New Antioxidant Ingredients from Brewery By-Products for Cosmetic Formulations
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Sample Preparation
2.3. Total Phenol Content Determination
2.4. Evaluation of the Antioxidant Activity
2.5. Evaluation of the Trolox Equivalent Antioxidant Capacity (DPPH)
2.6. Radical Cation Scavenging Activity and Reducing Power (ABTS)
2.7. Ferric-Ion-Reducing Antioxidant Parameter (FRAP)
- 50 mL 0.3M acetate buffer pH 3.6 (1.23 g of sodium acetate in 50 mL of water acidifying with acetic acid);
- 5 mL of stock solution of 5 mM TPTZ (2,4,6-Tripyridyl-s-triazine) (15.6 mg) in 40 mM HCl;
- 5 mL of 5 mM FeCl3·6 H2O (16.2 mg) in 40 mM HCl.
2.8. Cell Cultures
2.9. Cytotoxicity and Mitochondrial Activity
2.10. Intracellular ROS Formation
2.11. Statistical Analysis
3. Results and Discussion
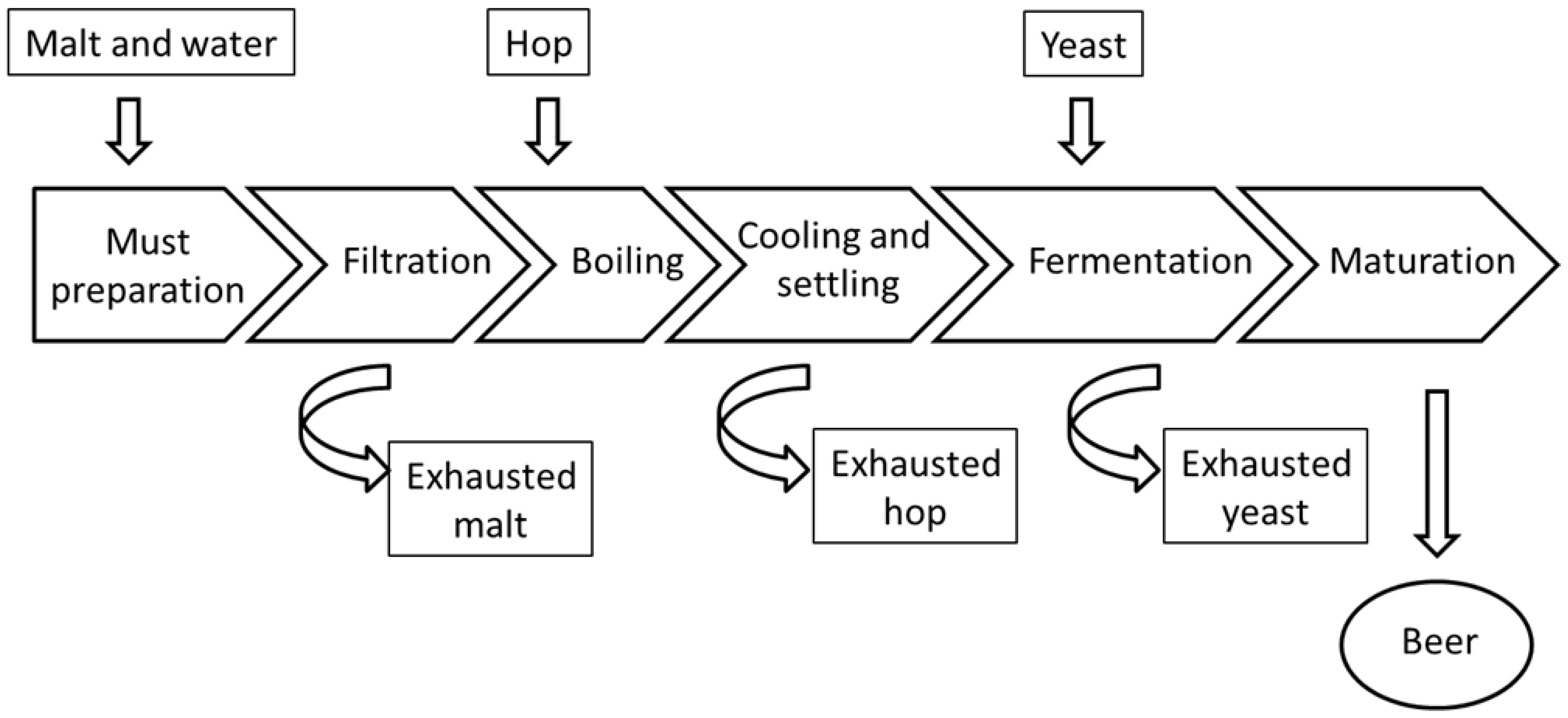
3.1. Brewing Process of Craft Beers under Study
3.2. Determination of Total Phenol Content
3.3. Evaluation of the Antioxidant Activities
