Vitiligo: A Review of Aetiology, Pathogenesis, Treatment, and Psychosocial Impact
Abstract
1. Introduction
2. Psychosocial Aspects of Vitiligo and Associated Disorders
2.1. Psychological Disorders
2.2. Systemic Disorders
2.2.1. Thyroid Disorders
2.2.2. Other Disorders
3. Aetiology and Pathogenesis
3.1. Autoimmune Theory
3.2. Genetic Theory
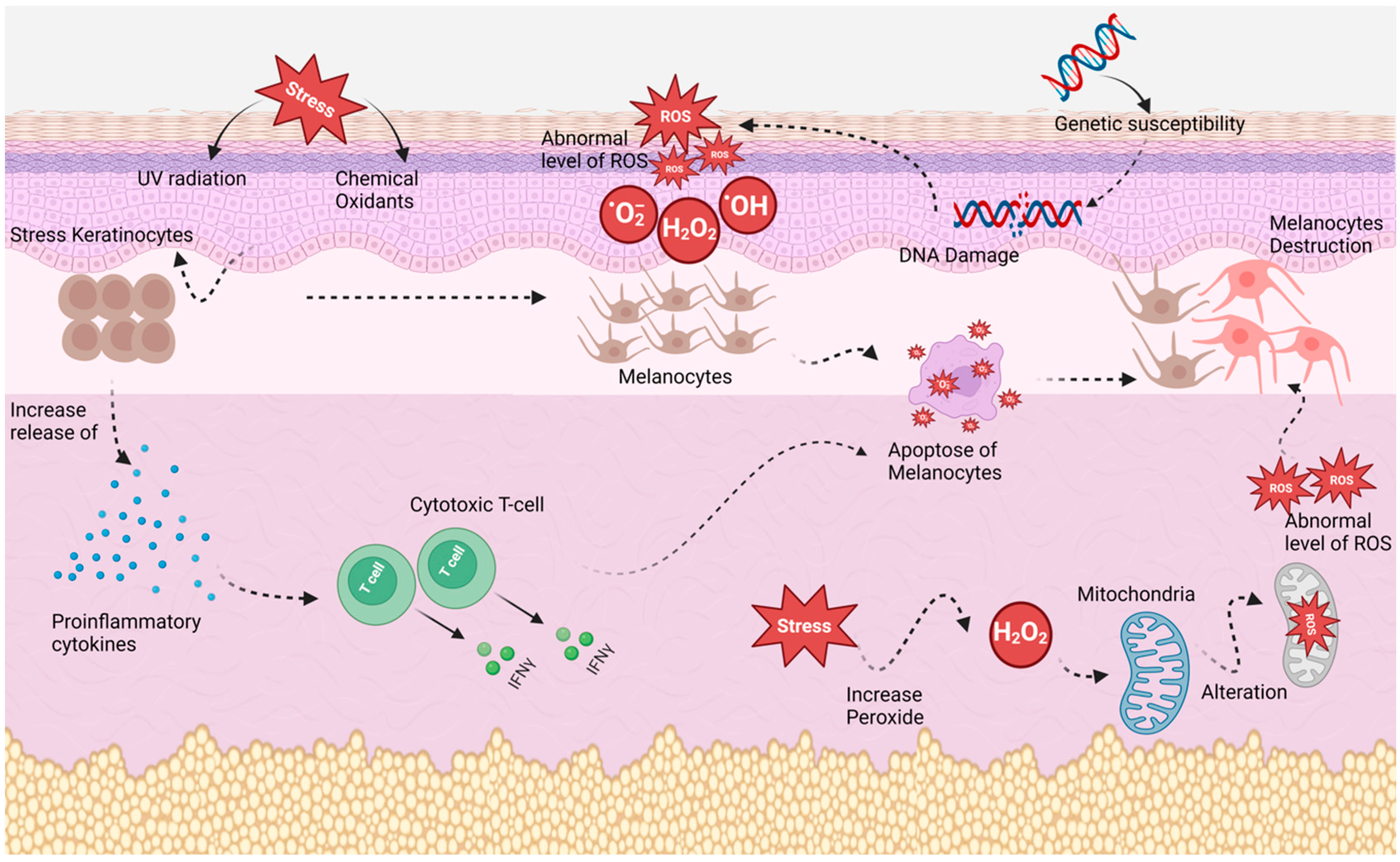
3.3. Oxidative Stress Theory
3.4. Neural Theory
3.5. Biochemical Theory
4. Classification
4.1. Segmental Vitiligo
4.2. Non-Segmental Vitiligo
4.3. Unclassified/Mixed Vitiligo
5. Diagnosis and Assessment of Severity
6. Vitiligo and Skin Cancer Connection
7. Treatment Options for Vitiligo
7.1. Pharmacotherapy
7.1.1. Immunomodulators
7.1.2. Vitamin D Analogues
7.2. Phototherapy
7.3. Surgery
7.4. Cosmetic Camouflage and Aesthetic Therapy
7.4.1. Temporary Camouflage
7.4.2. Permanent Camouflage
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arthi, G.; Lakshmanakumar, V.; Rathinam, J.; Sivaraman, D. Molecular docking analysis to validate the efficacy of phytocompounds of traditional siddha herb Glycyrrhiza glabra against enzyme tyrosinase for the treatment of vitiligo. TMR Integr. Med. 2022, 6, e22032. [Google Scholar] [CrossRef]
- Prasad, P.V.; Bhatnagar, V.K. Medico-historical study of “Kilasa”(vitiligo/leucoderma) a common skin disorder. Bull. Indian Inst. Hist. Med. 2003, 33, 113–127. [Google Scholar]
- Czajkowski, R.; Męcińska-Jundziłł, K. Current aspects of vitiligo genetics. Adv. Dermatol. Allergol. Postępy Dermatol. I Alergol. 2014, 31, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Medicine, S.P.C.C.J.o. Vitiligo. Clevel. Clin. J. Med. 2022. Available online: https://my.clevelandclinic.org/health/diseases/12419-vitiligo (accessed on 24 March 2023).
- Hartmann, A.; Lurz, C.; Hamm, H.; Bröcker, E.B.; Hofmann, U.B. Narrow-band UVB311 nm vs. broad-band UVB therapy in combination with topical calcipotriol vs. placebo in vitiligo. Int. J. Dermatol. 2005, 44, 736–742. [Google Scholar] [CrossRef]
- Mohr, N.; Petersen, J.; Kirsten, N.; Augustin, M. Epidemiology of Vitiligo–A Dual Population-Based Approach. Clin. Epidemiol. 2021, 13, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Marinho, F.d.S.; Cirino, P.V.; Fernandes, N.C. Clinical epidemiological profile of vitiligo in children and adolescents. An. Bras. Dermatol. 2013, 88, 1026–1028. [Google Scholar] [CrossRef] [PubMed]
- Krüger, C.; Schallreuter, K.U. A review of the worldwide prevalence of vitiligo in children/adolescents and adults. Int. J. Dermatol. 2012, 51, 1206–1212. [Google Scholar] [CrossRef]
- Zhang, Y.; Cai, Y.; Shi, M.; Jiang, S.; Cui, S.; Wu, Y.; Gao, X.-H.; Chen, H.-D. The prevalence of vitiligo: A meta-analysis. PLoS ONE 2016, 11, e0163806. [Google Scholar] [CrossRef]
