UV-Based Technologies for SARS-CoV2 Inactivation: Status and Perspectives
Abstract
:1. Introduction
1.1. Control of Microorganisms and Viruses by Chemical and Physical Treatments
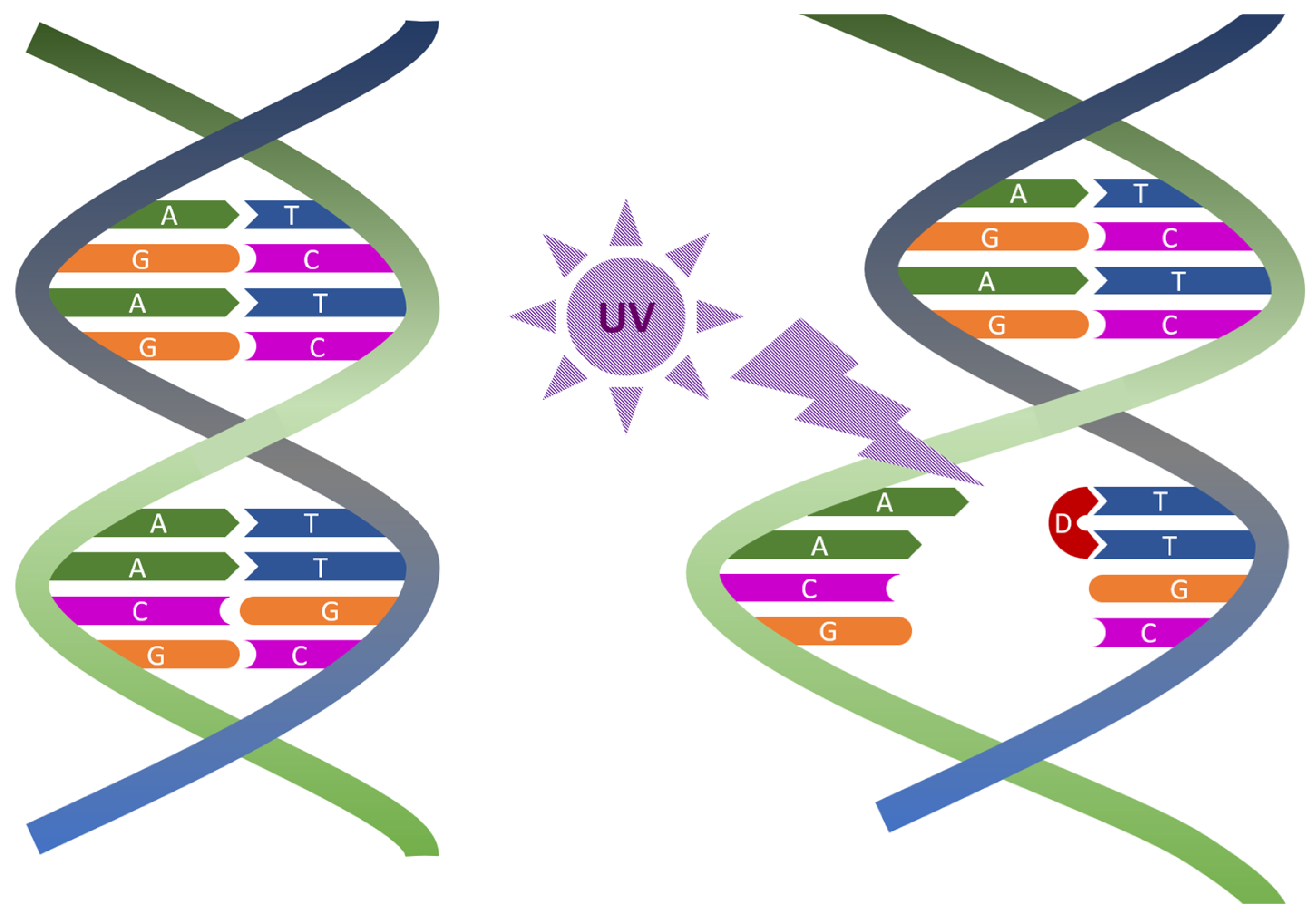
1.2. UV Irradiation in Viral Inactivation
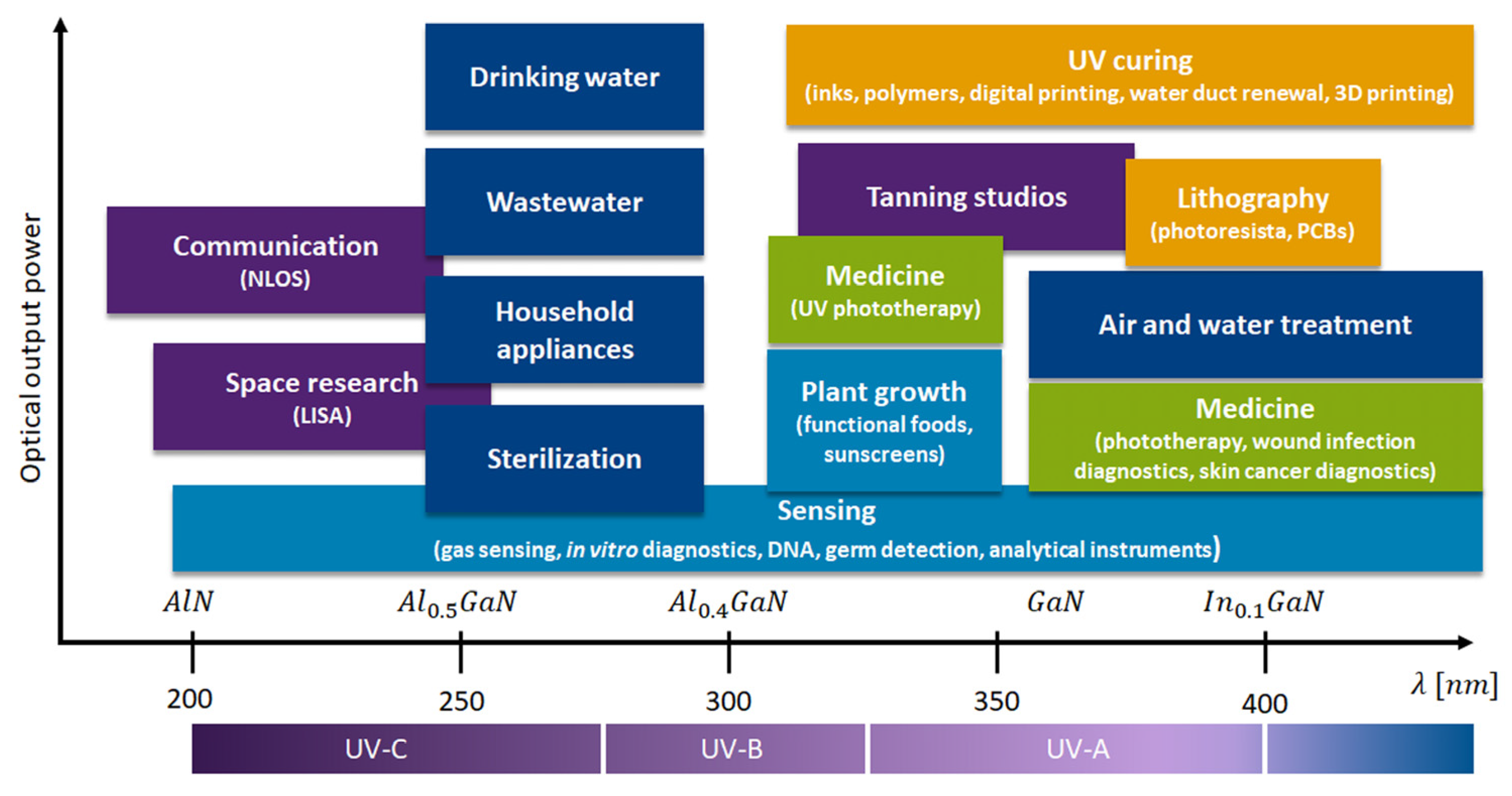
1.3. UVC Sources
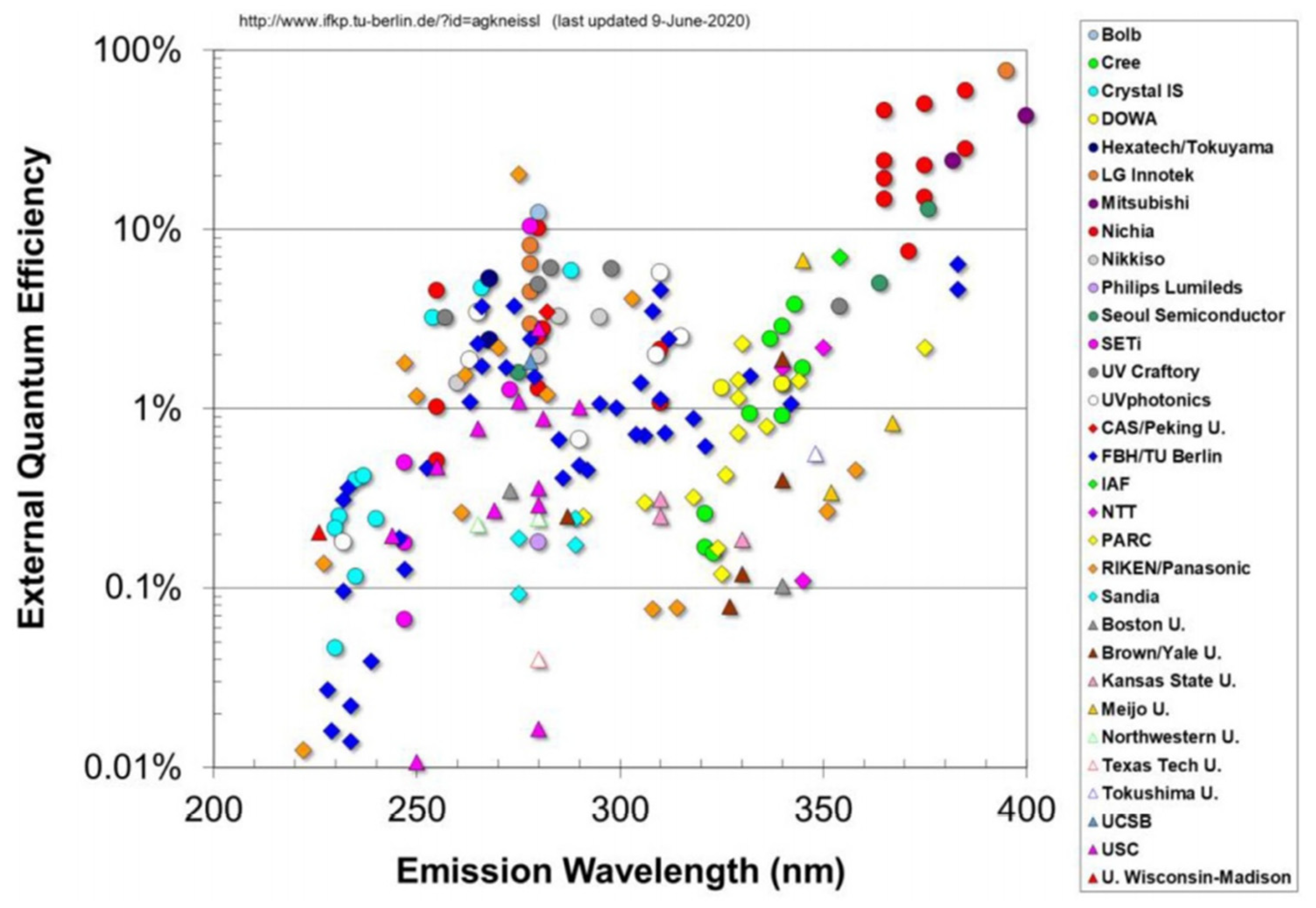
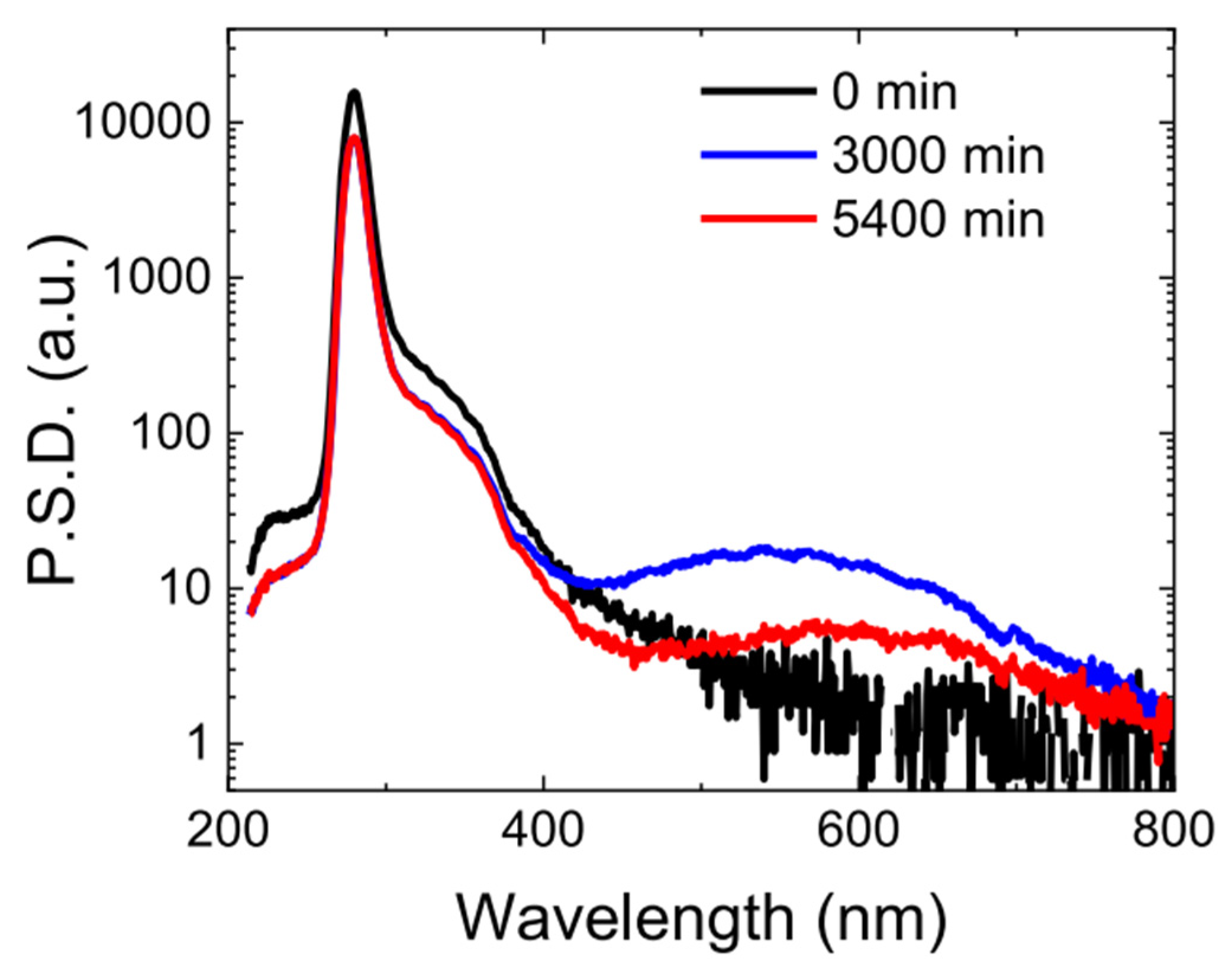
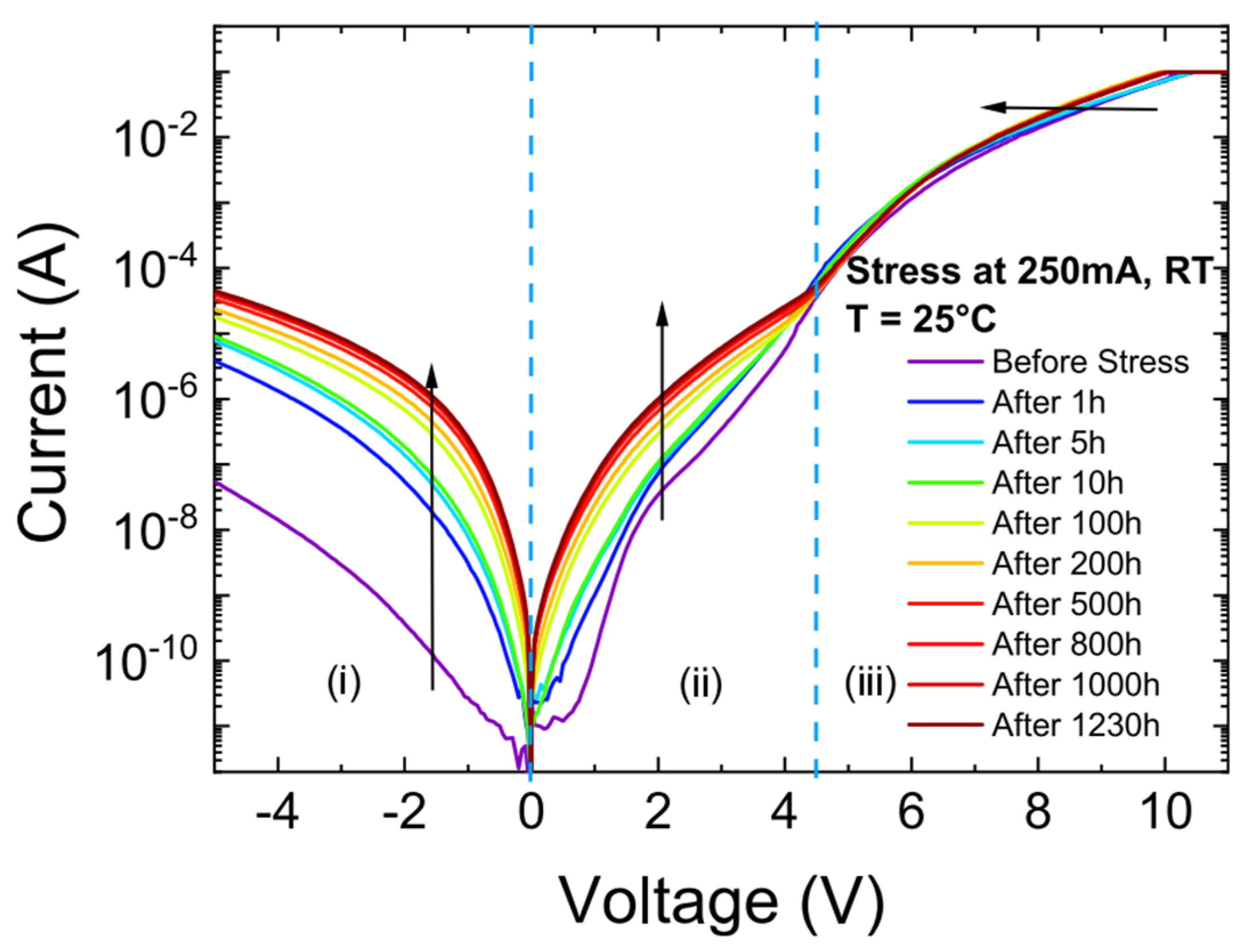
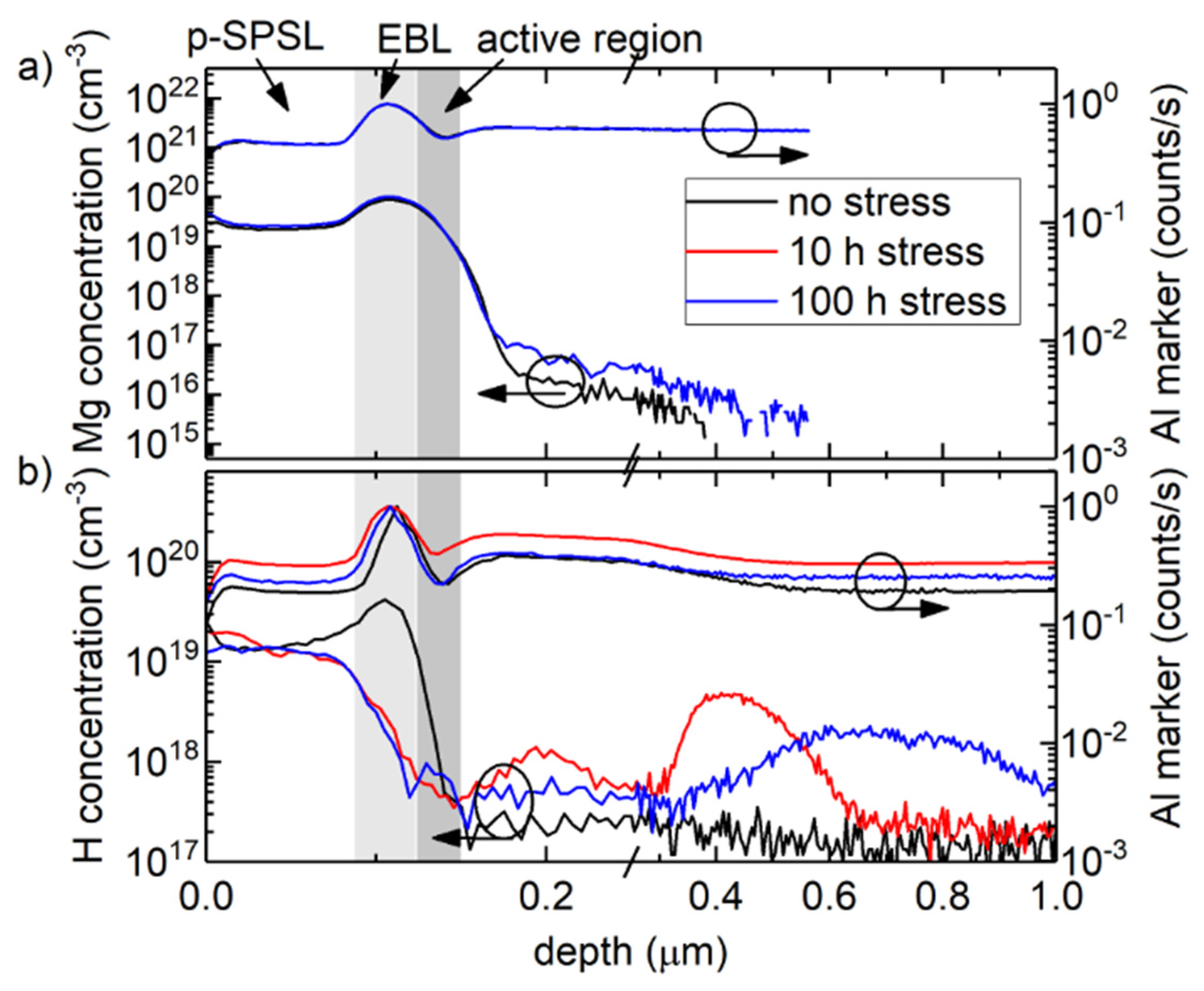
2. Challenges of UV LEDs
