The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims
Abstract
:1. Introduction
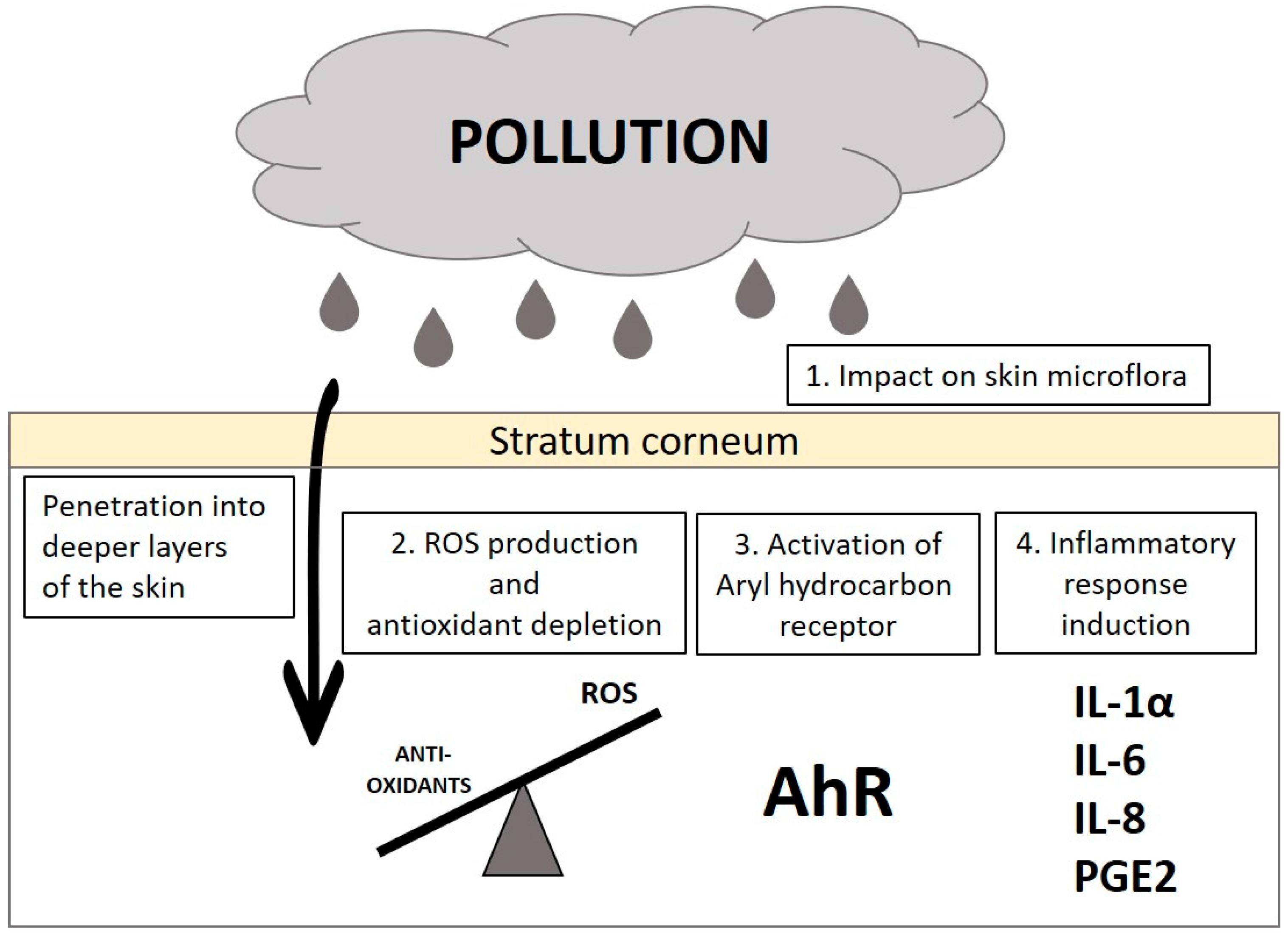
2. Effect of Pollutants on the Skin and Its Mechanism of Action
2.1. Impact on Skin Microflora
2.2. Generation of Reactive Oxygen Species (ROS)
2.3. Activation of the Aryl Hydrocarbon Receptor (AhR)
2.4. Induction of Inflammatory Cascade
3. Testing of Anti-Pollution Claims
3.1. In Vivo Efficacy Testing
3.2. In Vitro Efficacy Testing
4. Summary
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Ambient Air Pollution: A Global Assessment of Exposure and Burden of Disease; World Health Organization: Geneva, Switzerland, 2016; pp. 1–131. [Google Scholar]
- Baudouin, C.; Charveron, M.; Tarroux, R.; Gall, Y. Environmental pollutants and skin cancer. Cell Biol. Toxicol. 2002, 18, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Drakaki, E.; Dessinioti, C.; Antoniou, C.V. Air pollution and the skin. Front. Environ. Sci. 2014, 2, 1–6. [Google Scholar] [CrossRef]
- Dales, R.; Liu, L.; Wheeler, A.J.; Gilbert, N.L. Public health: Quality of indoor residential air and health. Can. Med. Assoc. J. 2008, 179, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Kim, S.; Lee, J.H.; Kim, J.; Han, Y.; Kim, Y.M.; Kim, G.B.; Jung, K.; Cheong, H.K.; Ahn, K. Indoor air pollution aggravates symptoms of atopic dermatitis in children. PLoS ONE 2015, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Misztal, P.K.; Nazaroff, W.W.; Goldstein, A.H. Volatile Organic Compound Emissions from Humans Indoors. Environ. Sci. Technol. 2016, 50, 12686–12694. [Google Scholar] [CrossRef] [PubMed]
- Folinsbee, L.J. Human Health Effects of Air Pollution. Environ. Health Perspect. 1992, 100, 45–56. [Google Scholar] [CrossRef]
- English, J.S.C.; Dawe, R.S.; Ferguson, J. Environmental effects and skin disease. Br. Med. Bull. 2003, 68, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Freiman, A.; Bird, G.; Metelitsa, A.I.; Barankin, B.; Lauzon, G.J. Cutaneous effects of smoking. J. Cutan. Med. Surg. 2004, 8, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.E.; Cho, D.; Park, H.J. Air pollution and skin diseases: Adverse effects of airborne particulate matter on various skin diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Koohgoli, R.; Hudson, L.; Naidoo, K.; Wilkinson, S.; Chavan, B.; Birch-Machin, M.A. Bad air gets under your skin. Exp. Dermatol. 2017, 26, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.H.; Kennedy, E.A.; Kong, H.H. Topographical and physiological differences of the skin mycobiome in health and disease. Virulence 2017, 8, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Mancebo, S.E.; Wang, S.Q. Recognizing the impact of ambient air pollution on skin health. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 2326–2332. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, M.A.; Pham, D.M.; Boussouira, B.; Bernard, D.; Camus, C.; Nguyen, Q.L. Evaluation of the impact of urban pollution on the quality of skin: A multicentre study in Mexico. Int. J. Cosmet. Sci. 2015, 37, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Puri, P.; Nandar, S.; Kathuria, S.; Ramesh, V. Effects of air pollution on the skin: A review. Indian J. Dermatol. Venerol. Leprol. 2017, 83, 415–423. [Google Scholar] [CrossRef]
