Topical Retinol Restores Type I Collagen Production in Photoaged Forearm Skin within Four Weeks
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of ROL Lotion
2.2. Topical ROL Treatment and Skin Tissue Procurement
2.3. RNA Extraction and Real-Time RT-PCR
2.4. Immunofluorescent Staining
2.5. proCOL1 Enzyme Immunoassay (EIA)
2.6. Assessment of Skin Erythema
2.7. Statistical Analysis
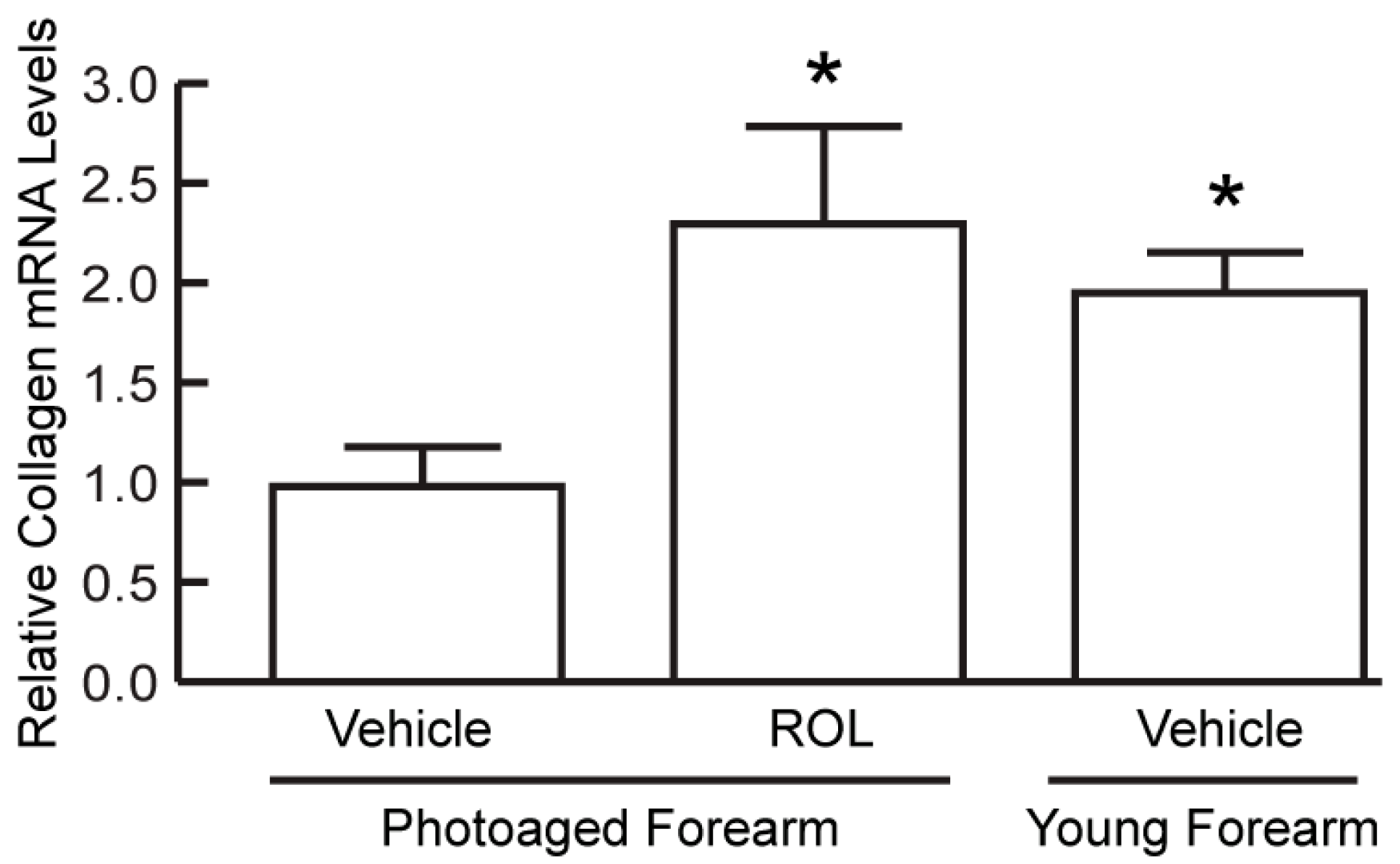
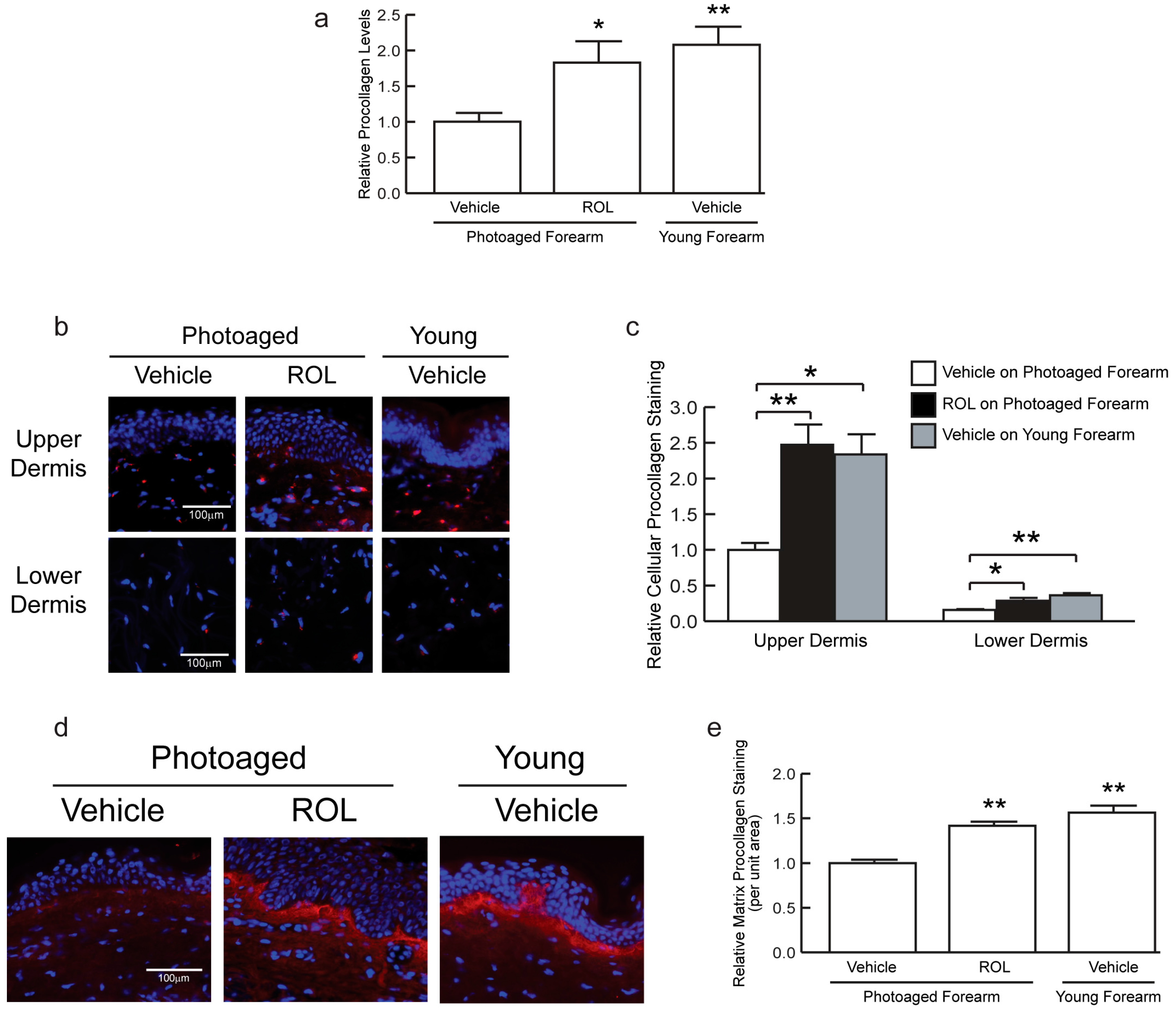
3. Results
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rittie, L.; Fisher, G. Natural and sun-induced aging of human skin. Cold Spring Harb. Perspect. Med. 2015, 5, a105370. [Google Scholar] [CrossRef] [PubMed]
- Yaar, M.; Gilchrest, B.A. Skin aging: Postulated mechanisms and consequent changes in structure and function. Clin. Geriatr. Med. 2001, 17, 617–630. [Google Scholar] [CrossRef]
- Canty, E.G.; Kadler, K.E. Procollagen trafficking, processing and fibrillogenesis. J. Cell Sci. 2005, 118, 1341–1353. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lei, D.; Swindell, W.R.; Xia, W.; Weng, S.; Fu, J.; Worthen, C.A.; Okubo, T.; Johnston, A.; Gudjonsson, J.E.; et al. Age-associated increase in skin fibroblast-derived prostaglandin E2 contributes to reduced collagen levels in elderly human skin. J. Investig. Dermatol. 2015, 135, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Wang, F.; Shao, Y.; Rittie, L.; Xia, W.; Orringer, J.S.; Voorhees, J.J.; Fisher, G.J. Enhancing structural support of the dermal microenvironment activates fibroblasts, endothelial cells, and keratinocytes in aged human skin in vivo. J. Investig. Dermatol. 