Modulation of Cutaneous Carotenoid Content via Ozone Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. Skin Carotenoid Quantification
2.2. Ozone Exposure
3. Results
3.1. Cutaneous Carotenoid Content
3.2. SCS Variations in Relation to the Portions of Fruits and Vegetables Consumed
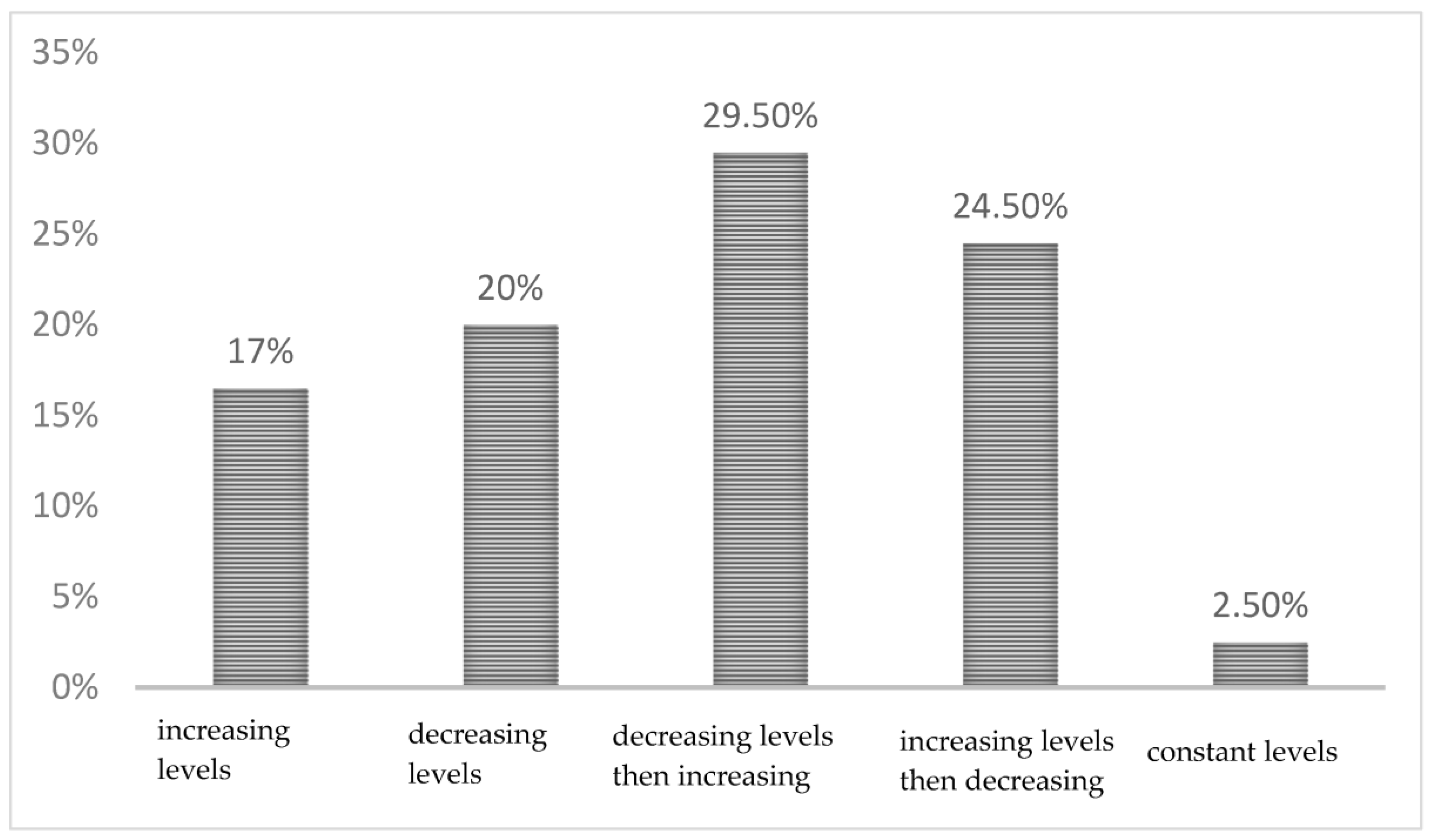
3.3. Variation in Carotenoids on the Whole Group after 15 min of Ozone Treatment
3.4. Variation in SCS Values Following Ozone Treatment in the Smoker and Non-Smoker Groups
3.5. Variations in Expected Carotenoids Based on the Consumption of Fruits and Vegetables
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carslaw, K.S.; Lee, L.A.; Reddington, C.L.; Pringle, K.J.; Rap, A.; Forster, P.M.; Mann, G.W.; Spracklen, D.V.; Woodhouse, M.T.; Regayre, L.A.; et al. Large contribution of natural aerosols to uncertainty in indirect forcing. Nature 2013, 503, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Chiu, Y.-H.; Li, Y.; Lin, T.-Y. Air Pollutant and Health-Efficiency Evaluation Based on a Dynamic Network Data Envelopment Analysis. Int. J. Environ. Res. Public Health 2018, 15, 2046. [Google Scholar] [CrossRef]
- Bais, A.F.; Bernhard, G.; McKenzie, R.L.; Aucamp, P.J.; Young, P.J.; Ilyas, M.; Jöckel, P.; Deushi, M. Ozone—climate interactions and effects on solar ultraviolet radiation. Photochem. Photobiol. Sci. 2019, 18, 602–640. [Google Scholar] [CrossRef]
- Ritz, B.; Hoffmann, B.; Peters, A. The Effects of Fine Dust, Ozone, and Nitrogen Dioxide on Health. Dtsch. Arztebl. Int. 2019, 51–52, 881–886. [Google Scholar] [CrossRef]
- Bassett, D.; Elbon-Copp, C.; Otterbein, S.; Barraclough-Mitchell, H.; Delorme, M.; Yang, H. Inflammatory cell availability affects ozone-induced lung damage. J. Toxicol. Environ. Health Part A 2001, 64, 547–565. [Google Scholar] [CrossRef] [PubMed]
- Khatri, S.B.; Peabody, J.; Burwell, L.; Harris, F.; Brown, L.S. Systemic Antioxidants and Lung Function in Asthmatics during High Ozone Season: A Closer Look at Albumin, Glutathione, and Associations with Lung Function. Clin. Transl. Sci. 2014, 7, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Staehelin, J. The two ozone problems: Too much in the troposphere, too little in the stratosphere. Schweiz. Rundsch. Med. Prax. Rev. Suisse Med. Prax. 1992, 81, 315–321. [Google Scholar]
- Valacchi, G.; van der Vliet, A.; Schock, B.C.; Okamoto, T.; Obermuller-Jevic, U.; E Cross, C.; Packer, L. Ozone exposure activates oxidative stress responses in murine skin. Toxicology 2002, 179, 163–170. [Google Scholar] [CrossRef]
