Laser-Assisted Drug Delivery on the Face: A Systematic Review of Indications and Outcomes
Abstract
1. Introduction
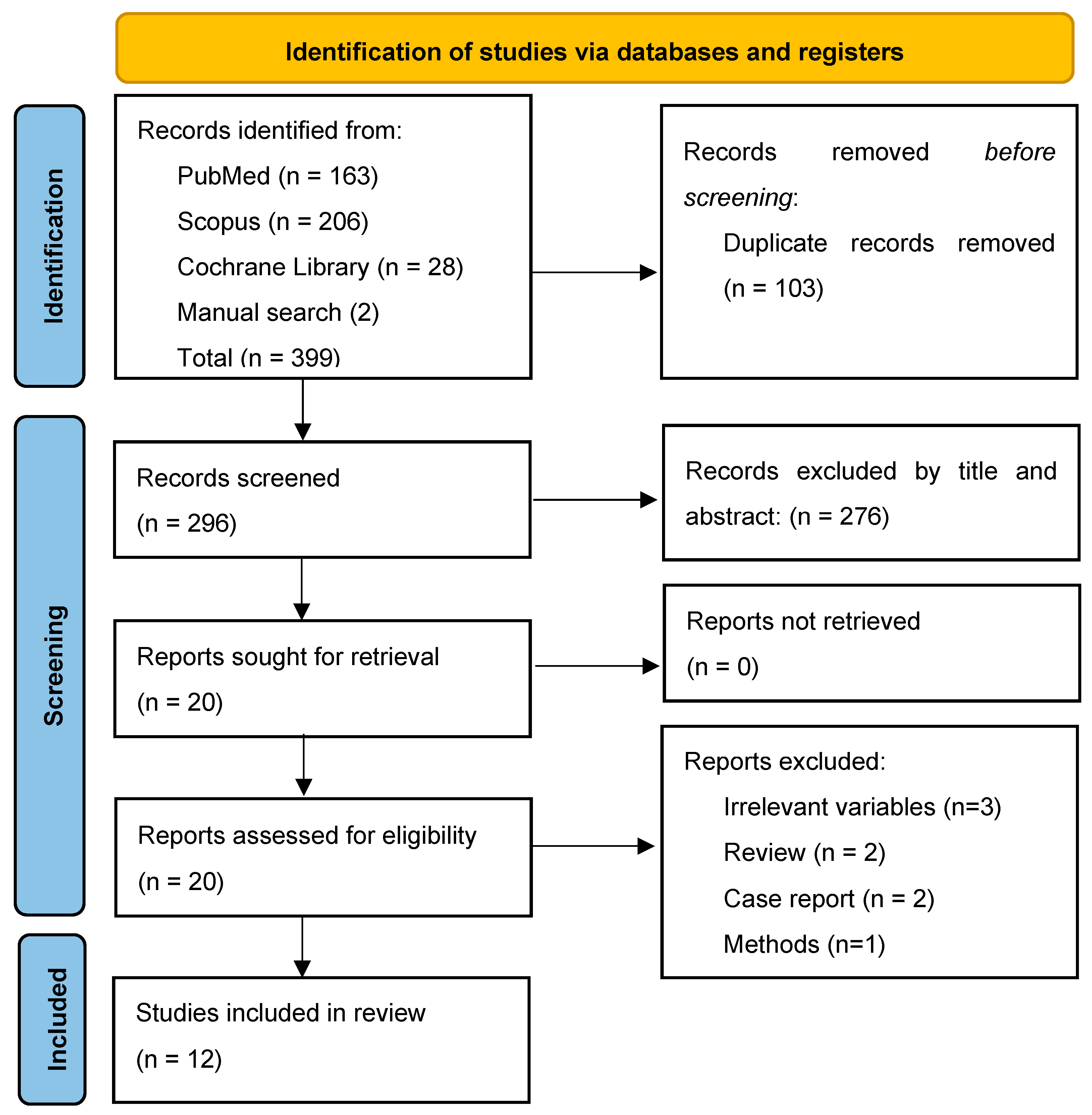
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility of Relevant Studies
2.3. Study Selection
2.4. Data Collection and Risk of Bias Assessment
2.5. Data Synthesis and Analysis
3. Results
3.1. General Study Characteristics
3.2. Patient Characteristics
3.3. LADD Characteristics
3.4. Outcomes
3.4.1. Melasma
3.4.2. Facial Rejuvenation and Rhytides
3.4.3. Other Applications
3.5. Complications
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wenande, E.; Anderson, R.R.; Haedersdal, M. Fundamentals of Fractional Laser-Assisted Drug Delivery: An in-Depth Guide to Experimental Methodology and Data Interpretation. Adv. Drug Deliv. Rev. 2020, 153, 169–184. [Google Scholar] [CrossRef]
- Elias, P.M.; Menon, G.K. Structural and Lipid Biochemical Correlates of the Epidermal Permeability Barrier. In Advances in Lipid Research; Elsevier: Amsterdam, The Netherlands, 1991; Volume 24, pp. 1–26. ISBN 978-0-12-024924-4. [Google Scholar]
- Chuang, S.-Y.; Lin, Y.-K.; Lin, C.-F.; Wang, P.-W.; Chen, E.-L.; Fang, J.-Y. Elucidating the Skin Delivery of Aglycone and Glycoside Flavonoids: How the Structures Affect Cutaneous Absorption. Nutrients 2017, 9, 1304. [Google Scholar] [CrossRef] [PubMed]
- Parhi, R.; Mandru, A. Enhancement of Skin Permeability with Thermal Ablation Techniques: Concept to Commercial Products. Drug Deliv. Transl. Res. 2021, 11, 817–841. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R.; Langer, R. Transdermal Drug Delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; John Wiley & Sons: Chichester, UK, 2019. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. J. Clin. Epidemiol. 2021, 134, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Yun, P.L.; Tachihara, R.; Anderson, R.R. Efficacy of Erbium:Yttrium-Aluminum-Garnet Laser-Assisted Delivery of Topical Anesthetic. J. Am. Acad. Dermatol. 2002, 47, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yao, C.; Zhang, H.; Li, L.; Song, Y. Efficacy and Safety of 755-Nm Picosecond Alexandrite Laser with Topical Tranexamic Acid versus Laser Monotherapy for Melasma and Facial Rejuvenation: A Multicenter, Randomized, Double-Blinded, Split-Face Study in Chinese Patients. Lasers Med. Sci. 2022, 37, 2879–2887. [Google Scholar] [CrossRef]
- Benzaquen, M.; Fongue, J.; Pauly, V.; Collet-Villette, A.-M. Laser-Assisted Hyaluronic Acid Delivery by Fractional Carbon Dioxide Laser in Facial Skin Remodeling: A Prospective Randomized Split-Face Study in France. Lasers Surg. Med. 2021, 53, 1166–1172. [Google Scholar] [CrossRef]
- Al-Dhalimi, M.A.; Yasser, R.H. Evaluation of the of the Efficacy of Fractional Erbium-Doped Yttrium Aluminum Garnet Laser-Assisted Drug Delivery of Kojic Acid in the Treatment of Melasma; A Split Face, Comparative Clinical Study. J. Cosmet. Laser Ther. 2021, 23, 65–71. [Google Scholar] [CrossRef]