3. State-of-the-Art Commercial Devices: Performance and Reliability
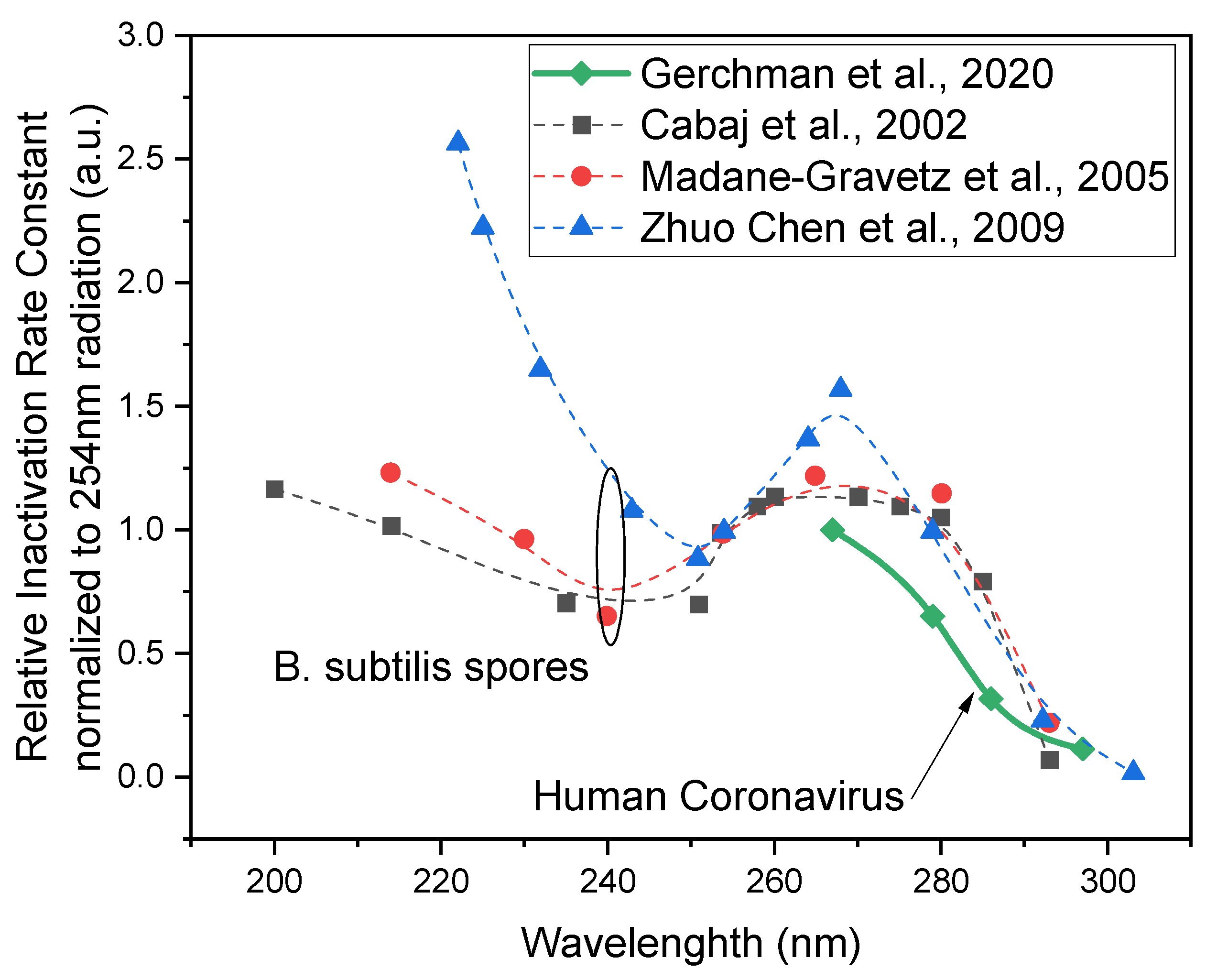
4. UV Disinfection for SARS-CoV2: Previous Reports
5. UV Disinfection for SARS-CoV2: Challenges and Perspectives for Massive Adoptions
5.1. Environmental Issues
5.2. Development of Technology to Fight Future Pandemics
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- WHO. COVID-19 Weekly Epidemiological Update 35; World Health Organization: Geneva, Switzerland, 2020; pp. 1–3. [Google Scholar]
