Contributions of Chondroitin Sulfate, Keratan Sulfate and N-linked Oligosaccharides to Inhibition of Neurite Outgrowth by Aggrecan
Abstract
1. Introduction
2. Materials and Methods
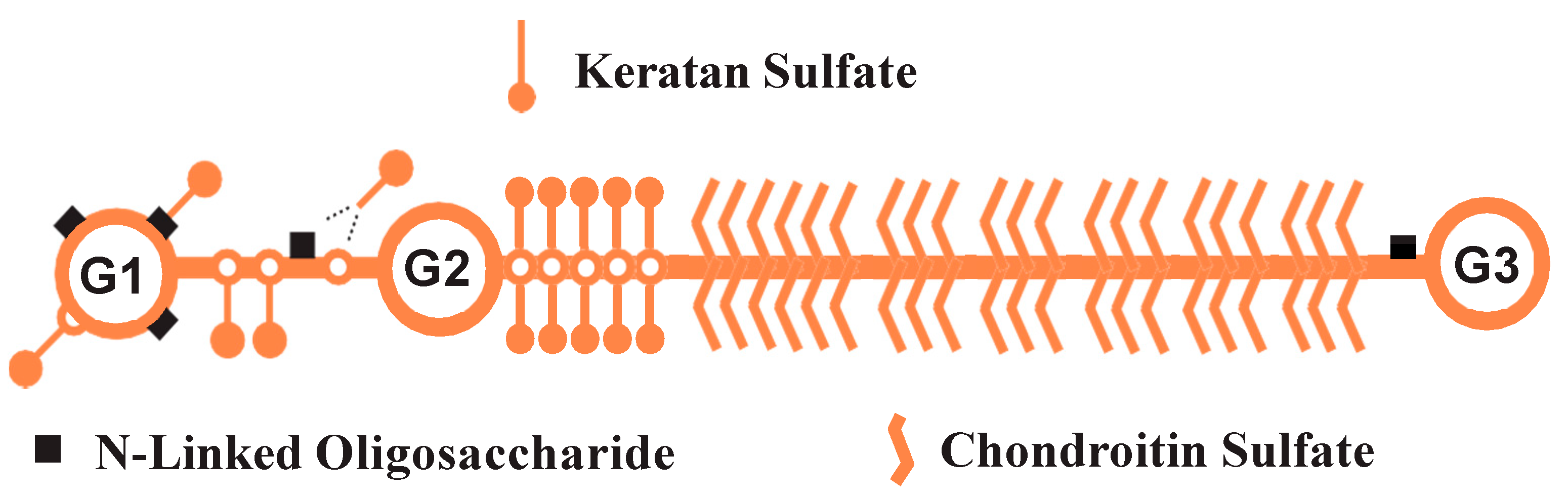
2.1. Preparation of Aggrecan
2.2. Enzyme Digestions
2.3. ELISA Assay
2.4. Neurite Outgrowth Assay
2.5. Image Acquisition
2.6. Data Analysis
3. Results
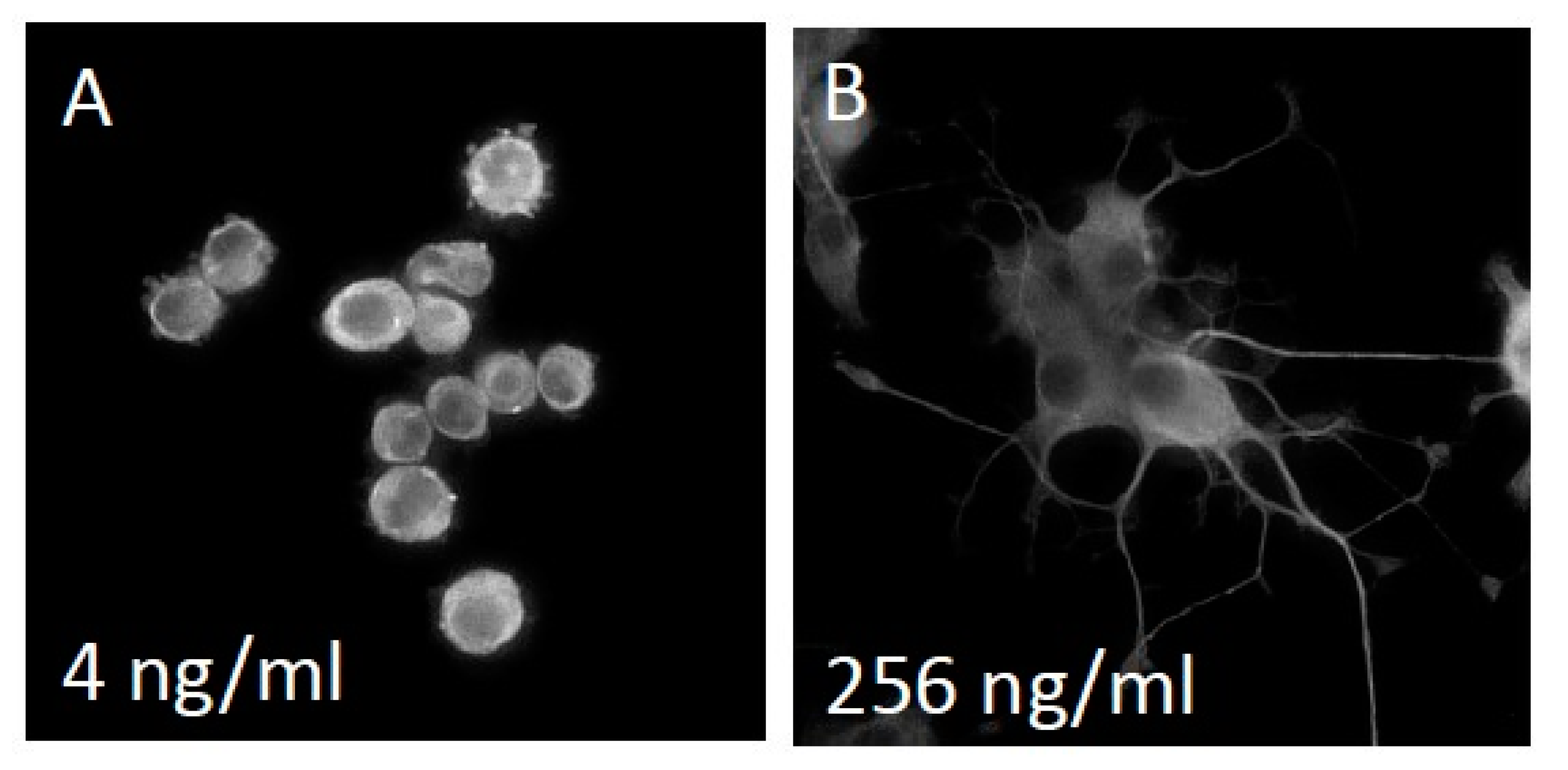
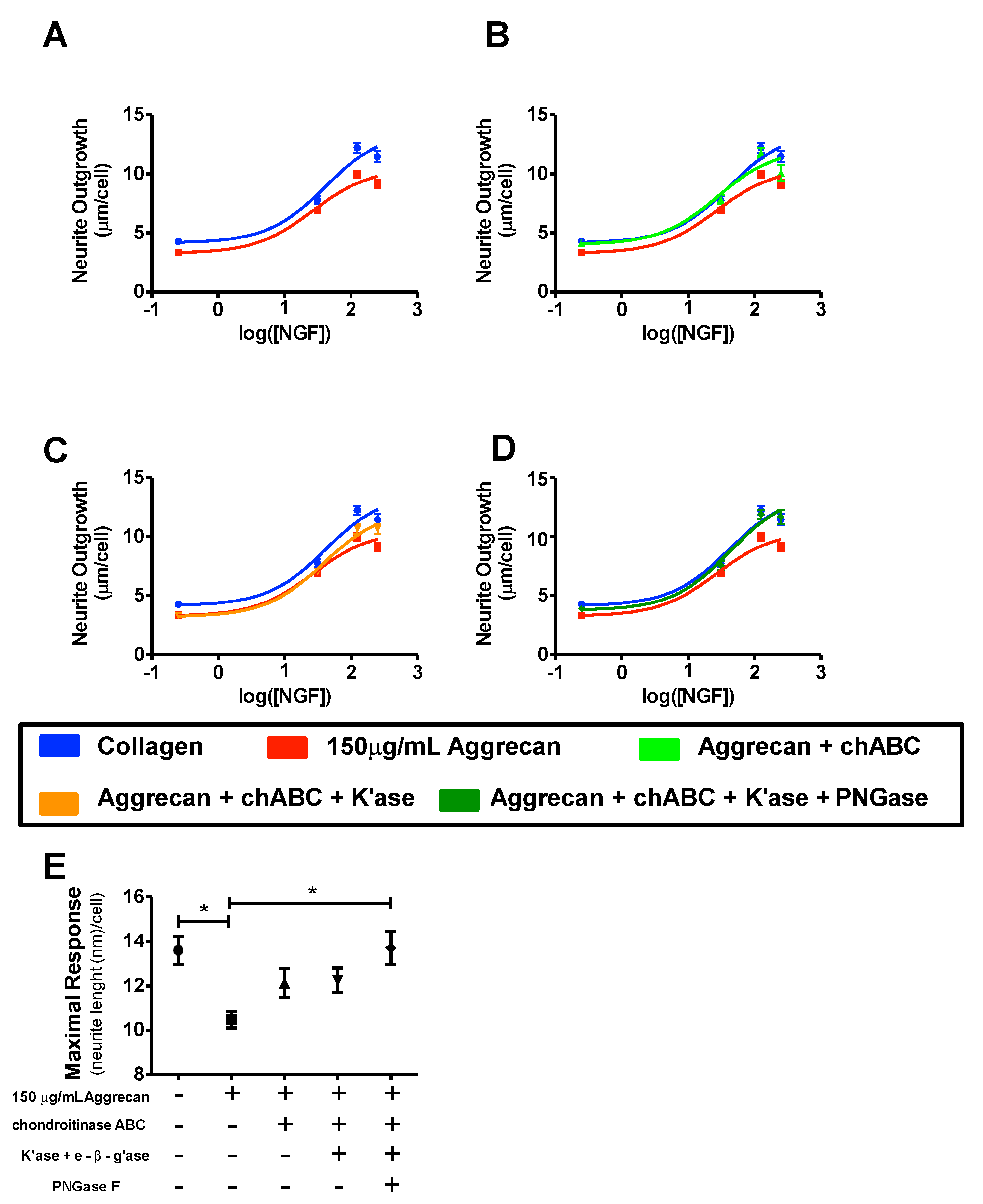
3.1. NS-1 Cells Extend Neurites in a Dose-Dependent Fashion When Exposed to Varying Amounts of NGF
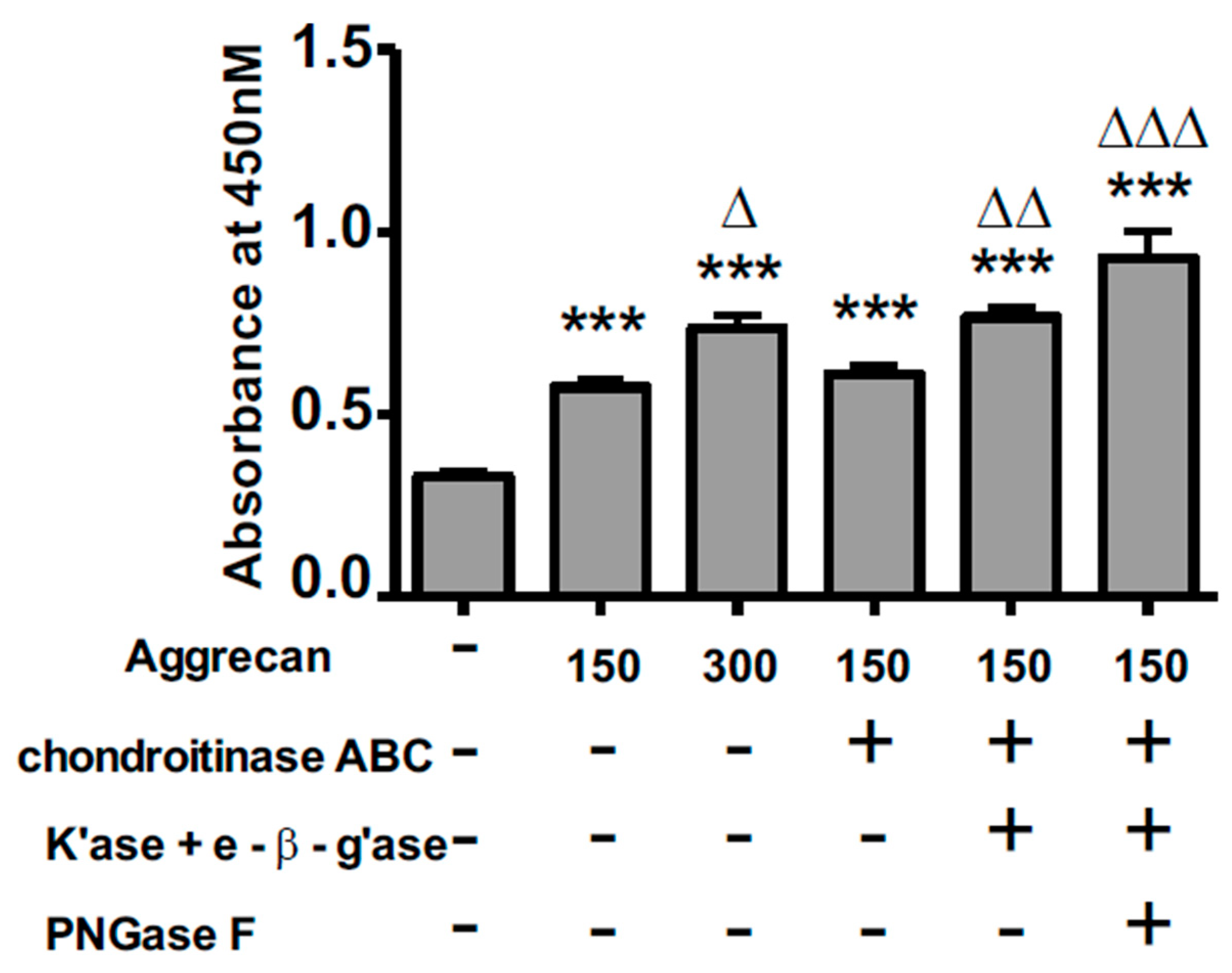
3.2. ELISA Assay Confirms Adsorption to Wells and Reveals Greater Access to G1-Domain Following Enzymatic Digestion
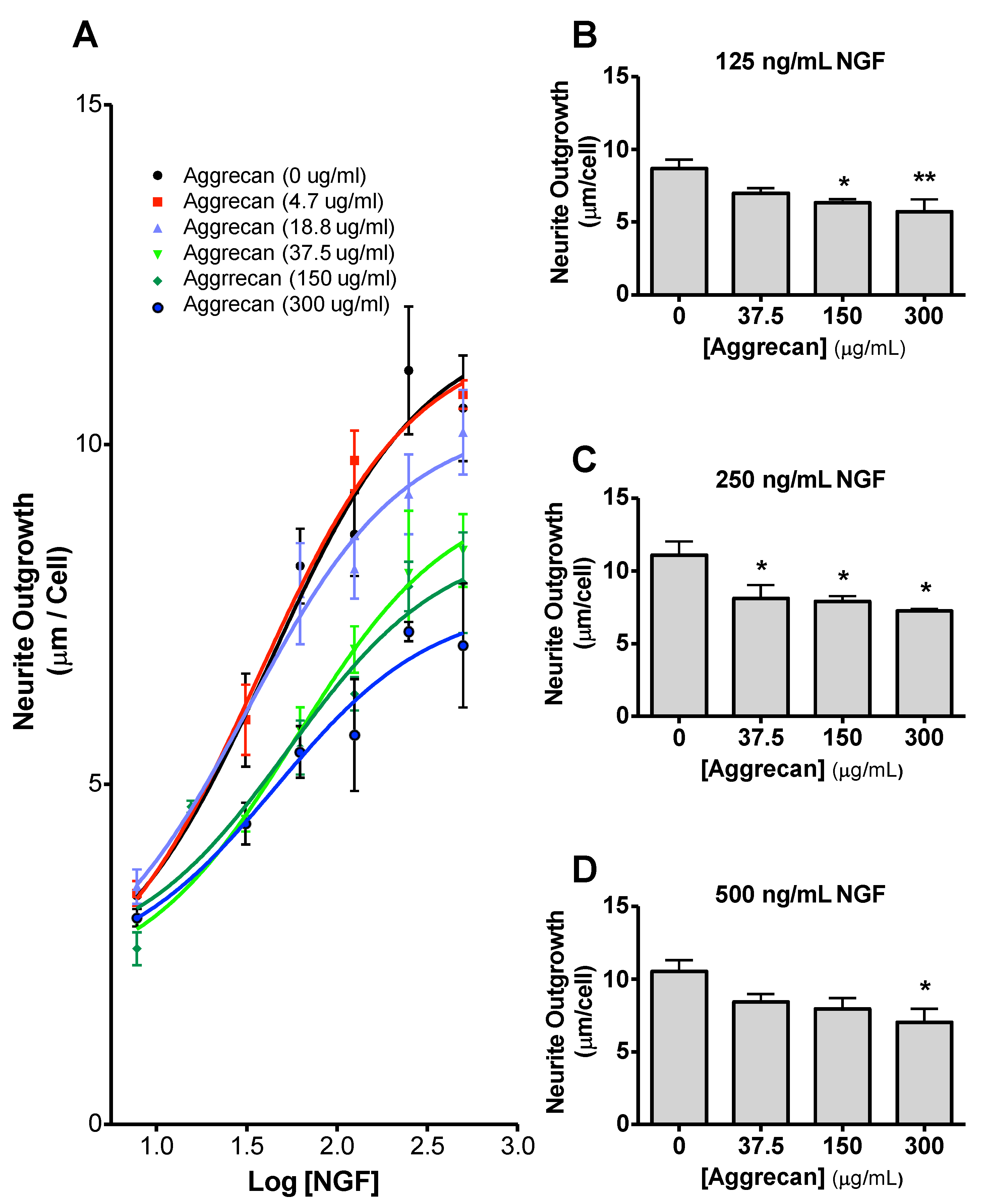
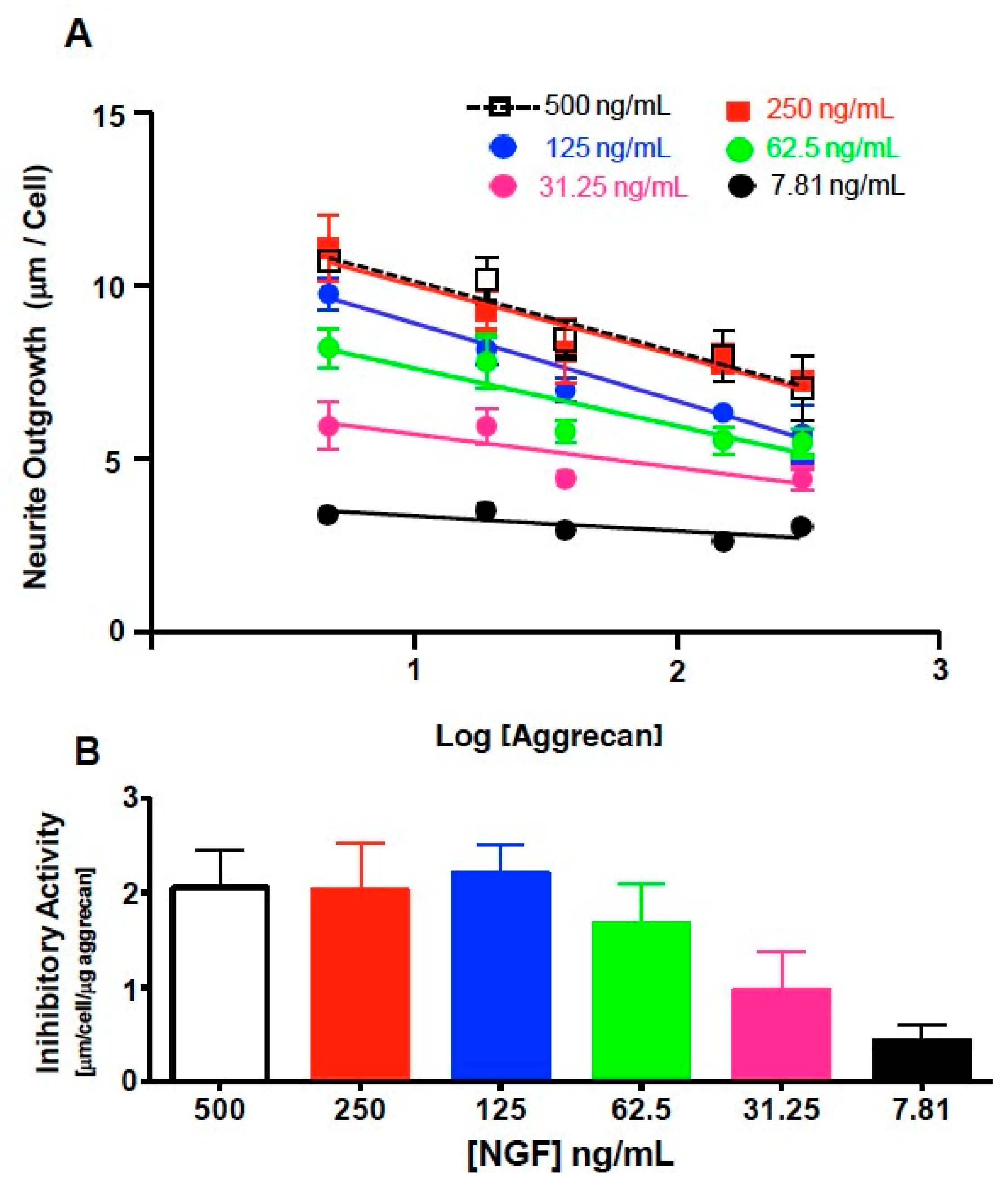
3.3. NS-1 Cell Neurite Outgrowth is Inhibited in a Concentration-Dependent Manner by Bound Aggrecan
3.4. Quantification of Inhibition of NGF-Stimulated Outgrowth Due to Aggrecan
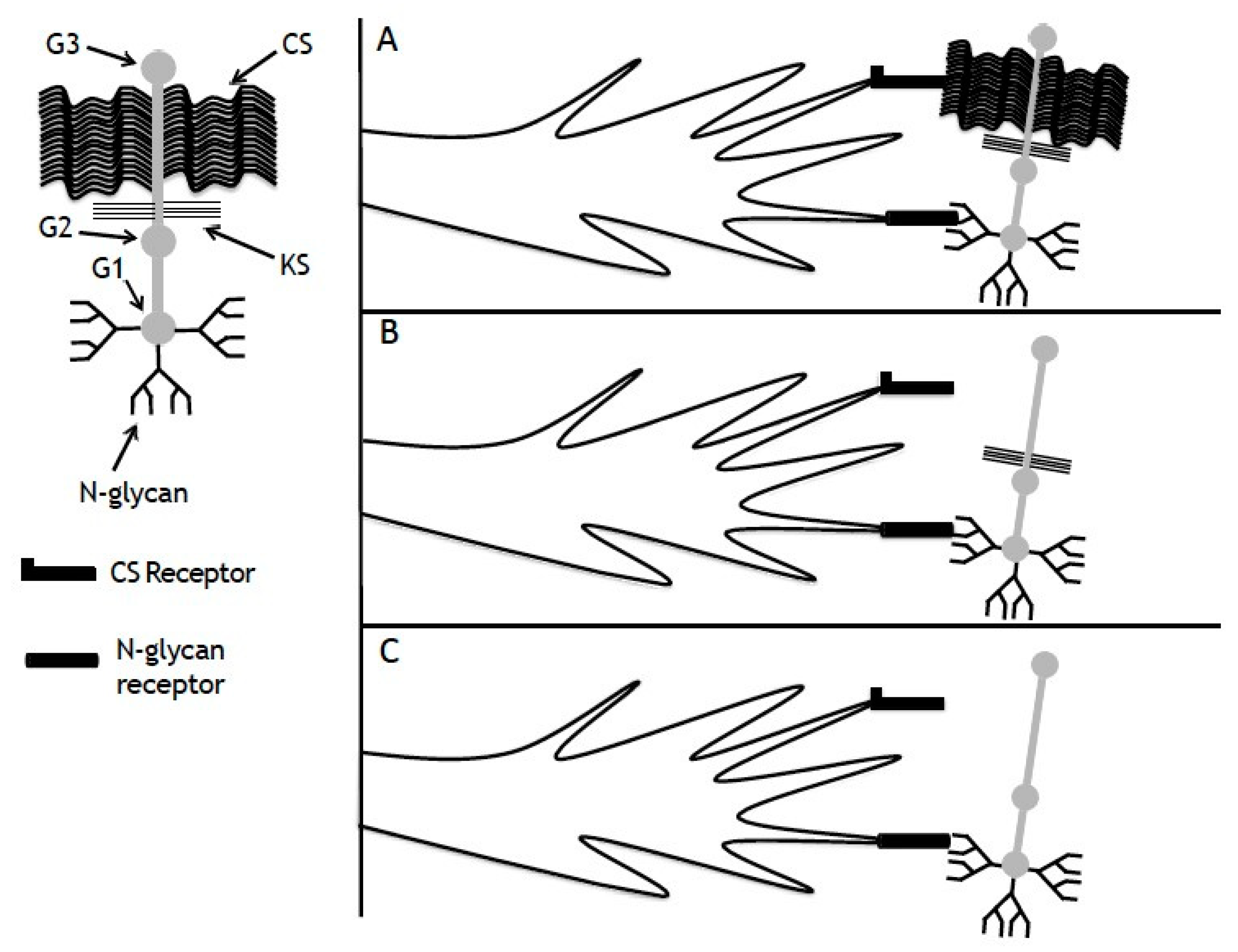
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kresse, H.; Schonherr, E. Proteoglycans of the extracellular matrix and growth control. J. Cell. Physiol. 2001, 189, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Beller, J.A.; Kulengowski, B.; Kobraei, E.M.; Curinga, G.; Calulot, C.M.; Bahrami, A.; Hering, T.M.; Snow, D.M. Comparison of sensory neuron growth cone and filopodial responses to structurally diverse aggrecan variants, In Vitro. Exp. Neurol. 2013, 247, 143–157. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Snow, D.M.; Lemmon, V.; Carrino, D.A.; Caplan, A.I.; Silver, J. Sulfated proteoglycans in astroglial barriers inhibit neurite outgrowth In Vitro. Exp. Neurol. 1990, 109, 111–130. [Google Scholar] [CrossRef]
- Snow, D.M.; Watanabe, M.; Letourneau, P.C.; Silver, J. A chondroitin sulfate proteoglycan may influence the direction of retinal ganglion cell outgrowth. Development 1991, 113, 1473–1485. [Google Scholar]
