Abstract
For some time, Brazil has been appointed as one of the greatest consumers of cosmetic products in the world. Although cosmetics may seem harmless, destined exclusively to enhance personal appearance or to clean and protect the skin, hair and nails, new studies and events are highlighting the need to evaluate the safety of such products. The present work interrelated the lifecycle of a cosmetic product with the safety trials and tests applicable to some cycle phases. From this information, a survey was made of accredited Conformity Assessment Bodies (CAB) and test facilities recognized by the General Coordination for Accreditation (CGCRE) which are competent respectively to carry out safety trials and tests of cosmetics. Twenty five competent laboratories were identified to carry out chemical and/or biological trials of cosmetics, according to the legislation ABNT ISO IEC 17025:2005, and 10 test facilities recognized by the Compliance Monitoring Program that can carry out tests of the development of a product for register purposes, aiming at human health and safety. It is interesting to notice that Brazil has accredited laboratories to carry out trials that are critical for the health of the population, such as the levels of heavy metals and the presence of pathogens. On the other hand, CGCRE does not have a program to recognize safety clinical trials. The importance of this kind of study is understood, considering the world history of adverse reactions and the great consumption of cosmetics in the country.
1. Introduction
1.1. Brief History of the Use of Cosmetic Products
Since the beginning of civilization, cosmetics have been included in daily body care, not only by the upper class of society, but also by the middle and lower classes [1].
There are registries from Ancient Egypt, Greece and the Roman Empire that women applied a reddish mineral or plant pigment on their cheeks and lips for embellishment. Thereby, lip cosmetics might have been around for thousands of years [2].
Papyrus, over 30 centuries, old revealed recipes destined to promote skin care and improve its complexion. These recipes announced the appearance of modern cosmetology.
Later on, doctors, philosophers and Greek and Roman poets, such as Hippocrates and Horace, showed especial interest in the ingredients destined to blanch or color the face [3].
During the 19th century, the area of cosmetics and pharmaceuticals greatly evolved due to the Industrial Revolution and the emergence of new technologies. During this period, methods for the elimination of wrinkles and the embellishment of the face emerged for the first time. The increasing search for beauty promoted the research of new cosmetic products, and with the beginning of the Industrial Era and the consequent increase of manufactured products, publicity campaigns were improved and changed from being simply informative to persuasive and aggressive, leading consumers to buy [4].
In 1984, Kligman [5], during a conference of the Chemical Cosmetic Society, used for the first time the term “cosmeceutical”, a reference to the products applied topically on the skin that were able to change its conditions. However, they are not considered drugs, focusing on the treatment of diseases, nor cosmetics, with the only goal of embellishing or improving the appearance of the skin [5,6]. That is, cosmeceuticals represent the “marriage” between these two products: cosmetics and pharmaceuticals.
Despite the development of new items for personal care and embellishment and also the diversity of the applications of cosmetic products (anti-wrinkle, cellulite reducer, beauty balm or blemish balm (BB) and color control or color-correcting (CC) creams, tanners, sunscreens, etc.), which can be classified as cosmeceuticals, in this work, the denominations and classifications according to the Brazilian Health Surveillance Agency (ANVISA), the organization body responsible for the regulation in this area, were adopted.
According to the ANVISA Directors’ Collegiate Resolution (RDC) No 7 of 10 February 2015 [7], products for personal hygiene, cosmetics and perfumes are preparations composed of natural or synthetic substances, for external use on many parts of the human body, skin, hair and scalp, nails, lips, external genitalia, teeth and oral mucosa, with the exclusive or main goal of cleaning, scenting, altering their appearance and/or correcting body odors and preventing them.
Because of regulatory issues, these products need to prove, at least, their safety in order to request an authorization for commercialization.
BB and CC creams are marketing terms coined that are used by some brands to mean beauty balm or blemish balm cream and color control or color-correcting cream, respectively.
1.2. The Brazilian Cosmetics Scenery
Nowadays, a huge diversity of cosmetic products can be noticed, not only for the usual issues of hygiene and personal care, such as soaps, shampoos, conditioners, deodorants and perfumes, but also those that improve the appearance of the consumer, such as anti-wrinkle and dark circles, skin blanchers, improvers of facial elasticity, sunscreens and hair dyes, among others.
The search for beauty can be considered global, especially among women, and Brazil is under the spotlight as one of the countries that consumes cosmetic products the most.
According to data from ABIHPEC (Brazilian Association of Personal Hygiene, Perfume and Cosmetics), Brazil is the third largest market of cosmetics consumption, only behind the USA and China, and moving away from Japan, in the fourth position. With earnings of R$101.7 billion (approximately US$25.5 billions), this sector/branch had nominal growth of 11% in 2014, when compared to the R$91.9 billion (approximately US$23 billion) in 2013 [8].
These earnings can be separated by different product categories, and the segments of perfumes and deodorants are considered groundbreakers [8].