- Grint, D.J.; Wing, K.; Williamson, E.; McDonald, H.I.; Bhaskaran, K.; Evans, D.; Evans, S.J.W.; Walker, A.J.; Hickman, G.; Nightingale, E.; et al. Case fatality risk of the SARS-CoV-2 variant of concern B.1.1.7 in England, 16 November to 5 February. Eurosurveillance 2021, 26, 2100256. [Google Scholar] [CrossRef]
- Jain, V.K.; Iyengar, K.P.; Garg, R.; Vaishya, R. Elucidating reasons of COVID-19 re-infection and its management strategies. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 1001–1006. [Google Scholar] [CrossRef]
- Stadnytskyi, V.; Bax, C.E.; Bax, A.; Anfinrud, P. The airborne lifetime of small speech droplets and their potential importance in SARS-CoV-2 transmission. Proc. Natl. Acad. Sci. USA 2020, 117, 11875–11877. [Google Scholar] [CrossRef]
- Wang, J.; Du, G. COVID-19 may transmit through aerosol. Irish J. Med. Sci. 2020, 189, 1143–1144. [Google Scholar] [CrossRef] [Green Version]
- Centers for Disease Control and Prevention Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008; Miscellaneous Inactivating Agents; CDC Website: Atlanta, GA, USA, 2013; pp. 9–13.
