Abstract
3D printing (3DP) is a manufacturing technology that produces 3D objects from a design file using layer-by-layer deposition of material. It has already found applications in the healthcare and pharmaceutical industries, while its use in the field of topical delivery has been extensively studied in the last two decades. The aim of this study is to provide a comprehensive overview of the 3DP-based developments in topical delivery, with special emphasis on its current and potential use in the cosmetic field. This review covers the principles and main types of 3DP technology, production, and characteristics of two key 3DP skin delivery platforms (patches and microneedles—MNs), as well as topical active materials used, focusing on those for cosmetic application. A comprehensive search of peer-reviewed articles in relevant databases was performed (including PubMed, Wiley Online Library, Multidisciplinary Digital Publishing Institute, Kosmet, ScienceDirect and Scopus) from 2004 to date. The study concludes that 3D printed MNs would gain prevalence over patches for both pharmaceutical and cosmetic applications and that dissolving MNs have the highest potential among them. The use of 3DP technology appears to be a promising approach to the development of efficient personalised cosmetic delivery platforms.
1. Introduction
Stratum corneum (SC) acts as an efficient barrier against physical, chemical, and microbiological xenophobes, preventing their penetration into the skin. However, this excellent barrier is a limiting factor for the penetration of cosmetic active ingredients (also known as actives) into the skin. Skin delivery from topical formulations is known to be very inefficient, with typical bioavailability of less than 2% of the applied dose [1]. A good example is caffeine, a well-studied cosmetic and pharmaceutical active ingredient, also a model hydrophilic compound in skin toxicology. Summarising a series of studies conducted with different topical caffeine formulations, a review article [2] has established that the highest penetration from conventional ointment formulations was only 0.0062%.
Therefore, it is crucial to explore all available means for more efficient delivery of topical (cosmetic and pharmaceutical) active ingredients into the skin. Many technologies have been studied and developed so far, including penetration enhancers, supersaturation, and a wide range of skin delivery systems (e.g., liposomes, niosomes, transfersomes, lipid nanoparticles, polymeric microparticles and nanoparticles, patches, and microneedles). One of the relatively recent approaches is the use of 3D printed platforms (carriers).
3D printing (3DP) is a manufacturing technology that produces 3D objects from a design file using layer-by-layer deposition of material. It offers some advantages over traditional manufacturing techniques, such as one-step fabrication and customisation [3]. In addition, 3D printing has shown potential in increasing skin delivery efficacy and user compliance [4].
The healthcare and medical industry has already benefited from 3DP with versatile applications, from 3D printed pharmaceuticals in solid and semisolid forms [5,6], to those with complex release profiles [7]. In addition, there are 3D printed medical devices, such as patient-specific implants and hydrogel grid wound dressings [8,9], many of them approved by the United States Food and Drug Administration (FDA) [10].
However, the number of applications of 3DP in skin delivery is relatively low, with limited choice of 3DP-specific materials being the biggest obstacle. This is because specific physico-chemical properties, such as photosensitivity or thermal sensitivity, are required for the solidification process of the inks during 3D printing in order to provide the structure of 3D objects; in addition, some 3D printing technologies require the ink to be within certain viscosity range [11]. Another obstacle is high initial investment necessary to increase the production output. Extensive studies in skin delivery have only been carried out in the last two decades [12], and have demonstrated a considerable potential of 3DP in this area.
There are already comprehensive review articles which cover different aspects on cosmetic skin delivery platforms [13,14], as well as 3D printed transdermal [3,11,12] and topical [15] drug delivery application. However, none of them discusses cosmetic applications of 3D printed skin delivery platforms in detail. Therefore, it was of interest to summarise the developments in this cutting-edge technology, with the view of its application to the delivery of cosmetic active ingredients.
2. Types of 3D Printing Technologies
Based on the process involved, the American Society for Testing and Materials (ASTM) has classified 3DP technology into seven types, the overview of which is given in several articles [16,17]. Among these methods, fused deposition modelling (FDM) and stereolithography (SLA) have been the most popular 3DP technologies for the fabrication of skin delivery platforms. In recent studies, digital light processing (DLP) and two photon-polymerisation (TPP) were also used [18,19]. In addition, ink jet printing is applied for the loading of active ingredients in the post-platform fabrication processes [20,21].
All currently used types of 3D processes could be classified into three broad categories: ink jet printing, extrusion-based and photopolymerisation-based, and are summarised in Table 1.

Table 1.
Three categories of common 3DP technologies [16,17,24].
2.1. Inkjet Printing
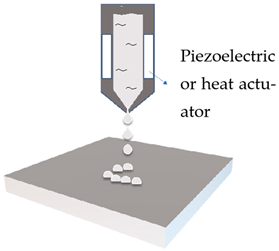
Inkjet printing is the general name for pattern-generating devices that could produce digitally controlled formulation and placement of small liquid drops onto a substrate. It can be further classified according to the droplet size and droplet formation method, which is achieved either by thermal or piezoelectric means [22].
Inkjet printing presents the earliest attempt to apply 3DP on the manufacturing of skin delivery devices, dating from early 2000s. The technology has already been widely studied for solid dosage forms, with high drug loading as one of its major benefits [23]. Inkjet printing has been mostly used as a two-dimensional printing method to deposit formulations onto skin delivery platforms that are made conventionally or with other 3DP technologies [20,21].
2.2. Extrusion-Based Printing
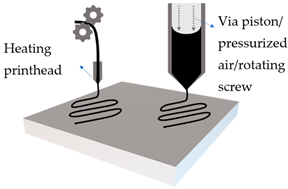
Extrusion-based printers are user-friendly, versatile, and inexpensive. The process involves melting of the printing material, which is ejected from nozzles on the printhead while it moves, and deposited in successive thin layers, enabling a bottom-up construction of 3D objects. The disadvantages of such a technology are low resolution, which depends on the size of the nozzle, and the possibility of deformation or collapsing of the object during printing [10].
Fused deposition modelling (FDM) is a common type of extrusion-based 3DP method, with various applications in the pharmaceutical industry, such as personalised tablets or capsules. An innovative study by Stevic et al. demonstrated the fabrication of lipsticks utilising this 3DP technology [25]. The printing process depends on the printing material and nozzle size, pressure difference, feed rate, printing temperature, and layer thickness [26]. FDM is commonly combined with hot melt extrusion (HME), which involves dispersing active molecules into a molten hydrophilic polymer before printing, thus avoiding a separate coating step. The process achieves high loading, but is limited to thermostable active ingredients. It was used, for example, to manufacture a personalised anti-acne patch with salicylic acid [27]. A follow-up study used polycaprolactone (PCL) mixed with antimicrobial metals to obtain effective wound dressings [28]. An investigation into the printability of a semi-solid form using FDM method has resulted in enhanced delivery of an anti-acne active [29].
In addition to the patch form, a considerable research effort (e.g., [26,30,31]) was devoted to the use of FDM in the production of microneedle patches for transdermal drug delivery. A frequent problem found in extrusion-based printing methods is the ‘stair-stepping’ effect due to relatively low resolution. To achieve a sharper tip and smoother surface required for the better penetration of microneedle (MN) arrays, additional post-printing steps are being used. Some studies attempted chemical etching as a post-treatment step and succeeded in achieving a tip diameter as low as 1 μm [31,32]. Wu et al. [33] have applied a post-stretching process on the cylindrical microneedle arrays in order to obtain sharp tips.
2.3. Photopolymersation
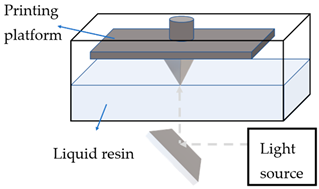
This process refers to the selective curing of liquid photopolymers by using a laser, light or ultraviolet (UV) emitter [17]. During printing, the laser head scans the surface of a photopolymerisable liquid, and the polymerisation occurs upon scanning to create a layer of solid, which is then lowered to a depth equivalent to the thickness of the previously formed layer. Since UV light penetrates further than each layer’s thickness, the lower layer becomes over-cured and fused with the upper layer, which leads to a high level of accuracy and resolution. Stereolithography (SLA), digital light processing (DLP), continuous liquid interface production (CLIP), and two-photon polymerisation (TPP) are examples of 3DP technologies based on photopolymersation. Due to the limited choice of printable materials and the toxicity of common photoinitiators, these processes are rarely used for solid dosage forms research, but are common for medical devices (e.g., implants), tissue engineering, and skin delivery platforms. Factors that affect the quality of the printed platform include exposure time, laser power, type of the printable material, and photoinitiator [10].
- Stereolithography (SLA)
SLA is the most studied photopolymerisation technology for the manufacturing of skin delivery platforms. It utilises a laser beam to draw lines of solidified polymer on the surface of a vat of liquid. The study by Goyanes et al. [27] compared the SLA and FDM production methods for an anti-acne patch with salicylic acid and found that SLA resulted in a smooth and flexible finish, good loading, and efficacy.
Due to its high accuracy and precision, SLA has been successfully used in the fabrication of MN arrays, combined with different methods for the loading of active ingredients, e.g., direct mixing with the polymer solution before polymerisation [27,34] and ink jet printing on the surface [20,21]. It was also used for the preparation of the MN moulds in the micro-moulding approach to the MN production [35]. Since SLA operates at mild temperatures, it was possible to incorporate proteins or peptides into the MN arrays using this method [21].
The largest drawback of SLA technology is that post-printing procedures are necessary to remove toxic substances, which results in additional processing time and possible introduction of solvents into the 3D platform.
- Digital light processing (DLP)
DPL is similar to SLA but has a different light source. It is normally an arc lamp with a high-definition liquid crystal display, which flashes an image on the surface of the photopolymer resin. Since the light source can be applied to the whole object in a single pass instead of a beam scanning, DLP is generally faster than SLA. In the study by Gittard et al. [36] the use of DLP for MNs with various geometries intended for wound healing was investigated, whereby the pulsed laser deposition was used to coat the MNs surface with silver and zinc oxide [36]. The limitations of DLP are the difficulties in drug loading and the lack of suitable non-toxic printing materials.
- Continuous liquid interface production (CLIP)
CLIP is another photopolymerisation-based 3DP technology with fast printing speed and high resolution, which involves continuous polymerisation, but not in a layer-by-layer fashion. UV radiation is continuously emitted into the liquid resin through an oxygen-permeable window. Oxygen-containing ‘dead-zones’ within the resin reservoir remain as liquid, while solidification only occurs in the designated region [37]. A recent review discussed the technology and highlighted the use in drug delivery in detail [38]. In addition to the drawbacks common to all photopolymersation methods, CLIP tends to be more expensive.
- Two-photon polymerisation (TPP)
TPP is used to build elaborate and complex structures in micro and nanoscale. This enables a potential use in the fabrication of drug delivery systems with complex release profiles, including both solid oral dosage forms and MNs. TPP technology prints 3D objects by a raster scanning of the femtosecond laser pulses, in a one-step manner. This technology can produce structures with details of less than 100 nm, which include ultrasharp MNs and hollow MNs [3]. Some early attempts have shown the potential of TPP in the fabrication of MNs with ceramic material [39,40]. The same group has published a review on the use of TPP in fabricating hollow and solid MNs, including specific antimicrobial MNs [41].
Subsequent studies have focused on the TPP-aided fabrication of MNs made from biocompatible polymers [19]. Due to the high precision of the hollow MNs produced by TPP, the researchers were able to fit them with smart medical devices for controlled drug delivery and lab-on-a-chip diagnostics [26]. TPP is the most promising photopolymerisation method for the fast production of ultraprecise MNs; however, it suffers from the same limitations related to the photopolymerisation process as SLA and DLP.
