Epigenetic Inheritance and Its Role in Evolutionary Biology: Re-Evaluation and New Perspectives
Abstract
:1. Introduction
2. What Is “Epigenetics”?
“Some employ epigenetics to explain changes in gene expression, others use it to refer to transgenerational effects and/or inherited expression states. This disagreement on a clear definition has made communication difficult, synthesis of epigenetic research across fields nearly impossible, and has in many ways biased methodologies and interpretations.”
3. A Survey of Epigenetic Research Papers: Methodology
- Searches of the PubMed database only identified papers whose title or abstract contained a search term; that is, the body of the paper was not part of the search. Thus, it is possible that papers discussing epigenetics in some form or other have been overlooked. However, in a sense, this eliminated trivial references to epigenetics and focused the survey on those papers that actually thought epigenetics was important enough to include in the abstract, if not the title itself.
- The choice of search terms reflects the author’s view of biology. Thus, for example, “evo-devo” may arguably not be a “discipline” within the life sciences, per se, but it does reflect an actively growing area of biology.
- This survey were carried out in February of 2016. The PubMed database is expanded daily, if not hourly. Indeed, estimates are that an average of ~1 paper a minute is added to this database, but in batches. Therefore, this survey is unique to that specific month and year and in this sense is not exactly replicable by the reader using the current on-line database. However, the incremental changes are unlikely to affect the outcomes of the survey or its conclusions, at least in the short term, given PubMed’s huge article database.
- Importantly, any survey is affected by the source of its data, and this one is no different. While PubMed contains an enormous number of scientific articles, as implied by its name, the database does primarily focus on biomedical research. Certainly, there are journals in the area of evolution and ecology that are not indexed by PubMed (though plant biology, for example, generally is well represented). Thus, there may be a bias towards underrepresentation of evolution papers in this database. On the other hand, as the survey goes on to reveal, the near complete absence of evolutionary papers mentioning epigenetics compared to the overall number of epigenetic papers speaks volumes about the failure to link epigenetics with biology, when considering the many life science journals in animal, plant and microbial science that are indexed by PubMed.
- No distinction was made between primary research papers and review papers in this survey.
- Collectively, the caveats in this survey do point out some sources of inaccuracy in the survey. For example, the secondary search terms are somewhat subjective choices made by the author. Moreover, in some limited instances, papers are undoubtedly double counted, e.g., a paper containing the words “epigenetic”, “illness” and “death” contributes to the paper count for the overall category of “disease”. Important to emphasize is that the goal of this survey was simply to seek out general patterns, not to provide an accurate statistical analysis that would characterize a true meta-analysis. However, given the enormous number of papers in the PubMed data base, nearly 50,000 of which reference epigenetics in some form or other, it is assumed that meta-patterns will emerge despite these inherent limitations of the analytical approach. To emphasize the general nature of these findings, data on percentages are reported to the nearest whole number (but even rounding to the nearest 10% would not obscure the emergent patterns, as is evident below).
