Bioactive Compounds in Anti-Diabetic Plants: From Herbal Medicine to Modern Drug Discovery
Abstract
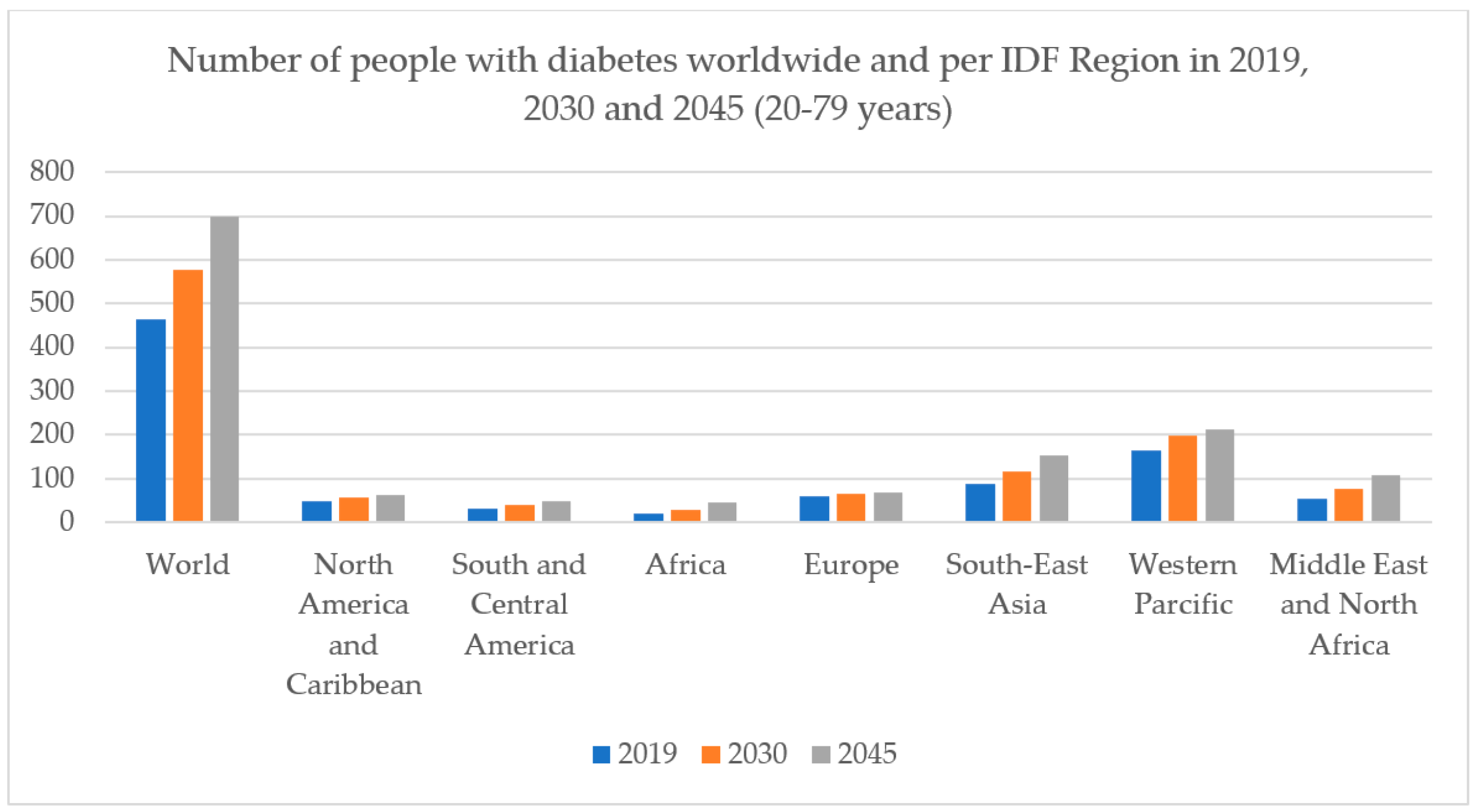
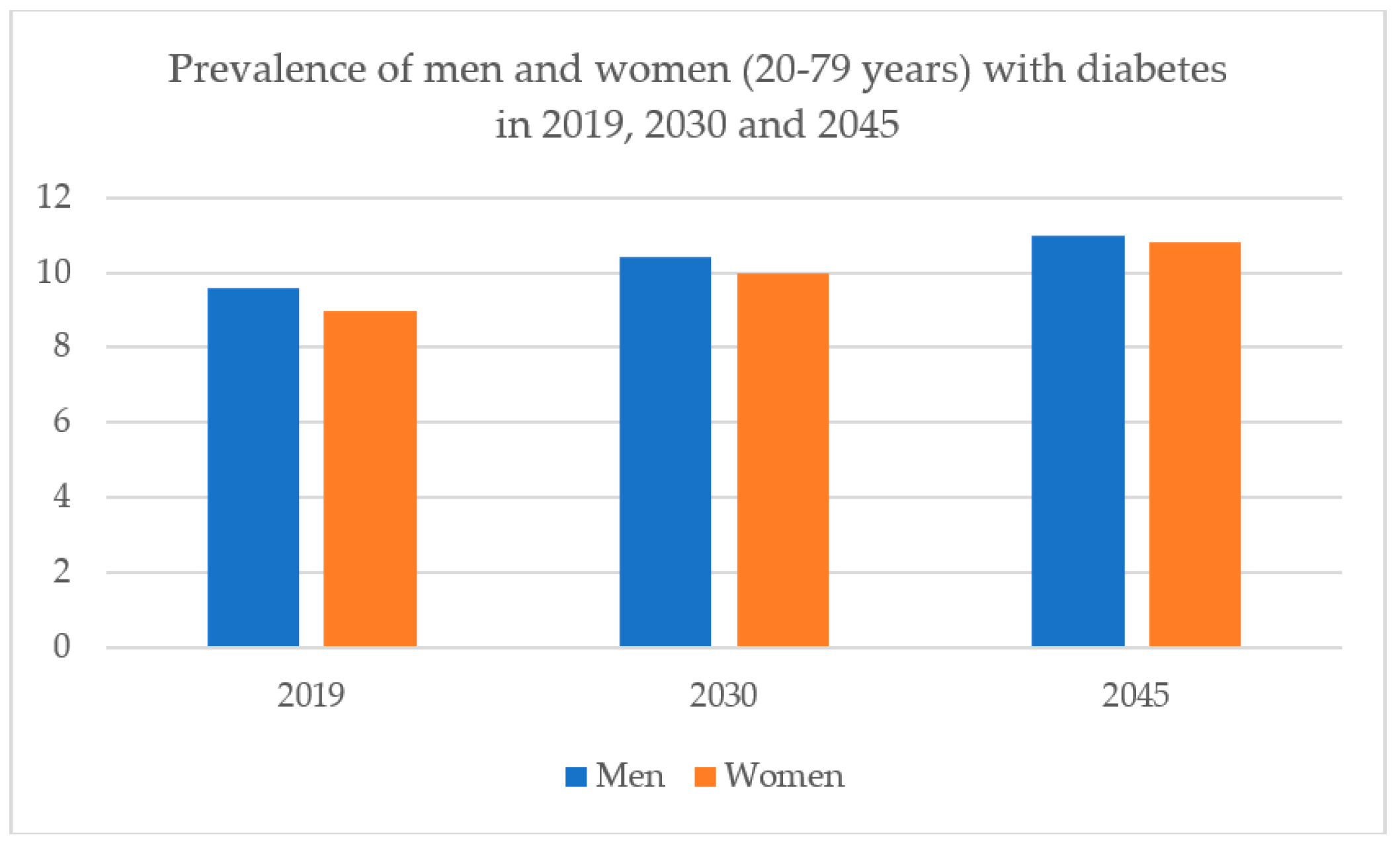
1. Introduction
2. Management of Diabetes Mellitus
3. Bioactive Compounds from Plants Having Type 2 Antidiabetic Activity
3.1. Bioactive Compounds Act as Insulin
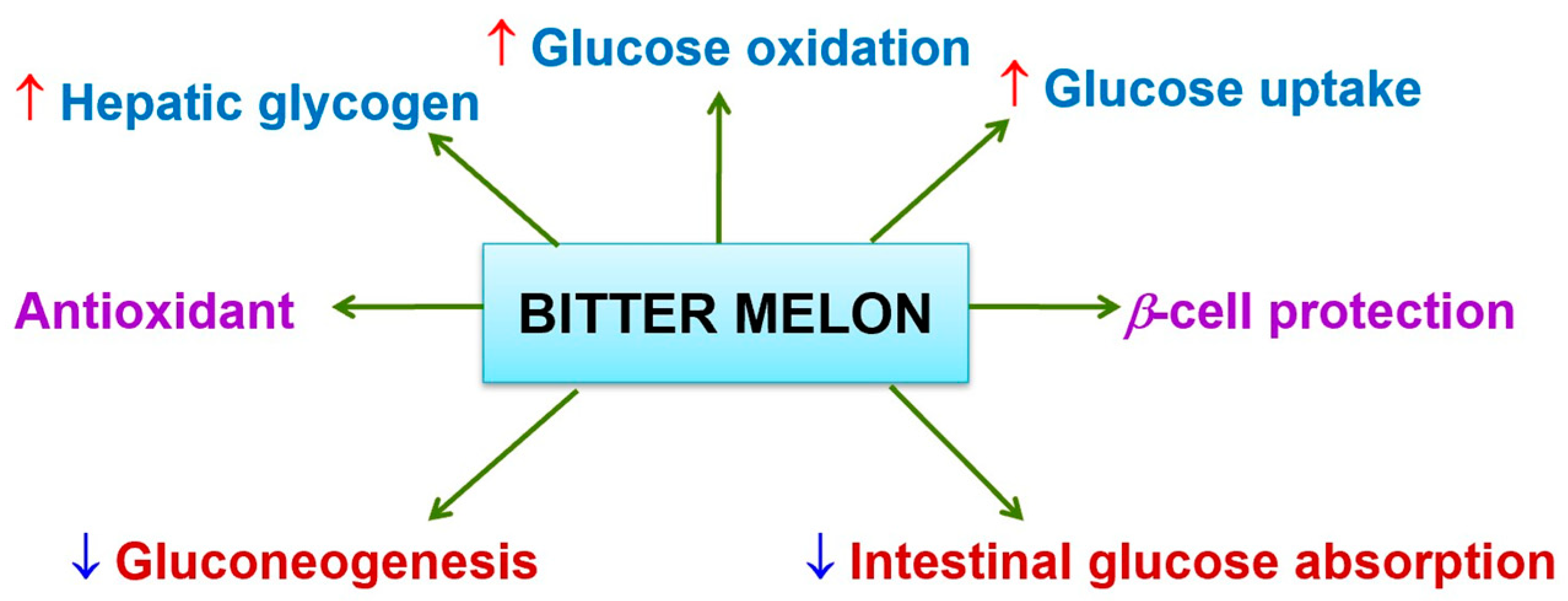
3.1.1. Momordica charantia (Bitter Melon)
Phytochemistry
Antidiabetic Activity
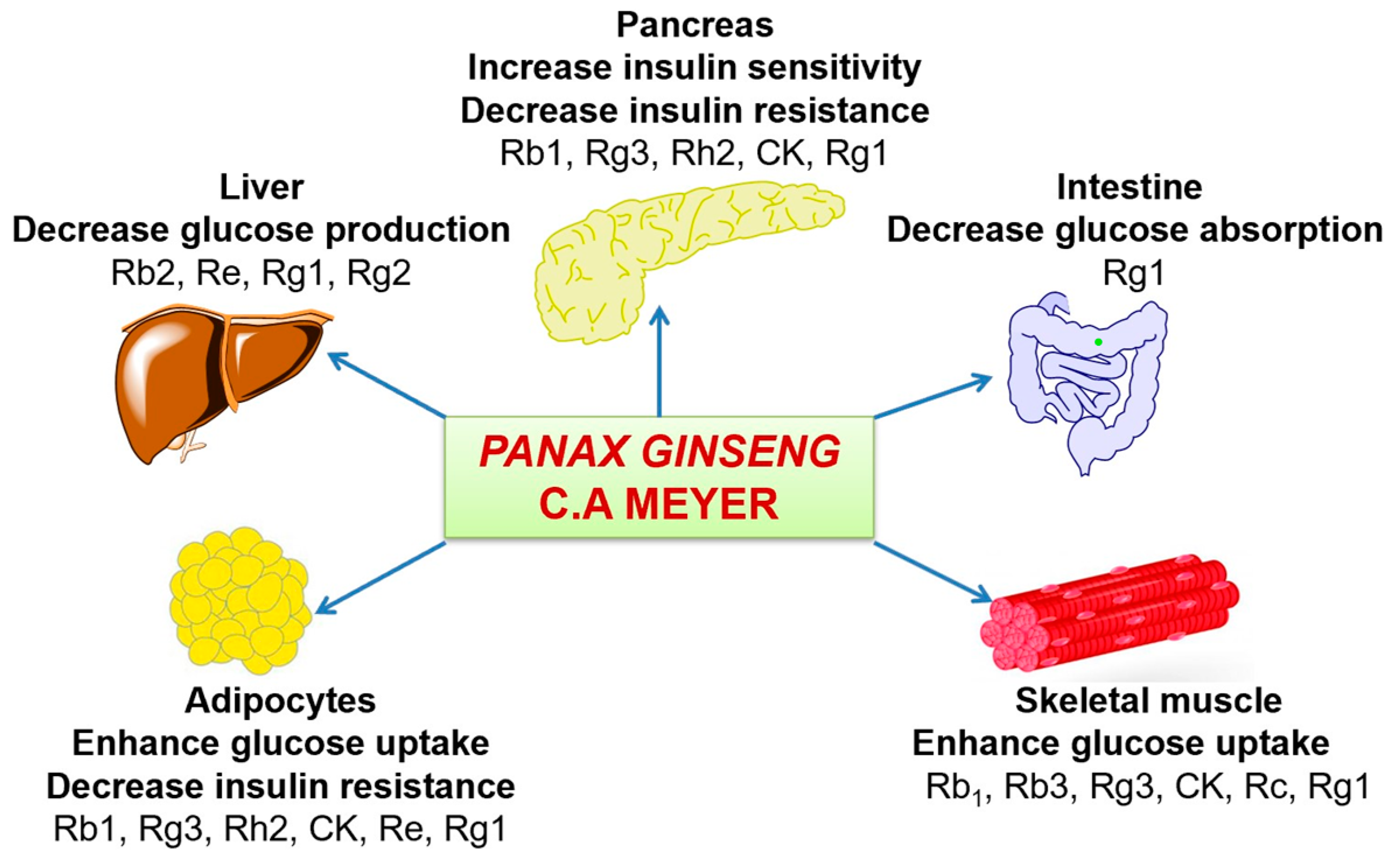
3.1.2. Panax ginseng C.A Meyer
Phytochemistry
Antidiabetic Activity
3.2. Bioactive Compounds Increase Insulin Secretion from Beta-Cells of Pancreas
3.2.1. Allium cepa (Allium)
Phytochemistry
Antidiabetic Activity
3.2.2. Allium sativum (Allium)
Phytochemistry
Antidiabetic Activity
3.2.3. Aloe vera L. Burm. (Asphodelaceae)
Phytochemistry
Antidiabetic Activity
3.3. Bioactive Compounds Regenerate of Beta-Cells of the Islets of the Pancreas
3.3.1. Pterocarpus marsupium (Fabaceae)
Phytochemistry
Antidiabetic Activity
3.3.2. Tinospora cordifolia (Menispermaceae)
Phytochemistry
Antidiabetic Activity
3.3.3. Tinospora crispa (Menispermaceae)
Phytochemistry
Antidiabetic Activity
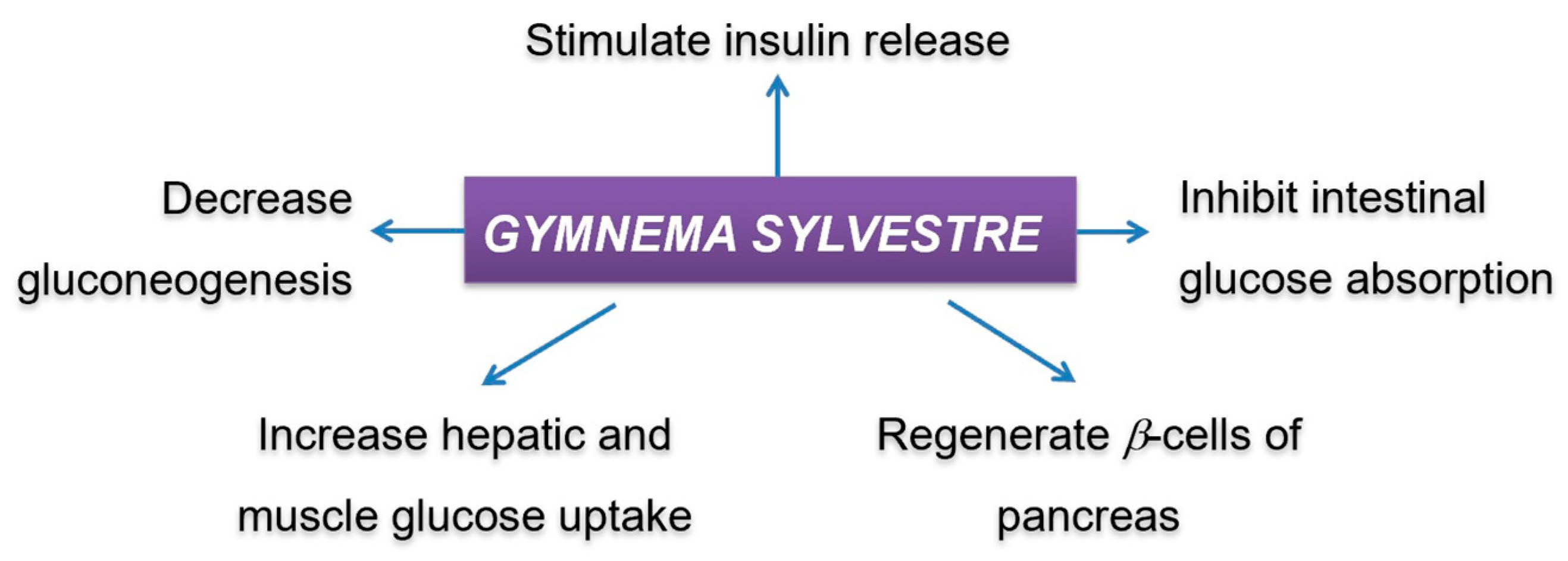
3.3.4. Gymnema sylvestre (Apocynaceae)
Phytochemistry
Antidiabetic Activity
3.4. Bioactive Compounds Reduce the Absorption of Glucose from Gastrointestinal Tract
3.4.1. Cyamposis tertragonoloba (Fabaceae)
Phytochemistry
Antidiabetic Activity
3.4.2. Ocimum sanctum L. (Lamiaceae)
Phytochemistry
Antidiabetic Activity
3.5. Bioactive Compounds Have Oxygen Radical Scavenging Activity
3.6. Bioactive Compounds Inhibit Alpha-Glucosidase and Alpha-Amylase Activities
3.6.1. Costus pictus (Zingiberacea)
Phytochemistry
Antidiabetic Activity
3.6.2. Phaseolus vulgaris (Leguminosae)
Phytochemistry
Antidiabetic Activity
3.6.3. Euphorbia hirta Linn. (Euphorbiaceae)
Phytochemistry
Anti-Diabetic Activity
3.7. Bioactive Compounds Increase Glucose Utilization
3.7.1. Zingiber officinale Rosc (Ginger)
Phytochemistry
Antidiabetic Activity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications; Department of Noncommunicable Disease Surveillance: Geneva, Switzerland, 1999. [Google Scholar]
- American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of Medical Care in Diabetes–2019. Diabetes Care 2019, 42 (Suppl. 1), S13–S28. [Google Scholar] [CrossRef]
- Heise, T.; Nosek, L.; Rønn, B.B.; Endahl, L.; Heinemann, L.; Kapitza, C.; Draeger, E. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes 2004, 53, 1614–1620. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, H.D.; Catalano, P.; Zhang, C.; Desoye, G.; Mathiesen, E.R.; Damm, P. Gestational diabetes mellitus. Nat. Rev. Dis. Primers 2019, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Faselis, C.; Katsimardou, A.; Imprialos, K.; Deligkaris, P.; Kallistratios, M.; Dimitriadis, K. Microvascular Complications of Type 2 Diabetes Mellitus. Curr. Vasc. Pharmacol. 2020, 18, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Mayfield, J. Diagnosis and classification of diabetes mellitus: New criteria. Am. Fam. Physician. 1998, 58, 1355–1362, 1369–1370. [Google Scholar] [PubMed]
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019. [Google Scholar]
- Medagama, A.B.; Bandara, R. The use of complementary and alternative medicines (CAMs) in the treatment of diabetes mellitus: Is continued use safe and effective? Nutr. J. 2014, 13, 102. [Google Scholar] [CrossRef]
- Alqathama, A.; Alluhiabi, G.; Baghdadi, H.; Aljahani, L.; Khan, O.; Jabal, S.; Makkawi, S.; Alhomoud, F. Herbal medicine from the perspective of type II diabetic patients and physicians: What is the relationship? BMC Complement. Med. Ther. 2020, 20, 65. [Google Scholar] [CrossRef]
- Kesavadev, J.; Saboo, B.; Sadikot, S.; Das, A.K.; Joshi, S.; Chawla, R.; Thacker, H.; Shankar, A.; Ramachandran, L.; Kalra, S. Unproven therapies for diabetes and their implications. Adv. Ther. 2017, 34, 60–77. [Google Scholar] [CrossRef]
- Knight, K. A systematic review of diabetes disease management programs. Am. J. Manag. Care 2005, 11, 242–250. [Google Scholar]
- Colberg, S.R.; Sigal, R.J.; Yardley, J.E.; Riddell, M.C.; Dunstan, D.W.; Dempsey, P.C.; Horton, E.S.; Castorino, K.; Tate, D.F. Physical activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016, 39, 2065–2079. [Google Scholar] [CrossRef]
- Khan, M.U. Lifestyle Modification in the Prevention of Type II Diabetes Mellitus. Oman Med. J. 2012, 27, 170–171. [Google Scholar] [CrossRef] [PubMed]
- Bhoyar, P.K.; Tripathi, A.K.; Baheti, J.R.; Biyani, D. Herbal Antidiabetics: A Review. Int. J. Res. Pharm. Sci. 2011, 2, 30–37. [Google Scholar]
- Wang, Z.; Wang, J.; Chan, P. Treating Type 2 Diabetes Mellitus with Traditional Chinese and Indian Medicinal Herbs. Evid. Based Complement. Altern. Med. 2013, 2013, 343594. [Google Scholar] [CrossRef] [PubMed]
- Thulé, P.M. Mechanisms of current therapies for diabetes mellitus type 2. Adv. Physiol. Educ. 2012, 36, 275–283. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A. Pharmacologic therapy for type 2 diabetes mellitus. Ann. Intern. Med. 1999, 131, 281–303. [Google Scholar] [CrossRef]
- Goldberg, R.B.; Kendall, D.M.; Deeg, M.A.; Buse, J.B.; Zagar, A.J.; Pinaire, J.A.; Tan, M.H.; Khan, M.A.; Perez, A.T.; Jacober, S.J.; et al. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care 2005, 28, 1547–1554. [Google Scholar] [CrossRef]