- Valacchi, G.; Pagnin, E.; Okamoto, T.; Corbacho, A.M.; Olano, E.; Davis, P.A.; van der Vliet, A.; Packer, L.; Cross, C.E. Induction of stress proteins and MMP-9 by 0.8ppm of ozone in murine skin. Biochem. Biophys. Res. Commun. 2003, 305, 741–746. [Google Scholar] [CrossRef]
- Valacchi, G.; Fortino, V.; Bocci, V. The dual action of ozone on the skin. Br. J. Dermatol. 2005, 153, 1096–1100. [Google Scholar] [CrossRef]
- Fortino, V.; Maioli, E.; Torricelli, C.; Davis, P.; Valacchi, G. Cutaneous MMPs are differently modulated by environmental stressors in old and young mice. Toxicol. Lett. 2007, 173, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Pecorelli, A.; McDaniel, D.H.; Wortzman, M.; Nelson, D.B. Protective effects of a comprehensive topical antioxidant against ozone-induced damage in a reconstructed human skin model. Arch. Dermatol. Res. 2021, 313, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Fuks, K.B.; Hüls, A.; Sugiri, D.; Altug, H.; Vierkötter, A.; Abramson, M.J.; Goebel, J.; Wagner, G.G.; Demuth, I.; Schikowski, T.; et al. Tropospheric ozone and skin aging: Results from two German cohort studies. Environ. Int. 2019, 124, 139–144. [Google Scholar] [CrossRef]
- Césarini, J.P.; Michel, L.; Maurette, J.M.; Adhoute, H.; Béjot, M. Immediate effects of UV radiation on the skin: Modification by an antioxidant complex containing carotenoids. Photodermatol. Photoimmunol. Photomed. 2003, 19, 182–189. [Google Scholar] [CrossRef]
- Lademann, J.; Meinke, M.C.; Sterry, W.; Darvin, M.E. Carotenoids in human skin. Exp. Dermatol. 2011, 20, 377–382. [Google Scholar] [CrossRef]
- Udensi, J.; Loughman, J.; Loskutova, E.; Byrne, H.J. Raman Spetroscopy of Carotenoids. compounds for clinical applications. Molecules 2022, 27, 9017. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, P.; Surmann, P.; Vallentin, G.; Fuhrmann, H. A small-scale method for quantitation of carotenoids in bacteria and yeasts. J. Microbiol. Methods 2007, 70, 142–149. [Google Scholar] [CrossRef]
- Fielding, J.M.; Rowley, K.G.; Cooper, P.; O’Dea, K. Increases in plasma lycopene concentration after consumption of tomatoes cooked with olive oil. Asia Pac. J. Clin. Nutr. 2005, 14, 131–136. [Google Scholar]
- Bohn, T. Carotenoids and Markers of Oxidative Stress in Human Observational Studies and Intervention Trials: Implications for Chronic Diseases. Antioxidants 2019, 8, 179. [Google Scholar] [CrossRef]
