Bradykinesia Detection in Parkinson’s Disease Using Smartwatches’ Inertial Sensors and Deep Learning Methods
Abstract
1. Introduction
- This study evaluates the potential of accelerometer and gyroscope sensors embedded in commodity smartwatches to detect bradykinesia severity using a set of standardized exercises. This approach can present an unobtrusive solution for bradykinesia monitoring in ambulatory and non-supervised environments using low-cost devices instead of using proprietary monitoring devices or sensors.
- Different feature extraction methodologies proposed in the related literature are reproduced and evaluated with the data collected using commodity smartwatches. This task is performed to compare the predictive power of approaches based on ML and DL. In addition, the potential of different data representations and data augmentation techniques is evaluated with the aim of improving the performance of the systems for automatic bradykinesia severity scoring.
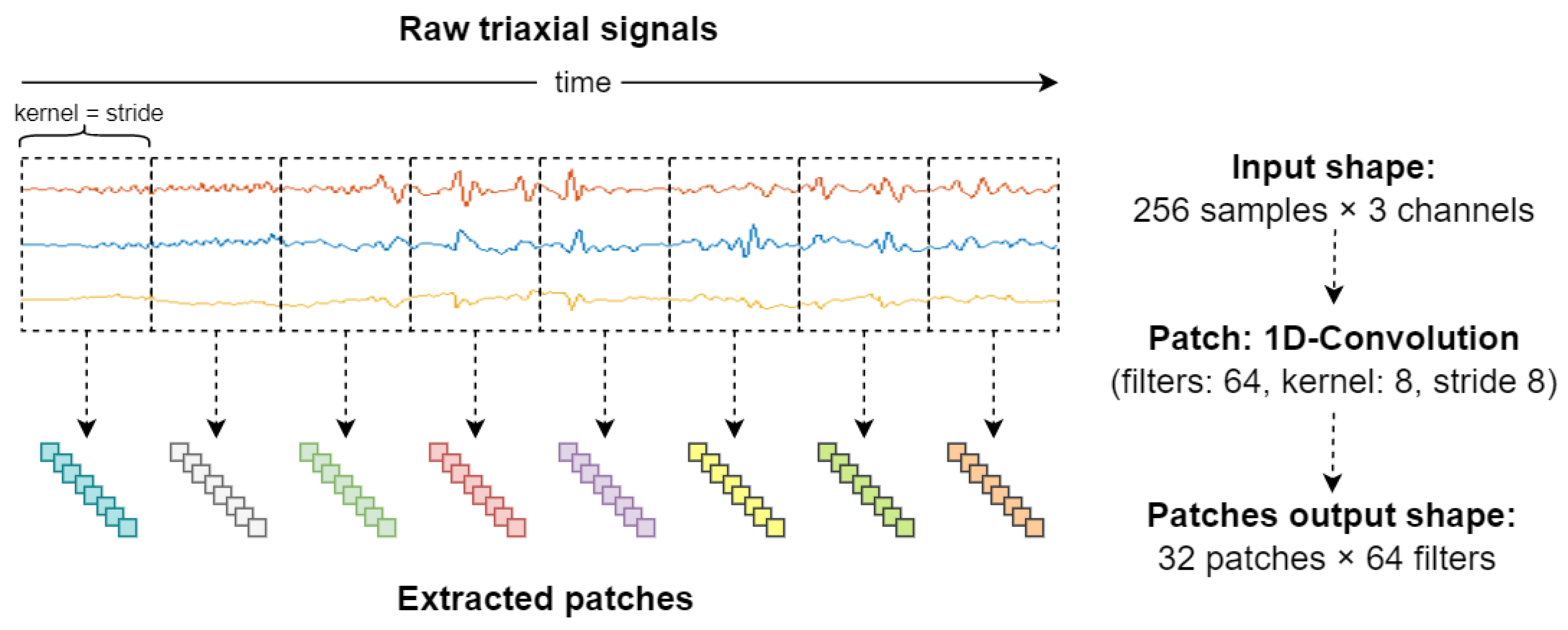
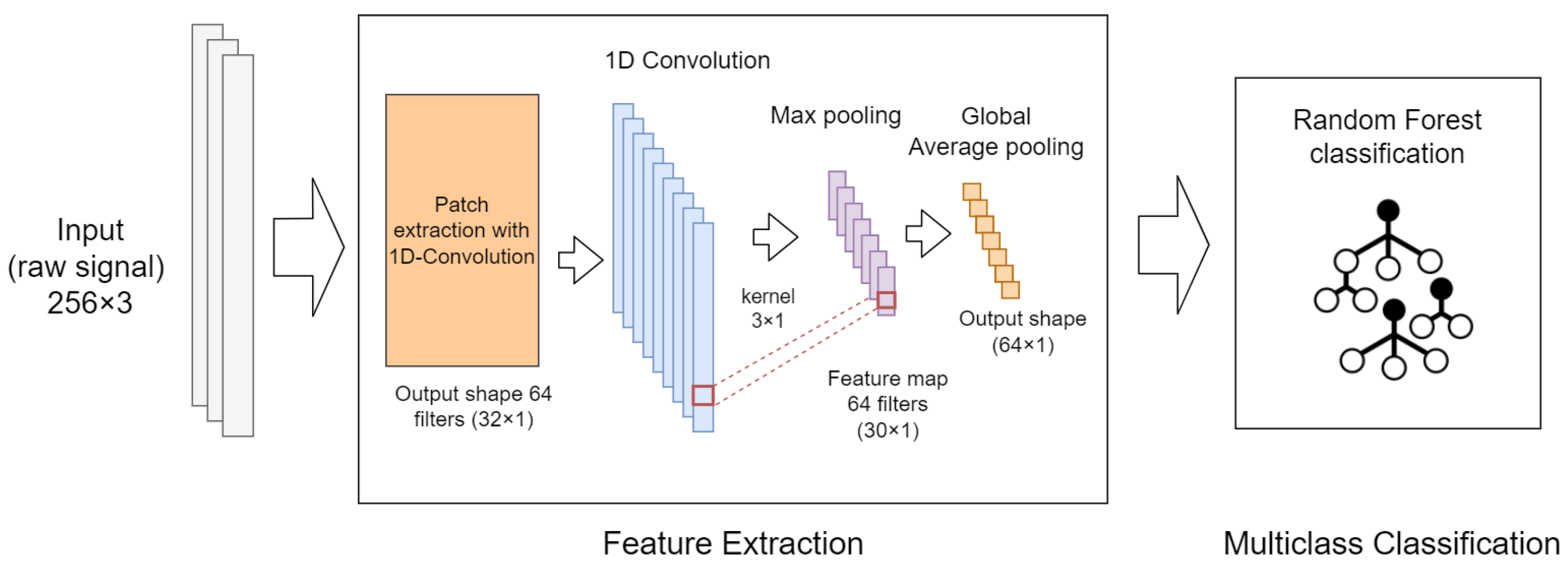
- This work also introduces the use of convolutional neural networks (CNN) with patch input (implemented with 1D-convolutional layers) for automatic temporal window contextualization. The patch input strategy is proposed as a mechanism to automatically split and project the data of a single (multi-channel) sliding window into another dimension that can be exploited by classification algorithms. Additionally, the proposed approach is evaluated using an end-to-end neural network, and in combination with a Random Forest (RF) classifier located at the top of the neural network.
- Finally, a methodology for the aggregation of a set of predictions (severity ratings) obtained from the classifiers during a single clinical visit is proposed and evaluated. This methodology