Hormetins as Novel Components of Cosmeceuticals and Aging Interventions
Abstract
:1. Introduction
2. Biogerontology in a Nutshell
3. Modulation of Aging through Hormesis
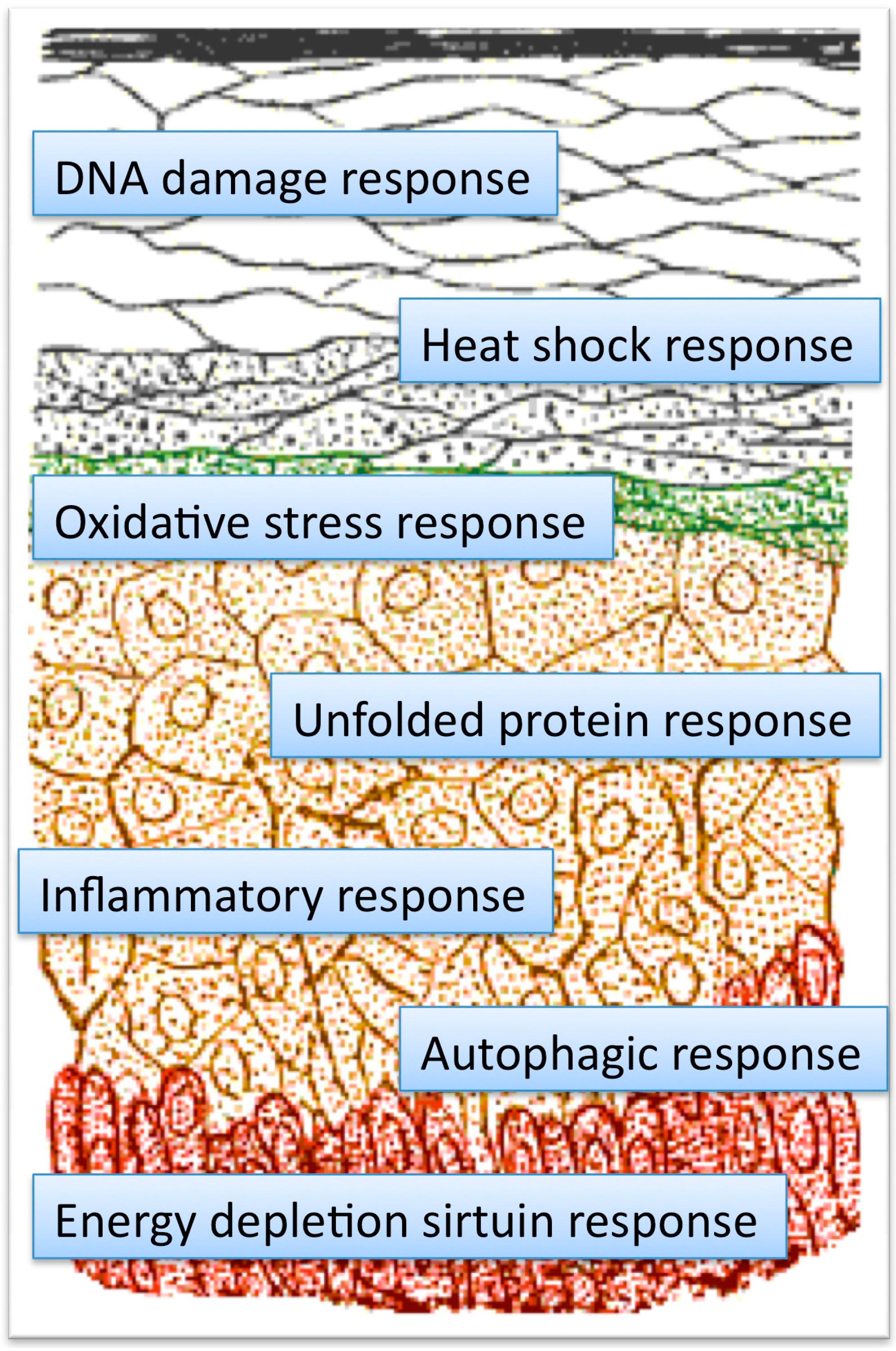
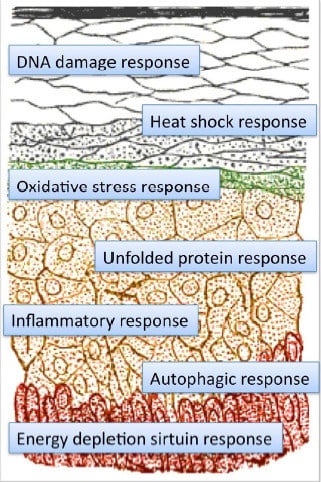
4. Hormesis and Hormetins in Cosmetics
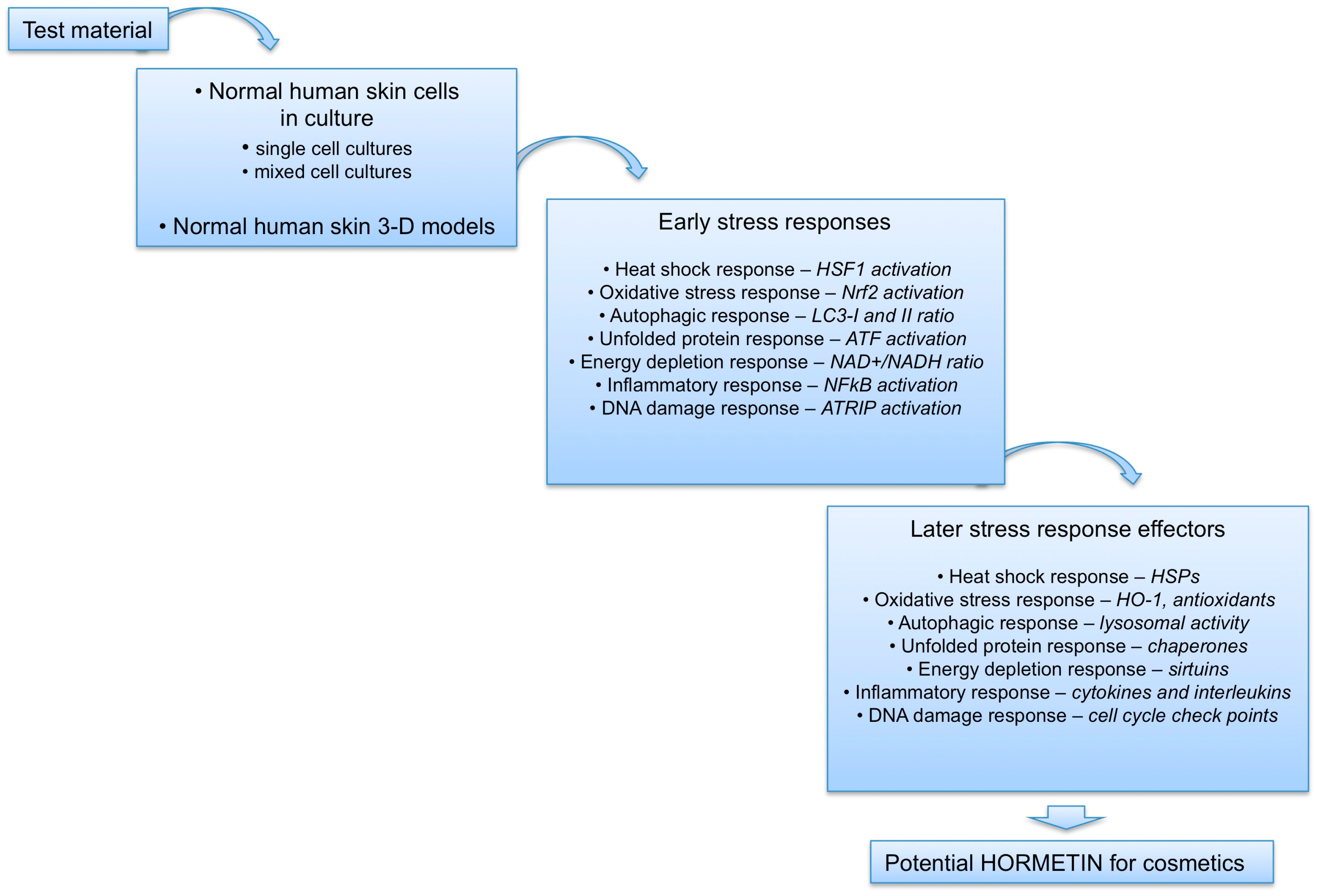
5. Conclusions and Perspectives
Conflicts of Interest
References
- Rattan, S.I.S. Healthy ageing, but what is health? Biogerontology 2013, 14, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. Aging is not a disease: Implications for intervention. Aging Dis. 2014, 5, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Otin, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Mitnitski, A.; Song, X.; Rockwood, K. Assessing biological aging: The origin of deficit accumulation. Biogerontology 2013, 14, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Muradian, K. “Pull and push back” concepts of longevity and life span extension. Biogerontology 2013, 14, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Hayflick, L. Biological aging is no longer an unsolved problem. Ann. NY Acad. Sci. 2007, 1100, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R. Understanding Ageing; Cambridge University Press: Cambridge, UK, 1995; p. 207. [Google Scholar]
- Holliday, R. Aging is no longer an unsolved problem in biology. Ann. NY Acad. Sci. 2006, 1067, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R. Genes and the evolution of longevities. Biogerontology 2009, 10, 1–2. [Google Scholar] [CrossRef] [PubMed]
- </i>Barzilai, N.; Guarente, L.; Kirkwood, T.B.; Partridge, L.; Rando, T.A.; Slagboom, P.E. The palce of genetics in ageing research. Nat. Rev. Genet. 2012, 13, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. Homeostasis, homeodynamics, and aging. In Encyclopedia of Gerontology, 2nd ed.; Birren, J., Ed.; Elsevier Inc.: Oxford, UK, 2007; pp. 696–699. [Google Scholar]
- Rattan, S.I.S. Biogerontology: From here to where? The Lord Cohen Medal Lecture-2011. Biogerontology 2012, 13, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. Increased molecular damage and heterogeneity as the basis of aging. Biol. Chem. 2008, 389, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R.; Rattan, S.I.S. Longevity mutants do not establish any “new science” of ageing. Biogerontology 2010, 11, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Yates, F.E. Order and complexity in dynamical systems: Homeodynamics as a generalized mechanics for biology. Math. Comput. Model. 1994, 19, 49–74. [Google Scholar] [CrossRef]