3. Types of 3D Printed Delivery Platforms
Skin patches and microneedles (MNs) have emerged as the two main types of 3D printed platforms. Due to the same principles of skin delivery of cosmetic and topical pharmaceutical formulations, the developments in both will be reviewed in this section.
- Skin patches
Skin patches are the most used and studied among all device-based skin delivery systems. They have a long history for treating skin conditions [42] and have also been used for transdermal delivery. A recent review article reported research work conducted on conventional skin patches, in terms of their active ingredients, materials, delivery enhancers, characterisation methods, and results [43]. Standard methods for the fabrication of skin patches include solvent, hydrogel, and hot melt-based means [44].
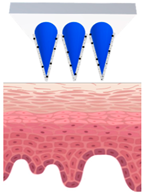
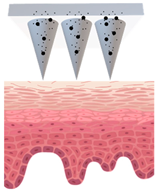
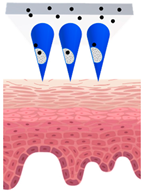
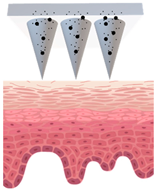
Patches closely adhere to the skin and could be designed with or without separate adhesive support, which will be either loaded with active ingredients or saturated with active ingredients from the reservoir [45], as demonstrated in Figure 1. The adhesive property of patches strongly affects the delivery of active ingredients, and in turn their efficacy [46,47]. They create a continuous occlusion which increases skin penetration by providing a strong driving force for the diffusion of active ingredients [48].

Figure 1.
Schematic diagram of three types of conventional multi-layered skin patches.
Theoretically, cosmetic patches have the potential to tackle many cosmetic skin problems, such as wrinkles, pigmentation, and the effects of aging [49]. The main disadvantage of conventional patches is the low quantity of active ingredients that could be loaded and delivered. The mechanism of drug absorption from a patch-like device starts with its release from the patch, its penetration and then storage in the stratum corneum, at which point it might crystalise and prevent further transport [50]. The next stage is diffusion of the active to the deeper layers of skin, and, if applicable, into the systemic circulation, causing a controlled delay of the therapeutic effect [50]. Another disadvantage is a possible inconsistent diffusion rate of the active ingredient from skin patches, which depends on the skin condition of individuals. This includes skin hydration state, age, and ethnicity, with the SC being a rate-limiting barrier. In addition, the delivery strongly depends on the type and physicochemical properties of the active [3].
Conventional adhesive patches have multilayer structures and are classified by the layer in which the drug/active has been loaded [46,47,51,52], as shown in Figure 1. Therefore, the fabrication involves a multi-stage process [53]. In contrast, 3D printed patches are mostly made of one material in a single layer, although there is a potential for the multi-layered printing. Due to the flexible nature of the 3D printed patches, researchers have been exploring the potential of drug-loaded 3DP mesh or 3DP grid patches for implantation within tissues and to support organs [54].
Overall, the development of 3DP patch platforms is an ongoing research area, with a potential to improve current skin delivery designs.
- Microneedles
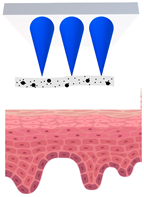
Microneedles (MNs) have evolved as a hybrid of two conventional skin delivery systems, skin patches and hypodermic injection needles, with some advantages of both, such as ease of administration, being minimally invasive, and enabling high bioavailability of active ingredients [55]. An MN platform is typically composed of micro-sized, needle-like structures attached to a patch (backing) for ease of application. This delivery platform is sometimes referred to as a MNP (microneedle patch). In comparison to patches and semisolid topical formulations, microneedles are more versatile and more efficient in delivering active ingredients into deeper layers of the skin. Instead of relying on passive diffusion, microneedles can actively enhance the delivery by piercing the epidermis (0.1–0.2 mm) and, if so designed, the dermis (1–2 mm), creating microscopic channels [56]. These microscopic punctures could overcome the skin barrier and provide an alternative route for enhanced transport of drug or cosmetic active in a painless, minimally invasive manner [57]. Table 2 provides a summary of the common types of MNs. Some novel MNs not only serve as a delivery platform, but also as a wearable therapeutic device for real time monitoring [58].

Table 2.
Classification of MNs [59,60,61,62].
A recent review paper [13] has provided a comprehensive report on the studies on cosmetic application of microneedles. It summarises MN materials and categorises MN studies in terms of the targeted skin problems.
3D printing could be used in three different ways in the manufacture of an MN platform: (1) to develop ‘male’ master moulds; (2) to coat active ingredients onto previously prepared MNs and (3) to print complete MN structures.
Research on 3D printed cosmetic microneedles is still in its early age. However, several published papers and patents have shown the feasibility of delivering both hydrophilic and lipophilic active ingredients by microneedles that are fabricated by methods other than 3DP [63,64,65], proving that this concept is viable. One study has compared the wrinkle improvement by two different delivery platforms (dissolving MNs and standard formulation, both containing hyaluronic acid); after eight weeks of treatment, the MNs have shown higher effectiveness [66].In general, all MNs enhance skin delivery via micro-channels they create, partly bypassing the skin barrier. In the case of wrinkle improvement, there is a second mechanism [67,68]: the perforations they create could induce elastin and collagen expression and deposition, stimulating the metabolism in the upper skin layers, as well as the natural healing of the skin.
Table 3 illustrates the mechanism of skin delivery of different types of 3D printed solid MNs, including coated MNs, dissolving/swellable MNs (DMNs), and hollow MNs. Further details and diagrams could be found in several review articles which focus on microneedle skin delivery platforms, including their characteristics and their typical delivery mechanisms [61,69,70].

Table 3.
Schematic diagrams of common types of 3D printed MNs before and after application.
There is an argument that dissolvable MNs (DMNs) might not be an ideal platform for cosmetic use, due to the potential loss of hydration through the perforations made in the skin. However, it has been shown that a novel design of DMNs, loaded with a barrier-restoring active ingredient, horse oil, has significantly improved dermal density, skin elasticity and moisturisation level [71].
Very few published studies have reported a detailed 3DP manufacture of the dissolving MNs, and none has studied the use of sugar-based biopolymers [72]. It remains challenging to fabricate DMNs with 3DP other than by micro-moulding methods, due to the lack of the printability of dissolvable polymers.
Fabrication Methods
Conventional MN fabrication methods can be classified into several categories: (1) moulding method, (2) lithography, (3) droplet-born air blowing (DAB) method, some followed by coating or deposition process to produce coated MNs [55,70,73,74,75].
As described by Kim et al., DAB was a popular DMN fabrication method [76], adopted by many researchers who have successfully fabricated DMNs and assessed their effects in improving skin delivery of cosmetic active ingredients [77,78,79,80]. This method was gradually abandoned with the development of centrifugal lithography (CL) [81] for DMN fabrication. Due to the self-shaping nature of viscous polymer solution, continuous transformation under centrifugal force is induced in the CL process. Polymer drops dispense on the inner plate of two parallel fixed plates, and, upon the separation of plates, DMNs with two different shapes have been formed on the top and bottom plates, respectively. Morphological observation, fracture force analysis, and in vitro skin penetration tests have shown that both DMNs platforms could achieve an efficient diffusion and permeation of active ingredients through the skin [82]. It is worth mentioning that no additional environmental stimulation is required for producing DMNs using CL. The usual problems related to other fabrication methods, such as the loss of activity of cosmetic ingredients when exposed to UV irradiation, heat, and air, do not exist in CL. However, CL-produced MN shapes are extremely limited, with little variation of the natural droplet shape, which points to the necessity of studying the use of 3DP technology in the fabrication of DMNs.
Apart from the use of FDM and SLA, the two most common 3DP technologies, microneedles have also been successfully produced using some novel 3DP technologies, including DLP, CLIP, and TPP.
An investigation on the use of high precision DLP for the 3D printing of hydrogel MNs in terms of the process parameters were performed by Yao et al. [18]. A dye rhodamine B was used as the model compound for the platform characterisation. Its loading was achieved through soaking of the DLP printed MN in the dye solution. The authors have concluded that the long exposure time enhances the stiffness of MNs, and that with the use of hydrogel, the drug loading capacity was greatly increased. There was also a significant decrease in the fabrication time, which only took a few minutes [18].
The DLP printing of personalised and flexible MN patches has been extensively studied by Lim et al. [83], e.g., the MN patches to treat the trigger finger, which is not achievable with conventional MNs [83]. Their more recent studies on printing MN periorbital patch focused on the relationship between geometries of these microneedles to their mechanical strength and skin penetration efficiency [84,85]. The fabrication involves two steps: the DLP printing of flat MN patches, which are then compressed against a FDM-printed curved substrate to generate flexible, curved MN patches. Acetyl-hexapeptide 3 (AHP-3) is a small peptide and anti-aging active, with very poor skin penetration due to its hydrophilicity and high molecular weight. With the aid of the optimised DLP-printed MN periorbital patch, enhanced anti-wrinkle effect was achieved with significantly improved AHP-3 delivery [84,85].
Different materials and geometries with various aspect ratios of MNs were attempted by Johnson and co-workers [37], using CLIP technology. Square pyramidal needle shape was found to be the most suitable design for encapsulating and delivering a wide range of active ingredients. That shape has been shown to effectively pierce the skin and achieve controlled release of drug, based on the fabrication process of less than 10 min [86]. Another more recent work successfully demonstrated a rapid fabrication method of DIP coating CLIP microneedles for transdermal delivery of therapeutic proteins, achieving a high degree of control over microneedle design parameters [37].
The recent study by Cordeiro et al. [19] has shown that highly precise and reproducible MNs could be successfully manufactured using TPP technology to make silicone MN moulds. MNs with various needle shapes and lengths were then produced by a micro-moulding method. Polyvinylpyrrolidone (PVP) and polyvinyl alcohol (PVA) liquid blend and Polyvinyl methyl ether/maleic acid (PVM/MA) copolymer and poly(ethylene glycol) (PEG) liquid blend were prepared and poured in the silicone moulds to produce dissolvable and ‘super-swellable’ MNs, respectively [19].
The above 3DP technologies generally have high printing resolution and precision, producing micro-sized needle shapes highly suitable for MN-type skin delivery systems. Although significant development in this area has been made, the research is still not widely carried out due to the cost and the need of specialist equipment. It is envisaged that more extensive research will be carried out with further development of 3DP technologies.
4. Materials Used in 3DP Platforms
This section reviews the materials used in the fabrication of 3D printed skin patches and microneedles, focusing specifically on recent sustainability trends by introducing biopolymers [87]. Common 3DP ink materials adopted for cosmetic-relevant application are listed in Table 4.

Table 4.
3DP materials (via direct fabrication only) relevant to cosmetic applications.
- Materials used in 3DP skin patches
Skin patches consist of the backbone support, normally made of polymers, into which the actives could be embedded in different ways.
In their pioneering study at the time, Goyanes et al. [27] used both FDM and SLA methods in the production of the cosmetic patch with anti-acne effect, containing salicylic acid. Several FDM printable soft polymers suitable for cosmetic use were tested, such as polycaprolactone (PCL), thermoplastic polyurethane (NinjaFlex®), and flexible polylactic acid (Flex EcoPLA™). In terms of the loading method, hot melt extrusion was used for the FDM route to dissolve polymer solution in organic solvent with salicylic acid at the concentration of 2% w/w. For the SLA route, the solutions were made up of two photo-linkable materials, poly(ethylene glycol) diacrylate (PEGDA) and poly(ethylene glycol) (PEG) as the photoinitiators, before salicylic acid was added. The results showed that the FDM method was not the right choice; a degradation of salicylic acid occurred due to the high extrusion temperature, while the lower extrusion temperature led to the nozzle blockage. The incorporation of drug also reduced the heat stability of the printable polymers. In contrast, the patches fabricated using SLA method were flexible and smooth, with a higher amount of salicylic acid than that obtained with FDM [27].
