Hyaluronic Acid in Facial Rehabilitation—A Narrative Review
Abstract
1. Introduction
2. Material and Methods
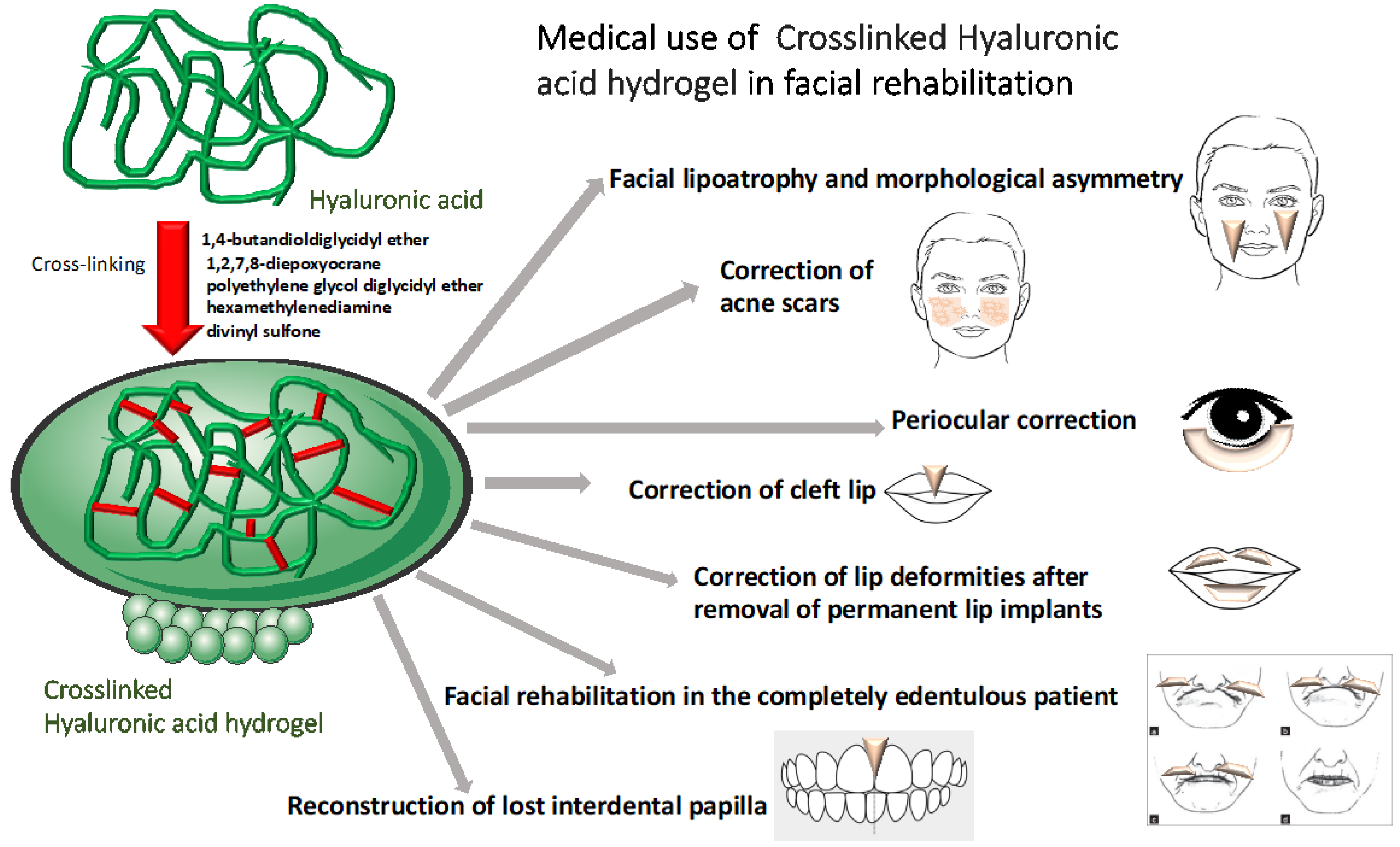
3. Medical Use in Facial Rehabilitation
3.1. Facial Lipoatrophy and Morphological Asymmetry
3.2. Correction of Depressed Post-Traumatic Scars
3.3. Correction of Acne Scars
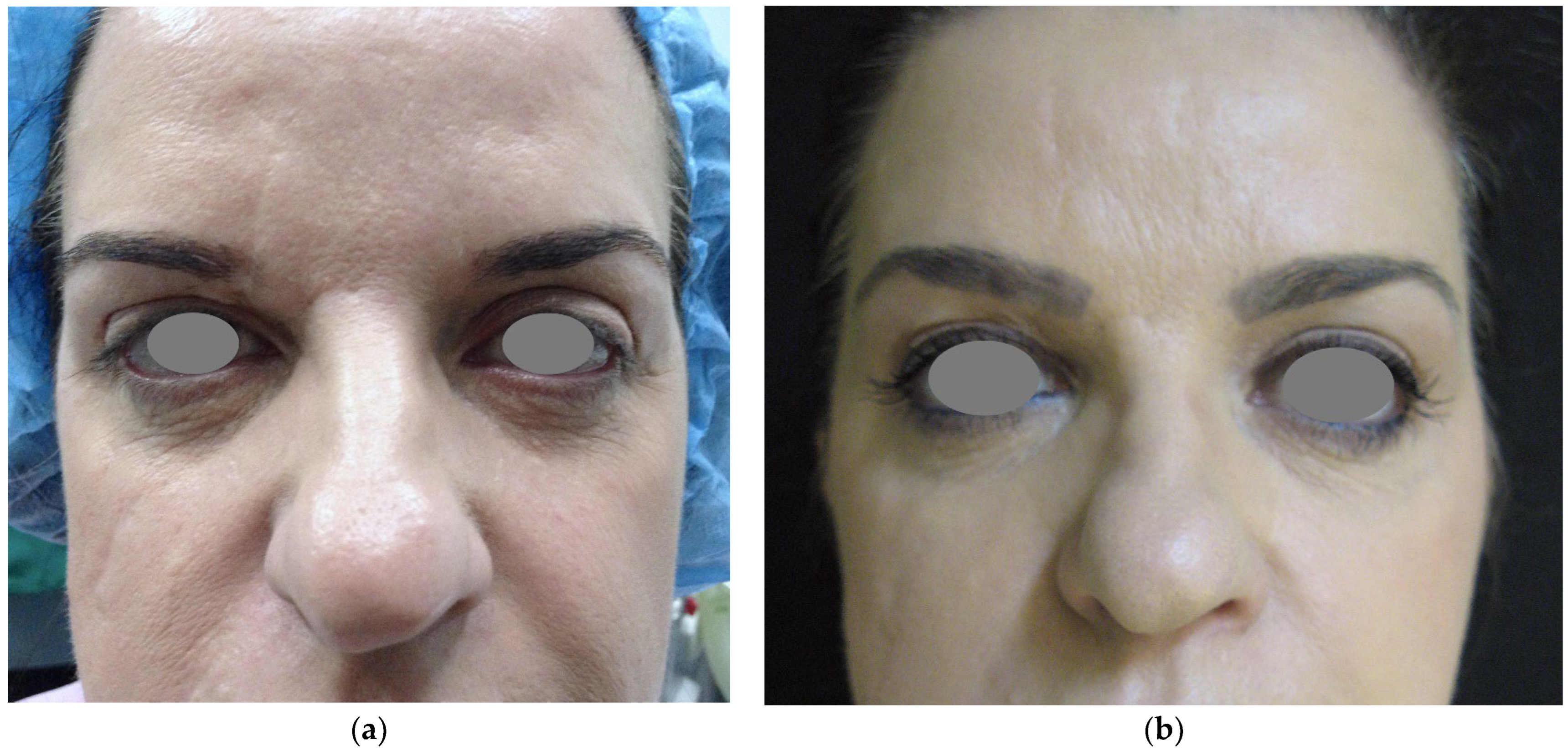
4. Periocular Correction
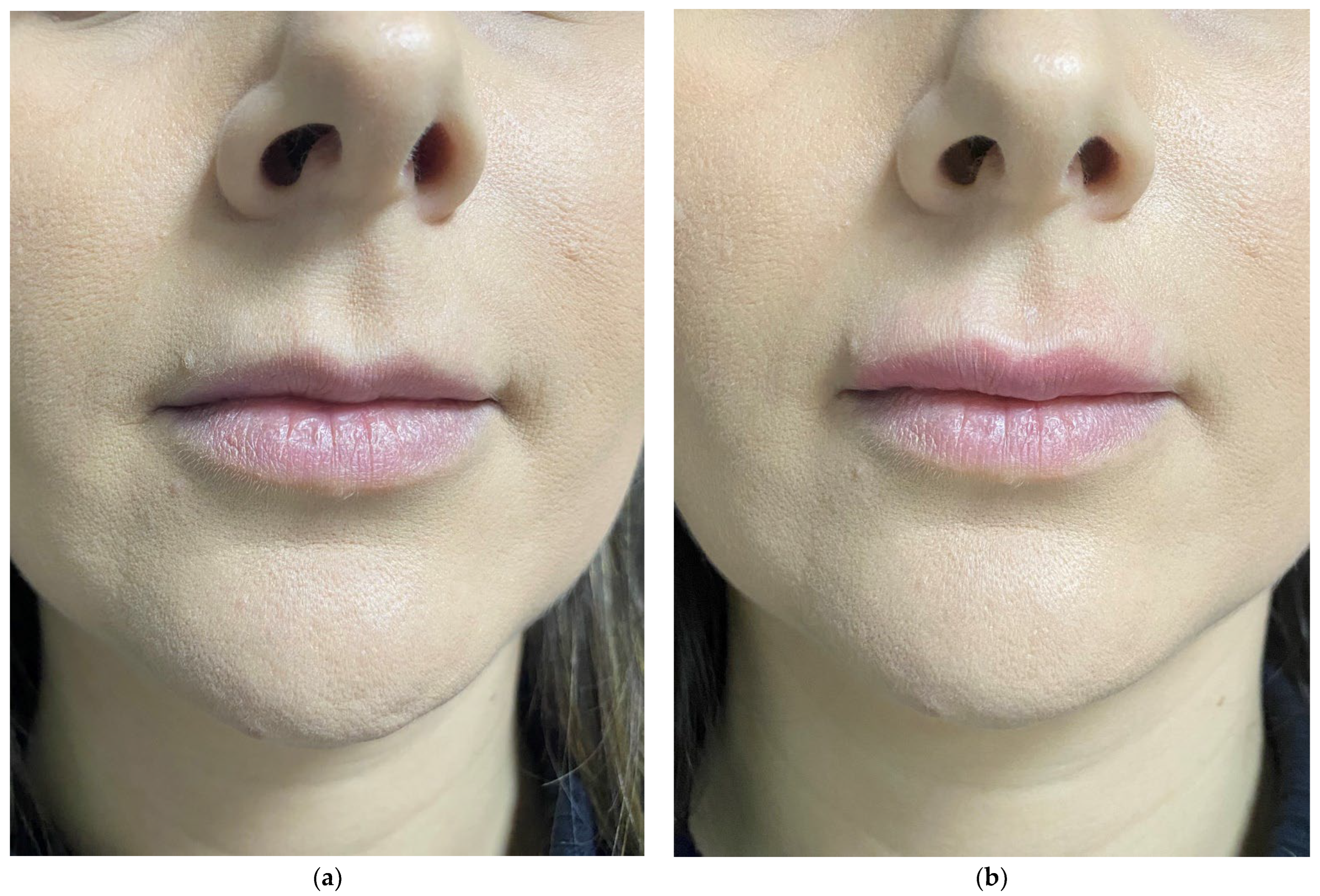
5. Correction of Cleft Lip
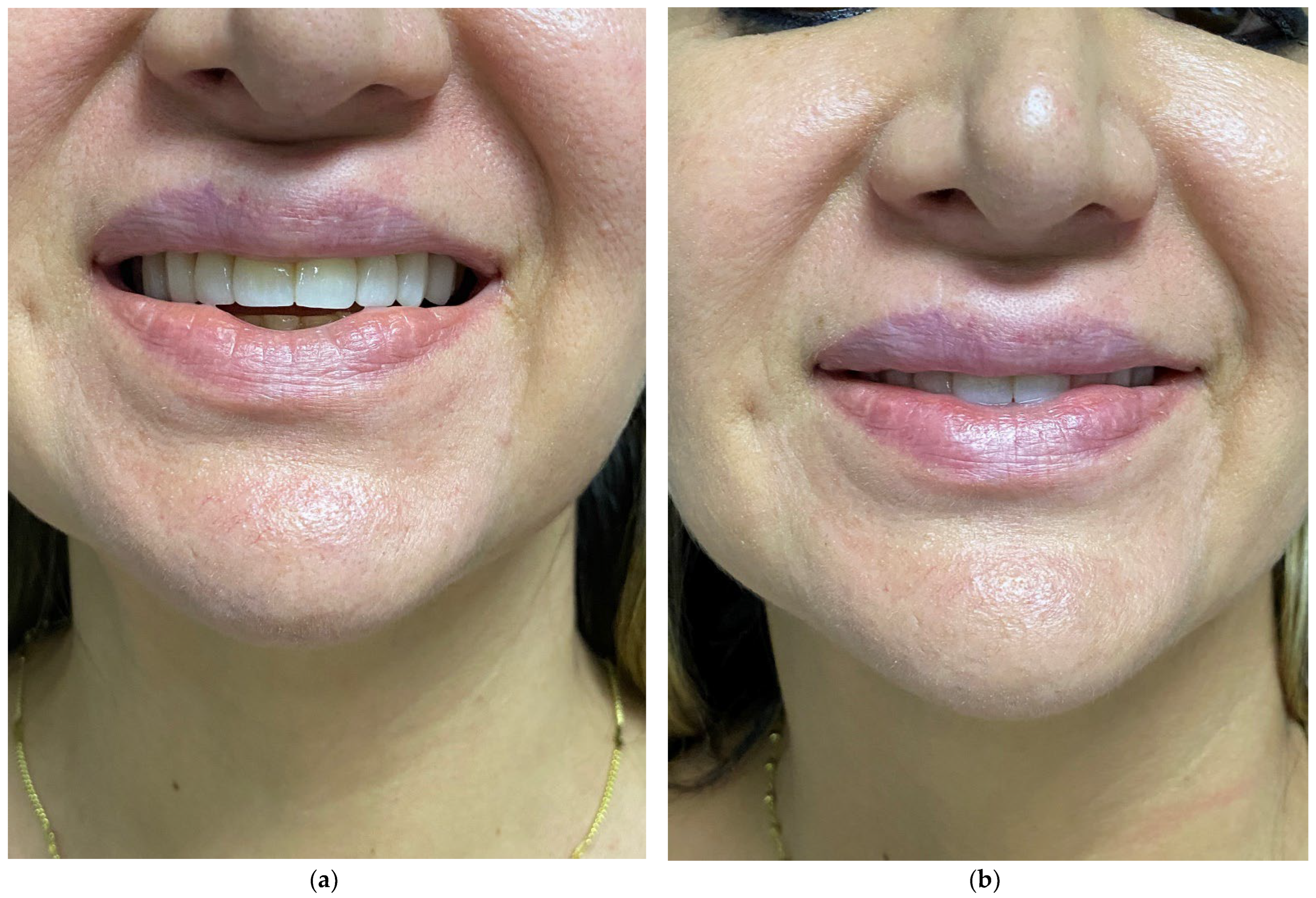
6. Correction of Lip Deformities after Removal of Permanent Lip Implants
7. Facial Rehabilitation in the Completely Edentulous Patient
8. Reconstruction of Lost Interdental Papilla
9. Reshaping Lower Jaw and Restoration after Delayed Diagnosis of Malar Arch Fracture
10. Ear Lobe Restoration
11. Nasal Obstruction
12. Facial Morphea
13. Limitations
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wollina, U.; Goldman, A. Hyaluronic acid dermal fillers: Safety and efficacy for the treatment of wrinkles, aging skin, body sculpturing and medical conditions. Clin. Med. Rev. Ther. 2011, 3, 107–121. [Google Scholar] [CrossRef]
- Zerbinati, N.; D’Este, E.; Farina, A.; Rauso, R.; Cherubino, M.; Calligaro, A. Morphological evidences following pegylated filler treatment in human skin. J. Biol. Regul. Homeost. Agents 2017, 31 (2 Suppl. 2), 79–85. [Google Scholar] [PubMed]
- Fundarò, S.P.; Salti, G.; Malgapo, D.M.H.; Innocenti, S. The rheology and physicochemical characteristics of hyaluronic acid fillers: Their clinical implications. Int. J. Mol. Sci. 2022, 23, 10518. [Google Scholar] [CrossRef] [PubMed]
- Kruk, D.; Rochowski, P.; Masiewicz, E.; Wilczynski, S.; Wojciechowski, M.; Broche, L.M.; Lurie, D.J. Mechanism of water dynamics in hyaluronic dermal fillers revealed by nuclear magnetic resonance relaxometry. Chemphyschem 2019, 20, 2816–2822. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, J.; Leemans, G.; Dhaene, K.; Forsyth, R.; Vanslembrouck, J.; Hatem, F.; Micheels, P. Spreading pattern and tissue response to hyaluronic acid gel injections in the subcutis: Ultrasound videos, ultrasound measurements, and histology. Aesthetic Surg. J. 2021, 41, 224–241. [Google Scholar] [CrossRef]
- Rosamilia, G.; Hamade, H.; Freytag, D.L.; Frank, K.; Green, J.B.; Devineni, A.; Gavril, D.L.; Hernandez, C.A.; Pavicic, T.; Cotofana, S. Soft tissue distribution pattern of facial soft tissue fillers with different viscoelastic properties. J. Cosmet. Dermatol. 2020, 19, 312–320. [Google Scholar] [CrossRef]
- Nadra, K.; André, M.; Marchaud, E.; Kestemont, P.; Braccini, F.; Cartier, H.; Kéophiphath, M.; Fanian, F. A hyaluronic acid-based filler reduces lipolysis in human mature adipocytes and maintains adherence and lipid accumulation of long-term differentiated human preadipocytes. J. Cosmet. Dermatol. 2021, 20, 1474–1482. [Google Scholar] [CrossRef]
- Wollina, U. Midfacial rejuvenation by hyaluronic acid fillers and subcutaneous adipose tissue—A new concept. Med. Hypotheses 2015, 84, 327–330. [Google Scholar] [CrossRef]
- Wollina, U.; Goldman, A. Facial vascular danger zones for filler injections. Dermatol. Ther. 2020, 33, e14285. [Google Scholar] [CrossRef]
- Walker, L.; Convery, C.; Davies, E.; Murray, G.; Croasdell, B. Consensus opinion for the management of soft tissue filler induced vision loss. J. Clin. Aesthetic Dermatol. 2021, 14, E84–E94. [Google Scholar]
