Phytochemical Analysis and Antioxidant, Antimicrobial, and Antiaging Activities of Ethanolic Seed Extracts of Four Mucuna Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Collecting and Identification of Plant Materials
2.2. Preparation of Mucuna Seed Extracts
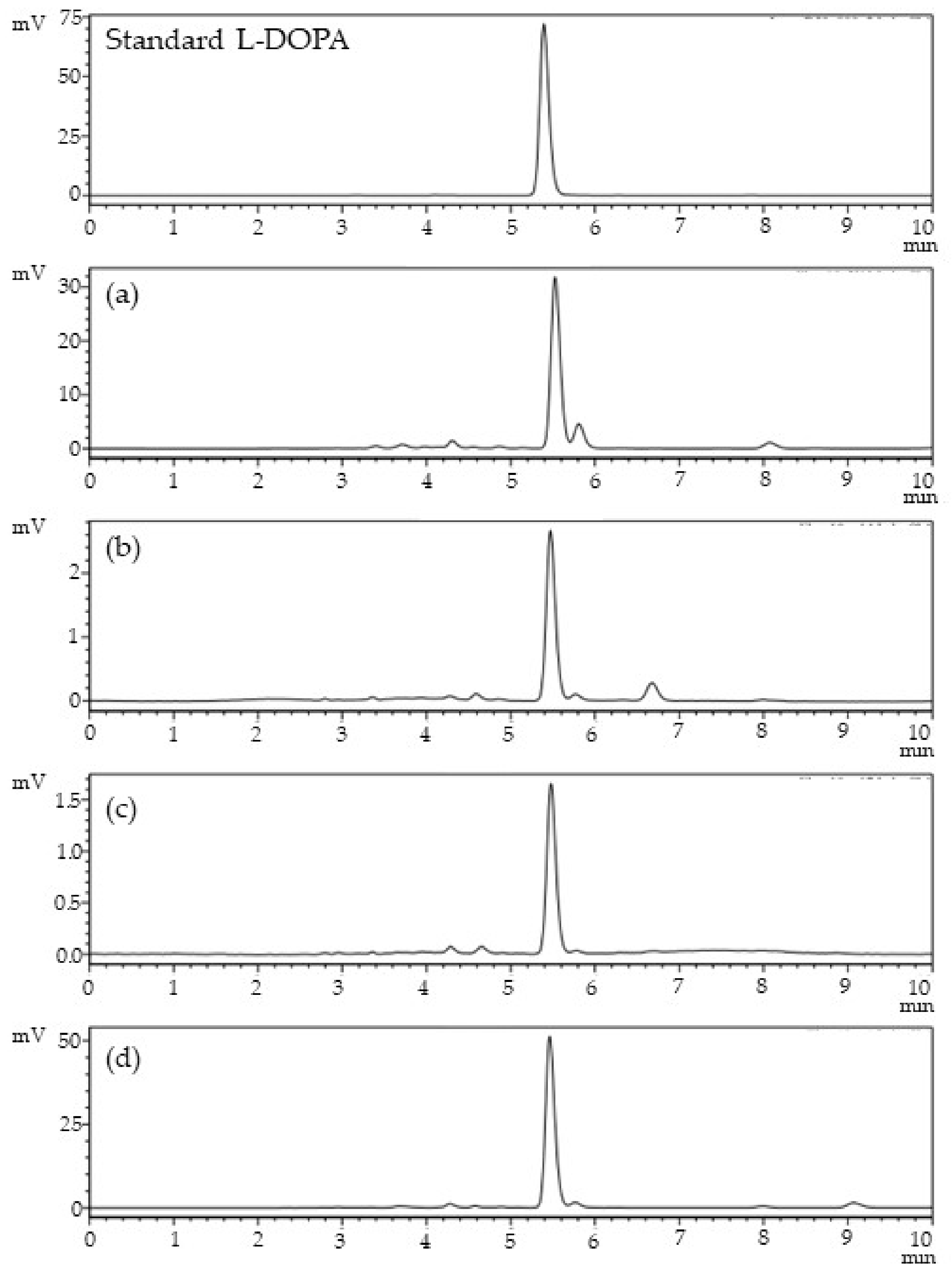
2.3. Determination of L-DOPA Content
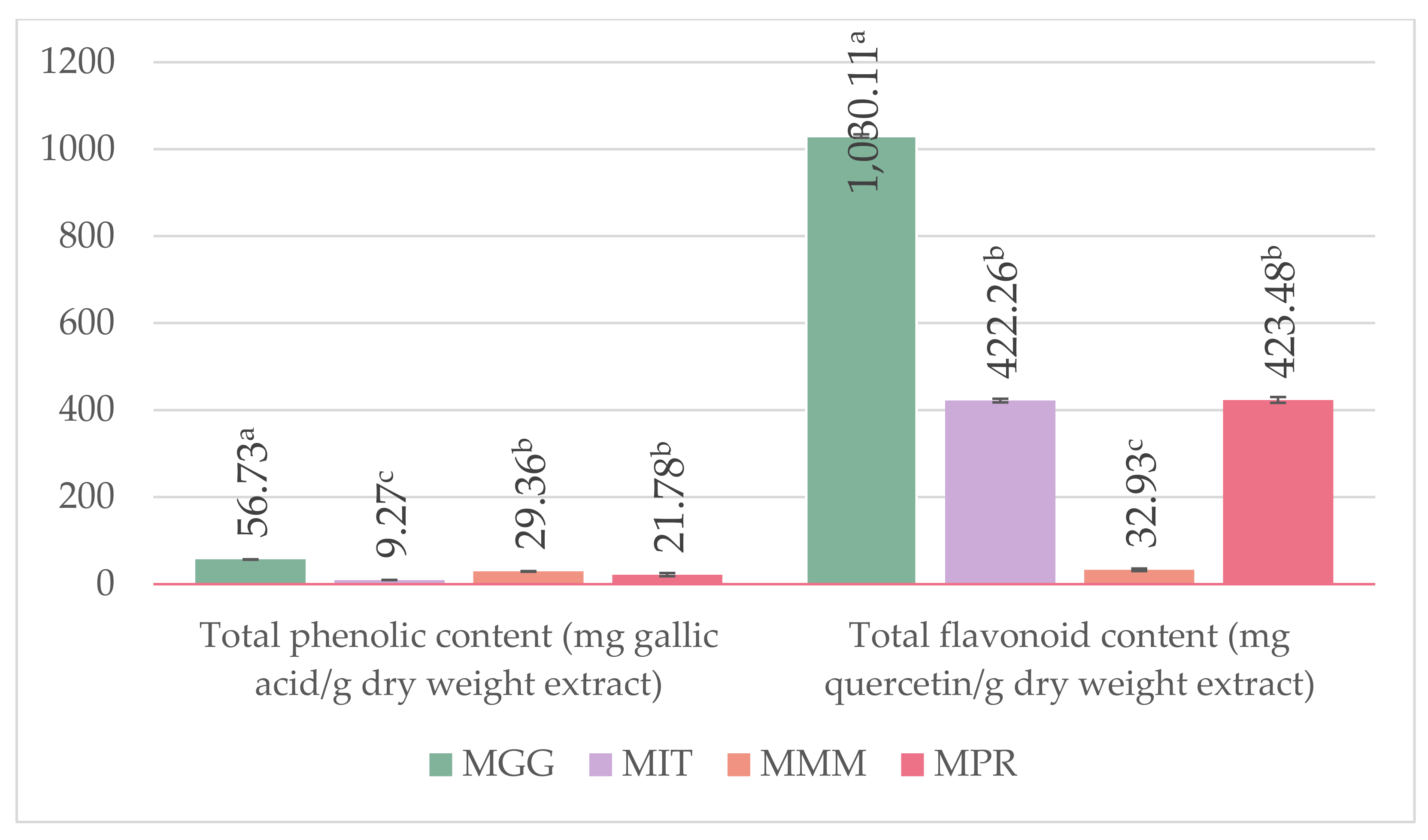
2.4. Determination of Total Phenolic Content
2.5. Determination of Total Flavonoid Content
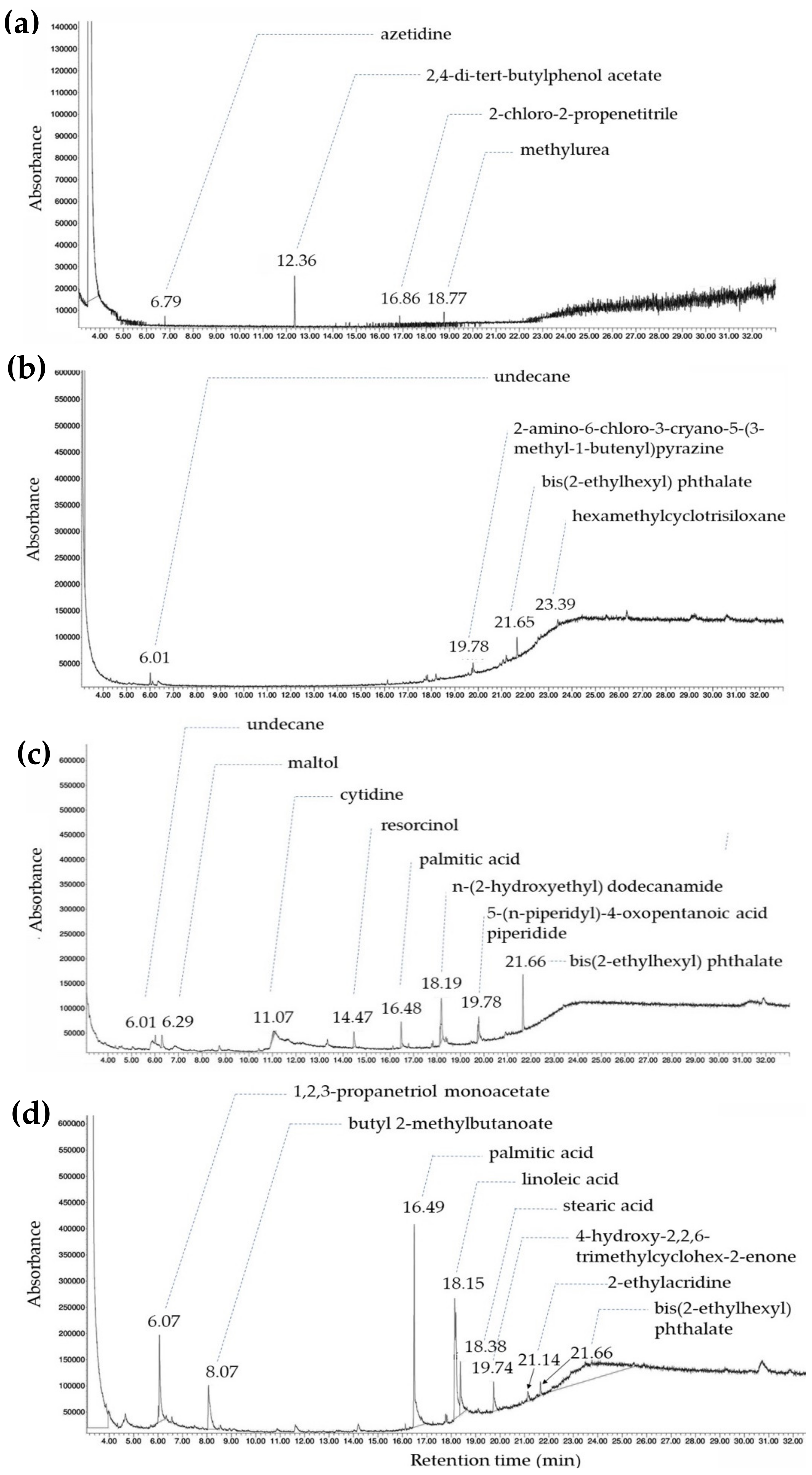
2.6. Gas Chromatography–Mass Spectrometry Analysis
2.7. Antimicrobial Activity Assay
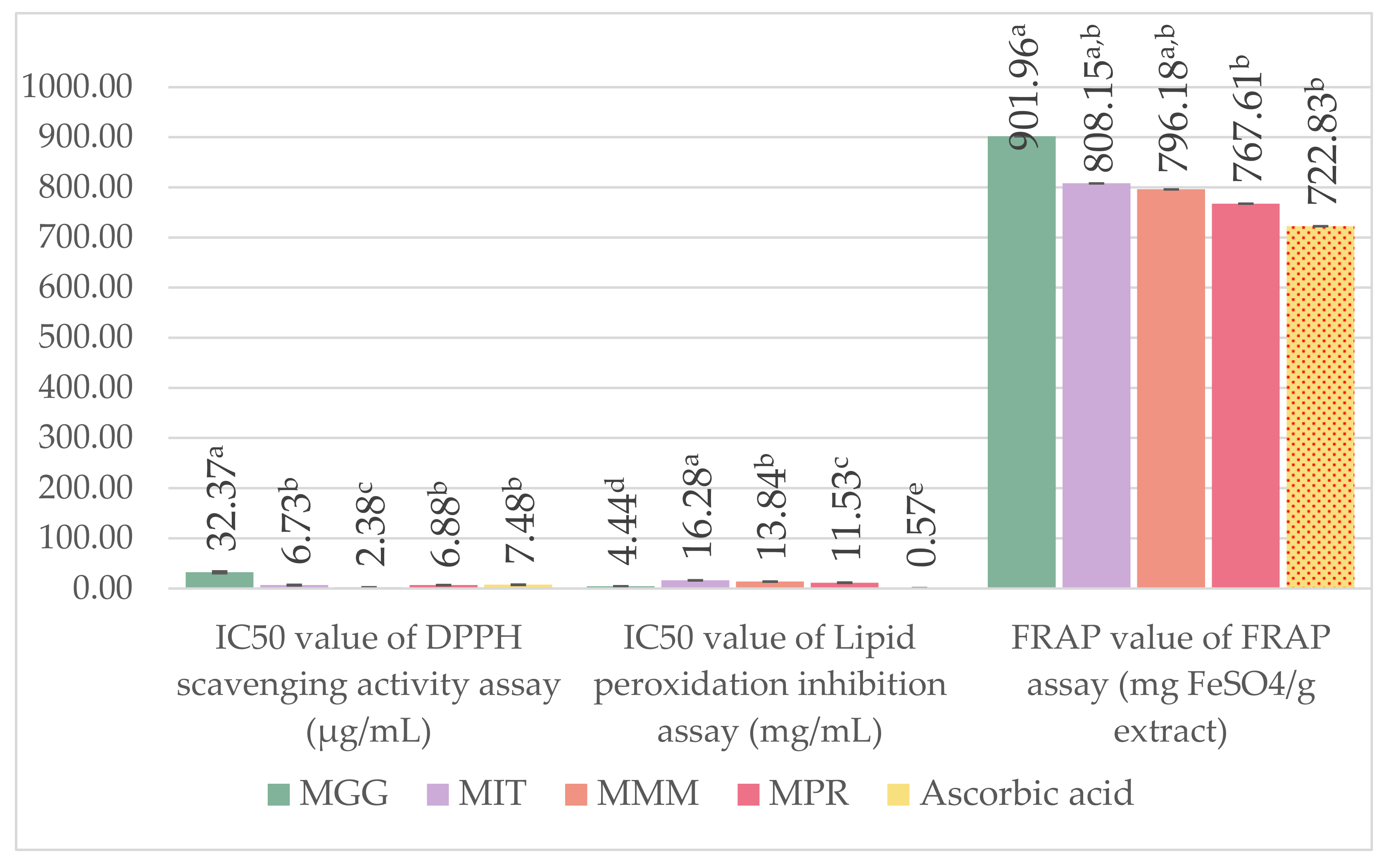
2.8. Antioxidant Activity Assay
2.8.1. 1,1-Diphenyl-2-Picrylhydrazyl (DPPH) Radical Scavenging Assay
2.8.2. Ferric Reducing Antioxidant Power (FRAP) Assay
2.8.3. Lipid Peroxidation Inhibition Assay Using Linoleic Acid Thiocyanate Method
2.9. Hyaluronidase Inhibitory Assay
2.10. Collagenase Inhibitory Activity
2.11. Elastase Inhibitory Activity
2.12. Statistical Analysis
3. Results and Discussions
3.1. Identification of Plant Materials Using a Mini-Barcode, ITS2 Intergenic Region
3.2. Determination of L-DOPA and, Total Phenolic, and Flavonoid Content
3.3. Phytochemical Analysis Using GC–MS
3.4. Skin-Related Antimicrobial Activities
3.5. Antioxidant Activity Assay
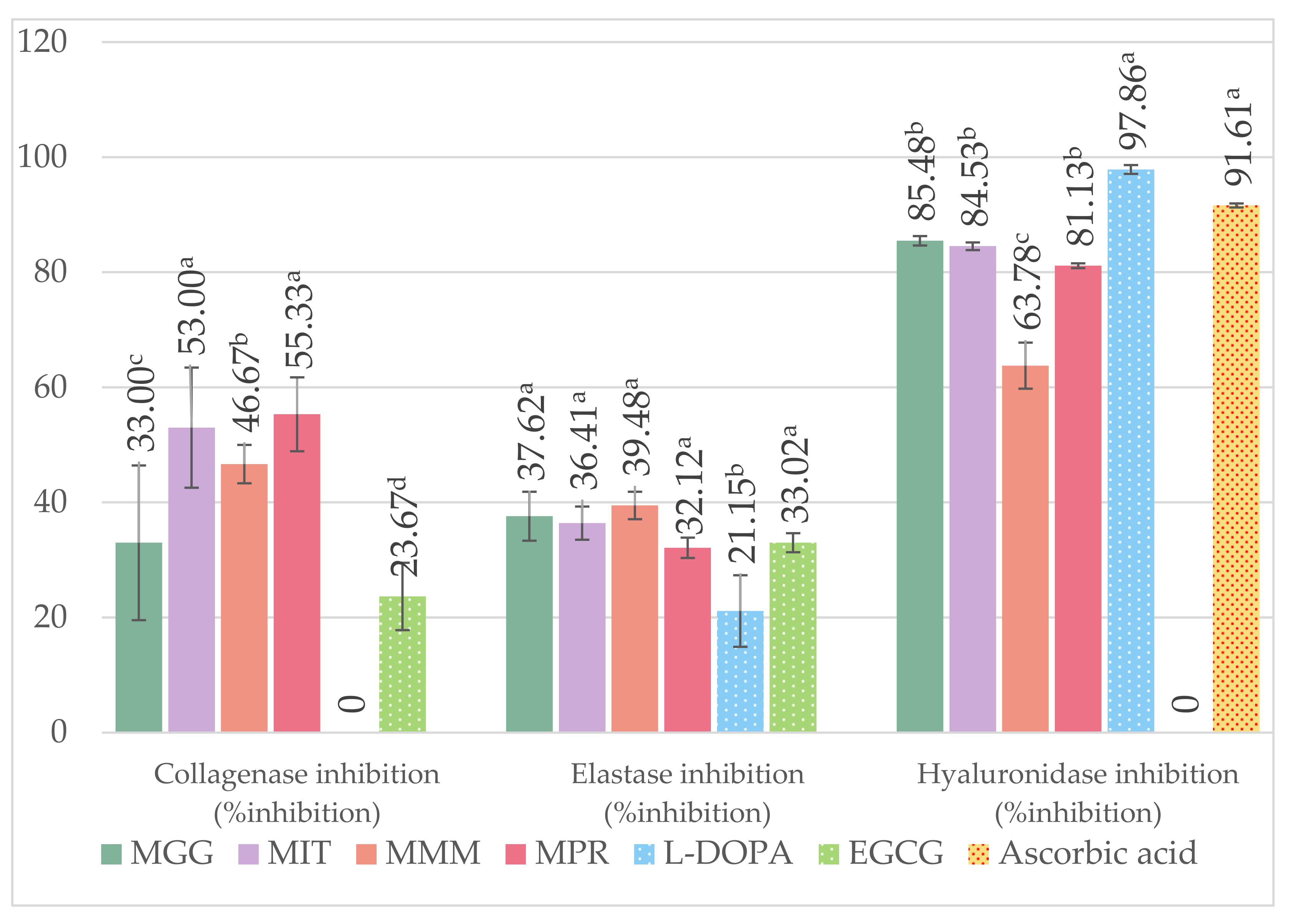
3.6. Anti-Aging Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moura, T.M.; Vatanparas, M.; Tozzi, A.M.G.A.; Forest, F.; Wilmot-Dear, M.; Simon, M.F.; Mansano, V.F.; Kajita, T.; Lewis, G.P. A Molecular Phylogeny and New Infrageneric Classification of Mucuna Adans. (Leguminosae-Papilionoideae) Including Insights from Morphology and Hypothesis about Biogeography. Int. J. Plant Sci. 2016, 177, 76–89. [Google Scholar] [CrossRef]
- Wilmot-Dear, C.M. Mucuna Adams (Leguminosae) in Thailand. Thai. For. Bull. Bot. 2008, 36, 114–139. [Google Scholar]
- Leeratiwong, C.; Jornead, S.; Satthaphorn, J.; Chantaranothai, P. A new record of Mucuna Adans. (Leguminosae) for Thailand. Thai. For. Bull. Bot. 2018, 46, 1–3. [Google Scholar] [CrossRef][Green Version]
- Sathyanarayana, N.; Leelamabika, M.; Mahesh, S.; Jaheer, M. AFLP assessment of genetic diversity among Indian Mucuna accessions. Physiol. Mol. Biol. Plants 2011, 17, 171–180. [Google Scholar] [CrossRef]