is carried out with the aim of improving the outcomes of the bradykinesia assessment by providing a single severity indicator of the motor function of the upper limbs.
2. Related Work
3. Materials and Methods
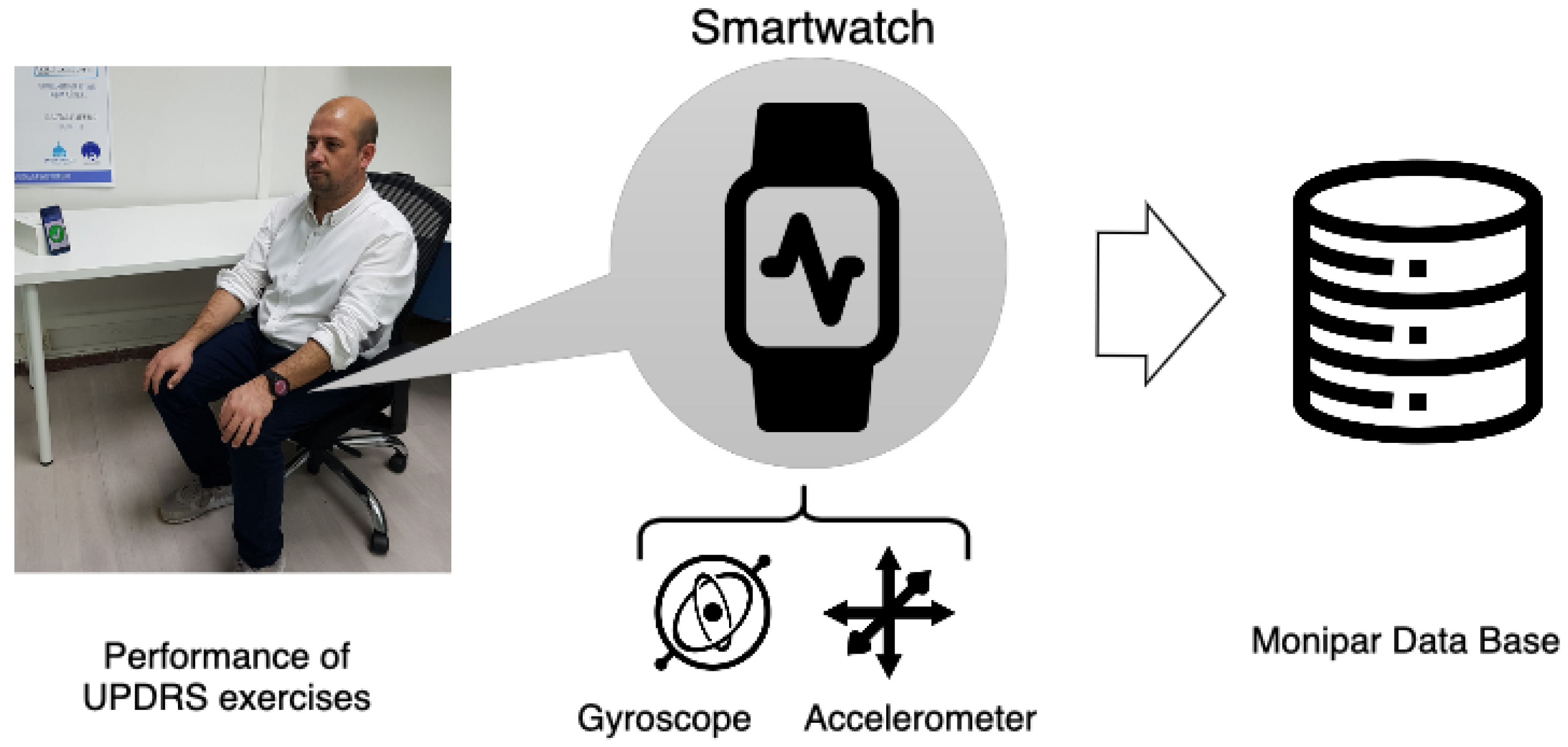
3.1. Bradykinesia Dataset
3.1.1. Data Acquisition
3.1.2. Data Labeling
3.2. Signal Preprocessing
3.2.1. Filtering and Segmentation
3.2.2. Feature Extraction
3.2.3. Data Augmentation
3.3. Classification Algorithms
3.4. Session-Based Analysis
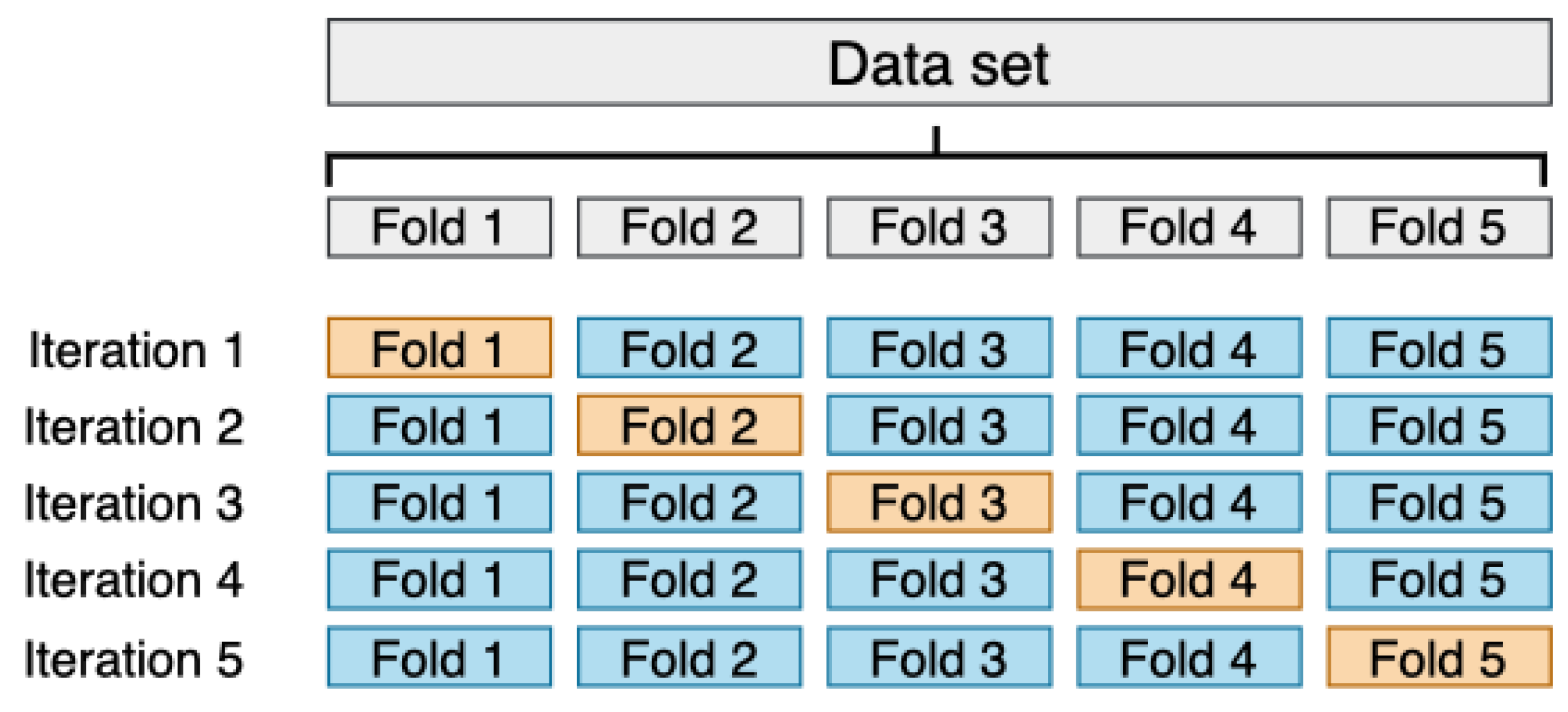
3.5. Evaluation Methodology
4. Experiments and Results
4.1. Baseline
4.2. Classification Methods
4.3. Results of the Session-Based Analysis
4.4. Summary of the Findings Observed in the Experiments
- The use of the gyroscope sensors presents better results in bradykinesia detection than the use of only the accelerometer data or the combination of accelerometer and gyroscope data.