- Wang, H.C.; Yu, N.; Wang, X.; Dong, R.; Long, X.; Feng, X.; Li, J.; Wu, W.T.L. Cerebral Embolism as a Result of Facial Filler Injections: A Literature Review. Aesthetic Surg. J. 2022, 42, NP162–NP175. [Google Scholar] [CrossRef]
- Soares, D.J. Bridging a century-old problem: The pathophysiology and molecular mechanisms of HA filler-induced vascular occlusion (FIVO)-implications for therapeutic interventions. Molecules 2022, 27, 5398. [Google Scholar] [CrossRef]
- Wang, H.C.; Yu, N.; Wang, X. False sense of safety: Blunt cannulas cause the majority of severe vascular complications in hyaluronic acid injection. Plast. Reconstr. Surg. 2021, 147, 694e–695e. [Google Scholar] [CrossRef]
- Goodman, G.J.; Magnusson, M.R.; Callan, P.; Roberts, S.; Hart, S.; Lin, F.; Rahman, E.; McDonald, C.B.; Liew, S.; Porter, C.; et al. Aspiration before tissue filler-an exercise in futility and unsafe practice. Aesthetic Surg. J. 2022, 42, 89–101. [Google Scholar] [CrossRef]
- Singh, A.; Deshmukh, P. Bell’s palsy: A review. Cureus 2022, 14, e30186. [Google Scholar] [CrossRef]
- Turki, A.; Abbas, K.S.; Makram, A.M.; Elfert, M.; Elmarabea, M.; El-Shahat, N.A.; Abdalshafy, H.; Sampong, A.; Chintalapati, S.; Huy, N.T. Epidemiology, clinical features, and treatment modalities of facial nerve palsy in COVID-19 patients: A systematic review. Acta Neurol. Belg. 2022, 122, 1419–1432. [Google Scholar] [CrossRef]
- Carey, D.; Liew, S.; Emery, S. Restorative interventions for HIV facial lipoatrophy. AIDS Rev. 2008, 10, 116–124. [Google Scholar]
- Guzman, N.; Vijayan, V. HIV-Associated Lipodystrophy; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Pavicic, T.; Ruzicka, T.; Korting, H.C.; Gauglitz, G. Monophasic, cohesive-polydensified-matrix crosslinking-technology-based hyaluronic acid filler for the treatment of facial lipoatrophy in HIV-infected patients. J. Drugs Dermatol. 2010, 9, 690–695. [Google Scholar]
- Wollina, U.; Goldman, A. Botulinum toxin A and/or soft tissue fillers for facial rehabilitation. Wien. Med. Wochenschr. 2017, 167, 92–95. [Google Scholar] [CrossRef]
- Kopera, D.; Ivezic-Schoenfeld, Z.; Federspiel, I.G.; Grablowitz, D.; Gehl, B.; Prinz, M. Treatment of facial lipoatrophy, morphological asymmetry, or debilitating scars with the hyaluronic acid dermal filler Princess® FILLER. Clin. Cosmet. Investig. Dermatol. 2018, 11, 621–628. [Google Scholar] [CrossRef]
- Grablowitz, D.; Ivezic-Schoenfeld, Z.; Federspiel, I.G.; Gehl, B.; Kopera, D.; Prinz, M. Long-term effectiveness of a hyaluronic acid soft tissue filler in patients with facial lipoatrophy, morphological asymmetry, or debilitating scars. J. Cosmet. Dermatol. 2020, 19, 2536–2541. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.N.; Goodman, G.J.; Rahman, E. Treatment of a traumatic atrophic depressed scar with hyaluronic acid fillers: A case report. Clin. Cosmet. Investig. Dermatol. 2017, 10, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Mashiko, T.; Mori, H.; Kato, H.; Doi, K.; Kuno, S.; Kinoshita, K.; Kunimatsu, A.; Ohtomo, K.; Yoshimura, K. Semipermanent volumization by an absorbable filler: Onlay injection technique to the bone. Plast. Reconstr. Surg. Glob. Open 2013, 1, e4–e14. [Google Scholar] [CrossRef] [PubMed]