- Park, S.J.; Park, J.W.; Seo, S.J.; Park, K.Y. Evaluating the Tolerance and Efficacy of Laser-Assisted Delivery of Tranexamic Acid, Niacinamide, and Kojic Acid for Melasma: A Single Center, Prospective, Split-Face Trial. Dermatol. Ther. 2022, 35, e15287. [Google Scholar] [CrossRef]
- Wanitphakdeedecha, R.; Sy-Alvarado, F.; Patthamalai, P.; Techapichetvanich, T.; Eimpunth, S.; Manuskiatti, W. The Efficacy in Treatment of Facial Melasma with Thulium 1927-Nm Fractional Laser-Assisted Topical Tranexamic Acid Delivery: A Split-Face, Double-Blind, Randomized Controlled Pilot Study. Lasers Med. Sci. 2020, 35, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Widianingsih, N.P.S.; Setyaningrum, T.; Prakoeswa, C.R.S. The Efficacy and Safety of Fractional Erbium Yag Laser Combined with Topical Amniotic Membrane Stem Cell (AMSC) Metabolite Product for Facial Rejuvenation: A Controlled, Split-Face Study. Dermatol. Rep. 2019, 11, S1. [Google Scholar] [CrossRef]
- Ibrahim, O.; Ionta, S.; Depina, J.; Petrell, K.; Arndt, K.A.; Dover, J.S. Safety of Laser-Assisted Delivery of Topical Poly-l-Lactic Acid in the Treatment of Upper Lip Rhytides: A Prospective, Rater-Blinded Study. Dermatol. Surg. 2019, 45, 968–974. [Google Scholar] [CrossRef]
- Badawi, A.M.; Osman, M.A. Fractional Erbium-Doped Yttrium Aluminum Garnet Laser-Assisted Drug Delivery of Hydroquinone in the Treatment of Melasma. Clin. Cosmet. Investig. Dermatol. 2018, 11, 13–20. [Google Scholar] [CrossRef]
- Waibel, J.S.; Mi, Q.-S.; Ozog, D.; Qu, L.; Zhou, L.; Rudnick, A.; Al-Niaimi, F.; Woodward, J.; Campos, V.; Mordon, S. Laser-Assisted Delivery of Vitamin C, Vitamin E, and Ferulic Acid Formula Serum Decreases Fractional Laser Postoperative Recovery by Increased Beta Fibroblast Growth Factor Expression. Lasers Surg. Med. 2016, 48, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, B.H.; Burnett, C.; Ozog, D. Prospective Randomized Controlled Study to Determine the Effect of Topical Application of Botulinum Toxin A for Crow’s Feet after Treatment with Ablative Fractional CO2 Laser. Dermatol. Surg. 2015, 41, S75–S81. [Google Scholar] [CrossRef]
- Gawdat, H.I.; Hegazy, R.A.; Fawzy, M.M.; Fathy, M. Autologous Platelet Rich Plasma: Topical Versus Intradermal After Fractional Ablative Carbon Dioxide Laser Treatment of Atrophic Acne Scars. Dermatol. Surg. 2014, 40, 152–161. [Google Scholar] [CrossRef]
- Seretis, K.; Goulis, D.; Demiri, E.C.; Lykoudis, E.G. Prevention of Seroma Formation Following Abdominoplasty: A Systematic Review and Meta-Analysis. Aesthetic Surg. J. 2017, 37, 316–323. [Google Scholar] [CrossRef]
- Seretis, K.; Boptsi, A.; Boptsi, E.; Lykoudis, E.G. The Efficacy of Wide-Awake Local Anesthesia No Tourniquet (WALANT) in Common Plastic Surgery Operations Performed on the Upper Limbs: A Case-Control Study. Life 2023, 13, 442. [Google Scholar] [CrossRef]
- Demiri, E.; Koliakos, G.; Goulis, D.G.; Seretis, K. Weight Reduction Following Abdominoplasty: A Systematic Review. Plast. Reconstr. Surg. 2013, 132, 314e–316e. [Google Scholar] [CrossRef]