- Zuo, J.; Neubauer, D.; Dyess, K.; Ferguson, T.A.; Muir, D. Degradation of chondroitin sulfate proteoglycan enhances the neurite-promoting potential of spinal cord tissue. Exp. Neurol. 1998, 154, 654–662. [Google Scholar] [CrossRef]
- Massey, J.M.; Amps, J.; Viapiano, M.S.; Matthews, R.T.; Wagoner, M.R.; Whitaker, C.M.; Alilain, W.; Yonkof, A.L.; Khalyfa, A.; Cooper, N.G.; et al. Increased chondroitin sulfate proteoglycan expression in denervated brainstem targets following spinal cord injury creates a barrier to axonal regeneration overcome by chondroitinase ABC and neurotrophin-3. Exp. Neurol. 2008, 209, 426–445. [Google Scholar] [CrossRef]
- Emerling, D.E.; Lander, A.D. Inhibitors and promoters of thalamic neuron adhesion and outgrowth in embryonic neocortex: Functional association with chondroitin sulfate. Neuron 1996, 17, 1089–1100. [Google Scholar] [CrossRef][Green Version]
- Faissner, A.; Clement, A.; Lochter, A.; Streit, A.; Mandl, C.; Schachner, M. Isolation of a neural chondroitin sulfate proteoglycan with neurite outgrowth promoting properties. J. Cell. Biol. 1994, 126, 783–799. [Google Scholar] [CrossRef]
- Johnson, W.E.; Caterson, B.; Eisenstein, S.M.; Hynds, D.L.; Snow, D.M.; Roberts, S. Human intervertebral disc aggrecan inhibits nerve growth In Vitro. Arthritis Rheum. 2002, 46, 2658–2664. [Google Scholar] [CrossRef]
- Condic, M.L.; Snow, D.M.; Letourneau, P.C. Embryonic neurons adapt to the inhibitory proteoglycan aggrecan by increasing integrin expression. J. Neurosci. 1999, 19, 10036–10043. [Google Scholar] [CrossRef]
- Kwok, J.C.; Carulli, D.; Fawcett, J.W. In Vitro modeling of perineuronal nets: Hyaluronan synthase and link protein are necessary for their formation and integrity. J. Neurochem. 2010, 114, 1447–1459. [Google Scholar] [CrossRef] [PubMed]
- Kwok, J.C.; Dick, G.; Wang, D.; Fawcett, J.W. Extracellular matrix and perineuronal nets in CNS repair. Dev. Neurobiol. 2011, 71, 1073–1089. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Giger, R.J. A new role for RPTPsigma in spinal cord injury: Signaling chondroitin sulfate proteoglycan inhibition. Sci. Signal. 2010, 3, pe6. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Tenney, A.P.; Busch, S.A.; Horn, K.P.; Cuascut, F.X.; Liu, K.; He, Z.; Silver, J.; Flanagan, J.G. PTPsigma is a receptor for chondroitin sulfate proteoglycan, an inhibitor of neural regeneration. Science 2009, 326, 592–596. [Google Scholar] [CrossRef]