- Richards, K.N.; Rashid, R.M. Twenty-four-month persistence of hyaluronic acid filler for an atrophic scar. J. Cosmet. Dermatol. 2011, 10, 311–312. [Google Scholar] [CrossRef] [PubMed]
- Kasper, D.A.; Cohen, J.L.; Saxena, A.; Morganroth, G.S. Fillers for postsurgical depressed scars after skin cancer reconstruction. J. Drugs Dermatol. 2008, 7, 486–487. [Google Scholar]
- Riccio, M.; Marchesini, A.; Senesi, L.; Skrami, E.; Gesuita, R.; De Francesco, F. Managing pathologic scars by injecting auto-cross-linked hyaluronic acid: A preliminary prospective clinical study. Aesthetic Plast. Surg. 2019, 43, 480–489. [Google Scholar] [CrossRef]
- Goodman, G.J.; Baron, J.A. Postacne scarring--a quantitative global scarring grading system. J. Cosmet. Dermatol. 2006, 5, 48–52. [Google Scholar] [CrossRef]
- Wollina, U.; Goldman, A. Fillers for the improvement in acne scars. Clin. Cosmet. Investig. Dermatol. 2015, 8, 493–499. [Google Scholar] [CrossRef]
- Hasson, A.; Romero, W.A. Treatment of facial atrophic scars with Esthélis, a hyaluronic acid filler with polydense cohesive matrix (CPM). J. Drugs Dermatol. 2010, 9, 1507–1509. [Google Scholar]
- Halachmi, S.; Ben Amitai, D.; Lapidoth, M. Treatment of acne scars with hyaluronic acid: An improved approach. J. Drugs Dermatol. 2013, 12, e121–e123. [Google Scholar]
- Goodman, G.J.; Van Den Broek, A. The modified tower vertical filler technique for the treatment of post-acne scarring. Australas. J. Dermatol. 2016, 57, 19–23. [Google Scholar] [CrossRef]
- Dierickx, C.; Larsson, M.K.; Blomster, S. Effectiveness and safety of acne scar treatment with nonanimal stabilized hyaluronic acid gel. Dermatol. Surg. 2018, 44 (Suppl. 1), S10–S18. [Google Scholar] [CrossRef]
- Artzi, O.; Cohen, S.; Koren, A.; Niv, R.; Friedman, O. Dual-plane hyaluronic acid treatment for atrophic acne scars. J. Cosmet. Dermatol. 2020, 19, 69–74. [Google Scholar] [CrossRef]
- Siperstein, R.; Nestor, E.; Meran, S.; Grunebaum, L. A split-face, blind, randomized placebo-controlled clinical trial investigating the efficacy and safety of hyaluronic acid filler for the correction of atrophic facial scars. J. Cosmet. Dermatol. 2022, 21, 3768–3778. [Google Scholar] [CrossRef]
- Akerman, L.; Mimouni, D.; Nosrati, A.; Hilewitz, D.; Solomon-Cohen, E. A combination of non-ablative laser and hyaluronic acid injectable for postacne scars: A novel treatment protocol. J. Clin. Aesthetic Dermatol. 2022, 15, 53–56. [Google Scholar]
- Taban, M.; Mancini, R.; Nakra, T.; Velez, F.G.; Ela-Dalman, N.; Tsirbas, A.; Douglas, R.S.; Goldberg, R.A. Nonsurgical management of congenital eyelid malpositions using hyaluronic acid gel. Ophthalmic Plast. Reconstr. Surg. 2009, 25, 259–263. [Google Scholar] [CrossRef]
- Martín-Oviedo, C.; García, I.; Lowy, A.; Scola, E.; Aristegui, M.; Scola, B. Hyaluronic acid gel weight: A nonsurgical option for the management of paralytic lagophthalmos. Laryngoscope 2013, 123, E91–E96. [Google Scholar] [CrossRef]
- Mancini, R.; Khadavi, N.M.; Goldberg, R.A. Nonsurgical management of upper eyelid margin asymmetry using hyaluronic acid gel filler. Ophthalmic Plast. Reconstr. Surg. 2011, 27, 1–3. [Google Scholar] [CrossRef]
- Malhotra, R. Deep orbital Sub-Q restylane (nonanimal stabilized hyaluronic acid) for orbital volume enhancement in sighted and anophthalmic orbits. Arch. Ophthalmol. 2007, 125, 1623–1629. [Google Scholar] [CrossRef]