2013, 133, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.H.; Seo, J.Y.; Choi, H.R.; Lee, M.K.; Youn, C.S.; Rhie, G.; Cho, K.H.; Kim, K.H.; Park, K.C.; Eun, H.C. Modulation of skin collagen metabolism in aged and photoaged human skin in vivo. J. Investig. Dermatol. 2001, 117, 1218–1224. [Google Scholar] [CrossRef] [PubMed]
- Geesin, J.C.; Darr, D.; Kaufman, R.; Murad, S.; Pinnell, S.R. Ascorbic acid specifically increases type I and type III procollagen messenger RNA levels in human skin fibroblast. J. Investig. Dermatol. 1988, 90, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Orringer, J.S.; Sachs, D.L.; Shao, Y.; Hammerberg, C.; Cui, Y.; Voorhees, J.J.; Fisher, G.J. Direct quantitative comparison of molecular responses in photodamaged human skin to fractionated and fully ablative carbon dioxide laser resurfacing. Dermatol. Surg. 2012, 38, 1668–1677. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Varani, J.; Voorhees, J.J. Looking older: Fibroblast collapse and therapeutic implications. Arch. Dermatol. 2008, 144, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Orringer, J.S.; Rittie, L.; Hamilton, T.; Karimipour, D.J.; Voorhees, J.J.; Fisher, G.J. Intraepidermal erbium:Yag laser resurfacing: Impact on the dermal matrix. J. Am. Acad. Dermatol. 2011, 64, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Voorhees, J.J. Molecular mechanisms of retinoid actions in skin. FASEB J. 1996, 10, 1002–1013. [Google Scholar] [PubMed]
- Darlenski, R.; Surber, C.; Fluhr, J.W. Topical retinoids in the management of photodamaged skin: From theory to evidence-based practical approach. Br. J. Dermatol. 2010, 163, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, B.A.; Unger, J.G.; Rohrich, R.J. Reversal of skin aging with topical retinoids. Plast. Reconstr. Surg. 2014, 133, 481e–490e. [Google Scholar] [CrossRef] [PubMed]