- Dickendesher, T.L.; Baldwin, K.T.; Mironova, Y.A.; Koriyama, Y.; Raiker, S.J.; Askew, K.L.; Wood, A.; Geoffroy, C.G.; Zheng, B.; Liepmann, C.D.; et al. NgR1 and NgR3 are receptors for chondroitin sulfate proteoglycans. Nat. Neurosci. 2012, 15, 703–712. [Google Scholar] [CrossRef]
- Brown, J.M.; Xia, J.; Zhuang, B.; Cho, K.S.; Rogers, C.J.; Gama, C.I.; Rawat, M.; Tully, S.E.; Uetani, N.; Mason, D.E.; et al. A sulfated carbohydrate epitope inhibits axon regeneration after injury. Proc. Natl. Acad. Sci. USA 2012, 109, 4768–4773. [Google Scholar] [CrossRef]
- Hering, T.M.; Kollar, J.; Huynh, T.D. Complete coding sequence of bovine aggrecan: Comparative structural analysis. Arch. Biochem. Biophys. 1997, 345, 259–270. [Google Scholar] [CrossRef]
- Monnier, P.P.; Sierra, A.; Schwab, J.M.; Henke-Fahle, S.; Mueller, B.K. The Rho/ROCK pathway mediates neurite growth-inhibitory activity associated with the chondroitin sulfate proteoglycans of the CNS glial scar. Mol. Cell. Neurosci. 2003, 22, 319–330. [Google Scholar] [CrossRef]
- Snow, D.M.; Mullins, N.; Hynds, D.L. Nervous system-derived chondroitin sulfate proteoglycans regulate growth cone morphology and inhibit neurite outgrowth: A light, epifluorescence, and electron microscopy study. Microsc. Res. Tech. 2001, 54, 273–286. [Google Scholar] [CrossRef]
- Lingor, P.; Teusch, N.; Schwarz, K.; Mueller, R.; Mack, H.; Bahr, M.; Mueller, B.K. Inhibition of Rho kinase (ROCK) increases neurite outgrowth on chondroitin sulphate proteoglycan In Vitro and axonal regeneration in the adult optic nerve in vivo. J. Neurochem. 2007, 103, 181–189. [Google Scholar] [CrossRef]