It is also important to highlight that the country has a strong representation for the sunscreen category. More and more, the population is becoming aware of the risks of sun exposure, contributing to a strong internal market of R$4.1 billion (approximately US$1 billion). As per the current President of ABIHPEC, João Carlos Basilio, “although Brazil was already a leader in Sun protection, last year (2014) we expanded our leadership” [8]. Mintel, the world’s leading market intelligence agency, estimates that the category will have grown 15% in 2015 [8].
Besides sunscreens, it is important to highlight in this work another significant segment in Brazil: children’s products. In this category, Brazil is the second largest consumer of the world. Over the last five years, the segment of children products had an average growth of 14%, reaching the earnings of R$4.5 billion (approximately US$1.1 billion) in 2014. Brazil is a leader of consuming children’s hair products, representing 24% of world consumption [8].
Due to the outbreak of dengue, chikungunya and zika that is currently happening in the country, diseases transmitted by the mosquito Aedes, the consumption of repellents skyrocketed. The earnings of the segment grew 50% when compared to last year, according to data from Nielsen Consultancy and published in the Brazilian newspaper “O Globo”. Sales reached R$217 million (approximately US$54.3 million), the biggest number in the history of the category [9].
1.3. A Brief History of Adverse Reactions to Cosmetics
Despite that, historically, cosmetics have been considered harmless and very popular since ancient times, two serious episodes that occurred in the USA in the 1930s drew attention to the importance of regulation for this kind of product in order to protect the health of the population [10].
The first episode was about the unwanted effects caused by the use of an eye mascara called Lash-Lure. According to the FDA (Food and Drug Administration), the product caused blindness in at least one woman and serious side effects in many others, such as ocular irritation and ulceration, when used to color the eyelashes and eyebrows during the 1920s and 1930s [11]. The technical report that shows the view of COLIPA (The European Cosmetic, Toiletry and Perfumery Association) shows that p-phenylenediamine (PPD), the ingredient present in Lash-Lure, is known as an extreme skin sensitizer, but not as a severe eye-damaging chemical that can cause blindness [12].
The second episode that led to serious consequences was the use by a large number of women of a depilatory cream called Koremlu Cream, which promised the definite removal of body hair. This product was formulated with thallium (a substance used in rat poison), and it caused hair loss, myalgia, arthralgia and neuritis in many consumers [10]. There were even reports of death. These episodes and other dramatic events with other types of products made the American society demand more rigid legislation, in order to protect the health of the population. This movement was the initial step for the establishment of the world’s best known health surveillance organization body, the FDA [10].
Later, in 1960, there was an outbreak in England caused by an ingredient present in soaps. It is estimated that 10,000 people experienced acute photo-dermatitis due to the presence of halogenated salicylanilides in soaps.
Between the 1950s and 1960s, also an inflammatory allergic reaction occurred in consumers in Europe and USA who used deodorants with zirconium.
In 1972, an incident in France occurred that caused great commotion. By accident, hexachlorophene was added to some batches of baby talcum powder. It caused intoxication symptoms, and high serum levels of this substance were observed. More than 204 babies were ill, and 36 died of respiratory failure.
At the end of the 1970s, there were many cases of contact photo-dermatitis related to synthetic fragrances present in perfumes, 6-methylcoumarin and musk ambrette.
At the end of the 1990s, there were many reports of adverse reactions due to henna temporary tattoos. Frequently, these products contain, besides henna, PPD, the same pigment used in Lash Lure mascara [10].
In January of 2016, the first case of erythema multiforme probably caused by octocrylene was reported [13]. This substance absorbs UVA and UVB radiation, and it is widely used in sunscreens.
Furthermore, in January of 2016, due to the wide use of repellents to prevent dengue, zika and chikungunya in Brazil, there were many cases of allergic reactions of consumers [14].
This entire context shows the importance of ensuring the safety of a cosmetic product when it is accessible to the population.
Furthermore, it is interesting to note that most of these ingredients are listed in the list of substances that are prohibited in the formulation of cosmetics in Brazil [15] and European Union countries [16]. Therefore, it is important to cross-check such lists before electing an ingredient in the formulation of a cosmetic product.
1.4. INMETRO’s Function in Ensuring Product Safety
The National Institute of Metrology, Quality and Technology (INMETRO) is a federal autarchy, with the mission of “ensuring reliability to the Brazilian society in measurements and products, through metrology and conformity assessment, promoting harmonization of consumer relations, innovation and competitiveness in the country” [17]. Among the duties and attributions of INMETRO are the accreditation activities, which are structured by CGCRE (General Coordination for Accreditation), the organization body competent to act as Accreditation Body for the Conformity Assessment Body (CAB) that is recognized by the Brazilian Government [18].