- Seretis, K. The Efficacy of Local Anesthesia for Postoperative Pain Control in Breast Augmentation Surgery: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials. Aesthetic Plast. Surg. 2022, 46, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Seretis, K.; Bounas, N. The Efficacy of Different Nerve Blocks on Postoperative Pain and Sequelae in Patients Undergoing Abdominoplasty: A Network Meta-Analysis. Aesthet Surg. J. 2023, 43, NP325–NP336. [Google Scholar] [CrossRef] [PubMed]
- Seretis, K.; Bounas, N.; Papaspyrou, F. Antibiotic Prophylaxis in Reduction Mammaplasty: A Network Meta-Analysis. Aesthetic Plast. Surg. 2023, 47, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, O.; Wenande, E.; Hogan, S.; Arndt, K.A.; Haedersdal, M.; Dover, J.S. Challenges to Laser-Assisted Drug Delivery: Applying Theory to Clinical Practice. Lasers Surg. Med. 2018, 50, 20–27. [Google Scholar] [CrossRef]
- Haak, C.S.; Christiansen, K.; Erlendsson, A.M.; Taudorf, E.H.; Thaysen-Petersen, D.; Wulf, H.C.; Haedersdal, M. Ablative Fractional Laser Enhances MAL-Induced PpIX Accumulation: Impact of Laser Channel Density, Incubation Time and Drug Concentration. J. Photochem. Photobiol. B 2016, 159, 42–48. [Google Scholar] [CrossRef]
- Oni, G.; Brown, S.A.; Kenkel, J.M. Can Fractional Lasers Enhance Transdermal Absorption of Topical Lidocaine in an in Vivo Animal Model? Lasers Surg. Med. 2012, 44, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Taudorf, E.H.; Lerche, C.M.; Erlendsson, A.M.; Philipsen, P.A.; Hansen, S.H.; Janfelt, C.; Paasch, U.; Anderson, R.R.; Haedersdal, M. Fractional Laser-Assisted Drug Delivery: Laser Channel Depth Influences Biodistribution and Skin Deposition of Methotrexate. Lasers Surg. Med. 2016, 48, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.L.; Ortner, V.K.; Haedersdal, M.; Olesen, U.H. Optical Imaging Visualizes a Homogeneous and Horizontal Band-Like Biodistribution of Large- and Small-Size Hydrophilic Compounds Delivered by Ablative Fractional Laser. Pharmaceutics 2022, 14, 1537. [Google Scholar] [CrossRef]
- Haak, C.S.; Hannibal, J.; Paasch, U.; Anderson, R.R.; Haedersdal, M. Laser-Induced Thermal Coagulation Enhances Skin Uptake of Topically Applied Compounds. Lasers Surg. Med. 2017, 49, 582–591. [Google Scholar] [CrossRef]
- Bay, C.; Lerche, C.M.; Ferrick, B.; Philipsen, P.A.; Togsverd-Bo, K.; Haedersdal, M. Comparison of Physical Pretreatment Regimens to Enhance Protoporphyrin IX Uptake in Photodynamic Therapy: A Randomized Clinical Trial. JAMA Dermatol. 2017, 153, 270–278. [Google Scholar] [CrossRef]
- Nieboer, M.J.; Meesters, A.A.; Almasian, M.; Georgiou, G.; de Rie, M.A.; Verdaasdonk, R.M.; Wolkerstorfer, A. Enhanced Topical Cutaneous Delivery of Indocyanine Green after Various Pretreatment Regimens: Comparison of Fractional CO2 Laser, Fractional Er:YAG Laser, Microneedling, and Radiofrequency. Lasers Med. Sci. 2020, 35, 1357–1365. [Google Scholar] [CrossRef]