- Rattan, S.I.S. Biogerontology: The next step. Ann. N.Y. Acad. Sci. 2000, 908, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Carnes, B.A. What is lifespan regulation and why does it exist? Biogerontology 2011, 12, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Carnes, B.A.; Olshansky, S.J.; Grahn, D. Biological evidence for limits to the duration of life. Biogerontology 2003, 4, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. Gerontogenes: Real or virtual? FASEB J. 1995, 9, 284–286. [Google Scholar] [PubMed]
- Rattan, S.I.S. Theories of biological aging: Genes, proteins and free radicals. Free Rad. Res. 2006, 40, 1230–1238. [Google Scholar] [CrossRef]
- Rattan, S.I.S. Hormesis in aging. Ageing Res. Rev. 2008, 7, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Baldwin, L.A. Toxicology rethinks its central belief. Nature 2003, 421, 891–892. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Iavicoli, I.; Calabrese, V. Hormesis: Why it is important to biogerontologists. Biogerontology 2012, 13, 215–235. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Jonas, W.B. Homeopathy: Clarifying its relationship to hormesis. Hum. Exp. Toxicol. 2010, 29, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Southam, C.M.; Ehrlich, J. Effects of extracts of western red-cedar heartwood on certain wood-decaying fungi in culture. Phytopathology 1943, 33, 517–524. [Google Scholar]
- Calabrese, E.J.; Bachmann, K.A.; Bailer, A.J.; Bolger, P.M.; Borak, J.; Cai, L.; Cedergreen, N.; Cherian, M.G.; Chiueh, C.C.; Clarkson, T.W.; et al. Biological stress response terminology: Integrating the concepts of adaptive response and preconditioning stress within a hormetic dose-response framework. Toxicol. Appl. Pharmacol. 2007, 222, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Le Bourg, E.; Rattan, S.I.S. Mild Stress and Healthy Aging: Applying Hormesis in Aging Research and Interventions; Springer: Dordrecht, The Netherlands, 2008. [Google Scholar]
- Mattson, M.P.; Calabrese, E. Hormesis—A Revolution in Biology, Toxicology and Medicine; Springer: New York, NY, USA, 2010. [Google Scholar]
- Rattan, S.I.S.; Le Bourg, E. Hormesis in Health and Disease; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Rattan, S.I.S.; Demirovic, D. Hormesis and aging. In Hormesis: A Revolution in Biology, Toxicology and Medicine; Mattson, M.P., Calabrese, E., Eds.; Springer: New York, NY, USA, 2009; pp. 153–175. [Google Scholar]
