Analyzing Possible Shifts in the Climatic Niche of Pomacea canaliculata Between Native and Chinese Ranges
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
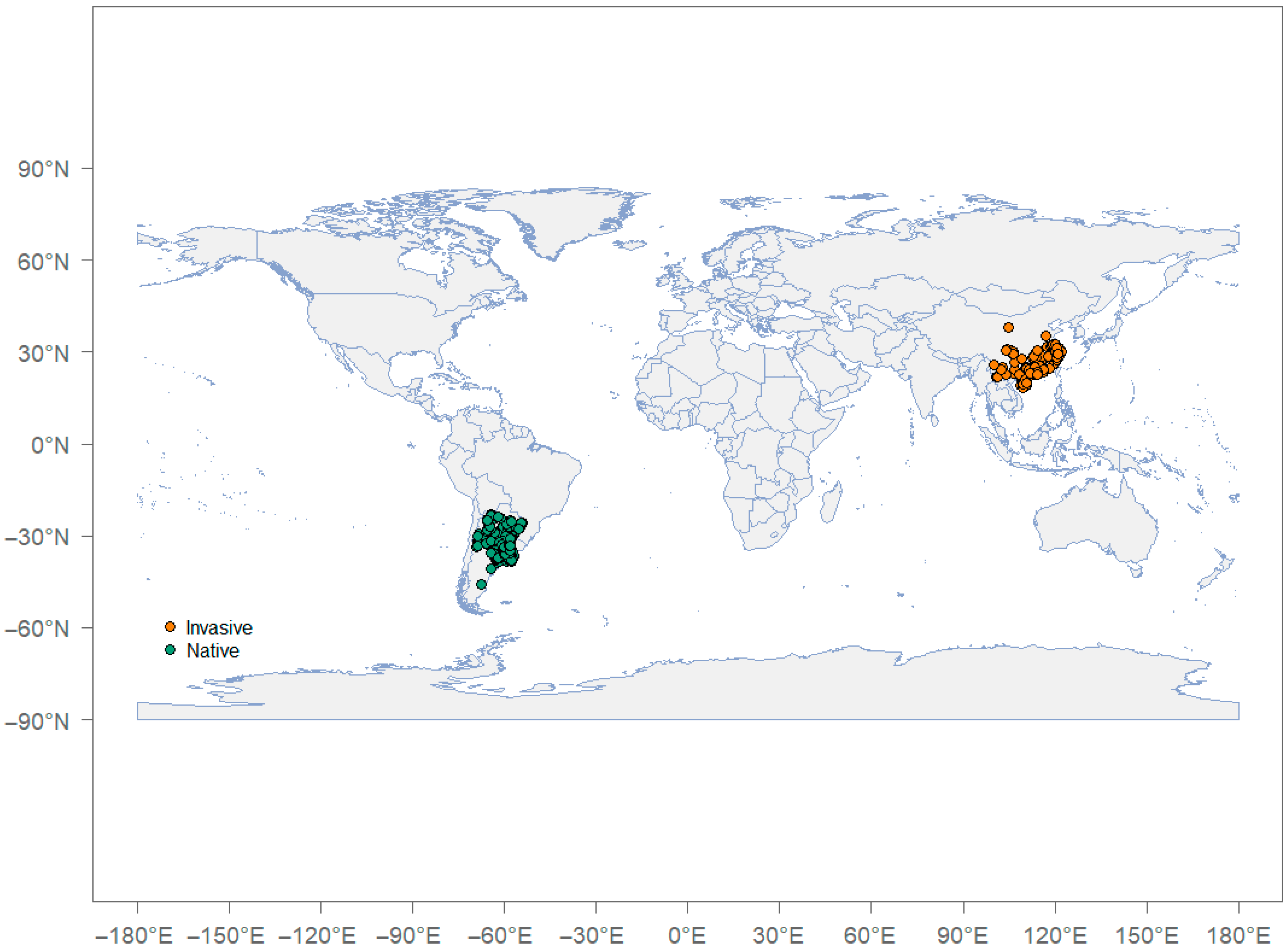
2.1. Occurrence Records (P. canaliculata Distribution Data)
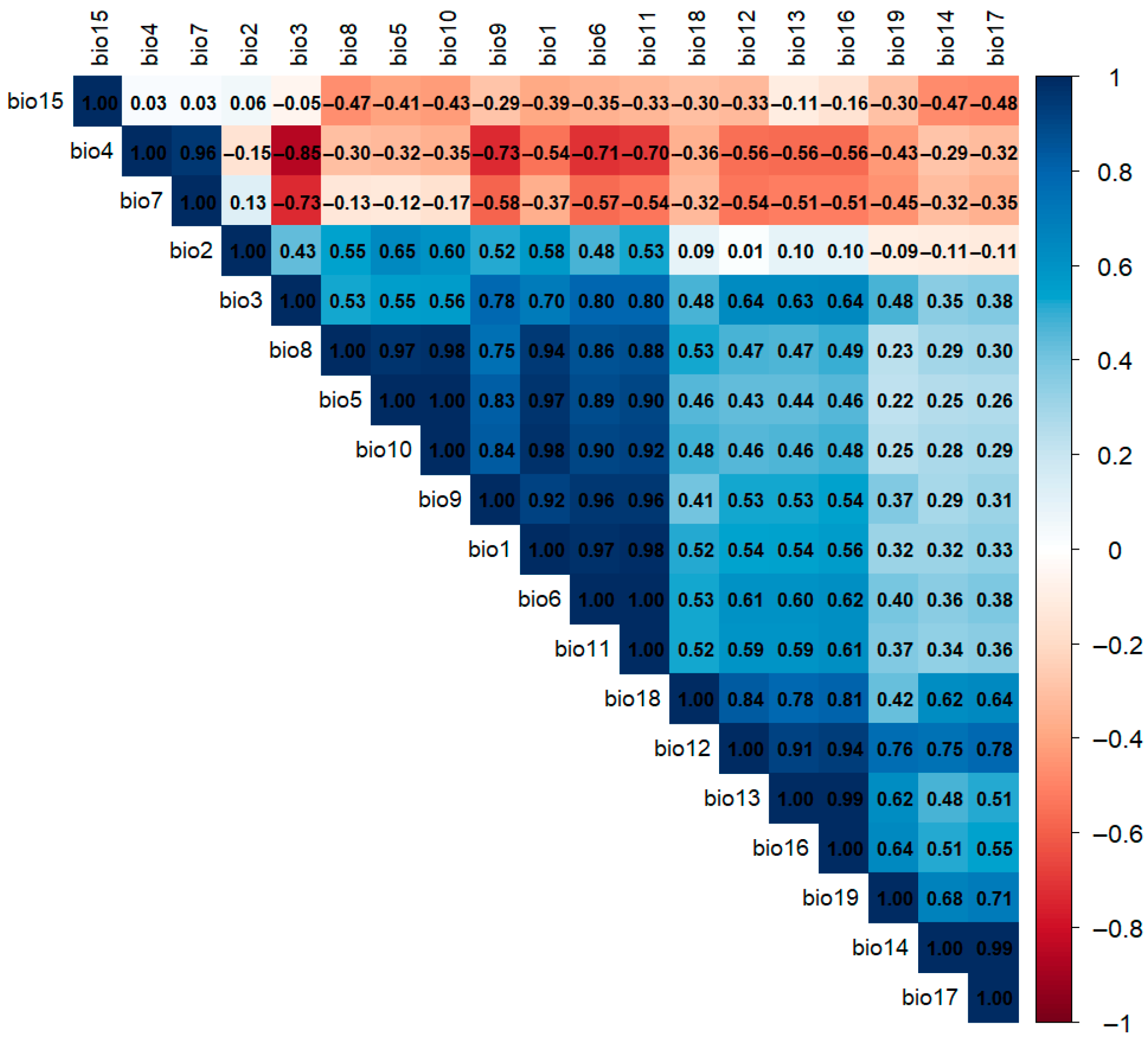
2.2. Climatic Variables
2.3. Climatic Niche Shift Analysis
3. Results
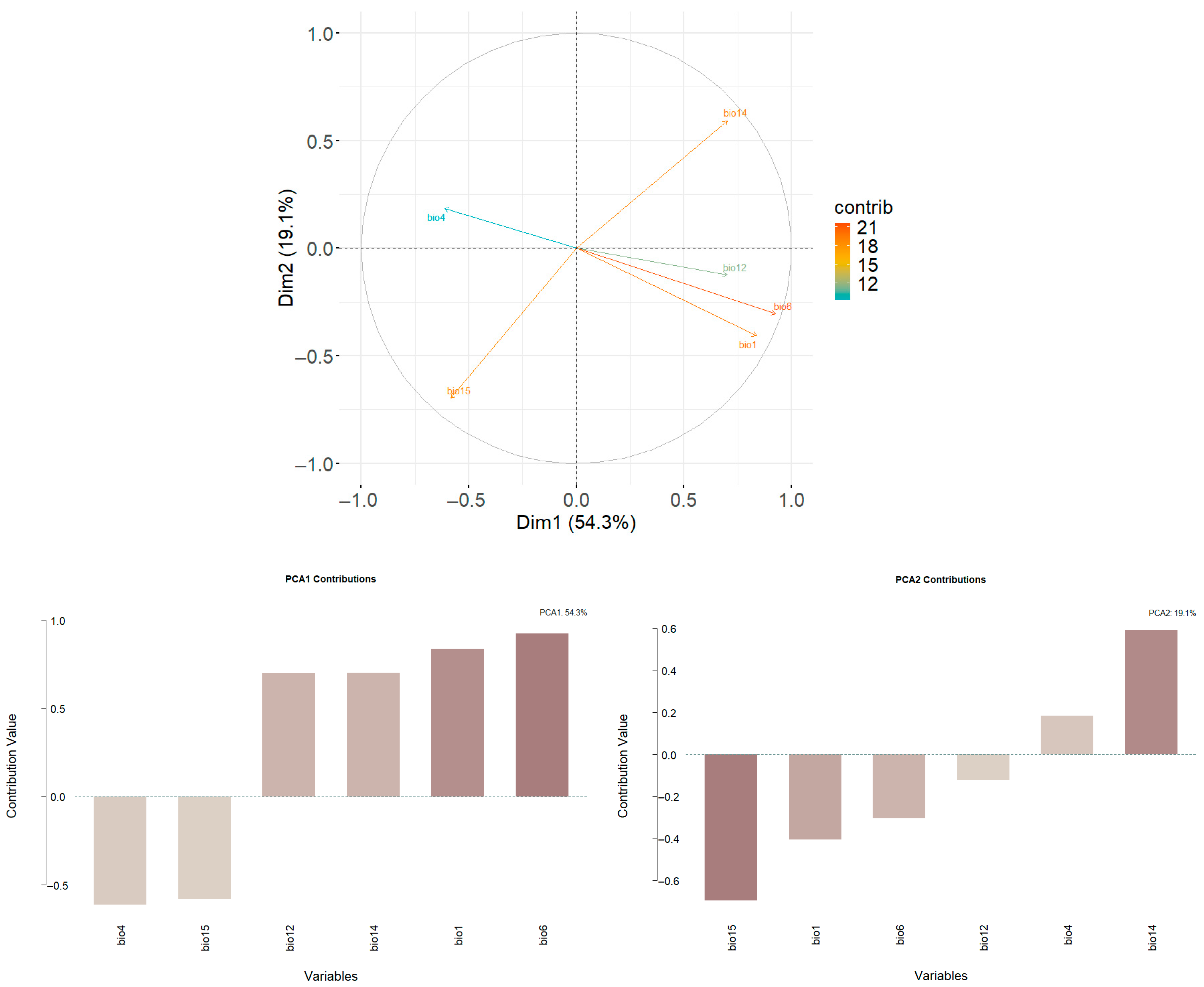
3.1. Predictors Responsible for Climatic Niche Shifts
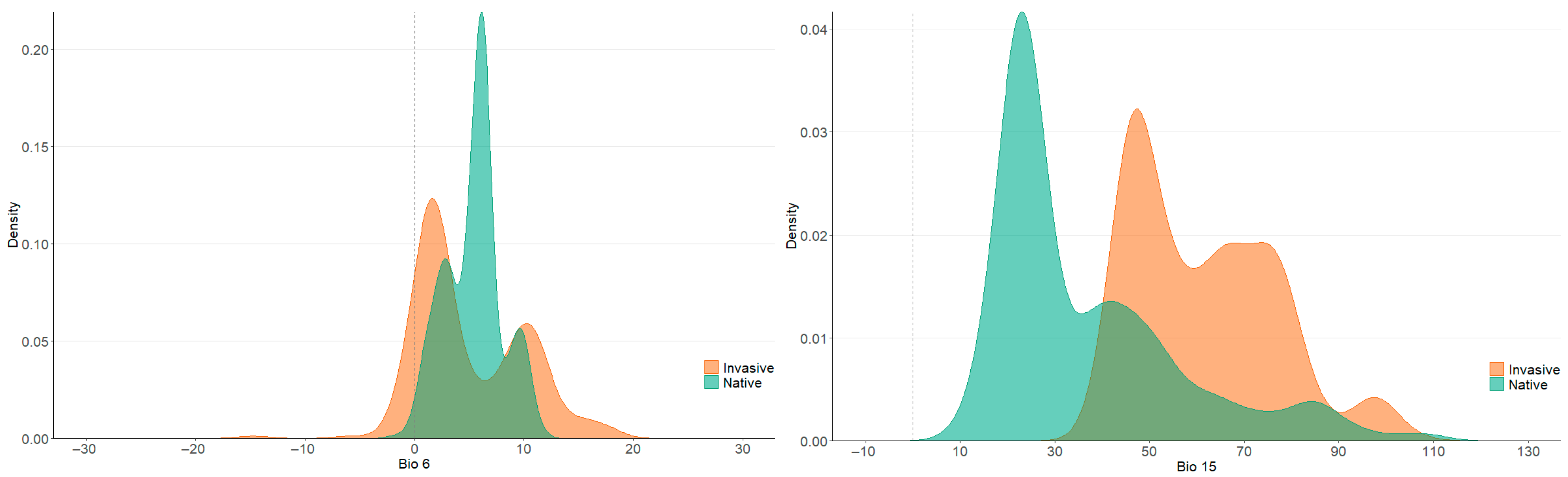
3.2. Climatic Niche Shifts
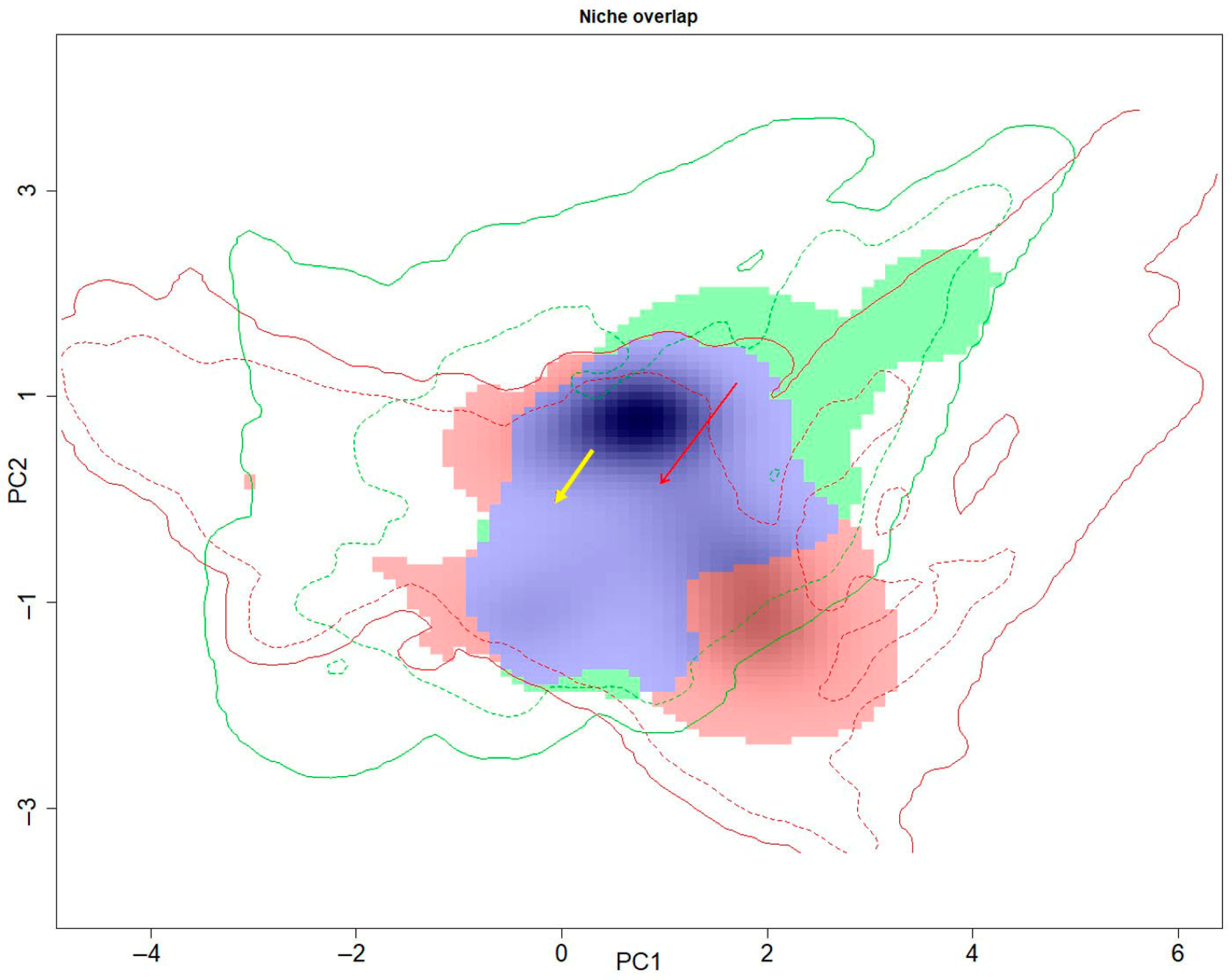
3.3. Niche Overlap
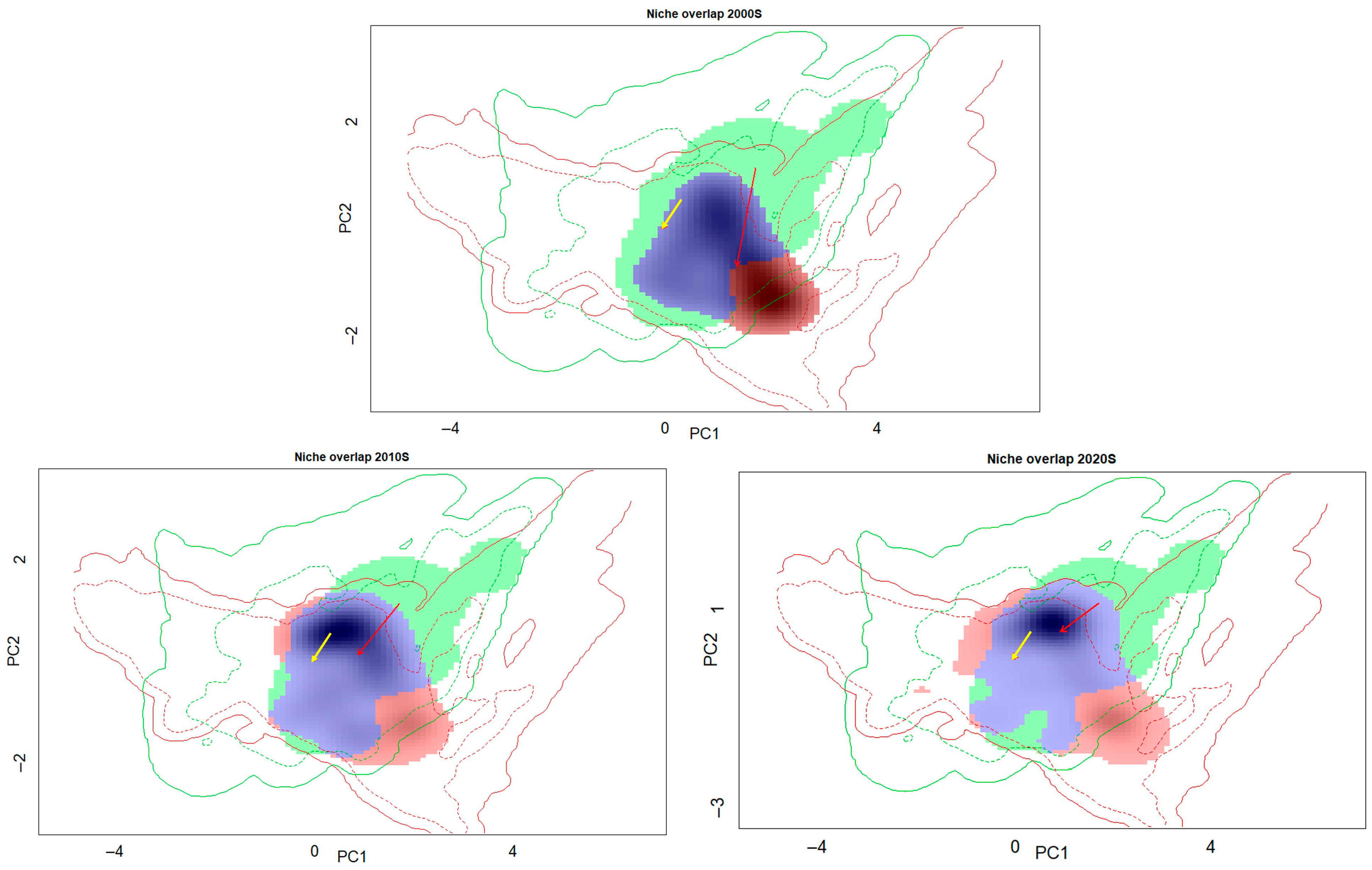
3.4. Climatic Niche Shifts Dynamics
4. Discussion
4.1. The Main Variables Driving Climatic Niche Shifts in P. canaliculata
4.2. Climatic Niche Shift Dynamics and Invasion Strategies of P. canaliculata
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. The Data Cited from GBIF
| Data Set | Link | |
| native distribution data of P. canaliculata. | iNaturalist contributors, iNaturalist (2025). iNaturalist Research-grade Observations. iNaturalist.org. Occurrence dataset https://doi.org/10.15468/ab3s5x (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=50c9509d-22c7-4a22-a47d-8c48425ef4a7&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. |
| Darrigran G, Damborenea C (2024). Zoología Invertebrados—Malacología. Version 1.2. Museo de La Plata. Occurrence dataset https://doi.org/10.15468/3h7xpk (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=93efdc85-10ca-448c-a3e3-01bce24479c9&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Seuffert M E, Martín P R (2024). Global records of the invasive freshwater apple snail Pomacea canaliculata (Lamarck, 1822). Version 1.3. Instituto de Ciencias Biológicas y Biomédicas del Sur (INBIOSUR). Occurrence dataset https://doi.org/10.15468/j4tbns (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=26820b90-367e-4029-b70c-db601c591128&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Tablado A, Rodríguez D (2021). Museo Argentino de Ciencias Naturales “Bernardino Rivadavia” (MACN). Invertebrates National Collection (MACNIn). Museo Argentino de Ciencias Naturales. Occurrence dataset https://doi.org/10.15468/uuz636 (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=6197c830-d9c7-11de-b793-b8a03c50a862&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Orrell T, Informatics and Data Science Center—Digital Stewardship (2025). NMNH Extant Specimen Records (USNM, US). Version 1.96. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/hnhrg3 (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=821cc27a-e3bb-4bc5-ac34-89ada245069d&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Carnegie Museums (2025). Carnegie Museum of Natural History—Mollusks. Occurrence dataset https://doi.org/10.15468/4rfubm (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=07ae2aa8-5031-4312-b26e-84a5c753daac&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Harvard University M, Morris P J (2025). Museum of Comparative Zoology, Harvard University. Version 162.476. Museum of Comparative Zoology, Harvard University. Occurrence dataset https://doi.org/10.15468/p5rupv (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=4bfac3ea-8763-4f4b-a71a-76a6f5f243d3&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| SysTax. SysTax—Zoological Collections. Occurrence dataset https://doi.org/10.15468/zyqkbl (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=7d8ed137-1d30-42f1-8b78-12a4957e4690&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Denver Museum of Nature & Science Marine Invertebrate Collection https://doi.org/10.15468/lyw1fq (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=5a6977c1-ece6-44b8-b71d-43cb1d4e0919&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| The International Barcode of Life Consortium (2024). International Barcode of Life project (iBOL). Occurrence dataset https://doi.org/10.15468/inygc6 (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=AR&dataset_key=040c5662-da76-4782-a48e-cdea1892d14c&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| invasive distribution (in China) data of P. canaliculata | European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk (2025). INSDC Sequences. Version 1.148. European Nucleotide Archive (EMBL-EBI). Occurrence dataset https://doi.org/10.15468/sbmztx (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=d8cd16ba-bb74-4420-821e-083f2bac17c2&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. |
| iNaturalist contributors, iNaturalist (2025). iNaturalist Research-grade Observations. iNaturalist.org. Occurrence dataset https://doi.org/10.15468/ab3s5x (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=50c9509d-22c7-4a22-a47d-8c48425ef4a7&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Seuffert M E, Martín P R (2024). Global records of the invasive freshwater apple snail Pomacea canaliculata (Lamarck, 1822). Version 1.3. Instituto de Ciencias Biológicas y Biomédicas del Sur (INBIOSUR). Occurrence dataset https://doi.org/10.15468/j4tbns (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=26820b90-367e-4029-b70c-db601c591128&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| The International Barcode of Life Consortium (2024). International Barcode of Life project (iBOL). Occurrence dataset https://doi.org/10.15468/inygc6 (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=040c5662-da76-4782-a48e-cdea1892d14c&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Zhou J, Wong L (2025). CBCGDF CCAfa Volunteer Observation Archive. Version 1.198. China Biodiversity Conservation and Green Development Foundation. Occurrence dataset https://doi.org/10.15468/wxze8b (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=1b26c417-366a-4eb0-a212-6ee8035d83fb&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Wong L, Chen S, Chen S (2025). CBCGDF-Green-Youth. Version 1.42. China Biodiversity Conservation and Green Development Foundation. Occurrence dataset https://doi.org/10.15468/8dteut (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=98d8d571-f83f-4076-8a41-86056198a969&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| naturgucker.de. NABU| naturgucker. Occurrence dataset https://doi.org/10.15468/uc1apo (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=6ac3f774-d9fb-4796-b3e9-92bf6c81c084&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. | |
| Illinois Natural History Survey (2025). Illinois Natural History Survey- Mollusk Collection. Occurrence dataset https://doi.org/10.15468/cu4scd (accessed on 2 August 2025) via GBIF.org. | https://www.gbif.org/occurrence/search?country=CN&dataset_key=763f9aee-060c-4d71-a538-16c383e53a4d&taxon_key=2292582&occurrence_status=present (accessed on 2 August 2025) via GBIF.org. |
References
- Pyšek, P.; Hulme, P.E.; Simberloff, D.; Bacher, S.; Blackburn, T.M.; Carlton, J.T.; Richardson, D.M. Scientists’ Warning on Invasive Alien Species. Biol. Rev. 2020, 95, 1511–1534. [Google Scholar] [CrossRef] [PubMed]
- Diagne, C.; Leroy, B.; Vaissière, A.C.; Gozlan, R.E.; Roiz, D.; Jarić, I.; Salles, J.M.; Bradshaw, C.J.A.; Courchamp, F. High and Rising Economic Costs of Biological Invasions Worldwide. Nature 2021, 592, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Simberloff, D.; Martin, J.-L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; et al. Impacts of Biological Invasions: What’s What and the Way Forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef]
- Gonçalves da Silva, A.; Kolokotronis, S.-O.; Wharton, D. Modeling the Eradication of Invasive Mammals Using the Sterile Male Technique. Biol. Invasions 2009, 12, 751–759. [Google Scholar] [CrossRef]
- Simberloff, D. How Much Information on Population Biology Is Needed to Manage Introduced Species? Conserv. Biol. 2003, 17, 83–92. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater Biodiversity: Importance, Threats, Status and Conservation Challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef]