- Zhao, Y.; Voyer, J.; Li, Y.; Kang, X.; Chen, X. Laser Microporation Facilitates Topical Drug Delivery: A Comprehensive Review about Preclinical Development and Clinical Application. Expert Opin. Drug Deliv. 2023, 20, 31–54. [Google Scholar] [CrossRef]
- Meesters, A.A.; Bakker, M.M.; de Rie, M.A.; Wolkerstorfer, A. Fractional CO2 Laser Assisted Delivery of Topical Anesthetics: A Randomized Controlled Pilot Study. Lasers Surg. Med. 2016, 48, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Wenande, E.; Gundavarapu, S.C.; Tam, J.; Bhayana, B.; Thomas, C.N.; Farinelli, W.A.; Vakoc, B.J.; Anderson, R.R.; Haedersdal, M. Local Vasoregulative Interventions Impact Drug Concentrations in the Skin after Topical Laser-Assisted Delivery. Lasers Surg. Med. 2022, 54, 1288–1297. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, N.J.; Grégoire, S.; Cubberley, R.; Duplan, H.; Eilstein, J.; Ellison, C.; Lester, C.; Fabian, E.; Fernandez, J.; Géniès, C.; et al. Measurement of the Penetration of 56 Cosmetic Relevant Chemicals into and through Human Skin Using a Standardized Protocol. J. Appl. Toxicol. 2020, 40, 403–415. [Google Scholar] [CrossRef]
- Seo, J.-W.; Kim, H.-J.; Song, K.-H. A Comparison of the Efficacy of Ablative Fractional Laser–Assisted Photodynamic Therapy According to Ablative Depth for Actinic Keratosis: A Single-Blinded, Randomized, Comparative, Prospective Study. J. Am. Acad. Dermatol. 2019, 81, 636–638. [Google Scholar] [CrossRef] [PubMed]
- Labadie, J.G.; Ibrahim, S.A.; Worley, B.; Kang, B.Y.; Rakita, U.; Rigali, S.; Arndt, K.A.; Bernstein, E.; Brauer, J.A.; Chandra, S.; et al. Evidence-Based Clinical Practice Guidelines for Laser-Assisted Drug Delivery. JAMA Dermatol. 2022, 158, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-R.; Seo, J.-W.; Kim, H.-J.; Song, K.-H. A Comparison of the Efficacy of Ablative Fractional Laser-Assisted Photodynamic Therapy According to the Density of the Ablative Laser Channel in the Treatment of Actinic Keratosis: A Prospective, Randomized, Controlled Trial. J. Am. Acad. Dermatol. 2021, 85, 750–752. [Google Scholar] [CrossRef]
- Kim, H.-J.; Song, K.-H. Ablative Fractional Laser–Assisted Photodynamic Therapy Provides Superior Long-Term Efficacy Compared with Standard Methyl Aminolevulinate Photodynamic Therapy for Lower Extremity Bowen Disease. J. Am. Acad. Dermatol. 2018, 79, 860–868. [Google Scholar] [CrossRef]
- Vrani, F.; Sotiriou, E.; Lazaridou, E.; Vakirlis, E.; Sideris, N.; Kirmanidou, E.; Apalla, Z.; Lallas, A.; Ioannides, D. Short Incubation Fractional CO 2 Laser-Assisted Photodynamic Therapy vs. Conventional Photodynamic Therapy in Field-Cancerized Skin: 12-Month Follow-up Results of a Randomized Intraindividual Comparison Study. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 79–83. [Google Scholar] [CrossRef]