- Sticozzi, C.; Pecorelli, A.; Romani, A.; Belmonte, G.; Cervellati, F.; Maioli, E.; Lila, M.A.; Cervellati, C.; Valacchi, G. Tropospheric ozone affects SRB1 levels via oxidative post-translational modifications in lung cells. Free Radic. Biol. Med. 2018, 126, 287–295. [Google Scholar] [CrossRef]
- Perrone, A.; Pintaudi, A.M.; Traina, A.; Carruba, G.; Attanzio, A.; Gentile, C.; Tesoriere, L.; Livrea, M.A. Raman Spectroscopic Measurements of Dermal Carotenoids in Breast Cancer Operated Patients Provide Evidence for the Positive Impact of a Dietary Regimen Rich in Fruit and Vegetables on Body Oxidative Stress and BC Prognostic Anthropometric Parameters: A Five-Year Study. Oxid. Med. Cell Longev. 2016. [Google Scholar] [CrossRef]
- Valacchi, G.; Pecorelli, A.; Belmonte, G.; Pambianchi, E.; Cervellati, F.; Lynch, S.; Krol, Y.; Oresajo, C. Protective Effects of Topical Vitamin C Compound Mixtures against Ozone-Induced Damage in Human Skin. J. Investig. Dermatol. 2017, 137, 1373–1375. [Google Scholar] [CrossRef] [PubMed]
- Packer, L.; Valacchi, G. Antioxidants and the Response of Skin to Oxidative Stress: Vitamin E as a Key Indicator. Ski. Pharmacol. Physiol. 2002, 15, 282–290. [Google Scholar] [CrossRef]
- Valacchi, G.; Sticozzi, C.; Zanardi, I.; Belmonte, G.; Cervellati, F.; Bocci, V.; Travagli, V. Ozone mediators effect on “in vitro” scratch wound closure. Free Radic. Res. 2016, 50, 1022–1031. [Google Scholar] [CrossRef]
- Mustafa, M.G. Biochemical basis of ozone toxicity. Free Radic. Biol. Med. 1990, 9, 245–265. [Google Scholar] [CrossRef]
- Moran, N.E.; Chang, J.; Stroh, R.; Zaidi, Y.; Hason, N.; Musaad, S.; O’connor, T. Noninvasive Reflection Spectroscopy Measurement of Skin Carotenoid Score in Infants Is Feasible and Reliable. J. Nutr. 2022, 152, 2966–2977. [Google Scholar] [CrossRef]
- Massenti, R.; Perrone, A.; Livrea, M.A.; Bianco, R.L. Regular consumption of fresh orange juice increases human skin carotenoid content. Int. J. Food Sci. Nutr. 2015, 66, 718–721. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, S.S.; Wengreen, H.J.; Dew, J. Skin Carotenoid Response to a High-Carotenoid Juice in Children: A Randomized Clinical Trial. J. Acad. Nutr. Diet. 2015, 115, 1771–1778. [Google Scholar] [CrossRef]
- Zidichouski, J.A.; Mastaloudis, A.; Poole, S.J.; Reading, J.C.; Smidt, C.R. Clinical Validation of a Noninvasive, Raman Spectroscopic Method to Assess Carotenoid Nutritional Status in Humans. J. Am. Coll. Nutr. 2009, 28, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Mastaloudis, A.; Subramaniam, P.; Wood, S.M.; Hester, S.N.; McGlade, E.C.; Renshaw, P.F.; Yurgelun-Todd, D.A. Supplementation with a putative calorie restriction mimetic micronutrient blend increases glutathione concentrations and improves neuroenergetics in brain of healthy middle-aged men and women. Free Radic. Biol. Med. 2020, 153, 112–121. [Google Scholar] [CrossRef]