A two-layer (PLA/phycocyanin–alginate) novel cosmetic patch was recently studied [115], using a simple casting method for patch fabrication. PLA has been extensively used in the FDM technology, so it was of interest to further study the printability of innovative PLA composite materials. The loaded active, spirulina extract (phycocyanin) has been proven to be safe for cosmetic use as an antioxidant. An optimised formulation has been shown to be a promising patch material, with excellent flexibility and the ability to fully release the active from the phycocyanin–alginate layer of the patch [115].
In the study by Rees et al. on sustainable 3DP materials [116], nanocellulose obtained from renewable sources was used to print a patch-like hydrogel matrix for wound healing. The patch was shown to be effective in inhibiting the growth of common wound pathogen, Pseudomonas aeruginosa. Hence, this platform could be used to tailor-made wound dressings with antimicrobial components. In addition, the porous structure of the 3D printed hydrogel matrix was highly suitable for the incorporation of topical actives, including those for cosmetic applications [116].
A study by Domínguez-Robles et al. [117] has produced a new material by combining naturally occurring polymer lignin with poly lactic acid (PLA). They have used modified hot melt extrusion and FDM technologies for the 3D printing of hydrogel patches for wound dressing. The novel material was proven to be very suitable for the fabrication of 3DP wound dressings, being flexible and able to carry high drug load. Since lignin is abundant, inexpensive, biocompatible, and possesses antioxidant properties, its mixture with PLA might have a wider potential, e.g., for the applications in food packaging, tissue culture, and implants [117].
One difficulty with the traditional cosmetic platform is the loading of a hydrophobic active into a hydrophilic backbone polymer. Sommer et al. [91] have partly solved this problem by 3D printing via direct ink writing (DIW), using an emulsion containing biocompatible soft material. Their oil-in-water emulsion, stabilised by chitosan-modified nano-silica particle, could host both hydrophilic and hydrophobic compounds. Moreover, by printing an active-loaded emulsion onto a regular ink emulsion, the authors achieved a precise positioning of actives within the 3D structure [90]. Since emulsion-based patch is soft and fits the undulated skin, this work opens a new approach for printing cosmetic patches.
In line with sustainability efforts, a recent work studied the effect of the incorporation of starch into alginate-based 3D hydrogel patches for topical delivery. The addition of starch increases pore sizes of the matrix after crosslinking, as well as the structural stability. In principle, the drug release rate from 3DP patches is dependent on drug solubility, the influx of the medium into the structure, and polymer(s) swelling. In this case, the in vitro release results showed that release of actives (chemical dye rhodamine B as a model hydrophobic compound) from the starch-incorporated patch was faster and 20% higher in comparison with the patch that did not contain starch [118].
From the sustainability point of view, it is preferable to use biopolymers in 3DP due to their biodegradability under environmental conditions. In addition, most of them provide flexibility and viscoelasticity to the printed patches, while some exhibit antibacterial effect. A major drawback of this type of 3DP patches is their relatively low rate of drug/active release, which is very difficult to improve [3].
- Materials used in 3DP microneedles
Microneedles must have sufficient strength to penetrate skin without breakage under the force of thumb [119]. The effectiveness of 3D printed microneedles is mostly determined by the material they are made of. A large variety of materials have been investigated for this purpose, including sugarcane (carboxymethylcellulose-based polymers) [120], glass, metal, carbon-based, and silicon-based polymers [11].
The materials so far studied for the fabrication of the active ingredient-loaded 3DP microneedles include solid polymers (e.g., PLA), hydrogels (e.g., hyaluronic acid), and dissolving polymers (e.g., polyvinyl alcohol) [51,69]. A list of materials that were used for the direct 3D printing of microneedles has been summarised in the work by Economidou and Douroumis [121]. The development of 3DP microneedles is still constrained, since the manufacturing process involves heating, UV, laser light or other instability-causing factors, and requires different viscosities for different 3DP technologies [70]. Selected materials must meet the requirements of non-toxicity and high biocompatibility, while being biodegradable and sustainably produced. Therefore, the development of novel 3D printable material is vital for the development of 3D printed microneedles.
In order to penetrate the skin, microneedles have to withstand a certain amount of force without breakage. One of the main concerns about 3D printed polymeric microneedles is their relatively low mechanical strength in comparison with conventional method of moulding. However, it has been shown that successful 3DP microneedles could have comparable mechanical strength to MNs that are constructed by conventional materials [122]. For example, it has been shown that microneedles produced using PLA filament with FDM technology have similar mechanical properties as conventional PLA microneedles made from multistage-micromoulding method [31,123].
The investigation by Tang et al. [124] explored the relationship between printing parameters and the quality of the 3D printed transdermal platform in terms of its surface finish and dimensional accuracy, using PLA as the printing material. The resolution and accuracy of printed microneedles were higher when selecting a thinner layer, a smaller infill width and an increasing spacing between needles, while a better surface finish could be achieved using smaller nozzle orifice [124]. The result also revealed that the process of HME and FDM caused temperature-induced degradation of the polymers.
Attempts have been made to alter the 3D printable materials in order to overcome the constraints of the technology. Sommer et al. [90] demonstrated a new approach to 3D printing of an oil-in-water emulsion stabilised with chitosan-modified silica. They used direct ink writing (DIW), which is a 3DP method that requires mild conditions. The only prerequisite is that the 3DP ink mixture must have a suitable viscoelasticity, which was achieved through optimisation of the stabiliser, followed by centrifugation to obtain an emulsion paste which would be used as the 3DP ink. The rheological study of the paste showed good alignment with the requirements for the 3DP material and revealed the mixture to be shear-thinning and elastic, with fast recovery [90].
Due to its excellent biocompatibility, viscoelasticity, skin moisturising effect [77] and non-immunogenicity, hyaluronic acid (HA) is the material of choice for the cosmetic dissolving microneedles (DMNs), either on its own or as the backbone material onto which to load other skin actives [125,126]. Recent work by Jang et al. [127] has shown that the active delivery achieved by the HA-based DMNs directly depends on the molecular weight of HA. With the same MN size, geometry and active load, high molecular weight HA performs better than low molecular weight, due to a higher degree of swelling when inserted into the skin. Another use of HA-based DMN was in anti-aging as a skin filler, via the mechanism of short-term epidermal expansion after insertion. Zhang et al. [128] have shown that the stability of the DMNs depends on the degree of HA crosslinking using 1,4-butanediol diglycidyl ether. A higher level of crosslinking produced more stable DMNs, which lasted longer in the skin (2–6 days), therefore it was considered a better filler [128].
With micro-moulding method to fabricate DMNs using 3DP, Shim et al. [129] have presented an innovative approach to optimising skin penetration and the dissolution rate of an active. They have evaluated the use of biocompatible, water-soluble polymers, such as carboxymethylcellulose, hyaluronic acid and polyvinylpyrrolidone (PVP), while the actives were ascorbic acid and acetyl hexapeptide-8. It has been shown that increasing the PVP content of the MNs leads to a faster dissolution of the actives in the skin, but also to their compromised skin penetration. Thus, an optimal composition of the printable materials must be established for each active [129].
DLP 3DP technology has been employed for micro-moulding fabrication method by several research groups [130,131]. In the study by El-Sayed et al. [131], a master mould was produced with DLP, followed by polydimethylsiloxane (PDMS) casting to produce a negative mould. The MN array was then prepared using drug-loaded polymer moulding, vacuuming and base-casting with PVA/sucrose, and its removal after drying. Several different shapes of MNs were also fabricated by DLP [131]. These studies present a useful alternative approach in this area.
Other base materials used in cosmetic DMNs include hydroxypropyl methylcellulose (HPMC) and a range of water-soluble polymers, including methylvinyl ether and maleic anhydride copolymer (MVA/MA), polyvinylpyrrolidone (PVP) and chitosan, as well as their combinations [132].
In respect of sustainability, some progress has been made in both the development of novel bioderived [133] or biocomposite [134] 3D printable materials and in the process of production of microneedles using 3DP technology. Clearly, abundant and biodegradable biomaterials should be more widely adopted, as well as 3DP technologies that do not require post-production processes. A close collaboration between these two fields of research is necessary in order to make significant developments in sustainability.
Among the reviewed MN types, dissolvable MNs have shown the best sustainability potential, since they could achieve the highest drug/cosmetic active loading, have a single-step application with minimal waste, and could be made of sustainable biomaterials [126].
5. Characterisation of 3DP Platforms
Testing for stability, safety, and efficacy is a fundamental requirement and must be carried out for both cosmetic and pharmaceutical products. In addition, there is a wide range of physico-mechanical characterisation methods, which could help formulators in their development work, enabling predictions of how products will behave during their production, storage, and use. Some of these methods have the potential to reveal the interactions between active materials and the components of the base. This in turn could explain the observed stability issues, the rate and extent of active ingredient release and ultimately the product efficacy.
Due to the novel nature of the 3DP platforms, a possible interaction (or the lack of it) between the carrier and the active is particularly important. Two aspects of 3DP platforms should be considered: characterisation of finished 3D printed products and characterisation for optimisation of printing process (including intermediates, such as printing filaments).
In terms of the final product, researchers normally report basic physical parameters of the 3DP platforms, including their morphology, geometry, density, and mechanical strength (patch stiffness). For optimising the printing process with extrusion-type 3D printers, the most commonly listed properties for filament polymers include molecular weight, which is measured by gel permeation chromatography (GPC) [124], as well as thermal properties and crystallinity, which are measured by differential scanning calorimetry (DSC) and X-ray diffraction, respectively [135]. These methods are used in addition to well-established standard characterisation techniques for active molecules via in vitro/in vivo permeability studies.
Regarding the 3DP patches, few characterisation methods have been used. Rheological properties, gel strength and bio-adhesive properties are key to an effective 3DP patch. Texture analysers and rheometers are mostly used to determine the printability of active -loaded ink, especially important for extrusion-based 3DP technologies [90]. In addition, the pH of the 3DP patch must be suitable for topical use and its pH value, when in contact with moisture, should be close to the pH of human skin [29].
For 3DP microneedles, the two properties that have drawn most attention are their geometry and their mechanical properties, which can be further divided into insertion force and mechanical strength (failure test), performed by theoretical simulation and/or experiments [74].
Various geometries of MNs have been studied to achieve more defined tip of microneedles, which directly relate to the ability to perforate the SC [136]. The most common shape of MNs are cones, with different aspect ratio, height, interspace, tip diameter and base diameter [121]. There is no standard for the best geometry; in addition, the performance of MNs vary depending on the materials and 3D printers used. Therefore, each parameter must be studied in relation to various ink formulations and 3DP parameters in order to be optimised. Pyramid, cross and spear shapes are also studied, obtained by SLA or other 3DP technologies that have higher resolution [26]. Although FDM printers are easy to use, fast and cost-effective, the technique is generally not suitable for printing the fine structures of MNs. The extrusion manner of printing makes it difficult for printed layers to adhere to one another when the printing area is very small (for sharp tips). Therefore, only shapes that gradually change from the bottom to top could be printed by the FDM method.
The observation of platform morphology and the measurement of their dimensions have been carried out using optical microscopy [31,137], scanning electron microscopy [34,137] and in vivo imaging techniques [138]. Image analysis is particularly useful, because it visualises the shape and uniformity of the MN array, allowing checking for any defects [132].