4. The Reach of Epigenetic Research in the Life Sciences
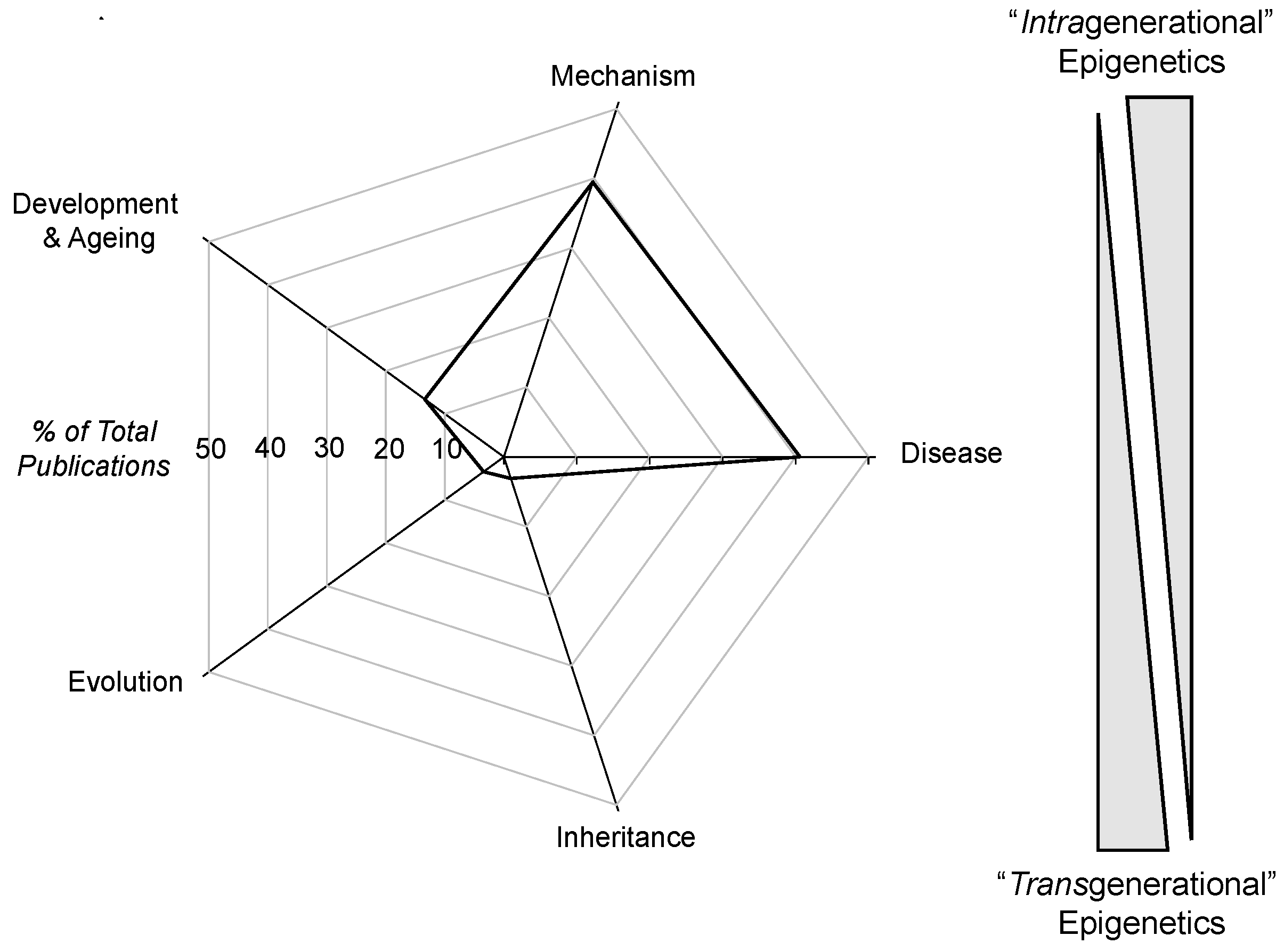
4.1. Intragenerational and Transgenerational Epigenetics
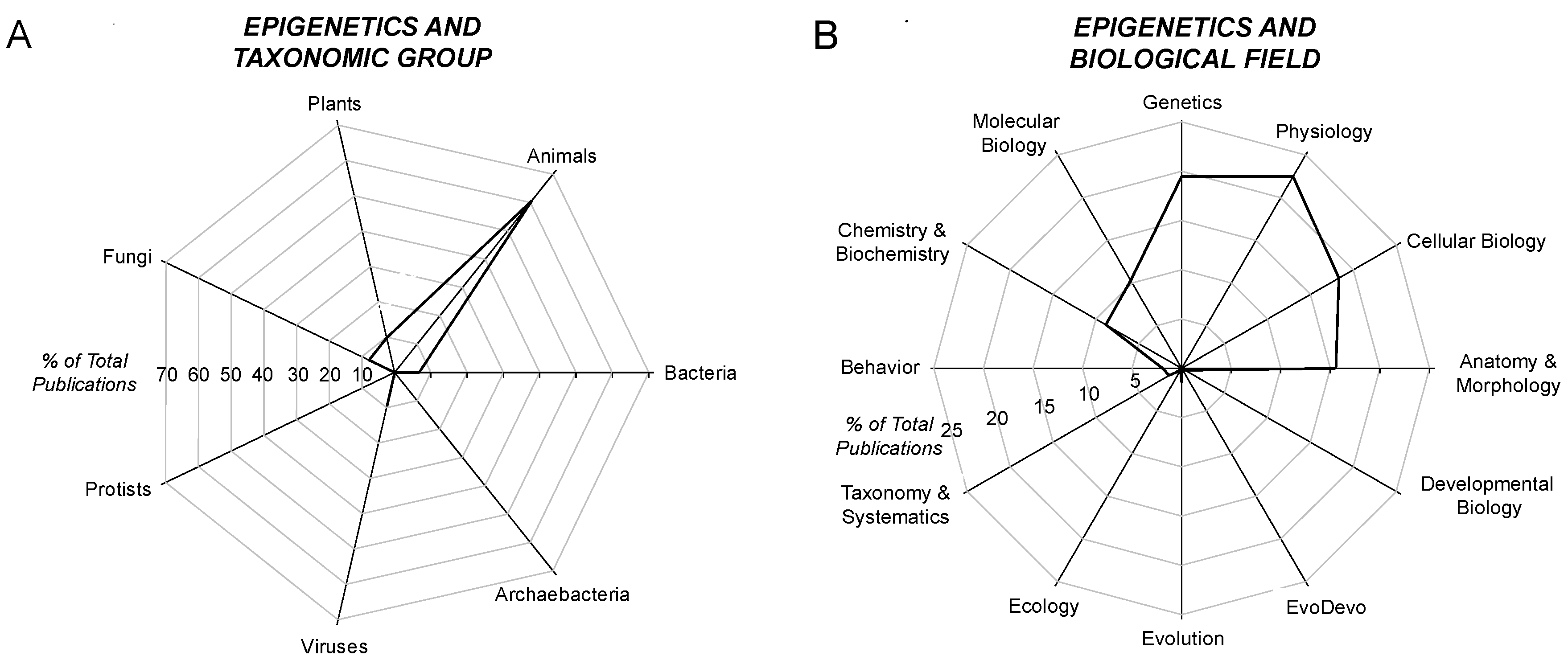
4.2. Epigenetics and Taxon
4.3. Epigenetics and the Biological Field
5. Investigating Epigenetic Inheritance: The Challenges
5.1. The Resource Cost of Epigenetic Studies
5.2. Elusive Epigenetic Mechanisms
5.3. The Complexities of Epigenetic “Dynamics”
5.4. Keep Calm and Carry on with Transgenerational Epigenetic Research
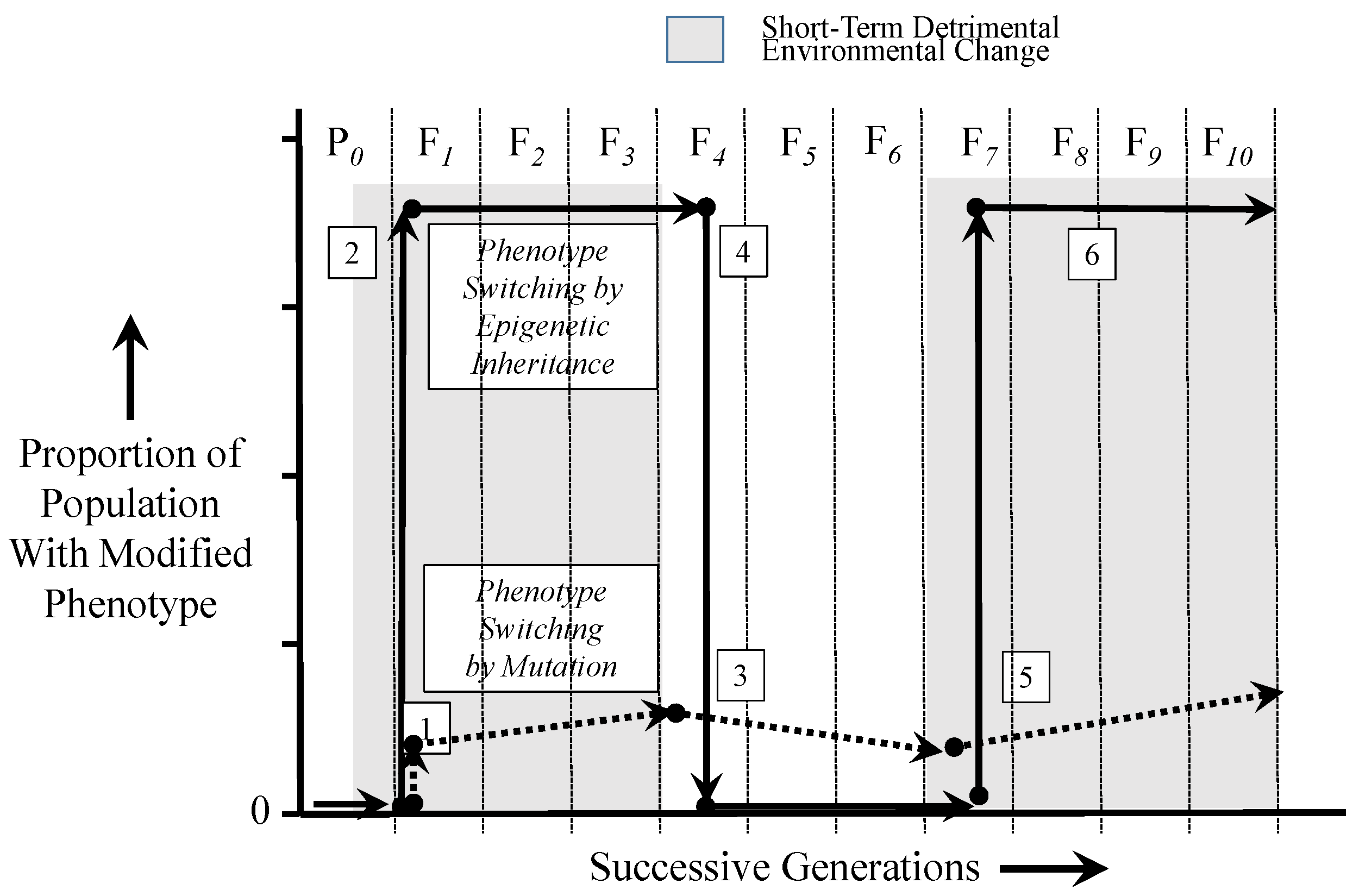
6. Epigenetics in a Dynamic Environment: Consequences of Rapid and Widely-Distributed Phenotype Switching
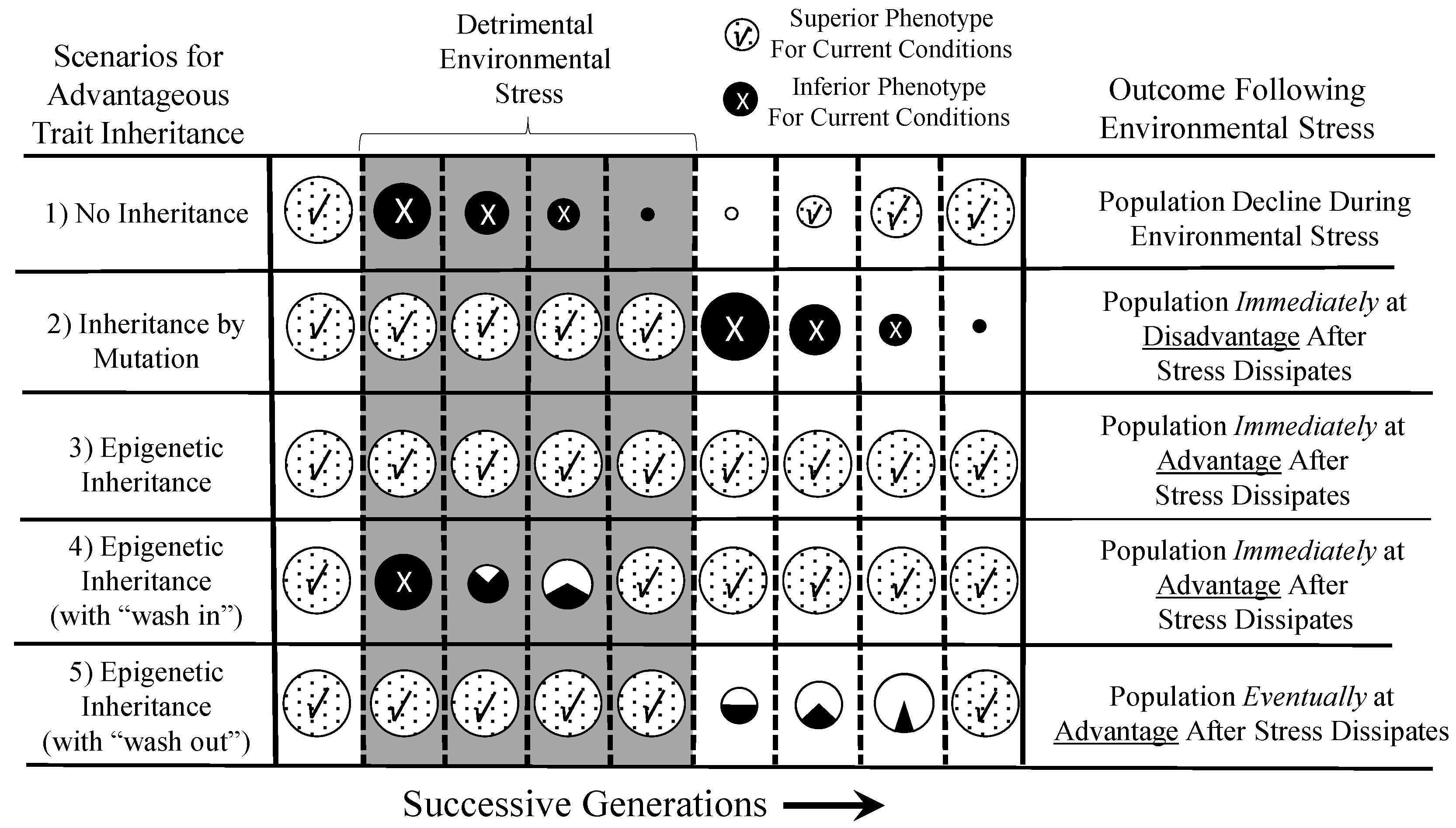
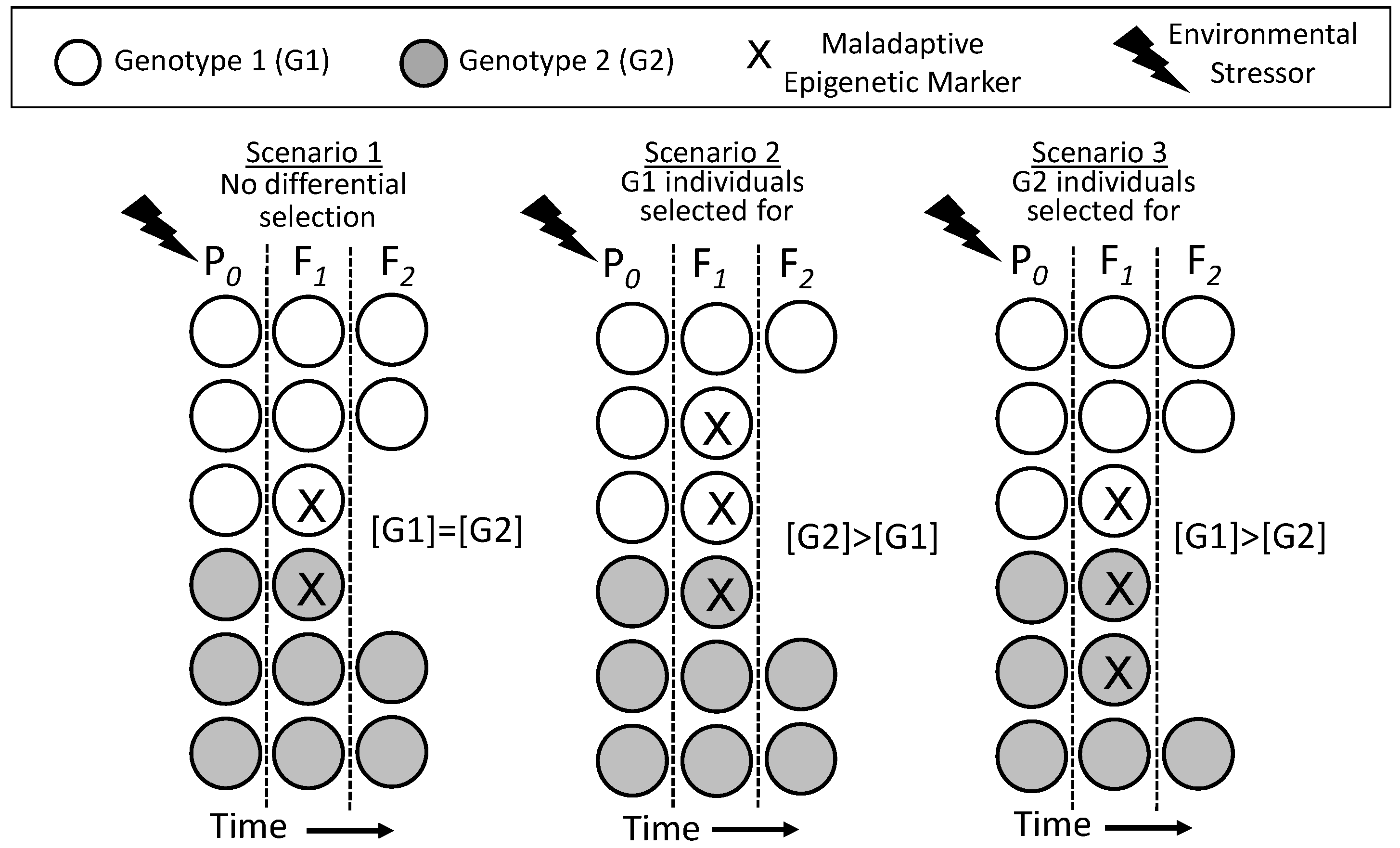
6.1. Epigenetically-Inherited Phenotypes: Neutral, Advantageous or Disadvantageous?
6.2. Comparing the Time Courses of Genetic and Epigenetic Inheritance
7. An “Epigenetic Advantage” in Changing Environments
7.1. Epigenetics in a Temporally-Complex Environment
7.2. Epigenetics in a Spatially-Complex Environment and the Exploitation of New Ecological Niches
8. A Role for Epigenetics in Evolution
8.1. Is Epigenetics Even Relevant to Evolution?
8.2. How Epigenetic Inheritance Influences Evolution
8.2.1. Beyond the Modern Evolutionary Synthesis
8.2.2. Direct Incorporation of an Epigenetically-Inherited Phenotype into the Genome
8.2.3. Indirect Epigenetic Effects on Evolution: Altered Gene Stability and Mutation Rates
8.2.4. Indirect Epigenetic Effects on Evolution: Altered Natural Selection
- (1)
- Phenotypes can be epigenetically inherited. Abundant evidence of epigenetic inheritance exists in numerous organisms, as already discussed.
- (2)
- Epigenetically-inherited phenotypes can be neutral, adaptive or maladaptive. Again, this has been discussed above in the context of epigenetically-inherited disease, for example.
- (3)
- Variation in genotype exists between members of a population. This is, of course, a basic tenet of Mendelian inheritance, although fitness is increasingly viewed as a complex combination of population density, genotype and genotype frequencies [98].
- (4)
- Variation in epigenetic markers exists between members of a population. There is increasing appreciation that there is variation between individuals within a population (and in an individual as a function of age) in the DNA methylation or post-translational acetylation, methylation, phosphorylation or ubiquitination of histones associated with the nucleosome cores and the phenotypes they ultimately produce; e.g., [53,55,64,78,81,106,125,132,134,135,136,137,138,139,140,141,142,143,144]. There are also spontaneous changes in DNA methylation patterns [89,145]. Thus, both the epigenome and genome of a population show heterogeneity to a greater or lesser extent.
- (5)