- DL approaches provided better results than classic ML classifiers fed with extracted features. In addition, the use of the proposed method (CNN with PI and RF classification) presents competitive results for bradykinesia severity detection at the window level.
- Data augmentation methods have a positive effect on classification performance and its effect is more pronounced in the DL-based approaches.
- The use of data transformation methods such as FFT does not provide better results, compared to raw inertial signals.
- Using DL approaches with an RF model as the final classification layer does not significantly improve performance.
- The aggregation of the window-level predictions from a single clinical visit presents a slight increase in the accuracy and increases the correlation between the automatic and the clinical evaluation.
5. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | Artificial intelligence |
| ADAM | Adaptive moment estimation |
| ADLs | Activities of daily living |
| AUC | Area under the curve |
| CNN | Convolutional neural network |
| CV | Cross-validation |
| DA | Data augmentation |
| DL | Deep learning |
| DNN | Deep neural network |
| FFT | Fast Fourier Transform |
| GAP | Global average pooling |
| kNN | k-nearest neighbors |
| IMU | Inertial measurement unit |
| LR | Linear regression |
| ML | Machine learning |
| MLP | Multi-layer perceptron |
| PCA | Principal component analysis |
| PD | Parkinson’s disease |
| QoL | Quality of life |
| ReLU | Rectified linear unit |
| RF | Random forest |
| ROC | Receiver operating characteristic |
| RMSE | Root mean square error |
| SMOTE | Synthetic minority over-sampling technique |
References
- Pringsheim, T.; Jette, N.; Frolkis, A.; Steeves, T.D. The prevalence of Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 2014, 29, 1583–1590. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Kalia, L.; Lang, A. Parkinson’s disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Hou, J.; Lai, E. Non-motor Symptoms of Parkinson’s Disease. Int. J. Gerontol. 2007, 1, 53–64. [Google Scholar] [CrossRef]
- Williams, D.; Watt, H.; Lees, A. Predictors of falls and fractures in bradykinetic rigid syndromes: A retrospective study. J. Neurol. Neurosurg. Psychiatry 2006, 77, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Bronte-Stewart, H. Postural instability in idiopathic Parkinson’s disease: The role of medication and unilateral pallidotomy. Brain 2002, 125, 2100–2114. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Lorincz, K.; Hughes, R.; Huggins, N.; Growdon, J.; Standaert, D.; Akay, M.; Dy, J.; Welsh, M.; Bonato, P. Monitoring motor fluctuations in patients with Parkinson’s disease using wearable sensors. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Davie, C. A review of Parkinson’s disease. Br. Med. Bull. 2008, 86, 109–127. [Google Scholar] [CrossRef]
- Fahn, S. Parkinson disease, the effect of levodopa, and the ELLDOPA trial. Arch. Neurol. 1999, 56, 529–535. [Google Scholar] [CrossRef]
- Olanow, C.W.; Stern, M.B.; Sethi, K. The scientific and clinical basis for the treatment of Parkinson’s disease. Neurology 2009, 72, S1–S136. [Google Scholar] [CrossRef] [PubMed]
- Parkinson Study Group. Evaluation of dyskinesias in a pilot, randomized, placebo-controlled trial of remacemide in advanced Parkinson disease. Arch. Neurol. 2001, 58, 1660–1668. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.J.; Daniel, S.E.; Blankson, S.; Lees, A.J. A clinicopathologic study of 100 cases of Parkinson’s disease. Arch. Neurol. 1993, 50, 140–148. [Google Scholar] [CrossRef]
- Berardelli, A. Pathophysiology of bradykinesia in Parkinson’s disease. Brain 2001, 124, 2131–2146. [Google Scholar] [CrossRef] [PubMed]