- Home, P.D.; Phil, D.; Pocock, S.J.; Beck-Nielsen, H.; Gomis, R.; Hanefeld, M.; Jones, N.P.; Komajdam, M.; McMurray, J.J.V. Rosiglitazone evaluated for cardiovascular outcomes-an interim analysis. N. Engl. J. Med. 2007, 357, 28–38. [Google Scholar] [CrossRef]
- Singh, S.; Loke, Y.K.; Furberg, C.D. Thiazolidinediones and heart failure: A teleo-analysis. Diabetes Care 2007, 30, 2148–2153. [Google Scholar] [CrossRef]
- Siconolfi-Baez, L.; Banerji, M.A.; Lebovitz, H.E. Characterization and significance of sulfonylurea receptors. Diabetes Care 1990, 13, 2–8. [Google Scholar] [CrossRef]
- Simonson, D.C.; Ferrannini, E.; Bevilacqua, S.; Smith, D.; Barrett, E.; Carlson, R.; DeFronzo, R. Mechanism of improvement in glucose metabolism after chronic glyburide therapy. Diabetes 1984, 33, 838–845. [Google Scholar] [CrossRef]
- Lebovitz, H.E. Thiazolidinediones: The Forgotten Diabetes Medications. Curr. Diabetes Rep. 2019, 19, 151. [Google Scholar] [CrossRef] [PubMed]
- Perfetti, R.; Ahmad, A. Novel sulfonylurea and non-sulfonylurea drugs to promote the secretion of insulin. Trends Endocrinol. Metab. 2000, 11, 218–223. [Google Scholar] [CrossRef]
- Hanefeld, M.; Bouter, K.P.; Dickinson, S.; Guitard, C. Rapid and short-acting mealtime insulin secretion with nateglinide controls both prandial and mean glycemia. Diabetes Care 2000, 23, 202–207. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jovanovic, L.; Dailey, G., III; Huang, W.C.; Strange, P.; Goldstein, B.J. Repaglinide in type 2 diabetes: A 24-week, fixed-dose efficacy and safety study. J. Clin. Pharmacol. 2000, 40, 49–57. [Google Scholar] [CrossRef]
- Yakubu, O.E.; Imo, C.; Shaibu, C.; Akighir, J.; Ameh, D.S. Effects of Wthanolic Leaf and Sterm-bark Extracts of Adansonia digitate in Alloxan-induced Diabetic Wistar Rats. J. Pharmacol. Toxicol. 2020, 15, 1–7. [Google Scholar] [CrossRef][Green Version]
- Chan, C.H.; Ngoh, G.C.; Yusoff, R. A brief review on anti-diabetic plants: Global distribution, active ingredients, extraction techniques and acting mechanisms. Pharmacogn. Rev. 2012, 6, 22–28. [Google Scholar] [CrossRef]
- Harris, M.I. Health care and health status and outcomes for patients with type 2 diabetes. Diabetes Care 2000, 23, 754–758. [Google Scholar] [CrossRef]
- Preethi, P.J. Herbal medicine for diabetes mellitus: A Review. Int. J. Phytopharm. 2013, 3, 1–22. [Google Scholar] [CrossRef]
- Evans, M. A Guide to Herbal Remedies; Orient Paperbacks: Delhi, India, 1994; ISBN-10: 8122201628. [Google Scholar]
- Patel, D.K.; Prasad, S.K.; Kumar, R.; Hemalatha, S. An overview on antidiabetic medicinal plants having insulin mimetic property. Asian Pac. J. Trop. Biomed. 2012, 2, 320–330. [Google Scholar] [CrossRef]
- Malviya, N. Antidiabetic Potential Of Medicinal Plants. Acta Pol. Pharm. 2010, 67, 113–118. [Google Scholar]
- Grover, J.K.; Yadav, S.; Vats, V. Medicinal plants of India with anti-diabetic potential. J. Ethnopharmacol. 2002, 81, 81–100. [Google Scholar] [CrossRef]
- Aggarwal, N.; Aggarwal, S. A Review of Recent Investigations on Medicinal Herbs Possessing AntiDiabetic Properties. J. Nutr. Disord. Ther. 2011. [Google Scholar] [CrossRef]
- Kayarohanam, S.; Kavimani, S. Current Trends of Plants Having Antidiabetic Activity: A Review. J. Bioanal. Biomed. 2015, 7, 55–65. [Google Scholar] [CrossRef]
- Sidhu, M.C.; Sharma, T. Medicinal Plants From Twelve Families Having Antidiabetic Activity: A Review. Am. J. PharmTech Res. 2013, 3, 36–52. [Google Scholar]
- Li, W.L.; Zheng, H.C.; Bukuru, J.; De Kimpe, N. Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J. Ethnopharmacol. 2004, 92, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Rathi, S.S.; Grover, J.K.; Vats, V. The effect of Momordica charantia and Mucuna pruriens in experimental diabetes and their effect on key metabolic enzymes involved in carbohydrate metabolism. Phytother. Res. 2002, 16, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Saeed, F.; Afzaal, M.; Niaz, B.; Arshad, M.U.; Tufail, T.; Hussain, M.B.; Javed, A. Bitter melon (Momordica charantia): A natural healthy vegetable. Int. J. Food Prop. 2018, 21, 1270–1290. [Google Scholar] [CrossRef]
- Dandawate, P.R.; Subramaniam, D.; Padhye, S.B.; Anant, S. Bitter Melon: A Panacea for Inflammation and Cancer. Chin. J. Nat. Med. 2016, 14, 81–100. [Google Scholar] [CrossRef]
- Ibrahim, T.A.; El-Hefnawy, H.M.; El-Hela, A.A. Antioxidant Potential and Phenolic Acid Content of Certain Cucurbitaceous Plants Cultivated in Egypt. Nat. Prod. Res. 2010, 24, 1537–1545. [Google Scholar] [CrossRef]
- Tan, S.P.; Stathopoulos, C.; Parks, S.; Roach, P. An Optimised Aqueous Extract of Phenolic Compounds from Bitter Melon with High Antioxidant Capacity. Antioxidants 2014, 3, 814–829. [Google Scholar] [CrossRef]
- Popovich, D.G.; Li, L.; Zhang, W. Bitter Melon (Momordica Charantia) Triterpenoid Extract Reduces Preadipocyte Viability, Lipid Accumulation and Adiponectin Expression in 3T3-L1 Cells. Food Chem. Toxicol. 2010, 48, 1619–1626. [Google Scholar] [CrossRef] [PubMed]
- Chuang, C.Y.; Hsu, C.; Chao, C.Y.; Wein, Y.S.; Kuo, Y.H.; Huang, C.J. Fractionation and identification of 9c, 11t, 13t-conjugated linolenic acid as an activator of PPARalpha in bitter gourd (Momordica charantia L.). J Biomed Sci. 2006, 13, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Zamhuri, K.F.; Yaacob, A.; Siong, C.H.; Selvarajah, M.; Nazrul, I.A.; Hakim, M. In Vitro Antidiabetic Activities and Chemical Analysis of Polypeptide-K and Oil Isolated from Seeds of Momordica Charantia (Bitter Gourd). Molecules 2012, 17, 9631–9640. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.C.; Ma, J.; Kavalier, A.; He, K.; Brillantes, A.M.; Kennelly, E.J. Saponins from the traditional medicinal plant Momordica charantia stimulate insulin secretion in vitro. Phytomedicine 2011, 19, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Sasa, M.; Inoue, I.; Shinoda, Y.; Takahashi, S.; Seo, M.; Komoda, T.; Awata, T.; Katayama, S. Activating effect of momordin, extract of bitter melon (Momordica Charantia L.), on the promoter of human PPAR delta. J. Atheroscler. Thromb. 2009, 16, 888–892. [Google Scholar] [CrossRef]
