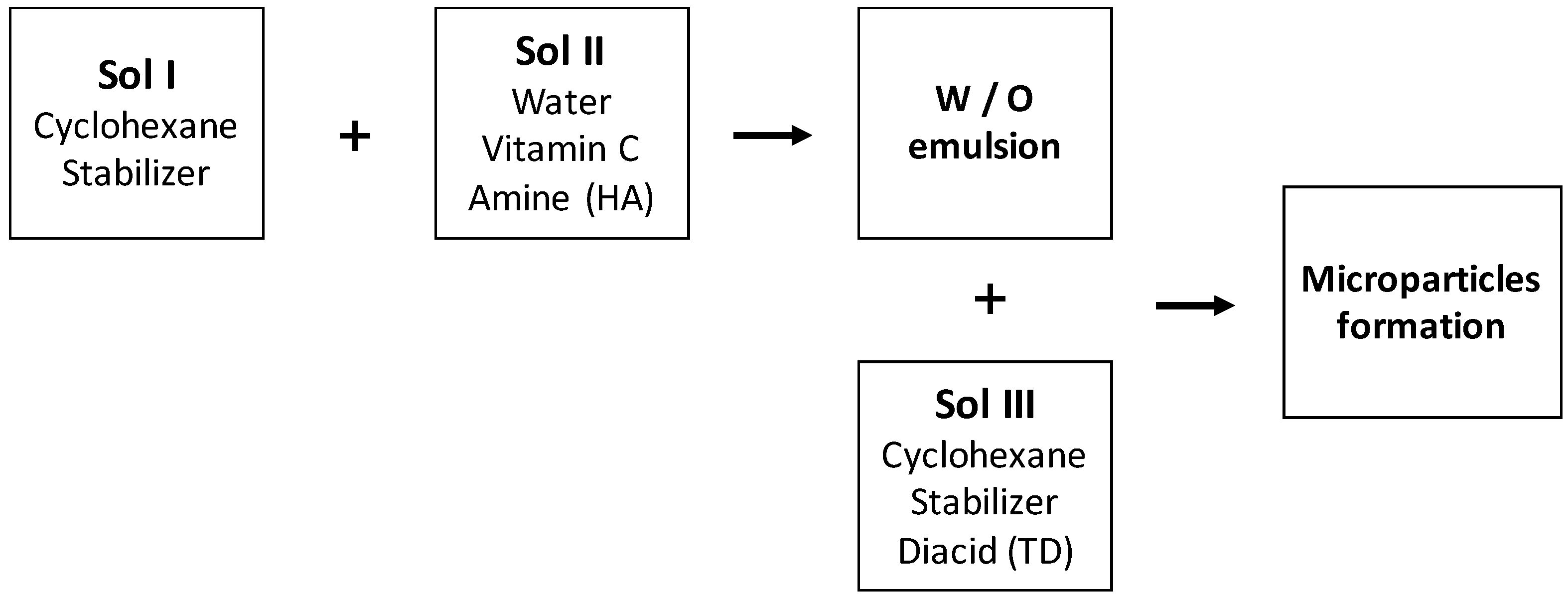
2.2. Preparation of Microparticles
The process of preparing microparticles is described on the
Figure 1. Briefly, three solutions were prepared separately:
Sol I: Organic phase containing 15 g/L of stabilizer (Arlacel 83);
Sol II: Aqueous phase containing 1,6-hexamethylenediamine (HA) and sodium hydroxide;
Sol III: Organic phase containing 15 g/L of stabiliser and terephtaloyl dichloride (TD).
Sol I and Sol II were mixed to form a water/oil (W/O) emulsion using a rotor stator stirrer (Ultra Turax IKA T18 basic). The stirring rate was 15,500 rpm for 10 min. Next, emulsion was transferred into a reactor vessel equipped with mechanical stirrer (280 rpm). Sol III was added, at different time, for the polycondensation reaction. After 3 h, the reaction was stopped by dilution with cyclohexane.
Particles were separated from cyclohexane using a centrifuge at 4000 rpm for 20 min and were washed three times with water.
It is important to note that amine group/hydroxide group ratio in Sol II should be 1:1 to neutralise the HCl liberated during the reaction. Furthermore, polyamide microparticles were prepared with a constant molar ratio diacid group/amine group at 3:1 [
41] in order to limit the residual amine monomers in the particles. Excess was removed by washing the capsules. A series of experimentations was conducted using DoE describe below (
Section 2.3).
2.3. Design of Experiment (DoE)
Previous works [
26,
32,
42] show that different parameters have important impacts on particle morphology (size, structure, porosity) and on encapsulation efficiency. To improve the process and have a better understanding of this phenomenon, a DoE (design of experiments) was carried out, with the goal to obtain particles with appropriate properties [
37,
40].