- Kumar, S.; Nayak, C.S.; Padhi, T.; Rao, G.; Rao, A.; Sharma, V.K.; Srinivas, C.R. Epidemiological pattern of psoriasis, vitiligo and atopic dermatitis in India: Hospital-based point prevalence. Indian Dermatol. Online J. 2014, 5, S6. [Google Scholar] [CrossRef]
- Ayanlowo, O.; Olumide, Y.M.; Akinkugbe, A.; Ahamneze, N.; Otike-Odibi, B.I.; Ekpudu, V.I.; Nnaji, T.; Akolawole, N.A. Characteristics of vitiligo in Lagos, Nigeria. West Afr. J. Med. 2009, 28, 118–121. [Google Scholar]
- Behl, P.N.; Bhatia, R.K. 400 cases of vitiligo. A clinico-therapeutic analysis. Indian J. Dermatol. 1972, 17, 51–56. [Google Scholar]
- Sehgal, V.; Srivastava, G. Vitiligo: Compendium of clinico-epidemiological features. Indian J. Dermatol. Venereol. Leprol. 2007, 73, 149–156. [Google Scholar] [CrossRef]
- Alkhateeb, A.; Fain, P.R.; Thody, A.; Bennett, D.C.; Spritz, R.A. Epidemiology of vitiligo and associated autoimmune diseases in Caucasian probands and their families. Pigment Cell Res. 2003, 16, 208–214. [Google Scholar] [CrossRef]
- Alikhan, A.; Felsten, L.M.; Daly, M.; Petronic-Rosic, V. Vitiligo: A comprehensive overview: Part I. Introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J. Am. Acad. Dermatol. 2011, 65, 473–491. [Google Scholar] [CrossRef]
- Nejad, S.B.; Qadim, H.H.; Nazeman, L.; Fadaii, R.; Goldust, M. Frequency of autoimmune diseases in those suffering from vitiligo in comparison with normal population. Pak. J. Biol. Sci. PJBS 2013, 16, 570–574. [Google Scholar] [CrossRef]
- Hann, S.K.; Lee, H.J. Segmental vitiligo: Clinical findings in 208 patients. J. Am. Acad. Dermatol. 1996, 35, 671–674. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef]
- Weinstock, M.A. Assessment of sun sensitivity by questionnaire: Validity of items and formulation of a prediction rule. J. Clin. Epidemiol. 1992, 45, 547–552. [Google Scholar] [CrossRef]
- Silverberg, J.I.; Silverberg, A.I.; Malka, E.; Silverberg, N.B. A pilot study assessing the role of 25 hydroxy vitamin D levels in patients with vitiligo vulgaris. J. Am. Acad. Dermatol. 2010, 62, 937–941. [Google Scholar] [CrossRef]
- Bibeau, K.; Pandya, A.G.; Ezzedine, K.; Jones, H.; Gao, J.; Lindley, A.; Harris, J.E. Vitiligo prevalence and quality of life among adults in Europe, Japan and the USA. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1831–1844. [Google Scholar] [CrossRef]
- Jayaprakasam, A.; Darvay, A.; Osborne, G.; McGibbon, D. Comparison of assessments of severity and quality of life in cutaneous disease. Clin. Exp. Dermatol. 2002, 27, 306–308. [Google Scholar] [CrossRef]
- Jowett, S.; Ryan, T. Skin disease and handicap: An analysis of the impact of skin conditions. Soc. Sci. Med. 1985, 20, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Silverberg, N.B. Association between vitiligo extent and distribution and quality-of-life impairment. JAMA Dermatol. 2013, 149, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Morrison, B.; Burden-Teh, E.; Batchelor, J.M.; Mead, E.; Grindlay, D.; Ratib, S. Quality of life in people with vitiligo: A systematic review and meta-analysis. Br. J. Dermatol. 2017, 177, e338–e339. [Google Scholar] [CrossRef] [PubMed]