- Sungthongwises, K. Diversity of Phosphate Solubilizing Bacteria under Rubber Intercropping. Asian J. Plant Sci. 2016, 15, 75–80. [Google Scholar] [CrossRef]
- Balogun, I.O.; Olatidoye, O.P. Chemical Composition and Nutritional Evaluation of Velvet Bean Seeds (Mucuna utilis) for Domestic Consumption and Industrial Utilization in Nigeria. Pak. J. Nutr. 2012, 11, 116–122. [Google Scholar] [CrossRef]
- Lampariello, L.R.; Cortelazzo, A.; Guerranti, R.; Sticozzi, C.; Valacchi, G. The Magic Velvet Bean of Mucuna pruriens. J. Tradit. Complement. Med. 2012, 2, 331–339. [Google Scholar] [CrossRef]
- Cilia, R.; Laguna, J.; Cassani, E.; Cereda, E.; Pozzi, N.G.; Isaias, I.U.; Contin, M.; Barichella, M.; Pezzoli, G. Mucuna pruriens in Pakinson disease: A double-blind, randomized, controlled, crossover study. Neurology 2017, 89, 432–438. [Google Scholar] [CrossRef]
- Pathak-Gandhi, N.; Vaidya, A.D.B. Management of Parkinson’s disease in Ayurveda: Medicinal plants and adjuvant measures. J. Ethnopharmacol. 2017, 197, 46–51. [Google Scholar] [CrossRef]
- Damodaran, M.; Ramaswamy, R. Isolation of l-3:4-dihydroxyphynylalanine from the seeds of Mucuna pruriens. Biochem. J. 1937, 31, 2149–2152. [Google Scholar] [CrossRef]
- Aware, C.; Patil, R.; Gaikwad, S.; Yadav, S.; Bapat, V.; Jadhav, J. Evaluation of L-dopa, proximate composition with in vitro anti-inflamatory and antioxidant activity of Mucuna macrocarpa beans: A future drug for Parkinson treatment. Asian Pac. J. Trop. Biomed. 2017, 7, 1097–1106. [Google Scholar] [CrossRef]
- Soumyanath, A.; Danne, T.; Hiller, A.; Ramachandran, S.; Shinto, L. Analysis of Levodopa Content in Commercial Mucuna pruriens Products Using High-Performance Liquid Chromatography with Fluorescence Detection. J. Altern. Complement. Med. 2018, 24, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Rene, M.; Suryawanshi, S.; Patil, R.; Aware, C.; Jadhav, R.; Gaikwad, S.; Singh, P.; Yadav, S.; Bapat, V.; Gurav, R.; et al. Exploring the proximate composition, antioxidant, anti-Parkinson’s and anti-inflammatory potential of two neglected and underutilized Mucuna species from India. S. Afr. J. Bot. 2019, 124, 304–310. [Google Scholar] [CrossRef]