- Feng, J.; Shen, S.; Song, X.; Xiang, W. Efficacy and Safety of Laser-Assisted Delivery of Tranexamic Acid for the Treatment of Melasma: A Systematic Review and Meta-analysis. J. Cosmet. Laser Ther. 2022, 24, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Hawwam, S.A.; Ismail, M.; El-Attar, Y.A. Split-Face Comparative Study between Intradermal Tranexamic Acid Injection Alone versus Intradermal Tranexamic Acid Injection Combined with Q-Switched Nd:YAG Laser in Melasma Treatment: Dermoscopic and Clinical Evaluation. Lasers Med. Sci. 2022, 37, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Muskat, A.; Kost, Y.; Balazic, E.; Cohen, J.L.; Kobets, K. Laser-Assisted Drug Delivery in the Treatment of Scars, Rhytids, and Melasma: A Comprehensive Review of the Literature. Aesthetic Surg. J. 2023, 43, NP181–NP198. [Google Scholar] [CrossRef] [PubMed]
- Truong, K.; Prasidha, I.; Wain, T. A Systematic Review of Randomised Controlled Trials Investigating Laser Assisted Drug Delivery for the Treatment of Keloid and Hypertrophic Scars. Lasers Med. Sci. 2022, 37, 47–59. [Google Scholar] [CrossRef]
- Ng, W.H.S.; Smith, S.D. Laser-Assisted Drug Delivery: A Systematic Review of Safety and Adverse Events. Pharmaceutics 2022, 14, 2738. [Google Scholar] [CrossRef]
- Prohaska, J.; Hohman, M.H. Laser Complications. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Oni, G.; Rasko, Y.; Kenkel, J. Topical Lidocaine Enhanced by Laser Pretreatment: A Safe and Effective Method of Analgesia for Facial Rejuvenation. Aesthetic Surg. J. 2013, 33, 854–861. [Google Scholar] [CrossRef][Green Version]
- Haedersdal, M.; Erlendsson, A.M.; Paasch, U.; Anderson, R.R. Translational Medicine in the Field of Ablative Fractional Laser (AFXL)-Assisted Drug Delivery: A Critical Review from Basics to Current Clinical Status. J. Am. Acad. Dermatol. 2016, 74, 981–1004. [Google Scholar] [CrossRef]
| Author, Year | Study Design | Evaluation | Patients | Skin Type, % | Topical Agent | Laser Type | Laser Mode | Indication |
|---|---|---|---|---|---|---|---|---|
| Li, Y. et al. (2022) [9] | RCT | Split-face | 37 | III 47% IV 53% | Tranexamic acid 2.5 mL 10% post-laser, and at 12 h, 24 h, 48 h | Picosecond Alexandrite 755 nm | Fluence 0.4 J/cm2, spot size 8 mm, frequency 5 Hz, pulse duration 750 × 10−12 s, 2000 pulses, 10–15% overlap | Melasma, facial rejuvenation |
| Benzaquen M. et al. (2021) [10] | RCT | Split-face | 20 | I 10% II 15% III 50% IV 25% | Hyaluronic Acid 20 mg/mL | Fr CO2 | Spot size 300 μm, power 10 W, density 5%, pulse duration 2.5 ms | Facial rejuvenation |
| Al-Dhalimi M.A et al. (2021) [11] | RCT | Split-face | 25 | III 20% IV 80% | Kojic Acid | Fr Er:YAG | Fluence 0.3 J/cm2, spot size 9 mm, frequency 4 Hz, pulse duration 0.3 ms. | Melasma |
| Park et al. (2021) [12] | PCS | Split-face | 25 | II–IV | Tranexamic acid 3%, Kojic acid 1%, Niacinamide 5% | Q-switched Nd:YAG | Fluence 1.1 J/cm2, spot size 8 mm, frequency 10 Hz | Melasma |
| Wanitphakdeedecha R. et al. (2020) [13] | RCT | Split-face | 46 | III 15% IV 82% V 3% | Tranexamic acid 1 mL 1.2% | Fr Thulium 1927 nm | Fluence 5 mJ, spot size 0.1 mm, pulse duration 1.7 ms, power 3 W | Melasma |