It is worth pointing out that the laboratory accreditation made by CGCRE is performed by the Laboratory Accreditation Division (DICLA), which acts on the following most important modalities: (i) laboratory accreditation of trials according to norm ABNT NBR ISO/IEC 17025:2005 [19]; and (ii) recognition of conformity to the principles of Good Laboratory Practice (GLP), according to the requirements established in NIT-Dicla-035: Principles of Good Laboratory Practice [20].
The accreditation is an attestation carried out by a third party related to a CAB (Conformity Assessment Body), which can be a laboratory, for instance, expressing a formal demonstration of its competence to carry out specific chores of conformity assessment [21].
The recognition of the principles of good practice are applicable to the test facilities (main unit where the studies are conducted, which can be labs, field installations or conservatories) and test units (unit where one or more phases of a study are carried out, which can be labs, field installations or conservatories) and constitute the recognition of the adherence levels of the test installation in relation to the good conduct principles.
The good conduct principles are composed by a quality system that covers the organizational process and the conditions in which animal, in vitro and environment safety studies are planned, developed, monitored, registered, filed and reported. They are constituted in tests demanded by regulatory authorities to evaluate and register cosmetic products, for instance, among others, aiming at the evaluation of risk to human health and the environment [20].
This means that every laboratory accredited by CGCRE is competent to carry out determined product trials, as well as the test facilities are recognized to carry out the determined tests. CGCRE evaluates CABs and has a Compliance Monitoring Program of Good Practice Principles in order to check the compliance of obligatory requirements in laboratories and test facilities.
Both accreditation and recognition add values of the product. If the accredited trials and recognized tests directly impact the safety of a cosmetic product, the approved products will have an additional safety requirement that offers more reliability to the products accessible to the consumers. This is one of the reasons why many national regulatory authorities are demanding the accreditation of laboratories that carry out trials for the product in their field. It should be noted in this work that ANVISA, the body responsible for the surveillance of cosmetic products, established the RDC No 12 of 16 December 2012, which determines that every laboratory has to be accredited by CGCRE in order to be part of REBLAS (Brazilian Net of Analytical Laboratories in Health) [22].
The goal of this work was to carry out a survey of the main national legislations related to trials and tests performed on cosmetic products and, from there, to map the areas already accredited and recognized by CGCRE, as well as to identify possible gaps in the programs related to the accreditation bodies.
2. Experimental Section
2.1. Materials
As a resource of the scientific basis to define the safety trials and lifecycles of a cosmetic product, guaranteed bibliographical references in this area were used, including books and scientific articles.
As a resource of research and information, the following materials were used:
- (i).
- ANVISA documents, which is the Brazilian regulatory authority for cosmetic products. In this context are included technical statements and guidelines, as well as RDC.
- (ii).
- Information of the INMETRO website, a division of the CGCRE site, which is the Brazilian accreditation body. This information is divided into two groups: accreditation and recognition programs provided by CGCRE; as well as the contents of the scopes of the accredited Conformity Assessment Bodies (CAB) and test facilities recognized by CGCRE.
2.2. Development of the Work
The gathering of information was made by mapping the requirements of ANVISA documents, related to safety trials on cosmetic products, and from there, linking them with constant information of the scope of the CABs and recognized test facilities.