- Rattan, S.I.S.; Demirovic, D. Hormesis can and does work in humans. Dose-Response 2010, 8, 58–63. [Google Scholar] [CrossRef]
- Rattan, S.I.S.; Demirovic, D. Hormesis as a mechanism for the anti-aging effects of calorie restriction. In Calorie Restriction, Aging and Longevity; Everitte, A.V., Rattan, S.I.S., Le Couteur, D.G., de Cabo, R., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 233–245. [Google Scholar]
- Rattan, S.I.S. Rationale and methods of discovering hormetins as drugs for healthy ageing. Expert Opin. Drug Discov. 2012, 7, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Abete, P.; Calabrese, E.; Ji, L.L.; Kristensen, T.; Le Bourg, E.; Loeschcke, V.; Morris, B.; Rengo, F.; Rattan, S.I.S.; Safwat, A.; et al. Mild stress and healthy aging: Perspectives for human beings. In Mild Stress and Healthy Aging: Applying Hormesis in Aging Research and Interventions; Le Bourg, E., Rattan, S.I.S., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 171–183. [Google Scholar]
- Whitmarsh, A.J. A central role for p38 mapk in the early trascriptional response to stress. BMC Biol. 2010, 8. [Google Scholar] [CrossRef]
- Majmundar, A.J.; Wong, W.J.; Simon, M.C. Hypoxia-inducible factors and the response to hypoxic stress. Mol. Cell 2010, 40, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Demirovic, D.; Rattan, S.I. Establishing cellular stress response profiles as biomarkers of homeodynamics, health and hormesis. Exp. Gerontol. 2013, 48, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Demirovic, D.; de Toda, I.M.; Rattan, S.I.S. Molecular stress response pathways as the basis of hormesis. In Hormesis in Health and Disease; Rattan, S.I.S., Le Bourg, E., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 227–241. [Google Scholar]
- Verbeke, P.; Fonager, J.; Clark, B.F.C.; Rattan, S.I.S. Heat shock response and ageing: Mechanisms and applications. Cell Biol. Int. 2001, 25, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Fonager, J.; Beedholm, R.; Clark, B.F.C.; Rattan, S.I.S. Mild stress-induced stimulation of heat shock protein synthesis and improved functional ability of human fibroblasts undergoing aging in vitro. Exp. Gerontol. 2002, 37, 1223–1238. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S.; Eskildsen-Helmond, Y.E.G.; Beedholm, R. Molecular mechanisms of anti-aging hormetic effects of mild heat stress on human cells. Nonlinearity Biol. Toxicol. Med. 2003, 2, 105–116. [Google Scholar] [CrossRef]