- Grice, E.A.; Kong, H.H.; Conlan, S.; Deming, C.B.; Davis, J.; Young, A.C.; Bouffard, G.G.; Blakesley, R.W.; Murray, P.R.; Green, E.D.; et al. Topographical and Temporal Diversity of the Human Skin Microbiome. Science 2009, 324, 1190–1192. [Google Scholar] [CrossRef] [PubMed]
- Grice, E.A.; Segre, J.A. The skin microbiome. Nat. Rev. Microbiol. 2011, 9, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Flores, G.E.; Caporaso, J.G.; Henley, J.B.; Rideout, J.R.; Domogala, D.; Chase, J.; Leff, J.W.; Vázquez-Baeza, Y.; Gonzalez, A.; Knight, R.; et al. Temporal variability is a personalized feature of the human microbiome. Genome Biol. 2014, 15, 531. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L.; Larcombe, D.-L.; Logan, A.C.; West, C.; Burks, W.; Caraballo, L.; Levin, M.; Van Etten, E.; Horwitz, P.; Kozyrskyj, A.; et al. The skin microbiome: Impact of modern environments on skin ecology, barrier integrity, and systemic immune programming. World Allergy Organ. J. 2017, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- He, Q.C.; Tavakkol, A.; Wietecha, K.; Begum-Gafur, R.; Ansari, S.A.; Polefka, T. Effects of environmentally realistic levels of ozone on stratum corneum function. Int. J. Cosmet. Sci. 2006, 28, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Valacchi, G.; Porada, E.; Rowe, B. Ambient ozone and bacterium Streptococcus: A link between cellulitis and pharyngitis. Int. J. Occup. Med. Environ. Health 2015, 28, 771–774. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Moyal, D.; Xiang, L.F.; Seité, S. Pollution and acne: Is there a link? Clin. Cosmet. Investig. Dermatol. 2017, 10, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.D.; Huang, B.-W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 2012, 24, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Ghio, A.J.; Carraway, M.S.; Madden, M.C. Composition of air pollution particles and oxidative stress in cells, tissues, and living systems. J. Toxicol. Environ. Heal. Part B Crit. Rev. 2012, 15, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Birch-Machin, M.A.; Bowman, A. Oxidative stress and ageing. Br. J. Dermatol. 2016, 175, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Thiele, J.J.; Traber, M.G.; Podda, M.; Tsang, K.; Cross, C.E.; Packer, L. Ozone depletes tocopherols and tocotrienols topically applied to murine skin. FEBS Lett. 1997, 401, 167–170. [Google Scholar] [CrossRef]
- Ahn, K. The role of air pollutants in atopic dermatitis. J. Allergy Clin. Immunol. 2014, 134, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Del Rosso, J.Q.; Levin, J. The clinical relevance of maintaining the functional integrity of the stratum corneum in both healthy and disease-affected skin. J. Clin. Aesthet. Dermatol. 2011, 4, 22–42. [Google Scholar] [PubMed]
- Isik, B.; Ceylan, A.; Isik, R. Oxidative stress in smokers and non-smokers. Inhal. Toxicol. 2007, 19, 767–769. [Google Scholar] [CrossRef] [PubMed]