The mapping was oriented by the lifecycle of a cosmetic product, as well as pertinent safety trials for each cycle phase.
3. Results and Discussion
3.1. Lifecycles of a Cosmetic Product
According to the consulted references, the best illustration of the lifecycle of a cosmetic product can be seen in Figure 1 [10].
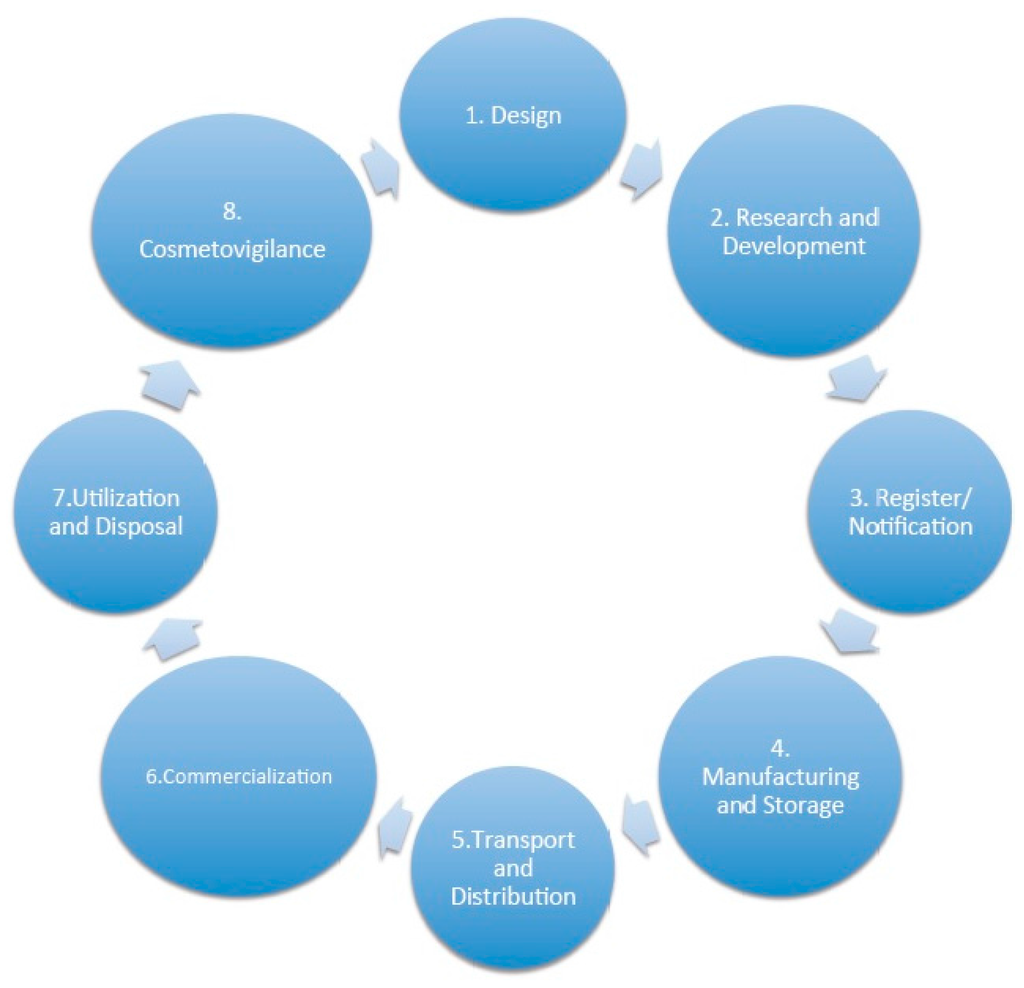
Figure 1.
Lifecycle of a cosmetic product.
Trials and tests in cosmetic products were identified in Phases 2–4.
Although Brazil has a specific resolution to implement a cosmetovigilance system, Phase 8 [23], it has not carried out a specific analysis of the tests to be performed at this stage; therefore, the need to perform a test for the cosmetic product and the specification of which test will depend on each case and the study to be conducted by the company. Several studies are available in the literature about the conduct of a study cosmetovigilance [24,25]. For this reason, Phase 8 has not been studied in this paper, although there is the possibility of carrying out a safety assessment involving testing in cosmetics.
3.1.1. Phase 2: Research and Development
Table 1 shows the results of technical requirements and applicable legislation to this cycle phase, as well as the location in this article of the results and discussions.

Table 1.
Relevant results of the research and development phase of a cosmetic product *.
3.1.2. Phase 3: Register and Notification
In Table 2 is the result of technical requirements and applicable legislation to this phase of the cycle, as well as the location of the results and discussions in this article.

Table 2.
Pertinent results of the register and notification phase of a cosmetic product.
3.1.3. Phase 4: Manufacturing and Storage
Table 3 shows the results of the technical requirements and applicable legislation to this phase of the cycle, as well as the location of the results in this article.

Table 3.
Pertinent results for the manufacturing and storage phase of a cosmetic product.
Table 4, Table 5, Table 6, Table 7 and Table 8 show the results regarding to phases 3 and 4 of the lifecycle of a cosmetic product.

Table 4.
Accredited Laboratories for the verification of organoleptic characteristics and physicochemical trials in cosmetics suggested by ANVISA [34].

Table 5.
Accredited laboratories for the determination of actives and other control components used in cosmetics.

Table 6.
Accredited laboratories for the evaluation of the stability of cosmetic products: organoleptic and physicochemical parameters.

Table 7.
Accredited laboratories for the stability evaluation of cosmetics: “challenge test”.

Table 8.
Accredited laboratories for the evaluation of the microbiological parameters of cosmetics.
It is important to point out that in this phase, the following parameters usually used in the production of cosmetics were not included: (i) cleanness validation and process; (ii) raw materials’ specifications (except when in the specific legislation, like the case of surfactants, pigments and preservatives (iii) trials on the intermediate and in the bulk product; (iv) specific control trials in the process; and (v) detection of prohibited substances.
Note for the following results, the laboratories in this study were coded as “Lab” followed by a number. It was not identified which test facilities were recognized by CGCRE.
3.2. Toxicological Evaluation of the Ingredients and Cosmetic Formula Per Se
With regard to toxicological data, the Scientific Committee on Consumer Safety (SCCS) has indicated the execution of the following trials for the evaluation of the toxicity of a cosmetic ingredient:
- (1)
- Acute systemic toxicity;
- (2)
- Corrosiveness and dermal irritation;
- (3)
- Skin sensitization;
- (4)
- Skin absorption/penetration;
- (5)
- Repeated-dose;
- (6)
- Mutagenicity/genotoxicity;
- (7)
- Sub-acute and sub-chronic toxicity;
- (8)
- Eye irritation;
- (9)
- Mucosa irritation;
- (10)
- Toxic effects induced by UV radiation (phototoxicity, genotoxicity, photoallergy);
- (11)
- Carcinogenicity;
- (12)
- Reproductive and development toxicity (teratogenicity);
- (13)
- Toxicokinetics and toxicodynamics.
