Guide snoRNAs: Drivers or Passengers in Human Disease?
Abstract
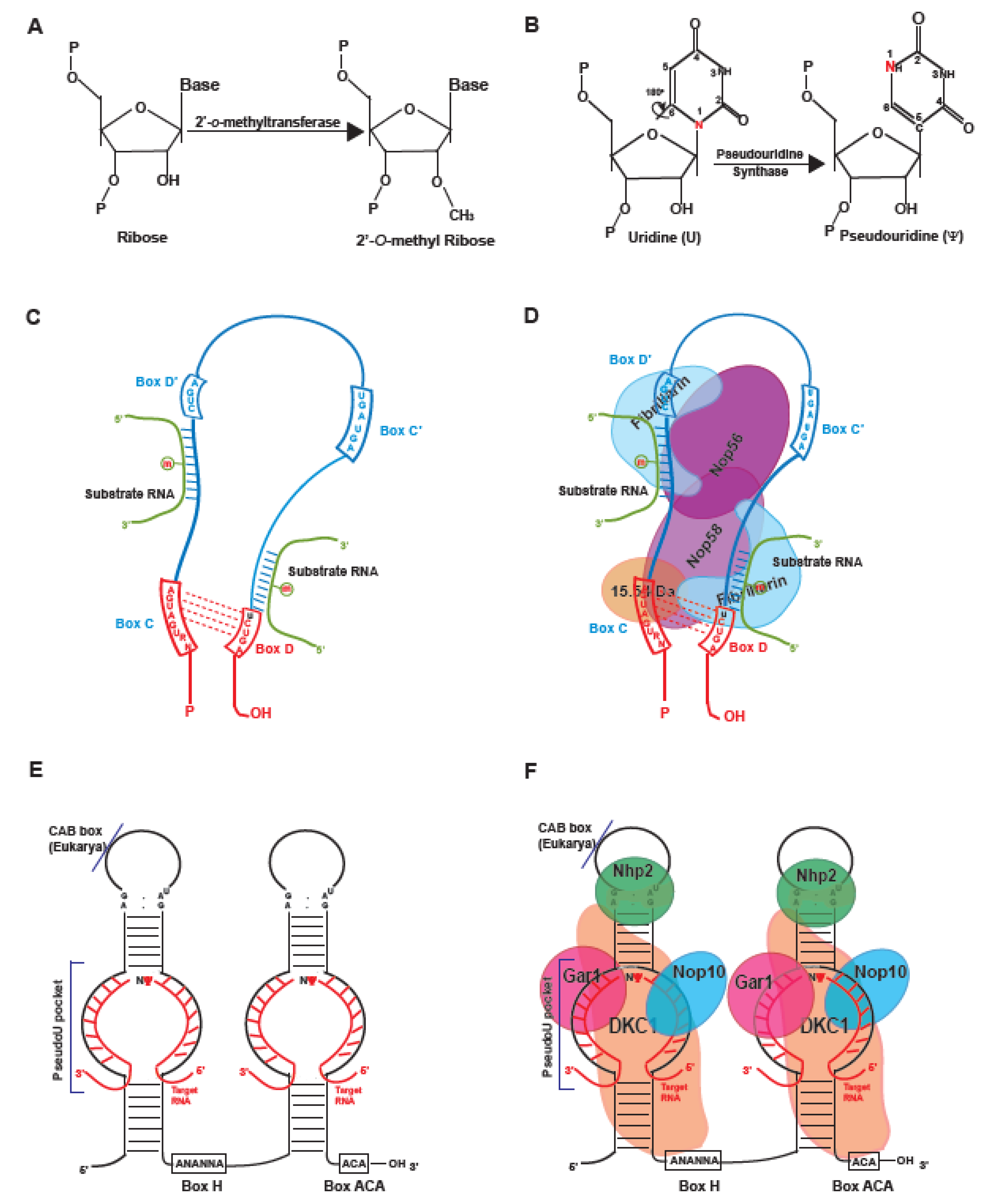
1. Introduction
2. Box H/ACA Guide RNAs in Human Disease
2.1. Box H/ACA Guide RNAs in Cancer
2.2. Box H/ACA Guide RNAs in Genetic Disease
2.3. Box H/ACA Guide RNA Host Gene Deregulation in Disease
3. Box C/D Guide RNAs in Human Disease
3.1. Box C/D Guide RNAs in Cancer
3.2. Box C/D Guide RNAs in Genetic Disease
3.3. Box C/D Guide RNAs in Other Disease
3.4. Box C/D Guide RNA Host Gene Deregulation in Disease
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blaby, I.K.; Majumder, M.; Chatterjee, K.; Jana, S.; Grosjean, H.; de Crecy-Lagard, V.; Gupta, R. Pseudouridine formation in archaeal RNAs: The case of Haloferax volcanii. RNA 2011, 17, 1367–1380. [Google Scholar] [CrossRef] [PubMed]
- Bortolin, M.L.; Kiss, T. Human U19 intron-encoded snoRNA is processed from a long primary transcript that possesses little potential for protein coding. RNA 1998, 4, 445–454. [Google Scholar] [PubMed]
- Decatur, W.A.; Fournier, M.J. rRNA modifications and ribosome function. Trends Biochem. Sci. 2002, 27, 344–351. [Google Scholar] [CrossRef]
- Limbach, P.A.; Crain, P.F.; McCloskey, J.A. Summary: The modified nucleosides of RNA. Nucleic Acids Res. 1994, 22, 2183–2196. [Google Scholar] [CrossRef] [PubMed]
- Ofengand, J. Ribosomal RNA pseudouridines and pseudouridine synthases. FEBS Lett. 2002, 514, 17–25. [Google Scholar] [CrossRef]
- Tollervey, D.; Lehtonen, H.; Jansen, R.; Kern, H.; Hurt, E.C. Temperature-sensitive mutations demonstrate roles for yeast fibrillarin in pre-rRNA processing, pre-rRNA methylation, and ribosome assembly. Cell 1993, 72, 443–457. [Google Scholar] [CrossRef]
- Cantara, W.A.; Crain, P.F.; Rozenski, J.; McCloskey, J.A.; Harris, K.A.; Zhang, X.; Vendeix, F.A.; Fabris, D.; Agris, P.F. The RNA Modification Database, RNAMDB: 2011 update. Nucleic Acids Res. 2011, 39, D195–D201. [Google Scholar] [CrossRef]
- Bachellerie, J.P.; Cavaille, J.; Huttenhofer, A. The expanding snoRNA world. Biochimie 2002, 84, 775–790. [Google Scholar] [CrossRef]
- Kiss, T. Small nucleolar RNA-guided post-transcriptional modification of cellular RNAs. EMBO J. 2001, 20, 3617–3622. [Google Scholar] [CrossRef]
- Rashid, R.; Aittaleb, M.; Chen, Q.; Spiegel, K.; Demeler, B.; Li, H. Functional requirement for symmetric assembly of archaeal box C/D small ribonucleoprotein particles. J. Mol. Biol. 2003, 333, 295–306. [Google Scholar] [CrossRef]
- Hamma, T.; Ferre-D’Amare, A.R. The box H/ACA ribonucleoprotein complex: Interplay of RNA and protein structures in post-transcriptional RNA modification. J. Biol. Chem. 2010, 285, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Terns, M.; Terns, R. Noncoding RNAs of the H/ACA family. Cold Spring Harb. Symp. Quant. Biol. 2006, 71, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Filipowicz, W.; Pogacic, V. Biogenesis of small nucleolar ribonucleoproteins. Curr. Opin. Cell Biol. 2002, 14, 319–327. [Google Scholar] [CrossRef]
- Herter, E.K.; Stauch, M.; Gallant, M.; Wolf, E.; Raabe, T.; Gallant, P. snoRNAs are a novel class of biologically relevant Myc targets. BMC Biol. 2015, 13, 25. [Google Scholar] [CrossRef] [PubMed]
- Jorjani, H.; Kehr, S.; Jedlinski, D.J.; Gumienny, R.; Hertel, J.; Stadler, P.F.; Zavolan, M.; Gruber, A.R. An updated human snoRNAome. Nucleic Acids Res. 2016, 44, 5068–5082. [Google Scholar] [CrossRef] [PubMed]
- Lestrade, L.; Weber, M.J. snoRNA-LBME-db, a comprehensive database of human H/ACA and C/D box snoRNAs. Nucleic Acids Res. 2006, 34, D158–D162. [Google Scholar] [CrossRef]
- Dieci, G.; Preti, M.; Montanini, B. Eukaryotic snoRNAs: A paradigm for gene expression flexibility. Genomics 2009, 94, 83–88. [Google Scholar] [CrossRef]
- Tollervey, D.; Kiss, T. Function and synthesis of small nucleolar RNAs. Curr. Opin. Cell. Biol. 1997, 9, 337–342. [Google Scholar] [CrossRef]
- Smith, C.M.; Steitz, J.A. Classification of gas5 as a multi-small-nucleolar-RNA (snoRNA) host gene and a member of the 5′-terminal oligopyrimidine gene family reveals common features of snoRNA host genes. Mol. Cell. Biol. 1998, 18, 6897–6909. [Google Scholar] [CrossRef]
