Hair Growth Promotion by Extracts of Inula Helenium and Caesalpinia Sappan Bark in Patients with Androgenetic Alopecia: A Pre-clinical Study Using Phototrichogram Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
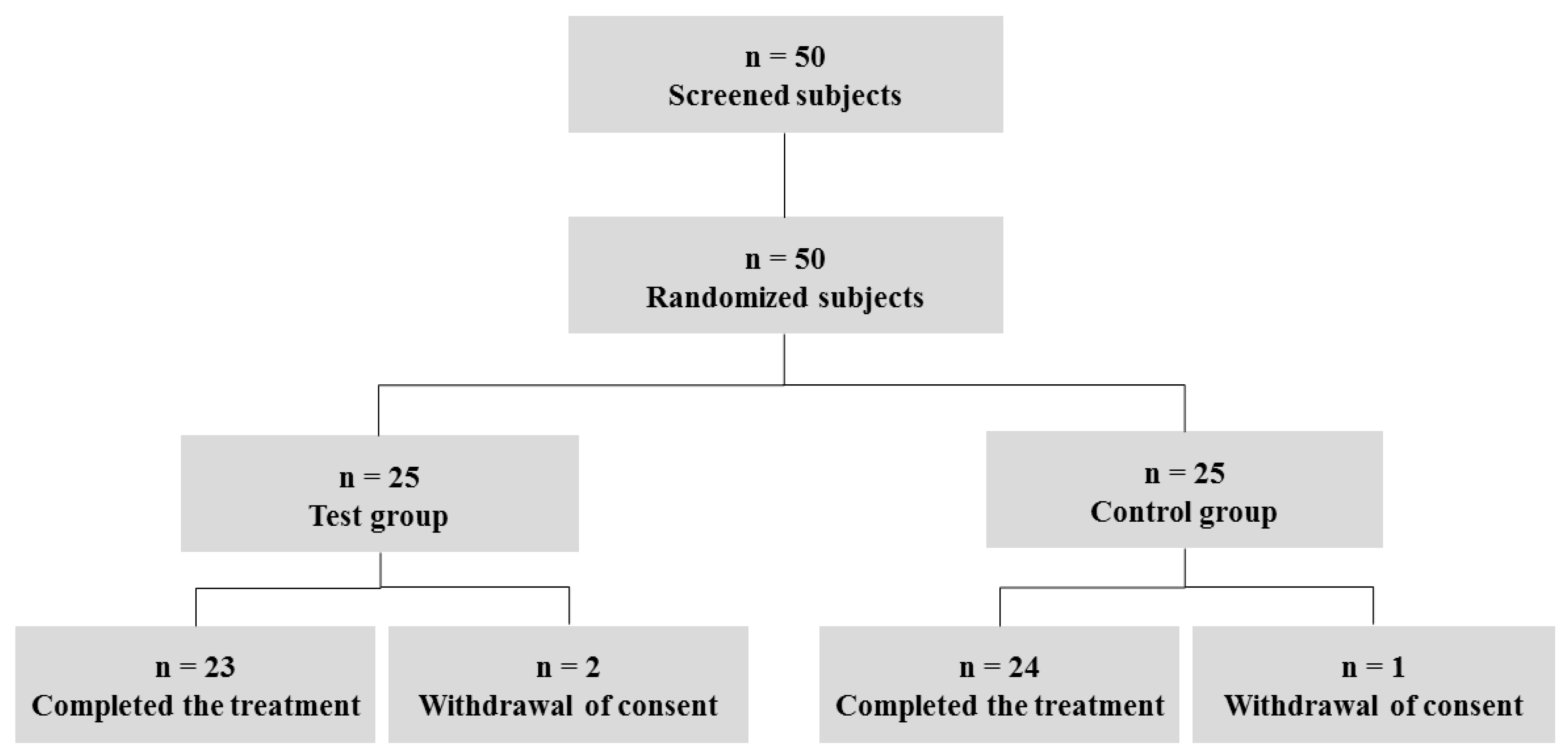
2.2. Subjects and Study Design
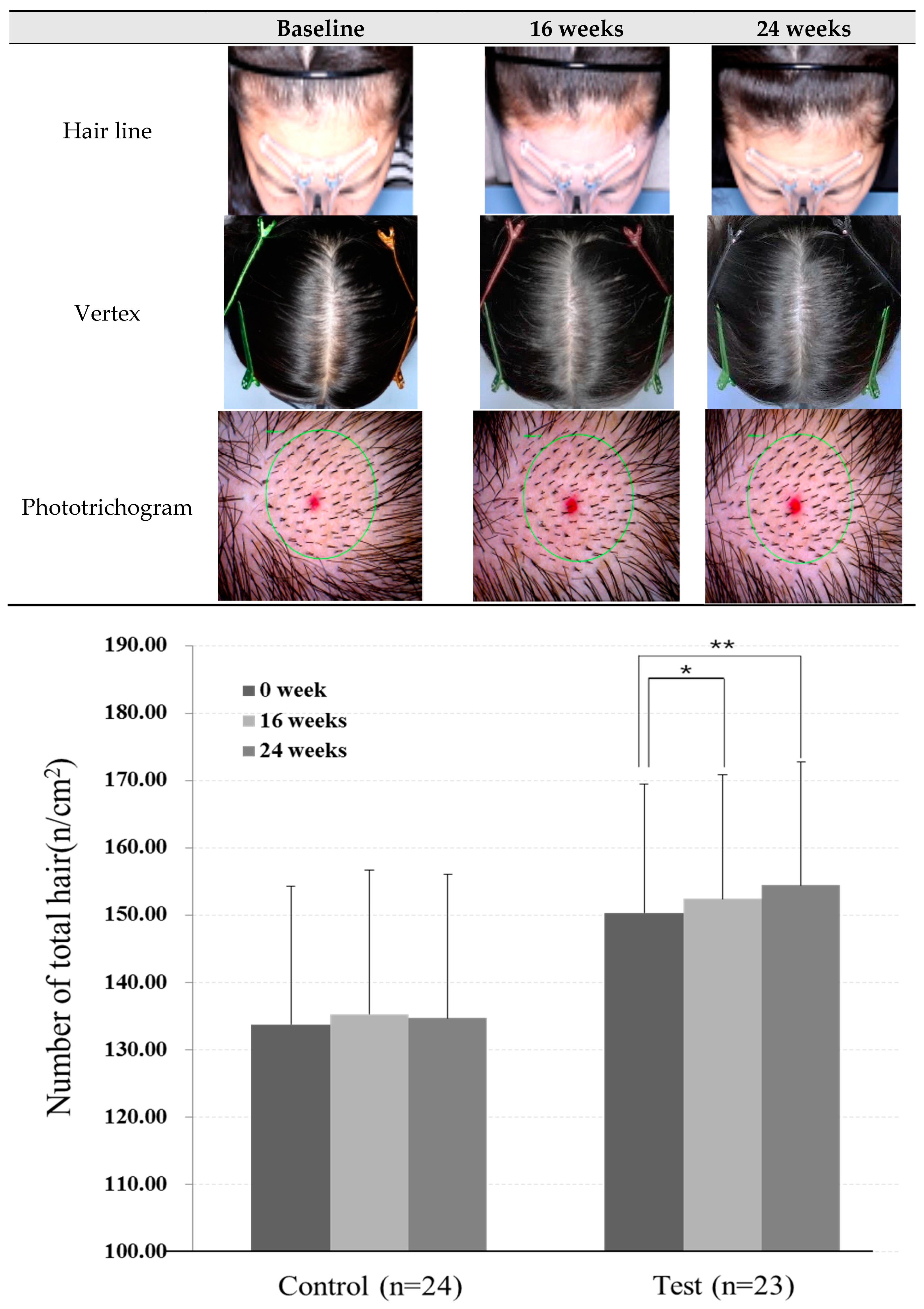
2.3. Measurement of Hair Density and Total Hair Counts
3. Statistical Analysis
4. Results
4.1. General Characteristics of the Subjects
4.2. Effects of Scalp Shampoo on Hair Density and Total Hair Counts
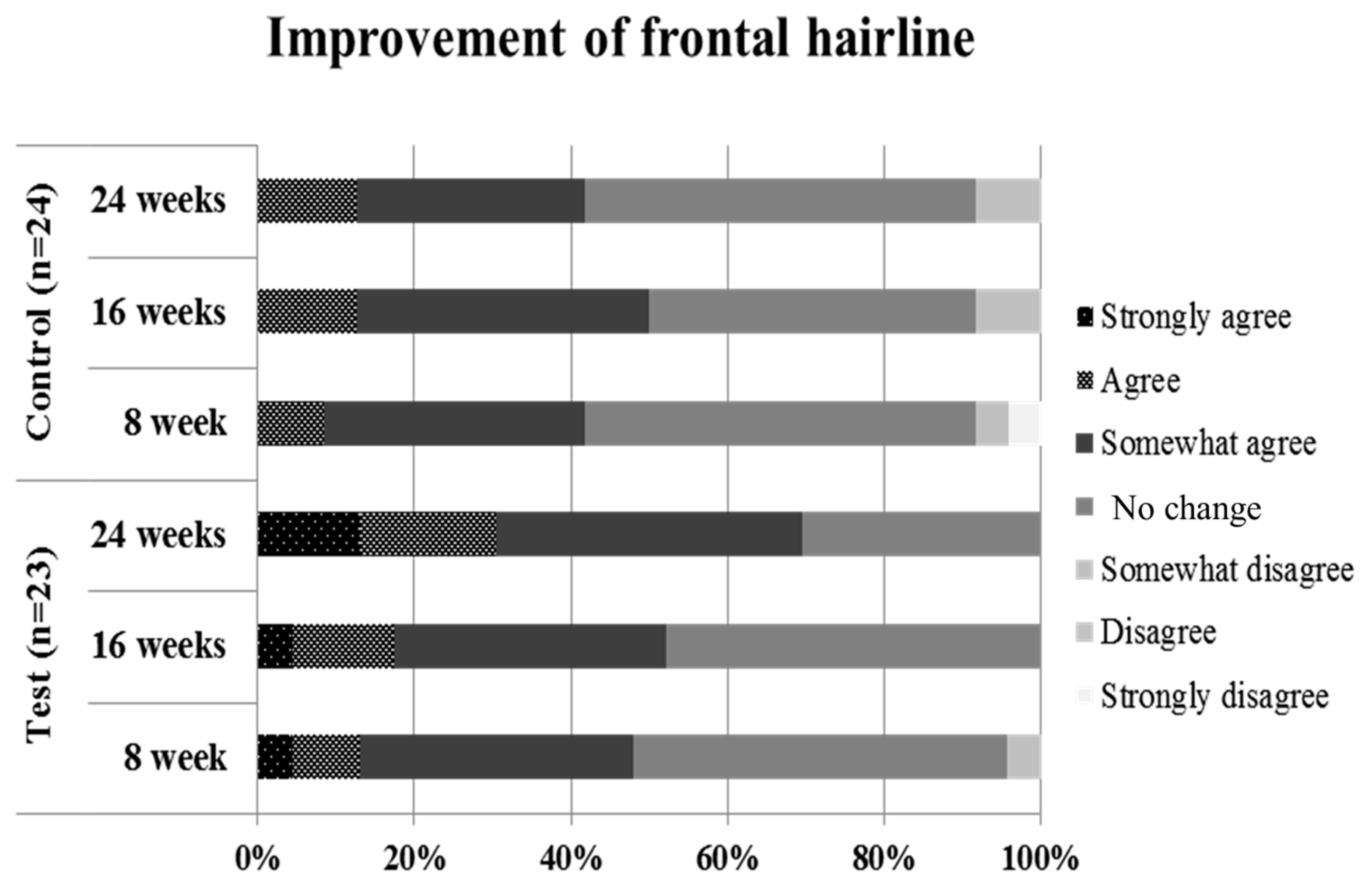
4.3. Assessment of Self-Questionnaires by Subjects
5. Discussion
Author Contributions
Funding
Acknowledgments
Availability of Data
Conflicts of Interest
References
- Adams, J.U. Raising hairs. Nat. Biotechnol. 2011, 29, 474–476. [Google Scholar] [CrossRef] [PubMed]
- Stenn, K.; Parimoo, S.; Zheng, Y.; Barrows, T.; Boucher, M.; Washenik, K. Bioengineering the hair follicle. Organogenesis 2007, 3, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Stenn, K.S.; Cotsarelis, G. Bioengineering the hair follicle: Fringe benefits of stem cell technology. Curr. Opin. Biotechnol. 2005, 16, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Anitha, B.; Inamadar, A.C.; Ragunatha, S. Finasteride-its impact on sexual function and prostate cancer. J. Cutan. Aesthetic Surg. 2009, 2, 12. [Google Scholar]