- Tay, E.; Olver, J. Intraorbital hyaluronic acid for enophthalmos. Ophthalmology 2008, 115, 1101–1101.e2. [Google Scholar] [CrossRef]
- Zamani, M.; Thyagarajan, S.; Olver, J.M. Adjunctive use of hyaluronic acid gel (Restylane Sub-Q) in anophthalmic volume deficient sockets and phthisical eyes. Ophthalmic Plast. Reconstr. Surg. 2010, 26, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Wadde, K.; Chowdhar, A.; Venkatakrishnan, L.; Ghodake, M.; Sachdev, S.S.; Chhapane, A. Protocols in the management of cleft lip and palate: A systematic review: Running Title: Management of Cleft Lip and Palate. J. Stomatol. Oral Maxillofac. Surg. 2022, 142, 101338. [Google Scholar] [CrossRef]
- Schweiger, E.S.; Riddle, C.C.; Tonkovic-Capin, V.; Aires, D.J. Successful treatment with injected hyaluronic acid in a patient with lip asymmetry after surgical correction of cleft lip. Dermatol. Surg. 2008, 34, 717–719. [Google Scholar] [CrossRef]
- Manzoni, A.P.D.; Venier, N.A.B. Hyaluronic acid filler correction of scars and asymmetries secondary to cleft lip repair. Dermatol. Surg. 2022, 48, 1264–1266. [Google Scholar] [CrossRef] [PubMed]
- Alighieri, C.; Bettens, K.; Roche, N.; Bruneel, L.; Van Lierde, K. Lipofilling in patients with a cleft lip (and palate)—A pilot study assessing functional outcomes and patients’ satisfaction with appearance. Int. J. Pediatr. Otorhinolaryngol. 2020, 128, 109692. [Google Scholar] [CrossRef]
- Goldman, A.; Wollina, U. Polymethylmethacrylate-induced nodules of the lips: Clinical presentation and management by intralesional neodymium:YAG laser therapy. Dermatol. Ther. 2019, 32, e12755. [Google Scholar] [CrossRef]
- Rauso, R.; Federico, F.; Zerbinati, N.; De Cicco, D.; Nicoletti, G.F.; Tartaro, G. Hyaluronic acid injections to correct lips deformity following surgical removal of permanent implant. J. Craniofac. Surg. 2020, 31, e604–e606. [Google Scholar] [CrossRef]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Wollina, U. Perioral rejuvenation: Restoration of attractiveness in aging females by minimally invasive procedures. Clin. Interv. Aging 2013, 8, 1149–1155. [Google Scholar] [CrossRef]
- Steenen, S.A.; Bauland, C.G.; van der Lei, B.; Su, N.; van Engelen, M.D.G.; Anandbahadoer-Sitaldin, R.D.; Koeiman, W.; Jawidan, T.; Hamraz, Y.; Lange, J. Head-to-head comparison of 4 hyaluronic acid dermal fillers for lip augmentation: A multicenter randomized, quadruple-blind, controlled clinical trial. J. Am. Acad. Dermatol. 2022, 88, 932–935. [Google Scholar] [CrossRef]
- Aubry, S.; Collart-Dutilleul, P.Y.; Renaud, M.; Batifol, D.; Montal, S.; Pourreyron, L.; Carayon, D. Benefit of hyaluronic acid to treat facial aging in completely edentulous patients. J. Clin. Med. 2022, 11, 5874. [Google Scholar] [CrossRef]
- Villalobos, V.; Garrido, M.; Reyes, A.; Fernández, C.; Diaz, C.; Torres, V.A.; González, P.A.; Cáceres, M. Aging envisage imbalance of the periodontium: A keystone in oral disease and systemic health. Front. Immunol. 2022, 13, 1044334. [Google Scholar] [CrossRef]
- Tian, W.; Gao, P.; Zong, D.P.; Liu, J.J.; Zhang, M.Y.; Wang, C.C.; Wang, Z.X.; Wang, J.M.; Niu, Y.Y.; Xiang, P. The oral bioaccessibility and gingival cytotoxicity of metal(loid)s in wild vegetables from mining areas: Implication for human oral health. Front. Nutr. 2022, 9, 1042300. [Google Scholar] [CrossRef]