- Askarian-Amiri, M.E.; Crawford, J.; French, J.D.; Smart, C.E.; Smith, M.A.; Clark, M.B.; Ru, K.; Mercer, T.R.; Thompson, E.R.; Lakhani, S.R.; et al. SNORD-host RNA Zfas1 is a regulator of mammary development and a potential marker for breast cancer. RNA 2011, 17, 878–891. [Google Scholar] [CrossRef]
- Maxwell, E.S.; Fournier, M.J. The small nucleolar RNAs. Annu. Rev. Biochem. 1995, 64, 897–934. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, R.; Suzuki, Y.; Takeuchi, N.; Wakaguri, H.; Ueda, T.; Sugano, S.; Nakai, K. Comprehensive detection of human terminal oligo-pyrimidine (TOP) genes and analysis of their characteristics. Nucleic Acids Res. 2008, 36, 3707–3715. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.Y.; Guo, P.; Boyd, J.; Sun, X.; Li, Q.; Zhou, W.; Dong, J.T. Implication of snoRNA U50 in human breast cancer. J. Genet. Genom. 2009, 36, 447–454. [Google Scholar] [CrossRef]
- Dong, X.Y.; Rodriguez, C.; Guo, P.; Sun, X.; Talbot, J.T.; Zhou, W.; Petros, J.; Li, Q.; Vessella, R.L.; Kibel, A.S.; et al. SnoRNA U50 is a candidate tumor suppressor gene at 6q14.3 with a mutation associated with clinically significant prostate cancer. Hum. Mol. Genet. 2008, 17, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- Gee, H.E.; Buffa, F.M.; Camps, C.; Ramachandran, A.; Leek, R.; Taylor, M.; Patil, M.; Sheldon, H.; Betts, G.; Homer, J.; et al. The small-nucleolar RNAs commonly used for microRNA normalisation correlate with tumour pathology and prognosis. Br. J. Cancer 2011, 104, 1168–1177. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Yu, L.; Mei, Y.; Guarnera, M.; Shen, J.; Li, R.; Liu, Z.; Jiang, F. Small nucleolar RNA signatures as biomarkers for non-small-cell lung cancer. Mol. Cancer 2010, 9, 198. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.T.; Farzaneh, F. Are snoRNAs and snoRNA host genes new players in cancer? Nat. Rev. Cancer 2012, 12, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Esteller, M. Non-coding RNAs in human disease. Nat. Rev. Genet. 2011, 12, 861–874. [Google Scholar] [CrossRef] [PubMed]
- McMahon, M.; Contreras, A.; Ruggero, D. Small RNAs with big implications: New insights into H/ACA snoRNA function and their role in human disease. Wiley Interdiscip. Rev. RNA 2015, 6, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Cohn, W.E. 5-Ribosyl uracil, a carbon-carbon ribofuranosyl nucleoside in ribonucleic acids. Biochim. Biophys. Acta 1959, 32, 569–571. [Google Scholar] [CrossRef]
- Ganot, P.; Bortolin, M.L.; Kiss, T. Site-specific pseudouridine formation in preribosomal RNA is guided by small nucleolar RNAs. Cell 1997, 89, 799–809. [Google Scholar] [CrossRef]
- Ni, J.; Tien, A.L.; Fournier, M.J. Small nucleolar RNAs direct site-specific synthesis of pseudouridine in ribosomal RNA. Cell 1997, 89, 565–573. [Google Scholar] [CrossRef]
- Kiss, A.M.; Jady, B.E.; Bertrand, E.; Kiss, T. Human box H/ACA pseudouridylation guide RNA machinery. Mol. Cell. Biol. 2004, 24, 5797–5807. [Google Scholar] [CrossRef]
- Baxter-Roshek, J.L.; Petrov, A.N.; Dinman, J.D. Optimization of ribosome structure and function by rRNA base modification. PLoS ONE 2007, 2, e174. [Google Scholar] [CrossRef] [PubMed]
- Matera, A.G.; Terns, R.M.; Terns, M.P. Non-coding RNAs: Lessons from the small nuclear and small nucleolar RNAs. Nat. Rev. Mol. Cell Biol. 2007, 8, 209–220. [Google Scholar] [CrossRef]
- Reichow, S.L.; Hamma, T.; Ferre-D’Amare, A.R.; Varani, G. The structure and function of small nucleolar ribonucleoproteins. Nucleic Acids Res. 2007, 35, 1452–1464. [Google Scholar] [CrossRef]
- Jady, B.E.; Ketele, A.; Kiss, T. Human intron-encoded Alu RNAs are processed and packaged into Wdr79-associated nucleoplasmic box H/ACA RNPs. Genes Dev. 2012, 26, 1897–1910. [Google Scholar] [CrossRef]
- Ofengand, J.; Bakin, A. Mapping to nucleotide resolution of pseudouridine residues in large subunit ribosomal RNAs from representative eukaryotes, prokaryotes, archaebacteria, mitochondria and chloroplasts. J. Mol. Biol. 1997, 266, 246–268. [Google Scholar] [CrossRef]
