Criticisms in the Development of High-Protection and Broad-Spectrum “Natural/Organic” Certifiable Sunscreen
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instruments
2.3. Analysis of Surface Morphology
2.4. Formulations
2.5. In Vitro Evaluation of Filtering Parameters
- E (λ) = erythema action spectrum (CIE-1987) at a wavelength λ.
- I (λ) = spectral irradiance received from the UV source at a wavelength λ.
- A (λ) = a monochromatic absorbance of the test product layer at a wavelength.
- d (λ) = wavelength step (1 nm).
- P (λ) = Persistent Pigment Darkening (PPD) action spectrum.
- I (λ) = spectral irradiance received from the UV source (UVA 320–400 nm for PPD testing).
- A (λ) = Mean monochromatic absorbance of the test product layer.
- C = Coefficient of adjustment.
- d (λ) = Wavelength step (1 nm).
2.6. Stability of the Formulas
2.7. Optical Analysis
2.8. Sunscreens’ Appearance and Transparency
3. Results
3.1. Chemical-Physical Parameters
3.2. Stability
3.3. Optical Microscopy
3.4. Morphological Study
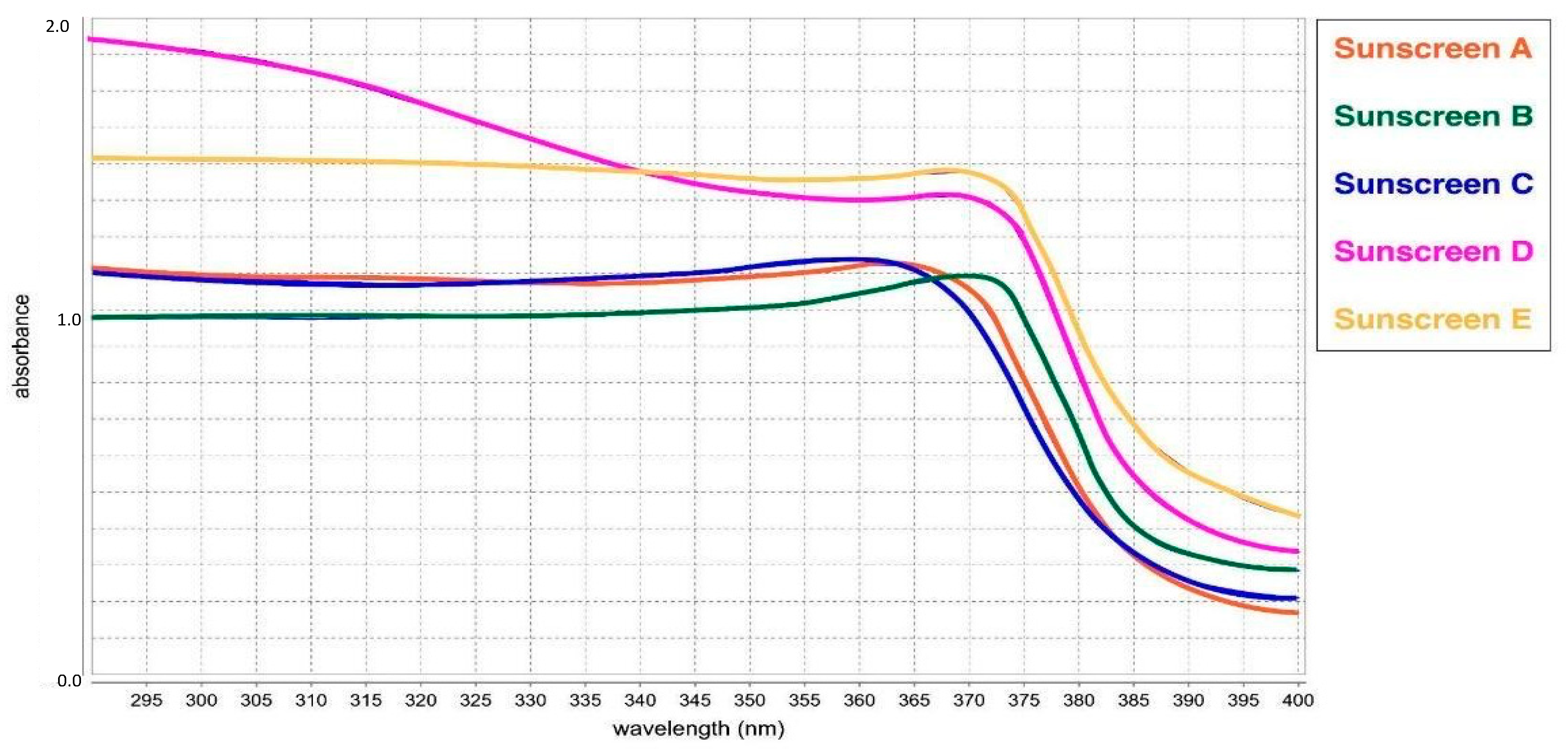
3.5. Analysis of UV Absorption Performance
3.6. Sunscreens’ Appearance and Transparency
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Acharya, S.; Bali, S.; Bhatia, B.S. Exploring Consumer Behavior towards Sustainability of Green Cosmetics. In Proceedings of the 2021 International Conference on Advances in Electrical, Computing, Communication and Sustainable Technologies (ICAECT), Bhilai, India, 19–20 February 2021; pp. 1–6. [Google Scholar] [CrossRef]
- Positano Gian Andrea, Natural and Sustainable Cosmetics: Research Results, Economic Survey, Cosmetica Italia Conference, Milan, Italy. 1 December 2020. Available online: https://www.cosmeticaitalia.it/export/sites/default/centro-studi/indagini-tematiche/erboristeria/Positano-Risultati-di-ricerca-sul-CCNS-2020.pdf (accessed on 21 February 2022).