- Haubrock, P.J.; Cuthbert, R.N.; Ricciardi, A.; Diagne, C.; Courchamp, F. Economic Costs of Invasive Bivalves in Freshwater Ecosystems. Divers. Distrib. 2022, 28, 1010–1021. [Google Scholar] [CrossRef]
- Schuler, M.S.; Hintz, W.D.; Jones, D.K.; Mattes, B.M.; Stoler, A.B.; Relyea, R.A. The Effects of Nutrient Enrichment and Invasive Mollusks on Freshwater Environments. Ecosphere 2020, 11, e03196. [Google Scholar] [CrossRef]
- Pablo, R.M. Interpopulation Variation in Life-History Traits of Pomacea canaliculata (Gastropoda: Ampullariidae) in Southwestern Buenos Aires Province, Argentina. Malacologia 2002, 44, 153–163. [Google Scholar]
- Mochida, O.S.A.M.U. Spread of Freshwater Pomacea snails (Pilidae, Mollusca) from Argentina to Asia. Micronesica 1991, 3, 51–62. [Google Scholar]
- Joshi, R.C.; Robert, H.C.; Leocadio, S.S. Biology and Management of Invasive Apple snails; Philippine Rice Research Institute: Muñoz, Philippine, 2017. [Google Scholar]
- Carlsson, N.O.L.; Brönmark, C.; Hansson, L.-A. Invading Herbivory: The Golden Apple snail Alters Ecosystem Functioning in Asian Wetlands. Ecology 2004, 85, 1575–1580. [Google Scholar] [CrossRef]
- Horgan, F.G.; Stuart, A.M.; Kudavidanage, E.P. Impact of Invasive Apple snails on the Functioning and Services of Natural and Managed Wetlands. Acta Oecologica 2014, 54, 90–100. [Google Scholar] [CrossRef]
- Lv, S.; Zhang, Y.; Steinmani, P.; Yang, G.-J.; Yang, K.; Zhou, X.-N.; Utzinger, J. The Emergence of Angiostrongyliasis in the People’s Republic of China: The Interplay between Invasive Snails, Climate Change and Transmission Dynamics. Freshw. Biol. 2011, 56, 717–734. [Google Scholar] [CrossRef]
- Matsukura, K.; Izumi, Y.; Yoshida, K.; Wada, T. Cold Tolerance of Invasive Freshwater Snails, Pomacea canaliculata, P. Maculata, and Their Hybrids Helps Explain Their Different Distributions. Freshw. Biol. 2015, 61, 80–87. [Google Scholar] [CrossRef]
- Hayes, K.A.; Joshi, R.C.; Thiengo, S.C.; Cowie, R.H. Out of South America: Multiple Origins of Non-Native Apple snails in Asia. Divers. Distrib. 2008, 14, 701–712. [Google Scholar] [CrossRef]
- Lowe, S.; Browne, M.; Boudjelas, S.; De Poorter, M. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database; Invasive Species Specialist Group: Auckland, New Zealand, 2000; Volume 12, p. 12. [Google Scholar]
- Lv, S.; Zhang, Y.; Liu, H.-X.; Hu, L.; Liu, Q.; Wei, F.-R.; Guo, Y.-H.; Steinmann, P.; Hu, W.; Zhou, X.-N.; et al. Phylogenetic Evidence for Multiple and Secondary Introductions of Invasive Snails: Pomacea Species in the People’s Republic of China. Divers. Distrib. 2012, 19, 147–156. [Google Scholar] [CrossRef]