- Khanna, P.; Jain, S.C.; Panagariya, A.; Dixit, V.P. Hypoglycemic activity of polypeptide-p from a plant source. J. Nat. Prod. 1981, 44, 648–655. [Google Scholar] [CrossRef]
- Dans, A.M.; Villarruz, M.V.; Jimeno, C.A.; Javelosa, M.A.U.; Chua, J.; Bautista, R.; Velez, G.G.B. The effect of Momordica charantia capsule preparation on glycemic control in type 2 diabetes mellitus needs further studies. J. Clin. Epidemiol. 2007, 60, 554–559. [Google Scholar] [CrossRef]
- Jiang, B.; Ji, M.; Liu, W.; Chen, L.; Cai, Z.; Zhao, Y.; Bi, X. Antidiabetic activities of a cucurbitane-type triterpenoid compound from Momordica charantia in alloxan-induced diabetic mice. Mol. Med. Rep. 2016, 14, 4865–4872. [Google Scholar] [CrossRef]
- Akhtar, N.; Khan, B.A.; Majid, A.; Khan, H.M.S.; Mahmood, T.; Gulfishan; Saeed, T. Pharmaceutical and biopharmaceutical evaluation of extracts from different plant parts of indigenous origin for their hypoglycemic responses in rabbits. Acta Pol. Pharm. 2011, 68, 919–925. [Google Scholar]
- Ma, J.; Whittaker, P.; Keller, A.C.; Mazzola, E.P.; Pawar, R.S.; White, K.D.; Callahan, J.H.; Kennelly, E.J.; Krynitsky, A.J.; Rader, J.I. Cucurbitane-type triterpenoids from Momordica charantia. Planta Med. 2010, 76, 1758–1761. [Google Scholar] [CrossRef]
- Singh, N.; Gupta, M.; Sirohi, P.; Varsha. Effects of alcoholic extract of Momordica charantia (Linn.) whole fruit powder on the pancreatic islets of alloxan diabetic albino rats. J. Environ. Biol. 2008, 29, 101–106. [Google Scholar] [PubMed]
- Singh, N.; Gupta, M. Regeneration of beta cells in islets of Langerhans of pancreas of alloxan diabetic rats by acetone extract of Momordica charantia (Linn.) (bitter gourd) fruits. Indian J. Exp. Biol. 2007, 45, 1055–1062. [Google Scholar] [PubMed]
- Mahmoud, M.F.; El Ashry, F.E.; El Maraghy, N.N.; Fahmy, A. Studies on the antidiabetic activities of Momordica charantia fruit juice in streptozotocin-induced diabetic rats. Pharm. Biol. 2017, 55, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Kim, H.Y. Bitter melon (Momordica charantia) extract suppresses cytokine induced activation of MAPK and NF-κB in pancreatic β-cells. Food Sci. Biotechnol. 2011, 20, 531–535. [Google Scholar] [CrossRef]
- Paul, A.; Raychaudhuri, S.S. Medicinal uses and molecular identification of two Momordica charantia varieties—A review. Ej Bio 2010, 6, 43–51. [Google Scholar]
- Poovitha, S.; Parani, M. In Vitro and in Vivo α-amylase and α-glucosidase Inhibiting Activities of the Protein Extracts from Two Varieties of Bitter Gourd (Momordica Charantia L.). BMC Complement. Altern. Med. 2016, 16, 185. [Google Scholar] [CrossRef]
- Attele, A.S.; Zhou, Y.P.; Xie, J.T.; Wu, J.A.; Zhang, L.; Dey, L.; Pugh, W.; Rue, P.A.; Polonsky, K.S.; Yuan, C.-S. Antidiabetic effects of Panax ginseng berry extract and the identification of an effective component. Diabetes 2002, 51, 1851–1858. [Google Scholar] [CrossRef]
- Park, J.D.; Rhee, D.K.; Lee, Y.H. Biological Activities and Chemistry of Saponins from Panax ginseng C. A. Meyer. Phytochem. Rev. 2005, 4, 159–175. [Google Scholar] [CrossRef]
- Ratan, Z.A.; Haidere, M.F.; Hong, Y.H.; Park, S.H.; Lee, J.-O.; Lee, J.; Cho, J.Y. Pharmacological potential of ginseng and its major component ginsenosides. J. Ginseng Res. 2020. [Google Scholar] [CrossRef]
- Konno, C.; Murakami, M.; Oshima, Y.; Hikino, H. Isolation and hypoglycemic activity of panaxans Q, R, S, T and U, glycans of Panax ginseng roots. J. Ethnopharmacol. 1985, 14, 69–74. [Google Scholar] [CrossRef]
- Kimura, M.; Waki, I.; Tanaka, O.; Nagai, Y.; Shibata, S. Pharmacological sequential trials for the fractionation of components with hypoglycemic activity in alloxan diabetic mice from ginseng radix. J. Pharmacobiodyn. 1981, 4, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Kobayashi, T.; Oura, H.; Kawashima, Y. Studies on the mechanism of the hypoglycemic activity of ginsenoside-Rb2 in streptozotocin-diabetic rats. Chem. Pharm. Bull. 1985, 33, 869–872. [Google Scholar] [CrossRef]
- Kim, H.-J.; Chae, I.-G.; Lee, S.-G.; Jeong, H.-J.; Lee, E.-J.; Lee, I.-S. Effects of Fermented Red Ginseng Extracts on Hyperglycemia in Streptozotocin-induced Diabetic Rats. J. Ginseng Res. 2010, 34, 104–112. [Google Scholar] [CrossRef]
- Shao, J.-W.; Jiang, J.-L.; Zou, J.-J.; Yang, M.-Y.; Chen, F.-M.; Zhang, Y.-J.; Jia, L. Therapeutic potential of ginsenosides on diabetes: From hypoglycemic mechanism to clinical trials. J. Funct. Foods 2020, 64, 103630. [Google Scholar] [CrossRef]
- Bai, L.; Gao, J.; Wei, F.; Zhao, J.; Wang, D.; Wei, J. Therapeutic Potential of Ginsenosides as an Adjuvant Treatment for Diabetes. Front. Pharmacol. 2018, 9, 423. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Sen, S.; Lui, E. Effect of ginseng therapy on diabetes and its chronic complications: Lessons learned. J. Altern. Complement. Med. 2017, 14, 20160166. [Google Scholar] [CrossRef] [PubMed]
- Bao, T.T. Effect of total saponin of Panax ginseng on alloxan diabetes in mice. Acta Pharm. Sin. 1981, 16, 618–620. [Google Scholar]