MN platforms are normally applied by pressing them into the skin with a thumb, hence MNs must have sufficient mechanical strength to provide efficient delivery of the actives into the skin [139]. The upper surface of the skin experiences viscoelastic deformation while being perforated with an increasing force. There is a minimal force necessary to punctuate the intact skin, which must not exceed the maximum force that an individual micro-sized needle can withstand, otherwise the needle will break or fracture before piercing the skin [132]. Therefore, it is important to consider mechanical properties of MNs when designing MN platforms.
A study by Davis et al. [136] first quantified the effect of geometry to the fracture force of MNs. Their theoretical and experimental analysis both led to the same conclusion: the insertion force varies linearly with the interfacial area of the needle tip [136]. It has been proven by many further studies that the smaller the tip diameter, the easier the perforation [34]. However, the tip diameter is limited by the resolution of the 3D printer, particularly for those using FDM technology. A recent study has shown that, by varying the tilted angle of the MN arrays during the SLA printing process, the tip diameter could be significantly changed [140]. Using the printing angle of 45°, the MNs appeared not only sharper but also without defects. However, the optimisation of printing quality and geometry accuracy differs significantly between the 3DP technologies, so it remains challenging to print sophisticated micro-sized needles.
The process of insertion of NMs into the skin has been evaluated by several methods. The penetration test using the membrane that mimics human skin was employed to determine the rate of piercing and the rate of needle breakage after the insertion [31]. Another approach used dye solution applied on the surface of the skin sample, before applying and removing MNs, and analysed the coloured holes produced. In the same study, when the insertion speed was kept constant at 0.5 mm/s, the predicted minimum insertion force through a multilayer skin structure obtained through modelling by Finite Element Analysis for each MN was above 0.03 N. This was consistent with their experimental result of 0.069 N and the literature [34].
Texture analyser has also been employed with skin samples to quantify the insertion of the MN platform, by reporting the continuous force and displacement of microneedle arrays fixed on the top of a moving probe [141]. The mechanical strength or fracture point of MNs were measured in various ways. Transversal, axial, and bending forces were exerted on the MN array to determine the point of mechanical failure by mechanical testers; the shear resistance was also measured [105]. It was found that the 3D printed MNs could be refined post-printing via etching (when using FDM) [31] and post-curing (when using SLA) [140].
Since transepidermal water loss (TEWL) reflects the integrity of the skin barrier, changes in TEWL have also been used to evaluate the effects of MNs penetration [29,142].
Comprehensive evaluation on the physico-mechanical properties of 3D printed platforms is important for their development and optimisation. For FDM-produced 3DP hydrogel patches and dissolving MNs, the addition of actives may significantly change rheological properties of the formulation, leading to a varied mechanical strength of the MNs after solidification process. In such cases, rheological characterisation is being used to evaluate and regulate their viscosity [68,105]. For developing dissolving MNs, it is vital to understand the process of MNs degradation, since the actives are released during this process. SEM provides information on any change in porosity and formation of cracks in the MN structure, while DSC and X-ray diffraction measure the change of crystallinity of the polymer. Since the crystalline region of the MN is where the integrity of the polymer structure was maintained, amorphous regions start to degrade or dissolve first [30].
6. Release and Skin Delivery of Actives Used in 3DP Platforms
This section presents a review of the release and penetration studies that have been performed on 3DP platforms in order to study them as carriers for pharmaceutical and cosmetic active molecules.
Even though a series of examples of 3D printed patches for wound healing have been discussed, the delivery mechanism is different from the one occurring in cosmetic application, since the application sites normally do not have a functioning skin barrier.
All published studies related to the use of 3D printing for the delivery of cosmetic active ingredients are summarised in Table 5, including potential ones. The use of standard delivery platforms (patches and MNs) is widely studied [13,14,49,106], but very few attempts have been made with 3D printed skin delivery platforms. Some methods normally used for tissue engineering, wound dressing, and food industry might be transferable for cosmetic applications.

Table 5.
An overview of cosmetic benefits, active ingredients and 3DP platforms investigated so far.
Skin permeation study is vital to determine the efficacy of 3DP delivery platform by providing information on any enhancement of the active ingredient delivery in comparison to other delivery systems [70,146]. Ideally, the performance of loaded 3DP platforms should be directly compared to non-loaded controls, or topical formulations containing the same active ingredient.
Skin permeation study, which could be performed in vivo or in vitro, is usually preceded by in vitro release study. Franz diffusion cells are the most widely used piece of equipment for the study of drug/cosmetic active release from the formulation, combined by various methods that determine the concentration of active molecules in the receptor medium after a given time period [77,139]. The mechanism of release could be studied by fitting the in vitro release data to various mathematical models, of which Korsmeyer-Peppas release model (1) is often used. It describes the relationship between the fraction of active ingredient released () and the release time (t), using the release rate constant (k). The analysis takes into account the order of kinetics (n) and the constant k, assessing how close the release data are to the theoretical model [35,48].
Other methods that are used include spectral imaging, e.g., confocal microscopy, which produces high-resolution images that visualise the site of skin penetration after the administration of MNs, including the geometry of MNs [84,131].
Confocal Raman spectroscopy (CRS) is an optical method that combines the principles of confocal microscopy and Raman spectroscopy for the study of in vivo penetration of topically applied substances. In a recent study [147], the skin penetration of niacinamide from various formulations was tested in vitro by the conventional Franz cells and in vivo using a quantitative CRS method under finite dose conditions; high correlation was found between the results. Krombholz and Lunter [148] have also applied the CRS method to the skin penetration of caffeine, showing the potential of achieving in situ measurement via CRS.
Another novel in vitro analytical approach, Time of Flight—Secondary Ion Mass Spectrometry (ToF-SIMS) imaging, was recently used in the testing of cosmetics, and shown to be capable of measuring multiple compounds in the skin, simultaneously and in one single experiment [149]. This would be a very useful technique for the in vivo assessment of the efficacy of 3DP platforms.
3D printed patches have simple geometry in general, such as film and grid/mesh. The earliest attempt to 3D-print a patch was the work by Goyanes et al. [27], with salicylic acid, an effective lipophilic keratolytic agent for acne treatment. A personalised nose-shape patch was created to provide complete occlusion and facilitate skin delivery of salicylic acid. The in vitro release of active from a standard formulation and a 3DP patch was performed using Franz diffusion cell. The initial release was slow, as expected for all delivery platforms. However, the SLA printed patch has shown a faster diffusion rate and a higher amount of total drug released within 3 h. An enhancement in salicylic acid delivery was observed by all 3DP types of patches in comparison to the standard formulation [27].
The follow-up study by the same group was performed to investigate the personalised wound dressing made by 3D scanning followed by FDM and containing a series of antimicrobial agents. In line to the intended purpose, the release of actives from wound dressings has been reported as a dissolution rate (not the amount), since the skin barrier of the wound was disrupted [28]. It was observed that a higher active loading has resulted in a higher release from the 3D printed delivery platforms, since there was less hindering by the polymer matrix. However, this conclusion is not applicable for actives that are chemically bonded to the ink material. Therefore, studying chemical interactions between the 3DP platform matrix and the loaded actives is necessary for better understanding of the release mechanisms and further optimisation.
3D printed wound dressings are the most studied patch-type skin delivery platforms. Biopolymers and hydrogels are the materials that have been tested for their printability by 3DP technologies. It was shown that rheological properties and swellability are the key features that determine the release of actives [105,116,137,150].
3D printing of emulsions into grids was performed by Sommer et al. [90], whereby a hydrophobic dye, unisol blue, was incorporated as model active. It is worth noting that the emulsion has the potential to incorporate and simultaneously deliver hydrophilic and hydrophobic actives to the skin [90].
Antioxidants are essential in anti-aging products, but also present in topical pharmaceuticals, hence they are the actives of interest for the 3DP platforms. One of them is lignin, an abundant natural antioxidant and antimicrobial ingredient, which was used in the 3DP patch intended for wound healing [117]. The printable filament was obtained by coating the PLA pellet with lignin solution, before applying hot melt extrusion. The resulting 3D printed patch was obtained by FDM. The product showed lower resistance to fracture and higher wettability than the one without lignin. In vitro release results showed that the delivery of the model drug, curcumin, was prolonged when loaded onto the 3D printed lignin-PLA mesh [117].
Recently, Wang et al. [29] developed a 3D printed hydrogel patch with cryptotanshinone (CPT), a quinoid diterpene with anti-acne effect. To counteract its poor water solubility, CPT was entrapped in non-ionic surfactant–based vesicles, niosomes, before dispersing them into the hydrogel for 3D printing [29]. A comparison of in vivo permeation results was performed between an active-free 3DP patch, a conventional CPT-loaded patch, and a 3DP CPT-loaded patch, in addition to a commercial adapalene product, as a positive control. The results showed that the 3DP patch has shown better hydration and anti-acne effect, with CPT being delivered to deeper layers of the skin, indicating a positive effect of niosomes. However, there was a lack of direct comparison of the release results between the gel formulation with CPT-loaded noisomes and the 3DP patch with niosomes, therefore, it is hard to validate that the 3DP printed patch itself was the reason for the improved results [29].
There is enough evidence to conclude that 3DP technology has the potential to increase the efficacy of topically applied actives, specifically in the form of microneedles, where two mechanisms combine to enhance skin delivery: occlusion and the formation of micro-punctures. Hence, it would be worth exploring the possibilities within the cosmetic field more extensively.
7. Research in Borderline Areas
Recently, 3D bioprinting has drawn considerable interest from the cosmetic industry in the areas of safety testing and characterisation of new products. 3D bioprinting refers to the extrusion of organic and biological materials in a layer-by-layer manner. The skin substitute could be produced by incorporating cells with bioinks such as collagen, gelatine, and alginate. A recent work by Olejnik et al. [151] has comprehensively reviewed the application of 3D bioprinting of skin.
The production of conventional cosmetic facial masks relies on the use of fossil-based fibres, which is not an environmentally friendly approach. Recently, a novel 3DP biodegradable cosmetic patch (beauty mask) was developed using the 3DP extrusion method. The material used for the fabric of mask was a blend of poly(hydroxyalkanoates) and starch [152].
An interesting recent study [153] has focused on reducing packaging material via 3D printing. The process involved the fabrication of montelukast sodium-loaded filaments by hot melt extrusion (HME), followed by extrusion-based 3D printing of transdermal patches directly onto packaging material. This has minimised contamination and reduced transportation issues. The material used as the backing sheet was aluminium, but biodegradable materials will be used in future work. Another study [135] adopted the same principle, using HME and FDM combined method to 3D print a skin patch directly on the polyester backing film. The results showed that the optimised formulation produced a skin patch that was smooth, uniform, and flexible, with a good release profile of the drug for the treatment of pulmonary tuberculosis [135].
A study by Zhang et al. [128] has revealed the concept of ‘all-purpose ice MNs’, capable of delivering macromolecules and living micro-organisms, which is extremely difficult to achieve with conventional skin delivery systems. The main principle was the freezing of water, present in all MN gel formulations, which produced the necessary needle sharpness. The micro-moulding method was used to prepare the MNs, which then could be easily detached from the mould after freezing. This solves a major problem presented by the viscoelastic nature of most gel formulations, which prevents them from being shaped into defined, micro-sized needles. Microneedles of less than 1 mm in length were manufactured successfully and have shown satisfactory skin penetration for all tested formulations. This study has overcome the limitations of standard 3D printable materials and might pave the way for the new generation of MNs. Because of its simplicity, reproducibility, and versatility, it has a considerable potential to be commercialised as a personalised skin delivery system [128].
8. Conclusions and Future Prospects
This review presents an overview and a critical assessment of the principles and main types of 3DP technology, production and characteristics of two key 3DP skin delivery platforms (patches and microneedles), and topical active ingredients used in these platforms. It considers relevant developments in a range of borderline areas, specifically in the medical area, aiming to inform and inspire prospective applications of 3D printing in cosmetics.