- Kierzek, E.; Malgowska, M.; Lisowiec, J.; Turner, D.H.; Gdaniec, Z.; Kierzek, R. The contribution of pseudouridine to stabilities and structure of RNAs. Nucleic Acids Res. 2014, 42, 3492–3501. [Google Scholar] [CrossRef]
- Penzo, M.; Montanaro, L. Turning Uridines around: Role of rRNA Pseudouridylation in Ribosome Biogenesis and Ribosomal Function. Biomolecules 2018, 8, 38. [Google Scholar] [CrossRef]
- Kehr, S.; Bartschat, S.; Tafer, H.; Stadler, P.F.; Hertel, J. Matching of Soulmates: Coevolution of snoRNAs and their targets. Mol. Biol. Evol. 2014, 31, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Krogh, N.; Jansson, M.D.; Hafner, S.J.; Tehler, D.; Birkedal, U.; Christensen-Dalsgaard, M.; Lund, A.H.; Nielsen, H. Profiling of 2′-O-Me in human rRNA reveals a subset of fractionally modified positions and provides evidence for ribosome heterogeneity. Nucleic Acids Res. 2016, 44, 7884–7895. [Google Scholar] [CrossRef] [PubMed]
- Taoka, M.; Nobe, Y.; Yamaki, Y.; Sato, K.; Ishikawa, H.; Izumikawa, K.; Yamauchi, Y.; Hirota, K.; Nakayama, H.; Takahashi, N.; et al. Landscape of the complete RNA chemical modifications in the human 80S ribosome. Nucleic Acids Res. 2018, 46, 9289–9298. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, G.A.; Filippova, J.A.; Komissarov, A.B.; Kuligina, E.V.; Richter, V.A.; Semenov, D.V. Regulatory role of small nucleolar RNAs in human diseases. BioMed Res. Int. 2015, 2015, 206849. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.S.; Lin, S.Y.; Lieu, A.S.; Wu, T.L. Differential expression of human 5S snoRNA genes. Biochem. Biophys. Res. Commun. 2002, 299, 196–200. [Google Scholar] [CrossRef]
- Mei, Y.P.; Liao, J.P.; Shen, J.; Yu, L.; Liu, B.L.; Liu, L.; Li, R.Y.; Ji, L.; Dorsey, S.G.; Jiang, Z.R.; et al. Small nucleolar RNA 42 acts as an oncogene in lung tumorigenesis. Oncogene 2012, 31, 2794–2804. [Google Scholar] [CrossRef]
- Okugawa, Y.; Toiyama, Y.; Toden, S.; Mitoma, H.; Nagasaka, T.; Tanaka, K.; Inoue, Y.; Kusunoki, M.; Boland, C.R.; Goel, A. Clinical significance of SNORA42 as an oncogene and a prognostic biomarker in colorectal cancer. Gut 2017, 66, 107–117. [Google Scholar] [CrossRef]
- Yoshida, K.; Toden, S.; Weng, W.; Shigeyasu, K.; Miyoshi, J.; Turner, J.; Nagasaka, T.; Ma, Y.; Takayama, T.; Fujiwara, T.; et al. SNORA21—An Oncogenic Small Nucleolar RNA, with a Prognostic Biomarker Potential in Human Colorectal Cancer. eBioMedicine 2017, 22, 68–77. [Google Scholar] [CrossRef]
- Li, G.; He, Y.; Liu, X.; Zheng, Z.; Zhang, M.; Qin, F.; Lan, X. Small nucleolar RNA 47 promotes tumorigenesis by regulating EMT markers in hepatocellular carcinoma. Minerva Med. 2017, 108, 396–404. [Google Scholar] [CrossRef]
- Wu, L.; Zheng, J.; Chen, P.; Liu, Q.; Yuan, Y. Small nucleolar RNA ACA11 promotes proliferation, migration and invasion in hepatocellular carcinoma by targeting the PI3K/AKT signaling pathway. Biomed. Pharmacother. 2017, 90, 705–712. [Google Scholar] [CrossRef]
- Qin, Y.; Meng, L.; Fu, Y.; Quan, Z.; Ma, M.; Weng, M.; Zhang, Z.; Gao, C.; Shi, X.; Han, K. SNORA74B gene silencing inhibits gallbladder cancer cells by inducing PHLPP and suppressing Akt/mTOR signaling. Oncotarget 2017, 8, 19980–19996. [Google Scholar] [CrossRef] [PubMed]
- Bellodi, C.; McMahon, M.; Contreras, A.; Juliano, D.; Kopmar, N.; Nakamura, T.; Maltby, D.; Burlingame, A.; Savage, S.A.; Shimamura, A.; et al. H/ACA small RNA dysfunctions in disease reveal key roles for noncoding RNA modifications in hematopoietic stem cell differentiation. Cell Rep. 2013, 3, 1493–1502. [Google Scholar] [CrossRef] [PubMed]
- Ronchetti, D.; Mosca, L.; Cutrona, G.; Tuana, G.; Gentile, M.; Fabris, S.; Agnelli, L.; Ciceri, G.; Matis, S.; Massucco, C.; et al. Small nucleolar RNAs as new biomarkers in chronic lymphocytic leukemia. BMC Med. Genom. 2013, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Ronchetti, D.; Todoerti, K.; Tuana, G.; Agnelli, L.; Mosca, L.; Lionetti, M.; Fabris, S.; Colapietro, P.; Miozzo, M.; Ferrarini, M.; et al. The expression pattern of small nucleolar and small Cajal body-specific RNAs characterizes distinct molecular subtypes of multiple myeloma. Blood Cancer J. 2012, 2, e96. [Google Scholar] [CrossRef] [PubMed]