- Mayne, S.T.; Cartmel, B.; Scarmo, S.; Lin, H.; Leffell, D.J.; Welch, E.; Ermakov, I.; Bhosale, P.; Bernstein, P.S.; Gellermann, W. Noninvasive assessment of dermal carotenoids as a biomarker of fruit and vegetable intake. Am. J. Clin. Nutr. 2010, 92, 794–800. [Google Scholar] [CrossRef]
- Ermakov, I.V.; Gellermann, W. Validation model for Raman based skin carotenoid detection. Arch. Biochem. Biophys. 2010, 504, 40–49. [Google Scholar] [CrossRef]
- Ermakov, I.V.; Ermakova, M.; Sharifzadeh, M.; Gorusupudi, A.; Farnsworth, K.; Bernstein, P.S.; Stookey, J.; Evans, J.; Arana, T.; Tao-Lew, L.; et al. Optical assessment of skin carotenoid status as a biomarker of vegetable and fruit intake. Arch. Biochem. Biophys. 2018, 646, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Chełchowska, M.; Laskowska-Klita, T.; Szymborski, J. Level of retinol and beta-carotene in plasma of smoking and non-smoking women. Wiad Lek. 2001, 54, 248–254. [Google Scholar] [PubMed]
- Coronel, J.; Pinos, I.; Amengual, J. β-carotene in Obesity Research: Technical Considerations and Current Status of the Field. Nutrients 2019, 11, 842. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.; Park, S.; Kim, H.; Kwon, O. Association of Carotenoids Concentration in Blood with Physical Performance in Korean Adolescents: The 2018 National Fitness Award Project. Nutrients 2020, 18, 1821. [Google Scholar] [CrossRef] [PubMed]
- Pryor, W.A. Mechanisms of radical formation from reactions of ozone with target molecules in the lung. Free Radic. Biol. Med. 1994, 17, 451–465. [Google Scholar] [CrossRef]
- Weber, C.; Erl, W.; Weber, K.S.C.; Weber, P.C. Effects of Oxidized Low Density Lipoprotein, Lipid Mediators and Statins on Vascular Cell Interactions. Clin. Chem. Lab. Med. 1999, 37, 243–254. [Google Scholar] [CrossRef]
- Valacchi, G.; Weber, S.U.; Luu, C.; Cross, C.E.; Packer, L. Ozone potentiates vitamin E depletion by ultraviolet radiation in the murine stratum corneum. FEBS Lett. 2000, 466, 165–168. [Google Scholar] [CrossRef]
- Valacchi, G.; Virgili, F.; Cervellati, C.; Pecorelli, A. OxInflammation: From Subclinical Condition to Pathological Biomarker. Front. Physiol. 2018, 9, 858. [Google Scholar] [CrossRef]