- Korkij, W.; Solatani, K.; Simjee, S.; Marcincin, P.G.; Chuang, T.Y. Tissue-specific autoantibodies and autoimmune disorders in vitiligo and alopecia areata: A retrospective study. J. Cutan. Pathol. 1984, 11, 522–530. [Google Scholar] [CrossRef]
- Hegedüs, L.; Heidenheim, M.; Gervil, M.; Hjalgrim, H.; Høier-Madsen, M. High frequency of thyroid dysfunction in patients with vitiligo. Acta Derm. Venereol. 1994, 74, 120–123. [Google Scholar]
- Daneshpazhooh, M.; Mostofizadeh, G.M.; Behjati, J.; Akhyani, M.; Robati, R.M. Anti-thyroid peroxidase antibody and vitiligo: A controlled study. BMC Dermatol. 2006, 6, 3. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, S.-X.; Zhang, F.-Y.; Yin, X.-Y.; Yang, S.; Xiao, F.-L.; Du, W.-H.; Wang, J.-F.; Lv, Y.-M.; Tang, H.-Y. The analysis of genetics and associated autoimmune diseases in Chinese vitiligo patients. Arch. Dermatol. Res. 2009, 301, 167–173. [Google Scholar] [CrossRef]
- Laberge, G.; Mailloux, C.M.; Gowan, K.; Holland, P.; Bennett, D.C.; Fain, P.R.; Spritz, R.A. Early disease onset and increased risk of other autoimmune diseases in familial generalized vitiligo. Pigment Cell Res. 2005, 18, 300–305. [Google Scholar] [CrossRef]
- Birlea, S.A.; Fain, P.R.; Spritz, R.A. A Romanian population isolate with high frequency of vitiligo and associated autoimmune diseases. Arch. Dermatol. 2008, 144, 310–316. [Google Scholar] [CrossRef]
- Jin, Y.; Mailloux, C.M.; Gowan, K.; Riccardi, S.L.; LaBerge, G.; Bennett, D.C.; Fain, P.R.; Spritz, R.A. NALP1 in vitiligo-associated multiple autoimmune disease. N. Engl. J. Med. 2007, 356, 1216–1225. [Google Scholar] [CrossRef]
- Ezzedine, K.; Lim, H.W.; Suzuki, T.; Katayama, I.; Hamzavi, I.; Lan, C.C.E.; Goh, B.K.; Anbar, T.; Silva de Castro, C.; Lee, A.Y. Revised classification/nomenclature of vitiligo and related issues: The Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res. 2012, 25, E1–E13. [Google Scholar] [CrossRef]
- Kemp, E.H.; Waterman, E.A.; Weetman, A.P. Autoimmune aspects of vitiligo. Autoimmunity 2001, 34, 65–77. [Google Scholar] [CrossRef]
- Dwivedi, M.; Kemp, E.H.; Laddha, N.C.; Mansuri, M.S.; Weetman, A.P.; Begum, R. Regulatory T cells in vitiligo: Implications for pathogenesis and therapeutics. Autoimmun. Rev. 2015, 14, 49–56. [Google Scholar] [CrossRef]
- Arora, A.K.; Kumaran, M.S. Pathogenesis of vitiligo: An update. Pigment Int. 2017, 4, 65. [Google Scholar]
- He, S.; Xu, J.; Wu, J. The promising role of chemokines in vitiligo: From oxidative stress to the autoimmune response. Oxidative Med. Cell. Longev. 2022, 2022, 8796735. [Google Scholar] [CrossRef]
- Ezzedine, K.; Le Thuaut, A.; Jouary, T.; Ballanger, F.; Taieb, A.; Bastuji-Garin, S. Latent class analysis of a series of 717 patients with vitiligo allows the identification of two clinical subtypes. Pigment Cell Melanoma Res. 2014, 27, 134–139. [Google Scholar] [CrossRef]