- Valleron, W.; Ysebaert, L.; Berquet, L.; Fataccioli, V.; Quelen, C.; Martin, A.; Parrens, M.; Lamant, L.; de Leval, L.; Gisselbrecht, C.; et al. Small nucleolar RNA expression profiling identifies potential prognostic markers in peripheral T-cell lymphoma. Blood 2012, 120, 3997–4005. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.S.; Stojanov, P.; Mermel, C.H.; Robinson, J.T.; Garraway, L.A.; Golub, T.R.; Meyerson, M.; Gabriel, S.B.; Lander, E.S.; Getz, G. Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 2014, 505, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Liang, F.; Qu, H.; Lin, Q.; Yang, Y.; Ruan, X.; Zhang, B.; Liu, Y.; Yu, C.; Zhang, H.; Fang, X.; et al. Molecular biomarkers screened by next-generation RNA sequencing for non-sentinel lymph node status prediction in breast cancer patients with metastatic sentinel lymph nodes. World J. Surg. Oncol. 2015, 13, 258. [Google Scholar] [CrossRef] [PubMed]
- Patil, P.; Kibiryeva, N.; Uechi, T.; Marshall, J.; O’Brien, J.E., Jr.; Artman, M.; Kenmochi, N.; Bittel, D.C. scaRNAs regulate splicing and vertebrate heart development. Biochim. Biophys. Acta 2015, 1852, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Parry, E.M.; Alder, J.K.; Lee, S.S.; Phillips, J.A., 3rd; Loyd, J.E.; Duggal, P.; Armanios, M. Decreased dyskerin levels as a mechanism of telomere shortening in X-linked dyskeratosis congenita. J. Med. Genet. 2011, 48, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Piekna-Przybylska, D.; Decatur, W.A.; Fournier, M.J. The 3D rRNA modification maps database: With interactive tools for ribosome analysis. Nucleic Acids Res. 2008, 36, D178–D183. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, C.; Ogren, A.; Kibiryeva, N.; Marshall, J.; O’Brien, J.E.; Kenmochi, N.; Bittel, D.C. The Role of scaRNAs in Adjusting Alternative mRNA Splicing in Heart Development. J. Cardiovasc. Dev. Dis. 2018, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- DiGiacomo, V.; Meruelo, D. Looking into laminin receptor: Critical discussion regarding the non-integrin 37/67-kDa laminin receptor/RPSA protein. Biol. Rev. Camb. Philos. Soc. 2016, 91, 288–310. [Google Scholar] [CrossRef] [PubMed]
- Boria, I.; Garelli, E.; Gazda, H.T.; Aspesi, A.; Quarello, P.; Pavesi, E.; Ferrante, D.; Meerpohl, J.J.; Kartal, M.; Da Costa, L.; et al. The ribosomal basis of Diamond-Blackfan Anemia: Mutation and database update. Hum. Mutat. 2010, 31, 1269–1279. [Google Scholar] [CrossRef] [PubMed]
- Garcon, L.; Ge, J.; Manjunath, S.H.; Mills, J.A.; Apicella, M.; Parikh, S.; Sullivan, L.M.; Podsakoff, G.M.; Gadue, P.; French, D.L.; et al. Ribosomal and hematopoietic defects in induced pluripotent stem cells derived from Diamond Blackfan anemia patients. Blood 2013, 122, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Trahan, C.; Dragon, F. Dyskeratosis congenita mutations in the H/ACA domain of human telomerase RNA affect its assembly into a pre-RNP. RNA 2009, 15, 235–243. [Google Scholar] [CrossRef]
- Schulten, H.J.; Bangash, M.; Karim, S.; Dallol, A.; Hussein, D.; Merdad, A.; Al-Thoubaity, F.K.; Al-Maghrabi, J.; Jamal, A.; Al-Ghamdi, F.; et al. Comprehensive molecular biomarker identification in breast cancer brain metastases. J. Transl. Med. 2017, 15, 269. [Google Scholar] [CrossRef]
- Cui, L.; Nakano, K.; Obchoei, S.; Setoguchi, K.; Matsumoto, M.; Yamamoto, T.; Obika, S.; Shimada, K.; Hiraoka, N. Small Nucleolar Noncoding RNA SNORA23, Up-Regulated in Human Pancreatic Ductal Adenocarcinoma, Regulates Expression of Spectrin Repeat-Containing Nuclear Envelope 2 to Promote Growth and Metastasis of Xenograft Tumors in Mice. Gastroenterology 2017, 153, 292–306. [Google Scholar] [CrossRef]