- Yang, Q.-Q.; Liu, S.-W.; He, C.; Yu, X.-P. Distribution and the Origin of Invasive Apple snails, Pomacea canaliculata and P. maculata (Gastropoda: Ampullariidae) in China. Sci. Rep. 2018, 8, 1185. [Google Scholar] [CrossRef] [PubMed]
- Seuffert, M.E.; Martín, P. Exceeding Its Own Limits: Range Expansion in Argentina of the Globally Invasive Apple snail Pomacea canaliculata. Hydrobiologia 2020, 848, 385–401. [Google Scholar] [CrossRef]
- Lei, J.; Chen, L.; Li, H. Using Ensemble Forecasting to Examine How Climate Change Promotes Worldwide Invasion of the Golden Apple snail (Pomacea canaliculata). Environ. Monit. Assess. 2017, 189, 404. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Cao, R.; Gong, X.; Feng, J. Large Shifts of Niche and Range in the Golden Apple snail (Pomacea canaliculata), an Aquatic Invasive Species. Ecosphere 2023, 14, e4391. [Google Scholar] [CrossRef]
- Davies, S.J.; Hill, M.P.; McGeoch, M.A.; Clusella-Trullas, S. Niche Shift and Resource Supplementation Facilitate an Amphibian Range Expansion. Divers. Distrib. 2018, 25, 154–165. [Google Scholar] [CrossRef]
- MacDougall, A.S.; Gilbert, B.; Levine, J.M. Plant Invasions and the Niche. J. Ecol. 2009, 97, 609–615. [Google Scholar] [CrossRef]
- Pearman, P.B.; Guisan, A.; Broennimann, O.; Randin, C.F. Niche Dynamics in Space and Time. Trends Ecol. Evol. 2008, 23, 149–158. [Google Scholar] [CrossRef]
- Petitpierre, B.; Kueffer, C.; Broennimann, O.; Randin, C.; Daehler, C.; Guisan, A. Climatic Niche Shifts Are Rare among Terrestrial Plant Invaders. Science 2012, 335, 1344–1348. [Google Scholar] [CrossRef]
- Guisan, A.; Petitpierre, B.; Broennimann, O.; Daehler, C.; Kueffer, C. Unifying Niche Shift Studies: Insights from Biological Invasions. Trends Ecol. Evol. 2014, 29, 260–269. [Google Scholar] [CrossRef]
- Pili, A.N.; Tingley, R.; Sy, E.Y.; Diesmos, M.L.L.; Diesmos, A.C. Niche Shifts and Environmental Non-Equilibrium Undermine the Usefulness of Ecological Niche Models for Invasion Risk Assessments. Sci. Rep. 2020, 10, 7972. [Google Scholar] [CrossRef] [PubMed]
- Bates, O.K.; Ollier, S.; Bertelsmeier, C. Smaller Climatic Niche Shifts in Invasive than Non-Invasive Alien Ant Species. Nat. Commun. 2020, 11, 5213. [Google Scholar] [CrossRef] [PubMed]
- Escoriza, D.; Ben Hassine, J.; Boix, D. Factors Regulating the Invasive Success of an Alien Frog: A Comparison of the Ecology of the Native and Alien Populations. Hydrobiologia 2014, 730, 127–138. [Google Scholar] [CrossRef]
- Wan, J.-Z.; Wang, C.-J.; Tan, J.; Yu, F.-H. Climatic Niche Divergence and Habitat Suitability of Eight Alien Invasive Weeds in China under Climate Change. Ecol. Evol. 2017, 7, 1541–1552. [Google Scholar] [CrossRef]