Based on the exponential growth in research on 3D printed MN technologies and the numerous associated advantages of this approach, it is envisaged that 3D printed MNs would gain prevalence over patches for both pharmaceutical and cosmetic applications. Dissolving MNs have shown the highest potential of the various MN types investigated so far, especially taking into account that comfort and convenience present major factors for consumer acceptance of cosmetic products.
Giving the limitations of conventional skin delivery systems [46,154], 3D printing might be able to provide personalised solutions. On the other hand, there are still difficulties in developing 3D printed skin delivery platforms, including the delivery of large molecules, skin irritation related to both MNs and patches, incomplete control of drug/cosmetic active release, and sustainability issues related to the raw materials used in 3DP. Due to the high cost of 3DP, when developing 3D printed skin delivery platforms, the priority will probably be given to personalised systems over mass production.
Although intense research has been carried out in the field of medical and pharmaceutical use of 3D printed microneedles during the last decade, the same does not apply to the field of cosmetic delivery. This indicates a large potential in developing efficient cosmetic delivery platforms using 3DP technology.
Author Contributions
Conceptualization, methodology, S.T. and Y.J.; writing—original draft preparation, Y.J.; writing—review and editing, S.T., A.B., M.S., M.J.U.; supervision, S.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hadgraft, J.; Lane, M.E. Advanced topical formulations (ATF). Int. J. Pharm. 2016, 514, 52–57. [Google Scholar] [CrossRef]
- Luo, L.; Lane, M.E. Topical and transdermal delivery of caffeine. Int. J. Pharm. 2015, 490, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Economidou, S.N.; Lamprou, D.A.; Douroumis, D. 3D printing applications for transdermal drug delivery. Int. J. Pharm. 2018, 544, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Menditto, E.; Orlando, V.; de Rosa, G.; Minghetti, P.; Musazzi, U.M.; Cahir, C.; Kurczewska-Michalak, M.; Kardas, P.; Costa, E.; Lobo, J.M.S.; et al. Patient centric pharmaceutical drug product design—The impact on medication adherence. Pharmaceutics 2020, 12, 44. [Google Scholar] [CrossRef]
- Martinez, P.R.; Goyanes, A.; Basit, A.W.; Gaisford, S. Fabrication of drug-loaded hydrogels with stereolithographic 3D printing. Int. J. Pharm. 2017, 532, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Trenfield, S.J.; Awad, A.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D Printing Pharmaceuticals: Drug Development to Frontline Care. Trends Pharmacol. Sci. 2018, 39, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.A.; Olawuni, D.; Kimbell, G.; Badruddoza, A.Z.M.; Hossain, M.S.; Sultana, T. Polymers for extrusion-based 3D printing of pharmaceuticals: A holistic materials–process perspective. Pharmaceutics 2020, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Chinga-Carrasco, G.; Ehman, N.V.; Filgueira, D.; Johansson, J.; Vallejos, M.E.; Felissia, F.E.; Håkansson, J.; Area, M.C. Bagasse—A major agro-industrial residue as potential resource for nanocellulose inks for 3D printing of wound dressing devices. Addit. Manuf. 2019, 28, 267–274. [Google Scholar] [CrossRef]
- Varaprasad, K.; Jayaramudu, T.; Kanikireddy, V.; Toro, C.; Sadiku, E.R. Alginate-based composite materials for wound dressing application:A mini review. Carbohydr. Polym. 2020, 236, 116025. [Google Scholar] [CrossRef] [PubMed]
- Souto, E.B.; Campos, J.C.; Filho, S.C.; Teixeira, M.C.; Martins-Gomes, C.; Zielinska, A.; Carbone, C.; Silva, A.M. 3D printing in the design of pharmaceutical dosage forms. Pharm. Dev. Technol. 2019, 24, 1044–1053. [Google Scholar] [CrossRef]
- Bird, D.; Eker, E.; Ravindra, N.M. 3D printing of pharmaceuticals and transdermal drug delivery—An overview. In Proceedings of the TMS 2019 148th Annual Meeting & Exhibition Supplemental Proceedings, San Antonio, TX, USA, 10–14 March 2019; Springer International Publishing: Cham, Switzerland, 2019; pp. 1563–1573. [Google Scholar]
- Elahpour, N.; Pahlevanzadeh, F.; Kharaziha, M.; Bakhsheshi-Rad, H.R.; Ramakrishna, S.; Berto, F. 3D printed microneedles for transdermal drug delivery: A brief review of two decades. Int. J. Pharm. 2021, 597, 120301. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yu, H.; Wang, L.; Shen, D.; Ni, Z.; Ren, S.; Lu, Y.; Chen, X.; Yang, J.; Hong, Y. Research progress on cosmetic microneedle systems: Preparation, property and application. Eur. Polym. J. 2022, 163, 110942. [Google Scholar] [CrossRef]
- Hu, X.; He, H. A review of cosmetic skin delivery. J. Cosmet. Dermatol. 2021, 20, 2020–2030. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.S.; Fantaus, S.S.; Guillot, A.J.; Melero, A.; Beck, R.C.R. 3D-Printed Products for Topical Skin Applications: From Personalized Dressings to Drug Delivery. Pharmaceutics 2021, 13, 1946. [Google Scholar] [CrossRef]
- Trenfield, S.J.; Awad, A.; Madla, C.M.; Hatton, G.B.; Firth, J.; Goyanes, A.; Gaisford, S.; Basit, A.W. Shaping the future: Recent advances of 3D printing in drug delivery and healthcare. Expert Opin. Drug Deliv. 2019, 16, 1081–1094. [Google Scholar] [CrossRef] [PubMed]
- Shahrubudin, N.; Lee, T.C.; Ramlan, R. An overview on 3D printing technology: Technological, materials, and applications. Procedia Manuf. 2019, 35, 1286–1296. [Google Scholar] [CrossRef]
- Yao, W.; Li, D.; Zhao, Y.; Zhan, Z.; Jin, G.; Liang, H.; Yang, R. 3D Printed Multi-Functional Hydrogel Microneedles Based on High-Precision Digital Light Processing. Micromachines 2019, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, A.S.; Tekko, I.A.; Jomaa, M.H.; Vora, L.; McAlister, E.; Volpe-Zanutto, F.; Nethery, M.; Baine, P.T.; Mitchell, N.; McNeill, D.W.; et al. Two-Photon Polymerisation 3D Printing of Microneedle Array Templates with Versatile Designs: Application in the Development of Polymeric Drug Delivery Systems. Pharm. Res. 2020, 37, 174. [Google Scholar] [CrossRef]
- Pere, C.P.P.; Economidou, S.N.; Lall, G.; Ziraud, C.; Boateng, J.S.; Alexander, B.D.; Lamprou, D.A.; Douroumis, D. 3D printed microneedles for insulin skin delivery. Int. J. Pharm. 2018, 544, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Economidou, S.N.; Pere, C.P.P.; Reid, A.; Uddin, M.J.; Windmill, J.F.C.; Lamprou, D.A.; Douroumis, D. 3D printed microneedle patches using stereolithography (SLA)for intradermal insulin delivery. Mater. Sci. Eng. C 2019, 102, 743–755. [Google Scholar] [CrossRef]
- Evans, S.E.; Harrington, T.; Rivero, M.C.R.; Rognin, E.; Tuladhar, T.; Daly, R. 2D and 3D inkjet printing of biopharmaceuticals—A review of trends and future perspectives in research and manufacturing. Int. J. Pharm. 2021, 599, 120443. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.; Haider, N.; Jain, K. 3D Printing in Personalized Drug Delivery. Curr. Pharm. Des. 2019, 24, 5062–5071. [Google Scholar] [CrossRef] [PubMed]
- Vithani, K.; Goyanes, A.; Jannin, V.; Basit, A.W.; Gaisford, S.; Boyd, B.J. An Overview of 3D Printing Technologies for Soft Materials and Potential Opportunities for Lipid-based Drug Delivery Systems. Pharm. Res. 2019, 36, 4. [Google Scholar] [CrossRef]
- Stevic, M.; Hill, P.; Tamburic, S. Exploring the use of 3D Printing Technology in the Fabrication of Personalised Lipstick Applicators. J. Dermatol. Cosmetol. 2017, 1, 00024. [Google Scholar] [CrossRef][Green Version]
- Yang, Q.; Zhong, W.; Xu, L.; Li, H.; Yan, Q.; She, Y.; Yang, G. Recent progress of 3D-printed microneedles for transdermal drug delivery. Int. J. Pharm. 2021, 593, 120106. [Google Scholar] [CrossRef] [PubMed]
- Goyanes, A.; Det-Amornrat, U.; Wang, J.; Basit, A.W.; Gaisford, S. 3D scanning and 3D printing as innovative technologies for fabricating personalized topical drug delivery systems. J. Control Release 2016, 234, 41–48. [Google Scholar] [CrossRef]
- Muwaffak, Z.; Goyanes, A.; Clark, V.; Basit, A.W.; Hilton, S.T.; Gaisford, S. Patient-specific 3D scanned and 3D printed antimicrobial polycaprolactone wound dressings. Int. J. Pharm. 2017, 527, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, L.; Xiang, S.; Jiang, C.; Wu, W.; Ruan, S.; Du, Q.; Chen, T.; Xue, Y.; Chen, H.; et al. Formulation and Characterization of a 3D-Printed Cryptotanshinone-Loaded Niosomal Hydrogel for Topical Therapy of Acne. AAPS PharmSciTech 2020, 21, 159. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.O.; Simon, G.P. Biodegradation of 3D-printed polylactic acid milliprojections under physiological conditions. J. Appl. Polym. Sci. 2020, 137. [Google Scholar] [CrossRef]
- Luzuriaga, M.A.; Berry, D.R.; Reagan, J.C.; Smaldone, R.A.; Gassensmith, J.J. Biodegradable 3D printed polymer microneedles for transdermal drug delivery. Lab Chip 2018, 18, 1223–1230. [Google Scholar] [CrossRef]
- Camović, M.; Biščević, A.; Brčić, I.; Borčak, K.; Bušatlić, S.; Ćenanović, N.; Dedović, A.; Mulalić, A.; Osmanlić, M.; Sirbubalo, M.; et al. Coated 3D printed PLA microneedles as transdermal drug delivery systems. In Proceedings of the CMBEBIH 2019, Banja Luka, Bosnia and Herzegovina, 16–18 May 2019; Springer International Publishing: Cham, Switzerland, 2019; pp. 735–742. [Google Scholar]
- Wu, M.; Zhang, Y.; Huang, H.; Li, J.; Liu, H.; Guo, Z.; Xue, L.; Liu, S.; Lei, Y. Assisted 3D printing of microneedle patches for minimally invasive glucose control in diabetes. Mater. Sci. Eng. C 2020, 117, 111299. [Google Scholar] [CrossRef] [PubMed]