- Chen, W.; Balan, P.; Popovich, D.G. Review of Ginseng Anti-diabetic Studies. Molecules 2019, 24, 4501. [Google Scholar] [CrossRef]
- Hyun, S.H.; Kim, S.W.; Won, H.; Youn, H.; Kyung, J.S.; Lee, Y.Y.; In, G.; Park, C.-K.; Han, C.-K. Physiological and pharmacological features of the non-saponin components in Korean Red Ginseng. J. Ginseng Res. 2020, 44, 527–537. [Google Scholar] [CrossRef]
- Zhou, P.; Xie, W.; He, S.; Sun, Y.; Meng, X.; Sun, G.; Sun, X. Ginsenoside Rb1 as an Anti-Diabetic Agent and Its Underlying Mechanism Analysis. Cells 2019, 8, 204. [Google Scholar] [CrossRef]
- World Health Organization. WHO Consultation on Selected Medicinal Plants, 2nd ed.; WHO: Ravello-Salerno, Italy, 1999. [Google Scholar]
- Kumari, K.; Mathew, B.C.; Augusti, K.T. Antidiabetic and hypolipidemic effects of S-methyl cysteine sulfoxide isolated from Allium cepa Linn. Indian J. Biochem. Biophys. 1995, 32, 49–54. [Google Scholar] [PubMed]
- Lakshmi, M.S.; Rani, K.S.S.; Reddy, U.T.K. A Review on Diabetes Mellitus and the Herbal Plants Used for Its Treatment. Asian J. Pharm. Clin. Res. 2012, 5, 15–21. [Google Scholar]
- Galal, E.E.; Gawad, M.A. Antidiabetic activity of Egyptian onion Allium cepa extract. J. Egypt. Med. Assoc. 1965, 48, 14–45. [Google Scholar]
- Kumari, K.; Augusti, K.T. Antidiabetic and antioxidant effects of S-methyl cysteine sulfoxide isolated from onions (Allium cepa Linn) as compared to standard drugs in alloxan diabetic rats. Indian. J. Exp. Biol. 2002, 40, 1005–1009. [Google Scholar]
- Das, S. Garlic–A Natural Source of Cancer Preventive Compounds. Asian Pac. J. Cancer Prev. 2002, 3, 305–311. [Google Scholar] [PubMed]
- Singh, V.; Kumar, R. Study of Phytochemical Analysis and Antioxidant Activity of Allium sativum of Bundelkhand Region. Int. J. Life Sci. Scienti. Res. 2017, 3, 1451–1458. [Google Scholar] [CrossRef]
- El-Saber Batiha, G.; Beshbishy, A.M.; Wasef, L.G.; Elewa, Y.H.; Al-Sagan, A.A.; El-Hack, M.E.A.; Taha, A.E.; Abd-Elhakim, Y.M.; Devkota, H.P. Chemical Constituents and Pharmacological Activities of Garlic (Allium sativum L.): A Review. Nutrients 2020, 12, 872. [Google Scholar] [CrossRef] [PubMed]
- Nasir, A.; Fatma, G.; Neshat, N.; Ahmad, M.A. Pharmacological and therapeutic attributes of garlic (Allium sativum Linn.) with special reference to Unani medicine—A review. J. Med. Plants Stud. 2020, 8, 6–9. [Google Scholar]
- Chauhan, A.; Sharma, P.K.; Srivastava, P.; Kumar, N. Plants having potential antidiabetic activity: A review. Der. Pharm. Lett. 2010, 2, 369–387. [Google Scholar]
- Bnouham, M.; Ziyyat, A.; Mekhfi, H. Medicinal plants with potential antidiabetic activity-a review of ten years of herbal medicine research (1990–2000). Int. J. Diabetes Metab. 2006, 14, 1–25. [Google Scholar] [CrossRef]
- Eidi, A.L.; Eidi, M.; Esmaeili, E. Antidiabetic effect of garlic (Allium sativum L.) in normal and streptozotocin-induced diabetic rats. Phytomedicine 2006, 13, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Martha, T.; Zainab, A.-A.; Khaled, A.-Q.; Lemia, S.; Muslim, A. Antidiabetic and hypolipidaemic properties of garlic (Allium sativum) in streptozotocin-induced diabetic rats. Int. J. Diabetes Metab. 2007, 15, 108–115. [Google Scholar]
- Adinortey, M.B.; Agbeko, R.; Boison, D.; Ekloh, W.; Kuatsienu, L.E.; Biney, E.E.; Kwarteng, J.; Nyarko, A.K. Phytomedicines Used for Diabetes Mellitus in Ghana: A Systematic Search and Review of Preclinical and Clinical Evidence. Evid. Based Complement. Altern. Med. 2019, 6021209. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M. Effect of aloes on blood glucose levels in normal and alloxan diabetic mice. J. Ethnopharmacol. 1990, 28, 215–220. [Google Scholar]
- Patel, D.K.; Patel, K.; Dhanabal, S.P. Phytochemical standardization of Aloe vera extract by HPTLC technique. J. Acute Dis. 2012, 47–50. [Google Scholar] [CrossRef]
- Yimam, M.; Zhao, J.; Corneliusen, B.; Pantier, M.; Brownell, L.; Jia, Q. Blood glucose lowering activity of aloe based composition, UP780, in alloxan induced insulin dependent mouse diabetes model. Diabetol. Metab. Syndr. 2014, 6, 61. [Google Scholar] [CrossRef]
- Kim, K.; Kim, H.; Kwon, J.; Lee, S.; Kong, H.; Im, S.-A.; Lee, Y.-H.; Lee, Y.-R.; Oh, S.-T.; Jo, T.H.; et al. Hypoglycemic and hypolipidemic effects of processed Aloe vera gel in a mouse model of non-insulin-dependent diabetes mellitus. Phytomedicine 2009, 16, 856–863. [Google Scholar] [CrossRef]
- Gupta, R.; Gupta, R.S. Effect of Pterocarpus Marsupium In Streptozotocin-Induced Hyperglycemic State In Rats: Comparison With Glibenclamide. Diabetol. Croat. 2009, 38, 39–45. [Google Scholar]
- Rahman, M.S.; Mujahid, M.D.; Siddiqui, M.A.; Rahman, M.A.; Arif, M.; Eram, S.; Khan, A.; Azeemuddin, M. Ethnobotanical uses, Phytochemistry and Pharmacological Activities of Pterocarpus marsupium: A review. Pharmacog. J. 2018, 10. [Google Scholar] [CrossRef]
- Chakravarthy, B.K.; Gupta, S.; Gambhir, S.S.; Gode, K.D. Pancreatic β-cell regeneration-A novel anti diabetic mechanism of Pterocarpus marsupium Roxb. Indian J. Pharmacol. 1980, 12, 123–127. [Google Scholar]
- Adinarayana, D.; Syamasundar, K.V. A new sesquiterpene alcohol from Pterocarpus marsupium. Phytochemistry 1982, 21, 1083–1085. [Google Scholar] [CrossRef]