- Oohira, A.; Matsui, F.; Katoh-Semba, R. Inhibitory effects of brain chondroitin sulfate proteoglycans on neurite outgrowth from PC12D cells. J. Neurosci. 1991, 11, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Snow, D.M.; Brown, E.M.; Letourneau, P.C. Growth cone behavior in the presence of soluble chondroitin sulfate proteoglycan (CSPG), compared to behavior on CSPG bound to laminin or fibronectin. Int. J. Dev. Neurosci. 1996, 14, 331–349. [Google Scholar] [CrossRef]
- Greene, L.A.; Tischler, A.S. Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc. Natl. Acad. Sci. USA 1976, 73, 2424–2428. [Google Scholar] [CrossRef] [PubMed]
- Yick, L.W.; Cheung, P.T.; So, K.F.; Wu, W. Axonal regeneration of Clarke’s neurons beyond the spinal cord injury scar after treatment with chondroitinase ABC. Exp. Neurol. 2003, 182, 160–168. [Google Scholar] [CrossRef]
- Barritt, A.W.; Davies, M.; Marchand, F.; Hartley, R.; Grist, J.; Yip, P.; McMahon, S.B.; Bradbury, E.J. Chondroitinase ABC promotes sprouting of intact and injured spinal systems after spinal cord injury. J. Neurosci. 2006, 26, 10856–10867. [Google Scholar] [CrossRef] [PubMed]
- Akyuz, N.; Rost, S.; Mehanna, A.; Bian, S.; Loers, G.; Oezen, I.; Mishra, B.; Hoffmann, K.; Guseva, D.; Laczynska, E.; et al. Dermatan 4-O-sulfotransferase1 ablation accelerates peripheral nerve regeneration. Exp. Neurol. 2013, 247, 517–530. [Google Scholar] [CrossRef]
- Imagama, S.; Sakamoto, K.; Tauchi, R.; Shinjo, R.; Ohgomori, T.; Ito, Z.; Zhang, H.; Nishida, Y.; Asami, N.; Takeshita, S.; et al. Keratan sulfate restricts neural plasticity after spinal cord injury. J. Neurosci. 2011, 31, 17091–17102. [Google Scholar] [CrossRef]
- Ito, Z.; Sakamoto, K.; Imagama, S.; Matsuyama, Y.; Zhang, H.; Hirano, K.; Ando, K.; Yamashita, T.; Ishiguro, N.; Kadomatsu, K. N-acetylglucosamine 6-O-sulfotransferase-1-deficient mice show better functional recovery after spinal cord injury. J. Neurosci. 2010, 30, 5937–5947. [Google Scholar] [CrossRef]
- Beller, J.A.; Hering, T.M.; Snow, D.M. High-throughput quantitative assay for analyzing neurite outgrowth on a uniform substratum: The cell substratum assay. In Neuromethods: Extracellular Matrix; Leach, J.B., Powell, E.M., Eds.; Humana Press: New York, NY, USA, 2014. [Google Scholar]