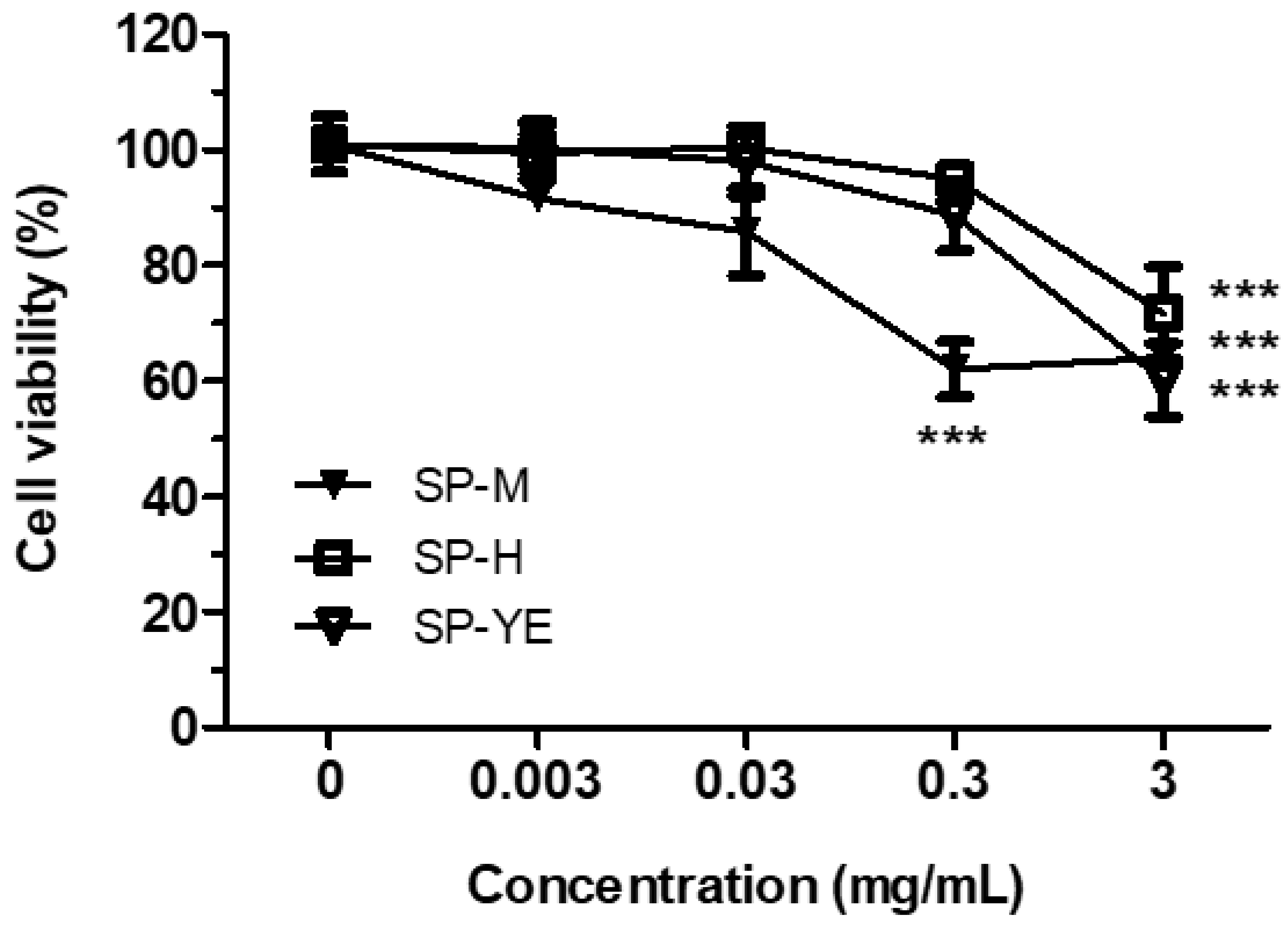
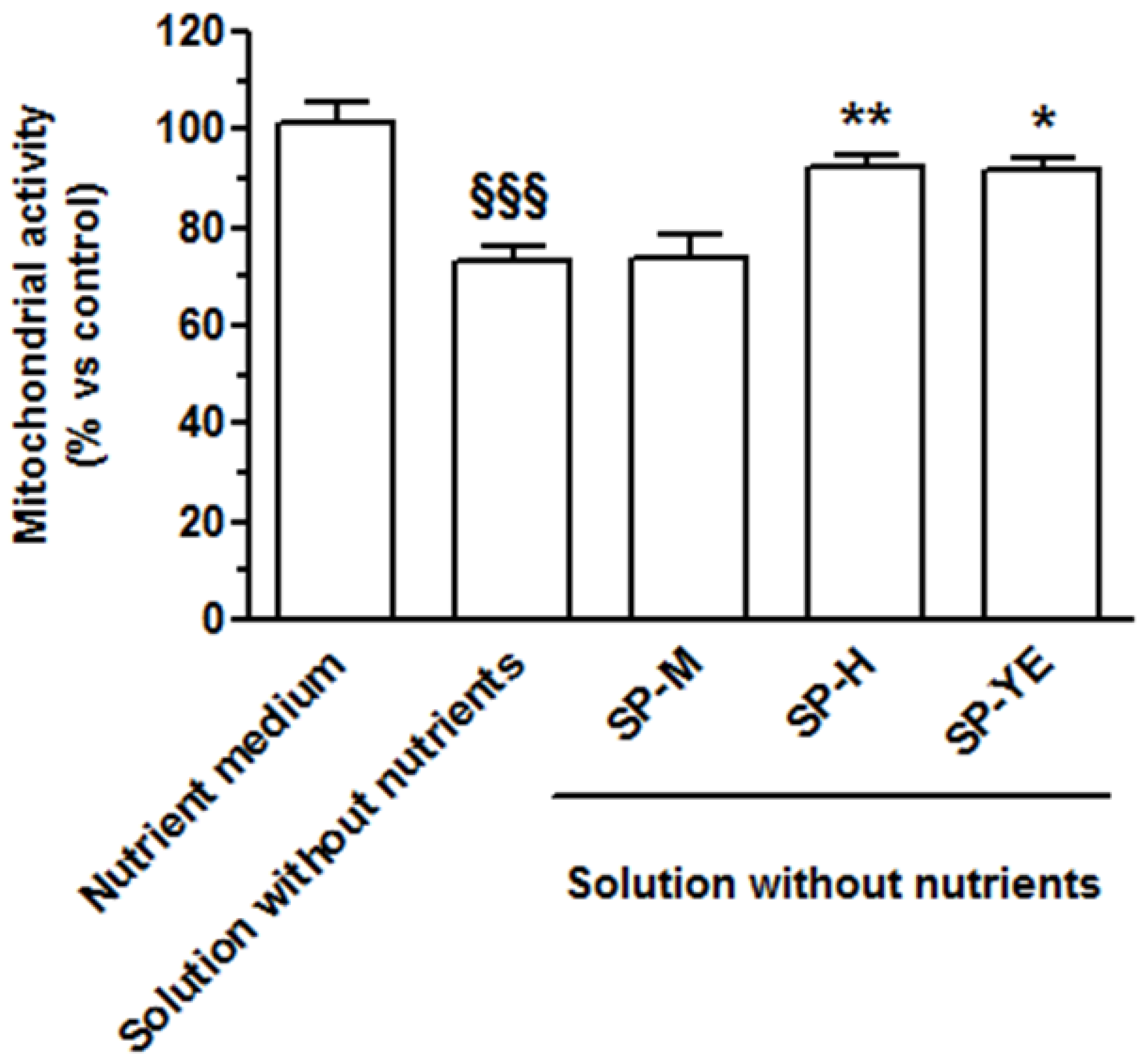
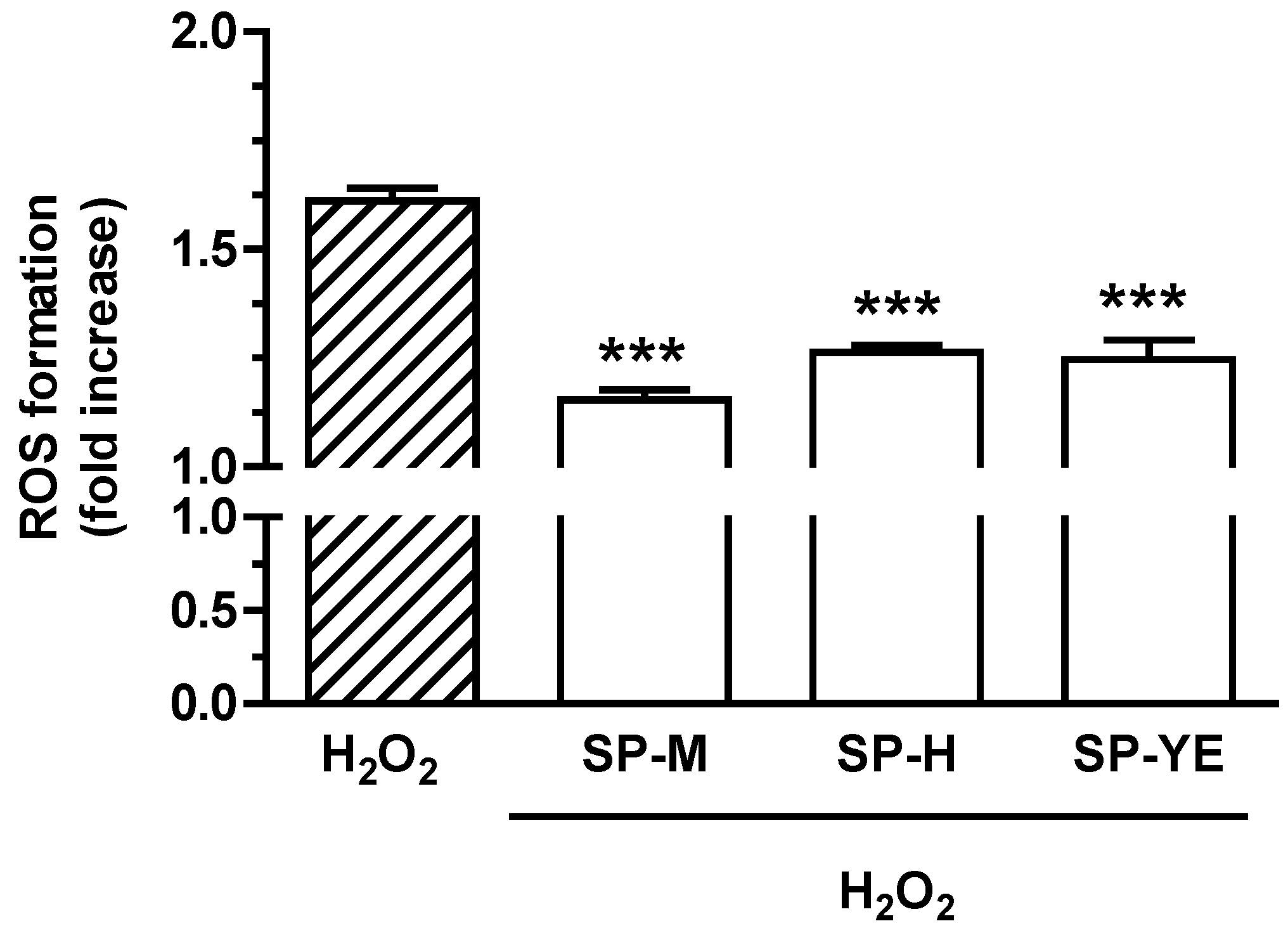
3.4. Bioactivity of Spent Extracts in Human Keratinocytes
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ascher, B. Global Beer: The Road To Monopoly; American Antititrust Institute: Washington, DC, USA, 2012; Available online: https://www.antitrustinstitute.org/work-product/global-beer-the-road-to-monopoly/ (accessed on 14 November 2012).