- Seuffert, M.E.; Martín, P.R. Global Distribution of the Invasive Apple snail Pomacea canaliculata: Analyzing Possible Shifts in Climatic Niche between Native and Invaded Ranges and Future Spread. Aquat. Sci. 2024, 86, 17. [Google Scholar] [CrossRef]
- Zhang, J.E.; Guo, J.; Zhao, B.L.; Qin, Z. Full-Spectrum Integrated Control Technology System Construction and Utilization of Pomacea canaliculata. Chin. J. Ecol. 2019, 38, 3831–3838. [Google Scholar] [CrossRef]
- Dao, X.Q.; Sun, T.; Deng, C.M.; Luo, X.R.; Zhou, W.; Yang, T.Y. Occurrence Patterns and Control Measures of Pomacea canaliculata in Yunnan Province. China Plant Prot. 2014, 34, 32–35. [Google Scholar]
- Du, L.N.; Davies, J.; Chen, X.Y.; Cui, G.H.; Yang, J.X. First Record of the Invasive Golden Apple snail (Pomacea canaliculata) in the Dianchi Lake Basin, China. Zool. Res. 2007, 28, 325–328. (In English) [Google Scholar]
- Huang, S.Z.; Wang, T.X. Preliminary Study on Biological Characteristics of Pomacea canaliculata in Yuping County, Guizhou Province. Beijing Agric. 2015, 14, 219. [Google Scholar]
- Ji, X.M.; Wang, A.X.; Fang, L.C.; Li, C.L.; Xu, L.; Yang, S.K.; Liu, X.B.; Zhong, L.; Liu, Y.M. Investigation on the Current Distribution of Pomacea canaliculata in the Lower Yangtze River Basin, China. Hubei Agric. Sci. 2020, 59, 111–116. [Google Scholar] [CrossRef]
- Liu, Y.F.; Li, F.; Li, Y.F.; Liu, W.H.; Zeng, Q.G.; Su, W.J. Distribution Status, Damage, and Diffusion Risk Warning of Pomacea canaliculata in Hunan Province. Acta Hydrobiol. Sin. 2011, 35, 1067–1071. [Google Scholar] [CrossRef]
- Wei, R.; Wu, C.D.; Xie, H.F.; Gong, W.R.; Du, Y.Z. Risk Analysis of the Invasive Pest Pomacea canaliculata in Jiangsu Province. J. Yangzhou Univ. (Agric. Life Sci. Ed.) 2021, 42, 119–124. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Zhu, L.X.; Hu, J.X.; Zhang, J.E. Distribution, Damage, and Control of Pomacea canaliculata in Different Regions of Fujian Province. Jiangsu Agric. Sci. 2014, 42, 136–140. [Google Scholar] [CrossRef]
- Dong, C.L. Biological and Ecological Characteristics of Pomacea canaliculata and Its Distribution and Harm Status in Guangxi. Master’s Thesis, Guangxi Normal University, Guilin, China, 2006. [Google Scholar]
- Li, Y.S.; Zeng, Y.L.; Xiang, Z.Q.; Wu, Q.; Yang, W. Occurrence and Control Measures of Pomacea canaliculata in Paddy Fields of Pingtang County. Cultiv. Plant. 2007, 4, 59. [Google Scholar]
- Liu, Y.M.; Li, C.L.; Jin, L.; Pei, X.; Chen, X.; Wang, A.; Ji, X.; Zhong, L. Investigation on the Northernmost Distribution of Pomacea canaliculata in the Upper Yangtze River Basin. Yangtze Veg. 2020, 12, 53–57. [Google Scholar]
- Qi, Q.Y. Damage Situation and Control Strategies of Four Invasive Alien Species in Zhenning County. South China Agric. 2023, 17, 224–226+230. [Google Scholar] [CrossRef]