- Cosmetica Italia, Annual Report 53rd Analysis of the Cosmetics Industry and Consumption in Italy in 2020. Available online: https://www.cosmeticaitalia.it/documenti/a_centrostudi/beauty_report/Rapporto-Annuale-21_EN.pdf (accessed on 14 May 2022).
- Cosmetica Italia Study Center, Congiunturale, Cosmetica Italia. 24 February 2022. Available online: https://www.cosmeticaitalia.it/export/sites/default/centro-studi/indagine-congiunturale/Congiunturale-2022-02-24.pdf (accessed on 14 May 2022).
- Masory, A. Millennials and Preferences in Beauty and Personal Care Products, AlixPartners. 2019. Available online: https://www.alixpartners.com/insights-impact/insights/millennials-preferences-beauty-personal-care-products/ (accessed on 14 May 2022).
- International Organization for Standardization. Guidelines on Technical Definitions and Criteria for Natural and Organic Cosmetic Ingredients and Products—Part 1: Definitions for Ingredients (ISO 16128-1:2016); IOS: Geneva, Switzerland, 2016. [Google Scholar]
- International Organization for Standardization. Guidelines on Technical Definitions and Criteria for Natural and Organic Cosmetic Ingredients and Products—Part 2: Criteria for Ingredients and Products (ISO 16128-2:2017); IOS: Geneva, Switzerland, 2017. [Google Scholar]
- COSMOS-Standard AISBL. COSMOS Position Statement Regarding ISO 16128, February 2018. Available online: https://cosmosstandard.files.wordpress.com/2018/08/cosmos-iso-position-paper-february-2018-2.pdf (accessed on 21 January 2022).
- Cvetkovska, A.D.; Manfredini, S.; Ziosi, P.; Molesini, S.; Dissette, V.; Magri, I.; Scapoli, C.; Carrieri, A.; Durini, E.; Vertuani, S. Factors affecting SPF in vitro measurement and correlation with in vivo results. Int. J. Cosmet. Sci. 2017, 39, 310–319. [Google Scholar] [CrossRef] [PubMed]
- COSMOS-standard AISBL, COSMOS Position on Mineral UV-Filters in Organic and Natural Cosmetics, 10 August 2016. Available online: https://cosmosstandard.files.wordpress.com/2014/08/cosmos-position-mineral-uv-filters-100816.pdf (accessed on 21 January 2022).