- Awartani, F.A.; Tatakis, D.N. Interdental papilla loss: Treatment by hyaluronic acid gel injection: A case series. Clin. Oral Investig. 2016, 20, 1775–1780. [Google Scholar] [CrossRef]
- Alhabashneh, R.; Alomari, S.; Khaleel, B.; Qinawi, H.; Alzaubi, M. Interdental papilla reconstruction using injectable hyaluronic acid: A 6 month prospective longitudinal clinical study. J. Esthet. Restor. Dent. 2021, 33, 531–537. [Google Scholar] [CrossRef]
- Lee, W.P.; Kim, H.J.; Yu, S.J.; Kim, B.O. Six month clinical evaluation of interdental papilla reconstruction with injectable hyaluronic acid gel using an image analysis system. J. Esthet. Restor. Dent. 2016, 28, 221–230. [Google Scholar] [CrossRef]
- Patil, S.C.; Dhalkari, C.D.; Indurkar, M.S. Hyaluronic acid: Ray of hope for esthetically challenging black triangles: A case series. Contemp. Clin. Dent. 2020, 11, 280–284. [Google Scholar] [CrossRef]
- Castro-Calderón, A.; Roccuzzo, A.; Ferrillo, M.; Gada, S.; González-Serrano, J.; Fonseca, M.; Molinero-Mourelle, P. Hyaluronic acid injection to restore the lost interproximal papilla: A systematic review. Acta Odontol. Scand. 2022, 80, 295–307. [Google Scholar] [CrossRef]
- Rauso, R.; Rugge, L.; Chirico, F.; Nicoletti, G.F.; Fragola, R.; Bove, P.; Zerbinati, N.; Giudice, G.L. Nonsurgical reshaping of the lower jaw with hyaluronic acid fillers: A retrospective case series. Dermatol. Pract. Concept. 2022, 12, e2022095. [Google Scholar] [CrossRef]
- Rauso, R.; Lo Giudice, G.; Zerbinati, N.; Tartaro, G. Hyaluronic acid injections as nonsurgical alternative in case of delayed diagnosis of malar arch fracture: Case report and literature review. Case Rep. Surg. 2019, 2019, 1360741. [Google Scholar] [CrossRef]
- Mowlavi, A.; Meldrum, D.G.; Kalkanis, J.; Wilhelmi, B.J.; Russell, R.C.; Zook, E.G. Surgical design and algorithm for correction of earlobe ptosis and pseudoptosis deformity. Plast. Reconstr. Surg. 2005, 115, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Zhang, Y.K.; Cao, Q.; Hou, Y.; Lv, W.; Fan, J.F. Clinical application of earlobe augmentation with hyaluronic acid filler in the Chinese population. Aesthetic Plast. Surg. 2017, 41, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Wollina, U. Filler-rejuvenation of the earlobes. J. Appl. Cosmetol. 2019, 37, 12–16. [Google Scholar]
- Arora, G.; Arora, S. Rejuvenating earlobe esthetics with dermal fillers. J. Cosmet. Dermatol. 2022, 21, 2788–2792. [Google Scholar] [CrossRef]
- Di Gregorio, C.; D’Arpa, S. Nonsurgical treatment of earlobe aging in Mowlavi stages I and II earlobe ptosis with Hyaluronic acid fillers. J. Cosmet. Dermatol. 2019, 18, 508–510. [Google Scholar] [CrossRef]
- Gümüş, N.; Acaban, M.B.; Demirbağ, H.O. Hyaluronic acid dermal filler promotes cartilage reshaping in rabbit ears. Aesthetic Plast. Surg. 2022, 46, 1932–1941. [Google Scholar] [CrossRef]
- Radulesco, T.; Winter, C.; Kestemont, P.; Braccini, F.; Castillo, L.; Michel, J. Liquid spreader grafts: Internal nasal valve opening with hyaluronic acid. Aesthetic Plast. Surg. 2022, 46, 2912–2916. [Google Scholar] [CrossRef]
- Gonzalez, C.D.; Pamatmat, J.J.; Goff, H.W. Safety and efficacy of dermal fillers in patients with connective tissue disease: A review. Dermatol. Surg. 2021, 47, 360–364. [Google Scholar] [CrossRef]