- Aquilani, B.; Laureti, T.; Poponi, S.; Secondi, L. Beer choice and consuption determinants when craft beers are tasted: An exploratory study of consumer preferences. Food Qual. Prefer. 2015, 41, 214–224. [Google Scholar] [CrossRef]
- Berkhout, B.; Bertling, L.; Bleeker, Y.; de Wit, W.; Kruis, G.; Stokkel, R.; Theuws, R. The Contributionmade by Beer to the European Economy; Ernst & Young: Amsterdam, The Netherlands, 2013; pp. 1–290. [Google Scholar]
- Sohrabvandi, S.; Mortazavian, A.M.; Rezaei, K. Health-related aspects of beer: A review. Int. J. Food Prop. 2012, 15, 350–373. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, W.; Lu, J.; Zhao, M. Phenolic profiles and antioxidant activities of commercial beers. Food Chem. 2010, 119, 1150–1158. [Google Scholar] [CrossRef]
- Ribeiro Tafulo, P.A.; Barbosa Queirós, R.; Delerue-Matos, C.M.; Sales, M.G.F. Control and comparison of the antioxidant capacity of beers. Food Res. Int. 2010, 43, 1702–1709. [Google Scholar] [CrossRef] [Green Version]
- Piazzon, A.; Forte, M.; Nardini, M. Characterization of phenolics content and antioxidant activity of different beer types. J. Agric. Food Chem. 2010, 58, 10677–10683. [Google Scholar] [CrossRef] [PubMed]
- Fărcaş, A.; Tofană, M.; Socaci, S.; Scrob, S.; Salanţă, L.; Borşa, A. Preliminary Study on Antioxidant Activity and Polyphenols Content in Discharged Waste from Beer Production. J. Agroaliment. Process. Technol. 2013, 19, 319–324. [Google Scholar]
- Zhao, H.; Fan, W.; Dong, J.; Lu, J.; Chen, J.; Shan, L.; Lin, Y.; Kong, W. Evaluation of antioxidant activities and total phenolic contents of typical malting barley varieties. Food Chem. 2008, 107, 296–304. [Google Scholar] [CrossRef]
- Quifer-Rada, P.; Vallverdu-Queralt, A.; Martinez-Huelamo, M.; Chiva-Blanch, G.; Jauregui, O.; Estruch, R.; Lamuela-Raventos, R. A comprehensive characterisation of beer polyphenols by high resolution mass spectrometry (LC-ESI-LTQ-Orbitrap-MS). Food Chem. 2015, 169, 336–343. [Google Scholar] [CrossRef]
- Cheiran, K.P.; Raimundo, V.P.; Manfroi, V.; Anzanello, M.J.; Kahmann, A.; Rodrigues, E.; Frazzon, J. Simultaneous identification of low-molecular weight phenolic and nitrogen compounds in craft beers by HPLC-ESI-MS/MS. Food Chem. 2019, 286, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.; Gigliobianco, M.R.; Peregrina, D.V.; Sagratini, G.; Censi, R.; Di Martino, P. Quantification of phenolic compounds in different types of crafts beers, worts, starting and spent ingredients by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2020, 1612, 460622. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagent. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Zorzetto, C.; Sanchez-Mateo, C.C.; Rabanal, R.M.; Lupidi, G.; Petrelli, D.; Vitali, L.A.; Bramucci, M.; Quassinti, L.; Caprioli, G.; Papa, F.; et al. Phytochemical analysis and in vitro biological activity of three Hypericum species from the Canary Islands (Hypericum reflexum, Hypericum canariense and Hypericum grandifolium). Fitoterapia 2015, 100, 95–109. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Venditti, A.; Bianco, A.; Quassinti, L.; Bramucci, M.; Lupidi, G.; Damiano, S.; Papa, F.; Vittori, S.; Maleci Bini, L.; Giuliani, C.; et al. Phytochemical Analysis, Biological Activity, and Secretory Structures of Stachys annua (L.) L. subsp. annua (Lamiaceae) from Central Italy. Chem. Biodivers. 2015, 12, 1172–1183. [Google Scholar] [CrossRef]
- Moon, J.-Y.; Ngoc, L.T.N.; Chae, M.; Van Tran, V.; Lee, Y.-C. Effects of Microwave-Assisted Opuntia humifusa Extract in Inhibiting the Impacts of Particulate Matter on Human Keratinocyte Skin Cell. Antioxidants 2020, 9, 271. [Google Scholar] [CrossRef] [Green Version]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Ornano, L.; Venditti, A.; Ballero, M.; Sanna, C.; Quassinti, L.; Bramucci, M.; Lupidi, G.; Papa, F.; Vittori, S.; Maggi, F.; et al. Chemopreventive and antioxidant activity of the chamazulene-rich essential oil obtained from Artemisia arborescens L. growing on the Isle of La Maddalena, Sardinia, Italy. Chem. Biodivers. 2013, 10, 1464–1474. [Google Scholar] [CrossRef]