- Kafi, R.; Kwak, H.S.; Schumacher, W.E.; Cho, S.; Hanft, V.N.; Hamilton, T.A.; King, A.L.; Neal, J.D.; Varani, J.; Fisher, G.J.; et al. Improvement of naturally aged skin with vitamin a (retinol). Arch. Dermatol. 2007, 143, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Fisher, G.J.; Voorhees, J.J. Photoaging and topical tretinoin: Therapy, pathogenesis, and prevention. Arch. Dermatol. 1997, 133, 1280–1284. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Ahn, B.N.; Kong, C.S.; Park, S.H.; Park, B.J.; Kim, S.K. Antiphotoaging effect of chitooligosaccharides on human dermal fibroblasts. Photodermatol. Photoimmunol. Photomed. 2012, 28, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.R.; Dixon, T.K.; Bhattacharyya, T.K. Effects of topicals on the aging skin process. Facial Plast. Surg. Clin. N. Am. 2013, 21, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.E.; Craven, N.M.; Kang, S.; Jones, C.J.; Kielty, C.M.; Griffiths, C.E. A short-term screening protocol, using fibrillin-1 as a reporter molecule, for photoaging repair agents. J. Investig. Dermatol. 2001, 116, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.E.; Ogden, S.; Cotterell, L.F.; Bowden, J.J.; Bastrilles, J.Y.; Long, S.P.; Griffiths, C.E. Effects of a cosmetic ‘anti-ageing’ product improves photoaged skin [corrected]. Br. J. Dermatol. 2009, 161, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Bergstrom, K.G. Beyond tretinoin: Cosmeceuticals for aging skin. J. Drugs Dermatol. 2009, 8, 674–677. [Google Scholar] [PubMed]