- Xenikakis, I.; Tzimtzimis, M.; Tsongas, K.; Andreadis, D.; Demiri, E.; Tzetzis, D.; Fatouros, D.G. Fabrication and finite element analysis of stereolithographic 3D printed microneedles for transdermal delivery of model dyes across human skin in vitro. Eur. J. Pharm. Sci. 2019, 137, 104976. [Google Scholar] [CrossRef] [PubMed]
- Amer, R.I.; El-Osaily, G.H.; Bakr, R.O.; el Dine, R.S.; Fayez, A.M. Characterization and Pharmacological Evaluation of Anti-Cellulite Herbal Product(s) Encapsulated in 3D-Fabricated Polymeric Microneedles. Sci. Rep. 2020, 10, 6316. [Google Scholar] [CrossRef]
- Gittard, S.D.; Miller, P.R.; Jin, C.; Martin, T.N.; Boehm, R.D.; Chisholm, B.J.; Stafslien, S.J.; Daniels, J.W.; Cilz, N.; Monteiro-Riviere, N.A.; et al. Deposition of antimicrobial coatings on microstereolithography-fabricated microneedles. JOM 2011, 63, 59–68. [Google Scholar] [CrossRef]
- Caudill, C.L.; Perry, J.L.; Tian, S.; Luft, J.C.; DeSimone, J.M. Spatially controlled coating of continuous liquid interface production microneedles for transdermal protein delivery. J. Control Release 2018, 284, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Pazhamannil, R.V.; Govindan, P. Current state and future scope of additive manufacturing technologies via vat photopolymerization. Mater. Today Proc. 2021, 43, 130–136. [Google Scholar] [CrossRef]
- Ovsianikov, A.; Chichkov, B.; Mente, P.; Monteiro-Riviere, N.A.; Doraiswamy, A.; Narayan, R.J. Two Photon Polymerization of Polymer-Ceramic Hybrid Materials for Transdermal Drug Delivery. Int. J. Appl. Ceram. Technol. 2007, 4, 22–29. [Google Scholar] [CrossRef]
- Doraiswamy, A.; Ovsianikov, A.; Gittard, S.D.; Monteiro-Riviere, N.A.; Crombez, R.; Montalvo, E.; Shen, W.; Chichkov, B.N.; Narayan, R.J. Fabrication of Microneedles Using Two Photon Polymerization for Transdermal Delivery of Nanomaterials. J. Nanosci. Nanotechnol. 2010, 10, 6305–6312. [Google Scholar] [CrossRef] [PubMed]
- Gittard, S.D.; Ovsianikov, A.; Chichkov, B.N.; Doraiswamy, A.; Narayan, R.J. Two-photon polymerization of microneedles for transdermal drug delivery. Expert Opin. Drug Deliv. 2010, 7, 513–533. [Google Scholar] [CrossRef]
- Anantrao, J.H.; Nath, P.A.; Nivrutti, P.R. Drug Penetration Enhancement Techniques in Transdermal Drug Delivery System: A Review. J. Pharm. Res. Int. 2021, 33, 46–61. [Google Scholar] [CrossRef]
- Chandan, S.; Nishant, T.; Bhupinder, K.; Manish, G. Recent advancements in transdermal patches. Int. J. Health Sci. 2022, 6, 6443–6460. [Google Scholar] [CrossRef]
- Pastore, M.N.; Kalia, Y.N.; Horstmann, M.; Roberts, M.S. Transdermal patches: History, development and pharmacology. Br. J. Pharmacol. 2015, 172, 2179–2209. [Google Scholar] [CrossRef] [PubMed]
- Kadam, C.Y.; Muchandi, A.; Alabade, P.P.; Narwade, P.P.; Khandwe, S.R. Transdermal Drug Delivery System: A Painless Method for Healthy Skin—A Review. Int. J. Sci. Dev. Res. 2022, 7, 123–130. [Google Scholar]
- Brooks, Z.; Goswami, T.; Neidhard-Doll, A.; Goswami, T. Transdermal drug delivery systems: Analysis of adhesion failure. J. Pharm. Biopharm. Res. 2022, 4, 256–270. [Google Scholar] [CrossRef]
- Cilurzo, F.; Gennari, C.G.M.; Minghetti, P. Adhesive properties: A critical issue in transdermal patch development. Expert Opin. Drug Deliv. 2012, 9, 33–45. [Google Scholar] [CrossRef]
- Brown, M.B.; Traynor, M.J.; Martin, G.P.; Akomeah, F.K. Transdermal drug delivery systems: Skin perturbation devices. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2008; pp. 119–139. [Google Scholar]
- Patravale, V.B.; Mandawgade, S.D. Novel cosmetic delivery systems: An application update. Int. J. Cosmet. Sci. 2008, 30, 19–33. [Google Scholar] [CrossRef]
- Hadgraft, J.; Lane, M.E. Drug crystallization—Implications for topical and transdermal delivery. Expert Opin. Drug Deliv. 2016, 13, 817–830. [Google Scholar] [CrossRef]
- Bird, D.; Ravindra, N.M. Transdermal Drug Delivery and Patches—An Overview. Med. Devices Sens. 2020, 3, e10069. [Google Scholar] [CrossRef]
- Prodduturi, S.; Sadrieh, N.; Wokovich, A.M.; Doub, W.H.; Westenberger, B.J.; Buhse, L. Transdermal delivery of fentanyl from matrix and reservoir systems: Effect of heat and compromised skin. J. Pharm. Sci. 2010, 99, 2357–2366. [Google Scholar] [CrossRef]
- Pawar, R.; Mishra, D.N.; Pawar, N. An Updated Review on Global Pharmaceutical Formulation Developments and Future Potential of Non-invasive Transdermal Drug Delivery System. Int. J. Pharm. Sci. Res. 2022, 13, 1896–1907. [Google Scholar]
- Reddy, R.D.P.; Sharma, V. Additive manufacturing in drug delivery applications: A review. Int. J. Pharm. 2020, 589, 119820. [Google Scholar] [CrossRef]
- Sirbubalo, M.; Tucak, A.; Muhamedagic, K.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Cekic, A.; Begic-Hajdarevic, D.; Husic, M.C.; Dervišević, A.; et al. 3D Printing—A “Touch-Button” Approach to Manufacture Microneedles for Transdermal Drug Delivery. Pharmaceutics 2021, 13, 924. [Google Scholar] [CrossRef]
- Hirao, T. Structure and function of skin from a cosmetic aspect. In Cosmetic Science and Technology: Theoretical Principles and Applications; Sakamoto, K., Lochhead, R., Maibach, H., Yamashita, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 673–683. [Google Scholar]
- Kochhar, J.S.; Tan, J.J.Y.; Kwang, Y.C.; Kang, L. Microneedles for Transdermal Drug Delivery; Springer International Publishing AG: Cham, Switzerland, 2019. [Google Scholar]
- Teymourian, H.; Tehrani, F.; Mahato, K.; Wang, J. Lab under the Skin: Microneedle Based Wearable Devices. Adv. Healthc. Mater. 2021, 10, e2002255. [Google Scholar] [CrossRef]
- Fonseca, D.F.S.; Vilela, C.; Silvestre, A.J.D.; Freire, C.S.R. A compendium of current developments on polysaccharide and protein-based microneedles. Int. J. Biol. Macromol. 2019, 136, 704–728. [Google Scholar] [CrossRef] [PubMed]
- Guillot, A.J.; Cordeiro, A.S.; Donnelly, R.F.; Montesinos, M.C.; Garrigues, T.M.; Melero, A. Microneedle-based delivery: An overview of current applications and trends. Pharmaceutics 2020, 12, 569. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.D.; Zhang, X.P.; Zhang, B.L.; Hao, Y.Y.; Guo, X.D. Safety Assessment of Microneedle Technology for Transdermal Drug Delivery: A Review. Adv. Ther. 2020, 3, 2000033. [Google Scholar] [CrossRef]
- Aldawood, F.K.; Andar, A.; Desai, S. A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications. Polymers 2021, 13, 2815. [Google Scholar] [CrossRef]
- Markiewicz, A.; Zasada, M.; Erkiert-Polguj, A.; Wieckowska-Szakiel, M.; Budzisz, E. An evaluation of the antiaging properties of strawberry hydrolysate treatment enriched with L-ascorbic acid applied with microneedle mesotherapy. J. Cosmet. Dermatol. 2019, 18, 129–135. [Google Scholar] [CrossRef]
- Serrano-Castañeda, P.; Escobar-Chávez, J.J.; Rodríguez-Cruz, I.M.; Melgoza-Contreras, L.M.; Martínez-Hernández, J. Microneedles as enhancer of drug absorption through the skin and applications in medicine and cosmetology. J. Pharm. Pharm. Sci. 2018, 21, 73–93. [Google Scholar] [CrossRef]
- Cohen, I.D.; Bratescu, D.; Althea, K.E.; Thomas, M. Dissolvable Microneedles Comprising One or More Encapsulated Cosmetic Ingredients. U.S. Patent Application No. US20140200509A1, 17 July 2014. [Google Scholar]
- Choi, S.Y.; Kwon, H.J.; Ahn, G.R.; Ko, E.J.; Yoo, K.H.; Kim, B.J.; Lee, C.; Kim, D. Hyaluronic acid microneedle patch for the improvement of crow’s feet wrinkles. Dermatol. Ther. 2017, 30, e12546. [Google Scholar] [CrossRef]
- Bhatnagar, S.; Dave, K.; Venuganti, V.V.K. Microneedles in the clinic. J. Control. Release 2017, 260, 164–182. [Google Scholar] [CrossRef]
- McCrudden, M.T.; Alkilani, A.Z.; McCrudden, C.M.; McAlister, E.; McCarthy, H.O.; Woolfson, A.D.; Donnelly, R.F. Design and physicochemical characterisation of novel dissolving polymeric microneedle arrays for transdermal delivery of high dose, low molecular weight drugs. J. Control. Release 2014, 180, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Gadeela, P.R.; Thathireddy, P.; Venuganti, V.V.K. Microneedle-based drug delivery: Materials of construction. J. Chem. Sci. 2019, 131, 90. [Google Scholar] [CrossRef]
- Kang, N.; Kim, S.; Lee, J.; Kim, K.; Choi, Y.; Oh, Y.; Kim, J.; Kim, D.; Park, J. Microneedles for drug delivery: Recent advances in materials and geometry for preclinical and clinical studies. Expert Opin. Drug Deliv. 2021, 18, 929–947. [Google Scholar] [CrossRef]
- Lee, C.; Eom, Y.A.; Yang, H.; Jang, M.; Jung, S.U.; Park, Y.O.; Lee, S.E.; Jung, H. Skin Barrier Restoration and Moisturization Using Horse Oil-Loaded Dissolving Microneedle Patches. Ski. Pharmacol. Physiol. 2018, 31, 163–171. [Google Scholar] [CrossRef]
- Koyani, R.D. Biopolymers for microneedle synthesis: From then to now. Biomanufacturing Rev. 2019, 4, 1. [Google Scholar] [CrossRef]
- Lee, K.; Lee, H.C.; Lee, D.; Jung, H. Drawing Lithography: Three-Dimensional Fabrication of an Ultrahigh-Aspect-Ratio Microneedle. Adv. Mater. 2010, 22, 483–486. [Google Scholar] [CrossRef]
- Ebrahiminejad, V.; Rad, Z.F.; Prewett, P.D.; Davies, G.J. Fabrication and testing of polymer microneedles for transdermal drug delivery. Beilstein J. Nanotechnol. 2022, 13, 629–640. [Google Scholar] [CrossRef]
- Sonetha, V.; Majumdar, S.; Shah, S. Step-wise micro-fabrication techniques of microneedle arrays with applications in transdermal drug delivery—A review. J. Drug Deliv. Sci. Technol. 2022, 68, 103119. [Google Scholar] [CrossRef]
- Kim, J.D.; Kim, M.; Yang, H.; Lee, K.; Jung, H. Droplet-born air blowing: Novel dissolving microneedle fabrication. J. Control. Release 2013, 170, 430–436. [Google Scholar] [CrossRef]