Design of experiments helps to test as many factors having main effects with a minimal number of tests (often). Among the different experimental designs, factorial designs are common because they are the simplest to implement and enable the very quick identification of interactions between factors. The basic principle is to assign to each factor (normalised) its lowest value (−1) and its highest value (+1). Thus, for k factors, we are left with a set of 2k possible values.
The first step is to take an interest in the responses and their interactions. There are currently different screening methods adapted to this problem, as the experiences of matrices by Plackett and Burman, full factorial design, the experience supersaturated matrices, screening in groups, bifurcation sequential, and so on.
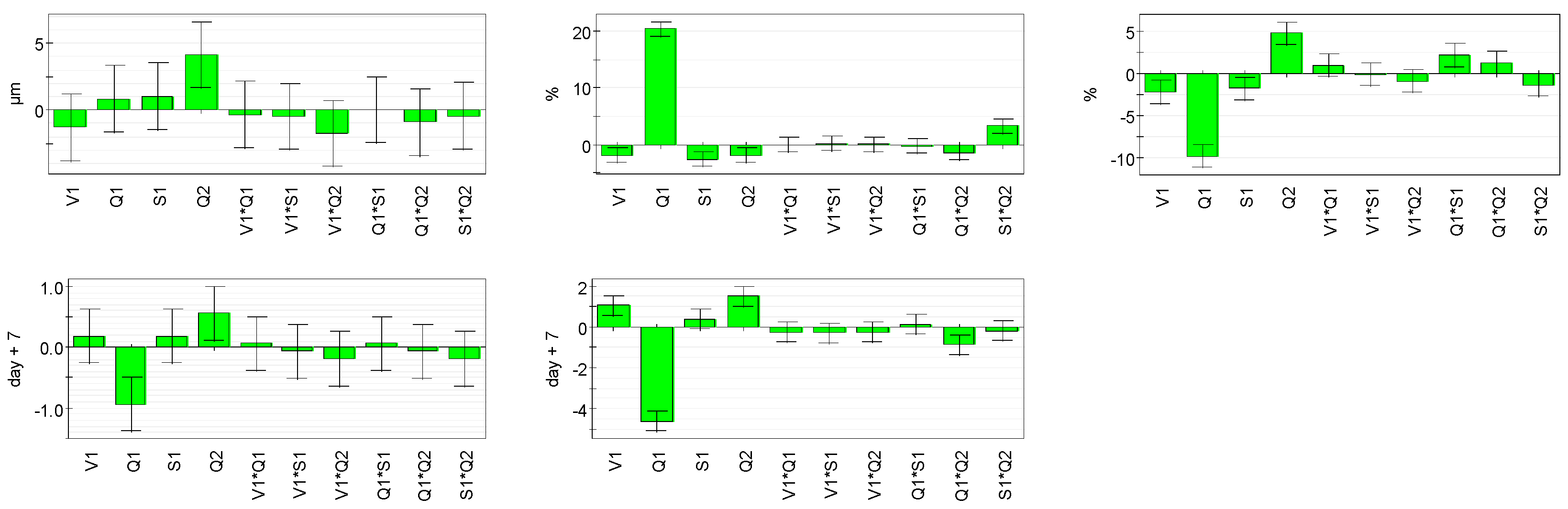
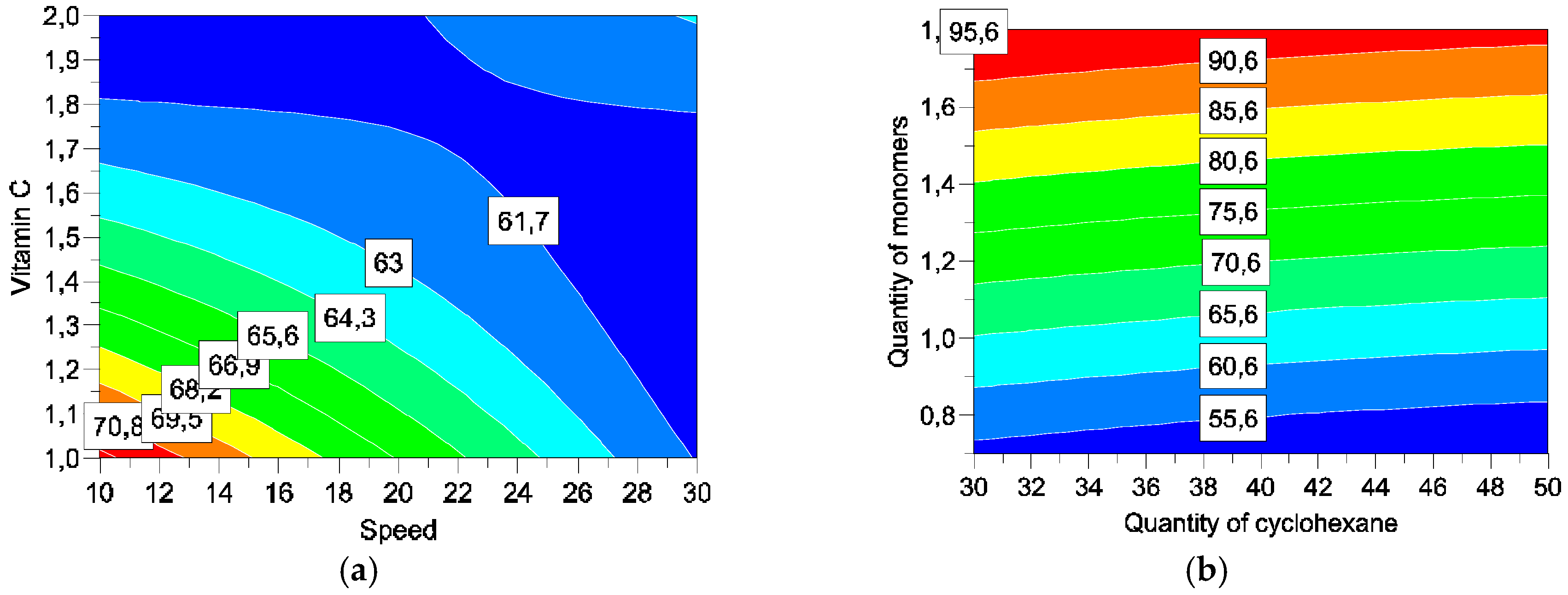
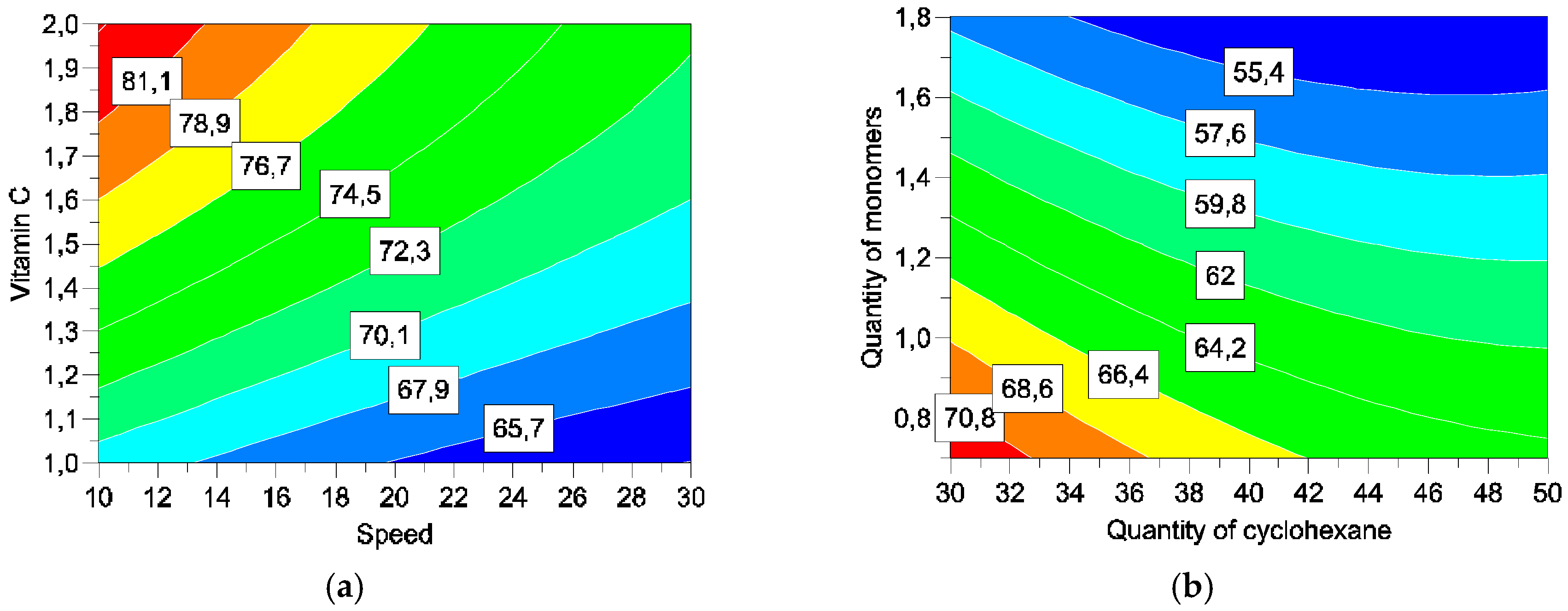
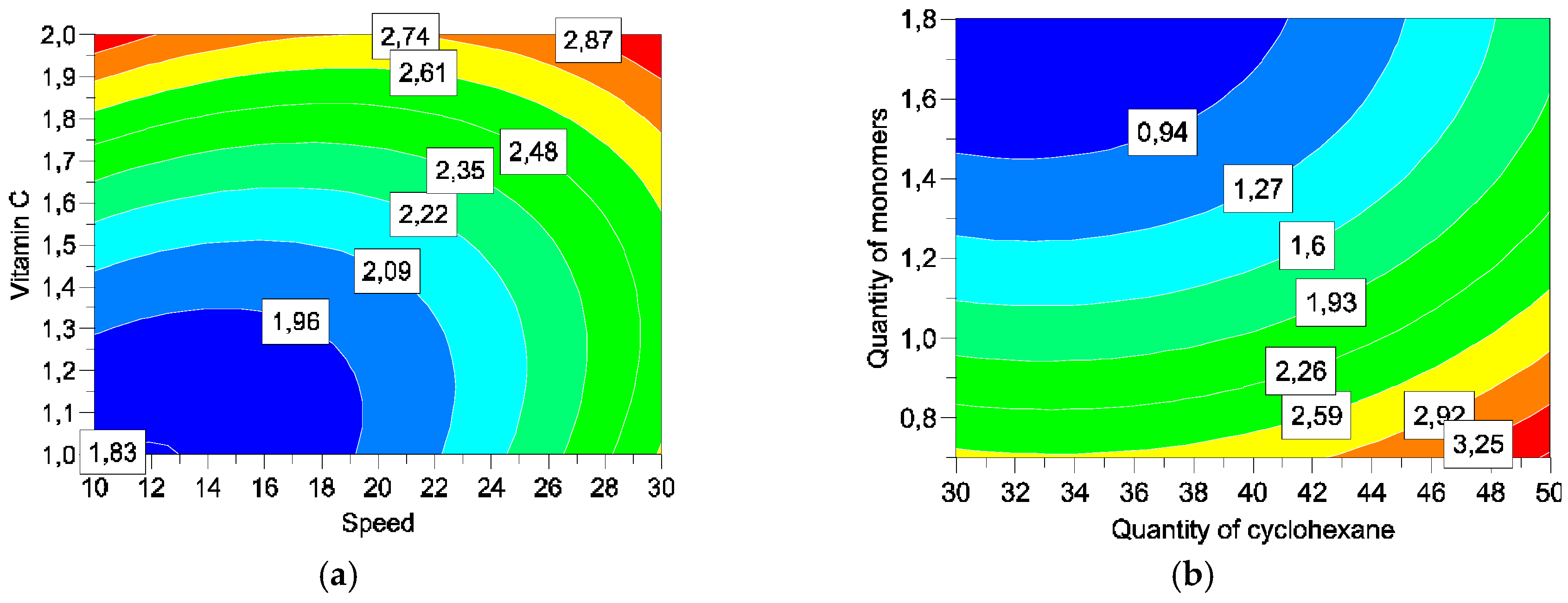
Four parameters were studied: concentrations of cyclohexane (
X1), monomers (
X2), and vitamin C (
X4), and speed of addition of solution III into the emulsion (
X3) (
Table 1).
Five responses were studied: particles size, encapsulation efficiency, encapsulation rates, vitamin C degradation, and vitamin C liberation. With such matrices/variables/parameters, a full factorial matrix was chosen to estimate the effects of the parameters and their interactions [
38]. Yates algorithm was used for the matrix construction [
43]. To have some statistics, 3 points were repeated in the middle of the experimental domain. The mathematical model calculated is a linear model:
where
is the average of the responses for all experiments,
are coefficients of the factors
X1,
X2, ...,
Xn, and
is the interaction between
n factors.