- Gigliobianco, M.R.; Campisi, B.; Vargas Peregrina, D.; Censi, R.; Khamitova, G.; Angeloni, S.; Caprioli, G.; Zannotti, M.; Ferraro, S.; Giovannetti, R.; et al. Optimization of the Extraction from Spent Coffee Grounds Using the Desirability Approach. Antioxidants 2020, 9, 370. [Google Scholar] [CrossRef] [PubMed]
- Ozanne, H.; Toumi, H.; Roubinet, B.; Landemarre, L.; Lespessailles, E.; Daniellou, R.; Cesaro, A. Laminarin Effects, a β-(1,3)-Glucan, on Skin Cell Inflammation and Oxidation. Cosmetics 2020, 7, 66. [Google Scholar] [CrossRef]
- Tarozzi, A.; Marchesl, A.; Hrelia, S.; Angeloni, C.; Andrisano, V.; Fiori, J.; Cantelli-Forti, G.; Hrella, P. Protective Effects of Cyanidin-3-O-β-glucopyranoside Against UVA-induced Oxidative Stress in Human Keratinocytes. Photochem. Photobiol. 2005, 81, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Li, B.B.; Smith, B.; Hossain, M. Extraction of phenolics from citrus peels: I. Solvent extraction method. Sep. Purif. Technol. 2006, 48, 182–188. [Google Scholar] [CrossRef]
- Alothman, M.; Bhat, R.; Karim, A.A. Antioxidant Capacity and Phenolic Content of Selected Tropical Fruits from Malaysia, Extracted with Different Solvents. Food Chem. 2009, 115, 785–788. [Google Scholar] [CrossRef]
- Palma, M.; Pineiro, Z.; Barroso, C.G. Stability of phenolic compounds during extraction with superheated solvents. J. Chromatogr. A 2001, 921, 169–174. [Google Scholar] [CrossRef]
- Plumb, G.W.; Price, K.R.; Williamson, G. Antioxidant properties of flavonol glycosides from tea. Redox. Rep. 1999, 4, 13–16. [Google Scholar] [CrossRef]
- Plumb, G.W.; Price, K.R.; Williamson, G. Antioxidant properties of flavonol glycosides from green beans. Redox. Rep. 1999, 4, 123–127. [Google Scholar] [CrossRef]
- Naidoo, K.; Hanna, R.; Birch-Machin, M.A. What is the role of mitochondrial dysfunction in skin photoaging? Exp. Dermatol. 2018, 27, 124–128. [Google Scholar] [CrossRef] [Green Version]
- Stout, R.; Birch-Machin, M. Mitochondria’s Role in Skin Ageing. Biology 2019, 8, 29. [Google Scholar] [CrossRef] [Green Version]
- Campos, A.M.; Lissi, E.A. Kinetics of the reaction between 2,2-azinobis(3-ethylbenzothiazoline)-6-sulfonic acid (ABTS) derived radical cation and phenols. Int. J. Chem. Kinet. 1996, 29, 219–224. [Google Scholar] [CrossRef]

| Beer | Type of Beer | Beer Alcohol Content (%, V/V) | Raw Materials | Malt Type * | Hops Type * | Yeast | Main Differences |
|---|---|---|---|---|---|---|---|
| EGO | Lager | 6.0 | Water, malt, hops, yeast | Type 1 | Perle + Saaz | Saccharomices Cerevisiae | Lowest malt and hop concentration |
| ALTER | Lager | 6.0 | Water, malt, hops, yeast | Type 1 + Type 2 | Perle + Saaz | Saccharomices Cerevisiae | Lowest malt and hop concentration |
| FIAT LUX | Amber | 7.5 | Water, malt, hops, yeast | Type 1 + Type 3 | Perle + Saaz | Saccharomices Cerevisiae | Intermediate malt and hop concentration |
| TRIPLO MALTO | Triple malt | 8.0 | Water, malt, hops, yeast | Type 1 + Type 4 | Perle + Saaz | Saccharomices Cerevisiae | Highest malt and hop concentration |
| UBI | Red | 9.0 | Water, malt, hops, yeast | Type 1 + Type 3 | Perle + Saaz | Saccharomices Cerevisiae | Intermediate malt and hop concentration |
| MAIOR | Black | 8.0 | Water, malt, hops, yeast | Type 1 + Type 3 + Type 5 | Perle + Saaz | Saccharomices Cerevisiae | Intermediate malt and hop concentration |
| Starting Malt | Wort | Spent Malt | Starting Hop | Wort after Hop | Spent Hop | Starting Yeast | Beer after Yeast | Spent Yeast | Beer | |
|---|---|---|---|---|---|---|---|---|---|---|
| Aspect | Dried solid | Turbid liquid | Humid solid | Dried solid | Turbid liquid | Humid solid | Dried solid | Turbid liquid | Humid solid | Turbid liquid |