- Gopalakrishnan, S.M.; Teusch, N.; Imhof, C.; Bakker, M.H.; Schurdak, M.; Burns, D.J.; Warrior, U. Role of Rho kinase pathway in chondroitin sulfate proteoglycan-mediated inhibition of neurite outgrowth in PC12 cells. J. Neurosci. Res. 2008, 86, 2214–2226. [Google Scholar] [CrossRef]
- Heinegard, D. Extraction, fractionation and characterization of proteoglycans from bovine tracheal cartilage. Biochim. Biophys. Acta 1972, 285, 181–192. [Google Scholar] [CrossRef]
- Rosenberg, L.; Choi, H.U.; Tang, L.H.; Pal, S.; Johnson, T.; Lyons, D.A.; Laue, T.M. Proteoglycans of bovine articular cartilage. The effects of divalent cations on the biochemical properties of link protein. J. Biol. Chem. 1991, 266, 7016–7024. [Google Scholar] [PubMed]
- Hering, T.M.; Beller, J.A.; Calulot, C.M.; Centers, A.; Snow, D.M. Proteoglycans of reactive rat cortical astrocyte cultures: Abundance of N-unsubstituted glucosamine-enriched heparan sulfate. Matrix Biol. 2015, 41, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Pool, M.; Thiemann, J.; Bar-Or, A.; Fournier, A.E. NeuriteTracer: A novel ImageJ plugin for automated quantification of neurite outgrowth. J. Neurosci. Methods 2008, 168, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Snow, D.M.; Letourneau, P.C. Neurite outgrowth on a step gradient of chondroitin sulfate proteoglycan (CS-PG). J. Neurobiol. 1992, 23, 322–336. [Google Scholar] [CrossRef]
- Greene, L.A. Nerve growth factor prevents the death and stimulates the neuronal differentiation of clonal PC12 pheochromocytoma cells in serum-free medium. J. Cell. Biol. 1978, 78, 747–755. [Google Scholar] [CrossRef]
- Bradbury, E.J.; Moon, L.D.; Popat, R.J.; King, V.R.; Bennett, G.S.; Patel, P.N.; Fawcett, J.W.; McMahon, S.B. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 2002, 416, 636–640. [Google Scholar] [CrossRef]