According to the Guidance for the testing of cosmetics ingredients [28], the first six tests have been considered mandatory, and the others are subject to evaluation.
The majority of the tests indicated for the toxicological evaluation of cosmetic ingredients is acknowledged by ANVISA, by other international regulatory authorities and by SCCS, especially the tests proposed by OECD (Organization for Economic Co-operation and Development), but are not limited to these institutions.
As suggested in the Guidelines for the Evaluation of the Safety of Cosmetic Products and also in other sources [10,27], it is important to consider the toxicological data of the finished product because the final formulation can interfere, as it facilitates total or partial absorption of the ingredients. Furthermore, the possible interactions resulting from the association between ingredients can influence the potential risk of a product [26].
The OECD is an international organization with the goal of promoting politics that aim at the economic development and social wellbeing of people throughout the world [54]. Among these politics are the Principles of Good Laboratory Practice (GLP).
OECD’s principles of GLP are directed to assure the high quality and reliability of the tests related to the safety of chemical substances and their preparations. The principles were created in a context that aimed to harmonize the test procedures with the Mutual Acceptance of Data (MAD) [55].
In the specific case of toxicological trials related to the cosmetic products, the application of the GLP principles contributes to diluting the technical barriers for commercialization and improves the protection level of human and environmental health.
As already explained, in Brazil, the CGCRE is the Brazilian authority for the conformity assessment monitoring of the GLP. In May of 2011, Brazil, obtained full adhesion of the OECD actions related to the MAD for the evaluation of chemical products, including non-clinical trials of environmental safety and human health for the substances: “agrotoxins and its components” and “industrial chemical products” [56].
In February of 2015, it was made official by OECD that CGCRE obtained an expansion of its scope for the cosmetics area [55].
That is to say that the tests destined for cosmetic products carried out by all Brazilian test facilities recognized by CGCRE would be accepted in all member countries of the OECD, as well as non-members with full adhesion to the MAD.
Searching through the INMETRO website, a total of 10 test facilities was identified as recognized by CGCRE for the GLP studies related to ingredients in cosmetic products. The quantities of the recognized facilities, followed by the respective tests, are as follows:
- (i)
- Seven test facilities are recognized for the realization of physicochemical tests;
- (ii)
- Five test facilities are recognized for the realization of toxicological tests;
- (iii)
- Five test facilities are recognized for the realization of mutagenicity tests;
- (iv)
- Two test facilities are recognized for the realization of cytotoxicity;
- (v)
- Two test facilities are recognized for the realization of analytic and clinical chemistry.
3.3. Clinical Trials
Cosmetic products may need clinical trials in humans, enabling companies to offer consumers the highest safety with lower risk, assuring the best conditions to use a product. From pre-clinical information, the safety for human use should be confirmed [26].
Clinical trials that aim at the confirmation of safety in humans consist of two large groups: compatibility studies (maximized conditions) and acceptance studies (real use conditions). The types and objectives of each of these studies are specified in the following Table 9.

Table 9.
Types and objectives of the compatibility and acceptance clinical trials related to the application of the study [26].
Despite the importance of the clinical trials, CGCRE does not have a recognition program of this type of study. In previous work carried out by the author in 2013 [57], no accreditation body was identified to have such a kind of program.
Considering the relevance of the results of clinical trials in the development of a cosmetic product, as well as the adopted controls, for example, the profile and choice of studied population, the rigid protocols of human studies and the ethical principles involved, the recognition of the safety clinical trials in cosmetic products deserves an additional study, in order to verify mainly:
- (i)
- if clinical trials are liable for recognition;
- (ii)
- if nowadays there already exists a recognition program for the clinical trials by an accreditation body and;
- (iii)
- if the recognition activity has been practiced in Brazil and in other countries by non-accreditation bodies.
4. Conclusions
In conclusion, the importance of the evaluation of the safety of a cosmetic product is evident, as well as the added value to the product when trials and tests are carried out by accredited laboratories and test facilities recognized by an accreditation body and the Brazilian authority that monitors GLP studies.
It is interesting to note that Brazil has accredited laboratories to carry out most of the trials of the determination of actives and control ingredients stipulated by ANVISA, also including the stability trials. On the other hand, the quantity of accredited laboratories seems small, when we analyze the great consumption of cosmetics, the size of the Brazilian population and the continental dimension of the country.
With regard the toxicological evaluation, we can say only that in Brazil, there are recognized testing facilities to carry out toxicological tests. However, it is not stated whether the recognized tests fully cover that which is determined by ANVISA. For in the case of the testing facilities, the scope is flexible, and this information is kept only for the installation and is kept confidential by the accreditation body (CGCRE). Thus, this information has not been mentioned nor explored in this paper.