| Widianingsih NPS. et al. (2019) [14] | RCT | Split-face | 9 | NR | Amniotic membrane stem cell metabolite product | Fr Er:YAG | Power 11 J/cm2, 2 multi shots, 0.5 sec interval | Facial rejuvenation (cheek) |
| Ibrahim, O. et al. (2019) [15] | PS | Pre/post | 10 | I–III | Poly-l-Lactic acid | Fr CO2 | Pulse energy 70 mJ, spot size 135 μm, density 10% | Facial rejuvenation (upper lip) |
| Badawi A.M. et al. (2018) [16] | RCT | Split-face | 32 | III 37% IV 40% V 23% | Hydroquinone 4% | Fr Er:YAG | Fluence 250–300 mJ, spot size 7 mm, frequency 4–5 Hz, 30–40 μm ablation depth | Melasma |
| Waibel J.S. et al. (2016) [17] | RCT | Split-face | 15 | NR | Vit C 15%, Vit E 1%, ferulic acid 0.5%, post-laser and 7 d | Fr CO2 | Pulse energy 90–125 mJ, spot size 10 mm, density 68% | Post-laser recovery |
| Mahmoud et al. (2015) [18] | RCT | Split-face | 10 | II–III | Botulinum toxin A | Fr CO2 | Pulse energy 100 mJ, size 6 (10 mm2), density 4 (100% coverage) | Facial rejuvenation (crow’s feet) |
| Gawdat et al. (2014) [19] | RCT | Split-face | 30 | III–V | ID PRP and topical PRP 2 mL | Fr CO2 | Power 15 W, dwell time 600 μs, spacing 700 μm, smart stack level 2 | Atrophic acne scars |
| Yun P.L. et al. (2002) [8] | RCT | Split-face | 12 | I–III | Lidocaine 30 g 5% cream | Er:YAG | Fluence 1.3 J/cm2, | Pre-laser analgesia |
| Author, Year | Indication | Evaluation Method | Duration, Evaluation Time | Outcomes | Complications |
|---|---|---|---|---|---|
| Li et al. (2022) [9] | 1. Melasma 2. Facial rejuvenation | MASI score, photographs, PROM (VAS, question.) | 3 sessions at 4–5 w intervals 1, 3, 6 m follow-up | Combi/monotherapy vs. baseline: MASI scores, dyschromia, skin texture. Significant decrease at all f/ups. Mono vs. combi therapy: SI at 1.3 m–NS at 6 m. Mono/combi therapy vs. baseline: Laxity, rhytids NS. Satisfaction higher in combi-half at 1 m. | Erythema, Dryness, PIH, Scaling: NS Irritation on combi-half.Redness, sensitivity higher on days 1–3 for combi-half. |
| Benzaquen et al. (2021) [10] | Facial rejuvenation | Clinical, photographs, PROM (VAS) | 1 session 3 m follow-up | NS difference in skin texture, firmness, radiance, and fine lines. | Erythema, edema, crusting NS. |
| Al-Dhalimi et al. (2021) [11] | Melasma | MASI score, photographs, PROM | 6 sessions at 2 w intervals 3 m follow-up | Combi/monotherapy vs. baseline: MASI scores SI. Combi vs. monotherapy: MASI NS, PGA, and patient satisfaction SI at 3 m. | Erythema, tingling/stinging sensation NS. |
| Park et al. (2021) [12] | Melasma | MASI score, photographs, PROM | 5 sessions at 2 w intervals 1 m follow up | Combi vs. monotherapy: hemi-MASI SI. Patient satisfaction SI at 1 m. | Self-limiting erythema NS. |
| Wanitphakdeedecha et al. (2020) [13] | Melasma | mMASI/MI score, photo-graphs, PROM (VAS) | 4 sessions at 1 w intervals 6 m follow-up | Combi/monotherapy vs. baseline: mMASI, MI scores SI at all time points (1 w-6 m). Combi vs. monotherapy: MI SI at 6 m, mMASI NS. Patient satisfaction SI both sides at all time points. | Mild PIH NS (4 TXA, 3 control side). |