- Lane, T.K.; Cheung, J.; Schaffer, J.V. Parry-Romberg syndrome with coexistent morphea. Dermatol. Online J. 2008, 14, 21. [Google Scholar] [CrossRef]
- Choksi, A.N.; Orringer, J.S. Linear morphea-induced atrophy treated with hyaluronic acid filler injections. Dermatol. Surg. 2011, 37, 880–883. [Google Scholar] [CrossRef]
- Thareja, S.K.; Sadhwani, D.; Alan Fenske, N. En coup de sabre morphea treated with hyaluronic acid filler. Report of a case and review of the literature. Int. J. Dermatol. 2015, 54, 823–836. [Google Scholar] [CrossRef]
- Sivek, R.; Emer, J. Use of a blunt-tipped microcannula for soft tissue filler injection in the treatment of linear scleroderma (en coup de sabre). Dermatol. Surg. 2014, 40, 1439–1441. [Google Scholar] [CrossRef]
- Sharquie, K.E.; Al-Jaralla, F.; Sharquie, I.K. Intralesional injection of hyaluronic acid as a long lasting therapy of morphea sclerosis. Am. J. Dermatol. Venereol. 2019, 8, 45–48. [Google Scholar] [CrossRef]
- Owczarczyk-Saczonek, A.; Kasprowicz-Furmańczyk, M.; Kruszewska, A.; Krajewska-Włodarczyk, M.; Bechtold, A.; Klimek, P.; Placek, W. The correction of facial morphea lesions by hyaluronic acid: A case series and literature review. Dermatol. Ther. 2020, 10, 1423–1434. [Google Scholar] [CrossRef]
- Sharad, J. Hyaluronic acid filler injection for localized scleroderma—Case report and review of literature on filler injections for localized scleroderma. Clin. Cosmet. Investig. Dermatol. 2022, 15, 1627–1637. [Google Scholar] [CrossRef]
- Moore, R.M.; Mueller, M.A.; Hu, A.C.; Evans, G.R.D. Asymptomatic Stroke After Hyaluronic Acid Filler Injection: Case Report and Literature Review. Aesthetic Surg. J. 2021, 41, NP602–NP608. [Google Scholar] [CrossRef] [PubMed]
- Rzany, B.; Becker-Wegerich, P.; Bachmann, F.; Erdmann, R.; Wollina, U. Hyaluronidase in the correction of hyaluronic acid-based fillers: A review and a recommendation for use. J. Cosmet. Dermatol. 2009, 8, 317–323. [Google Scholar] [CrossRef]
- Zhang-Nunes, S.; Ryu, C.; Cahill, K.; Straka, D.; Nabavi, C.; Czyz, C.; Foster, J. Prospective in vivo evaluation of three different hyaluronic acid gels to varying doses of hyaluronidase with long-term follow-up. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 874–880. [Google Scholar] [CrossRef]
- Vartanian, A.J.; Frankel, A.S.; Rubin, M.G. Injected hyaluronidase reduces restylane-mediated cutaneous augmentation. Arch. Facial Plast. Surg. 2005, 7, 231–237. [Google Scholar] [CrossRef]
- Kato, K.; Inoue, E.; Tanaka, S.; Kawamoto, H. Increase in the incidence of acute inflammatory reactions to injectable fillers during COVID-19 era. J. Cosmet. Dermatol. 2022, 21, 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.E.; Amerson, E.; Rosenbach, M.; Lipoff, J.B.; Moustafa, D.; Tyagi, A.; Desai, S.R.; French, L.E.; Lim, H.W.; Thiers, B.H.; et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: A registry-based study of 414 cases. J. Am. Acad. Dermatol. 2021, 85, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Aryanian, Z.; Balighi, K.; Emadi, S.N.; Razavi, Z.; Hatami, P.; Afshar, Z.M. COVID-19 vaccines and filler reactions: Should it be considered as a concern? J. Cosmet. Dermatol. 2022, 21, 6564–6567. [Google Scholar] [CrossRef]