- Tavares, R.L.; Silvo, A.S.; Campo, A.R.N.; Schuler, A.R.P.; Aquino, J.S. Nutritional Composition, phytochemicals and microbiological quality of the legume, Mucuna pruriens. Afr. J. Biotechnol. 2015, 14, 676–682. [Google Scholar]
- Das, D.; Das, S.; Pandey, M.; Bhattacharyay, D. In Silico Analysis of Phytochemicals from Mucuna pruriens (L.) DC against Mycobacterium tuberculosis Causing Tuberculosis. EJMP 2020, 31, 19–24. [Google Scholar] [CrossRef]
- Johnson, S.L.; Park, H.Y.; DaSilva, N.A.; Vattem, D.A.; Ma, H.; Seeram, N.P. Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster. Nutrients 2018, 10, 1139. [Google Scholar] [CrossRef]
- Laviada-Castillo, R.E.; Segura-Campos, M.R.; Chan-Zapata, I.; Torres-Romero, J.C.; Guillermo-Cordero, J.L.; Arana-Argáez, V.E. Immunosuppressive effects of protein derivatives from Mucuna pruriens on a streptozotocin-induced type 1 diabetes murine model. J. Food Biochem. 2009, 43, e12834. [Google Scholar] [CrossRef]
- Jimoh, M.A.; Idris, O.A.; Jimoh, M.O. Cytotoxicity, Phytochemical, Antiparasitic Screening, and Antioxidant Activities of Mucuna pruriens (Fabaceae). Plants 2020, 9, 1249. [Google Scholar] [CrossRef]
- Dhanani, T.; Singh, R.; Shah, S.; Kumari, P.; Kumar, S. Comparison of green extraction methods with conventional extraction method for extract yield, L-DOPA concentration and antioxidant activity of Mucuna pruriens seed. Green Chem. Lett. Rev. 2015, 8, 43–48. [Google Scholar] [CrossRef]
- Shanmugavel, G.; Krishnamoorthy, G. In vitro evaluation of the antibacterial activity of alcoholic extract from Mucuna pruriens seed. Int. J. Herb. Med. 2015, 2, 7–9. [Google Scholar]
- Mutwedu, V.B.; Ayagirwe, R.B.B.; Mwema, L.M.; Butseme, S.; Kashosi, T.; Mitima, B.; Manyawu, G.J.; Nyongesa, A.W. Effect of dietary inclusion of small quantities of Mucuna pruriens seed meal on sexual behavior, semen characteristics in rabbit bucks (Oryctolagus curiculus). Trop. Anim. Health Prod. 2019, 51, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Ashidi, J.S.; Owagboriaye, F.O.; Yaya, F.B.; Payne, D.E.; Lawal, O.I.; Owa, S.O. Assessment of reproductive function in male albino rat fed dietary meal supplemented with Mucuna pruriens seed powder. Heliyon 2019, 5, e02716. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Vukmanovic-Stejic, M. Skin barrier immunity and ageing. Immunology 2019, 160, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Duan, E. Fighting against skin aging: The way from bench to bedside. Cell Transplant. 2018, 27, 729–738. [Google Scholar] [CrossRef]
- Parrado, C.; Mercado-Saenz, S.; Perez-Davo, A.; Gilaberte, Y.; Gonzalez, S.; Juarranz, A. Environmental stressors on skin aging mechanistic insights. Front. Pharmacol. 2019, 10, 759. [Google Scholar] [CrossRef]
- Tobin, D.J. Introduction to skin aging. J. Tissue Viability 2017, 26, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.S.; Dreesen, O. Biomarkers of cellular senescence and skin aging. Front. Genet. 2018, 9, 247–260. [Google Scholar] [CrossRef]
- Shin, J.-W.; Kwon, S.-H.; Choi, J.-Y.; Na, J.-I.; Huh, C.-H.; Choi, H.-R.; Park, K.-C. Molecular mechanisms of dermal aging and antiaging approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef]
- Baumann, L. Skin ageing and its treatment. J. Pathol. 2007, 211, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Toole, B.P. Hyaluronan: From extracellular glue to pericellular cue. Nat. Rev. Cancer 2004, 4, 528–539. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Derm.-Endocrinol. 2012, 4, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yao, H.; Han, J.; Liu, C.; Song, J.; Shi, L.; Zhu, Y.; Ma, X.; Gao, T.; Pang, X.; et al. Validation of the ITS2 Region as a Novel DNA Barcode for Identifying Medicinal Plant Species. PLoS ONE 2010, 5, e8613. [Google Scholar] [CrossRef] [PubMed]
- Sheidai, M.; Tabaripour, R.; Talebi, S.M.; Noormohammadi, Z.; Koohdar, F. Adulteration in medicinally important plant species of Ziziphora in Iran market: DNA barcoding approach. Ind. Crops Prod. 2019, 130, 627–633. [Google Scholar] [CrossRef]
- Poomanee, W.; Wattananapakasem, I.; Panjan, W.; Kiattisin, K. Optimizing anthocyanins extraction and the effect of cold plasma treatment on the anti-aging potential of purple glutinous rice (Oryza sativa L.) extract. Cereal Chem. 2021, 98, 571–582. [Google Scholar] [CrossRef]