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products. Current Consolidated Version: 01/10/2021. Available online: https://eur-lex.europa.eu/eli/reg/2009/1223/2021-10-01 (accessed on 21 January 2022).
- Egambaram, O.P.; Pillai, S.K.; Ray, S.S. Materials Science Challenges in Skin UV Protection: A Review. Photochem. Photobiol. 2020, 96, 779–797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berardesca, E.; Zuberbier, T.; Viera, M.S.; Marinovich, M. Review of the safety of octocrylene used as an ultraviolet filter in cosmetics. J. Eur. Acad. Dermatol. Venereol. 2019, 33 (Suppl. 7), 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernauer, U.; Bodin, L.; Chaudhry, Q.; Coenraads, P.J.; Dusinska, M.; Ezendam, J.; Gaffet, E.; Galli, C.L.; Granum, B.; Panteri, E.; et al. SCCS OPINION ON Octocrylene—SCCS/1627/21—Final Opinion. Scientific Committee for Consumer Safety (SCCS, EC). 2021, Scientific Committee for Consumer Safety (SCCS, EC). Available online: https://ec.europa.eu/health/system/files/2021-04/sccs_o_249_0.pdf (accessed on 21 January 2022).
- Moeller, M.; Pawlowski, S.; Petersen-Thiery, M.; Miller, I.B.; Nietzer, S.; Heisel-Sure, Y.; Kellermann, M.Y.; Schupp, P.J. Challenges in Current Coral Reef Protection—Possible Impacts of UV Filters Used in Sunscreens, a Critical Review. Front. Mar. Sci. 2021, 8, 383. [Google Scholar] [CrossRef]
- Miller, I.B.; Pawlowski, S.; Kellermann, M.Y.; Petersen-Thiery, M.; Moeller, M.; Nietzer, S.; Schupp, P.J. Toxic effects of UV filters from sunscreens on coral reefs revisited: Regulatory aspects for “reef safe” products. Environ. Sci. Eur. 2021, 33, 74. [Google Scholar] [CrossRef]
- Radice, M.; Manfredini, S.; Ziosi, P.; Dissette, V.; Buso, P.; Fallacara, A.; Vertuani, S. Herbal extracts, lichens and biomolecules as natural photo-protection alternatives to synthetic UV filters. A systematic review. Fitoterapia 2016, 114, 144–162. [Google Scholar] [CrossRef] [PubMed]
- Surber, C.; Plautz, J.; Dähnhardt-Pfeiffer, S.; Osterwalder, U. Size matters! issues and challenges with nanoparticulate UV filters. Curr. Probl. Dermatol. 2021, 55, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Battistin, M.; Dissette, V.; Bonetto, A.; Durini, E.; Manfredini, S.; Marcomini, A.; Casagrande, E.; Brunetta, A.; Ziosi, P.; Molesini, S.; et al. A New Approach to UV Protection by Direct Surface Functionalization of TiO2 with the Antioxidant Polyphenol Dihydroxyphenyl Benzimidazole Carboxylic Acid. Nanomaterials 2020, 10, 231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dudley, D.K.; Laughlin, S.A.; Osterwalder, U. Spectral homeostasis—The fundamental requirement for an ideal sunscreen. Chall. Sun Prot. 2021, 55, 72–92. [Google Scholar] [CrossRef]