- Zhou, C.; Zang, D.; Jin, Y.; Wu, H.; Liu, Z.; Du, J.; Zhang, J. Mutation in ribosomal protein L21 underlies hereditary hypotrichosis simplex. Hum. Mutat. 2011, 32, 710–714. [Google Scholar] [CrossRef]
- Steinbusch, M.M.; Fang, Y.; Milner, P.I.; Clegg, P.D.; Young, D.A.; Welting, T.J.; Peffers, M.J. Serum snoRNAs as biomarkers for joint ageing and post traumatic osteoarthritis. Sci. Rep. 2017, 7, 43558. [Google Scholar] [CrossRef]
- Bosco, A.; Ehteshami, S.; Panyala, S.; Martinez, F.D. Interferon regulatory factor 7 is a major hub connecting interferon-mediated responses in virus-induced asthma exacerbations in vivo. J. Allergy Clin. Immunol. 2012, 129, 88–94. [Google Scholar] [CrossRef]
- Irizar, H.; Munoz-Culla, M.; Saenz-Cuesta, M.; Osorio-Querejeta, I.; Sepulveda, L.; Castillo-Trivino, T.; Prada, A.; Lopez de Munain, A.; Olascoaga, J.; Otaegui, D. Identification of ncRNAs as potential therapeutic targets in multiple sclerosis through differential ncRNA—mRNA network analysis. BMC Genom. 2015, 16, 250. [Google Scholar] [CrossRef] [PubMed]
- Mannoor, K.; Shen, J.; Liao, J.; Liu, Z.; Jiang, F. Small nucleolar RNA signatures of lung tumor-initiating cells. Mol. Cancer 2014, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- Lan, T.; Ma, W.; Hong, Z.; Wu, L.; Chen, X.; Yuan, Y. Long non-coding RNA small nucleolar RNA host gene 12 (SNHG12) promotes tumorigenesis and metastasis by targeting miR-199a/b-5p in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2017, 36, 11. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Yoshikawa, R.; Harada, H.; Harada, Y.; Ishida, A.; Yamazaki, T. Long noncoding RNA, CCDC26, controls myeloid leukemia cell growth through regulation of KIT expression. Mol. Cancer 2015, 14, 90. [Google Scholar] [CrossRef]
- Alawi, F.; Lin, P. Dyskerin is required for tumor cell growth through mechanisms that are independent of its role in telomerase and only partially related to its function in precursor rRNA processing. Mol. Carcinog. 2011, 50, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Deryusheva, S.; Gall, J.G. Orchestrated positioning of post-transcriptional modifications at the branch point recognition region of U2 snRNA. RNA 2018, 24, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Weiss, K.; Terhal, P.A.; Cohen, L.; Bruccoleri, M.; Irving, M.; Martinez, A.F.; Rosenfeld, J.A.; Machol, K.; Yang, Y.; Liu, P.; et al. De Novo Mutations in CHD4, an ATP-Dependent Chromatin Remodeler Gene, Cause an Intellectual Disability Syndrome with Distinctive Dysmorphisms. Am. J. Hum. Genet. 2016, 99, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Marnef, A.; Richard, P.; Pinzon, N.; Kiss, T. Targeting vertebrate intron-encoded box C/D 2′-O-methylation guide RNAs into the Cajal body. Nucleic Acids Res. 2014, 42, 6616–6629. [Google Scholar] [CrossRef]
- Vare, V.Y.; Eruysal, E.R.; Narendran, A.; Sarachan, K.L.; Agris, P.F. Chemical and Conformational Diversity of Modified Nucleosides Affects tRNA Structure and Function. Biomolecules 2017, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Pacilli, A.; Ceccarelli, C.; Trere, D.; Montanaro, L. SnoRNA U50 levels are regulated by cell proliferation and rRNA transcription. Int. J. Mol. Sci. 2013, 14, 14923–14935. [Google Scholar] [CrossRef] [PubMed]
- Michel, C.I.; Holley, C.L.; Scruggs, B.S.; Sidhu, R.; Brookheart, R.T.; Listenberger, L.L.; Behlke, M.A.; Ory, D.S.; Schaffer, J.E. Small nucleolar RNAs U32a, U33, and U35a are critical mediators of metabolic stress. Cell Metab. 2011, 14, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Bortolin-Cavaille, M.L.; Cavaille, J. The SNORD115 (H/MBII-52) and SNORD116 (H/MBII-85) gene clusters at the imprinted Prader-Willi locus generate canonical box C/D snoRNAs. Nucleic Acids Res. 2012, 40, 6800–6807. [Google Scholar] [CrossRef] [PubMed]
