Retinol Has a Skin Dehydrating Effect That Can Be Improved by a Mixture of Water-Soluble Polysaccharides
Abstract
1. Introduction
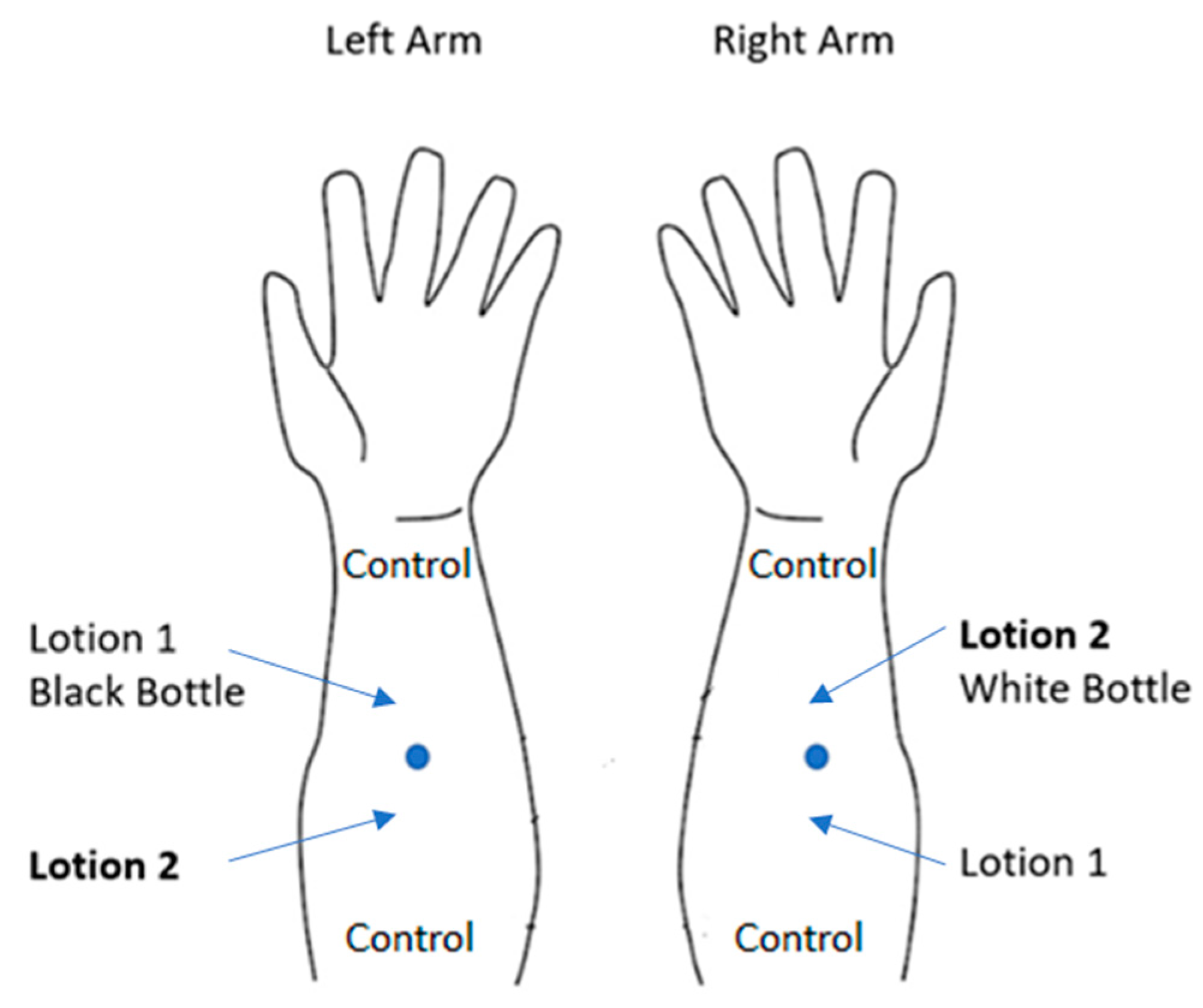
2. Materials and Methods
3. Results and Discussion
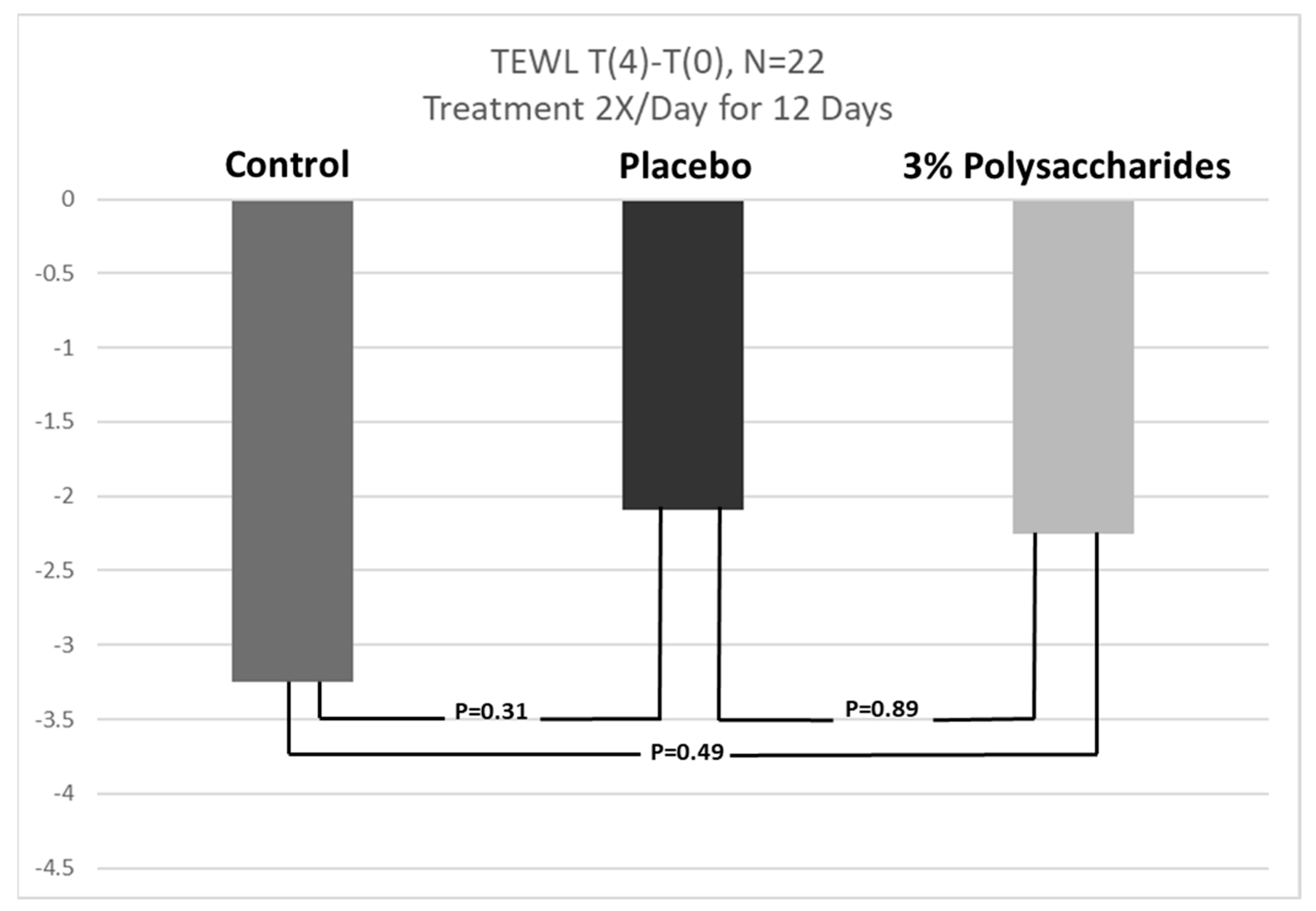
3.1. Transepidermal Water Loss (TEWL) Response
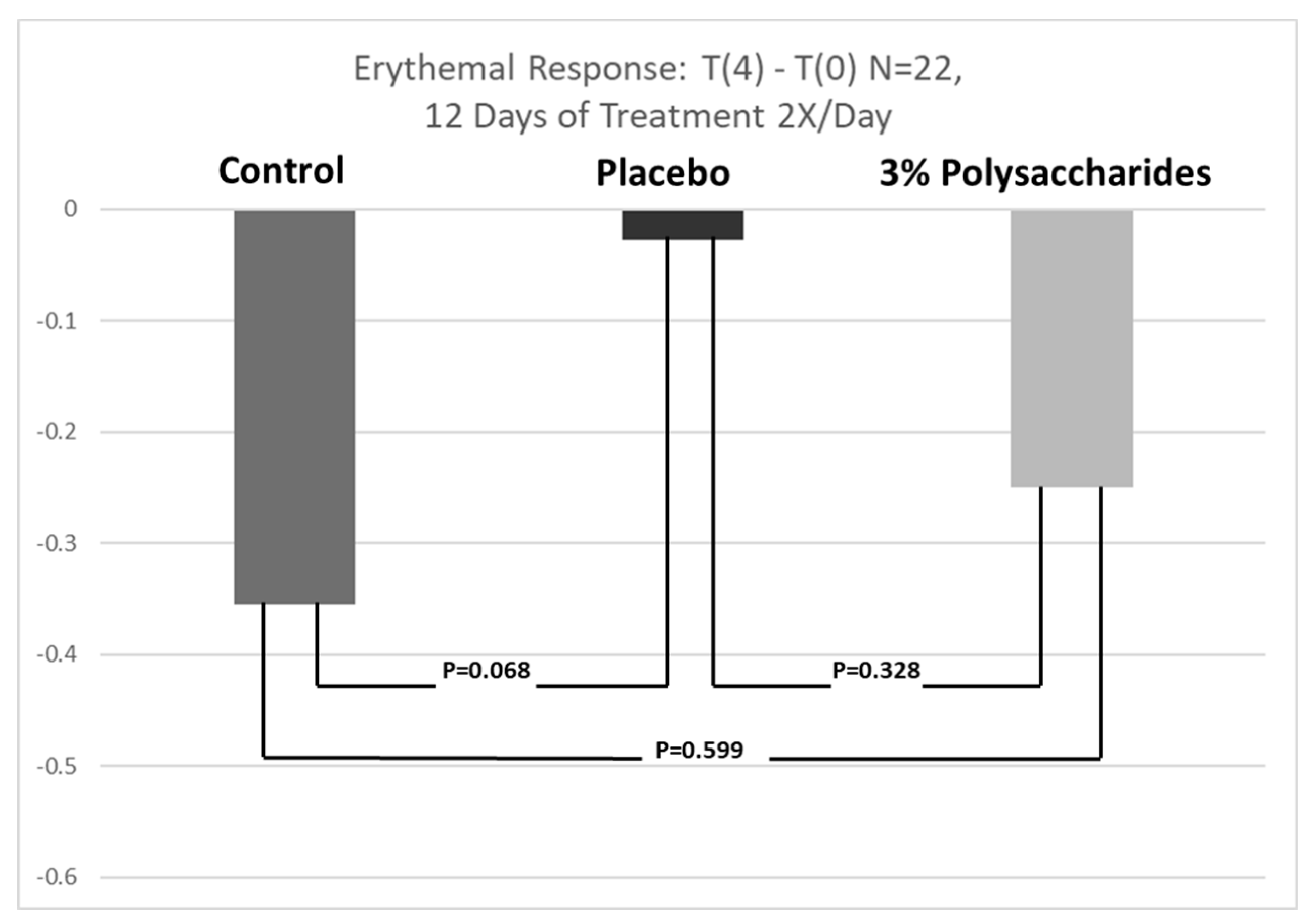
3.2. Erythema Response
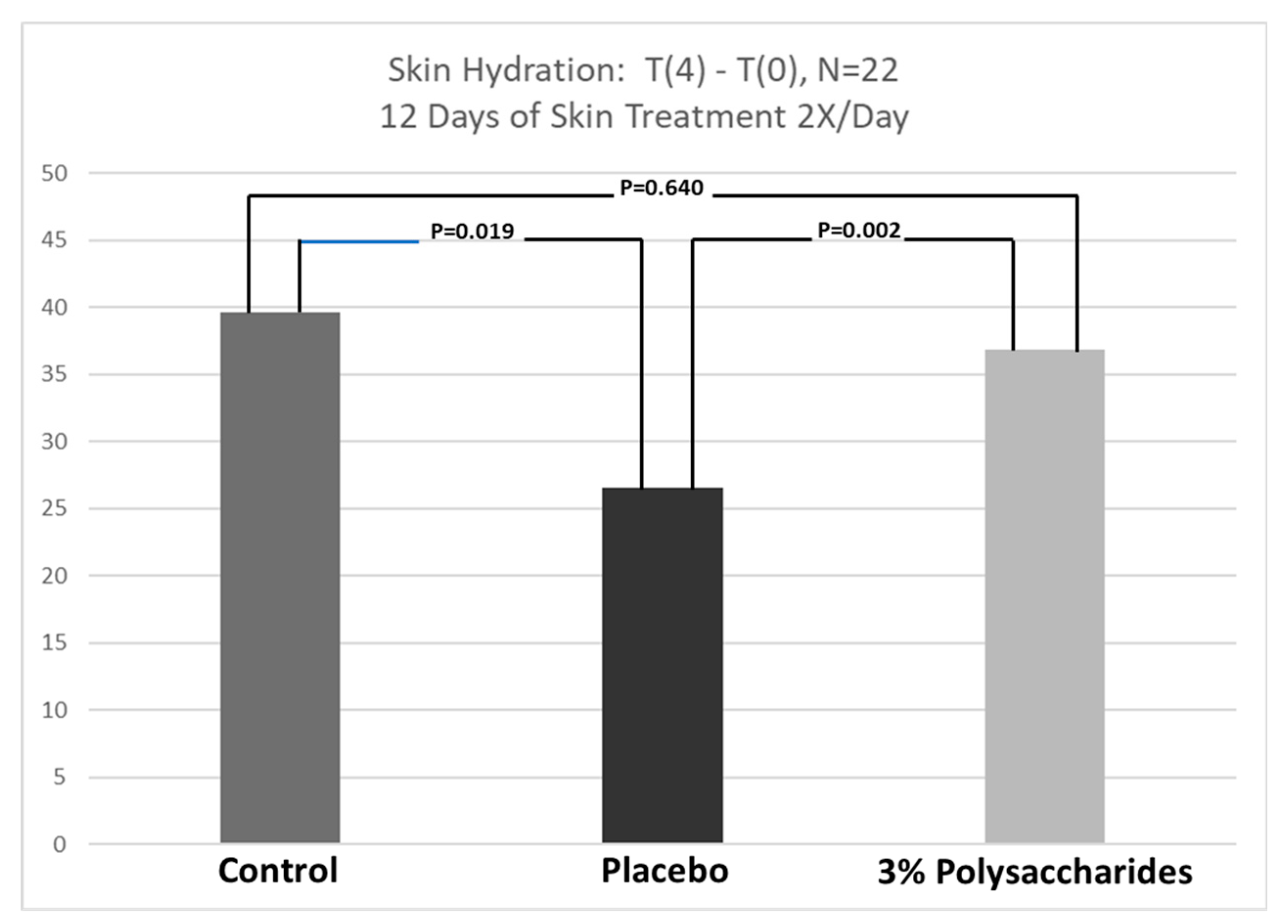
3.3. Skin Hydration Response
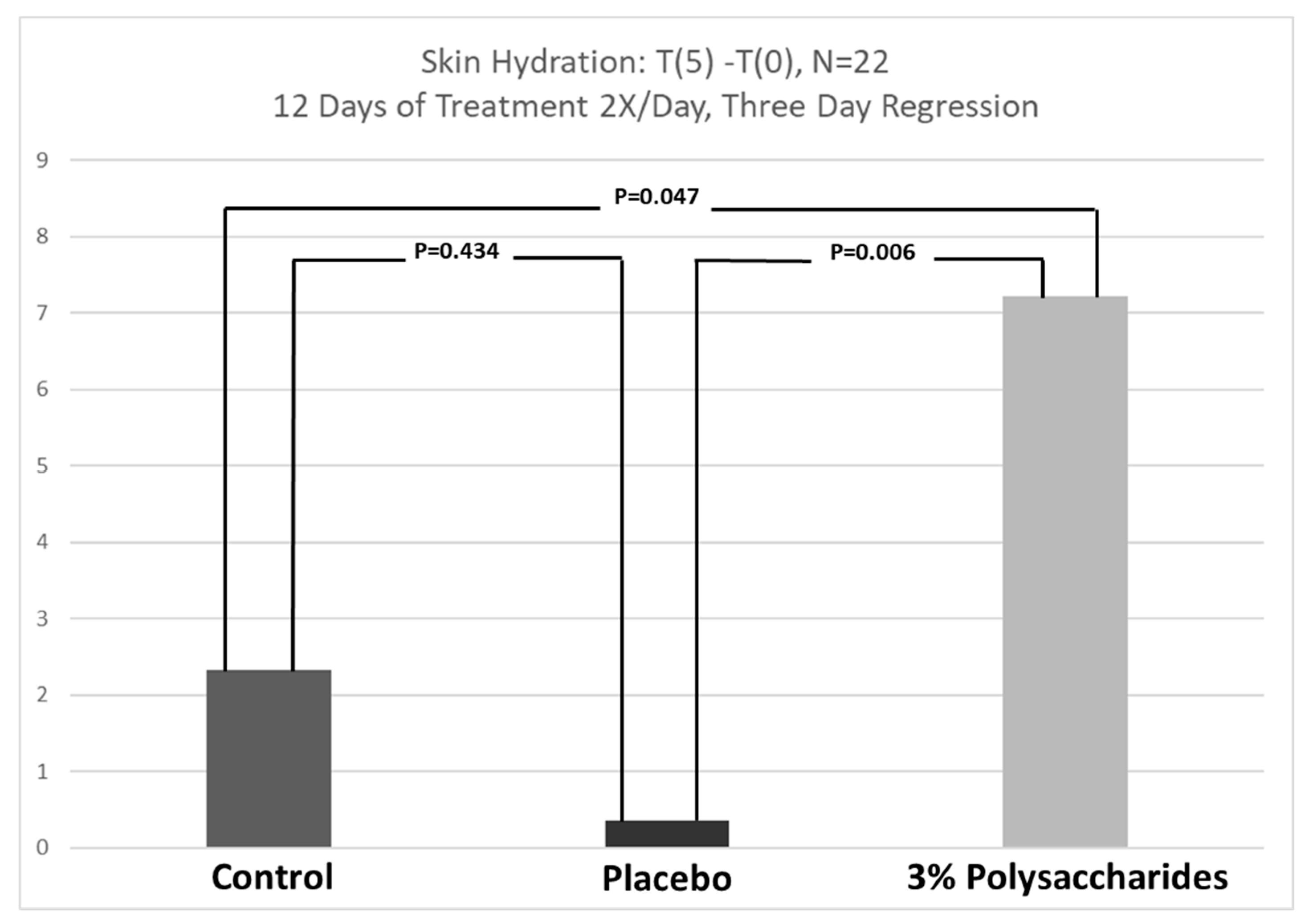
3.4. Extended Skin Hydration Regression Study Response
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fisher, G.J.; Esmann, J.; Griffiths, C.E.; Talwar, H.S.; Duell, E.A.; Hammerberg, C.; Elder, J.T.; Finkel, L.J.; Karabin, G.D.; Nickokoloff, B.J. Cellular, Immunologic and Biochemical Characterization of Topical Retinoic Acid-Treated Human Skin. J. Investig. Dermatol. 1991, 96, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Fluhr, J.W.; Vienne, M.P.; Lauze, C.; Dupuy, P.; Gehring, W.; Gloor, M. Tolerance Profile of Retinol, Retinaldehyde and Retinoic Acid Under Maximized and Long-Term Clinical Conditions. Dermatology 1999, 199 (Suppl. 