- Lee, H.; McKeon, R.J.; Bellamkonda, R.V. Sustained delivery of thermostabilized chABC enhances axonal sprouting and functional recovery after spinal cord injury. Proc. Natl. Acad. Sci. USA 2010, 107, 3340–3345. [Google Scholar] [CrossRef]
- Massey, J.M.; Hubscher, C.H.; Wagoner, M.R.; Decker, J.A.; Amps, J.; Silver, J.; Onifer, S.M. Chondroitinase ABC digestion of the perineuronal net promotes functional collateral sprouting in the cuneate nucleus after cervical spinal cord injury. J. Neurosci. 2006, 26, 4406–4414. [Google Scholar] [CrossRef]
- Huang, W.C.; Kuo, W.C.; Cherng, J.H.; Hsu, S.H.; Chen, P.R.; Huang, S.H.; Huang, M.C.; Liu, J.C.; Cheng, H. Chondroitinase ABC promotes axonal re-growth and behavior recovery in spinal cord injury. Biochem. Biophys. Res. Commun. 2006, 349, 963–968. [Google Scholar] [CrossRef]
- Nakamae, T.; Tanaka, N.; Nakanishi, K.; Kamei, N.; Sasaki, H.; Hamasaki, T.; Yamada, K.; Yamamoto, R.; Mochizuki, Y.; Ochi, M. Chondroitinase ABC promotes corticospinal axon growth in organotypic cocultures. Spinal Cord 2009, 47, 161–165. [Google Scholar] [CrossRef][Green Version]
- Garwood, J.; Schnadelbach, O.; Clement, A.; Schutte, K.; Bach, A.; Faissner, A. DSD-1-proteoglycan is the mouse homolog of phosphacan and displays opposing effects on neurite outgrowth dependent on neuronal lineage. J. Neurosci. 1999, 19, 3888–3899. [Google Scholar] [CrossRef] [PubMed]
- Fidler, P.S.; Schuette, K.; Asher, R.A.; Dobbertin, A.; Thornton, S.R.; Calle-Patino, Y.; Muir, E.; Levine, J.M.; Geller, H.M.; Rogers, J.H.; et al. Comparing astrocytic cell lines that are inhibitory or permissive for axon growth: The major axon-inhibitory proteoglycan is NG2. J. Neurosci. 1999, 19, 8778–8788. [Google Scholar] [CrossRef] [PubMed]
- Cole, G.J.; McCabe, C.F. Identification of a developmentally regulated keratan sulfate proteoglycan that inhibits cell adhesion and neurite outgrowth. Neuron 1991, 7, 1007–1018. [Google Scholar] [CrossRef]