- Within a population, a particular set of epigenetic markers may be distributed completely independently from a particular genotype. That is, a particular genotype in a population may or may not be associated with a particular set of epigenetic markers and the associated phenotype.
8.3. Traditional Genetic Inheritance of Mechanisms for Epigenetic Inheritance
9. Conclusions and A Possible Future for Epigenetic Research
Acknowledgments
Conflicts of Interest
References and Notes
- Waddington, C.H. The epigenotype. Endeavour 1942, 1, 18–20. [Google Scholar] [CrossRef] [PubMed]
- Burggren, W.W.; Crews, D. Epigenetics in comparative biology: Why we should pay attention. Integr. Comp. Biol. 2014, 54, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, E.E.; Skinner, M.K. Environmentally induced epigenetic transgenerational inheritance of reproductive disease. Biol. Reprod. 2015, 93. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K.; Guerrero-Bosagna, C.; Haque, M.M. Environmentally induced epigenetic transgenerational inheritance of sperm epimutations promote genetic mutations. Epigenetics 2015, 10, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K. Role of epigenetics in developmental biology and transgenerational inheritance. Birth. Defects. Res. C. Embryo Today 2011, 93, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K. Environmental Epigenetics and a Unified Theory of the Molecular Aspects of Evolution: A Neo-Lamarckian Concept that Facilitates Neo-Darwinian Evolution. Genome Biol. Evol. 2015, 7, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Ho, D.H.; Burggren, W.W. Epigenetics and transgenerational transfer: A physiological perspective. J. Exp. Biol. 2010, 213, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Crews, D.; Gillette, R.; Miller-Crews, I.; Gore, A.C.; Skinner, M.K. Nature, nurture and epigenetics. Mol. Cell. Endocrinol. 2014, 398, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Relton, L.R.; Smith, G.D. Is epidemiology ready for epigenetics? Int. J. Epidemiol. 2012, 41, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Recio, O.; Toro, M.A.; Bach, A. Past, present, and future of epigenetics applied to livestock breeding. Front. Genet. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Deans, C.; Maggert, K.A. What do you mean, "epigenetic"? Genetics 2015, 199, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.R. Epigenetics and memigenetics. Cell Mol. Life Sci. 2014, 71, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Tammen, S.A.; Friso, S.; Choi, S.W. Epigenetics: The link between nature and nurture. Mol. Aspects Med. 2013, 34, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Dupont, C.; Armant, D.R.; Brenner, C.A. Epigenetics: Definition, mechanisms and clinical perspective. Semin. Reprod. Med. 2009, 27, 351–357. [Google Scholar]