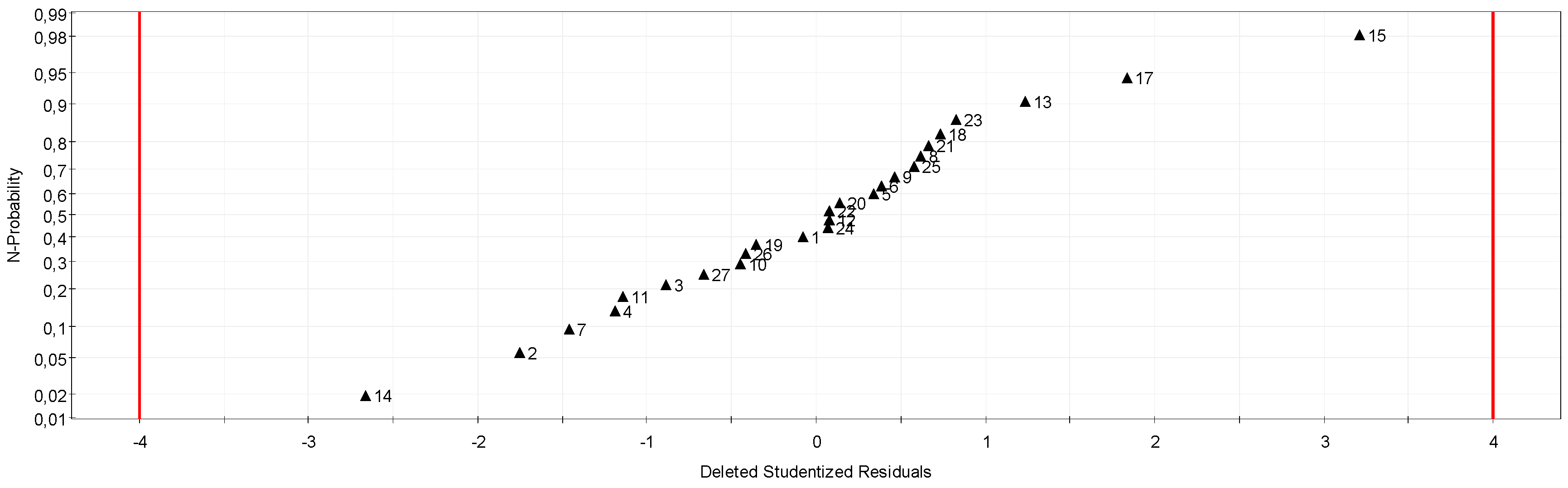
By solving the equation system, the model coefficients were obtained and, therefore, the equation of the model itself. To evaluate the explanatory power of this model, several statistical techniques were used.
● Variance analysis
The purpose of this analysis is to decompose the sum of squared deviations from the mean of the responses (
TSS) measured in two amounts: the sum of the deviations from the average of the calculated responses (
W) and the residual sum of squares (
RSS) [
44].
● Lack of fit
This section examines the variances of two terms of the residue: the fit and experimental error. The adjustment is the difference between the selected model and the real model that we do not know: lack of fit. The experimental error is measured by the standard deviation of several repetitions made under the same conditions: pure error.
● Summary of fit
R2 is the coefficient of determination. It is defined as the ratio of the sum of squared deviations from the mean of the calculated responses divided by the sum of squared deviations from the mean of the measured responses.
where
is the measured response,
is the calculated response, and
is the mean of the responses.
If the model is simply average, the numerator is null as well as the coefficient of determination R2. If the model completely explains the measured responses, the numerator equals the denominator and R2 coefficient is 1. The closer R2 is to 1, the better the model explains the measured responses.
R2-adjusted is a statistical value whose objective is to compare various mathematical models; it considers the number of coefficient in the model
where
n is the number of experiments.
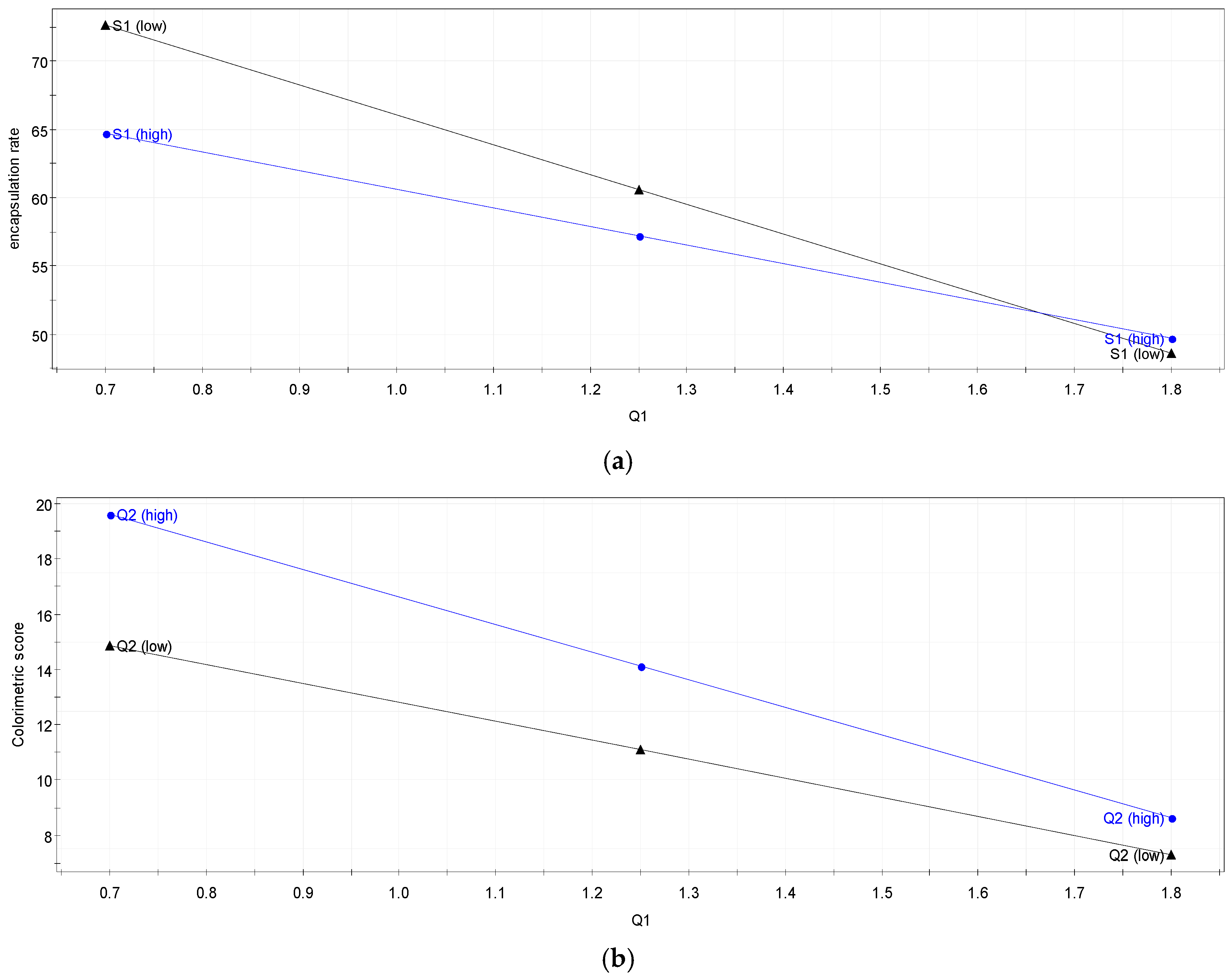
Q2 is a measure of how well the model will predict the responses, and is expressed in the same unit as R2. For each response, Q2 is computed and represents the percent variation of Y that is predictive. Q2 value of 0.6 or larger indicates that the model has good predictive ability and will have small prediction error.