In the survey, the importance of clinical trials was also detected, although the Brazilian accreditation body does not have a recognition program for this type of study.
Acknowledgments
Firstly, I thank INMETRO for providing the platform for access to the scientific papers, to Patricia Weigert for proofreading the article and for excellent contributions and to Cecilia Bedin for the impeccable translation from Portuguese to English.
Conflicts of Interest
The author declares no conflict of interest.
References
- Hall, B.; Tozer, S.; Safford, B.; Coroama, M.; Steiling, W.; Leneveu-Duchemin, M.C.; McNamara, C.; Gibney, M. European consumer exposure to cosmetic products, a framework for conducting population exposure assessments. Food Chem. Toxicol. 2007, 45, 2097–2108. [Google Scholar] [CrossRef] [PubMed]
- Brown, V.J. Metals in lip products a cause for concern? Environ. Health Perspect. 2013, 121, A196. [Google Scholar] [CrossRef] [PubMed]
- Barata, E.A.F. Cosmetology—Basic Principles. In Basic Concepts; Tecnopress Editoring and Publicity Ltda: São Paulo, Brazil, 1995. [Google Scholar]
- Gonçalves, J.P.R.V. Pharmacy and Cosmetics in 19th Century in Portugal. Master’s Thesis, Lusophone University of Humanities and Technologies, Lisbon, Portugal, 2013; p. 95. [Google Scholar]
- Klingman, A.M. What are cosmeceuticals? In Procedures in Cosmetic Dermatology: Cosmeceuticals; Draelos, Z.D., Ed.; Elsevier: Philadelphia, PA, USA, 2005; pp. 1–2. [Google Scholar]
- Anunciato, T.P.; da Rocha Filho, P.A. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J. Cosmet. Dermatol. 2012, 11, 51–54. [Google Scholar] [CrossRef] [PubMed]
- ANVISA. RDC No 7, of 10 February 2015. About Technical Requirements for the Regularization of Personal Hygiene Products, Cosmetics and Perfumes; ANVISA: Brasilia, Brazil, 2015. [Google Scholar]
- Abihpec. Brazilian Cosmetics Market Grows 11% in 2014. Available online: http://brazilbeautynews.com/mercado-brasileiro-de-cosmeticos-cresceu-de-11-em,630 (accessed on 26 September 2015). (In Portuguese)
- Use of Repellents Skyrockets in Brazil Due to Dengue and Zika Outbreaks. Available online: http://cbn.globoradio.globo.com/editorias/economia/2016/02/07/VENDAS-DE-REPELENTES-NO-BRASIL-DISPARAM-COM-SURTO-DE-DENGUE-E-ZIKA.htm (accessed on 10 February 2016). (In Portuguese)
- Costa, A. The International Cosmeceutical Treaty. In Foundations for the Cosmeceutical World; Guanabara Koogan: Rio de Janeiro, Brazil, 2012. [Google Scholar]
- Bad Reaction to Cosmetics? Tell FDA, March 2011. Available online: http://www.fda.gov/downloads/For-Consumers/ConsumerUpdates/UCM248686.pdf (accessed on 27 September 2015). (In Portuguese)
- SCCS—Scientific Committee on Consumer Safety. Opinion on p-Phenylenediamine, COLIPA No A7. Available online: http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_094.pdf (accessed on 26 May 2016).
- Farquharson, A.A.; Stoopler, E.T.; Houston, A.M.; Brown, R.S. Erythema multiforme major secondary to a cosmetic facial cream: First case report. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 121, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Natural Repellent Causes Allergic Reactions in Many Consumers. Available online: http://www.clickguarulhos.com.br/repelente-natural-causa-reacao-alergica-em-varios-usuarios (accessed on 10 February 2016). (In Portuguese)
- ANVISA. RDC No 48, 16 March 2006. Approves the Technical Regulation about the List of Substances That Can Not Be Used in Personal Care Products, Cosmetics and Perfumes; ANVISA: Brasilia, Brazil, 2006. [Google Scholar]
- EU (European Union). Annex II of Regulation (EC) No. 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32009R1223 (accessed on 26 May 2016).