1), 57–60. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Date, A.; Patravale, V.; Korting, H.C.; Roeder, A.; Weindl, G. Retinoids in the Treatment of Skin Aging: An Overview of Clinical Efficacy. Clin. Interv. Aging 2006, 1, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Kligman, A.M.; Willis, I.A. New Formula for Depigmenting Human Skin. Arch. Dermatol. 1975, 111, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Duell, E.A.; Fisher, G.J.; Datta, S.C.; Wang, Z.Q.; Reddy, A.P.; Tavakkol, A.; Yi, J.Y.; Griffiths, C.E.M.; Elder, J.T.; et al. Application of Retinol to Human Skin In Vivo Induces Epidermal Hyperplasia and Cellular Retinoid Binding Proteins Characteristic of Retinoic Acid but Without Measurable Retinoic Acid Levels or Irritation. J. Investig. Dermatol. 1995, 105, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; He, T.; Fisher, G.J.; Voorhees, J.J.; Quan, T. Molecular Basis of Retinol Anti-Aging Properties in Naturally Aged Human Skin In Vivo. Int. J. Cosmet. Sci. 2017, 39, 56–65. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, D.H.; Mazur, C.; Wortzman, M.S. Efficacy and tolerability of a double-conjugated retinoid cream vs 1.0% retinol cream or 0.025% tretinoin cream in subjects with mild to severe photoaging. J. Cosmet. Dermatol. 2017, 16, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Gruber, J.V.; Stojkoska, V. NLRP Inflammasomes and Induced Skin Inflammation, Barrier Recovery and Extended Skin Hydration. Int. J. Cosmet. Sci. 2020, 42, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Gruber, J.V. Inflammasomes: Key to the Engine of Skin Inflammaging. Pers. Care N. Am. 2020, 3, 27–33. [Google Scholar]
- Gankande, T.U.; Duke, J.M.; Wood, F.; Wallace, H.J. Interpretation of the DermaLab Combo® Pigmentation and Vascularity Measurements in Burn Scar Assessment: An Exploratory Analysis. Burns 2015, 41, 1176–1185. [Google Scholar] [CrossRef] [PubMed]
- Hua, W.; Fan, L.M.; Dai, R.; Luan, M.; Xie, H.; Li, A.Q.; Li, L. Comparison of two series of non-invasive instruments used for the skin physiological properties measurements: The DermaLab® from Cortex Technology vs the series of detectors from Courage & Khazaka. Skin Res. Technol. 2017, 23, 70–78. [Google Scholar] [PubMed]