The plans examined previously had only two levels of study factor, and mathematical models used were of the first degree with respect to each factor. These plans were used to allow factor screening and sometimes led to simple but adequate modelling. Yet, the modelling must go through mathematical models of the second degree to be improved. We used the plans for response surfaces. Here we will discuss a composite design. The response surface model (RSM) was used to optimise critical parameters of this formulation (amount of solvent, concentration of monomers, quantity of vitamin C). One of the main objectives of this model is the determination of the optimum settings of the control variables. Having a good-fitting model that provides an adequate representation of the mean response leads to a maximum or minimum response over a certain region of interest. The mathematical model applied to the plans used for response surface model is a second-degree model with two-way interactions; below is the global equation to solve the problem:
where
is the average of the responses for all experiments;
is the coefficient of the factors
X1,
X2, ...,
Xn;
is the interaction between factors
m and
n; and
is the curvature of the response.
Twenty-seven experiments were performed according to the surface response models method (Equation (5)) with a complete factorial matrix (16 experiments) (
Table 3), 8 points on the axis and 3 points in domain’s centre.
All regressions were analysed by Modde software. The regression quality was evaluated by the same statistical indicator.
After verifying the validity of the model by classical statistical tests [
45], the vitamin C encapsulation could be predicted in each point of the defined domain. Analysis of variance (ANOVA) was performed to determine the significance of the fitted equation. The multiple correlation coefficient statistic (
R2) revealed the part of the variation of
Y1 explained by the model.
2.4. Particles’ Characterisation
Formulated microparticles are characterised by the mean particle size and morphology, encapsulation efficiency, and the release of vitamin C. These characterisations are used as an answer for the screening design.
2.4.1. Particle Size and Morphology (Y1)
Particle sizes were measured by laser scattering (Malvern Mastersizer 3000, Malvern, Malvern, UK) and were characterised by their volume mean diameter (D43). Particle morphology was studied by scanning electron microscopy (SEM) (Hitachi S800 Microscope, Hitachi, Schaumburg, IL, USA).