- Kang, S.; Duell, E.A.; Fisher, G.J.; Datta, S.C.; Wang, Z.Q.; Reddy, A.P.; Tavakkol, A.; Yi, J.Y.; Griffiths, C.E.; Elder, J.T.; et al. Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoic acid but without measurable retinoic acid levels or irritation. J. Investig. Dermatol. 1995, 105, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.E.; Kang, S.; Ellis, C.N.; Kim, K.J.; Finkel, L.J.; Ortiz-Ferrer, L.C.; White, G.M.; Hamilton, T.A.; Voorhees, J.J. Two concentrations of topical tretinoin (retinoic acid) cause similar improvement of photoaging but different degrees of irritation. A double-blind, vehicle-controlled comparison of 0.1% and 0.025% tretinoin creams. Arch. Dermatol. 1995, 131, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, M.; Wang, P.; Sachs, D.; Xu, Y.; Xu, Y.; Voorhees, J.J.; Fisher, G.J.; Li, Y. Topical Retinol Restores Type I Collagen Production in Photoaged Forearm Skin within Four Weeks. Cosmetics 2016, 3, 35. https://doi.org/10.3390/cosmetics3040035
Sun M, Wang P, Sachs D, Xu Y, Xu Y, Voorhees JJ, Fisher GJ, Li Y. Topical Retinol Restores Type I Collagen Production in Photoaged Forearm Skin within Four Weeks. Cosmetics. 2016; 3(4):35. https://doi.org/10.3390/cosmetics3040035
Chicago/Turabian StyleSun, Min, Peiru Wang, Dana Sachs, Yang Xu, Yiru Xu, John J. Voorhees, Gary J. Fisher, and Yong Li. 2016. "Topical Retinol Restores Type I Collagen Production in Photoaged Forearm Skin within Four Weeks" Cosmetics 3, no. 4: 35. https://doi.org/10.3390/cosmetics3040035
APA StyleSun, M., Wang, P., Sachs, D., Xu, Y., Xu, Y., Voorhees, J. J., Fisher, G. J., & Li, Y. (2016). Topical Retinol Restores Type I Collagen Production in Photoaged Forearm Skin within Four Weeks. Cosmetics, 3(4), 35. https://doi.org/10.3390/cosmetics3040035