- Halagappa, K.; Girish, H.N.; Srinivasan, B.P. The study of aqueous extract of Pterocarpus marsupium Roxb. on cytokine TNF-α in type 2 diabetic rats. Indian J. Pharmacol. 2010, 42, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.; Khalid, P.; Khan, M.M.; Rastogi, A.K.; Kidwai, J.R. Insulin like activity in (-) epicatechin. Acta Diabetol. Lat. 1989, 26, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.; Nayak, P.; Prusty, S.K.; Sahu, P.K. Phytochemistry and pharmacology of Tinospora cordifolia: A Review. Syst. Rev. Pharm. 2018, 9, 70–78. [Google Scholar] [CrossRef]
- Sharma, P.; Dwivedee, B.P.; Bisht, D.; Dash, A.K.; Kumar, D. The chemical constituents and diverse pharmacological importance of Tinospora cordifolia. Heliyon 2019, 5, e024372. [Google Scholar] [CrossRef]
- Kumar, D.V.; Geethanjali, B.; Avinash, K.O.; Kumar, J.R.; Basalingappa, K.M. Tinospora cordifolia: The antimicrobial property of the leaves of amruthaballi. J. Bacteriol. Mycol. Open Access 2017, 5, 363–371. [Google Scholar] [CrossRef]
- Rajalakshmi, M.; Anita, R. β-cell regenerative efficacy of a polysaccharide isolated from methanolic extract of Tinospora cordifolia stem on streptozotocin-indued diabetic Wistae rats. Chem. Biol. Interact. 2016, 243, 45–53. [Google Scholar] [CrossRef]
- Sangeetha, M.K.; Balaji Raghavendran, H.R.; Gayathri, V.; Vasanthi, H.R. Tinospora cordifolia attenuates oxidative stress and distorted carbohydrat metabolism in experimentally induced type 2 diabetes in rats. J. Nat. Med. 2011, 65, 544–550. [Google Scholar] [CrossRef]
- Patel, M.B.; Mishra, S. Hypoglycemic activity of alkaloidal fraction of Tinospora cordifolia. Phytomedicine 2011, 18, 1045–1052. [Google Scholar] [CrossRef]
- Ahmad, W.; Jantan, I.; Bukhari, S.N.A. Tinosporaa crispa (L.) Hook. F. & Thomson: A Review of Its Ethnobotanical, Phytochemical, and Pharmacological Aspects. Front. Pharmacol. 2016, 7. [Google Scholar] [CrossRef]
- Pathak, A.K.; Jain, D.C.; Sharma, R.P. Chemistry and biological activities of the Genera Tinospora. Pharm. Biol. 1995, 33, 277–287. [Google Scholar] [CrossRef]
- Noor, H.; Ashcroft, S.J. Pharmacological characterisation of the antihyperglycaemic properties of Tinospora crispa extract. J. Ethnopharmacol. 1998, 62, 7–13. [Google Scholar] [CrossRef]
- Choudhary, M.I.; Ismail, M.; Ali, Z.; Shaari, K.; Lajis, N.H.; Rahman, A. Alkaloidal constituents of Tinospora crispa. Nat. Prod. Commun. 2010, 5, 1747–1750. [Google Scholar] [CrossRef] [PubMed]
- Umi Kalsom, Y.; Noor, H. Flavone O-glycosides from Tinospora crispa. Fitoterapia 1995, 66, 280. [Google Scholar]
- Koay, Y.C.; Amir, F. A Review of the Secondary Metabolites and Biological Activities of Tinospora crispa (Menispermaceae). Trop. J. Pharm. Res. 2013, 12, 641–649. [Google Scholar] [CrossRef]
- Noipha, K.; Ratanachaiyavong, S.; Purintrapiban, J.; Herunsalee, A.; Ninla-aesong, P. Effect of Tinospora crispa on glucose uptake in skeletal muscle: Role of glucose transporter 1 expression and extracellular signal-regulated kinase1/2 activation. Asian Biomed. 2011, 5, 361–369. [Google Scholar] [CrossRef]
- Klangjareonchai, T.; Roongpisuthipong, C. The Effect of Tinospora crispa on Serum Glucose and Insulin Levels in Patients with Type 2 Diabetes Mellitus. Hindawi Publ. Corp. J. Biomed. Biotechnol. 2012, 808762. [Google Scholar] [CrossRef]
- Tiwari, P.; Mishra, B.N.; Sangwan, N.S. Phytochemical and pharmacological properties of Gymnema sylvestre: An important medicinal plant. BioMed Res. Int. 2014, 2014, 830285. [Google Scholar] [CrossRef]
- Khan, F.; Sarker, M.M.R.; Ming, L.C.; Mohamed, I.N.; Zhao, C.; Sheikh, B.Y.; Tsong, H.F.; Rashid, M.A. Comprehensive Review on Phytochemicals, Pharmacological and Clinical Potentials of Gymnema sylvestre. Front. Pharmacol. 2019, 10, 1223. [Google Scholar] [CrossRef]
- Sathya, S.; Kokilavani, R.; Gurusamy, K. Hypoglycemic effect of Gymnema sylvestre (retz.,) R.Br leaf in normal and alloxan induced diabetic rats. Anc. Sci. Life 2008, 28, 12–14. [Google Scholar]
- Persaud, S.J.; Al-Majed, H.; Raman, A.; Jones, P.M. Gymnema sylvestre stimulates insulin release in vitro by increased membrane permeability. J. Endocrinol. 1999, 163, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Ananda, U.; Nandyb, S.; Mundhra, A.; Das, N.; Pandey, D.K.; Dey, A. A review on antimicrobial botanicals, phytochemicals and natural resistance modifying agents from Apocynaceae family: Possible therapeutic approaches against multidrug resistance in pathogenic microorganisms. Drug Resist. Updat. 2020, 51, 100695. [Google Scholar] [CrossRef] [PubMed]
- Spasov, A.A.; Samokhina, M.P.; Bulanov, A.E. Antidiabetic properties of Gymnema sylvestre (a review). Pharm. Cham. J. 2008, 42, 626. [Google Scholar] [CrossRef]
- Laha, S.; Paul, S. Gymnema sylvestre (Gurmar): A Potent Herb with Anti-Diabetic and Antioxidant Potential. Pharmacog. J. 2019, 11, 201–206. [Google Scholar] [CrossRef]
- Sharma, P.; Dubey, G.; Kaushik, S. Chemical and Medico-biological profile of Cyamopsis tetragonoloba (L) Taub: An overview. J. Appl. Pharm. Sci. 2011, 1, 32–37. [Google Scholar]
- Rahman, A.U.; Zaman, K. Medicinal plants with hypoglycemic activity. J. Ethnopharmacol. 1989, 26, 1–55. [Google Scholar] [CrossRef]
- Hayat, M.; Mukhtar, S.H.; Ansari, Z.A.B.; Naved, T. Antihyperglycemic Activity of Cyamopsis tetragonoloba. Beans on Blood Glucose Levels in Alloxan-Induced Diabetic Rats. Pharm. Biol. 2006, 44, 10–13. [Google Scholar] [CrossRef]
- Gandhi, G.R.; Vanlalhruaia, P.; Stalin, A.; Irudayaraj, S.S.; Ignacimuthu, S.; Paulraj, M.G. Polyphenols-rich Cyamopsis tetragonoloba (L.) Taub. beans show hypoglycemic and β-cells protective effects in type 2 diabetic rats. Food Chem. Toxicol. 2014, 66, 358–365. [Google Scholar] [CrossRef]
- Das, S.K.; Vasudevan, D.M. Tulsi: The Indian holy power plant. Nat. Prod. Rad. 2006, 5, 279–283. [Google Scholar]