- Beedholm, R.; Clark, B.F.C.; Rattan, S.I.S. Mild heat stress stimulates proteasome and its 11s activator in human fibroblasts undergoing aging in vitro. Cell Stress Chaperones 2004, 9, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Banhegyi, G.; Baumeister, P.; Benedetti, A.; Dong, D.; Fu, Y.; Lee, A.S.; li, J.; Mao, C.; Margittai, E.; Ni, M.; et al. Endoplasmic reticulum stress. Ann. NY Acad. Sci. 2007, 1113, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Markaki, M.; Tavernarakis, N. The role of autophagy in genetic pathways influencing ageing. Biogerontology 2011, 12, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Ryter, S.W.; Choi, A.M.K. Autophagy: An integral component of the mammalian stress response. J. Biochem. Pharmacol. Res. 2013, 1, 176–188. [Google Scholar] [PubMed]
- Morris, B.J. Seven sirtuins for seven deadly diseases of aging. Free Radic. Biol. Med. 2013, 56, 133–171. [Google Scholar] [CrossRef] [PubMed]
- Chirumbolo, S. Possible role of nf-kappaB in hormesis during ageing. Biogerontology 2012, 13, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S.; Kryzch, V.; Schnebert, S.; Perrier, E.; Carine Nizard, C. Hormesis-based anti-aging products: A case study of a novel cosmetic. Dose-Response 2013, 11, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Sonneborn, J.S. Mimetics of hormetic agents: Stress-resistance triggers. Dose-Response 2010, 8, 97–121. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T.; Tsuchiya, T.; Komatsu, T.; Mori, R.; Hayashi, H.; Shimano, H.; Spindler, S.R.; Shimokawa, I. Development of a bioassay to screen for chemicals mimicking the anti-aging effects of calorie restriction. Biochem. Biophys. Res. Commun. 2010, 401, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Gohil, V.M.; Sheth, S.A.; Nilsson, R.; Wojtovich, A.P.; Lee, J.H.; Perocchi, F.; Chen, W.; Clish, C.B.; Ayata, C.; Brookes, P.S.; et al. Nutrient-sensitized screening for drugs that shift energy metabolism from mitochondrial respiration to glycolysis. Nat. Biotechnol. 2010, 28, 249–255. [Google Scholar] [PubMed]