| Lyophilisation after receipt | No | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes |
| Milling | Yes | No | Yes | Yes | No | No | Yes | No | No | No |
| Extraction in water at room temperature for 24 h | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Extraction in ethanol at room temperature for 24 h | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Centrifuged | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Lyophilisation after extraction | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Storage at −20 °C in 50 mL polyethylene vials with screw cap | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| FOLIN | Starting Malt | Wort * | Spent malt | Starting Hops: Perle | Starting Hop:Saaz | Starting Hop: Perle and Saaz | Wort after Hops | Spent Hops | Starting Yeast | Beer after Yeast * | Spent Yeast | Beer * |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | mg GAE/g | ||
| Water extracts | ||||||||||||
| Ego | 14.687 ± 4.958 | 7.164 ± 0.476 | 14.102 ± 3.074 | 108.583 ± 24.863 | 93.703 ± 5.135 | 92.506 ± 0.446 | 9.263 ± 8.419 | 15.983 ± 0.594 | 53.844 ± 0.539 | 17.672 ± 1.950 | 38.949 ± 0.983 | 18.961 ± 1.082 |
| Alter | 12.119 ± 1.273 | 9.699 ± 0.488 | 14.286 ± 0.511 | 92.299 ± 0.919 | 11.335 ± 2.234 | 15.833 ± 0.860 | 26.708 ± 2.714 | 36.655 ± 2.412 | 25.493 ± 0.287 | |||
| Fiat lux | 11.672 ± 1.814 | 9.097 ± 0.646 | 9.618 ± 2.888 | 101.318 ± 0.839 | 15.463 ± 3.057 | 13.175 ± 0.711 | 22.950 ± 3.422 | 38.076 ± 3.437 | 23.147 ± 5.996 | |||
| Triplo malto | 15.794 ± 2.528 | 6.899 ± 0.819 | 11.798 ± 0.749 | 104.397 ± 2.911 | 19.063 ± 1.390 | 13.346 ± 0.433 | 35.600 ± 1.474 | 44.521 ± 1.777 | 35.822 ± 0.147 | |||
| Ubi | 14.711 ± 0.891 | 12.785 ± 1.855 | 11.456 ± 0.335 | 96.692 ± 1.944 | 14.001 ± 0.617 | 14.825 ± 3.861 | 21.871 ± 1.242 | 36.404 ± 2.858 | 22.320 ± 0.882 | |||
| Maior | 16.568 ± 2.412 | 12.664 ± 1.391 | 10.444 ± 1.309 | 94.343 ± 2.004 | 20.068 ± 7.339 | 15.476 ± 2.871 | 33.868 ± 8.933 | 31.837 ± 1.347 | 30.927 ± 0.667 | |||
| 70° ethanol extracts | ||||||||||||
| Ego | 28.523 ± 0.697 | 7.164 ± 0.476 | 20.077 ± 2.533 | 155.229 ± 9.768 | 138.247 ± 11.788 | 140.757 ± 3.514 | 9.263 ± 8.419 | 7.941 ± 0.371 | 9.302 ±0.721 | 17.672 ± 1.950 | 22.833 ± 1.309 | 18.961 ± 1.082 |
| Alter | 53.230 ± 9.361 | 9.699 ± 0.488 | 26.463 ± 2.410 | 140.537 ± 1.326 | 11.335 ± 2.234 | 8.947 ± 0.474 | 26.708 ± 2.714 | 18.889 ± 0.575 | 25.493 ± 0.287 | |||
| Fiat lux | 33.382 ± 1.043 | 9.097 ± 0.646 | 14.325 ± 1.242 | 142.929 ± 10.688 | 15.463 ± 3.057 | 7.397 ± 0.433 | 22.950 ± 3.422 | 23.348 ± 4.209 | 23.147 ± 5.996 | |||
| Triplo malto | 28.101 ± 1.052 | 6.899 ± 0.819 | 12.763 ± 0.178 | 145.847 ± 13.759 | 19.063 ± 1.390 | 7.902 ± 1.910 | 35.600 ± 1.474 | 21.623 ± 1.077 | 35.822 ± 0.147 | |||
| Ubi | 31.947 ± 3.696 | 12.785 ± 1.855 | 16.456 ± 0.443 | 143.761 ± 5.302 | 14.001 ± 0.617 | 8.103 ±0.432 | 21.871 ± 1.242 | 19.678 ± 1.028 | 22.320 ± 0.882 | |||
| Maior | 72.143 ± 1.866 | 12.664 ± 1.391 | 37.569 ± 1.730 | 143.668 ± 4.229 | 20.068 ± 7.339 | 8.723 ±0.572 | 33.868 ± 8.933 | 14.274 ± 1.507 | 30.927 ± 0.667 | |||
| DPPH | Starting Malt | Wort * | Spent Malt | Starting Hops: Perle | Starting Hops: Saaz | Starting Hops: Perle and Saaz | Wort after Hops | Spent Hops | Starting Yeast | Beer after Yeast | Spent Yeast | Beer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | |
| Water extracts | ||||||||||||
| Ego | 12.281 ± 4.497 | 2.549 ± 0.191 | 3.212 ± 0.212 | 89.243 ± 17.157 | 72.382 ± 10.257 | 72.467 ± 11.425 | 4.233 ± 1.123 | 3.114 ± 0.112 | 9.191 ± 0.001 | 5.324 ± 0.278 | 18.991 ± 1.009 | 5.544 ± 0.324 |
| Alter | 23.982 ± 5.624 | 1.946 ± 0.048 | 4.215 ± 0.025 | 73.152 ± 2.655 | 3.544 ± 1.022 | 2.645 ± 0.692 | 5.466 ± 0.255 | 12.757 ± 3.429 | 5.268 ± 1.277 | |||
| Fiat lux | 21.98 ± 2.785 | 1.822 ± 0.085 | 2.528 ± 0.068 | 75.681 ± 14.134 | 4.387 ± 1.850 | 3.704 ± 0.071 | 6.124 ± 1.243 | 17.991 ± 1.639 | 6.320 ± 1.123 | |||