- Mace, K.; Saxod, R.; Feuerstein, C.; Sadoul, R.; Hemming, F.J. Chondroitin and keratan sulfates have opposing effects on attachment and outgrowth of ventral mesencephalic explants in culture. J. Neurosci. Res 2002, 70, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Charvet, I.; Hemming, F.J.; Feuerstein, C.; Saxod, R. Mosaic distribution of chondroitin and keratan sulphate in the developing rat striatum: Possible involvement of proteoglycans in the organization of the nigrostriatal system. Brain Res. Dev. Brain. Res. 1998, 109, 229–244. [Google Scholar] [CrossRef]
- Helenius, A.; Aebi, M. Intracellular functions of N-linked glycans. Science 2001, 291, 2364–2369. [Google Scholar] [CrossRef] [PubMed]
- Dennis, J.W. N-linked oligosaccharide processing and tumor cell biology. Semin. Cancer Biol. 1991, 2, 411–420. [Google Scholar]
- Varki, A. Biological roles of oligosaccharides: All of the theories are correct. Glycobiology 1993, 3, 97–130. [Google Scholar] [CrossRef]
- Takahashi, M.; Tsuda, T.; Ikeda, Y.; Honke, K.; Taniguchi, N. Role of N-glycans in growth factor signaling. Glycoconj. J. 2004, 20, 207–212. [Google Scholar] [CrossRef]
- Parkkinen, J.; Raulo, E.; Merenmies, J.; Nolo, R.; Kajander, E.O.; Baumann, M.; Rauvala, H. Amphoterin, the 30-kDa protein in a family of HMG1-type polypeptides. Enhanced expression in transformed cells, leading edge localization, and interactions with plasminogen activation. J. Biol. Chem. 1993, 268, 19726–19738. [Google Scholar]
- Srikrishna, G.; Huttunen, H.J.; Johansson, L.; Weigle, B.; Yamaguchi, Y.; Rauvala, H.; Freeze, H.H. N -Glycans on the receptor for advanced glycation end products influence amphoterin binding and neurite outgrowth. J. Neurochem. 2002, 80, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Guo, H.B.; Kamar, M.; Abbott, K.; Troupe, K.; Lee, J.K.; Alvarez-Manilla, G.; Pierce, M. N-acetylglucosaminyltranferase VB expression enhances beta1 integrin- dependent PC12 neurite outgrowth on laminin and collagen. J. Neurochem. 2006, 97, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Horstkorte, R.; Schachner, M.; Magyar, J.P.; Vorherr, T.; Schmitz, B. The fourth immunoglobulin-like domain of NCAM contains a carbohydrate recognition domain for oligomannosidic glycans implicated in association with L1 and neurite outgrowth. J. Cell. Biol. 1993, 121, 1409–1421. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.R.; Fawcett, J.W. Combination treatment with chondroitinase ABC in spinal cord injury--breaking the barrier. Neurosci. Bull. 2013, 29, 477–483. [Google Scholar] [CrossRef]
- Hilton, B.J.; Lang, B.T.; Cregg, J.M. Keratan sulfate proteoglycans in plasticity and recovery after spinal cord injury. J. Neurosci. 2012, 32, 4331–4333. [Google Scholar] [CrossRef]
- Jones, L.L.; Tuszynski, M.H. Spinal cord injury elicits expression of keratan sulfate proteoglycans by macrophages, reactive microglia, and oligodendrocyte progenitors. J. Neurosci. 2002, 22, 4611–4624. [Google Scholar] [CrossRef]
- McAdams, B.D.; McLoon, S.C. Expression of chondroitin sulfate and keratan sulfate proteoglycans in the path of growing retinal axons in the developing chick. J. Comp. Neurol 1995, 352, 594–606. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hering, T.M.; Beller, J.A.; Calulot, C.M.; Snow, D.M. Contributions of Chondroitin Sulfate, Keratan Sulfate and N-linked Oligosaccharides to Inhibition of Neurite Outgrowth by Aggrecan. Biology 2020, 9, 29. https://doi.org/10.3390/biology9020029
Hering TM, Beller JA, Calulot CM, Snow DM. Contributions of Chondroitin Sulfate, Keratan Sulfate and N-linked Oligosaccharides to Inhibition of Neurite Outgrowth by Aggrecan. Biology. 2020; 9(2):29. https://doi.org/10.3390/biology9020029
Chicago/Turabian StyleHering, Thomas M., Justin A. Beller, Christopher M. Calulot, and Diane M. Snow. 2020. "Contributions of Chondroitin Sulfate, Keratan Sulfate and N-linked Oligosaccharides to Inhibition of Neurite Outgrowth by Aggrecan" Biology 9, no. 2: 29. https://doi.org/10.3390/biology9020029
APA StyleHering, T. M., Beller, J. A., Calulot, C. M., & Snow, D. M. (2020). Contributions of Chondroitin Sulfate, Keratan Sulfate and N-linked Oligosaccharides to Inhibition of Neurite Outgrowth by Aggrecan. Biology, 9(2), 29. https://doi.org/10.3390/biology9020029