- Zargoosh, Z.; Ghavam, M.; Bacchetta, G.; Tavili, A. Effects of ecological factors on the antioxidant potential and total phenol content of Scrophularia stricta Boiss. Sci. Rep. 2019, 9, 16021. [Google Scholar] [CrossRef]
- Yaraksa, N.; Anunthawan, T.; Theansungnoen, T.; Daduang, S.; Araki, T.; Dhiravisit, A.; Thammasirirak, S. Design and synthesis of cationic antibacterial peptide based on Leucrocin I sequence, antibacterial peptide from crocodile (Crocodylus siamensis) white blood cell extracts. J. Antibiot. 2014, 67, 205–212. [Google Scholar] [CrossRef]
- Theansungnoen, T.; Yaraksa, N.; Daduang, S.; Dhiravisit, A.; Thammasirirak, S. Purification and characterization of antioxidant peptides from leukocyte extract of Crocodylus siamensis. Protein J. 2014, 33, 24–31. [Google Scholar] [CrossRef]
- Nitthikan, N.; Leelapornpisid, P.; Natakankitkul, S.; Chaiyana, W.; Mueller, M.; Viernstein, H.; Kiattisin, K. Improvement of stability and transdermal delivery of bioactive compounds in green robusta coffee beans extract loaded nanostructured lipid carriers. J. Nanotechnol. 2018, 2018, 7865024. [Google Scholar] [CrossRef]
- Widowati, W.; Rani, A.P.; Hamzah, R.A.; Arumwardana, S.; Afifah, E.; Kusuma, H.S.W.; Rihibiha, D.D.; Nufus, H.; Amalia, A. Antioxidant and antiaging assays of Hibiscus sabdariffa extract and its compounds. Nat. Prod. Sci. 2017, 23, 192–200. [Google Scholar] [CrossRef]
- Thring, T.S.A.; Hili, P.; Naughton, D.P. Anti-collagenase, anti-elastase and anti-oxidant activities of extracts from 21 plants. BMC Complement. Med. Ther. 2009, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Drouet, S.; Garros, L.; Hano, C.; Tungmunnithum, D.; Renouard, S.; Hagège, D.; Maunit, B.; Laine, È. A Critical View of Different Botanical, Molecular, and Chemical Techniques Used in Authentication of Plant Materials for Cosmetic Applications. Cosmetics 2018, 5, 30. [Google Scholar] [CrossRef]
- Narayana, D.B.; Johnson, S.T. DNA barcoding in authentication of herbal raw materials, extracts and dietary supplements: A perspective. Plant Biotechnol. Rep. 2019, 13, 201–210. [Google Scholar] [CrossRef]
- Moorhouse-Gann, R.J.; Dunn, J.C.; de Vere, N.; Goder, M.; Cole, N.; Hipperson, H.; Symondson, W.O.C. New universal ITS2 primers for high-resolution herbivory analyses using DNA metabarcoding in both tropical and temperate zones. Sci. Rep. 2018, 8, 8542. [Google Scholar] [CrossRef]