- Guerrero-Bosagna, C.M.; Skinner, M.K. Epigenetic transgenerational effects of endocrine disruptors on male reproduction. Semin. Reprod. Med. 2009, 27, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, K.J.; von Holdt, B.M.; Sork, V.L. Epigenetics in ecology and evolution: What we know and what we need to know. Mol. Ecol. 2016. [Google Scholar] [CrossRef]
- Martos, S.N.; Tang, W.Y.; Wang, Z. Elusive inheritance: Transgenerational effects and epigenetic inheritance in human environmental disease. Prog. Biophys. Mol. Biol. 2015, 118, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Gissis, S.; Jablonka, E. Trarnsformations of Lamarckism; MIT Press: Cambridge, MA, USA, 2011; pp. 1–457. [Google Scholar]
- Bateson, P. The impact of the organism on its descendants. Genet. Res. Int. 2012. [Google Scholar] [CrossRef] [PubMed]
- Popper, K. Objective Knowledge: An evolutionary approach; Oxford University Press: Oxford, UK, 1979. [Google Scholar]
- Nelson, T.C.; Groth, K.D.; Sotherland, P.R. Maternal investment and nutrient use affect phenotype of American alligator and domestic chicken hatchlings. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2010, 157, 19–27. [Google Scholar]
- Reed, W.L.; Clark, M.E. Beyond maternal effects in birds: Responses of the embryo to the environment. Integr. Comp. Biol. 2011, 51, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Ho, D.H. Transgenerational epigenetics: The role of maternal effects in cardiovascular development. Integr. Comp. Biol. 2014, 54, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Seemann, F.; Peterson, D.R.; Witten, P.E.; Guo, B.S.; Shanthanagouda, A.H.; Ye, R.R.; Zhang, G.; Au, D.W. Insight into the transgenerational effect of benzo[a]pyrene on bone formation in a teleost fish (Oryzias latipes). Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2015, 178, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Clarke, H.J.; Vieux, K.F. Epigenetic inheritance through the female germ-line: The known, the unknown, and the possible. Semin. Cell Dev. Biol. 2015, 43, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Jinha, A.E. Article 50 million: An estimate of the number of scholarly articles in existince. Learn. Publ. 2010, 23, 258–263. [Google Scholar] [CrossRef]
- Prokopuk, L.; Western, P.S.; Stringer, J.M. Transgenerational epigenetic inheritance: Adaptation through the germline epigenome? Epigenomics 2015, 7, 829–846. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Chillaron, J.C.; Ramon-Krauel, M.; Ribo, S.; Diaz, R. Transgenerational epigenetic inheritance of diabetes risk as a consequence of early nutritional imbalances. Proc. Nutr. Soc. 2016, 75, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Not all would agree with these taxonomic choices of search terms, but, again, the purpose of this meta-analysis is to determine relative frequencies and patterns of various taxa in epigenetic studies, not to provide a comprehensive quantitative analysis.