- Vingerhoets, F.; Schulzer, M.; Calne, D.; Snow, B. Which clinical sign of Parkinson’s disease best reflects the nigrostriatal lesion? Ann. Neurol. 1997, 41, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Monje, M.H.G.; Foffani, G.; Obeso, J.; Sànchez-Ferro, Á. New Sensor and Wearable Technologies to Aid in the Diagnosis and Treatment Monitoring of Parkinson’s Disease. Annu. Rev. Biomed. Eng. 2019, 21, 111–143. [Google Scholar] [CrossRef] [PubMed]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. Off. J. Mov. Disord. Soc. 2008, 23, 2129–2170. [Google Scholar] [CrossRef]
- Borzì, L.; Varrecchia, M.; Sibille, S.; Olmo, G.; Artusi, C.A.; Fabbri, M.; Rizzone, M.G.; Romagnolo, A.; Zibetti, M.; Lopiano, L. Smartphone-Based Estimation of Item 3.8 of the MDS-UPDRS-III for Assessing Leg Agility in People with Parkinson’s Disease. IEEE Open J. Eng. Med. Biol. 2020, 1, 140–147. [Google Scholar] [CrossRef]
- Espay, A.J.; Hausdorff, J.M.; Sánchez-Ferro, Á.; Klucken, J.; Merola, A.; Bonato, P.; Paul, S.S.; Horak, F.B.; Vizcarra, J.A.; Mestre, T.A.; et al. A Roadmap for Implementation of Patient-Centered Digital Outcome Measures in Parkinson’s disease Obtained Using Mobile Health Technologies. Mov. Disord. 2019, 34, 657–663. [Google Scholar] [CrossRef]
- Luis-Martínez, R.; Monje, M.H.; Antonini, A.; Sánchez-Ferro, Á.; Mestre, T.A. Technology-enabled care: Integrating multidisciplinary care in Parkinson’s disease through digital technology. Front. Neurol. 2020, 11, 575975. [Google Scholar] [CrossRef]
- Virginia Anikwe, C.; Friday Nweke, H.; Chukwu Ikegwu, A.; Adolphus Egwuonwu, C.; Uchenna Onu, F.; Rita Alo, U.; Wah Teh, Y. Mobile and wearable sensors for data-driven health monitoring system: State-of-the-art and future prospect. Expert Syst. Appl. 2022, 202, 117362. [Google Scholar] [CrossRef]
- Tauţan, A.M.; Ionescu, B.; Santarnecchi, E. Artificial intelligence in neurodegenerative diseases: A review of available tools with a focus on machine learning techniques. Artif. Intell. Med. 2021, 117, 102081. [Google Scholar] [CrossRef] [PubMed]
- Rovini, E.; Maremmani, C.; Cavallo, F. How Wearable Sensors Can Support Parkinson’s Disease Diagnosis and Treatment: A Systematic Review. Front Neurosci. 2017, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Hubble, R.; Naughton, G.; Silburn, P.; Cole, M.H. Wearable Sensor Use for Assessing Standing Balance and Walking Stability in People with Parkinson’s Disease: A Systematic Review. PLoS ONE 2015, 10, e0123705. [Google Scholar] [CrossRef]
- Borzì, L.; Olmo, G.; Artusi, C.A.; Fabbri, M.; Rizzone, M.G.; Romagnolo, A.; Zibetti, M.; Lopiano, L. A new index to assess turning quality and postural stability in patients with Parkinson’s disease. Biomed. Signal Process. Control 2020, 62, 102059. [Google Scholar] [CrossRef]
- Mei, J.; Desrosiers, C.; Frasnelli, J. Machine Learning for the Diagnosis of Parkinson’s Disease: A Review of Literature. Front. Aging Neurosci. 2021, 13, 633752. [Google Scholar] [CrossRef]
- Cubo, E.; Mir Rivera, P.; Sánchez Ferro, Á. Manual SEN de Nuevas Tecnologías en Trastornos del Movimiento; Ediciones SEN: Madrid, Spain, 2021. [Google Scholar]
- Heldman, D.A.; Urrea-Mendoza, E.; Lovera, L.C.; Schmerler, D.A.; Garcia, X.; Mohammad, M.E.; McFarlane, M.C.U.; Giuffrida, J.P.; Espay, A.J.; Fernandez, H.H. App-based bradykinesia tasks for clinic and home assessment in Parkinson’s disease: Reliability and responsiveness. J. Park. Dis. 2017, 7, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, R.I.; Kotschet, K.; Arfon, S.; Xu, Z.M.; Johnson, W.; Drago, J.; Evans, A.; Kempster, P.; Raghav, S.; Horne, M.K. Automated assessment of bradykinesia and dyskinesia in Parkinson’s disease. J. Park. Dis. 2012, 2, 47–55. [Google Scholar] [CrossRef]