- Kim, M.; Yang, H.; Kim, H.; Jung, H.; Jung, H. Novel cosmetic patches for wrinkle improvement: Retinyl retinoate- and ascorbic acid-loaded dissolving microneedles. Int. J. Cosmet. Sci. 2014, 36, 207–212. [Google Scholar] [CrossRef]
- Kim, S.; Yang, H.; Kim, M.; Baek, J.H.; Kim, S.J.; An, S.M.; Koh, J.S.; Seo, R.; Jung, H. 4-n-butylresorcinol dissolving microneedle patch for skin depigmentation: A randomized, double-blind, placebo-controlled trial. J. Cosmet. Dermatol. 2016, 15, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Dangol, M.; Kang, G.; Lahiji, S.F.; Yang, H.; Jang, M.; Ma, Y.; Li, C.; Lee, S.G.; Kim, C.H.; et al. Enhanced Transdermal Delivery by Combined Application of Dissolving Microneedle Patch on Serum-Treated Skin. Mol. Pharm. 2017, 14, 2024–2031. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Yang, H.; Kim, S.; Kim, M.; Kang, H.; Kim, N.; An, S.; Koh, J.; Jung, H. Evaluation of the anti-wrinkle effect of an ascorbic acid-loaded dissolving microneedle patch via a double-blind, placebo-controlled clinical study. Int. J. Cosmet. Sci. 2016, 38, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kim, S.; Jang, M.; Kim, H.; Lee, S.; Kim, Y.; Eom, Y.A.; Kang, G.; Chiang, L.; Baek, J.H.; et al. Two-phase delivery using a horse oil and adenosine-loaded dissolving microneedle patch for skin barrier restoration, moisturization, and wrinkle improvement. J. Cosmet. Dermatol. 2019, 18, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Song, C.; Baik, S.; Kim, D.; Hyeon, T.; Kim, D. Device-assisted transdermal drug delivery. Adv. Drug Deliv. Rev. 2018, 127, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Ng, J.Y.; Kang, L. Three-dimensional printing of a microneedle array on personalized curved surfaces for dual-pronged treatment of trigger finger. Biofabrication 2017, 9, 015010. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Tiew, W.J.; Zhang, J.; Ho, P.C.L.; Kachouie, N.N.; Kang, L. Geometrical optimisation of a personalised microneedle eye patch for transdermal delivery of anti-wrinkle small peptide. Biofabrication 2020, 12, 035003. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Kathuria, H.; Amir, M.H.B.; Zhang, X.; Duong, H.T.T.; Ho, P.C.L.; Kang, L. High resolution photopolymer for 3D printing of personalised microneedle for transdermal delivery of anti-wrinkle small peptide. J. Control. Release 2021, 329, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.R.; Caudill, C.L.; Tumbleston, J.R.; Bloomquist, C.J.; Moga, K.A.; Ermoshkin, A.; Shirvanyants, D.; Mecham, S.J.; Luft, J.C.; DeSimone, J.M. Single-step fabrication of computationally designed microneedles by continuous liquid interface production. PLoS ONE 2016, 11, e0162518. [Google Scholar] [CrossRef] [PubMed]
- Zamboulis, A.; Michailidou, G.; Koumentakou, I.; Bikiaris, D.N. Polysaccharide 3D Printing for Drug Delivery Applications. Pharmaceutics 2022, 14, 145. [Google Scholar] [CrossRef] [PubMed]
- Diañez, I.; Gallegos, C.; la Fuente, E.B.; Martínez, I.; Valencia, C.; Sánchez, M.C.; Diaz, M.; Franco, J. 3D printing in situ gelification of κ-carrageenan solutions: Effect of printing variables on the rheological response. Food Hydrocoll. 2019, 87, 321–330. [Google Scholar] [CrossRef]
- Boonlai, W.; Tantishaiyakul, V.; Hirun, N. Characterization of κ-carrageenan/methylcellulose/cellulose nanocrystal hydrogels for 3D bioprinting. Polym. Int. 2021, 71, 181–191. [Google Scholar] [CrossRef]
- Sommer, M.R.; Alison, L.; Minas, C.; Tervoort, E.; Rühs, P.A.; Studart, A.R. 3D printing of concentrated emulsions into multiphase biocompatible soft materials. Soft Matter. 2017, 13, 1794–1803. [Google Scholar] [CrossRef] [PubMed]
- Aranaz, I.; Acosta, N.; Civera, C.; Elorza, B.; Mingo, J.; Castro, C.; de Los Llanos Gandía, M.; Caballero, A.H. Cosmetics and Cosmeceutical Applications of Chitin, Chitosan and Their Derivatives. Polymers 2018, 10, 213. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, M.; McConnell, M.; Cabral, J.; Ali, M.A. Chitosan hydrogels in 3D printing for biomedical applications. Carbohydr. Polym. 2021, 260, 117768. [Google Scholar] [CrossRef]
- Camcı, Y.; Türk, S.; Gepek, E.; İyibilgin, O.; Özsoy, M.İ. Fabrication and characterization of innovative chitosan/doxorubicin coated 3D printed microneedle patch for prolonged drug delivery. J. Appl. Polym. Sci. 2022, 139. [Google Scholar] [CrossRef]
- Bukhari, S.N.A.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S.; Sohail, M.; Ramli, N.A.; Thu, H.E.; Hussain, Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: A review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695. [Google Scholar] [CrossRef]
- Graça, M.F.P.; Miguel, S.P.; Cabral, C.S.D.; Correia, I.J. Hyaluronic acid—Based wound dressings: A review. Carbohydr. Polym. 2020, 241, 116364. [Google Scholar] [CrossRef]
- Maiz-Fernández, S.; Barroso, N.; Pérez-Álvarez, L.; Silván, U.; Vilas-Vilela, J.L.; Lanceros-Mendez, S. 3D printable self-healing hyaluronic acid/chitosan polycomplex hydrogels with drug release capability. Int. J. Biol. Macromol. 2021, 188, 820–832. [Google Scholar] [CrossRef]
- Mbituyimana, B.; Liu, L.; Ye, W.; Boni, B.O.O.; Zhang, K.; Chen, J.; Thomas, S.; Vasilievich, R.V.; Shi, Z.; Yang, G. Bacterial cellulose-based composites for biomedical and cosmetic applications: Research progress and existing products. Carbohydr. Polym. 2021, 273, 118565. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, D.J.; Maliha, M.; Raghuwanshi, V.S.; Browne, C.; Mouterde, L.M.M.; Simon, G.P.; Allais, F.; Garnier, G. Diethyl sinapate-grafted cellulose nanocrystals as nature-inspired UV filters in cosmetic formulations. Mater. Today Bio 2021, 12, 100126. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Teong, Z.K.; Bakir, A.N.; Sajab, M.S.; Kaco, H. Extending Cellulose-Based Polymers Application in Additive Manufacturing Technology: A Review of Recent Approaches. Polymers 2020, 12, 1876. [Google Scholar] [CrossRef]
- Li, V.C.; Dunn, C.K.; Zhang, Z.; Deng, Y.; Qi, H.J. Direct Ink Write (DIW) 3D Printed Cellulose Nanocrystal Aerogel Structures. Sci. Rep. 2017, 7, 8018. [Google Scholar] [CrossRef]
- Xiong, S.; Zhang, X.; Lu, P.; Wu, Y.; Wang, Q.; Sun, H.; Heng, B.C.; Bunpetch, V.; Zhang, S.; Ouyang, H. A Gelatin-sulfonated Silk Composite Scaffold based on 3D Printing Technology Enhances Skin Regeneration by Stimulating Epidermal Growth and Dermal Neovascularization. Sci. Rep. 2017, 7, 4288. [Google Scholar] [CrossRef] [PubMed]
- Al-Nimry, S.; Dayah, A.A.; Hasan, I.; Daghmash, R. Cosmetic, Biomedical and Pharmaceutical Applications of Fish Gelatin/Hydrolysates. Mar. Drugs 2021, 19, 145. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Kumar, H.; Tian, Z.; Jin, X.; Holzman, J.F.; Menard, F.; Kim, K. Visible Light Photoinitiation of Cell-Adhesive Gelatin Methacryloyl Hydrogels for Stereolithography 3D Bioprinting. ACS Appl. Mater. Interfaces 2018, 10, 26859–26869. [Google Scholar] [CrossRef]
- Liu, J.; Tagami, T.; Ozeki, T. Fabrication of 3D-Printed Fish-Gelatin-Based Polymer Hydrogel Patches for Local Delivery of PEGylated Liposomal Doxorubicin. Mar. Drugs 2020, 18, 325. [Google Scholar] [CrossRef]
- Karavasili, C.; Tsongas, K.; Andreadis, I.I.; Andriotis, E.G.; Papachristou, E.T.; Papi, R.M.; Tzetzis, D.; Fatouros, D.G. Physico-mechanical and finite element analysis evaluation of 3D printable alginate-methylcellulose inks for wound healing applications. Carbohydr. Polym. 2020, 247, 116666. [Google Scholar] [CrossRef]
- Hajleh, M.N.A.; Al-Samydai, A.A.; Al-Dujaili, E.A.S. Nano, micro particulate and cosmetic delivery systems of polylactic acid: A mini review. J. Cosmet. Dermatol. 2020, 19, 2805–2811. [Google Scholar] [CrossRef]
- Rydz, J.; Sikorska, W.; Musioł, M.; Janeczek, H.; Włodarczyk, J.; Misiurska-Marczak, M.; Łęczycka, J.; Kowalczuk, M. 3D-Printed Polyester-Based Prototypes for Cosmetic Applications—Future Directions at the Forensic Engineering of Advanced Polymeric Materials. Materials 2019, 12, 994. [Google Scholar] [CrossRef]
- Blanco, I. End-life Prediction of Commercial PLA Used for Food Packaging through Short Term TGA Experiments: Real Chance or Low Reliability? Chin. J. Polym. Sci. 2014, 32, 681–689. [Google Scholar] [CrossRef]
- Kesente, M.; Kavetsou, E.; Roussaki, M.; Blidi, S.; Loupassaki, S.; Chanioti, S.; Siamandoura, P.; Stamatogianni, C.; Philippou, E.; Papaspyrides, C.J.B.; et al. Encapsulation of Olive Leaves Extracts in Biodegradable PLA Nanoparticles for Use in Cosmetic Formulation. Bioengineering 2017, 4, 75. [Google Scholar] [CrossRef] [PubMed]
- Ausejo, J.G.; Rydz, J.; Musioł, M.; Sikorska, W.; Sobota, M.; Włodarczyk, J.; Adamus, G.; Janeczek, H.; Kwiecień, I.; Hercog, A.; et al. A comparative study of three-dimensional printing directions: The degradation and toxicological profile of a PLA/PHA blend. Polym. Degrad. Stab. 2018, 152, 191–207. [Google Scholar] [CrossRef]
- Asthana, N.; Pal, K.; Aljabali, A.A.A.; Tambuwala, M.M.; de Souza, F.G.; Pandey, K. Polyvinyl alcohol (PVA) mixed green–clay and aloe vera based polymeric membrane optimization: Peel-off mask formulation for skin care cosmeceuticals in green nanotechnology. J. Mol. Struct. 2021, 1229, 129592. [Google Scholar] [CrossRef]
- Badri, W.; Miladi, K.; Eddabra, R.; Fessi, H.; Elaissari, A. Elaboration of Nanoparticles Containing Indomethacin: Argan Oil for Transdermal Local and Cosmetic Application. J. Nanomater. 2015, 16, 113. [Google Scholar] [CrossRef]
- Yang, S.; Jeong, J.; Lim, Y.; Park, J. Synthesis and characterization of PVP microneedle patch using metal bioelectrodes for novel drug delivery system. Mater. Des. 2021, 201, 109485. [Google Scholar] [CrossRef]