- Verma, S. Chemical constituents and pharmacological action of Ocimum sanctum (Indian holy basil-Tulsi). J. Phytopharmacol. 2016, 5, 205–207. [Google Scholar]
- Rahman, S.; Islam, R.; Kamruzzaman, Md.; Alam, M.K. Ocimum sanctum L.: A review of phytochemical and pharmacological profile. Am. Drug Discov. Dev. 2011, 1–15. [Google Scholar] [CrossRef]
- Singh, D.; Chaudhuri, P.K. A review on phytochemical and pharmacological properties of Holy basil (Ocimum sanctum L.). Ind. Crops Prod. 2018, 118, 367–382. [Google Scholar] [CrossRef]
- Hannan, J.M.; Marenah, L.; Ali, L.; Rokeya, B.; Flatt, P.R.; Abdel-Wahab, Y.H. Ocimum sanctum leaf extracts stimulate insulin secretion from perfused pancreas, isolated islets and clonal pancreatic beta-cells. J. Endocrinol. 2006, 189, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Hannan, J.M.A.; Ojo, O.O.; Ali, L.; Rokeya, B.; Khaleque, J.; Akhter, M.; Flatt, P.R.; Abdel-Wahab, Y.H.A. Actions Underlying Antidiabetic Effects of Ocimum sanctum Leaf Extracts in Animal Models of Type 1 and Type 2 Diabetes. Eur. J. Med. Plants 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Patil, R.; Patil, R.; Ahirwar, B.; Ahirwar, D. Isolation and characterization of anti-diabetic component (bioactivity-guided fractionation) from Ocimum sanctum L. (Lamiaceae) aerial part. Asian Pac. J. Trop. Med. 2011, 4, 278–282. [Google Scholar] [CrossRef]
- Kapoor, S. Ocimum sanctum: A therapeutic role in diabetes and the metabolic syndrome. Horm. Metab. Res. 2008, 40, 296. [Google Scholar] [CrossRef]
- Kim, H.Y.; Sin, S.M.; Lee, S.; Cho, K.M.; Cho, E.J. The Butanol Fraction of Bitter Melon (Momordica charantia) Scavenges Free Radicals and Attenuates Oxidative Stress. Prev. Nutr. Food Sci. 2013, 18, 18–22. [Google Scholar] [CrossRef]
- Tsai, T.H.; Huang, C.J.; Wu, W.H.; Huang, W.C.; Chyuan, J.H.; Tsai, P.J. Antioxidant, cell-protective, and anti-melanogenic activities of leaf extracts from wild bitter melon (Momordica charantia Linn. var. abbreviata Ser.) cultivars. Bot. Stud. 2014, 55, 78. [Google Scholar] [CrossRef]
- Rathi, S.S.; Grover, J.K.; Vikrant, V.; Biswas, N.R. Prevention of Experimental Diabetic Cataract by Indian Ayurvedic Plant Extracts. Phytother. Res. 2002, 16, 774–777. [Google Scholar] [CrossRef]
- Kwon, Y.I.; Apostolidis, E.; Kim, Y.C.; Shetty, K. Health benefits of traditional corn, beans and pumpkin: In vitro studies for hyperglycemia and hypertension management. J. Med. Food 2007, 10, 266–275. [Google Scholar] [CrossRef]
- Kazeem, M.I.; Ashafa, A.O.T. In-vitro antioxidant and antidiabetic potentials of Dianthus basuticus Burtt Davy whole plant extract. J. Herb. Med. 2015, 5, 158–164. [Google Scholar] [CrossRef]
- Bhutkar, M.; Bhise, S.B. In vitro assay of alpha amylase inhibitory activity of some indigenous plants. Int. J. Chem. Sci. 2012, 10, 457–462. [Google Scholar] [CrossRef]
- Mahmood, N. A review of α-amylase inhibitors on weight loss and glycemic control in pathological state such as obesity and diabetes. Comp. Clin. Path. 2016, 25, 1253–1264. [Google Scholar] [CrossRef]
- de Souza, P.M.; de Oliveira e Magalhães, P. Application of microbial α-amylase in industry—A review. Braz. J. Microbiol. 2010, 41, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Radha Devi, G.M. A Comprehensive Review On Costus Pictus D. Don. Int. J. Pharm. Sci. Res. 2019, 10, 3187–3195. [Google Scholar] [CrossRef]
- Prakash, K.; Harini, H.; Rao, A.; Rao, P.N. A review on Isulin plant (Costus igneus Nak). Pharmacogn. Rev. 2014, 8, 67–72. [Google Scholar] [CrossRef]
- Jothivel, N.; Ponnusamy, S.P.; Appachi, M.; Singaravel, S.; Rasilingam, D.; Deivasigamani, K.; Thangavel, S. Anti-diabetic activity of methanol leaf extract of Costus pictus D. Don in alloxan-induced diabetic rats. J. Health Sci. 2007, 53, 655–663. [Google Scholar] [CrossRef]
- Peasari, J.R.; Motamarry, S.S.; Varma, K.S.; Anitha, P.; Potti, R.B. Chromatographic analysis of phytochemicals in Costus igneus and computational studies of flavonoids. Inform. Med. Unlocked 2018, 13, 34–40. [Google Scholar] [CrossRef]
- Sidhu, A.K.; Wani, S.; Tamboli, P.S.; Patil, S.N. In Vitro Evaluation of Anti-Diabetic Activity of Leaf and Callus Extracts of Costus pictus. Int. J. Recent Sci. Res. 2014, 3, 1622–1625. [Google Scholar]
- Jayasri, M.A.; Gunasekaran, S.; Radha, A.; Mathew, T.L. Antidiabetic effect of Costus pictus leaves in normal and streptozotocin-induced diabetes rats. Int. J. Diabetes Metab. 2008, 16, 117–122. [Google Scholar]
- Radha, J.M.A.; Mathew, T.L. α–amylase and α-glucosidase inhibitory activity of Costus pictus D.Don in the Management of diabetes. J. Herb. Med. Toxicol. 2009, 3, 91–94. [Google Scholar]
- Ramu, F.A.; Kumar, S.R. Scientific evaluation of traditionally known insulin plant Costus species for the treatment of diabetes in human. Int. J. Curr. Res. Biosci. Plant Biol. 2016, 3, 87–91. [Google Scholar] [CrossRef]
- Gireesh, G.; Thomas, S.K.; Joseph, B.; Paulose, C.S. Antihyperglycemic and insulin secretory activity of Costus pictus leaf extract in streptozotocin induced diabetic rats and in in vitro pancreatic islet culture. J. Ethnopharmacol. 2009, 123, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Al-Romaiyan, A.; Jayasri, M.A.; Mathew, T.L.; Huang, G.-C.; Amiel, S.; Jones, P.M.; Persaud, S.J. Costus pictus extracts stimulate insulin secretion from mouse and human islets of Langerhans in vitro. Cell Physiol. Biochem. 2010, 26, 1051–1058. [Google Scholar] [CrossRef]