- Dunnewold, R.J.; Hoff, J.I.; van Pelt, H.C.; Fredrikze, P.Q.; Wagemans, E.A.; van Hilten, B.J. Ambulatory quantitative assessment of body position, bradykinesia, and hypokinesia in Parkinson’s disease. J. Clin. Neurophysiol. 1998, 15, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Salarian, A. Ambulatory Monitoring of Motor Functions in Patients with Parkinson’s Disease Using Kinematic Sensors; Technical Report; EPFL: Lausanne, Switzerland, 2006. [Google Scholar]
- Shawen, N.; O’Brien, M.K.; Venkatesan, S.; Lonini, L.; Simuni, T.; Hamilton, J.L.; Ghaffari, R.; Rogers, J.A.; Jayaraman, A. Role of data measurement characteristics in the accurate detection of Parkinson’s disease symptoms using wearable sensors. J. Neuroeng. Rehabil. 2020, 17, 52. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, N.; Demanuele, C.; Zhang, H.; Volfson, D.; Ho, B.; Erb, M.K.; Patel, S. Development of digital biomarkers for resting tremor and bradykinesia using a wrist-worn wearable device. NPJ Digit. Med. 2020, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Parisi, F.; Ferrari, G.; Giuberti, M.; Contin, L.; Cimolin, V.; Azzaro, C.; Albani, G.; Mauro, A. Body-Sensor-Network-Based Kinematic Characterization and Comparative Outlook of UPDRS Scoring in Leg Agility, Sit-to-Stand, and Gait Tasks in Parkinson’s Disease. IEEE J. Biomed. Health Inform. 2015, 19, 1777–1793. [Google Scholar] [CrossRef]
- Giuberti, M.; Ferrari, G.; Contin, L.; Cimolin, V.; Azzaro, C.; Albani, G.; Mauro, A. Automatic UPDRS Evaluation in the Sit-to-Stand Task of Parkinsonians: Kinematic Analysis and Comparative Outlook on the Leg Agility Task. IEEE J. Biomed. Health Inform. 2015, 19, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Aghanavesi, S.; Bergquist, F.; Nyholm, D.; Senek, M.; Memedi, M. Motion sensor-based assessment of Parkinson’s disease motor symptoms during leg agility tests: Results from levodopa challenge. IEEE J. Biomed. Health Inform. 2019, 24, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Bengio, Y. Deep learning of representations: Looking forward. In Statistical Language and Speech Processing, Proceedings of the First International Conference, SLSP 2013, Tarragona, Spain, 29–31 July 2013; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–37. [Google Scholar]
- Glorot, X.; Bengio, Y. Understanding the difficulty of training deep feedforward neural networks. In Proceedings of the Thirteenth International Conference on Artificial Intelligence and Statistics, Sardinia, Italy, 13–15 May 2010; Teh, Y.W., Titterington, M., Eds.; PMLR: Sardinia, Italy, 2010; Volume 9, pp. 249–256. [Google Scholar]
- Eskofier, B.M.; Lee, S.I.; Daneault, J.F.; Golabchi, F.N.; Ferreira-Carvalho, G.; Vergara-Diaz, G.; Sapienza, S.; Costante, G.; Klucken, J.; Kautz, T.; et al. Recent machine learning advancements in sensor-based mobility analysis: Deep learning for Parkinson’s disease assessment. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 655–658. [Google Scholar]
- Park, D.J.; Lee, J.W.; Lee, M.J.; Ahn, S.J.; Kim, J.; Kim, G.L.; Ra, Y.J.; Cho, Y.N.; Jeong, W.B. Evaluation for Parkinsonian Bradykinesia by deep learning modeling of kinematic parameters. J. Neural Transm. 2021, 128, 181–189. [Google Scholar] [CrossRef]
- Pfister, F.M.; Um, T.T.; Pichler, D.C.; Goschenhofer, J.; Abedinpour, K.; Lang, M.; Endo, S.; Ceballos-Baumann, A.O.; Hirche, S.; Bischl, B.; et al. High-Resolution Motor State Detection in Parkinson’s Disease Using Convolutional Neural Networks. Sci. Rep. 2020, 10, 5860. [Google Scholar] [CrossRef]
- Lonini, L.; Dai, A.; Shawen, N.; Simuni, T.; Poon, C.; Shimanovich, L.; Daeschler, M.; Ghaffari, R.; Rogers, J.A.; Jayaraman, A. Wearable sensors for Parkinson’s disease: Which data are worth collecting for training symptom detection models. Npj Digit. Med. 2018, 1, 64. [Google Scholar] [CrossRef]