2.4.2. Encapsulation Efficiency (Y2) and Yield (Y3)
Prior to HPLC analysis, particles were dispersed in water and broken using a rotor stator stirrer at 24,000 rpm for 10 min.
Figure 2 shows polyamide microparticles before and after treatment.
Vitamin C analysis was conducted by HPLC using an Agilent 1260 Infinity LC (Agilent Technologies, Saint Laurant, QC, Canada) with a UV detector and Phenomenex Synergi 4 µm hydro RP 80 Å column (150 mm × 4.6 mm ×4 µm). The following parameters were used:
Encapsulation efficiency is defined as the ratio between the quantities of vitamin C experimentally measured (
) in the capsules and the quantity of vitamin C initially introduced (
):
Encapsulation yield is defined as the ratio between vitamin C quantities experimentally measured in the capsules and the capsules’ weight (weight of polymers (
) and vitamin C (
)):
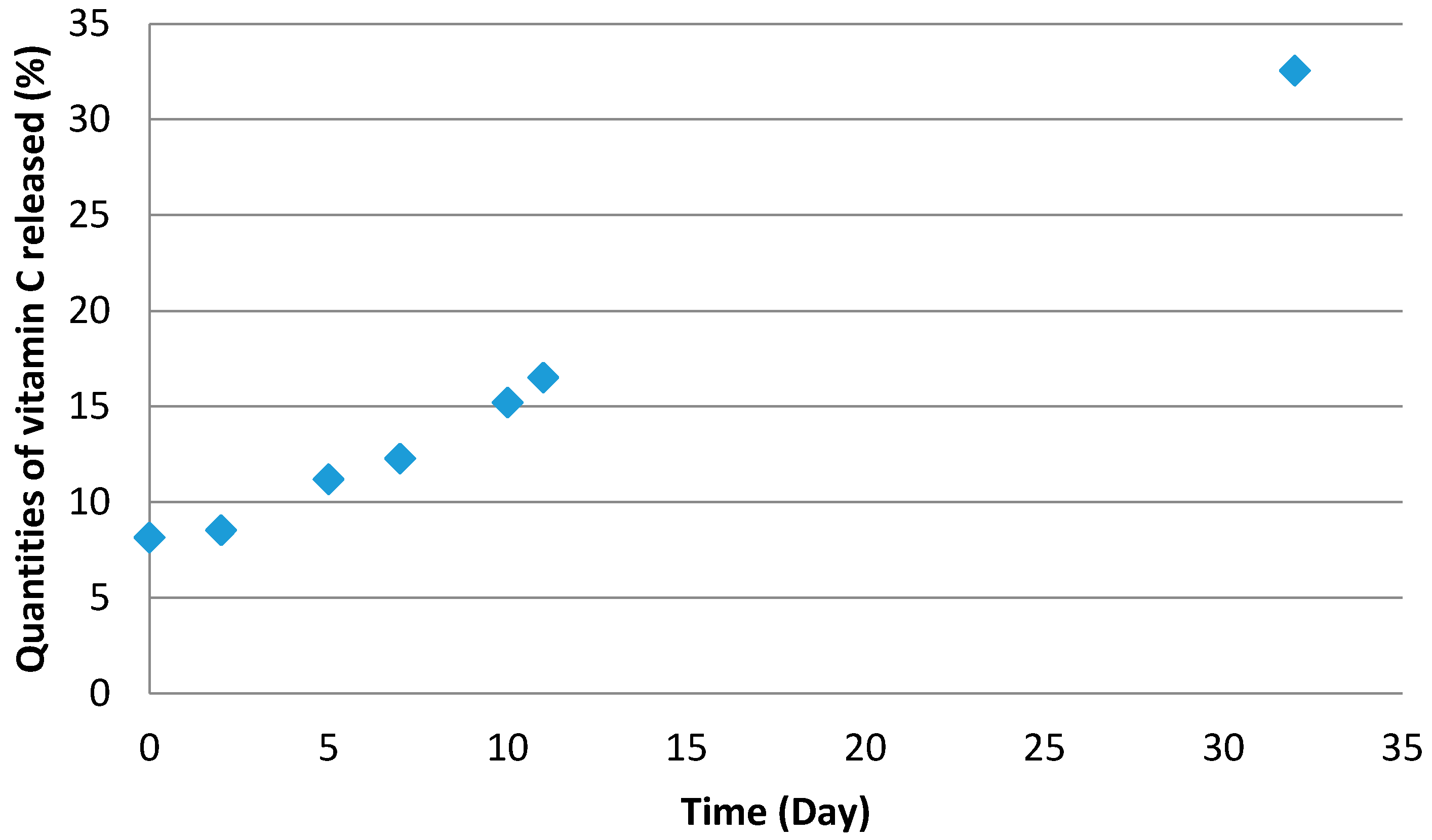
2.4.3. Release of Vitamin C (Y4)
Vitamin C release was measured by HPLC with the following procedure:
Capsules were dispersed in water and stored away from light. Several samples were taken after 1, 3, 7, 15, and 30 days. Each sample was analysed by HPLC following the previous method.