- Seok, J.; Kim, T.S.; Kwon, H.J.; Lee, S.P.; Kang, M.H.; Kim, B.J.; Kim, M.N. Efficacy of Cistanche tubulosa and Laminaria japonica extracts (MK-R7) supplement in preventing patterned hair loss and promoting scalp health. Clin. Nutr. Res. 2015, 4, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Stenn, K.S.; Paus, R. Controls of hair follicle cycling. Physiol. Rev. 2001, 81, 449–494. [Google Scholar] [CrossRef]
- Inui, S.; Fukuzato, Y.; Nakajima, T.; Yoshikawa, K.; Itami, S. Identification of androgen-inducible TGF-beta1 derived from dermal papilla cells as a key mediator in androgenetic alopecia. J. Investig. Dermatol. Symp. Proc. 2003, 8, 69–71. [Google Scholar] [CrossRef]
- Kwon, O.S.; Han, J.H.; Yoo, H.G.; Chung, J.H.; Cho, K.H.; Eun, H.C.; Kim, K.H. Human hair growth enhancement in vitro by green tea epigallocatechin-3-gallate (EGCG). Phytomedicine Int. J. Phytother. Phytopharm. 2007, 14, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.E. Molecular mechanisms regulating hair follicle development. J. Investig. Dermatol. 2002, 118, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.E.; Choi, H.C.; Nam, G.; Choi, B.Y. Costunolide promotes the proliferation of human hair follicle dermal papilla cells and induces hair growth in C57BL/6 mice. J. Cosmet. Dermatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.E.; Choi, H.C.; Lee, I.