- Sigcha, L.; Pavón, I.; Costa, N.; Costa, S.; Gago, M.; Arezes, P.; López, J.M.; De Arcas, G. Automatic Resting Tremor Assessment in Parkinson’s Disease Using Smartwatches and Multitask Convolutional Neural Networks. Sensors 2021, 21, 291. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1998, 50, 318. [Google Scholar] [CrossRef]
- Bao, L.; Intille, S.S. Activity recognition from user-annotated acceleration data. In Pervasive Computing, Proceedings of the Second International Conference, PERVASIVE 2004, Vienna, Austria, 21–23 April 2004; Springer: Berlin/Heidelberg, Germany, 2004; pp. 1–17. [Google Scholar]
- Bonato, P.; Sherrill, D.M.; Standaert, D.G.; Salles, S.S.; Akay, M. Data mining techniques to detect motor fluctuations in Parkinson’s disease. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2004; Volume 2, pp. 4766–4769. [Google Scholar]
- Channa, A.; Ifrim, R.C.; Popescu, D.; Popescu, N. A-WEAR bracelet for detection of hand tremor and bradykinesia in Parkinson’s patients. Sensors 2021, 21, 981. [Google Scholar] [CrossRef]
- San-Segundo, R.; Navarro-Hellín, H.; Torres-Sánchez, R.; Hodgins, J.; De la Torre, F. Increasing robustness in the detection of freezing of gait in Parkinson’s disease. Electronics 2019, 8, 119. [Google Scholar] [CrossRef]
- Sigcha, L.; Costa, N.; Pavón, I.; Costa, S.; Arezes, P.; López, J.; De Arcas, G. Deep Learning Approaches for Detecting Freezing of Gait in Parkinson’s Disease Patients through On-Body Acceleration Sensors. Sensors 2020, 20, 1895. [Google Scholar] [CrossRef]
- Sigcha, L.; Borzì, L.; Pavón, I.; Costa, N.; Costa, S.; Arezes, P.; López, J.M.; De Arcas, G. Improvement of Performance in Freezing of Gait detection in Parkinson’s Disease using Transformer networks and a single waist-worn triaxial accelerometer. Eng. Appl. Artif. Intell. 2022, 116, 105482. [Google Scholar] [CrossRef]
- Chawla, N.V.; Bowyer, K.W.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic minority over-sampling technique. J. Artif. Intell. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
- Um, T.T.; Pfister, F.M.J.; Pichler, D.; Endo, S.; Lang, M.; Hirche, S.; Fietzek, U.; Kulić, D. Data augmentation of wearable sensor data for parkinson’s disease monitoring using convolutional neural networks. In Proceedings of the 19th ACM International Conference on Multimodal Interaction, Glasgow, UK, 13–17 November 2017. [Google Scholar] [CrossRef]
- Jost, L. Entropy and diversity. Oikos 2006, 113, 363–375. [Google Scholar] [CrossRef]
- Trockman, A.; Kolter, J.Z. Patches are all you need? arXiv 2022, arXiv:2201.09792. [Google Scholar]
- Hassani, A.; Walton, S.; Shah, N.; Abuduweili, A.; Li, J.; Shi, H. Escaping the big data paradigm with compact transformers. arXiv 2021, arXiv:2104.05704. [Google Scholar]
- Li, L.; Jamieson, K.; DeSalvo, G.; Rostamizadeh, A.; Talwalkar, A. Hyperband: A Novel Bandit-Based Approach to Hyperparameter Optimization. J. Mach. Learn. Res. 2017, 18, 6765–6816. [Google Scholar]
- Kingma, D.P.; Ba, J. Adam: A Method for Stochastic Optimization. arXiv 2014, arXiv:1412.6980. [Google Scholar]
- Japkowicz, N.; Shah, M. Evaluating Learning Algorithms: A Classification Perspective; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]




| Feature Set | Number of Features | Description of the Features |
|---|---|---|
| Shawen et al. [31] | 74 | - Time domain features (24) |
| - Frequency domain features (24) | ||
| - Features extracted from the derivatives of the signals (16) | ||
| - Entropy (4) | ||
| - Peak correlation between signals (3) | ||
| - Cross-correlation delay (3) | ||
| Channa et al. [46] | 43 | - Time domain features (28) |
| - Frequency domain features (12) | ||
| - Peak correlation between signals (3) | ||
| Filtered signal (256-sample window) | 768 (256 × 3) | Filtered signal obtained from the triaxial sensors (accelerometer or gyroscope). |