- Ronsseray, S. Paramutation phenomena in non-vertebrate animals. Semin. Cell Dev. Biol. 2015, 44, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Andrewartha, S.J.; Burggren, W.W. Transgenerational variation in metabolism and life-history traits induced by maternal hypoxia in Daphnia magna. Physiol. Biochem. Zool. 2012, 85, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, K.; Twyman, R.M.; Vilcinskas, A. Insects as models to study the epigenetic basis of disease. Prog. Biophys. Mol. Biol. 2015, 118, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Schatten, H.; Sun, Q.Y. Environmental epigenetic inheritance through gametes and implications for human reproduction. Hum. Reprod. Update 2015, 21, 194–208. [Google Scholar] [CrossRef] [PubMed]
- Padilla, P.A.; Garcia, A.M.; Ladage, M.L.; Toni, L.S. Caenorhabditis elegans: An old genetic model can learn new epigenetic tricks. Integr. Comp. Biol. 2014, 54, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Perfus-Barbeoch, L.; Castagnone-Sereno, P.; Reichelt, M.; Fneich, S.; Roquis, D.; Pratx, L.; Cosseau, C.; Grunau, C.; Abad, P. Elucidating the molecular bases of epigenetic inheritance in non-model invertebrates: The case of the root-knot nematode Meloidogyne incognita. Front. Physiol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, S.; Thompson, M.B.; Seebacher, F. Energetic cost of a meal in a frequent feeding lizard. Comp. Biochem. Physiol. 2003, 135, 377–382. [Google Scholar] [CrossRef]
- Iglesias, F.M.; Cerdan, P.D. Maintaining Epigenetic Inheritance During DNA Replication in Plants. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Sun, B.; Looi, L.; Xu, Y.; Gan, E.S.; Huang, J.; Ito, T. Co-ordination of Flower Development Through Epigenetic Regulation in Two Model Species: Rice and Arabidopsis. Plant Cell Physiol. 2015, 56, 830–842. [Google Scholar] [CrossRef] [PubMed]
- Jobson, M.A.; Jordan, J.M.; Sandrof, M.A.; Hibshman, J.D.; Lennox, A.L.; Baugh, L.R. Transgenerational Effects of Early Life Starvation on Growth, Reproduction, and Stress Resistance in Caenorhabditis elegans. Genetics 2015, 201, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Burggren, W.W. Epigenetics as a source of variation in comparative animal physiology—or—Lamarck is lookin’ pretty good these days. J. Exp. Biol. 2014, 217, 682–689. [Google Scholar] [CrossRef] [PubMed]
- Burggren, W.W. Dynamics of epigenetic phenomena: Intergenerational and intragenerational phenotype 'washout'. J. Exp. Biol. 2015, 218, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Corrales, J.; Thornton, C.; White, M.; Willett, K.L. Multigenerational effects of benzo[a]pyrene exposure on survival and developmental deformities in zebrafish larvae. Aquat. Toxicol. 2014, 148, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Saab, B.J.; Mansuy, I.M. Neurobiological disease etiology and inheritance: An epigenetic perspective. J. Exp. Biol. 2014, 217, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Vaiserman, A.M. Epigenetic engineering and its possible role in anti-aging intervention. Rejuvenation Res. 2008, 11, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Felling, R.J.; Song, H. Epigenetic mechanisms of neuroplasticity and the implications for stroke recovery. Exp. Neurol. 2015, 268, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Goddard, M.E.; Whitelaw, E. The use of epigenetic phenomena for the improvement of sheep and cattle. Front Genet. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Bilichak, A.; Kovalchuk, I. Transgenerational response to stress in plants and its application for breeding. J. Exp. Bot. 2016, 67, 2081–2092. [Google Scholar] [CrossRef] [PubMed]
- López-Arredondo, D.; González-Morales, S.I.; Bello-Bello, E.; Alejo-Jacuinde, G.; Herrera, L. Engineering food crops to grow in harsh environments. F1000Res. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Sun, Y.; Shen, K.; Sun, S.; Wang, J.; Jiang, T.; Cao, S.; Josiah, S.M.; Pang, J.; Lin, X.; et al. Immediate Genetic and Epigenetic Changes in F1 Hybrids Parented by Species with Divergent Genomes in the Rice Genus (Oryza). PLoS ONE 2015, 10, e0132911. [Google Scholar] [CrossRef] [PubMed]
- Springer, N.M.; McGinnis, K.M. Paramutation in evolution, population genetics and breeding. Semin. Cell Dev. Biol. 2015, 44, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Martinez Bautista, N.; Burggren, W.W.; University of North Texas, Denton, TX, USA. Unpublished data. 2016.