- Mendelsohn, A.R.; Larrick, J.W. Fibroblast growth factor-21 is a promising dietary restriction mimetic. Rejuvenation Res. 2012, 15, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Pallauf, K.; Rimbach, G. Autophagy, polyphenols and healthy ageing. Ageing Res. Rev. 2013, 12, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Andujar, I.; Recio, M.C.; Giner, R.M.; Rios, J.L. Cocoa polyphenols and their potential benefits for human health. Oxid. Med. Cell Longev. 2012, 2012. [Google Scholar] [CrossRef]
- Chondrogianni, N.; Kapeta, S.; Chinou, I.; Vassilatou, K.; Papassideri, I.; Gonos, E.S. Anti-ageing and rejuvenating effects of quercetin. Exp. Gerontol. 2010, 45, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.F.; Pereira-Wilson, C.; Rattan, S.I. Curcumin induces heme oxygenase-1 in normal human skin fibroblasts through redox signaling: Relevance for anti-aging intervention. Mol. Nutr. Food Res. 2011, 55, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Phromnoi, K.; Yadav, V.R.; Chaturvedi, M.M.; Aggarwal, B.B. Targeting inflammatory pathways by flavonoids for prevention and treatment of cancer. Planta Med. 2010, 76, 1044–1063. [Google Scholar] [CrossRef] [PubMed]
- Niu, P.; Liu, L.; Gong, Z.; Tan, H.; Wang, F.; Yuan, J.; Feng, Y.; Wei, Q.; Tanguay, R.M.; Wu, T. Overexpressed heat shock protein 70 protects cells against DNA damage caused by ultraviolet c in a dose-dependent manner. Cell Stress Chaperones 2006, 11, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Leontieva, O.V.; Blagosklonny, M.V. Hypoxia and gerosuppression: The mTOR saga continues. Cell Cycle 2012, 11, 3926–3931. [Google Scholar] [CrossRef] [PubMed]
- Han, S.B.; Kwon, S.S.; Jeong, Y.M.; Yu, E.R.; Park, S.N. Physical characterization and in vitro skin permeation of solid lipid nanoparticles for transdermal delivery of quercetin. Int. J. Cosmet. Sci. 2014, 36, 588–597. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rattan, S.I.S. Hormetins as Novel Components of Cosmeceuticals and Aging Interventions. Cosmetics 2015, 2, 11-20. https://doi.org/10.3390/cosmetics2010011
Rattan SIS. Hormetins as Novel Components of Cosmeceuticals and Aging Interventions. Cosmetics. 2015; 2(1):11-20. https://doi.org/10.3390/cosmetics2010011
Chicago/Turabian StyleRattan, Suresh I. S. 2015. "Hormetins as Novel Components of Cosmeceuticals and Aging Interventions" Cosmetics 2, no. 1: 11-20. https://doi.org/10.3390/cosmetics2010011
APA StyleRattan, S. I. S. (2015). Hormetins as Novel Components of Cosmeceuticals and Aging Interventions. Cosmetics, 2(1), 11-20. https://doi.org/10.3390/cosmetics2010011