| Contextual FFT | 384 (128 × 3) | Concatenated single-side FFT of two consecutive windows. A single window (256 samples) was divided into 2 windows of 128 samples before FFT computation. |
| Feature Set | Sensor Data | Accuracy | Precision | Recall | F1-Score | AUC |
|---|---|---|---|---|---|---|
| Accel | 0.731 | 0.721 | 0.731 | 0.692 | 0.889 | |
| Shawen et al. [31] | Gyro | 0.762 | 0.753 | 0.762 | 0.727 | 0.907 |
| Accel + Gyro | 0.720 | 0.705 | 0.720 | 0.673 | 0.886 | |
| Accel | 0.749 | 0.745 | 0.749 | 0.716 | 0.905 | |
| Channa et al. [46] | Gyro | 0.783 | 0.773 | 0.783 | 0.755 | 0.909 |
| Accel + Gyro | 0.749 | 0.746 | 0.749 | 0.718 | 0.893 |
| Method | Data Representation | Classifier | Accuracy | Precision | Recall | F1-Score | AUC |
|---|---|---|---|---|---|---|---|
| Baseline | Channa et al. [46] | RF (100) | 0.783 | 0.773 | 0.783 | 0.755 | 0.909 |
| Baseline + SMOTE | Channa et al. [46] | RF (100) | 0.792 | 0.702 | 0.667 | 0.680 | 0.912 |
| CNN | Raw signal | CNN + MLP | 0.675 | 0.661 | 0.675 | 0.618 | 0.687 |
| Contextual CNN 1 | FFT contextual | CNN + MLP | 0.629 | 0.561 | 0.629 | 0.563 | 0.706 |
| CNN(PI) + DA 2 | Raw signal | CNN (PI) + MLP | 0.826 | 0.733 | 0.751 | 0.738 | 0.943 |
| CNN(PI) + RF + DA 2 | Raw signal | CNN (PI) + RF (100) | 0.835 | 0.750 | 0.748 | 0.746 | 0.939 |
| Assessment Method | Accuracy | Precision | Recall | F1-Score | r | RMSE |
|---|---|---|---|---|---|---|
| Window-level | 0.835 | 0.750 | 0.748 | 0.746 | 0.82 | 0.77 |
| Session-based | 0.857 | 0.733 | 0.728 | 0.716 | 0.94 | 0.46 |
| Study | Sensor (Location) | Task | ML Model (Input) | Accuracy | AUC | r | RMSE |
|---|---|---|---|---|---|---|---|
| [17] | Smartphone (thigh) | Leg agility | MLP (features) | 0.77 | 0.92 | 0.92 | 0.42 |
| [31] | Smartwatch, IMU (wrist, hand) | gait, upper limbs exercises | RF (features) | - | 0.65 | - | - |
| [33] | IMU (thighs) | Leg agility | kNN (features + PCA) | 0.430 | - | 0.640 | - |
| [35] | IMU (ankles) | Leg agility | SVM (features) | - | - | 0.83 | 0.53 |
| [38] | IMU (forearm) | upper limbs exercises | CNN (raw data) | 0.91 (binary) | - | - | - |
| [39] | IMU (wrist, fingers) | upper limbs exercises | LR (features) | - | 0.93 | 0.85 | - |
| [40] | Accelerometer (wrist) | ADLs | CNN (raw data + DA) | - | - | 0.83 | - |
| [41] | IMU (hand) | gait, upper limbs exercises | RF (features) | - | 0.73 | - | - |
| Proposed | Smartwatch (wrist) | upper limbs exercises | CNN + RF (raw data) | 0.86 | 0.94 | 0.94 | 0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sigcha, L.; Domínguez, B.; Borzì, L.; Costa, N.; Costa, S.; Arezes, P.; López, J.M.; De Arcas, G.; Pavón, I. Bradykinesia Detection in Parkinson’s Disease Using Smartwatches’ Inertial Sensors and Deep Learning Methods. Electronics 2022, 11, 3879. https://doi.org/10.3390/electronics11233879
Sigcha L, Domínguez B, Borzì L, Costa N, Costa S, Arezes P, López JM, De Arcas G, Pavón I. Bradykinesia Detection in Parkinson’s Disease Using Smartwatches’ Inertial Sensors and Deep Learning Methods. Electronics. 2022; 11(23):3879. https://doi.org/10.3390/electronics11233879
Chicago/Turabian StyleSigcha, Luis, Beatriz Domínguez, Luigi Borzì, Nélson Costa, Susana Costa, Pedro Arezes, Juan Manuel López, Guillermo De Arcas, and Ignacio Pavón. 2022. "Bradykinesia Detection in Parkinson’s Disease Using Smartwatches’ Inertial Sensors and Deep Learning Methods" Electronics 11, no. 23: 3879. https://doi.org/10.3390/electronics11233879
APA StyleSigcha, L., Domínguez, B., Borzì, L., Costa, N., Costa, S., Arezes, P., López, J. M., De Arcas, G., & Pavón, I. (2022). Bradykinesia Detection in Parkinson’s Disease Using Smartwatches’ Inertial Sensors and Deep Learning Methods. Electronics, 11(23), 3879. https://doi.org/10.3390/electronics11233879