- Rashmi, R.V.; Sathyanarayana, N.; Vidya, S.M. Validation of DNA barcoding markers in common Mucuna species of India for taxonomy and pharmacognosy applications. Plant Gene 2017, 12, 98–104. [Google Scholar]
- Demidchik, V. Mechanisms of oxidative stress in plants: From classical chemistry to cell biology. Environ. Exp. Bot. 2015, 109, 212–228. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Chelliah, R.; Hu, X.; Oh, D.H.; Kathiresan, K.; Wang, M.H. Antioxidant, Anti-Lung Cancer, and Anti-Bacterial Activities of Toxicodendron vernicifluum. Biomolecules 2019, 9, 127. [Google Scholar] [CrossRef]
- Wang, J.; Liu, H.; Gao, H.; Zhao, J.; Zhao, L.; Han, J.; Yu, Z.; Yang, F. Antimicrobial and antioxidant activities of the flower essential oil of Halimodendron halodendron. Nat. Prod. Commun. 2011, 6, 1749–1753. [Google Scholar] [CrossRef]
- Ahmad, S.; Ullah, F.; Sadiq, A.; Ayaz, M.; Imran, M.; Ali, I.; Zeb, A.; Ullah, F.; Shah, M.R. Chemical composition, antioxidant and anticholinesterase potentials of essential oil of Rumex hastatus D. Don collected from the North West of Pakistan. BMC Complement Altern. Med. 2016, 16, 29. [Google Scholar] [CrossRef]
- Babaiwa, U.F.; Erharuyi, O.; Falodun, A.; Akerele, J.O. Antimicrobial activity of ethyl acetate extract of Citrullus lanatus seeds. Trop. J. Pharm. Res. 2017, 16, 1631–1636. [Google Scholar] [CrossRef]
- Yang, J.F.; Liang, M.T.; Yang, C.H.; Gao, Z.J.; Wu, Y.W.; Chuang, L.Y. Chromatographic-mass spectrometric analysis of ethanol extract of Maesa perlaria var. formosana. Trop. J. Pharm. Res. 2016, 15, 1025–1029. [Google Scholar] [CrossRef]
- Butler, C.R.; Beck, E.M.; Harris, A.; Huang, Z.; McAllister, L.A.; am Ende, C.W.; Fennell, K.; Foley, T.L.; Fonseca, K.; Hawrylik, S.J.; et al. Azetidine and Piperidine Carbamates as Efficient, Covalent Inhibitors of Monoacylglycerol Lipase. J. Med. Chem. 2017, 60, 9860–9873. [Google Scholar] [CrossRef] [PubMed]
- Manjuntha, B.K.; Patil, H.S.R.; Vidya, S.M.; Kekuda, T.R.P.; Mukunda, S.; Davikar, R. Studies on the antibacterial activity of Mucuna monosperma. Indian Drugs 2006, 43, 150–152. [Google Scholar]