- Esser, C.; Bargen, I.; Weighardt, H.; Haarmann-Stemmann, T.; Krutmann, J. Functions of the aryl hydrocarbon receptor in the skin. Semin. Immunopathol. 2013, 35, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Merches, K.; Haarmann-Stemmann, T.; Weighardt, H.; Krutmann, J.; Esser, C. AHR in the skin: From the mediator of chloracne to a therapeutic panacea? Curr. Opin. Toxicol. 2017, 2, 79–86. [Google Scholar] [CrossRef]
- Tauchi, M.; Hida, A.; Negishi, T.; Katsuoka, F.; Noda, S.; Mimura, J.; Hosoya, T.; Yanaka, A.; Aburatani, H.; Fujii-kuriyama, Y.; et al. Constitutive expression of aryl hydrocarbon receptor in keratinocytes causes in ammatory skin lesions. Society 2005, 25, 9360–9368. [Google Scholar] [CrossRef]
- Kim, H.O.; Kim, J.H.; Chung, B.Y.; Choi, M.G.; Park, C.W. Increased expression of the aryl hydrocarbon receptor in patients with chronic inflammatory skin diseases. Exp. Dermatol. 2014, 23, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Luecke, S.; Backlund, M.; Jux, B.; Esser, C.; Krutmann, J.; Rannug, A. The aryl hydrocarbon receptor (AHR), a novel regulator of human melanogenesis. Pigment Cell Melanoma Res. 2010, 23, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Morita, A.; Torii, K.; Maeda, A.; Yamaguchi, Y. Molecular Basis of Tobacco Smoke-Induced Premature Skin Aging. J. Investig. Dermatology Symp. Proc. 2009, 14, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Abel, J.; Haarmann-Stemmann, T. An introduction to the molecular basics of aryl hydrocarbon receptor biology. Biol. Chem. 2010, 391, 1235–1248. [Google Scholar] [CrossRef] [PubMed]
- Palermo, C.M.; Hernando, J.I.M.; Dertinger, S.D.; Kende, A.S.; Gasiewicz, T.A. Identification of Potential Aryl Hydrocarbon Receptor Antagonists in Green Tea. Chem. Res. Toxicol. 2003, 16, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Portugal-Cohen, M.; Soroka, Y.; Ma’or, Z.; Oron, M.; Zioni, T.; Brégégère, F.M.; Neuman, R.; Kohen, R.; Milner, Y. Protective effects of a cream containing Dead Sea minerals against UVB-induced stress in human skin. Exp. Dermatol. 2009, 18, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kang, Z.; Jiang, S.; Zhao, J.; Yan, S.; Xu, F.; Xu, J. Effects of ambient fine particles PM2.5 on human HaCaT cells. Int. J. Environ. Res. Public Health 2017, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Portugal-Cohen, M.; Oron, M.; Cohen, D.; Ma’or, Z. Antipollution skin protection—A new paradigm and its demonstration on two active compounds. Clin. Cosmet. Investig. Dermatol. 2017, 10, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, G.; Takahara, M.; Uchi, H.; Takeuchi, S.; Mitoma, C.; Moroi, Y.; Furue, M. An environmental contaminant, benzo(a)pyrene, induces oxidative stress-mediated interleukin-8 production in human keratinocytes via the aryl hydrocarbon receptor signaling pathway. J. Dermatol. Sci. 2011, 62, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Perin, F.; Blanché, C. Study design for anti-pollution claims. Personal Care Magazine, 16 May 2017; 23–25. [Google Scholar]