The results are given in percentage of liberate active ingredient as a function of the time.
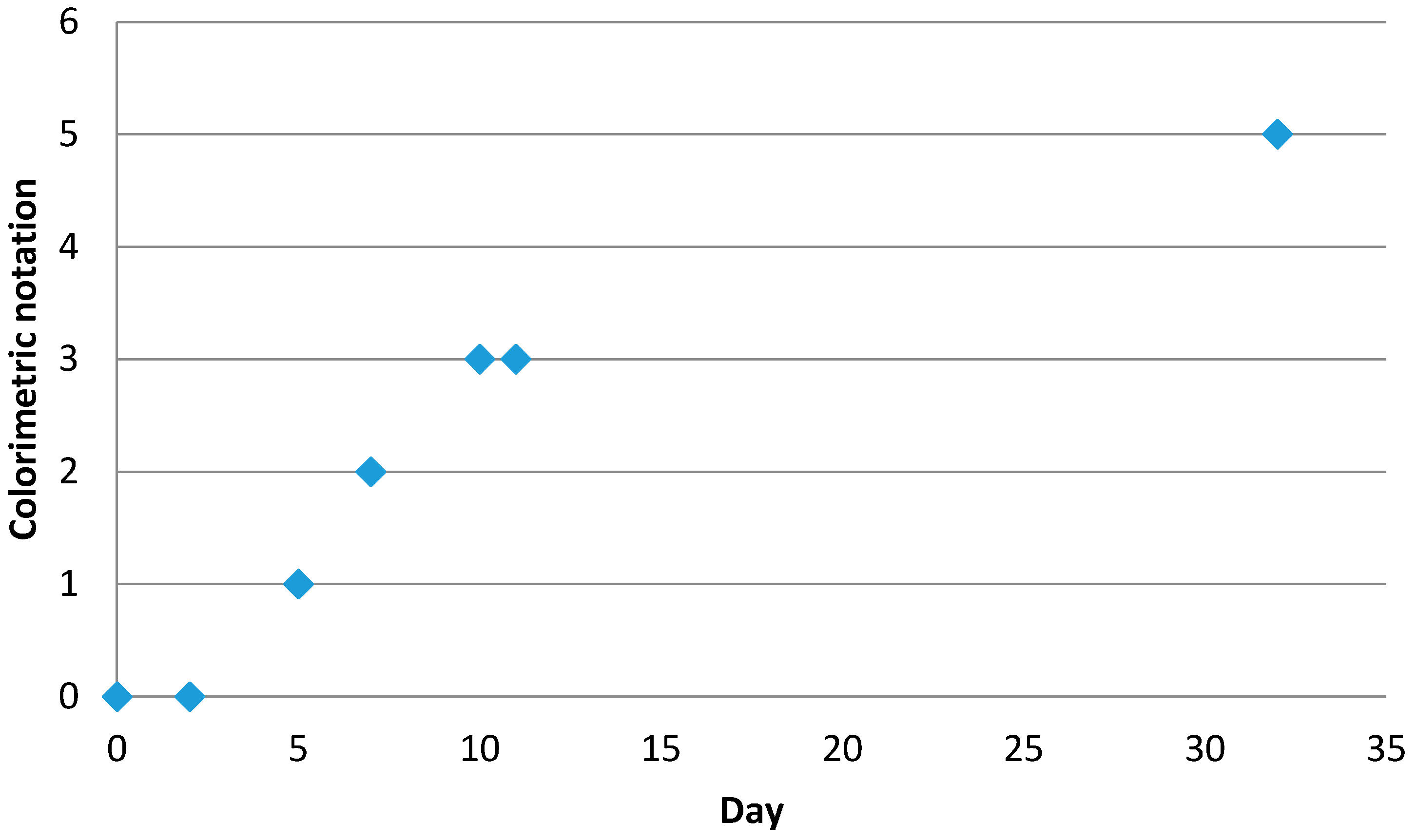
2.4.4. Colourimetric Score (Y5)
Capsules were dispersed in water and stored in light. The colour of each sample was observed at different times (1, 3, 7, 15, and 30 days). The solution turned brown due to vitamin C degradation through the Maillard reaction. A score between 1 (white solution with no degradation) and 10 (dark brown solution) was assigned to all solutions.