- Reghu, R.; James, E. Epidemiological profile and treatment pattern of vitiligo in a tertiary care teaching hospital. Children 2011, 2, 2–5. [Google Scholar]
- Boniface, K.; Seneschal, J.; Picardo, M.; Taïeb, A. Vitiligo: Focus on clinical aspects, immunopathogenesis, and therapy. Clin. Rev. Allergy Immunol. 2018, 54, 52–67. [Google Scholar] [CrossRef]
- Passeron, T.; Ortonne, J.-P. Physiopathology and genetics of vitiligo. J. Autoimmun. 2005, 25, 63–68. [Google Scholar] [CrossRef]
- Wolff, K.; Goldsmith, L.A.; Katz, S.I.; Gilchrest, B.A.; Paller, A.S.; Leffell, D.J. Fitzpatrick’s Dermatology in General Medicine; McGraw-Hill: New York, NY, USA, 2008. [Google Scholar]
- Picardo, M.; Dell’Anna, M.L.; Ezzedine, K.; Hamzavi, I.; Harris, J.E.; Parsad, D.; Taieb, A. Vitiligo (Primer). Nat. Rev. Dis. Prim. 2015, 1, 1–16. [Google Scholar] [CrossRef]
- Ezzedine, K.; Diallo, A.; Léauté-Labrèze, C.; Séneschal, J.; Prey, S.; Ballanger, F.; Alghamdi, K.; Cario-André, M.; Jouary, T.; Gauthier, Y. Halo naevi and leukotrichia are strong predictors of the passage to mixed vitiligo in a subgroup of segmental vitiligo. Br. J. Dermatol. 2012, 166, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Ezzedine, K.; Gauthier, Y.; Léauté-Labrèze, C.; Marquez, S.; Bouchtnei, S.; Jouary, T.; Taieb, A. Segmental vitiligo associated with generalized vitiligo (mixed vitiligo): A retrospective case series of 19 patients. J. Am. Acad. Dermatol. 2011, 65, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Lotti, T.; Gori, A.; Zanieri, F.; Colucci, R.; Moretti, S. Vitiligo: New and emerging treatments. Dermatol. Ther. 2008, 21, 110–117. [Google Scholar] [CrossRef]
- Lakhani, R.; Prakash, C.; Tiwari, S.; Purohit, S.; Paliwal, V.; Mathur, D.K.; Bhargava, P. Scoring system in dermatology: A review. IOSR 2016, 15, 89–99. [Google Scholar] [CrossRef]
- Oulehri, A.; Elloudi, S.; Baybay, H.; Douhi, Z.; Mernissi, F.Z. Vitiligo and skin cancer: What’s the link? Our Derm. Online 2020, 11, e144. [Google Scholar] [CrossRef]
- Staples, M.P.; Elwood, M.; Burton, R.C.; Williams, J.L.; Marks, R.; Giles, G.G. Non-melanoma skin cancer in Australia: The 2002 national survey and trends since 1985. Med. J. Aust. 2006, 184, 6–10. [Google Scholar] [CrossRef]
- Gloster, H.M., Jr.; Neal, K. Skin cancer in skin of color. J. Am. Acad. Dermatol. 2006, 55, 741–760. [Google Scholar] [CrossRef]
- Rodrigues, M. Skin cancer risk (nonmelanoma skin cancers/melanoma) in vitiligo patients. Dermatol. Clin. 2017, 35, 129–134. [Google Scholar] [CrossRef]
- Schallreuter, K.U.; Tobin, D.J.; Panske, A. Decreased photodamage and low incidence of non-melanoma skin cancer in 136 sun-exposed caucasian patients with vitiligo. Dermatology 2002, 204, 194–201. [Google Scholar] [CrossRef]