- Natarajan, K.; Narayanan, N.; Ravichandran, N. Pharmacognostical characterization, phytochemical and antimicrobial evaluation of Mucuna cochinchinensis (lour.) Cheval seeds. J. Pharm. Res. 2013, 6, 318–323. [Google Scholar] [CrossRef]
- Longhi, J.G.; Perez, E.; de Lima, J.J.; Cândido, L.M.B. In vitro evaluation of Mucuna pruriens (L.) DC. antioxidant activity. Braz. J. Pharm. Sci. 2011, 47, 535–544. [Google Scholar] [CrossRef]
- Dhanasakaran, M.; Tharakan, B.; Manyam, B.V. Antiparkinson drug—Mucuna pruriens shows antioxidant and metal chelating activity. Phytother. Res. 2008, 22, 6–11. [Google Scholar] [CrossRef]
- Lefèvre-Utile, A.; Braun, C.; Haftek, M.; Aubin, F. Five functional aspects of the epidermal barrier. Int. J. Mol. Sci. 2021, 22, 11676. [Google Scholar] [CrossRef]
- Félix, R.; Valentão, P.; Andrade, P.B.; Félix, C.; Novais, S.C.; Lemos, M.F. Evaluating the in vitro potential of natural extracts to protect lipids from oxidative damage. Antioxidants 2020, 9, 231. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkarm, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef]
- Fulop, T.; Khalil, A.; Larbi, A. The role of elastin peptides in modulating the immune response in aging and age-related diseases. Pathol. Biol. 2012, 60, 28–33. [Google Scholar] [CrossRef]
- Bravo, K.; Alzate, F.; Osorio, E. Fruits of selected wild and cultivated Andean plants as sources of potential compounds with antioxidant and anti-aging activity. Ind. Crops Prod. 2016, 85, 341–352. [Google Scholar] [CrossRef]
- Pathania, R.; Chawla, P.; Khan, H.; Kaushik, R.; Khan, M.A. An assessment of potential nutritive and medicinal properties of Mucuna pruriens: A natural food legume. Biotech 2020, 10, 261. [Google Scholar] [CrossRef] [PubMed]
- Santhosh, M.S.; Hemshekhar, M.; Sunitha, K.; Thushara, R.M.; Jnaneshwari, S.; Kemparaju, K.; Girish, K.S. Snake venom induced local toxicities: Plant secondary metabolites as an auxiliary therapy. Mini-Rev. Med. Chem. 2013, 13, 106–123. [Google Scholar] [CrossRef] [PubMed]

| Sample Codes | Expected Species | Collected Dates | Localities (District, Province) |
|---|---|---|---|
| MGG | M.gigantea (Willd.) DC. | 24 November 2017 | Khuan Niang, Songkhla |
| MIT | M.interrupta Gagnep. | 10 June 2017 | Mae Sariang, Mae Hong Son |