- Peterson, G.; Rapaka, S.; Koski, N.; Kearney, M.; Ortblad, K.; Tadlock, L. A robust sebum, oil, and particulate pollution model for assessing cleansing efficacy of human skin. Int. J. Cosmet. Sci. 2017, 39, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Schlay, S.; Slotta, U. Efficient skin protection against negative environmental influences by breathable vegan silk polypeptides. Sofw. J. 2016, 142, 14–19. [Google Scholar]
- Bielfeldt, S.; Boehling, A.; Springmann, G.; Wilhelm, K. Pollution Protection and the Skin—Testing Strategies. Househ. Pers. Care Today, 2016, 11, 81–84. [Google Scholar]
- Levannier, K.L.; Planel, E.; Gu, W.; Yu, X.J. Baicalin Protects against Cigarette Smoke. In Proceedings of the 29th Congress of the International Federation of Societies of Cosmetic Chemists, Orlando, FL, USA, 31 October–2 November 2016. [Google Scholar]
- Valacchi, G.; Sticozzi, C.; Belmonte, G.; Chen, N.; Krol, Y.; Oresajo, C. Vitamin C compound mixtures prevent ozone-induced oxidative damage in human keratinocytes as initial assessment of pollution protection. PLoS ONE 2015, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Facchini, G.; Pinheiro, A.L.T.A.; da Silva, M.S.; Bonner, M.Y.; Arbiser, J.; Eberlin, S. Honokiol protects skin cells against inflammation, collagenolysis, apoptosis, and senescence caused by cigarette smoke damage. Int. J. Dermatol. 2017, 56, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zheng, L.; Tuo, J.; Liu, Q.; Zhang, X.; Xu, Z.; Liu, S.; Sui, G. Analysis of PM2.5-induced cytotoxicity in human HaCaT cells based on a microfluidic system. Toxicol. In Vitro 2017, 43, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hubaux, R.; Weisgerber, F.; Salmon, M. In vitro assays to study the effects of air pollutants on skin: Exposure to urban dust and cigarette smoke extract. In Proceedings of the 23rd IFSCC Conference, Montreux, Switzerland, 21–23 September 2015. [Google Scholar]
- Gimenez, A.; Davi, C.; Canadas, E.; Alminana, N.; Delgado, R. Finding new solutions against pollution. Sofw. J. 2016, 142, 20–25. [Google Scholar]
- Iddamalgoda, A.; Biswas, K.B.; Tanaka, K.; Takayama, S. Protection against pollution-induced skin ageing by a natural cosmetic ingredient. Sofw. J. 2016, 142, 8–13. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rembiesa, J.; Ruzgas, T.; Engblom, J.; Holefors, A. The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims. Cosmetics 2018, 5, 4. https://doi.org/10.3390/cosmetics5010004
Rembiesa J, Ruzgas T, Engblom J, Holefors A. The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims. Cosmetics. 2018; 5(1):4. https://doi.org/10.3390/cosmetics5010004
Chicago/Turabian StyleRembiesa, Jadwiga, Tautgirdas Ruzgas, Johan Engblom, and Anna Holefors. 2018. "The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims" Cosmetics 5, no. 1: 4. https://doi.org/10.3390/cosmetics5010004
APA StyleRembiesa, J., Ruzgas, T., Engblom, J., & Holefors, A. (2018). The Impact of Pollution on Skin and Proper Efficacy Testing for Anti-Pollution Claims. Cosmetics, 5(1), 4. https://doi.org/10.3390/cosmetics5010004