- Battistin, M.; Durini, E.; Dissette, V.; Bonetto, A.; Marcomini, A.; Casagrande, E.; Brunetta, A.; Ziosi, P.; Molesini, S.; Gavioli, R.; et al. Synthesis and Characterization of New Multifunctional Self-Boosted Filters for UV Protection: ZnO Complex with Dihydroxyphenyl Benzimidazole Carboxylic Acid. Molecules 2019, 24, 4546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, S.; Huang, J.; Jiang, X.; Huang, Y.; Zhu, X.; Cai, Z. Environmental fate and toxicity of sunscreen-derived inorganic ultraviolet filters in aquatic environments: A review. Nanomaterials 2022, 12, 699. [Google Scholar] [CrossRef] [PubMed]
- Pal, A.; Hadagalli, K.; Bhat, P.; Goel, V.; Mandal, S. Hydroxyapatite—A promising sunscreen filter. J. Aust. Ceram. Soc. 2020, 56, 345–351. [Google Scholar] [CrossRef]
- Carella, F.; Degli Esposti, L.; Adamiano, A.; Iafisco, M. The Use of Calcium Phosphates in Cosmetics, State of the Art and Future Perspectives. Materials 2021, 14, 6398. [Google Scholar] [CrossRef] [PubMed]
- Nery, É.M.; Martinez, R.M.; Velasco, M.V.R.; Baby, A.R. A short review of alternative ingredients and technologies of inorganic UV filters. J. Cosmet. Dermatol. 2021, 20, 1061–1065. [Google Scholar] [CrossRef]
- Mueen, R.; Lerch, M.; Cheng, Z.; Konstantinov, K. Na-doped ZnO UV filters with reduced photocatalytic activity for sunscreen applications. J. Mater. Sci. 2020, 55, 2772–2786. [Google Scholar] [CrossRef]
- International Organization for Standardization. Determination of Sunscreen UVA Photoprotection In Vitro (ISO 24443:2012); IOS: Geneva, Switzerland, 2012. [Google Scholar]
- Commission Recommendation of 22 September 2006 on the Efficacy of Sunscreen Products and the Claims Made Relating Thereto. (Notified Under Document Number C(2006) 4089). Off. J. Eur. Union 2006, 265, 39–43. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:265:0039:0043:en:PDF (accessed on 21 January 2022).
- Reinosa, J.J.; Leret, P.; Álvarez-Docio, C.M.; Del Campo, A.; Fernández, J.F. Enhancement of UV absorption behavior in ZnO-TiO2 composites. Bol. Soc. Esp. Ceram. Vidr. 2016, 55, 55–62. [Google Scholar] [CrossRef] [Green Version]
- Lu, P.J.; Fang, S.W.; Cheng, W.L.; Huang, S.C.; Huang, M.C.; Cheng, H.F. Characterization of titanium dioxide and zinc oxide nanoparticles in sunscreen powder by comparing different measurement methods. J. Food Drug Anal. 2018, 26, 1192–1200. [Google Scholar] [CrossRef] [PubMed]








| INCI Name | Description | CAS Number | Ratio of Solid (%) | Average Particle Size Declared by the Producer |
|---|---|---|---|---|
| ZINC OXIDE | Uncoated zinc oxide powder | 1314-13-2 | 100 | 500 nm |
| ZINC OXIDE, SODIUM LAUROYL GLUTAMATE, LYSINE, MAGNESIUM CHLORIDE | Zinc oxide coated sodium lauroyl glutamate and lysine and Magnesium Chloride | 1314-13-2 | N.A. | 294 nm |
| 29923-31-7 | ||||
| 70-54-2 | ||||
| 7786-30-3 | ||||
| ZINC OXIDE, STEARIC ACID | Zinc oxide coated with stearic acid | 1314-13-2 | N.A. | 150 nm |
| 57-11-4 | ||||