- Gruber, J.V.; Holtz, R. In vitro expression of NLRP inflammasome-induced active Caspase-1 expression in normal human epidermal keratinocytes (NHEK) by various exogenous threats and subsequent inhibition by naturally derived ingredient blends. J. Inflamm. Res. 2019, 12, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.A.; Yan, M.D.; Lin, H.T.V.; Li, K.L.; Lin, Y.C. Toxicological evaluation of low molecular weight Fucoidan in vitro and in vivo. Mar. Drugs 2016, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Ozanne, H.; Toumi, H.; Roubinet, B.; Landemarre, L.; Lespessailles, E.; Daniellou, R.; Cesaro, A. Laminarin effects, a β-(1,3)-Glucan, on skin cell inflammation and oxidation. Cosmetics 2020, 7, 66. [Google Scholar] [CrossRef]
- Petruk, G.; Di Lorenzo, F.; Imbimbo, P.; Silipo, A.; Bonina, A.; Rizza, L.; Piccoli, R.; Monti, D.M.; Lanzetta, R. Protective effect of Opuntia ficus-indica L. cladodes against UVA-induced oxidative stress in normal human keratinocytes. Bioorg. Med. Chem. Lett. 2017, 27, 5485–5489. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.S. Larch arabinogalactan: Clinical relevance of a novel immune enhancing polysaccharide. Altern. Med. Rev. 1999, 4, 96–103. [Google Scholar] [PubMed]
- Novak, M.; Vetvicka, V. Beta-glucans, history, and the present: Immunomodulatory aspects and mechanisms of action. J. Immunotoxicol. 2008, 5, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Bekhradnia, S.; Naz, I.; Lund, R.; Effenberg, C.; Appelhaus, D.; Sande, S.A.; Nystrom, B. Characterization of oligosaccharide-functionalized hyperbranched poly(ethylene imine) and their complexes with retinol in aqueous solution. J. Colloid Interface Sci. 2015, 458, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, R.K.; Bojanowski, K. Bakuchiol: A retinol-like functional compound revealed by gene expression profiling and clinically proven to have anti-aging effects. Int. J. Cosmet. Sci. 2014, 36, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Li, W.H.; Wong, H.K.; Serrano, J.; Randhawa, M.J.; Kaur, S.; Southhall, M.D.; Parsa, R. Topical stabilized retinol treatment induces the expression of HAS genes and HA production in human skin In vitro and in vivo. Arch. Dermatol. Res. 2017, 309, 275–283. [Google Scholar] [CrossRef] [PubMed]
| Ingredient | INCI Name | Quantity (%) | |
|---|---|---|---|
| Placebo | |||
| Water | Aqua | 95 | |
| Jeesperse®ICE-T®-LB-T-NS | Sodium Acrylate/Sodium Acrylol Dimethyl Taurate Copolymer (and) Cetyl Alcohol (and) Glycol Stearate (and) Glyceryl Stearate (and) Caprylic/Capric Triglyceride | 3.5 | |
| Jeecide® Phenoxy | Phenoxyethanol | 1 | |
| Retinol 10 SU | Retinol (and) Glycine Soja (Soybean) Oil | 0.5 | |
| Active | |||
| Water | Aqua | 92 | |
| Jeesperse®ICE-T®-LB-T-NS | Sodium Acrylate/Sodium Acrylol Dimethyl Taurate Copolymer (and) Cetyl Alcohol (and) Glycol Stearate (and) Glyceryl Stearate (and) Caprylic/Capric Triglyceride | 3.5 | |
| Jeecide® Phenoxy | Phenoxyethanol | 1 | |
| Retinol 10 SU | Retinol (and) Glycine Soja (Soybean) Oil | 0.5 | |