-C.; Yuk, D.Y.; Lee, H.; Choi, B.Y. 3-Deoxysappanchalcone promotes proliferation of human hair follicle dermal papilla cells and hair growth in C57BL/6 mice by modulating Wnt/β-catenin and STAT signaling. Biomol. Ther. 2016, 24, 572. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Ro, B.I.; Hong, S.P.; Bak, H.; Sim, W.Y.; Kim, D.W.; Park, J.K.; Ihm, C.W.; Eun, H.C.; Kwon, O.S.; et al. A new classification of pattern hair loss that is universal for men and women: Basic and specific (BASP) classification. J. Am. Acad. Dermatol. 2007, 57, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.; Lee, S.; Yoo, M.; Park, W.S.; Lee, S.; Boo, Y.; Koh, J.S. Effects of a new mild shampoo for preventing hair loss in Asian by a simple hand-held phototrichogram technique. Int. J. Cosmet. Sci. 2011, 33, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Choi, B.Y. Costunolide-A Bioactive Sesquiterpene Lactone with Diverse Therapeutic Potential. Int. J. Mol. Sci. 2019, 20, 2926. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Test Group n = 23 | Control Group n = 24 | p-Value | |
|---|---|---|---|---|
| n (%) | n (%) | |||
| Sex | Male | 7 (30.43) | 6 (25.00) | |