- Rando, O.J.; Verstrepen, K.J. Timescales of genetic and epigenetic inheritance. Cell 2007, 128, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Klironomos, F.D.; Berg, J.; Collins, S. How epigenetic mutations can affect genetic evolution: Model and mechanism. Bioessays 2013, 35, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.A.; Martin, M.D.; Nguyen, M.; Mendelson, T.C. Epigenetic divergence as a potential first step in darter speciation. Mol. Ecol. 2016, 25, 1883–1894. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.M.; Medrano, M.; Bazaga, P. Comparative spatial genetics and epigenetics of plant populations: Heuristic value and a proof of concept. Mol. Ecol. 2016, 25, 1653–1664. [Google Scholar] [CrossRef] [PubMed]
- Flatscher, R.; Frajman, B.; Schönswetter, P.; Paun, O. Environmental heterogeneity and phenotypic divergence: Can heritable epigenetic variation aid speciation? Genet. Res. Int. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Eichten, S.R.; Briskine, R.; Song, J.; Li, Q.; Swanson-Wagner, R.; Hermanson, P.J.; Waters, A.J.; Starr, E.; West, P.T.; Tiffin, F.; et al. Epigenetic and genetic influences on DNA methylation variation in maize populations. Plant Cell 2013, 25, 2783–2797. [Google Scholar] [CrossRef] [PubMed]
- Vastenhouw, N.L.; Brunschwig, K.; Okihara, K.L.; Müller, F.; Tijsterman, M.; Plasterk, R.H. Gene expression: Long-term gene silencing by RNAi. Nature 2006, 442. [Google Scholar] [CrossRef] [PubMed]
- Cubas, P.; Vincent, C.; Coen, E. An epigenetic mutation responsible for natural variation in floral symmetry. Nature 1999, 401, 157–161. [Google Scholar] [PubMed]
- Richards, C.L.; Bossdorf, O.; Pigliucci, M. What role does heritable epigenetic variation play in phenotypic evolution? BioScience 2010, 60, 232–237. [Google Scholar] [CrossRef]
- Hartl, D.L.; Clark, A.G. Principles of Population Genetics; Sinauer Associates: Sunderland, MA, USA, 2007. [Google Scholar]
- Dubin, M.J.; Zhang, P.; Meng, D.; Remigereau, M.S.; Osborne, E.J.; Paolo, C.F.; Drewe, P.; Kahles, A.; Jean, G.; Vilhjálmsson, B.; et al. DNA methylation in Arabidopsis has a genetic basis and shows evidence of local adaptation. Elife 2015, 4, e05255. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Arcelus, M.; Lappalainen, T.; Montgomery, S.B.; Buil, A.; Ongen, H.; Yurovsky, A.; Bryois, J.; Giger, T.; Romano, L.; Planchon, A.; et al. Passive and active DNA methylation and the interplay with genetic variation in gene regulation. Elife 2013, 2, e00523. [Google Scholar] [PubMed]
- Schmitz, R.J.; Schultz, M.D.; Urich, M.A.; Nery, J.R.; Pelizzola, M.; Libiger, O.; Alix, A.; McCosh, R.B.; Chen, H.; Schork, N.J.; et al. Patterns of population epigenomic diversity. Nature 2013, 495, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Brandvain, Y.; Wright, S.I. The Limits of Natural Selection in a Nonequilibrium World. Trends Genet. 2016, 32, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Taute, K.M.; Gude, S.; Nghe, P.; Tans, S.J. Evolutionary constraints in variable environments, from proteins to networks. Trends Genet. 2014, 30, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Bell, G. Fluctuating selection: The perpetual renewal of adaptation in variable environments. Philos. Trans. R. Soc. Lond. B. 2010, 365, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Angers, B.; Castonguay, E.; Massicotte, R. Environmentally induced phenotypes and DNA methylation: How to deal with unpredictable conditions until the next generation and after. Mol. Ecol. 2010, 19, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Bonduriansky, R.; Crean, A.J.; Day, T. The implications of nongenetic inheritance for evolution in changing environments. Evol. Appl. 2012, 5, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Dolinoy, D.C.; Huang, D.; Jirtle, R.L. Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc. Natl. Acad. Sci. USA 2007, 104, 13056–13061. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Geng, Y.; Li, B.; Chen, J.; Yang, J. Genome-wide DNA methylation alterations of Alternanthera philoxeroides in natural and manipulated habitats: Implications for epigenetic regulation of rapid responses to environmental fluctuation and phenotypic variation. Plant Cell Environ. 2010, 33, 1820–1827. [Google Scholar] [CrossRef] [PubMed]
- Soubry, A. Epigenetic inheritance and evolution: A paternal perspective on dietary influences. Prog. Biophys. Mol. Biol. 2015, 118, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Jablonka, E. Epigenetic inheritance and plasticity: The responsive germline. Prog. Biophys. Mol. Biol. 2013, 111, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Badyaev, A.V. Epigenetic resolution of the 'curse of complexity' in adaptive evolution of complex traits. J. Physiol. 2014, 592, 2251–2260. [Google Scholar] [CrossRef] [PubMed]
- Roux, F.; Colomé-Tatché, M.; Edelist, C.; Wardenaar, R.; Guerche, P.; Hospital, F.; Colot, V.; Jansen, R.C.; Johannes, F. Genome-wide epigenetic perturbation jump-starts patterns of heritable variation found in nature. Genetics 2011, 188, 1015–1017. [Google Scholar] [CrossRef] [PubMed]
- De Vos, M.G.; Poelwijk, F.J.; Battich, N.; Ndika, J.D.; Tans, S.J. Environmental dependence of genetic constraint. PLoS Genet. 2013, 9, e1003580. [Google Scholar] [PubMed]
- Ho, D.H.; Burggren, W.W. Parental hypoxic exposure confers offspring hypoxia resistance in zebrafish (Danio rerio). J. Exp. Biol. 2012, 215, 4208–4216. [Google Scholar] [CrossRef] [PubMed]
- Schrey, A.W.; Richards, C.L. Within-genotype epigenetic variation enables broad niche width in a flower living yeast. Mol. Ecol. 2012, 21, 2559–2561. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.M.; Pozo, M.I.; Bazaga, P. Jack of all nectars, master of most: DNA methylation and the epigenetic basis of niche width in a flower-living yeast. Mol. Ecol. 2012, 21, 2602–2616. [Google Scholar] [CrossRef] [PubMed]
- Preite, V.; Snoek, L.B.; Oplaat, C.; Biere, A.; van der Putten, W.H.; Verhoeven, K.J.F. The epigenetic footprint of poleward range-expanding plants in apomictic dandelions. Mol. Ecol. 2015, 24, 4406–4418. [Google Scholar] [CrossRef] [PubMed]
- Kilvitis, H.J.; Alvarez, M.; Foust, C.M.; Schrey, A.W.; Robertson, M.; Richards, C.L. Ecological epigenetics. Adv. Exp. Med. Biol. 2014, 781, 191–210. [Google Scholar] [PubMed]
- Robertson, M.; Richards, C. Opportunities and challenges of next-generation sequencing applications in ecological epigenetics. Mol. Ecol. 2015, 24, 3799–3801. [Google Scholar] [CrossRef] [PubMed]
- Richards, E.J. Population epigenetics. Curr. Opin. Genet. Dev. 2008, 18, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Bossdorf, O.; Richards, C.L.; Pigliucci, M. Epigenetics for ecologists. Ecol. Lett. 2008, 11, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, J. The epigenetics heretic. Science 2014, 343, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Heard, E.; Martienssen, R.A. Transgenerational epigenetic inheritance: myths and mechanisms. Cell 2014, 157, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Jablonka, E.; Lamb, M.J.; Zeligowski, A. Evolution in Four Dimensions: Genetics, Epigenetic, Behavioral, and Symbolic Variation in the History of Life; MIT Press: Cambridge, MA, USA, 2006; pp. 1–472. [Google Scholar]
- Smith, G.D. Epidemiology, epigenetics and the 'Gloomy Prospect': Embracing randomness in population health research and practice. Int. J. Epidemiol. 2011, 40, 537–562. [Google Scholar] [CrossRef] [PubMed]
- Weigel, D.; Colot, V. Epialleles in plant evolution. Genome Biol. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
