon | 245 | |
| [17] | 55 × 55 | 4.0–4.5 | hexagonal patch | 50–250 | RT-5880 | 352 | |
| [32] | 35 × 35 | 2.25–2.5 | monopole antenna | 0–190 | Textile | 350 | |
| this work | 30 × 15 | 6–6.4 | HSSTPA | 0–5000 | RT-5880 | 55.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kose, U.; Sili, G.; Doken, B.; Saygili, E.S.; Akleman, F.; Kartal, M. A New Hybrid Sensor Design Based on a Patch Antenna with an Enhanced Sensitivity Using Frequency-Selective Surfaces (FSS) in the Microwave Region for Non-Invasive Glucose Concentration Level Monitoring. Electronics 2026, 15, 427. https://doi.org/10.3390/electronics15020427
Kose U, Sili G, Doken B, Saygili ES, Akleman F, Kartal M. A New Hybrid Sensor Design Based on a Patch Antenna with an Enhanced Sensitivity Using Frequency-Selective Surfaces (FSS) in the Microwave Region for Non-Invasive Glucose Concentration Level Monitoring. Electronics. 2026; 15(2):427. https://doi.org/10.3390/electronics15020427
Chicago/Turabian StyleKose, Umut, Guliz Sili, Bora Doken, Emre Sedar Saygili, Funda Akleman, and Mesut Kartal. 2026. "A New Hybrid Sensor Design Based on a Patch Antenna with an Enhanced Sensitivity Using Frequency-Selective Surfaces (FSS) in the Microwave Region for Non-Invasive Glucose Concentration Level Monitoring" Electronics 15, no. 2: 427. https://doi.org/10.3390/electronics15020427
APA StyleKose, U., Sili, G., Doken, B., Saygili, E. S., Akleman, F., & Kartal, M. (2026). A New Hybrid Sensor Design Based on a Patch Antenna with an Enhanced Sensitivity Using Frequency-Selective Surfaces (FSS) in the Microwave Region for Non-Invasive Glucose Concentration Level Monitoring. Electronics, 15(2), 427. https://doi.org/10.3390/electronics15020427