- Kishore, S.; Stamm, S. The snoRNA HBII-52 regulates alternative splicing of the serotonin receptor 2C. Science 2006, 311, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Cavaille, J.; Seitz, H.; Paulsen, M.; Ferguson-Smith, A.C.; Bachellerie, J.P. Identification of tandemly-repeated C/D snoRNA genes at the imprinted human 14q32 domain reminiscent of those at the Prader-Willi/Angelman syndrome region. Hum. Mol. Genet. 2002, 11, 1527–1538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yang, M.; Marks, P.; White, L.M.; Hurtig, M.; Mi, Q.S.; Divine, G.; Gibson, G. Serum non-coding RNAs as biomarkers for osteoarthritis progression after ACL injury. Osteoarthritis Cartilage 2012, 20, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Kishikawa, T.; Otsuka, M.; Ohno, M.; Yoshikawa, T.; Takata, A.; Koike, K. Circulating RNAs as new biomarkers for detecting pancreatic cancer. World J. Gastroenterol. 2015, 21, 8527–8540. [Google Scholar] [CrossRef]
- Kasai, H.; Nadano, D.; Hidaka, E.; Higuchi, K.; Kawakubo, M.; Sato, T.A.; Nakayama, J. Differential expression of ribosomal proteins in human normal and neoplastic colorectum. J. Histochem. Cytochem. 2003, 51, 567–574. [Google Scholar] [CrossRef]
- Nadano, D.; Ishihara, G.; Aoki, C.; Yoshinaka, T.; Irie, S.; Sato, T.A. Preparation and characterization of antibodies against human ribosomal proteins: Heterogeneous expression of S11 and S30 in a panel of human cancer cell lines. Jpn. J. Cancer Res. 2000, 91, 802–810. [Google Scholar] [CrossRef]
- Zou, A.E.; Ku, J.; Honda, T.K.; Yu, V.; Kuo, S.Z.; Zheng, H.; Xuan, Y.; Saad, M.A.; Hinton, A.; Brumund, K.T.; et al. Transcriptome sequencing uncovers novel long noncoding and small nucleolar RNAs dysregulated in head and neck squamous cell carcinoma. RNA 2015, 21, 1122–1134. [Google Scholar] [CrossRef]
- Mourtada-Maarabouni, M.; Pickard, M.R.; Hedge, V.L.; Farzaneh, F.; Williams, G.T. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in breast cancer. Oncogene 2009, 28, 195–208. [Google Scholar] [CrossRef]
- Xu, L.; Ziegelbauer, J.; Wang, R.; Wu, W.W.; Shen, R.F.; Juhl, H.; Zhang, Y.; Rosenberg, A. Distinct Profiles for Mitochondrial t-RNAs and Small Nucleolar RNAs in Locally Invasive and Metastatic Colorectal Cancer. Clin. Cancer Res. 2016, 22, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Corral, L.; Mateos, M.V.; Corchete, L.A.; Sarasquete, M.E.; de la Rubia, J.; de Arriba, F.; Lahuerta, J.J.; Garcia-Sanz, R.; San Miguel, J.F.; Gutierrez, N.C. Genomic analysis of high-risk smoldering multiple myeloma. Haematologica 2012, 97, 1439–1443. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Bracken, C.P.; Pillman, K.A.; Lawrence, D.M.; Goodall, G.J.; Callen, D.F.; Neilsen, P.M. p53 Represses the Oncogenic Sno-MiR-28 Derived from a SnoRNA. PLoS ONE 2015, 10, e0129190. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Han, L.; Wei, J.; Zhang, K.; Shi, Z.; Duan, R.; Li, S.; Zhou, X.; Pu, P.; Zhang, J.; et al. SNORD76, a box C/D snoRNA, acts as a tumor suppressor in glioblastoma. Sci. Rep. 2015, 5, 8588. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chang, L.; Wang, H.; Ma, W.; Peng, Q.; Yuan, Y. Clinical significance of C/D box small nucleolar RNA U76 as an oncogene and a prognostic biomarker in hepatocellular carcinoma. Clin. Res. Hepatol. Gastroenterol. 2018, 42, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Wang, H.; Han, L.; Jing, W.; Zhou, X.; Liu, Z. Up-regulation of small nucleolar RNA 78 is correlated with aggressive phenotype and poor prognosis of hepatocellular carcinoma. Tumour Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, L.M.; Wu, H.; Tian, G.; Dai, S.L.; Zhao, R.Y.; Shan, B.E. C/D-Box Snord105b Promotes Tumorigenesis in Gastric Cancer via ALDOA/C-Myc Pathway. Cell. Physiol. Biochem. 2018, 45, 2471–2482. [Google Scholar] [CrossRef] [PubMed]
- Krogh, N.; Kongsbak-Wismann, M.; Geisler, C.; Nielsen, H. Substoichiometric ribose methylations in spliceosomal snRNAs. Org. Biomol. Chem. 2017, 15, 8872–8876. [Google Scholar] [CrossRef] [PubMed]
- Brameier, M.; Herwig, A.; Reinhardt, R.; Walter, L.; Gruber, J. Human box C/D snoRNAs with miRNA like functions: Expanding the range of regulatory RNAs. Nucleic Acids Res. 2011, 39, 675–686. [Google Scholar] [CrossRef]