- Spaulding, E.H. Alcohol as a Surgical Disinfectant: Pros and cons of a much discussed topic. AORN J. 1964, 2, 67–71. [Google Scholar] [CrossRef]
- Seymour Stanton Block. Disinfection, Sterilization, and Preservation, 4th ed.; Lea & Febiger: Philadelphia, PA, USA, 1991; ISBN1 10: 0812113640. ISBN2 13: 9780812113648. [Google Scholar]
- Rutala, W.A.; Weber, D.J. Uses of inorganic hypochlorite (bleach) in health-care facilities. Clin. Microbiol. Rev. 1997, 10, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Goldust, M.; Abdelmaksoud, A.; Navarini, A.A. Hand disinfection in the combat against COVID-19. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e454–e455. [Google Scholar] [CrossRef]
- Al-Sayah, M.H. Chemical disinfectants of COVID-19: An overview. J. Water Health 2020, 18, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hudson, J.B.; Sharma, M.; Vimalanathan, S. Development of a Practical Method for Using Ozone Gas as a Virus Decontaminating Agent. Ozone Sci. Eng. 2009, 31, 216–223. [Google Scholar] [CrossRef]
- Dubuis, M.-E.; Dumont-Leblond, N.; Laliberté, C.; Veillette, M.; Turgeon, N.; Jean, J.; Duchaine, C. Ozone efficacy for the control of airborne viruses: Bacteriophage and norovirus models. PLoS ONE 2020, 15, e0231164. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Bong, C.; Lim, W.; Bae, P.K.; Abafogi, A.T.; Baek, S.H.; Shin, Y.B.; Bak, M.S.; Park, S. Fast and Easy Disinfection of Coronavirus-Contaminated Face Masks Using Ozone Gas Produced by a Dielectric Barrier Discharge Plasma Generator. Environ. Sci. Technol. Lett. 2021, 8, 339–344. [Google Scholar] [CrossRef]
- Cutler, T.D.; Zimmerman, J.J. Ultraviolet irradiation and the mechanisms underlying its inactivation of infectious agents. Anim. Health Res. Rev. 2011, 12, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, W. Ultraviolet Germicidal Irradiation Handbook; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-642-01998-2. [Google Scholar]
- Gerchman, Y.; Mamane, H.; Friedman, N.; Mandelboim, M. UV-LED disinfection of Coronavirus: Wavelength effect. J. Photochem. Photobiol. B Biol. 2020, 212, 112044. [Google Scholar] [CrossRef]
- Chen, R.Z.; Craik, S.A.; Bolton, J.R. Comparison of the action spectra and relative DNA absorbance spectra of microorganisms: Information important for the determination of germicidal fluence (UV dose) in an ultraviolet disinfection of water. Water Res. 2009, 43, 5087–5096. [Google Scholar] [CrossRef]
- Buonanno, M.; Welch, D.; Shuryak, I.; Brenner, D.J. Far-UVC light (222 nm) efficiently and safely inactivates airborne human coronaviruses. Sci. Rep. 2020, 10, 1–8. [Google Scholar] [CrossRef]
- Torres, A.E.; Lyons, A.B.; Narla, S.; Kohli, I.; Kohli, I.; Parks-Miller, A.; Ozog, D.; Hamzavi, I.H.; Lim, H.W. Ultraviolet-C and other methods of decontamination of filtering facepiece N-95 respirators during the COVID-19 pandemic. Photochem. Photobiol. Sci. 2020, 19, 746–751. [Google Scholar] [CrossRef]
- Biasin, M.; Bianco, A.; Pareschi, G.; Cavalleri, A.; Cavatorta, C.; Fenizia, C.; Galli, P.; Lessio, L.; Lualdi, M.; Tombetti, E.; et al. UV-C irradiation is highly effective in inactivating SARS-CoV-2 replication. Sci. Rep. 2021, 11, 1–7. [Google Scholar] [CrossRef]
- Cabaj, A.; Sommer, R.; Pribil, W.; Haider, T. The spectral UV sensitivity of microorganisms used in biodosimetry. Water Supply 2002, 2, 175–181. [Google Scholar] [CrossRef]
- Mamane-Gravetz, H.; Linden, K.G.; Cabaj, A.; Sommer, R. Spectral sensitivity of Bacillus subtilis spores and MS2 coliphage for validation testing of ultraviolet reactors for water disinfection. Environ. Sci. Technol. 2005, 39, 7845–7852. [Google Scholar] [CrossRef]
- Hylander, L.D.; Goodsite, M.E. Environmental costs of mercury pollution. Sci. Total Environ. 2006, 368, 352–370. [Google Scholar] [CrossRef]
- Asif Khan, M. AlGaN multiple quantum well based deep UV LEDs and their applications. Phys. Status Solidi 2006, 203, 1764–1770. [Google Scholar] [CrossRef]
- Shatalov, M.; Jain, R.; Saxena, T.; Dobrinsky, A.; Shur, M. Development of Deep UV LEDs and Current Problems in Material and Device Technology. Semicond. Semimet. 2017, 96, 45–83. [Google Scholar] [CrossRef]
- Trivellin, N.; Monti, D.; De Santi, C.; Buffolo, M.; Meneghesso, G.; Zanoni, E.; Meneghini, M. Current induced degradation study on state of the art DUV LEDs. Microelectron. Reliab. 2018, 88–90, 868–872. [Google Scholar] [CrossRef]
- Amano, H.; Collazo, R.; De Santi, C.; Einfeldt, S.; Funato, M.; Glaab, J.; Hagedorn, S.; Hirano, A.; Hirayama, H.; Ishii, R.; et al. The 2020 UV emitter roadmap. J. Phys. D Appl. Phys. 2020, 53, 503001. [Google Scholar] [CrossRef]
- Endruweit, A.; Johnson, M.S.; Long, A.C. Curing of composite components by ultraviolet radiation: A review. Polym. Compos. 2006, 27, 119–128. [Google Scholar] [CrossRef]
- Hockberger, P.E. A History of Ultraviolet Photobiology for Humans, Animals and Microorganisms. Photochem. Photobiol. 2007, 76, 561–579. [Google Scholar] [CrossRef]
- Dreyer, C.; Mildner, F. Application of LEDs for UV-Curing; Springer Series in Materials Science; Springer International Publishing: Cham, Switzerland, 2016; pp. 415–434. [Google Scholar]