| Female | 16 (69.57) | 18 (75.00) | ||
| Age | Mean ± SD | 40.33 ± 7.88 | 41.08 ± 6.74 | 0.448 |
| Median | 42.00 | 43.00 | ||
| Min, Max | 24.00, 52.00 | 25.00, 51.00 | ||
| 20–29 | 3 (13.04) | 2 ( 8.33) | ||
| 30–39 | 6 (26.09) | 6 (25.00) | ||
| 40–49 | 12 (52.17) | 15 (62.50) | ||
| ≥50 | 2 ( 8.70) | 1 ( 4.17) | ||
| (a) | Baseline | 8 weeks | 16 weeks | 24 weeks | |
| Control group (n = 24) | Mean 1 | 0.00 | 0.00 | 0.04 | 0.00 |
| SD | 0.00 | 0.00 | 0.20 | 0.29 | |
| p-value | - | 1.000 | 0.317 | 1.000 | |
| Test group (n = 23) | Mean 1 | 0.00 | 0.04 | 0.09 | 0.13 |
| SD | 0.00 | 0.21 | 0.29 | 0.34 | |
| p-value | - | 0.317 | 0.157 | 0.083 | |
| (b) | Baseline | 8 weeks | 16 weeks | 24 weeks | |
| Control group (n = 24) | Mean (n/cm2) 2 | 133.75 | - | 135.29 | 134.71 |
| SD | 20.54 | - | 21.45 | 21.34 | |
| p-value 3 | - | - | 0.085 | 0.285 | |
| Test group (n = 23) | Mean (n/cm2) 2 | 145.70 | - | 147.87 | 150.00 |
| SD | 23.82 | - | 23.21 | 22.83 | |
| p-value 3 | - | - | 0.044 * | 0.003 ** | |
| Group | Item | p-Value 1 | ||
|---|---|---|---|---|
| 8 weeks | 16 weeks | 24 weeks | ||
| Control vs. Test | Improvement of vertex hair | 0.706 | 0.207 | 0.230 |
| Improvement of frontal hairline 1 | 0.524 | 0.574 | 0.025 * | |
| Improvement of hair loss | 0.703 | 0.537 | 0.119 | |