| Triplo malto | 9.527 ± 1.770 | 8.503 ± 5.179 | 2.433 ± 0.063 | 75.977 ± 4.072 | 12.798 ± 2.150 | 3.604 ± 0.125 | 15.177 ± 1.178 | 28.593 ± 1.257 | 14.785 ± 2.235 | |||
| Ubi | 21.647 ± 1.173 | 5.451 ± 0.277 | 2.507 ± 0.123 | 77.485 ± 16.279 | 10.143 ± 1.244 | 3.381 ± 0.142 | 13.043 ± 0.124 | 20.125 ± 2.674 | 14.244 ± 1.466 | |||
| Maior | 20.647 ± 1.167 | 4.480 ± 1.725 | 3.449 ± 0.055 | 75.739 ± 5.987 | 8.527 ± 2.326 | 3.313 ± 0.052 | 10.078 ± 1.255 | 23.459 ± 3.242 | 9.127 ± 0.675 | |||
| 70° ethanol extracts | ||||||||||||
| Ego | 22.339 ± 0.256 | 2.549 ± 0.191 | 9.036 ± 0.191 | 354.182 ± 78.223 | 258.252 ± 27.154 | 327.241 ± 77.993 | 4.233 ± 1.123 | 7.579 ± 0.436 | 7.483 ± 0.707 | 5.324 ± 0.278 | 32.989 ± 5.936 | 5.544 ± 0.324 |
| Alter | 20.379 ± 0.234 | 1.946 ± 0.048 | 7.022 ± 1.241 | 309.025 ± 66.471 | 3.544 ± 1.022 | 6.949 ± 0.586 | 5.466 ± 0.255 | 28.988 ± 3.934 | 5.268 ± 1.277 | |||
| Fiat lux | 39.954 ± 0.145 | 1.822 ± 0.085 | 9.174 ± 1.347 | 341.825 ± 36.986 | 4.387 ± 1.850 | 6.149 ± 0.252 | 6.124 ± 1.243 | 30.263 ± 16.218 | 6.320 ± 1.123 | |||
| Triplo malto | 22.339 ± 0.067 | 8.503 ± 5.179 | 8.124 ± 1.256 | 283.059 ± 31.749 | 12.798 ± 2.150 | 8.055 ± 1.822 | 15.177 ± 1.178 | 32.026 ± 18.567 | 14.785 ± 2.235 | |||
| Ubi | 40.765 ± 0.145 | 5.451 ± 0.277 | 9.756 ± 1.266 | 270.852 ± 25.782 | 10.143 ± 1.244 | 9.412 ± 0.142 | 13.043 ± 0.124 | 58.675 ± 11.566 | 14.244 ± 1.466 | |||
| Maior | 42.387 ± 0.893 | 4.480 ± 1.725 | 10.244 ± 1.345 | 260.854 ± 36.245 | 8.527 ± 2.326 | 6.759 ± 0.227 | 10.078 ± 1.255 | 19.799 ± 2.453 | 9.127 ± 0.675 | |||
| ABTS | Starting Malt | Wort | Spent Malt | Starting Hops: Perle | Starting Hops: Saaz | Starting Hops: Perle and Saaz | Wort after Hops | Spent Hops | Starting Yeast | Beer after Yeast | Spent Yeast | Beer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | |
| Water extracts | ||||||||||||
| Ego | 25.801 ± 2.499 | 47.671 ± 3.022 | 14.667 ± 3.096 | 2233.942 ± 238.077 | 1954.786 ± 259.234 | 636.489 ± 20.753 | 141.376 ± 62.439 | 8.257 ± 1.318 | 177.450 ± 4.346 | 322.082 ± 104.791 | 35.475 ± 0.942 | 31.683 ± 2.487 |
| Alter | 22.699 ± 0.955 | 76.374 ± 6.201 | 13.865 ± 15.869 | 491.661 ± 4.664 | 23.974 ± 5.377 | 3.905 ± 0.347 | 226.365 ± 38.859 | 30.553 ± 3.346 | 37.485 ± 1.723 | |||
| Fiat lux | 24.045 ± 1.849 | 61.555 ± 5.267 | 17.855 ± 4.889 | 527.328 ± 31.529 | 49.818 ± 29.808 | 7.967 ± 1.381 | 251.736 ± 25.611 | 28.107 ± 4.855 | 43.687 ± 3.508 | |||
| Triplo malto | 21.389 ± 2.694 | 33.621 ± 2.996 | 18.973 ± 10.953 | 618.662 ± 2.42 | 115.768 ± 25.891 | 9.743 ± 0.730 | 188.430 ± 11.987 | 51.308 ± 3.046 | 34.704 ± 0.607 | |||
| Ubi | 25.519 ± 0.721 | 98.108 ± 13.203 | 18.890 ± 2.038 | 424.874 ± 28.439 | 214.435 ± 18.075 | 7.150 ± 0.754 | 122.265 ± 14.521 | 22.373 ± 4.954 | 33.742 ± 4.285 | |||
| Maior | 46.823 ± 0.031 | 112.802 ± 9.703 | 16.890 ± 5.036 | 314.732 ± 9.467 | 57.636 ± 23.309 | 6.704 ± 0.409 | 255.149 ± 56.934 | 26.103 ± 1.652 | 28.452 ± 1.507 | |||
| 70° ethanol extracts | ||||||||||||
| Ego | 50.669 ± 1.066 | 47.671 ± 3.022 | 20.341 ± 2.163 | 6547.220 ± 174.500 | 5405.892 ± 127.897 | 2189.582 ± 329.231 | 141.376 ± 62.439 | 5.408 ± 2.858 | 65.906 ± 4.600 | 322.082 ± 104.791 | 47.139 ± 0.321 | 31.683 ± 2.487 |
| Alter | 41.305 ± 2.778 | 76.374 ± 6.201 | 21.722 ± 2.163 | 1807.521 ± | 23.974 ± 5.377 | 5.256 ± 4.568 | 226.365 ± 38.859 | 41.372 ± 0.623 | 37.485 ± 1.723 | |||
| Fiat lux | 44.566 ± 6.738 | 61.555 ± 5.267 | 18.306 ± 0.436 | 3935.707 ± 108.798 | 49.818 ± 29.808 | 4.724 ± 0.491 | 251.736 ± 25.611 | 41.194 ± 3.149 | 43.687 ± 3.508 | |||
| Triplo malto | 68.801 ± 1.381 | 33.621 ± 2.996 | 17.356 ± 0.228 | 3859.089 ± 87.956 | 115.768 ± 25.891 | 6.479 ± 0.359 | 188.430 ± 11.987 | 45.35 ± 2.476 | 34.704 ± 0.607 | |||
| Ubi | 76.198 ± 7.221 | 98.108 ± 13.203 | 18.879 ± 2.753 | 1158.326 ± 46.978 | 214.435 ± 18.075 | 5.987 ± 1.750 | 122.265 ± 14.521 | 36.723 ± 3.798 | 33.742 ± 4.285 | |||