- INMETRO. What is Inmetro? Rio de Janeiro, Brazil, 2016. Available online: http://www.inmetro.gov.br/inmetro/oque.asp (accessed on 26 May 2016). (In Portuguese)
- Ministry of Development, Industry and Foreign Trade. Imetro´s Bylaws. Ordinance No 165, of 02 April 2013; Ministry of Development, Industry and Foreign Trade: Brasilia, Brazil, 2013. [Google Scholar]
- INMETRO. Accreditation of Laboratories (ABNT NBR ISO/IEC 17025:2005). Rio de Janeiro, Brazil, 2016. Available online: http://inmetro.gov.br/credenciamento/acre_lab.asp (accessed on 26 May 2016). (In Portuguese)
- INMETRO. GLP Monitoring. Available online: http://www.inmetro.gov.br/monitoramento_BPL/reconhecimento_BPL.asp (accessed on 16 January 2016). (In Portuguese)
- ABNT. Conformity Assessment—Vocabulary and General Principles; ABNT NBR ISO/IEC 17.000:2005; ABNT: Rio de Janeiro, Brazil, 2005. [Google Scholar]
- ANVISA. ANVISA RDC No 12, of 16 February 2012. Order About the Brazilian Network of Analytical Laboratories in Health (REBLAS); ANVISA: Brasilia, Brazil, 2012. [Google Scholar]
- ANVISA. RDC No 332, of 1 December 2005. The Manufacturers and/or Importers of Personal Care Products and Cosmetics Perfumes, Installed in the National Territory Shall Implement a Cosmetovigilance System from 31 December 2005; ANVISA: Brasilia, Brazil, 2005. [Google Scholar]
- Zweers, P.G.M.A.; Gilmour, N.J.; Hepburn, P.A.; Gerritsen, R.F.; van Puijenbroek, E.P. Causality methods in Cosmetovigilance: Comparison of Colipa and PLM versus global introspection. Regul. Toxicol. Pharmacol. 2012, 63, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Giovanni, C.D.; Arcoraci, V.; Gambardella, L.; Sautebin, L. Cosmetovigilance survey: Are cosmetics considered safe by consumers? Pharmacol. Res. 2006, 53, 16–21. [Google Scholar] [CrossRef] [PubMed]
- ANVISA. Guidelines for the Evaluation of Cosmetic Products Safety, 2nd ed.; Brazilian Health Surveillance Agency (ANVISA): Brasilia, Brazil, 2012. [Google Scholar]
- Nohynek, G.; Antignac, E.; Re, T.; Toutain, H. Review Safety assessment of personal care products/cosmetics and their ingredients. Toxicol. Appl. Pharmacol. 2010, 243, 239–259. [Google Scholar] [CrossRef] [PubMed]
- SCCS—Scientific Committee on Consumer Safety. The SCCS Notes of Guidance for the Testing of Cosmetic Ingredients and Their Safety Evaluation, 9th Edition, Adopted on 29 September 2015. Available online: http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_190.pdf (accessed on 26 May 2016).
- ANVISA. RDC No 15, of 24 April 2015. About Technical Requirements for the Concession of Registry of Personal Hygiene Products, Cosmetics and Children Perfumes; ANVISA: Brasilia, Brazil, 2015. [Google Scholar]
- ANVISA. RDC No 44, of 9 August 2012. Approves Mercosul Technical Regulation about “List of Pigments Permitted for Personal Hygiene Products, Cosmetics and Perfumes” and Other Arrangements; ANVISA: Brasilia, Brazil, 2012. [Google Scholar]
- ANVISA. RDC No 29, of 1 June 2012. Approves Mercosul Technical Regulation about “List of Conservative Substances Permitted for Personal Hygiene Products, Cosmetics and Perfumes” and Other Arrangements; ANVISA: Brasilia, Brazil, 2012. [Google Scholar]
- ANVISA. RDC No 03, of 18 January 2012. Mercosul Technical Regulation about “List of Substances That Personal Hygiene Products, Cosmetics and Perfumes Should not Have, Except in Established Conditions and Restrictions”; ANVISA: Brasilia, Brazil, 2012. [Google Scholar]
- ANVISA. RDC No 47, of 16 March 2006. Technical Regulation “List of Ultraviolet Filters Permitted for Personal Hygiene Products, Cosmetics and Perfumes”; ANVISA: Brasilia, Brazil, 2006. [Google Scholar]
- ANVISA. Guidelines of Quality Control of Cosmetic Products—An Approach about Physical and Chemical Trials, 2nd ed.; ANVISA: Brasilia, Brazil, 2008. [Google Scholar]
- ANVISA. Theme Series—Cosmetics, Quality 1: Guidelines of the Cosmetics Stability; ANVISA: Brasilia, Brazil, 2004; Volume 1. [Google Scholar]
- ANVISA. RDC No 481, 23 September 1999. Establishes Parameters for the Microbiological Control of Personal Hygiene, Cosmetics and Perfumes; ANVISA: Brasilia, Brazil, 1999. [Google Scholar]