- Pan, Y.Z.; Zhou, A.; Hu, Z.; Yu, A.M. Small nucleolar RNA-derived microRNA hsa-miR-1291 modulates cellular drug disposition through direct targeting of ABC transporter ABCC1. Drug Metab. Dispos. Biol. Fate Chem. 2013, 41, 1744–1751. [Google Scholar] [CrossRef]
| Guide RNA | Target rRNA Position (Uridine) | Host Gene | Disease Associated with the RNA | Level Compared to Normal | Ref. |
|---|---|---|---|---|---|
| snoRA2A (ACA2A) | 28S-4263, -4282 | KANSL2 (KAT8 regulatory NSL complex subunit 2) | Breast cancer | Up | [66] |
| snoRA6 (ACA6); snoRA62 (E2) | 28S-3616, -4438, and -3830, -3832 | RPSA (ribosomal protein SA, 67 kD laminin receptor) | Isolated congenital asplenia | Down-regulation of RPSA due to mutations | [62] |
| snoRA15 (ACA15) | 18S-1367 | CCT6A (chaperonin containing TCP1, subunit 6A (zeta 1)) | Acute myeloblastic, acute lymphoblastic and peripheral T-cell lymphoma, X-Linked DC | Down | [29,52] |
| snoRA21 (ACA21) | 28S-4401, 18S-1499 | RPL23 (ribosomal protein L23) | Colorectal cancer | Up | [48] |
| snoRA22 (ACA22) | 28S-4966 (snoRA33 *), -4975 | CCT6P1 (chaperonin containing TCP1 subunit 6 pseudogene 1) | DC | Down | [65] |
| snoRA23 (ACA23) | 28S-3737, -4331 | IPO7 (importin 7) | Pancreatic Cancer, DC | Up | [43,67] |
| snoRA24 (ACA24) | 18S-609, -863, 18S-1045 (snoRA24B *) | SNHG8 (Small Nucleolar RNA Host Gene 8) | Acute myeloblastic, acute and chronic lymphoblastic, and peripheral T-cell lymphoma, X-Linked DC | Down | [29,52] |
| snoRA27 (ACA27) | 28S-3694, -4522 | RPL21 (ribosomal protein L21) | HHS | - | [68] |
| snoRA31 (ACA31) | 28S-3713, 18S-218 | TPT1 (Tumor protein, translationally-controlled 1) | Musculoskeletal aging and osteoarthritis | Up | [69] |
| snoRA36 (ACA36) | 18S-105, -1244 | DKC1 (dyskeratosis congenita 1, dyskerin) | DC, X-Linked DC | Down | [65] |
| snoRA40 (ACA40) | 28S-4546, 18S-1174 | TAF1D (TATA-box binding protein-associated factor, RNA pol I subunit D) | Multiple myeloma of the TC1 subgroup, Asthma, Multiple sclerosis | Up | [54,70,71] |
| snoRA42 (ACA42) | 18S-109, -572 (snoRA80 *) | KHDC4 (KH Domain Containing 4) | NSCLC, X-Linked DC | Up | [46,72] |
| snoRA44 (ACA44) | 18S-686, -822, -897 | SNHG12 (small nucleolar RNA host gene 12) | Hepatocellular carcinoma | Host gene up-regulation | [73] |
| snoRA47 (HBI-115) | 28S-1766 | ZBED3 ( zinc finger BED-type containing 3) | Hepatocellular carcinoma | Up | [49] |
| snoRA48 (ACA48) | 28S-3797 | SENP3-EIF4A1 (eukaryotic translation initiation factor 4A1) | Breast Cancer | Down | [29] |
| snoRA56 (ACA56) | 28S-1664, 18S-296 | DKC1 | DC, X-Linked DC | Down | [52,59] |
| snoRA64 (U64) | 28S-4975 | RPS2 (ribosomal protein S2) | Multiple myeloma, Prostate cancer, X-DC | Up | [29,52,54] |
| snoRA66 (U66) | 18S-119 | RPL5 (ribosomal protein L5) | Diamond-Blackfan Anemia | Up | [63] |
| snoRA67 (U67) | 18S-1445 | SENP3-EIF4A1 | X-Linked DC | Down | [52] |
| snoRA71C, D (U71C, U71D) | 18S-406 | SNHG17 (small nucleolar RNA host gene 17) | Myelofibrosis, a variant is expressed in HNSCC | Down | [53,56] |
| snoRA74A (U19) | 28S-3741, -3743 and U3-8 | MATR3 (Matrin 3) | Astrocytoma, Gallbladder cancer | Up | [51,74] |
| snoRA81 (HBI-61) | 28S-4606 | EIF4A2 (eukaryotic translation initiation factor 4A, isoform 2) | DC | Down | [75] |
| Guide RNA | Target snRNA Position (Uridine) | Host Gene | Disease Associated with the RNA | Level Compared to Normal | Ref. |
|---|---|---|---|---|---|
| scaRNA1 (ACA35) | U2-89 | PPP1R8 (protein phosphatase 1 regulatory subunit 8) | Tetralogy of Fallot (TOF), a heart condition in children | Down | [61] |
| scaRNA4 (ACA26) | U2-41 | KHDC4 (KH domain containing 4, pre-mRNA splicing factor) | TOF | Down | [61,76] |
| scaRNA8 (U92) | U2-34, -43, -44 | HAUS6 (HAUS augmin like complex subunit 6) | TOF | Down | [61,76] |
| scaRNA11 (ACA57) | U5-43 | CHD4 (Chromodomain helicase DNA binding protein 4) | Sifrim-Hitz-Weiss syndrome | Host gene mutation | [77] |
| scaRNA13 (U93) | U2-54, U5-51 | SNHG170 (small nucleolar RNA host gene 170) | DC, Congenital heart defects | Down | [58] |