- Teulings, H.E.; Overkamp, M.; Ceylan, E.; Nieuweboer-Krobotova, L.; Bos, J.D.; Nijsten, T.; Wolkerstorfer, A.W.; Luiten, R.M.; Van der Veen, J.P.W. Decreased risk of melanoma and nonmelanoma skin cancer in patients with vitiligo: A survey among 1307 patients and their partners. Br. J. Dermatol. 2013, 168, 162–171. [Google Scholar] [CrossRef]
- Jin, Y.; Birlea, S.A.; Fain, P.R.; Gowan, K.; Riccardi, S.L.; Holland, P.J.; Mailloux, C.M.; Sufit, A.J.D.; Hutton, S.M.; Amadi-Myers, A. Variant of TYR and autoimmunity susceptibility loci in generalized vitiligo. New Engl. J. Med. 2010, 362, 1686–1697. [Google Scholar] [CrossRef]
- Teulings, H.-E.; Limpens, J.; Jansen, S.N.; Zwinderman, A.H.; Reitsma, J.B.; Spuls, P.I.; Luiten, R.M. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: A systematic review and meta-analysis. J. Clin. Oncol. 2015, 33, 773–781. [Google Scholar] [CrossRef]
- James, W.D.; Berger, T.G.; Elston, D.M. Andrew’s Diseases of the Skin: Clinical Dermatol; Saunders Elsevier: Philadelphia, PA, USA, 2006; Volume 10. [Google Scholar]
- López, M. Treatment of skin disease. Comprehensive therapeutic strategies. Dermatol. Venez. 2006, 44, 76–1398. [Google Scholar]
- Garg, B.J.; Saraswat, A.; Bhatia, A.; Katare, O.P. Topical treatment in vitiligo and the potential uses of new drug delivery systems. Indian J. Dermatol. Venereol. Leprol. 2010, 76, 231. [Google Scholar]
- Saputra, M.A.R.; Purwoko, I.H.; Toruan, T.L. The Role of Topical Vitamin D in Vitiligo: A Narrative Literature Review. Biosci. Med. J. Biomed. Transl. Res. 2022, 6, 2516–2526. [Google Scholar]
- Jha, A.K.; Karki, S. Pigmentary disorders; vitiligo and melasma in context of south asian countries: A psychosocio-cosmetic challenge. Int. J. Dermatol. Clin. Res. 2015, 1, 24–30. [Google Scholar] [CrossRef]
- Akdeniz, N.; Yavuz, I.H.; Gunes Bilgili, S.; Ozaydın Yavuz, G.; Calka, O. Comparison of efficacy of narrow band UVB therapies with UVB alone, in combination with calcipotriol, and with betamethasone and calcipotriol in vitiligo. J. Dermatol. Treat. 2014, 25, 196–199. [Google Scholar] [CrossRef]
- Köse, O.; Arca, E.; Kurumlu, Z. Mometasone cream versus pimecrolimus cream for the treatment of childhood localized vitiligo. J. Dermatol. Treat. 2010, 21, 133–139. [Google Scholar] [CrossRef]
- Ho, N.; Pope, E.; Weinstein, M.; Greenberg, S.; Webster, C.; Krafchik, B. A double-blind, randomized, placebo-controlled trial of topical tacrolimus 0.1% vs. clobetasol propionate 0.05% in childhood vitiligo. Br. J. Dermatol. 2011, 165, 626–632. [Google Scholar] [CrossRef]