- Abu-Reidah, I.M.; Arráez-Román, D.; Lozano-Sánchez, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phytochemical characterisation of green beans (Phaseolus vulgaris L.) by using high-performance liquid chromatography coupled with time-of-flight mass spectrometry. Phytochem. Anal. 2013, 24, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Bollini, R.; Chrispeels, M.J. Characterization and subcellular localization of vicilin and phytohemagglutinin, the two major reserve proteins of Phaseolus vulgaris L. Planta 1978, 142, 291–298. [Google Scholar] [CrossRef]
- Los, F.G.B.; Zielinski, A.A.F.; Wojeicchowski, J.P.; Nogueira, A.; Demiate, I.M. Beans (Phaseolus vulgaris L.): Whole seeds with complex chemical composition. Curr. Opin. Food Sci. 2018, 19, 63–71. [Google Scholar] [CrossRef]
- Ishimoto, M.; Kitamura, K. Growth inhibitory effects of an alpha-amylase inhibitor from the kidney bean, Phaseolus vulgaris (L.) on three species of Bruchid (Cleoptera: Bruchidae). Appl. Ent. Zool. 1989, 24, 281–286. [Google Scholar]
- Ngoh, Y.; Gan, C. Identification of Pinto bean peptides with inhibitory effects on alfa-amylase and angiotensin converting enzyme (ACE) activities using an integrated bioinformatics assisted approach. Food Chem. 2018, 267, 124–131. [Google Scholar] [CrossRef]
- Nolan, R.; Shannon, O.M.; Robinson, N.; Joel, A.; Houghton, D.; Malcomson, F.C. It’s No Has Bean: A Review of the Effects of White Kidney Bean Extract on Body Composition and Metabolic Health. Nutrients 2020, 12, 1398. [Google Scholar] [CrossRef]
- Pueyo’, J.J.; Hunt, D.C.; Chrispeels, M.J. Activation of Bean (Phaseolus vulgaris) α-Amylase Inhibitor Requires Proteolytic Processing of the Proprotein. Plant Physiol. 1993, 101, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Barrett, M.L.; Udani, J.K. A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): A review of clinical studies on weight loss and glycemic control. Nutr. J. 2011, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Micheli, L.; Lucarini, E.; Trallori, E.; Avagliano, C.; De Caro, C.; Russo, R.; Calignano, A.; Ghelardini, C.; Pacini, A.; Di Cesare Mannelli, L. Phaseolus vulgaris L. extract: Alpha-amylase Inhibtion against Metabolic Syndrome in Mice. Nutrients 2019, 11, 1778. [Google Scholar] [CrossRef] [PubMed]
- Sivaji, A. Euphorbia hirta linn-a review on traditional uses, phytochemistry and pharmacology. World J. Pharm. Res. 2014, 3, 180–205. [Google Scholar]
- Widharna, R.M.; Soemardji, A.A.; Wirasutisna, K.R.; Kardono, L.B.S. Anti diabetes mellitus activity in vivo of ethanolic extract and ethyl acetate fraction of Euphorbia hirta L. herb. Int. J. Pharmacol. 2010, 6, 231–240. [Google Scholar] [CrossRef]
- Kumar, S.; Malhotra, R.; Kumar, D. Euphorbia hirta: Its chemistry, traditional and medicinal uses, and pharmacological activities. Pharmacogn. Rev. 2010, 4, 58–61. [Google Scholar] [CrossRef]
- Tran, N.; Tran, M.; Truong, H.; Le, L. Spray-drying microencapsulation of high concentration of bioactive compounds fragments from Euphorbia hirta L. extract and their effect on diabetes mellitus. Food 2020, 9, 881. [Google Scholar] [CrossRef]
- Sheliya, M.A.; Rayhana, B.; Ali, A.; Pillai, K.K.; Aeri, V.; Sharma, M.; Mir, S.R. Inhibition of α-glucosidase by new prenylated flavonoids from Euphorbia Hirta, L. herb. J. Ethnopharmacol. 2015, 176, 1–8. [Google Scholar] [CrossRef]
- Sheliya, M.A.; Begum, R.; Pillai, K.K.; Aeri, V.; Mir, S.R.; Ali, A.; Sharma, M. In vitro α-glucosidase and α-amylase inhibition by aqueous, hydroalcoholic, and alcoholic extract of Euphorbia hirta L. Drug Dev. Ther. 2016, 7, 26–30. [Google Scholar] [CrossRef]
- Trinh, Q.; Le, L. An investigation of antidiabetic activities of bioactive compounds in Euphorbia hirta Linn using molecular docking and pharmacophore. Med. Chem. Res. 2014, 23, 2033–2045. [Google Scholar] [CrossRef]
- Nasry, M.R.; Abo-Youssef, A.M.; Abd El-Latif, H.A. Anti-diabetic activity of the petroleum ether extract of Guar gum in streptozotocin-induced diabetic rats: A comparative study. Beni-Suef Univ. J. Basic Appl. Sci. 2013, 2, 51–59. [Google Scholar] [CrossRef][Green Version]
- Khattab, H.A.; El-Shitany, N.A.; Abdallah, I.Z.; Yousef, F.M.; Alkreathy, H.M. Antihyperglycemic Potential of Grewia asiatica Fruit Extract against Streptozotocin-Induced Hyperglycemia in Rats: Anti-Inflammatory and Antioxidant Mechanisms. Oxid. Med. Cell. Longev. 2015, 549743. [Google Scholar] [CrossRef]
- Wei, C.K.; Tsai, Y.H.; Korinek, M.; Hung, P.H.; El-Shazly, M.; Cheng, Y.B.; Wu, Y.C.; Hsieh, T.J.; Chang, F.R. 6-Paradol and 6-Shogaol, the Pungent Compounds of Ginger, Promote Glucose Utilization in Adipocytes and Myotubes, and 6-Paradol Reduces Blood Glucose in High-Fat Diet-Fed Mice. Int. J. Mol. Sci. 2017, 18, 168. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef]
- Imtiyaz, S.; Rahman, K.; Sultana, A.; Tariq, M.; Chaudhary, S.S. Zingiber officinale Rosc.: A traditional herb with medicinal properties. Tang 2013, 3. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, J.; Zhang, Y. Research Progress on Chemical Constituents of Zingiber officinale Roscoe. Hindawi BioMed Res. Int. 2019, 5370823. [Google Scholar] [CrossRef]
- Sharma, M.; Shukla, S. Hypoglycaemic effect of ginger. J. Res. Indian Yoga Homoeop. 1977, 12, 127–130. [Google Scholar]