| scaRNA23 (ACA12) | U6-40 | COP1 (COP1, E3 ubiquitin ligase) | Breast Cancer | UP | [57] |
| Guide RNA | Target rRNA/snRNA Position | Host Gene | Disease Associated with the RNA | Level Compared to Normal | Ref. |
|---|---|---|---|---|---|
| SNORD7 (Z30) | U6-A47 | LINC02001 (long intergenic non-protein coding RNA 2001) | Tetralogy of Fallot (TOF), a heart condition in children | Down | [61] |
| SNORD8 (mgU6-53) | U6-A53 | CHD8 (chromo-domain helicase DNA binding protein 8) | TOF | Down | [61] |
| SNORD9 (mgU6-53B) | U6-A53 | CHD8 | TOF | Down | [61] |
| SNORD19 (HBII-108) | 18S-G683 (SNORD136) * | GNL3 (G protein nucleolar 3) | Colorectal cancer | Up | [91] |
| SNORD25 (U25) | 18S-G1490 | SNHG1 (small nucleolar RNA host gene 1) | Multiple Myeloma | Up | [92] |
| SNORD27 (U27) | 18S-A27 | SNHG1 | Multiple Myeloma | Up | [92] |
| SNORD28 (U28) | 18S-C1391 | SNHG1 | Breast tumors | Up | [93] |
| SNORD30 (U30) | 28S-A3804 18S-C1383 | SNHG1 | Multiple Myeloma | Up | [92] |
| SNORD31 (U31) | 28S-G4166 | SNHG1 | Multiple Myeloma | Up | [92] |
| SNORD32 (U32) | 28S-A1511 18S-G1328 | RPL13 (ribosomal protein L13) | Secondary plasma cell leukemia | Down | [54] |
| SNORD33A (U33A) | 18S-U1326 | RPL13 | NSCLC | Up | [26] |
| SNORD35A (U35A) | 28S-C4506 | RPL13 | Colorectal carcinomas, Head and neck cancer | Up | [87,89] |
| SNORD35B (U35B) | 28S-C4506 | RPS11 (ribosomal protein S11) | Colorectal carcinomas, HNSCC | Up | [87,89] |
| SNORD38A (U38A) SNORD38B (U38B) | 28S-A1858 | RPS8 (ribosomal protein S8) | ACL | Up | [85] |
| SNORD43 (U43) | 18S-C1703 | RPL3 (ribosomal protein L3) | Breast cancer and HNSCC | Patient-specific variability | [25] |
| SNORD44 (U44) | 18S-A166 | GAS5 (Growth arrest-specific transcript 5) | Breast cancer and HNSCC | Down | [25] |
| SNORD48 (U48) | 28S-C2279 | KIF24 (Kinesin Family Member 24) | ACL | Up | [85] |
| SNORD50A (U50) | 28S-C2848, 28S-G2863 | SNHG5 (small nucleolar RNA host gene 5) | Breast cancer, prostate cancer, colon cancer | Down | [23,24,80] |
| SNORD66 (HBII-142) | 18S-C1272 | EIF4G1 (Eukaryotic translation initiation factor 4 gamma, 1) | NSCLC | Up | [26] |
| SNORD67 (HBII-166) | U6-C60 | CKAP5 (cytoskeleton associated protein 5) | TOF | Down | [61] |
| SNORD71 (HBII-239) | 5.8S-U14 | AP1G1 (adaptor-related protein complex 1 subunit gamma 1) | T cell lymphoma | Down | [55] |
| SNORD76 (U76) | 28S-A2350 | GAS5 | Hepatocellular carcinoma, glioblastoma and NSCLC | Up in NSCLC | [94,95] |
| SNORD78 (U78) | 28S-G4593 | GAS5 | Hepatocellular carcinoma | Up | [96] |
| SNORD94 (U94) | U6-C62 | PTCD3 (pentatricopeptide repeat domain 3) | TOF | Down | [61] |
| SNORD98 (HBII-419) | 18S-G867 | CCAR1 (cell division cycle and apoptosis regulator 1) | Colorectal cancer | Up | [91] |
| SNORD105B | 18S-U799 | PPAN (peter pan homolog) | Gastric cancer | Up | [97] |
| Guide RNA | Target snRNA Position | Host Gene | Disease Associated with the RNA | Level Compared to Normal | Ref. |
|---|---|---|---|---|---|
| scaRNA2 (HBII-382) | U2-G25 and G61 | Independent transcriptional unit | Congenital heart defect and TOF | Down | [58,61] |
| scaRNA9 (Z32) | U2-G19, A30 | Centrosomal Protein 295 | Chronic lymphocytic leukemia and TOF | Down | [54,61] |
| scaRNA17 (U91) | U4-C8 and U12-G22 | SNHG22 (small nucleolar RNA host gene 22) | Chronic lymphocytic leukemia | Down | [54,98] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deogharia, M.; Majumder, M. Guide snoRNAs: Drivers or Passengers in Human Disease? Biology 2019, 8, 1. https://doi.org/10.3390/biology8010001
Deogharia M, Majumder M. Guide snoRNAs: Drivers or Passengers in Human Disease? Biology. 2019; 8(1):1. https://doi.org/10.3390/biology8010001
Chicago/Turabian StyleDeogharia, Manisha, and Mrinmoyee Majumder. 2019. "Guide snoRNAs: Drivers or Passengers in Human Disease?" Biology 8, no. 1: 1. https://doi.org/10.3390/biology8010001
APA StyleDeogharia, M., & Majumder, M. (2019). Guide snoRNAs: Drivers or Passengers in Human Disease? Biology, 8(1), 1. https://doi.org/10.3390/biology8010001