| Widianingsih et al. (2019) [14] | Facial rejuvenation | Photographs, skin analyzer | 3 sessions at 4 w intervals 3 m follow-up | Pores, wrinkles, pigmentation, skin tone. NS improvement in Combi vs. monotherapy (slightly better on AMSC side). | Erythema, mild pain, acne eruption NS. |
| Ibrahim et al. (2019) [15] | Facial rejuvenation | Photographs, PROM | 3 sessions at 2 m intervals 3 m follow-up | SI clinically, post-treatment by both blinded raters and patients. | 2 patients withdrew due to exuberant erythematous response, and prolonged erythema after 1st session. |
| Badawi et al. (2018) [16] | Melasma | MASI score, photographs, dermoscopy, PROM | 6 sessions at 2 w intervals 2 w, 14 w follow-up | Combi/monotherapy vs. baseline: SI at 2 w. Combi vs. monotherapy: MASI SI at 2 w. | Erythema, crusting on laser side. Burning sensation, itching NS. Recurrence 2 pts on both facial sides. |
| Waibel et al. (2016) [17] | Post-laser recovery | Photographs, PROM (question.) | 1 session 1 w follow-up | Combi vs. monotherapy: NS clinical difference. bFGF statistical difference on combi-side. | No side effects are mentioned. |
| Mahmoud et al. (2015) [18] | Facial rejuvenation | Photographs, PROM (question.) | 1 session 1 m follow-up | Dynamic rhytids: SI only for combi-side.Static rhytids: NS for both sides. Patient satisfaction SI combi-side at 1 m. | Pain, erythema, swelling (NS between sides). |
| Gawdat et al. (2014) [19] | Atrophic Acne Scars | Photographs, OCT, PROM (scale) | 3 sessions at 1 m intervals 4 m follow-up | Clinical improvement, OCT significant in combi vs. monotherapy. NS difference in the improvement grade, downtime between Intradermal and topical PRP. | Erythema, edema, crusting, PIH, acneform eruption SI on combi-side. Pain significantly greater for Laser +ID than +topical PRP or laser alone. |
| Yun et al. (2002) [8] | Pre-laser analgesia | PROM (VAS) | 1 session post each laser pass, and at 3 h | Combi-treatment superior, especially during the second pass. NS pain difference at 3 h. | No complications. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seretis, K.; Papaioannou, K. Laser-Assisted Drug Delivery on the Face: A Systematic Review of Indications and Outcomes. Cosmetics 2023, 10, 122. https://doi.org/10.3390/cosmetics10050122
Seretis K, Papaioannou K. Laser-Assisted Drug Delivery on the Face: A Systematic Review of Indications and Outcomes. Cosmetics. 2023; 10(5):122. https://doi.org/10.3390/cosmetics10050122
Chicago/Turabian StyleSeretis, Konstantinos, and Konstantina Papaioannou. 2023. "Laser-Assisted Drug Delivery on the Face: A Systematic Review of Indications and Outcomes" Cosmetics 10, no. 5: 122. https://doi.org/10.3390/cosmetics10050122
APA StyleSeretis, K., & Papaioannou, K. (2023). Laser-Assisted Drug Delivery on the Face: A Systematic Review of Indications and Outcomes. Cosmetics, 10(5), 122. https://doi.org/10.3390/cosmetics10050122