- Carr, W.W. Topical calcineurin inhibitors for atopic dermatitis: Review and treatment recommendations. Pediatr. Drugs 2013, 15, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.A.; Mohammed, G.F.; Gomaa, A.H.A.; Eyada, M.M.K. Carbon dioxide laser plus topical 5-fluorouracil: A new combination therapeutic modality for acral vitiligo. J. Cosmet. Laser Ther. 2015, 17, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Attwa, E.M.; Khashaba, S.A.; Ezzat, N.A. Evaluation of the additional effect of topical 5-fluorouracil to needling in the treatment of localized vitiligo. J. Cosmet. Dermatol. 2020, 19, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Pazyar, N.; Hatami, M.; Yaghoobi, R.; Parvar, S.Y.; Radmanesh, M.; Hadibarhaghtalab, M. The efficacy of adding topical 5-fluorouracil to micro-needling in the treatment of vitiligo: A randomized controlled trial. J. Cosmet. Dermatol. 2023, 22, 1513–1520. [Google Scholar] [CrossRef]
- Pasricha, J.S.; Khaitan, B.K. Oral mini-pulse therapy with betamethasone in vitiligo patients having extensive or fast-spreading disease. Int. J. Dermatol. 1993, 32, 753–757. [Google Scholar] [CrossRef]
- Lotti, T.; Agarwal, K.; Podder, I.; Satolli, F.; Kassir, M.; Schwartz, R.A.; Wollina, U.; Grabbe, S.; Navarini, A.A.; Mueller, S.M. Safety of the current drug treatments for vitiligo. Expert Opin. Drug Saf. 2020, 19, 499–511. [Google Scholar] [CrossRef]
- Chang, H.-C.; Hsu, Y.-P.; Huang, Y.-C. The effectiveness of topical calcineurin inhibitors compared with topical corticosteroids in the treatment of vitiligo: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2020, 82, 243–245. [Google Scholar] [CrossRef]
- Dillon, A.B.; Sideris, A.; Hadi, A.; Elbuluk, N. Advances in vitiligo: An update on medical and surgical treatments. J. Clin. Aesthetic Dermatol. 2017, 10, 15. [Google Scholar]
- Daniel, B.S.; Wittal, R. Vitiligo treatment update. Australas. J. Dermatol. 2015, 56, 85–92. [Google Scholar] [CrossRef]
- Singh, H.; Kumaran, M.S.; Bains, A.; Parsad, D. A randomized comparative study of oral corticosteroid minipulse and low-dose oral methotrexate in the treatment of unstable vitiligo. Dermatology 2015, 231, 286–290. [Google Scholar] [CrossRef]
- Qi, F.; Liu, F.; Gao, L. Janus kinase inhibitors in the treatment of vitiligo: A review. Front. Immunol. 2021, 12, 790125. [Google Scholar] [CrossRef]
- Birlea, S.; Birlea, M.; Cimponeriu, D.; Apostol, P.; Cosgarea, R.; Gavrila, L.; Tigan, S.; Costin, G.; Das, P. Autoimmune diseases and vitamin D receptor Apa-I polymorphism are associated with vitiligo in a small inbred Romanian community. Acta Derm. Venereol. 2006, 86, 209–214. [Google Scholar] [CrossRef]
- Imran, M.; Jin, X.; Ali, M.; Tapfumaneyi, P.; Lelasseur, P.; Carlo, L.; Jude, A.; Bourg, A.L.; Panchal, B.; Dick, A. The Pandemic and Your Skin—Direct and Indirect Impact of COVID-19. Cosmetics 2023, 10, 34. [Google Scholar] [CrossRef]
- Parsad, D.; Saini, R.; Verma, N. Combination of PUVAsol and topical calcipotriol in vitiligo. Dermatology 1998, 197, 167–170. [Google Scholar] [CrossRef]
- Parsad, D.; Kanwar, A. Topical vitamin D3 analogues in the treatment of vitiligo. Pigment Cell Melanoma Res. 2009, 22, 487–488. [Google Scholar] [CrossRef]