- Abdulrazaq, N.B.; Cho, M.M.; Win, N.N.; Zaman, R.; Rahman, M.T. Beneficial effects of ginger (Zingiber officinale) on carbohydrate metabolism in streptozotocin-induced diabetic rats. Br. J. Nutr. 2012, 108, 1194–1201. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Tran, V.H.; Duke, C.C.; Roufogalis, B.D. Gingerols of Zingiber officinale enhance glucose uptake by increasing cell surface GLUT4 in cultured L6 myotubes. Planta Med. 2012, 78, 1549–1555. [Google Scholar] [CrossRef]
- Oboh, G.; Akinyemi, A.J.; Ademiluyi, A.O.; Adefegha, S. Inhibitory effects of aqueous extract of two varieties of ginger on some key enzymes linked to type-2 diabetes in vitro. J. Food Nutr. Res. 2010, 49, 14–20. [Google Scholar]
- Akinyemi, A.J.; Ademiluyi, A.O.; Oboh, G. Inhibition of angiotensin-1-converting enzyme activity by two varieties of ginger (Zingiber officinale) in rats fed a high cholesterol diet. J. Med. Food 2014, 17, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Sahardi, N.F.N.M.; Makpol, S. Ginger (Zingiber officinale Roscoe) in the Prevention of Ageing and Degenerative Diseases: Review of Current Evidence. Evid. Based Complement. Altern. Med. 2019, 5054395. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Vo, T.-H.; Le, T.; Pham, D.; Nguyen, T.; Le, P.; Nguyen, A.; Nguyen, T.; Nguyen, T.-N.; Nguyen, V.; Do, H.; et al. VIETHERB: A Database for Vietnamese Herbal Species. J. Chem. Inf. Model. 2019, 59, 1–9. [Google Scholar] [CrossRef]
- Thanh-Hoang, N.-V.; Loc, N.; Nguyet, D.; Thien-Ngan, N.; Khang, T.; Cao, H.; Le, L. Plant Metabolite Databases: From Herbal Medicines to Modern Drug Discovery. J. Chem. Inf. Model. 2020, 60, 1101–1110. [Google Scholar]












| Bioactive Compounds | Antidiabetic Effects |
|---|---|
| Polypeptide-p | Act as Insulin-like protein, decrease blood glucose level |
| Momordicosides | Enhance the uptake of glucose |
| Saponins | Stimulate insulin secretion, a lower blood glucose level |
| Conjugated linolenic acid | Release intestinal GLP-1 |
| Momordin | PPAR δ activation |
| 9c, 11t, 13t conjugated linolenic acid | PPAR α activation |
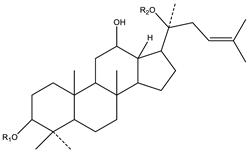 | 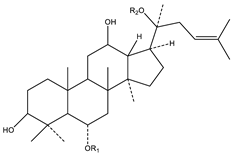 |
| A. Protopanaxadiol | B. Protopanaxatriol |
| R2 = Ra1, Ra2, Ra3, Rb1, Rb2, Rb3, Rc, Rd, Rg2, Rg3, Rs1, Rs2, etc. | R2 = Re, Rf, Rg1, Rg2, Rh1, etc. |
| Phytoconstituents | Classification |
|---|---|
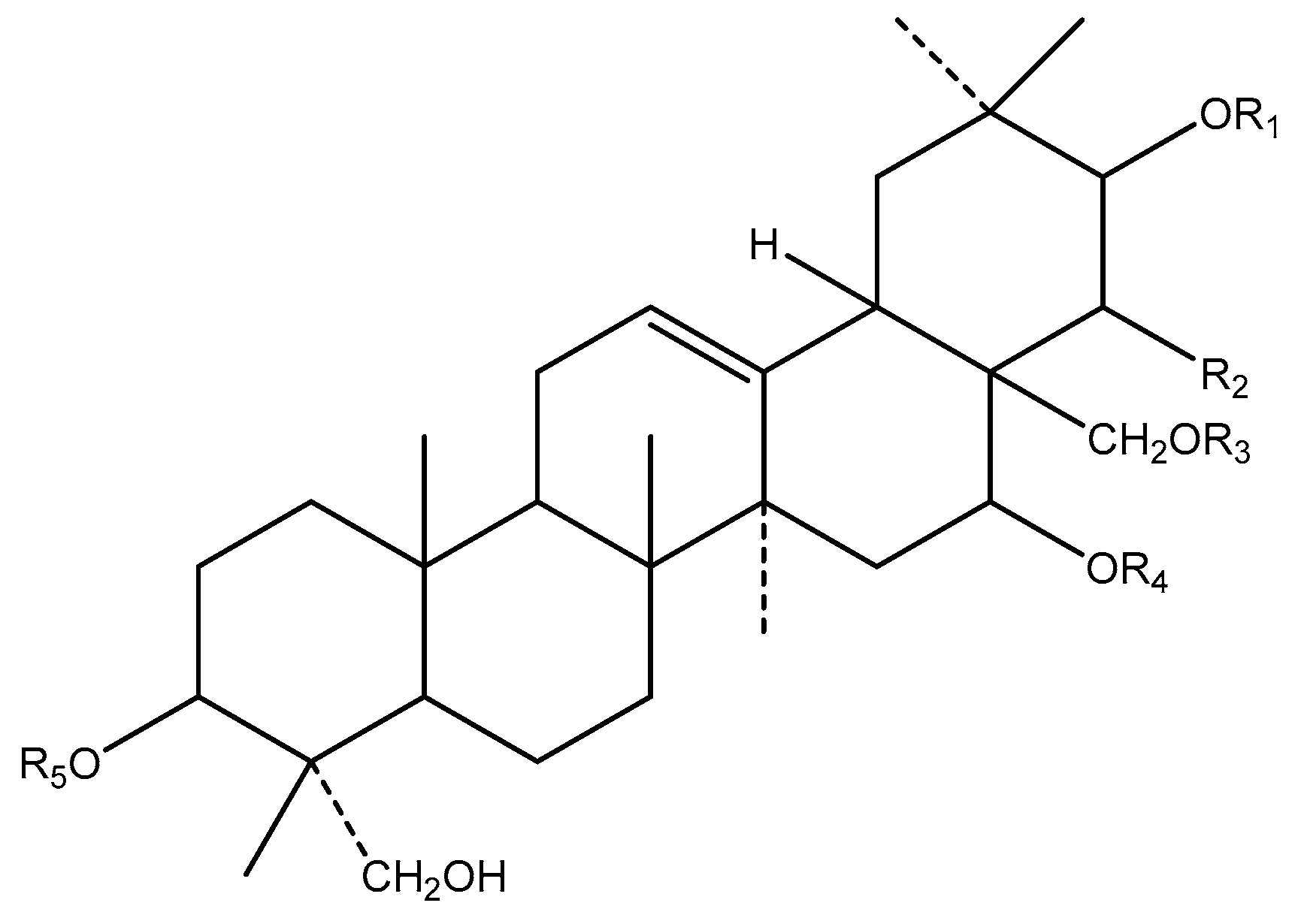
| Triterpene saponins | Gymnemic acids-acylated (tiglolyl, methylbutyroyl) derivatives of deacylgymnemic acid (DAGA) which is a 3-O-β-glucouronide of gymnemagenin (3β, 16β, 21β, 22α, 23, 28-hexahydroxy-olean-12-ene) |
| Oleanane saponins | Gymnemic acids and gymnemasaponins |
| Dammarene saponins | Gymnemosides A, B, C, D, E, and F |
| Gurmarin | A novel 35-amino-acid peptide with a 4209 molecular weight |
| Triterpenoidsaponins | |
| Gymnemasins A | 3-O [β-d-glucopyranosyl (1-3)-β-d-glucopyranosyl]-22-O-tiglyol gymnemanol |
| Gymnemasins B | 3-O-[β-d-glucopyranosyl-(1-3)-β-d-glucuro-nopyranosyl]-gymnemanol |
| Gymnemasins C | glucuronopyranosyl-22-O-tigloyl-gymnemanol |
| Gymnemasins D | 3-O-β-d-glucopyranosyl-gymnemanol |
| Gymnemanol | 3,β-16,β-22, α-23-28-pentahydroxyolean-12-ene |
| Gymmestrogenin | Pentahydroxytriterpene |
| Flavonol glycoside | Kaempferol 3-O-β-d-glucopyranosyl-(1-4)-α-l-rhamnopyranosyl-(1-6)- β-d-galactopyranoside |
| Sterols | Stigmasterol |
| Group | Phytochemical Compound |
|---|---|
| Phenolic acids | Hydroxybezoic acid and derivatives flavonoids, anthocyanins, flavonols, flavanols, isoflavones, flavanones, proanthocyanidins, and tannins |
| Hydroxycinnamic acid and derivatives | |
| Flavonoids | Orientin, isoerientin, rutin, myricetin, luteolin, quercetin, kaempferol, myricetin-3-rhamnoside, hyperoside, isorhamnetin-3-glucoside, isoquercitirn |
| Proteins | Vicilin, phytohenmagglutinin, alpha-amylase inhibitor (α-AI1, α-AI2, and α-AIL) |
| Group | Compound Name | Structure of Compound |
|---|---|---|
| Flavonols | Quercetin | 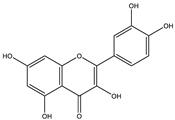 |
| Kaempferol |  | |
| Leucoanthocyanidins | Leucocyanidin | 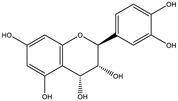 |
| Flavonoid glycosides | Rutin | 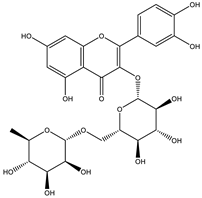 |
| Quercitrin | 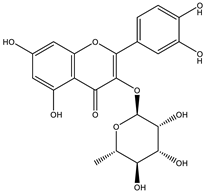 | |
| Myricitrin | 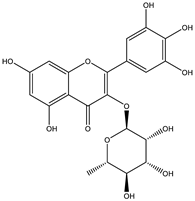 | |
| Afzelin |  | |
| Luteolin-7-O-glucoside | 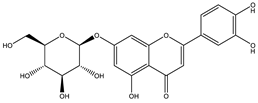 | |
| Isoquercitrin | 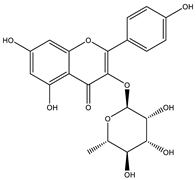 | |
| Acids | Syringic acid | 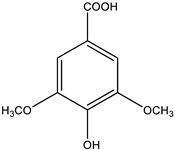 |
| Ellagic acid | 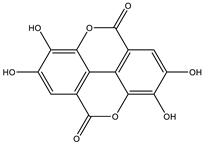 | |
| Gallic acid | 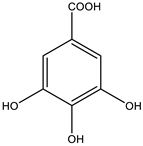 | |
| Shikimic acid |  | |
| Terpenoids | 2β,16α-dihydroxy-ent-kaurane | 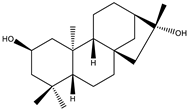 |
| Sterols | β-sitosterol | 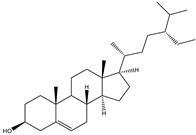 |
| Stigmasterol | 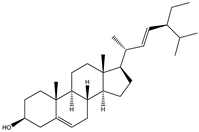 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, N.; Pham, B.; Le, L. Bioactive Compounds in Anti-Diabetic Plants: From Herbal Medicine to Modern Drug Discovery. Biology 2020, 9, 252. https://doi.org/10.3390/biology9090252
Tran N, Pham B, Le L. Bioactive Compounds in Anti-Diabetic Plants: From Herbal Medicine to Modern Drug Discovery. Biology. 2020; 9(9):252. https://doi.org/10.3390/biology9090252
Chicago/Turabian StyleTran, Ngan, Bao Pham, and Ly Le. 2020. "Bioactive Compounds in Anti-Diabetic Plants: From Herbal Medicine to Modern Drug Discovery" Biology 9, no. 9: 252. https://doi.org/10.3390/biology9090252
APA StyleTran, N., Pham, B., & Le, L. (2020). Bioactive Compounds in Anti-Diabetic Plants: From Herbal Medicine to Modern Drug Discovery. Biology, 9(9), 252. https://doi.org/10.3390/biology9090252