| TITANIUM DIOXIDE (NANO), ALUMINUM HYDROXIDE, STEARIC ACID | highly micronized titanium dioxide coated with aluminum hydroxide and stearic acid | 13463-67-7 | 78 | 15 nm |
| 21645-51-2 | ||||
| 57-11-4 | ||||
| TITANIUM DIOXIDE, SILICA, JOJOBA ESTERS | Titanium dioxide coated with silica and jojoba esters | 13463-67-7 | 76-81 | 500 nm (aggregate size) |
| 7631-86-9 | ||||
| 61789-91-1 |
| Phase | Ingredient (INCI Name) | Function | Sunscreen Formula A % in Formula | Sunscreen Formula B % in Formula | Sunscreen Formula C % in Formula |
|---|---|---|---|---|---|
| A | CAPRYLIC/CAPRIC TRIGLYCERIDE | Emollient | 10.00 | 10.00 | 10.00 |
| A | ISOAMYL COCOATE | Emollient | 10.00 | 10.00 | 10.00 |
| A | JOJOBA ESTERS HELIANTHUS ANNUUS SEED CERA POLYGLYCERIN-3 ACACIA DECURRENS FLOWER CERA | Emollient | 5.00 | 5.00 | 5.00 |
| A | TOCOPHEROL HELIANTHUS ANNUUS SEED OIL | Antioxidant | 0.20 | 0.20 | 0.20 |
| A | POLYHYDROXYSTEARIC ACID CAPRYLIC/CAPRIC TRIGLYCERIDE ISOSTEARIC ACID LECITHIN POLYGLYCERYL-3 POLYRICINOLEATE | Dispersant | 1.25 | 1.25 | 1.25 |
| A | ZINC OXIDE | UV Filter | 25.00 | 0 | 0 |
| A | ZINC OXIDE SODIUM LAUROYL GLUTAMATE LYSINE MAGNESIUM CHLORIDE | UV Filter | 0 | 25.00 | 0 |
| A | ZINC OXIDE, STEARIC ACID | UV Filter | 0 | 0 | 25.00 |
| B | POLYGLYCERYL-2 OLEATE POLYHYDROXYSTEARIC ACID POLYGLYCERYL-2 STEARATE | W/O Emulsifier | 5.00 | 5.00 | 5.00 |
| C | AQUA | to 100 | to 100 | to 100 | |
| C | GLYCERIN | Humectant | 5.00 | 5.00 | 5.00 |
| C | SODIUM CHLORIDE | Stabilizer | 0.60 | 0.60 | 0.60 |
| C | MAGNESIUM SULFATE | Stabilizer | 0.60 | 0.60 | 0.60 |
| C | BENZYL ALCOHOL | Preservative | 0.90 | 0.90 | 0.90 |
| C | DEHYDROACETIC ACID | Preservative | 0.10 | 0.10 | 0.10 |
| C | XANTHAN GUM | Viscosity modifier | 0.10 | 0.10 | 0.10 |
| C | CITRIC ACID | pH regulator | 0.03 | 0.03 | 0.03 |
| Phase | Ingredient (INCI Name) | Function | Sunscreen Formula D: % in Formula | Sunscreen Formula E: % in Formula |
|---|---|---|---|---|
| A | CAPRYLIC/CAPRIC TRIGLYCERIDE | Emollient | 10.00 | 10.00 |
| A | ISOAMYL COCOATE | Emollient | 10.00 | 10.00 |
| A | JOJOBA ESTERS HELIANTHUS ANNUUS SEED CERA POLYGLYCERIN-3 ACACIA DECURRENS FLOWER CERA | Emollient | 5.00 | 5.00 |
| A | TOCOPHEROL HELIANTHUS ANNUUS SEED OIL | Antioxidant | 0.20 | 0.20 |
| A | POLYHYDROXYSTEARIC ACID CAPRYLIC/CAPRIC TRIGLYCERIDE ISOSTEARIC ACID LECITHIN POLYGLYCERYL-3 POLYRICINOLEATE | Dispersant | 1.25 | 1.25 |
| A | ZINC OXIDE SODIUM LAUROYL GLUTAMATE LYSINE MAGNESIUM CHLORIDE | UV Filter | 25.00 | 25.00 |
| A | TITANIUM DIOXIDE (nano) ALUMINUM HYDROXIDE STEARIC ACID | UV Filter | 8.00 | 0 |
| A | TITANIUM DIOXIDE, SILICA, JOJOBA ESTERS | UV Filter | 0 | 8.00 |
| B | POLYGLYCERYL-2 OLEATE POLYHYDROXYSTEARIC ACID POLYGLYCERYL-2 STEARATE | W/O Emulsifier | 5.00 | 5.00 |
| C | AQUA | to 100 | to 100 | |
| C | GLYCERIN | Humectant | 5.00 | 5.00 |