- Stotz, K. Extended evolutionary psychology: The importance of transgenerational developmental plasticity. Front. Psychol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Dickins, T.E.; Rahman, Q. The extended evolutionary synthesis and the role of soft inheritance in evolution. Proc. Biol. Sci. 2012, 279, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Danchin, E.; Charmantier, A.; Champagne, F.A.; Mesoudi, A.; Pujol, B.; Blanchet, S. Beyond DNA: Integrating inclusive inheritance into an extended theory of evolution. Nat. Rev. Genet. 2011, 12, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Danchin, E.; Pocheville, A. Inheritance is where physiology meets evolution. J. Physiol. 2014, 592, 2307–2317. [Google Scholar] [CrossRef] [PubMed]
- Laland, K.N.; Uller, T.; Feldman, M.W.; Sterelny, K.; Müller, G.B.; Moczek, A.; Jablonka, E.; John Odling-Smee, J. The extended evolutionary synthesis: Its structure, assumptions and predictions. Proc. Biol. Sci. 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Boero, F. From Darwin's Origin of Species toward a theory of natural history. F1000Prime Rep. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Noble, D. Evolution beyond neo-Darwinism: A new conceptual framework. J. Exp. Biol. 2015. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tricker, P.J. Transgenerational inheritance or resetting of stress-induced epigenetic modifications: Two sides of the same coin. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Reznick, D. Hard and Soft Selection Revisited: How Evolution by Natural Selection Works in the Real World. J. Hered. 2016, 107, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Pigliucci, M. An extended synthesis for evolutionary biology. Ann. N. Y. Acad. Sci. 2009, 1168, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Holland, M.L.; Rakyan, V.K. Transgenerational inheritance of non-genetically determined phenotypes. Biochem. Soc. Trans. 2013, 41, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Anava, S.; Posner, R.; Rechavi, O. The soft genome. Worm 2014, 3, e989798. [Google Scholar] [CrossRef] [PubMed]
- Moya, C.; Boyd, R.; Henrich, J. Reasoning About Cultural and Genetic Transmission: Developmental and Cross-Cultural Evidence From Peru, Fiji, and the United States on How People Make Inferences About Trait Transmission. Top. Cogn. Sci. 2015, 7, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Torday, J.S. On the evolution of development. Trends Dev. Biol. 2014, 8, 17–37. [Google Scholar] [PubMed]
- Laland, K.; Uller, T.; Feldman, M.; Sterelny, K.; Müller, G.B.; Moczek, A.; Jablonka, E.; Odling-Smee, J.; Wray, G.A.; Hoekstra, H.E.; et al. Does evolutionary theory need a rethink? Nature 2014, 514, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Day, T.; Bonduriansky, R. A unified approach to the evolutionary consequences of genetic and nongenetic inheritance. Am. Nat. 2011, 178, E18–E36. [Google Scholar] [CrossRef] [PubMed]
- Hauser, M.T.; Aufsatz, W.; Jonak, C.; Luschnig, C. Transgenerational epigenetic inheritance in plants. Biochim. Biophys. Acta 2011, 1809, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Mendizabal, I.; Keller, T.E.; Zeng, J.; Yi, S.V. Epigenetics and evolution. Integr. Comp. Biol. 2014, 54, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, S. The decline of soft inheritance. In Transformations of Lamarckism; Gissis, S., Jablonka, E., Eds.; MIT Press: Cambridge, MA, USA, 2011; pp. 121–126. [Google Scholar]
- Wilkins, A. Why did the Modern Synthesis give short shrift to "Soft Inheritance"? In Transformations of Lamarckism; Gissis, S., Jablonka, E., Eds.; MIT Press: Cambridge, MA, USA, 2011; pp. 127–132. [Google Scholar]
- Katz, D.J.; Edwards, T.M.; Reinke, V.; Kelly, W.G. A C. elegans LSD1 demethylase contributes to germline immortality by reprogramming epigenetic memory. Cell 2009, 137, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Rechavi, O.; Houri-Ze'evi, L.; Anava, S.; Goh, W.S.; Kerk, S.Y.; Hannon, G.J.; Hobert, O. Starvation-induced transgenerational inheritance of small RNAs in C. elegans. Cell 2014, 158, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Pilu, R. Paramutation phenomena in plants. Semin. Cell Dev. Biol. 2015, 44, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Borges, F.; Martienssen, R.A. The expanding world of small RNAs in plants. Nat. Rev. Mol. Cell. Biol. 2015, 16, 727–741. [Google Scholar] [CrossRef] [PubMed]
- Boffelli, D.; Martin, D.I. Epigenetic inheritance: A contributor to species differentiation? DNA Cell Biol. 2012, 31, S11–S16. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.D.; O'Neill, R.J. Chromosomes, conflict, and epigenetics: Chromosomal speciation revisited. Annu. Rev. Genomics Hum. Genet. 2010, 11, 291–316. [Google Scholar] [CrossRef] [PubMed]
- Rebollo, R.; Horard, B.; Hubert, B.; Vieira, C. Jumping genes and epigenetics: Towards new species. Gene 2010, 454, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R. The inheritance of epigenetic defects. Science 1987, 238, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Bonduriansky, R.; Day, T. Nongenetic inheritance and the evolution of costly female preference. J. Evol. Biol. 2013, 26, 76–87. [Google Scholar] [PubMed]
- Rehan, V.K.; Liu, J.; Naeem, E.; Tian, J.; Sakurai, R.; Kwong, K.; Akbari, O.; Torday, J.S. Perinatal nicotine exposure induces asthma in second generation offspring. BMC Med. 2012, 10. [Google Scholar] [CrossRef] [PubMed]
- Schorderet, D.F.; Gartler, S.M. Analysis of CpG suppression in methylated and nonmethylated species. Proc. Natl. Acad. Sci. USA 1992, 89, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Misawa, K.; Kamatani, N.; Kikuno, R.F. The universal trend of amino acid gain-loss is caused by CpG hypermutability. J. Mol. Evol. 2008, 67, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Mugal, C.F.; Arndt, P.F.; Holm, L.; Ellegren, H. Evolutionary consequences of DNA methylation on the GC content in vertebrate genomes. G3 (Bethesda) 2015, 5, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Borstnik, B.; Pumpernik, D. The apparent enhancement of CpG transversions in primate lineage is a consequence of multiple replacements. J. Bioinform. Comput. Biol. 2014, 12. [Google Scholar] [CrossRef] [PubMed]
- Bird, A.P. DNA methylation and the frequency of CpG in animal DNA. Nucleic Acids Res. 1980, 8, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Dubrovina, A.S.; Kiselev, K.V. Age-associated alterations in the somatic mutation and DNA methylation levels in plants. Plant Biol. (Stuttg) 2016, 18, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumari, R.; Sharma, V.; Sharma, V. Roles, and establishment, maintenance and erasing of the epigenetic cytosine methylation marks in plants. J. Genet. 2013, 92, 629–666. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.T.; Kim, D.K.; Cockayne, D.A.; Belousov, A.; Bitter, H.; Cho, M.H.; Duvoix, A.; Edwards, L.D.; Lomas, D.A.; Miller, B.E.; et al. Systemic soluble receptor for advanced glycation endproducts is a biomarker of emphysema and associated with AGER genetic variants in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2013, 188, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.; Cao, Y.; Qin, J.; Song, X.; Zhang, Q.; Shi, Y.; Cao, L. DNA methylation, its mediators and genome integrity. Int. J. Biol. Sci. 2015, 11, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.X.; Hackett, J.A.; Nestor, C.; Dunican, D.S.; Madej, M.; Reddington, J.P.; Pennings, S.; Harrison, D.J.; Meehan, R.R. Apoptosis and DNA methylation. Cancers 2011, 3, 1798–1820. [Google Scholar] [CrossRef] [PubMed]