- Oikawa, A.; Nakayasu, M. Stimulation of melanogenesis in cultured melanoma cells by calciferols. FEBS Lett. 1974, 42, 32–35. [Google Scholar] [CrossRef]
- Tomita, Y.; Torinuki, W.; Tagami, H. Stimulation of human melanocytes by vitamin D 3 possibly mediates skin pigmentation after sun exposure. J. Investig. Dermatol. 1988, 90, 882–884. [Google Scholar] [CrossRef]
- Xu, Q.; Du, J.; He, P.; Zhang, J.; Zhu, T. Effects of 1alpha, 25-dihydroxyvitamin D (3) and UVB on cell proliferation and melanin synthesis of cultured human melanocyte. Beijing Da Xue Xue Bao Yi Xue Ban J. Peking Univ. Health Sci. 2004, 36, 483–486. [Google Scholar]
- Oh, S.H.; Kim, T.; Jee, H.; Do, J.E.; Lee, J.H. Combination treatment of non-segmental vitiligo with a 308-nm xenon chloride excimer laser and topical high-concentration tacalcitol: A prospective, single-blinded, paired, comparative study. J. Am. Acad. Derm. 2011, 65, 428–430. [Google Scholar] [CrossRef]
- Abdel-Malek, Z.A.; Ross, R.; Trinkle, L.; Swope, V.; Pike, J.W.; Nordlund, J.J. Hormonal effects of vitamin D3 on epidermal melanocytes. J. Cell. Physiol. 1988, 136, 273–280. [Google Scholar] [CrossRef]
- Ermis, O.; Alpsoy, E.; Cetin, L.; Yilmaz, E. Is the efficacy of psoralen plus ultraviolet A therapy for vitiligo enhanced by concurrent topical calcipotriol? A placebo-controlled double-blind study. Br. J. Dermatol. 2001, 145, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, M.; Kaur, I.; Kumar, B. Effect of topical calcipotriol, betamethasone dipropionate and their combination in the treatment of localized vitiligo. J. Eur. Acad. Dermatol. Venereol. 2006, 20, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.A.; Hassan, G.F.; Elgendy, H.Y.; Al-shenawy, H.A. Evaluation of the efficacy of transdermal drug delivery of calcipotriol plus betamethasone versus tacrolimus in the treatment of vitiligo. J. Cosmet. Dermatol. 2019, 18, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Bouceiro Mendes, R.; Alpalhão, M.; Filipe, P. UVB phototherapy in the treatment of vitiligo: State of the art and clinical perspectives. Photodermatol. Photoimmunol. Photomed. 2022, 38, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Yones, S.S.; Palmer, R.A.; Garibaldinos, T.M.; Hawk, J.L. Randomized double-blind trial of treatment of vitiligo: Efficacy of psoralen–UV-A therapy vs narrowband–UV-B therapy. Arch. Dermatol. 2007, 143, 578–584. [Google Scholar] [CrossRef]
- Lotti, T.; Berti, S.; Moretti, S. Vitiligo therapy. Expert Opin. Pharmacother. 2009, 10, 2779–2785. [Google Scholar] [CrossRef]
- Savin, J.A. The hidden face of dermatology. Clin. Exp. Dermatol. 1993, 18, 393–395. [Google Scholar] [CrossRef]
- Mulekar, S.V.; Isedeh, P. Surgical interventions for vitiligo: An evidence-based review. Br. J. Dermatol. 2013, 169, 57–66. [Google Scholar] [CrossRef]