- Park, Y.; Park, J.; Chu, G.S.; Kim, K.S.; Sung, J.H.; Kim, B. Transdermal delivery of cosmetic ingredients using dissolving polymer microneedle arrays. Biotechnol. Bioprocess Eng. 2015, 20, 543–549. [Google Scholar] [CrossRef]
- Adli, S.A.; Ali, F.; Azmi, A.S.; Anuar, H.; Nasir, N.A.M.; Hasham, R.; Idris, M.K.H. Development of biodegradable cosmetic patch using a polylactic acid/phycocyanin-alginate composite. Polymers 2020, 12, 1669. [Google Scholar] [CrossRef]
- Rees, A.; Powell, L.C.; Chinga-Carrasco, G.; Gethin, D.T.; Syverud, K.; Hill, K.E.; Thomas, D.W. 3D Bioprinting of Carboxymethylated-Periodate Oxidized Nanocellulose Constructs for Wound Dressing Applications. BioMed Res. Int. 2015, 2015, 925757. [Google Scholar] [CrossRef]
- Domínguez-Robles, J.; Martin, N.K.; Fong, M.L.; Stewart, S.A.; Irwin, N.J.; Rial-Hermida, M.I.; Donnelly, R.F.; Larrañeta, E. Antioxidant pla composites containing lignin for 3D printing applications: A potential material for healthcare applications. Pharmaceutics 2019, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Bom, S.; Santos, C.; Barros, R.; Martins, A.M.; Paradiso, P.; Cláudio, R.; Pinto, P.; Ribeiro, H.; Marto, J. Effects of starch incorporation on the physicochemical properties and release kinetics of alginate-based 3D hydrogel patches for topical delivery. Pharmaceutics 2020, 12, 719. [Google Scholar] [CrossRef] [PubMed]
- Nagarkar, R.; Singh, M.; Nguyen, H.X.; Jonnalagadda, S. A review of recent advances in microneedle technology for transdermal drug delivery. J. Drug Deliv. Sci. Technol. 2020, 59, 101923. [Google Scholar] [CrossRef]
- Loizidou, E.Z.; Williams, N.A.; Barrow, D.A.; Eaton, M.J.; McCrory, J.; Evans, S.L.; Allender, C.J. Structural characterisation and transdermal delivery studies on sugar microneedles: Experimental and finite element modelling analyses. Eur. J. Pharm. Biopharm. 2015, 89, 224–231. [Google Scholar] [CrossRef]
- Economidou, S.N.; Douroumis, D. 3D printing as a transformative tool for microneedle systems: Recent advances, manufacturing considerations and market potential. Adv. Drug Deliv. Rev. 2021, 173, 60–69. [Google Scholar] [CrossRef]
- Wallis, M.; Al-Dulimi, Z.; Tan, D.K.; Maniruzzaman, M.; Nokhodchi, A. 3D printing for enhanced drug delivery: Current state-of-the-art and challenges. Drug Dev. Ind. Pharm. 2020, 46, 1385–1401. [Google Scholar] [CrossRef]
- Cha, K.J.; Kim, T.; Park, S.J.; Kim, D.S. Simple and cost-effective fabrication of solid biodegradable polymer microneedle arrays with adjustable aspect ratio for transdermal drug delivery using acupuncture microneedles. J. Micromechanics Microengineering 2014, 24, 115015. [Google Scholar] [CrossRef]
- Tang, T.O.; Holmes, S.; Dean, K.; Simon, G.P. Design and fabrication of transdermal drug delivery patch with milliprojections using material extrusion 3D printing. J. Appl. Polym. Sci. 2020, 137, 48777. [Google Scholar] [CrossRef]
- Zvezdin, V.; Kasatkina, T.; Kasatkin, I.; Gavrilova, M.; Kazakova, O. Microneedle patch based on dissolving, detachable microneedle technology for improved skin quality of the periorbital region. Part 2: Clinical Evaluation. Int. J. Cosmet. Sci. 2020, 42, 429–435. [Google Scholar] [CrossRef]
- Zvezdin, V.; Peno-Mazzarino, L.; Radionov, N.; Kasatkina, T.; Kasatkin, I. Microneedle patch based on dissolving, detachable microneedle technology for improved skin quality—Part 1: Ex vivo safety evaluation. Int. J. Cosmet. Sci. 2020, 42, 369–376. [Google Scholar] [CrossRef]
- Jang, M.; Baek, S.; Kang, G.; Yang, H.; Kim, S.; Jung, H. Dissolving microneedle with high molecular weight hyaluronic acid to improve skin wrinkles, dermal density and elasticity. Int. J. Cosmet. Sci. 2020, 42, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Fu, X.; Chen, G.; Wang, Y.; Zhao, Y. Versatile Ice Microneedles for Transdermal Delivery of Diverse Actives. Adv. Sci. 2021, 8, e2101210. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.S.; Hwang, Y.M.; Park, S.G.; Lee, C.K.; Kang, N.G. Role of Polyvinylpyrrolidone in Dissolving Microneedle for Efficient Transdermal Drug Delivery: In vitro and Clinical Studies. Bull. Korean Chem. Soc. 2018, 39, 789–793. [Google Scholar] [CrossRef]
- Fang, J.; Liu, C.; Hsu, R.; Chen, Y.; Chiang, W.; Wang, H.D.; Hu, S.-H. Transdermal Composite Microneedle Composed of Mesoporous Iron Oxide Nanoraspberry and PVA for Androgenetic Alopecia Treatment. Polymers 2020, 12, 1392. [Google Scholar] [CrossRef]
- El-Sayed, N.; Vaut, L.; Schneider, M. Customized fast-separable microneedles prepared with the aid of 3D printing for nanoparticle delivery. Eur. J. Pharm. Biopharm. 2020, 154, 166–174. [Google Scholar] [CrossRef]
- Aung, N.N.; Ngawhirunpat, T.; Rojanarata, T.; Patrojanasophon, P.; Opanasopit, P.; Pamornpathomkul, B. HPMC/PVP Dissolving Microneedles: A Promising Delivery Platform to Promote Trans-Epidermal Delivery of Alpha-Arbutin for Skin Lightening. AAPS PharmSciTech 2020, 21, 25. [Google Scholar] [CrossRef]
- Wasti, S.; Adhikari, S. Use of Biomaterials for 3D Printing by Fused Deposition Modeling Technique: A Review. Front. Chem. 2020, 8, 315. [Google Scholar] [CrossRef]
- le Duigou, A.; Correa, D.; Ueda, M.; Matsuzaki, R.; Castro, M. A review of 3D and 4D printing of natural fibre biocomposites. Mater. Des. 2020, 194, 108911. [Google Scholar] [CrossRef]
- Chaudhari, V.S.; Malakar, T.K.; Murty, U.S.; Banerjee, S. Extruded filaments derived 3D printed medicated skin patch to mitigate destructive pulmonary tuberculosis: Design to delivery. Expert Opin. Drug Deliv. 2020, 18, 301–313. [Google Scholar] [CrossRef]
- Davis, S.P.; Landis, B.J.; Adams, Z.H.; Allen, M.G.; Prausnitz, M.R. Insertion of microneedles into skin: Measurement and prediction of insertion force and needle fracture force. J. Biomech. 2004, 37, 1155–1163. [Google Scholar] [CrossRef]
- Arany, P.; Róka, E.; Mollet, L.; Coleman, A.W.; Perret, F.; Kim, B.; Kovács, R.; Kazsoki, A.; Zelkó, R.; Gesztelyi, R.; et al. Fused depositionmodeling 3D printing: Test platforms for evaluating post-fabrication chemical modifications and in-vitro biological properties. Pharmaceutics 2019, 11, 277. [Google Scholar]
- Hao, Y.Y.; Yang, Y.; Li, Q.Y.; Zhang, X.P.; Shen, C.B.; Zhang, C.; Cui, Y.; Guo, X.D. Effect of polymer microneedle pre-treatment on drug distributions in the skin in vivo. J. Drug Target. 2020, 28, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lahiji, S.F.; Jang, J.; Jang, M.; Jung, H. Micro-pillar integrated dissolving microneedles for enhanced transdermal drug delivery. Pharmaceutics 2019, 11, 402. [Google Scholar] [CrossRef] [PubMed]
- Economidou, S.N.; Pere, C.P.P.; Okereke, M.; Douroumis, D. Optimisation of Design and Manufacturing Parameters of 3D Printed Solid Microneedles for Improved Strength, Sharpness, and Drug Delivery. Micromachines 2021, 12, 117. [Google Scholar] [CrossRef]
- Economidou, S.N.; Uddin, M.J.; Marques, M.J.; Douroumis, D.; Sow, W.T.; Li, H.; Reid, A.; Windmill, J.F.; Podoleanu, A. A Novel 3D Printed Hollow Microneedle Microelectromechanical System for Controlled, Personalised Transdermal Drug Delivery. Addit. Manuf. 2020, 38, 101815. [Google Scholar]
- Donnelly, R.F.; Singh, T.R.R.; Garland, M.J.; Migalska, K.; Majithiya, R.; McCrudden, C.M.; Kole, P.L.; Mahmood, T.M.T.; McCarthy, H.O.; Woolfson, A.D. Hydrogel-forming microneedle arrays for enhanced transdermal drug delivery. Adv. Funct. Mater. 2012, 22, 4879–4890. [Google Scholar] [CrossRef]
- Ratz-Łyko, A.; Arct, J. Resveratrol as an active ingredient for cosmetic and dermatological applications: A review. J. Cosmet. Laser Ther. 2019, 21, 84–90. [Google Scholar] [CrossRef]
- Siddique, S.; Parveen, Z.; Ali, Z.; Zaheer, M. Qualitative and Quantitative Estimation of Hydroquinone in Skin Whitening Cosmetics. J. Cosmet. Dermatol. Sci. Appl. 2012, 2, 224–228. [Google Scholar] [CrossRef]
- Tabriz, A.G.; Douroumis, D. Recent advances in 3D printing for wound healing: A systematic review. J. Drug Deliv. Sci. Technol. 2022, 74, 103564. [Google Scholar] [CrossRef]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef]
- Iliopoulos, F.; Lane, M.E.; Caspers, P.; Puppels, G. Franz cell diffusion testing and quantitative confocal raman spectroscopy: In vitro-in vivo correlation. Pharmaceutics 2020, 12, 887. [Google Scholar] [CrossRef]
- Krombholz, R.; Lunter, D. A new method for in-situ skin penetration aAnalysis by confocal raman microscopy. Molecules 2020, 25, 4222. [Google Scholar] [CrossRef] [PubMed]
- Munem, M.; Djuphammar, A.; Sjölander, L.; Hagvall, L.; Malmberg, P. Animal- free skin permeation analysis using mass spectrometry imaging. Toxicol. Vitr. 2021, 71, 105062. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tan, C.; Li, L. Review of 3D printable hydrogels and constructs. Mater. Des. 2018, 159, 20–38. [Google Scholar] [CrossRef]
- Olejnik, A.; Semba, J.A.; Kulpa, A.; Dańczak-Pazdrowska, A.; Rybka, J.D.; Gornowicz-Porowska, J. 3D Bioprinting in Skin Related Research: Recent Achievements and Application Perspectives. ACS Synth. Biol. 2022, 11, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.; Panariello, L.; Morganti, P.; Danti, S.; Baroni, A.; Lazzeri, A.; Fusco, A.; Donnarumma, G. Skin-Compatible Biobased Beauty Masks Prepared by Extrusion. J. Funct. Biomater. 2020, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Azizoğlu, E.; Özer, Ö. Fabrication of Montelukast sodium loaded filaments and 3D printing transdermal patches onto packaging material. Int. J. Pharm. 2020, 587, 119588. [Google Scholar] [CrossRef]
- Alkilani, A.; McCrudden, M.T.; Donnelly, R. Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Barrier Properties of the Stratum Corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).