- Signorini, M.; Liew, S.; Sundaram, H.; De Boulle, K.L.; Goodman, G.J.; Monheit, G.; Wu, Y.; Trindade de Almeida, A.R.; Swift, A.; Vieira Braz, A.; et al. Global Aesthetics Consensus: Avoidance and management of complications from hyaluronic acid fillers-evidence- and opinion-based review and consensus recommendations. Plast. Reconstr. Surg. 2016, 137, 961e–971e. [Google Scholar] [CrossRef]
- Philipp-Dormston, W.G.; Bergfeld, D.; Sommer, B.M.; Sattler, G.; Cotofana, S.; Snozzi, P.; Wollina, U.; Hoffmann, K.P.J.; Salavastru, C.; Fritz, K. Consensus statement on prevention and management of adverse effects following rejuvenation procedures with hyaluronic acid-based fillers. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1088–1095. [Google Scholar] [CrossRef]
- Galadari, H.; Krompouzos, G.; Kassir, M.; Gupta, M.; Wollina, U.; Katsambas, A.; Lotti, T.; Jafferany, M.; Navarini, A.A.; Vasconcelos Berg, R.; et al. Complication of soft tissue fillers: Prevention and management review. J. Drugs Dermatol. 2020, 19, 829–832. [Google Scholar] [CrossRef]
- Heydenrych, I.; De Boulle, K.; Kapoor, K.M.; Bertossi, D. The 10-Point Plan 2021: Updated concepts for improved procedural safety during facial filler treatments. Clin. Cosmet. Investig. Dermatol. 2021, 14, 779–814. [Google Scholar] [CrossRef]
| HA concentration | The total concentration per mL depends on crosslinked (non-soluble) and extractable (soluble) HA in each HAF |
| Crosslinking | Depends on crosslinking agent, ratio of crosslinking agent to the HA concentration, and the ratio of crosslinked agent to the total amount of the crosslinking agent |
| Hydration | The capacity of HAF to swell when in contact to phosphate-buffered NaCl |
| Elastic module (G’) | Describes the shape memory of an HAF gel under pressure |
| Viscous modulus (G’’) | Describes the behavior of HAF under shear forces (friction) |
| Complex modulus (G*) | Describes the resistance of HAF to deformation under shear forces |
| Cohesivity | A measure of internal adhesion strength |
| Tangential delta (tan δ) | The ratio of viscous to elastic quality |
| Author(s) | n | Indication(s) | HAF (HA Concentration) | Outcome |
|---|---|---|---|---|
| Pavicic et al. (2010) [19] | 2 | HIV-associated facial lipoatrophy (FAL) | Belotero balance (22.5 mg/mL) | Improvement during 24 months |
| Wollina and Goldman (2017) [20] | 2 | 1× Bell’s palsy, 1× after orthognathic surgery | Belotero balance (22.5 mg/mL) | Improvement for 6–9 months for palsy in combination with repeated treatments of botulinum toxin type A |
| Kopera et al. (2018) [21] | 53 | FAL, morphological asymmetry (MA), debilitating scars (DS) | Princess (23 mg/mL) | Success rate 100% (FLA and MA), touch-up after 2 weeks, for DS 94% follow-up 24 weeks |
| Grablowitz et al. (2020) [22] | 60 | FLA, MA, and DS | Princess (23 mg/mL) | Success rate 98% at 4 weeks, 80% at week 24, and 61% at week 36 |
| Author (s) | n | Treatment | Outcome |
|---|---|---|---|
| Hasson and Romero (2010) [30] | 12 | Linear threading, serial puncture, or a combination of both | Improvement moderate (27%), good (57%), and excellent (17%) |
| Halachmi et al. (2013) [31] | 12 | Low-viscosity HAF after laser treatment and discrete depressed scars | Immediate visual improvement |
| Goodman and Van den Broek (2016) [32] | 5 | Modified tower-technique, two injections, 3-month follow-up | Static objective grading scale improvement from 3.2 to 2.6 |