- Anbar, T.S.; Hegazy, R.A.; Picardo, M.; Taieb, A. Beyond vitiligo guidelines: Combined stratified/personalized approaches for the vitiligo patient. Exp. Dermatol. 2014, 23, 219–223. [Google Scholar] [CrossRef]
- Tapfumaneyi, P.; Imran, M.; Mohammed, Y.H.; Roberts, M. Recent advances and future prospective of topical and transdermal delivery systems. Front. Drug Deliv. 2022, 25. [Google Scholar] [CrossRef]
- Van Geel, N.; Ongenae, K.; De Mil, M.; Vander Haeghen, Y.; Vervaet, C.; Naeyaert, J.M. Double-blind placebo-controlled study of autologous transplanted epidermal cell suspensions for repigmenting vitiligo. Arch. Dermatol. 2004, 140, 1203–1208. [Google Scholar] [CrossRef][Green Version]
- Sarveswari, K.N. Cosmetic camouflage in vitiligo. Indian J. Dermatol. 2010, 55, 211. [Google Scholar] [CrossRef]
- Tanioka, M.; Yamamoto, Y.; Kato, M.; Miyachi, Y. Camouflage for patients with vitiligo vulgaris improved their quality of life. J. Cosmet. Dermatol. 2010, 9, 72–75. [Google Scholar] [CrossRef]
- Kaliyadan, F.; Kumar, A. Camouflage for patients with vitiligo. Indian J. Dermatol. Venereol. Leprol. 2012, 78, 8. [Google Scholar] [CrossRef]
- Sugathan, P.; Nejeeba, R. A new comouflage for vitiligo. Indian J. Dermatol. Venereol. Leprol. 1991, 57, 45. [Google Scholar]
- Rajatanavin, N.; Suwanachote, S.; Kulkollakarn, S. Dihydroxyacetone: A safe camouflaging option in vitiligo. Int. J. Dermatol. 2008, 47, 402–406. [Google Scholar] [CrossRef]
- Yoon, J.; Sun, Y.-W.; Kim, T.-H. Complementary and alternative medicine for vitiligo. In Vitiligo: Manage Therapy; BoD–Books on Demand: Kyungnam, Republic of Korea, 2011; pp. 143–158. [Google Scholar]
- Holme, S.A.; Beattie, P.E.; Fleming, C.J. Cosmetic camouflage advice improves quality of life. Br. J. Dermatol. 2002, 147, 946–949. [Google Scholar] [CrossRef]
| Types of Vitiligo | Subtypes |
|---|---|
| Segmental | Bisegmental/Unisegemntal/Plurisegmental |
| Non-segmental | Generalised/Acrofacial/Universal/Mucosal/Mixed |
| Unclassified/Undetermined | Focal/Mucosal (one site) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
AL-smadi, K.; Imran, M.; Leite-Silva, V.R.; Mohammed, Y. Vitiligo: A Review of Aetiology, Pathogenesis, Treatment, and Psychosocial Impact. Cosmetics 2023, 10, 84. https://doi.org/10.3390/cosmetics10030084
AL-smadi K, Imran M, Leite-Silva VR, Mohammed Y. Vitiligo: A Review of Aetiology, Pathogenesis, Treatment, and Psychosocial Impact. Cosmetics. 2023; 10(3):84. https://doi.org/10.3390/cosmetics10030084
Chicago/Turabian StyleAL-smadi, Khadeejeh, Mohammad Imran, Vania R. Leite-Silva, and Yousuf Mohammed. 2023. "Vitiligo: A Review of Aetiology, Pathogenesis, Treatment, and Psychosocial Impact" Cosmetics 10, no. 3: 84. https://doi.org/10.3390/cosmetics10030084
APA StyleAL-smadi, K., Imran, M., Leite-Silva, V. R., & Mohammed, Y. (2023). Vitiligo: A Review of Aetiology, Pathogenesis, Treatment, and Psychosocial Impact. Cosmetics, 10(3), 84. https://doi.org/10.3390/cosmetics10030084