- Tian, L.; Li, F.B.; He, W.F.; Cheng, X.L.; Xu, Y.B.; Li, M. Research Progress on Invasion Status and Control Methods of Pomacea canaliculata in Zhejiang Province. Mod. Agric. Sci. Technol. 2022, 5, 77–85. [Google Scholar]
- Zhou, Z.X.; Chen, X. Occurrence and Control of Pomacea canaliculata in Sandu County. Plant Dr. 2005, 6, 6. [Google Scholar]
- Aiello-Lammens, M.E.; Boria, R.A.; Radosavljevic, A.; Vilela, B.; Anderson, R.P. SpThin: An R Package for Spatial Thinning of Species Occurrence Records for Use in Ecological Niche Models. Ecography 2015, 38, 541–545. [Google Scholar] [CrossRef]
- Broennimann, O.; Fitzpatrick, M.C.; Pearman, P.B.; Petitpierre, B.; Pellissier, L.; Yoccoz, N.G.; Thuiller, W.; Fortin, M.-J.; Randin, C.; Zimmermann, N.E.; et al. Measuring Ecological Niche Overlap from Occurrence and Spatial Environmental Data. Glob. Ecol. Biogeogr. 2011, 21, 481–497. [Google Scholar] [CrossRef]
- Warren, D.L.; Glor, R.E.; Turelli, M. Environmental niche equivalency versus conservatism: Quantitative approaches to niche evolution. Evolution 2008, 62, 2868–2883. [Google Scholar] [CrossRef]
- Rödder, D.; Engler, J.O. Quantitative Metrics of Overlaps in Grinnellian Niches: Advances and Possible Drawbacks. Glob. Ecol. Biogeogr. 2011, 20, 915–927. [Google Scholar] [CrossRef]
- Di Cola, V.; Broennimann, O.; Petitpierre, B.; Breiner, F.T.; D’Amen, M.; Randin, C.; Engler, R.; Pottier, J.; Pio, D.; Dubuis, A.; et al. Ecospat: An R Package to Support Spatial Analyses and Modeling of Species Niches and Distributions. Ecography 2017, 40, 774–787. [Google Scholar] [CrossRef]
- Joshi, R.C.; Xavier, V.P. The Rice Apple snail in Spain: A Review. Int. Pest Control 2017, 59, 106. [Google Scholar]
- Keane, R.M.; Crawley, M.J. Exotic Plant Invasions and the Enemy Release Hypothesis. Trends Ecol. Evol. 2002, 17, 164–170. [Google Scholar] [CrossRef]
- Joshi, R.C.; Leocadio, S.S. Global Advances in Ecology and Management of Golden Apple snails; Philippine Rice Research Institute: Muñoz, Philippine, 2006. [Google Scholar]
- Sakai, A.K.; Allendorf, F.W.; Holt, J.S.; Lodge, D.M.; Molofsky, J.; With, K.A.; Weller, S.G. The Population Biology of Invasive Species. Annu. Rev. Ecol. Syst. 2001, 32, 305–332. [Google Scholar] [CrossRef]
- Davidson, A.M.; Jennions, M.; Nicotra, A.B. Do Invasive Species Show Higher Phenotypic Plasticity than Native Species And, If So, Is It Adaptive? A Meta-Analysis. Ecol. Lett. 2011, 14, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Colautti, R.I.; MacIsaac, H.J. A Neutral Terminology to Define “Invasive” Species. Divers. Distrib. 2004, 10, 135–141. [Google Scholar] [CrossRef]