- Schirripa Spagnolo, G.; Cozzella, L.; Simonetti, C. Banknote security using a biometric-like technique: A hylemetric approach. Meas. Sci. Technol. 2010, 21, 055501. [Google Scholar] [CrossRef]
- Won, W.-S.; Tran, L.G.; Park, W.-T.; Kim, K.-K.; Shin, C.S.; Kim, N.; Kim, Y.-J.; Yoon, Y.-J. UV-LEDs for the Disinfection and Bio-Sensing Applications. Int. J. Precis. Eng. Manuf. 2018, 19, 1901–1915. [Google Scholar] [CrossRef]
- Kemény, L.; Varga, E.; Novak, Z. Advances in phototherapy for psoriasis and atopic dermatitis. Expert Rev. Clin. Immunol. 2019, 15, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Vieyra-Garcia, P.A.; Wolf, P. A deep dive into UV-based phototherapy: Mechanisms of action and emerging molecular targets in inflammation and cancer. Pharmacol. Ther. 2021, 222, 107784. [Google Scholar] [CrossRef] [PubMed]
- Glickman, G.; Byrne, B.; Pineda, C.; Hauck, W.W.; Brainard, G.C. Light Therapy for Seasonal Affective Disorder with Blue Narrow-Band Light-Emitting Diodes (LEDs). Biol. Psychiatry 2006, 59, 502–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paradiso, R.; Proietti, S. Light-Quality Manipulation to Control Plant Growth and Photomorphogenesis in Greenhouse Horticulture: The State of the Art and the Opportunities of Modern LED Systems. J. Plant Growth Regul. 2021. [Google Scholar] [CrossRef]
- Schreiner, M.; Martínez-Abaigar, J.; Glaab, J.; Jansen, M. UV-B Induced Secondary Plant Metabolites. Opt. Photonik 2014, 9, 34–37. [Google Scholar] [CrossRef]
- Shur, M.S.; Gaska, R. Deep-Ultraviolet Light-Emitting Diodes. IEEE Trans. Electron. Devices 2010, 57, 12–25. [Google Scholar] [CrossRef]
- Žukauskas, A.; Kurilčik, N.; Vitta, P.; Juršėnas, S.; Bakienė, E.; Gaska, R. Optimization of a UV light-Emitting Diode Based Fluorescence-Phase Sensor; Carrano, J.C., Zukauskas, A., Eds.; Optically Based Biological and Chemical Detection for Defence III: Stockholm, Sweden, 2006; p. 63980Y. [Google Scholar]
- Mori, M.; Hamamoto, A.; Takahashi, A.; Nakano, M.; Wakikawa, N.; Tachibana, S.; Ikehara, T.; Nakaya, Y.; Akutagawa, M.; Kinouchi, Y. Development of a new water sterilization device with a 365 nm UV-LED. Med. Biol. Eng. Comput. 2007, 45, 1237–1241. [Google Scholar] [CrossRef]
- Würtele, M.A.; Kolbe, T.; Lipsz, M.; Külberg, A.; Weyers, M.; Kneissl, M.; Jekel, M. Application of GaN-based ultraviolet-C light emitting diodes—UV LEDs—for water disinfection. Water Res. 2011, 45, 1481–1489. [Google Scholar] [CrossRef]
- Kowalski, W.J.; Walsh, T.J.; Petraitis, V. COVID-19 Coronavirus Ultraviolet Susceptibility 2020 COVID-19 Coronavirus Ultraviolet Susceptibility; PurpleSun Inc.: Long Island City, NY, USA, 2020; pp. 1–4. [Google Scholar] [CrossRef]
- Inagaki, H.; Saito, A.; Sugiyama, H.; Okabayashi, T.; Fujimoto, S. Rapid inactivation of SARS-CoV-2 with deep-UV LED irradiation. Emerg. Microbes Infect. 2020, 9, 1744–1747. [Google Scholar] [CrossRef]
- Ran, Z.; Yao, M.; Li, S. Comparison of UVA-LED and UVC-LED for Water Disinfection: Inactivation of Escherichia coli; Sustainable Development of Water and Environment. ICSDWE 2020; Jeon, H.Y., Ed.; Springer: Cham, Switzerland, 2020; pp. 39–49. [Google Scholar]
- Chatterley, C.; Linden, K.G. UV-LED Irradiation Technology for Point-of-Use Water Disinfection. Proc. Water Environ. Fed. 2009, 2009, 222–225. [Google Scholar] [CrossRef] [Green Version]
- Pramanik, P.; Das, S.; Adhikary, A.; Chaudhuri, C.R.; Bhattacharyya, A. Design and implementation of water purification system based on deep ultraviolet light emitting diodes and a multi-pass geometry reactor. J. Water Health 2020, 18, 306–313. [Google Scholar] [CrossRef]
- Anyaogu, K.C.; Ermoshkin, A.A.; Neckers, D.C.; Mejiritski, A.; Grinevich, O.; Fedorov, A.V. Performance of the light emitting diodes versus conventional light sources in the UV light cured formulations. J. Appl. Polym. Sci. 2007, 105, 803–808. [Google Scholar] [CrossRef]
- Hao, G.-D.; Tamari, N.; Obata, T.; Kinoshita, T.; Inoue, S. Electrical determination of current injection and internal quantum efficiencies in AlGaN-based deep-ultraviolet light-emitting diodes. Opt. Express 2017, 25, A639. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Liu, G.; Zhang, J.; Arif, R.A.; Tansu, N. Analysis of internal quantum efficiency and current injection efficiency in III-nitride light-emitting diodes. IEEE/OSA J. Disp. Technol. 2013, 9, 212–225. [Google Scholar] [CrossRef]
- Cho, J.; Schubert, E.F.; Kim, J.K. Efficiency droop in light-emitting diodes: Challenges and countermeasures. Laser Photon. Rev. 2013, 7, 408–421. [Google Scholar] [CrossRef]
- Piva, F.; De Santi, C.; Deki, M.; Kushimoto, M.; Amano, H.; Tomozawa, H.; Shibata, N.; Meneghesso, G.; Zanoni, E.; Meneghini, M. Modeling the degradation mechanisms of AlGaN-based UV-C LEDs: From injection efficiency to mid-gap state generation. Photon. Res. 2020, 8, 1786. [Google Scholar] [CrossRef]
- Yun, J.; Shim, J.I.; Hirayama, H. Analysis of efficiency droop in 280-nm AlGaN multiple-quantum-well light-emitting diodes based on carrier rate equation. Appl. Phys. Express 2015, 8, 022104. [Google Scholar] [CrossRef]