| Maior | 97.207 ± 19.118 | 112.802 ± 9.703 | 10.097 ± 0.761 | 1353.216 ± 10.055 | 57.636 ± 23.309 | 5.789 ± 1.098 | 255.149 ± 56.934 | 32.814 ± 1.543 | 28.452 ± 1.507 | |||
| FRAP | Starting Malt | Wort | Spent Malt | Starting Hops: Perle | Starting Hops: Saaz | Starting Hops: Perle And Saaz | Wort after Hops | Spent Hops | Starting Yeast | Beer after Yeast | Spent Yeast | Beer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | TEAC (µmol TE/g) | |
| Water extracts | ||||||||||||
| Ego | 68.336 ± 2.637 | 26.036 ± 0.876 | 67.712 ± 1.439 | 332.045 ± 91.163 | 377.423 ± 45.244 | 141.194 ± 4.131 | 52.563 ± 1.576 | 102.657 ± 3.987 | 71.045 ± 5.859 | 88.967 ± 4.544 | 123.906 ± 1.915 | 109.843 ± 2.186 |
| Alter | 80.394 ± 1.025 | 28.144 ± 2.140 | 65.103 ± 0.607 | 131.694 ± 1.736 | 58.827 ± 3,005 | 101.557 ± 4.878 | 84.849 ± 6.334 | 103.154 ± 1.558 | 110.550 ± 1.830 | |||
| Fiat lux | 56.121 ± 1.480 | 25.395 ± 2.761 | 66.535 ± 1.079 | 136.897 ± 5.023 | 57.941 ± 12.189 | 90.999 ± 14.971 | 67.022 ± 2.444 | 106.609 ± 0.937 | 115.380 ± 0.051 | |||
| Triplo malto | 59.357 ± 0.904 | 29.073 ± 0.651 | 69.598 ± 2.014 | 151.032 ± 0.179 | 35.117 ± 0.650 | 87.898 ± 9.373 | 49.944 ± 0,082 | 124.793 ± 7.251 | 104.046 ± 7.680 | |||
| Ubi | 60.627 ± 2.158 | 30.191 ± 0.254 | 63.027 ± 2.711 | 138.153 ± 5.390 | 49.744 ± 2.123 | 97.256 ± 5.046 | 43.891 ± 0.378 | 136.719 ± 2.908 | 112.839 ± 0.457 | |||
| Maior | 59.996 ± 1.432 | 29.587 ± 0.824 | 43.388 ± 0.952 | 111.194 ± 3.844 | 74.441 ± 10.748 | 95.247± 4.456 | 99.051 ± 15.886 | 130.168 ± 14.966 | 125.159 ± 1.237 | |||
| 70° ethanol extracts | ||||||||||||
| Ego | 37.723 ± 0.987 | 26.036 ± 0.876 | 38.772 ± 1.639 | 120.133 ± 44.166 | 110.257 ± 37.122 | 120.605 ± 1.123 | 52.563 ± 1.576 | 32.245 ± 1.238 | 44.494 ± 0.501 | 88.967 ± 4.544 | 73.707 ± 1.224 | 109.843 ± 2.186 |
| Alter | 54.343 ± 2.835 | 28.144 ± 2.140 | 35.607 ± 0.560 | 121.971 ± 11.649 | 58.827 ± 3,005 | 31.236 ± 1.168 | 84.849 ± 6.334 | 82.229 ± 2.623 | 110.550 ± 1.830 | |||
| Fiat lux | 40.644 ± 6.405 | 25.395 ± 2.761 | 30.618 ± 0.425 | 53.718 ± 65.966 | 57.941 ± 12.189 | 33.813 ± 0.977 | 67.022 ± 2.444 | 74.299 ± 3.587 | 115.380 ± 0.051 | |||
| Triplo malto | 38.048 ± 0.159 | 29.073 ± 0.651 | 30.953 ± 0.041 | 211.213 ± 1.222 | 35.117 ± 0.650 | 29.281 ± 1.013 | 49.944 ± 0,082 | 70.214 ± 2.305 | 104.046 ± 7.680 | |||
| Ubi | 47.207 ± 0.418 | 30.191 ± 0.254 | 33.143 ± 0.179 | 81.798 ± 0.068 | 49.744 ± 2.123 | 30.911 ± 0.634 | 43.891 ± 0.378 | 77.751 ± 2.05 | 112.839 ± 0.457 | |||
| Maior | 33.932 ± 4.415 | 29.587 ± 0.824 | 36.506 ± 0.559 | 70.116 ± 0.923 | 74.441 ± 10.748 | 29.812 ± 0.913 | 99.051 ± 15.886 | 72.187 ± 0.679 | 125.159 ± 1.237 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Censi, R.; Vargas Peregrina, D.; Gigliobianco, M.R.; Lupidi, G.; Angeloni, C.; Pruccoli, L.; Tarozzi, A.; Di Martino, P. New Antioxidant Ingredients from Brewery By-Products for Cosmetic Formulations. Cosmetics 2021, 8, 96. https://doi.org/10.3390/cosmetics8040096
Censi R, Vargas Peregrina D, Gigliobianco MR, Lupidi G, Angeloni C, Pruccoli L, Tarozzi A, Di Martino P. New Antioxidant Ingredients from Brewery By-Products for Cosmetic Formulations. Cosmetics. 2021; 8(4):96. https://doi.org/10.3390/cosmetics8040096
Chicago/Turabian StyleCensi, Roberta, Dolores Vargas Peregrina, Maria Rosa Gigliobianco, Giulio Lupidi, Cristina Angeloni, Letizia Pruccoli, Andrea Tarozzi, and Piera Di Martino. 2021. "New Antioxidant Ingredients from Brewery By-Products for Cosmetic Formulations" Cosmetics 8, no. 4: 96. https://doi.org/10.3390/cosmetics8040096
APA StyleCensi, R., Vargas Peregrina, D., Gigliobianco, M. R., Lupidi, G., Angeloni, C., Pruccoli, L., Tarozzi, A., & Di Martino, P. (2021). New Antioxidant Ingredients from Brewery By-Products for Cosmetic Formulations. Cosmetics, 8(4), 96. https://doi.org/10.3390/cosmetics8040096