| Dierickx et al. (2018) [33] | 12 | Low-viscosity HAF three times, 4 weeks apart, follow-up to 36 weeks | Scar severity, self-esteem, and self-confidence improved |
| Artzi et al. 2020 [34] | 12 | Dual-plane technique, two times, 4 weeks apart | 4 weeks apart improved by 2.5 |
| Siperstein et al. (2022) [35] | 15 | Split-face study, 1mL HAF vs. 1 mL saline, one touch-up, follow-up 90 days | Quantitative global scarring grading system: –6.6 (HAF), −1.7 (saline) |
| Akerman et al. (2022) [36] | 12 | Four sessions non-ablative laser, immediate after session one and three injection of high-viscose HAF | Mild to moderate improvement in all patients and overall scar appearance improved by 1.88 |
| Author (s) | n | Injections Schedule | Outcome (6 Months) |
|---|---|---|---|
| Awartani and Tatakis (2016) [55] | 9 | 0.2 mL day 0 and day 21 | Lost interdental papilla area reduced from 1.2 to 0.7 |
| Lee et al. (2016) [56] | 10 | Five injections in 3 weeks | 29 of 43 sites complete restoration and 14 of 43 sites improved |
| Patil et al. (2020) [57] | 5 | <0.2 mL repeated 2–3 times | BTA decrease 0.25 mm2, BTH decrease 0.85 mm2, and BTW decrease 0.34 mm2 |
| Alhabashneh et al. (2021) [58] | 12 | 0.2 mL day 0 and day 21 | BTH reduction 29% |
| Author (s) | n | Treatment | Duration of Effects |
|---|---|---|---|
| Qian et al. (2017) [63] | 19 | One injection, HAF 0.3–0.5 mL | 6–9 Months |
| Wollina (2019) [64] | 26 | One injection, HAF ≤ 0.5 mL | 9–12 months |
| Di Gregorio and D’Arpa (2019) [66] | 16 | One injection, five patients with a touch-up after 5–6 months | 14 months |
| Author (s) | n | Treatment | Outcome (Duration of Volumizing Effect) |
|---|---|---|---|
| Lane et al. (2008) [70] | 1 | HAF | 2 months |
| Choksi and Orringer (2011) [71] | 1 | Restylane Perlane 1 mL (20 mg HAF/mL), repeated after 5 months | 5 months |
| Mashiko et al. (2013) [24] | 1 | Restylane SubQ 1 mL (20 mg/mL) followed by Macrolane (20 mg/mL) | 12 months |
| Thareja et al. (2013) [72] | 1 | HAF, repeated after 6 months | 6 months |
| Sivek and Emer (2014) [73] | 1 | Juvederm Ultra Plus 0.8 mL (24 mg/mL) | >9 months |
| Sharquie et al. (2019) [74] | 16 | HAF, 1-2 times, 4 weeks apart | 2–24 months (stable disease, n = 6) and ≤2 months (unstable disease, n = 10) |
| Owczarczyk-Saczonek et al. (2020) [75] | 2 | HAF 0.5 mL once or twice, one week apart | Up to 1.5 years |
| Sharad (2022) [76] | 1 | Restylane Lyft 1.0 mL (20 mg/mL) plus Restylane 0.5 mL (20 mg/mL) 4 weeks later | 12 months |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wollina, U.; Kocic, H.; Goldman, A. Hyaluronic Acid in Facial Rehabilitation—A Narrative Review. Cosmetics 2023, 10, 61. https://doi.org/10.3390/cosmetics10020061
Wollina U, Kocic H, Goldman A. Hyaluronic Acid in Facial Rehabilitation—A Narrative Review. Cosmetics. 2023; 10(2):61. https://doi.org/10.3390/cosmetics10020061
Chicago/Turabian StyleWollina, Uwe, Hristina Kocic, and Alberto Goldman. 2023. "Hyaluronic Acid in Facial Rehabilitation—A Narrative Review" Cosmetics 10, no. 2: 61. https://doi.org/10.3390/cosmetics10020061
APA StyleWollina, U., Kocic, H., & Goldman, A. (2023). Hyaluronic Acid in Facial Rehabilitation—A Narrative Review. Cosmetics, 10(2), 61. https://doi.org/10.3390/cosmetics10020061