| Hair satisfaction | 0.715 | 0.238 | 0.140 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, H.C.; Nam, G.W.; Jeong, N.H.; Choi, B.Y. Hair Growth Promotion by Extracts of Inula Helenium and Caesalpinia Sappan Bark in Patients with Androgenetic Alopecia: A Pre-clinical Study Using Phototrichogram Analysis. Cosmetics 2019, 6, 66. https://doi.org/10.3390/cosmetics6040066
Choi HC, Nam GW, Jeong NH, Choi BY. Hair Growth Promotion by Extracts of Inula Helenium and Caesalpinia Sappan Bark in Patients with Androgenetic Alopecia: A Pre-clinical Study Using Phototrichogram Analysis. Cosmetics. 2019; 6(4):66. https://doi.org/10.3390/cosmetics6040066
Chicago/Turabian StyleChoi, Hyoung Chul, Gae Won Nam, Noh Hee Jeong, and Bu Young Choi. 2019. "Hair Growth Promotion by Extracts of Inula Helenium and Caesalpinia Sappan Bark in Patients with Androgenetic Alopecia: A Pre-clinical Study Using Phototrichogram Analysis" Cosmetics 6, no. 4: 66. https://doi.org/10.3390/cosmetics6040066
APA StyleChoi, H. C., Nam, G. W., Jeong, N. H., & Choi, B. Y. (2019). Hair Growth Promotion by Extracts of Inula Helenium and Caesalpinia Sappan Bark in Patients with Androgenetic Alopecia: A Pre-clinical Study Using Phototrichogram Analysis. Cosmetics, 6(4), 66. https://doi.org/10.3390/cosmetics6040066