| StrataPhix™ POLY | Water (and) Glycerin (and) Laminaria Digitata Extract (and) Opuntia Ficus-Indica Stem Extract (and) Galactoarabininan (and) Schizophyllan | 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gruber, J.V.; Stojkoska, V.; Riemer, J. Retinol Has a Skin Dehydrating Effect That Can Be Improved by a Mixture of Water-Soluble Polysaccharides. Cosmetics 2020, 7, 80. https://doi.org/10.3390/cosmetics7040080
Gruber JV, Stojkoska V, Riemer J. Retinol Has a Skin Dehydrating Effect That Can Be Improved by a Mixture of Water-Soluble Polysaccharides. Cosmetics. 2020; 7(4):80. https://doi.org/10.3390/cosmetics7040080
Chicago/Turabian StyleGruber, James V., Venera Stojkoska, and Jed Riemer. 2020. "Retinol Has a Skin Dehydrating Effect That Can Be Improved by a Mixture of Water-Soluble Polysaccharides" Cosmetics 7, no. 4: 80. https://doi.org/10.3390/cosmetics7040080
APA StyleGruber, J. V., Stojkoska, V., & Riemer, J. (2020). Retinol Has a Skin Dehydrating Effect That Can Be Improved by a Mixture of Water-Soluble Polysaccharides. Cosmetics, 7(4), 80. https://doi.org/10.3390/cosmetics7040080