- Putiri, E.L.; Robertson, K.D. Epigenetic mechanisms and genome stability. Clin. Epigenetics 2011, 2, 299–314. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Zhu, B.; Li, M.; Wang, L.; Xu, L.; Zhang, H.; Zheng, S.; Qi, B.; Han, F.; Liu, B. Extensive and heritable epigenetic remodeling and genetic stability accompany allohexaploidization of wheat. Genetics 2011, 188, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Fischer, M.; Colot, V.; Bossdorf, O. Epigenetic variation creates potential for evolution of plant phenotypic plasticity. New Phytol. 2013, 197, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Sznajder, B.; Sabelis, M.W.; Egas, M. How Adaptive Learning Affects Evolution: Reviewing Theory on the Baldwin Effect. Evol. Biol. 2012, 39, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sun, K.; Jiang, T.; Feng, J. Natural epigenetic variation in bats and its role in evolution. J. Exp. Biol. 2015, 218, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Tabansky, I.; Stern, J.N.; Pfaff, D.W. Implications of Epigenetic Variability within a Cell Population for "Cell Type" Classification. Front Behav. Neurosci. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Alonso, C.; Pérez, R.; Bazaga, P.; Medrano, M.; Herrera, C.M. Individual variation in size and fecundity is correlated with differences in global DNA cytosine methylation in the perennial herb Helleborus foetidus (Ranunculaceae). Am. J. Bot. 2014, 101, 1309–1313. [Google Scholar] [CrossRef] [PubMed]
- Oey, H.; Isbel, L.; Hickey, P.; Ebaid, B.; Whitelaw, E. Genetic and epigenetic variation among inbred mouse littermates: Identification of inter-individual differentially methylated regions. Epigenetics Chromatin. 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Lévesque, M.L.; Casey, K.F.; Szyf, M.; Ismaylova, E.; Ly, V.; Verner, M.P.; Suderman, M.; Brendgen, M.; Vitaro, F.; Dionne, G.; et al. Genome-wide DNA methylation variability in adolescent monozygotic twins followed since birth. Epigenetics 2014, 9, 1410–1421. [Google Scholar] [CrossRef] [PubMed]
- Lemmers, R.J.; Goeman, J.J.; van der Vliet, P.J.; van Nieuwenhuizen, M.P.; Balog, J.; Vos-Versteeg, M.; Camano, P.; Ramos Arroyo, M.A.; Jerico, I.; Rogers, M.T.; et al. Inter-individual differences in CpG methylation at D4Z4 correlate with clinical variability in FSHD1 and FSHD2. Hum. Mol. Genet. 2015, 24, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Mathers, J.C. Session 2: Personalised nutrition. Epigenomics: A basis for understanding individual differences? Proc. Nutr. Soc. 2008, 67, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.B.; Abdallah, B.Y.; Horne, S.D.; Liu, G.; Bremer, S.W.; Heng, H.H. Genetic and Epigenetic heterogeneity in Cancer; John Wiley and Sons Ltd.: Chichester, UK, 2011. [Google Scholar]
- Heyn, H.; Moran, S.; Hernando-Herraez, I.; Sergi Sayols, S.; Gomez, A.; Sandoval, J.; Monk, D.; Hata, K.; Marques-Bonet, T.; Wang, L.; et al. DNA methylation contributes to natural human variation. Genome Res. 2013, 23, 1363–1372. [Google Scholar] [CrossRef] [PubMed]
- Whitelaw, N.C.; Whitelaw, E. How lifetimes shape epigenotype within and across generations. Hum. Mol. Genet. 2006, 15, R131–R137. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.J.; Tricker, P.J. Epigenomic plasticity within populations: Its evolutionary significance and potential. Heredity (Edinb) 2010, 105, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Van der Graaf, A.; Wardenaar, R.; Neumann, D.A.; Taudt, A.; Shaw, R.G.; Jansen, R.C.; Schmitz, R.J.; Colomé-Tatché, M.; Johannes, F. Rate, spectrum, and evolutionary dynamics of spontaneous epimutations. Proc. Natl. Acad. Sci. USA 2015, 112, 6676–6681. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, T.W.; Frankenhuis, W.E. Adaptive explanations for sensitive windows in development. Front Zool. 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Carretero, A.; Ditrich, H.; Pérez-Aparicio, F.J.; Splechtna, H.; Ruberte, J. Development and degeneration of the arterial system in the mesonephros and metanephros of chicken embryos. Anat. Rec. 1995, 243, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Kopp, M.; Matuszewski, S. Rapid evolution of quantitative traits: Theoretical perspectives. Evol. Appl. 2014, 7, 169–191. [Google Scholar] [CrossRef] [PubMed]
- Diaz, S.A.; Viney, M. The evolution of plasticity of dauer larva developmental arrest in the nematode Caenorhabditis elegans. Ecol. Evol. 2015, 5, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, S.; Baumberger, R.; Grossniklaus, U. Epigenetic variation, inheritance, and selection in plant populations. Cold Spring Harb. Symp. Quant. Biol. 2012, 77, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Hernando-Herraez, I.; Garcia-Perez, R.; Sharp, A.J.; Marques-Bonet, T. DNA Methylation: Insights into Human Evolution. PLoS Genet. 2015, 11, e1005661. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X. Comparative epigenomics: A powerful tool to understand the evolution of DNA methylation. New Phytol. 2016, 210, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Kooke, R.; Keurentjes, J.J. Epigenetic variation contributes to environmental adaptation of Arabidopsis thaliana. Plant Signal Behav. 2015, 10, e1057368. [Google Scholar] [CrossRef] [PubMed]
- Bintu, L.; Yong, J.; Antebi1, Y.E.; McCue, K.; Kazuki, Y.; Uno, N.; Oshimura, M.; Elowitz, M.B. Dynamics of epigenetic regulation at the single-cell level. Science 2016, 351, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Massah, S.; Beischlag, T.V.; Prefontaine, G.G. Epigenetic events regulating monoallelic gene expression. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 337–358. [Google Scholar] [CrossRef] [PubMed]
- Vogt, G. Stochastic developmental variation, an epigenetic source of phenotypic diversity with far-reaching biological consequences. J. Biosci. 2015, 40, 159–204. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Form of Inheritance | |
|---|---|---|
| Genetic | Epigenetic | |
| Rapidity of Appearance in the Population | Full onset in the F1 generation | Onset in the F1 generation, unless there is a “wash in” effect, in which case, full phenotypic switching may develop over multiple generations |
| Numbers of Affected F1 Individuals in the Population | Typically one or at most a few F1 individuals, with a slow increase in the allele (if an advantageous phenotype) or a decrease (if a deleterious phenotype) over a large number of generations | Many F1 individuals receiving the same epigenetic markers |
| Longevity in the Population | Permanent, until the individual with the altered allele is eliminated by natural selection or the allele is modified by additional mutation | Transient, with continued epigenetic inheritance dependent on continued exposure to the environmental stressor, resulting in modified epigenetic markers (incorporation of epigenetically-inherited phenotypes into the epigenome is still being debated) |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burggren, W. Epigenetic Inheritance and Its Role in Evolutionary Biology: Re-Evaluation and New Perspectives. Biology 2016, 5, 24. https://doi.org/10.3390/biology5020024
Burggren W. Epigenetic Inheritance and Its Role in Evolutionary Biology: Re-Evaluation and New Perspectives. Biology. 2016; 5(2):24. https://doi.org/10.3390/biology5020024
Chicago/Turabian StyleBurggren, Warren. 2016. "Epigenetic Inheritance and Its Role in Evolutionary Biology: Re-Evaluation and New Perspectives" Biology 5, no. 2: 24. https://doi.org/10.3390/biology5020024
APA StyleBurggren, W. (2016). Epigenetic Inheritance and Its Role in Evolutionary Biology: Re-Evaluation and New Perspectives. Biology, 5(2), 24. https://doi.org/10.3390/biology5020024