- Johnson, D.M.; Liebhold, A.M.; Tobin, P.C.; Bjørnstad, O.N. Allee Effects and Pulsed Invasion by the Gypsy Moth. Nature 2006, 444, 361–363. [Google Scholar] [CrossRef]
- Arim, M.; Abades, S.R.; Neill, P.E.; Lima, M.; Marquet, P.A. Spread dynamics of invasive species. Proc. Natl. Acad. Sci. USA 2006, 103, 374–378. [Google Scholar] [CrossRef]
- Prentis, P.J.; Wilson, J.R.U.; Dormontt, E.E.; Richardson, D.M.; Lowe, A.J. Adaptive Evolution in Invasive Species. Trends Plant Sci. 2008, 13, 288–294. [Google Scholar] [CrossRef] [PubMed]
| Bio 1 | annual mean temperature | Bio 11 | mean temperature of the coldest quarter |
| Bio 2 | mean diurnal temperature range | Bio 12 | annual precipitation |
| Bio 3 | isothermally | Bio 13 | precipitation of the wettest month |
| Bio 4 | temperature seasonality | Bio 14 | precipitation of the driest month |
| Bio 5 | maximum temperature of the warmest month | Bio 15 | precipitation seasonality |
| Bio 6 | minimum temperature of the coldest month | Bio 16 | precipitation of the wettest quarter |
| Bio 7 | temperature of annual range | Bio 17 | precipitation of the driest quarter |
| Bio 8 | mean temperature of the wettest quarter | Bio 18 | precipitation of the warmest quarter |
| Bio 9 | mean temperature of the driest quarter | Bio 19 | precipitation of the coldest quarter |
| Bio 10 | mean temperature of the warmest quarter |
| Schoener’s D | Expansion | Stability | Unfilling | |
|---|---|---|---|---|
| All | 0.0467 | 0.1975 | 0.8024 | 0.1153 |
| 2000s | 0.0175 | 0.2511 | 0.7488 | 0.5315 |
| 2010s | 0.0285 | 0.1181 | 0.8818 | 0.3313 |
| 2020s | 0.0418 | 0.1514 | 0.8485 | 0.1916 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, R.; Gao, Y.; Wang, R.; Liu, S.; Yang, Q.; Li, Y.; Lin, L. Analyzing Possible Shifts in the Climatic Niche of Pomacea canaliculata Between Native and Chinese Ranges. Biology 2025, 14, 1127. https://doi.org/10.3390/biology14091127
Zhang R, Gao Y, Wang R, Liu S, Yang Q, Li Y, Lin L. Analyzing Possible Shifts in the Climatic Niche of Pomacea canaliculata Between Native and Chinese Ranges. Biology. 2025; 14(9):1127. https://doi.org/10.3390/biology14091127
Chicago/Turabian StyleZhang, Ran, Yue Gao, Rui Wang, Shigang Liu, Qianqian Yang, Yuan Li, and Longshan Lin. 2025. "Analyzing Possible Shifts in the Climatic Niche of Pomacea canaliculata Between Native and Chinese Ranges" Biology 14, no. 9: 1127. https://doi.org/10.3390/biology14091127
APA StyleZhang, R., Gao, Y., Wang, R., Liu, S., Yang, Q., Li, Y., & Lin, L. (2025). Analyzing Possible Shifts in the Climatic Niche of Pomacea canaliculata Between Native and Chinese Ranges. Biology, 14(9), 1127. https://doi.org/10.3390/biology14091127