- De Santi, C.; Meneghini, M.; Tibaldi, A.; Vallone, M.; Goano, M.; Bertazzi, F.; Verzellesi, G.; Meneghesso, G.; Zanoni, E. Physical Mechanisms Limiting the Performance and the Reliability of GaN-Based LEDs; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; ISBN 9780081019436. [Google Scholar]
- Piva, F.; De Santi, C.; Deki, M.; Kushimoto, M.; Amano, H.; Tomozawa, H.; Shibata, N.; Meneghesso, G.; Zanoni, E.; Meneghini, M. Stability and degradation of AlGaN-based UV-B LEDs: Role of doping and semiconductor defects. Microelectron. Reliab. 2019, 100–101, 113418. [Google Scholar] [CrossRef]
- Haller, C.; Carlin, J.F.; Jacopin, G.; Martin, D.; Butté, R.; Grandjean, N. Burying non-radiative defects in InGaN underlayer to increase InGaN/GaN quantum well efficiency. Appl. Phys. Lett. 2017, 111, 262101. [Google Scholar] [CrossRef] [Green Version]
- Nagai, S.; Yamada, K.; Hirano, A.; Ippommatsu, M.; Ito, M.; Morishima, N.; Aosaki, K.; Honda, Y.; Amano, H.; Akasaki, I. Development of highly durable deep-ultraviolet AlGaN-based LED multichip array with hemispherical encapsulated structures using a selected resin through a detailed feasibility study. Jpn. J. Appl. Phys. 2016, 55, 082101. [Google Scholar] [CrossRef]
- Trivellin, N.; Monti, D.; Piva, F.; Buffolo, M.; De Santi, C.; Zanoni, E.; Meneghesso, G.; Meneghini, M. Degradation processes of 280 nm high power DUV LEDs: Impact on parasitic luminescence. Jpn. J. Appl. Phys. 2019, 58, SCCC19. [Google Scholar] [CrossRef]
- Yoshikawa, A.; Hasegawa, R.; Morishita, T.; Nagase, K.; Yamada, S.; Grandusky, J.; Mann, J.; Miller, A.; Schowalter, L.J. Improve efficiency and long lifetime UVC LEDs with wavelengths between 230 and 237 nm. Appl. Phys. Express 2020, 13, 022001. [Google Scholar] [CrossRef]
- Monti, D.; De Santi, C.; Da Ruos, S.; Piva, F.; Glaab, J.; Rass, J.; Einfeldt, S.; Mehnke, F.; Enslin, J.; Wernicke, T.; et al. High-Current Stress of UV-B (In)AlGaN-Based LEDs: Defect-Generation and Diffusion Processes. IEEE Trans. Electron. Devices 2019, 66, 3387–3392. [Google Scholar] [CrossRef]
- Glaab, J.; Haefke, J.; Ruschel, J.; Brendel, M.; Rass, J.; Kolbe, T.; Knauer, A.; Weyers, M.; Einfeldt, S.; Guttmann, M.; et al. Degradation effects of the active region in UV-C light-emitting diodes. J. Appl. Phys. 2018, 123. [Google Scholar] [CrossRef]
- Ruschel, J.; Glaab, J.; Beidoun, B.; Ploch, N.L.; Rass, J.; Kolbe, T.; Knauer, A.; Weyers, M.; Einfeldt, S.; Kneissl, M. Current-induced degradation and lifetime prediction of 310 nm ultraviolet light-emitting diodes. Photon. Res. 2019, 7, B36. [Google Scholar] [CrossRef]
- Glaab, J.; Ruschel, J.; Kolbe, T.; Knauer, A.; Rass, J.; Cho, H.K.; Lobo Ploch, N.; Kreutzmann, S.; Einfeldt, S.; Weyers, M.; et al. Degradation of (In)AlGaN-Based UVB LEDs and Migration of Hydrogen. IEEE Photon. Technol. Lett. 2019, 31, 529–532. [Google Scholar] [CrossRef]
- Meneghini, M.; Barbisan, D.; Rodighiero, L.; Meneghesso, G.; Zanoni, E. Analysis of the physical processes responsible for the degradation of deep-ultraviolet light emitting diodes. Appl. Phys. Lett. 2010, 97, 14–17. [Google Scholar] [CrossRef]
- Heilingloh, C.S.; Aufderhorst, U.W.; Schipper, L.; Dittmer, U.; Witzke, O.; Yang, D.; Zheng, X.; Sutter, K.; Trilling, M.; Alt, M.; et al. Susceptibility of SARS-CoV-2 to UV irradiation. Am. J. Infect. Control 2020, 48, 1273–1275. [Google Scholar] [CrossRef] [PubMed]
- Bormann, M.; Alt, M.; Schipper, L.; van de Sand, L.; Otte, M.; Meister, T.L.; Dittmer, U.; Witzke, O.; Steinmann, E.; Krawczyk, A. Disinfection of sars-cov-2 contaminated surfaces of personal items with uvc-led disinfection boxes. Viruses 2021, 13, 598. [Google Scholar] [CrossRef]
- Liu, S.; Luo, W.; Li, D.; Yuan, Y.; Tong, W.; Kang, J.; Wang, Y.; Li, D.; Rong, X.; Wang, T.; et al. Sec-Eliminating the SARS-CoV-2 by AlGaN Based High Power Deep Ultraviolet Light Source. Adv. Funct. Mater. 2021, 31, 2008452. [Google Scholar] [CrossRef]
- Kitagawa, H.; Nomura, T.; Nazmul, T.; Omori, K.; Shigemoto, N.; Sakaguchi, T.; Ohge, H. Effectiveness of 222-nm ultraviolet light on disinfecting SARS-CoV-2 surface contamination. Am. J. Infect. Control 2021, 49, 299–301. [Google Scholar] [CrossRef]
- Sabino, C.P.; Sellera, F.P.; Sales-Medina, D.F.; Machado, R.R.G.; Durigon, E.L.; Freitas-Junior, L.H.; Ribeiro, M.S. UV-C (254 nm) lethal doses for SARS-CoV-2. Photodiagnosis Photodyn. Ther. 2020, 32, 101995. [Google Scholar] [CrossRef]
- Minamikawa, T.; Koma, T.; Suzuki, A.; Mizuno, T.; Nagamatsu, K.; Arimochi, H.; Tsuchiya, K.; Matsuoka, K.; Yasui, T.; Yasutomo, K.; et al. Quantitative evaluation of SARS-CoV-2 inactivation using a deep ultraviolet light-emitting diode. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Trivellin, N.; Buffolo, M.; Onelia, F.; Pizzolato, A.; Barbato, M.; Orlandi, V.T.; Del Vecchio, C.; Dughiero, F.; Zanoni, E.; Meneghesso, G.; et al. Inactivating SARS-CoV-2 Using 275 nm UV-C LEDs through a Spherical Irradiation Box: Design, Characterization and Validation. Materials 2021, 14, 2315. [Google Scholar] [CrossRef] [PubMed]
- OMS Water, Sanitation, Hygiene and Waste Management for the COVID-19 Virus; World Health Organization: Geneva, Switzerland, 2020; pp. 1–9.