| MMM | M.monosperma Wight | 24 May 2017 | Mae Sot, Tak |
| MPR | M. pruriens (L.) DC. | 3 September 2017 | Ban Na Doem, Surat Thani |
| Sample Codes | Species of BLAST Results | Accession Number | Max Score | Percent Identity (%) |
|---|---|---|---|---|
| MGG | M.gigantea | LC494611 | 612 | 99.11 |
| MIT | M.interrupta | HM355841 | 675 | 98.94 |
| MMM | M.monosperma | AB775135 | 625 | 98.06 |
| MPR | M. pruriens | KX057890 | 730 | 99.50 |
| Extracts | C. acnes DMST 14916 | S. aureus TISTR 746 | S. epidermidis TISTR 2141 | C. albicans TISTR 5239 | ||||
|---|---|---|---|---|---|---|---|---|
| MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | MIC (mg/mL) | MFC (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | |
| MGG | Nd. | Nd. | Nd. | Nd. | Nd. | Nd. | Nd. | Nd. |
| MIT | Nd. | Nd. | Nd. | Nd. | Nd. | Nd. | 8.20 | 16.40 |
| MMM | Nd. | Nd. | 8.20 | 16.40 | 8.20 | 16.40 | 4.10 | 8.20 |
| MPR | Nd. | Nd. | 4.10 | 4.10 | Nd. | Nd. | Nd. | Nd. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theansungnoen, T.; Nitthikan, N.; Wilai, M.; Chaiwut, P.; Kiattisin, K.; Intharuksa, A. Phytochemical Analysis and Antioxidant, Antimicrobial, and Antiaging Activities of Ethanolic Seed Extracts of Four Mucuna Species. Cosmetics 2022, 9, 14. https://doi.org/10.3390/cosmetics9010014
Theansungnoen T, Nitthikan N, Wilai M, Chaiwut P, Kiattisin K, Intharuksa A. Phytochemical Analysis and Antioxidant, Antimicrobial, and Antiaging Activities of Ethanolic Seed Extracts of Four Mucuna Species. Cosmetics. 2022; 9(1):14. https://doi.org/10.3390/cosmetics9010014
Chicago/Turabian StyleTheansungnoen, Tinnakorn, Nichcha Nitthikan, Mayuramas Wilai, Phanuphong Chaiwut, Kanokwan Kiattisin, and Aekkhaluck Intharuksa. 2022. "Phytochemical Analysis and Antioxidant, Antimicrobial, and Antiaging Activities of Ethanolic Seed Extracts of Four Mucuna Species" Cosmetics 9, no. 1: 14. https://doi.org/10.3390/cosmetics9010014
APA StyleTheansungnoen, T., Nitthikan, N., Wilai, M., Chaiwut, P., Kiattisin, K., & Intharuksa, A. (2022). Phytochemical Analysis and Antioxidant, Antimicrobial, and Antiaging Activities of Ethanolic Seed Extracts of Four Mucuna Species. Cosmetics, 9(1), 14. https://doi.org/10.3390/cosmetics9010014