- ANVISA. RDC No 48, 25 October 2013. Approves the Technical Regulation of Good Practice in Manufacturing Personal Hygiene Products, Cosmetics and Perfumes; ANVISA: Brasilia, Brazil, 2013. [Google Scholar]
- ANVISA. Catec Technical Report, No 1, 15 December 2008. Use of Lead Acetate in Hair Dyes; ANVISA: Brasilia, Brazil, 2008. [Google Scholar]
- ANVISA. Catec Technical Report, No 7, 28 September 2001 (updated in 16 February 2006). Use of Alpha-Hydroxiacids in Cosmetics; ANVISA: Brasilia, Brazil, 2001. [Google Scholar]
- ANVISA. Catec Technical Statements, No 1, 29 January 2002. Use of Methylxantines in Cosmetic Preparations; ANVISA: Brasilia, Brazil, 2002. [Google Scholar]
- ANVISA. Catec Technical Report, No 6, 21 December 2010. Use of Camphor in Cosmetic Products; ANVISA: Brasilia, Brazil, 2010. [Google Scholar]
- ANVISA. Catec Technical Report, No 5, 22 March 2002 (updated in 07/05/2011). Cosmetic Products for Hair Blanching; ANVISA: Brasilia, Brazil, 2002. [Google Scholar]
- ANVISA. Resolution, No 19, 10 April 2013. About Technical Requirements for the Concession of Registries of Cosmetic Products and Repellents; ANVISA: Brasilia, Brazil, 2013. [Google Scholar]
- ANVISA. Catec Technical Report, No 2, 23 December 2009. Use of DEET and Other Repellent Substances; ANVISA: Brasilia, Brazil, 2009. [Google Scholar]
- Carcinogenic Potential of Sodium Lauryl Sulphate. Available online: http://www.anvisa.gov.br/cosmeticos/informa/parecer_lauril.htm (accessed on 20 September 2015). (In Portuguese)
- ANVISA. Catec Technical Report, No 8, 1 November 2005. Menthol in Cosmetic Products; ANVISA: Brasilia, Brazil, 2005. [Google Scholar]
- ANVISA. Catec Technical Report, No 1, 26 July 2006. Inclusion of “Methylisothiazolinone” in the Approved Conservatives List; ANVISA: Brasilia, Brazil, 2006. [Google Scholar]
- ANVISA. Catec Technical Report, No 10, 25 October 2002. Oral Hygiene Products Indicated for Dentine Hypersensitization; ANVISA: Brasilia, Brazil, 2002. [Google Scholar]
- ANVISA. Catec Technical Report, No 4, 6 July 2005. Use of Potassium Salts (Nitrate, Citrate e Chloride) in Products for Dentine Hypersensitization; ANVISA: Brasilia, Brazil, 2005. [Google Scholar]
- ANVISA. Catec Technical Report, No 6, 23 August 2005. Toxicological Evaluation of Methyl Salicylate; ANVISA: Brasilia, Brazil, 2005. [Google Scholar]
- ANVISA. Catec Technical Report, No 2, 13 January 2010. Use of Turpentine in Cosmetics; ANVISA: Brasilia, Brazil, 2010. [Google Scholar]
- ANVISA. Catec Technical Report, No 5, 21 December 2010. Use of Urea in Cosmetic Products; ANVISA: Brasilia, Brazil, 2010. [Google Scholar]
- ANVISA. Catec Technical Report, No 4, 21 December 2010. Use of Retinoids in Cosmetic Products; ANVISA: Brasilia, Brazil, 2010. [Google Scholar]
- The Organisation for Economic Co-operation and Development (OECD). Available online: http://www.oecd.org/about/ (accessed on 17 January 2016).
- OECD. Good Laboratory Practice (GLP). Available online: http://www.oecd.org/chemicalsafety/testing/goodlaboratorypracticeglp.htm (accessed on 17 January 2016).
- GLP Monitoring. Available online: http://www.inmetro.gov.br/monitoramento_BPL/historico.asp?iacao=imprimir (accessed on 16 January 2016). (In Portuguese)
- Almeida, L.S. Proposal of Scope of Clinical Assays of Safety and Effectiveness of Cosmetics Products. J. Phys. Conf. Ser. 2015, 575. [Google Scholar] [CrossRef]
- ANVISA. Catec Technical Report, No 6, de 28 September 2001. Use of the Term “for Sensitive Skin” in Cosmetic Products; ANVISA: Brasilia, Brazil, 2001. [Google Scholar]
- ANVISA. Catec Technical Report, No 5, 23 August 2005. Toxicological Evaluation of Methyl Nicotinate; ANVISA: Brasilia, Brazil, 2005. [Google Scholar]
- ANVISA. Catec Technical Report, No 3, 29 June 2001 (updated in 28 June 2004). Use of Vitamin C in Cosmetic Products; ANVISA: Brasilia, Brazil, 2001. [Google Scholar]
- ANVISA. Catec Technical Report, No 5, 28 September 2001. Use of the “Hypoallergenic” in Personal Hygiene Products, Cosmetics and Perfumes; ANVISA: Brasilia, Brazil, 2001. [Google Scholar]
- ANVISA. Catec Technical Report, No 1, 28 May 2004 (updated in 20 May 2005). Intimate Hygiene Products; ANVISA: Brasilia, Brazil, 2004. [Google Scholar]
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).