- Hu, Y.; Cheng, H. Mercury risk from fluorescent lamps in China: Current status and future perspective. Environ. Int. 2012, 44, 141–150. [Google Scholar] [CrossRef]
- Education, I.M.; Hampshire, N.; Island, R.; Lamps, F. IMERC Fact Sheet Mercury Use in Lighting Types of Mercury Lamps; NEWMOA, Northeast Waste Management Officials’ Association: Boston, MA, USA, 2001; pp. 1–14. [Google Scholar]
- Beyer, R.M.; Manica, A.; Mora, C. Shifts in global bat diversity suggest a possible role of climate change in the emergence of SARS-CoV-1 and SARS-CoV-2. Sci. Total Environ. 2021, 767, 145413. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, Z.; Wang, M.Y.; Sun, W. What caused the outbreak of COVID-19 in China: From the perspective of crisis management. Int. J. Environ. Res. Public Health 2020, 17, 3279. [Google Scholar] [CrossRef] [PubMed]
- Mazzoleni, S.; Turchetti, G.; Ambrosino, N. The COVID-19 outbreak: From “black swan” to global challenges and opportunities. Pulmonology 2020, 26, 117–118. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, C.; Mildner, F. III-Nitride Ultraviolet Emitters; Kneissl, M., Rass, J., Eds.; Springer Series in Materials Science; Springer International Publishing: Cham, Switzerland, 2016; Volume 227, ISBN 978-3-319-24098-5. [Google Scholar]
- Khan, M.H.; Yadav, H. Sanitization During and After COVID-19 Pandemic: A Short Review. Trans. Indian Natl. Acad. Eng. 2020, 5, 617–627. [Google Scholar] [CrossRef]



| Type of Lamp | Wavelength | UVC Efficiency | Power Density | Useful Life |
|---|---|---|---|---|
| Low pressure | 254 nm | 35% | 0.8–1.3 W/cm | 12,000 h |
| Amalgam | 254 nm | 33% | 1–6 W/cm | >12,000 h |
| Medium pressure | Broad | 15% | 80–300 W/cm | 1000–5000 h |
| Tech. | λ (Nm) | Dose (Mj/Cm2) | Log Reduction | Notes | Ref. |
|---|---|---|---|---|---|
| LED | 280 ± 5 | 3.75 37.5 75 112.5 225 | 0.9 3.1 >3.3 >3.3 >3.3 | Aliquots of virus stock (150 μL) were placed in the centre of a 60 mm Petri dish and irradiated with 3.75 mW/cm2 at a work distance of 20 mm for a range of times | [46] |
| Tubes | 254 365 | 1048 292 | >4 1 | A viral stock at a concentration of 5 × 106 TCID50/mL was irradiated with UV light up to 30 min; UV exposure was performed by separate or combined irradiation with UVC and/or UVA of 600 μL virus stock in 24-well plates | [67] |
| LED | 254 280 | 11.7 39 44.1 147 21.06 70.2 10 min, 4 cm dist. | 0.93 (glass, metal) 1 (plastic) 1.1 (glass) 1.13 (plastic) 1.83 (metal) >4 (glass, plastic) >4 (glass, plastic, metal) 1.6 (glass) 1.9 (plastic) 2.1 (metal) 1.67 (glass) 2.23 (metal) 2.27 (plastic) >4 (glass, plastic, metal) | A measurement of the emitted light intensity at 4 cm distance would have required drilling a hole into the bottom of the box, damaging the box; different materials (glass, metal, plastic) tested as carriers for viral contamination; the carriers were positioned at different distances from the UVC-LEDs | [68] |
| LED | 275 | 94 | >4 | To obtain a high output power density, the UVC LED chips are integrated into an array of 15 × 13, with a whole area of 3 × 3.6 cm2 | [69] |
| LED | 267 279 286 297 | 6 7 13 32 | 3 3 3 3 | The human Coronavirus OC43 (HCoV-OC43) was used as a surrogate to the SARS-CoV-2; for each irradiation experiment 50 μL of virus suspension was placed in each well of a black 24-well plate | [19] |
| Tubes | 222 | 3 | ~2.5 | Viral suspension (100 μL) containing ca. 5 × 106 TCID50/mL were loaded onto a sterile polystyrene plate (9 cm diameter) and spread into a circle of approx. 7 cm. The inoculum was allowed to dry under sterile laminar flow in a biosafety cabinet at room temperature; in contrast to the viable SARS-CoV-2, the copy number of SARS-CoV-2 RNA determined by RT-qPCR was not reduced even after 30 mJ/cm2 of 222 nm UVC irradiation | [70] |
| Tubes | 254 | 0.016 0.706 6.556 31.88 108.714 | 1 2 3 4 5 | within less than a second, UVC irradiation was able to inactivate more than 99% of SARS-CoV-2 viral particles | [71] |
| Tubes | 254 | 3.7 16.9 84.4 | 3 6 6 | different virus concentration were tested; a dose of 3.7 mJ/cm2 was not enough for a high virus concentration and a dose of 16.9 mJ/cm2 was required in order to avoid virus replication | [23] |
| LED | 265 280 300 | 1.8 3 23 | 3 3 3 | for each irradiation experiment, 100 μL of virus inoculum was placed in a defined well (E4) of a 96-well plate, and the plate was exposed to the designated irradiation wavelength and time | [72] |
| LED | 275 | 8.31 | 3 | Analysis carried out on different surfaces of sport balls | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trivellin, N.; Piva, F.; Fiorimonte, D.; Buffolo, M.; De Santi, C.; Orlandi, V.T.; Dughiero, F.; Meneghesso, G.; Zanoni, E.; Meneghini, M. UV-Based Technologies for SARS-CoV2 Inactivation: Status and Perspectives. Electronics 2021, 10, 1703. https://doi.org/10.3390/electronics10141703
Trivellin N, Piva F, Fiorimonte D, Buffolo M, De Santi C, Orlandi VT, Dughiero F, Meneghesso G, Zanoni E, Meneghini M. UV-Based Technologies for SARS-CoV2 Inactivation: Status and Perspectives. Electronics. 2021; 10(14):1703. https://doi.org/10.3390/electronics10141703
Chicago/Turabian StyleTrivellin, Nicola, Francesco Piva, Davide Fiorimonte, Matteo Buffolo, Carlo De Santi, Viviana Teresa Orlandi, Fabrizio Dughiero, Gaudenzio Meneghesso, Enrico Zanoni, and Matteo Meneghini. 2021. "UV-Based Technologies for SARS-CoV2 Inactivation: Status and Perspectives" Electronics 10, no. 14: 1703. https://doi.org/10.3390/electronics10141703
APA StyleTrivellin, N., Piva, F., Fiorimonte, D., Buffolo, M., De Santi, C., Orlandi, V. T., Dughiero, F., Meneghesso, G., Zanoni, E., & Meneghini, M. (2021). UV-Based Technologies for SARS-CoV2 Inactivation: Status and Perspectives. Electronics, 10(14), 1703. https://doi.org/10.3390/electronics10141703