| C | SODIUM CHLORIDE | Stabilizer | 0.60 | 0.60 |
| C | MAGNESIUM SULFATE | Stabilizer | 0.60 | 0.60 |
| C | BENZYL ALCOHOL | Preservative | 0.90 | 0.90 |
| C | DEHYDROACETIC ACID | Preservative | 0.10 | 0.10 |
| C | XANTHAN GUM | Viscosity modifier | 0.10 | 0.10 |
| C | CITRIC ACID | pH regulator | 0.03 | 0.03 |
| Formulation | Density (g/mL) | Viscosity (cP), 25 °C, Discharge Time | Viscosity (cP), RT, 1 Month after | Viscosity (cP), 45 °C, 1 Month after |
|---|---|---|---|---|
| Sunscreen Formula A | 1.23 | 24,000 (spindle RV06, 10 rpm) | 250,000 (spindle RV06, 10 rpm) | 390,000 (spindle RV06, 10 rpm) |
| Sunscreen Formula B | 1.22 | 40,000 (spindle RV06, 10 rpm) | 74,000 (spindle RV06, 10 rpm) | 110,000 (spindle RV 06, 10 rpm) |
| Sunscreen Formula C | 1.23 | 125,000 (spindle T-E95, 10 rpm) | 360,000 (spindle T-E95, 10 rpm) | 540,000 (spindle T-E95, 10 rpm) |
| Sunscreen Formula D | 1.31 | 38,000 (spindle RV06, 10 rpm) | 135,000 (spindle RV06, 10 rpm) | 380,000 (spindle RV06, 10 rpm) |
| Sunscreen Formula E | 1.30 | 32,000 (spindle RV06, 10 rpm) | 72,000 (spindle RV06, 10 rpm) | 260,000 (spindle RV 06, 10 rpm) |
| Formulation | SPF | UVAPF | SPF Label | SPF Label/ UVAPF | λ Critical (nm) |
|---|---|---|---|---|---|
| sunscreen formula A | 12.26 | 7.52 | 10 | 1.33 | 374 |
| sunscreen formula B | 11.23 | 8.82 | 10 | 0.89 | 377 |
| sunscreen formula C | 11.86 | 7.29 | 10 | 1.37 | 374 |
| sunscreen formula D | 35.42 | 12.44 | 30 | 2.41 | 375 |
| sunscreen formula E | 25.79 | 19.16 | 30 | 1.57 | 379 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tortini, G.; Ziosi, P.; Cesa, E.; Molesini, S.; Baldini, E.; De Lucia, D.; Rossi, C.; Durini, E.; Vertuani, S.; Manfredini, S. Criticisms in the Development of High-Protection and Broad-Spectrum “Natural/Organic” Certifiable Sunscreen. Cosmetics 2022, 9, 56. https://doi.org/10.3390/cosmetics9030056
Tortini G, Ziosi P, Cesa E, Molesini S, Baldini E, De Lucia D, Rossi C, Durini E, Vertuani S, Manfredini S. Criticisms in the Development of High-Protection and Broad-Spectrum “Natural/Organic” Certifiable Sunscreen. Cosmetics. 2022; 9(3):56. https://doi.org/10.3390/cosmetics9030056
Chicago/Turabian StyleTortini, Guido, Paola Ziosi, Elena Cesa, Sonia Molesini, Erika Baldini, Daniela De Lucia, Caterina Rossi, Elisa Durini, Silvia Vertuani, and Stefano Manfredini. 2022. "Criticisms in the Development of High-Protection and Broad-Spectrum “Natural/Organic” Certifiable Sunscreen" Cosmetics 9, no. 3: 56. https://doi.org/10.3390/cosmetics9030056
APA StyleTortini, G., Ziosi, P., Cesa, E., Molesini, S., Baldini, E., De Lucia, D., Rossi, C., Durini, E., Vertuani, S., & Manfredini, S. (2022). Criticisms in the Development of High-Protection and Broad-Spectrum “Natural/Organic” Certifiable Sunscreen. Cosmetics, 9(3), 56. https://doi.org/10.3390/cosmetics9030056