- Valacchi, G.; Pecorelli, A.; Mencarelli, M.; Maioli, E.; Davis, P. Beta-carotene prevents ozone-induced proinflammatory markers in murine skin. Toxicol. Ind. Health 2009, 25, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.E.; Mohn, E.S.; Hason, N.; Erdman, J.W., Jr.; Johnson, E.J. Intrinsic and Extrinsic Factors Impacting Absorption, Metabolism, and Health Effects of Dietary Carotenoids. Adv. Nutr. 2018, 9, 465–492. [Google Scholar] [CrossRef] [PubMed]
- Smeland, S.; Bjerknes, T.; Malaba, L.; Eskild, W.; Norum, K.R.; Blomhoff, R. Tissue distribution of the receptor for plasma retinol-binding protein. Biochem. J. 1995, 305, 419–424. [Google Scholar] [CrossRef]
- Ermakov, I.V.; Sharifzadeh, M.; Ermakova, M.; Gellermann, W. Resonance Raman detection of carotenoid antioxidants in living human tissue. J. Biomed. Opt. 2005, 10, 064028. [Google Scholar] [CrossRef] [PubMed]
- Shephard, R.J.; Urch, B.; Silverman, F.; Corey, P.N. Interaction of ozone and cigarette. Environ. Res. 1983, 31, 125–137. [Google Scholar] [CrossRef]
- Darvin, M.E.; Sterry, W.; Lademann, J.; Vergou, T. The Role of Carotenoids in Human Skin. Molecules 2011, 16, 10491–10506. [Google Scholar] [CrossRef]






| N (%) | T0 h SCS (Average ± DS) | p-Value | |
|---|---|---|---|
| Gender | |||
| Male | 32 | 48,288 ± 13,667 | N.S. |
| Female | 68 | 50,395 ± 15,487 | |
| BMI | |||
| Normal weight | 86 | 49,032 ± 15,614 | p < 0.05 |
| Overweight | 14 | 49,066 ± 12,452 | |
| Smoker | |||
| Yes | 21 | 43,181 ± 14,555 | p < 0.05 |
| No | 79 | 50,411 ± 14,977 | |
| Exercise | |||
| Yes | 44 | 46,334 ± 10,182 | p < 0.05 |
| No | 56 | 50,950 ± 17,075 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervellati, F.; Benedusi, M.; Mastaloudis, A.; Nagliati, V.; Valacchi, G. Modulation of Cutaneous Carotenoid Content via Ozone Exposure. Cosmetics 2023, 10, 97. https://doi.org/10.3390/cosmetics10040097
Cervellati F, Benedusi M, Mastaloudis A, Nagliati V, Valacchi G. Modulation of Cutaneous Carotenoid Content via Ozone Exposure. Cosmetics. 2023; 10(4):97. https://doi.org/10.3390/cosmetics10040097
Chicago/Turabian StyleCervellati, Franco, Mascia Benedusi, Angela Mastaloudis, Vittoria Nagliati, and Giuseppe Valacchi. 2023. "Modulation of Cutaneous Carotenoid Content via Ozone Exposure" Cosmetics 10, no. 4: 97. https://doi.org/10.3390/cosmetics10040097
APA StyleCervellati, F., Benedusi, M., Mastaloudis, A., Nagliati, V., & Valacchi, G. (2023). Modulation of Cutaneous Carotenoid Content via Ozone Exposure. Cosmetics, 10(4), 97. https://doi.org/10.3390/cosmetics10040097








