New Record of Dendronephthya sp. (Family: Nephtheidae) from Mediterranean Israel: Evidence for Tropicalization?
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
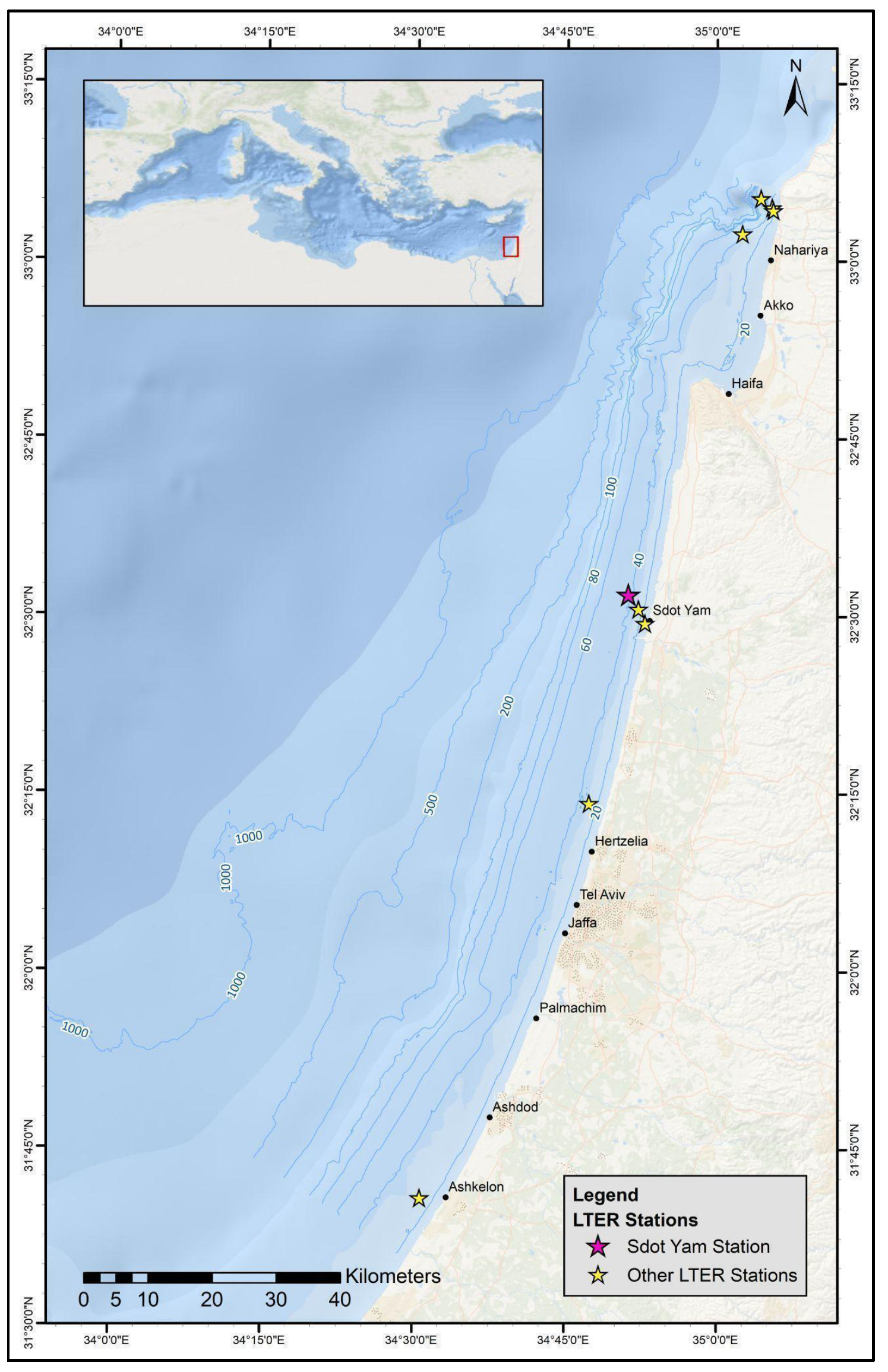
2.1. Study Site and Sample Collection
2.2. DNA Extraction
2.3. Sclerite Morphological Analysis
3. Results
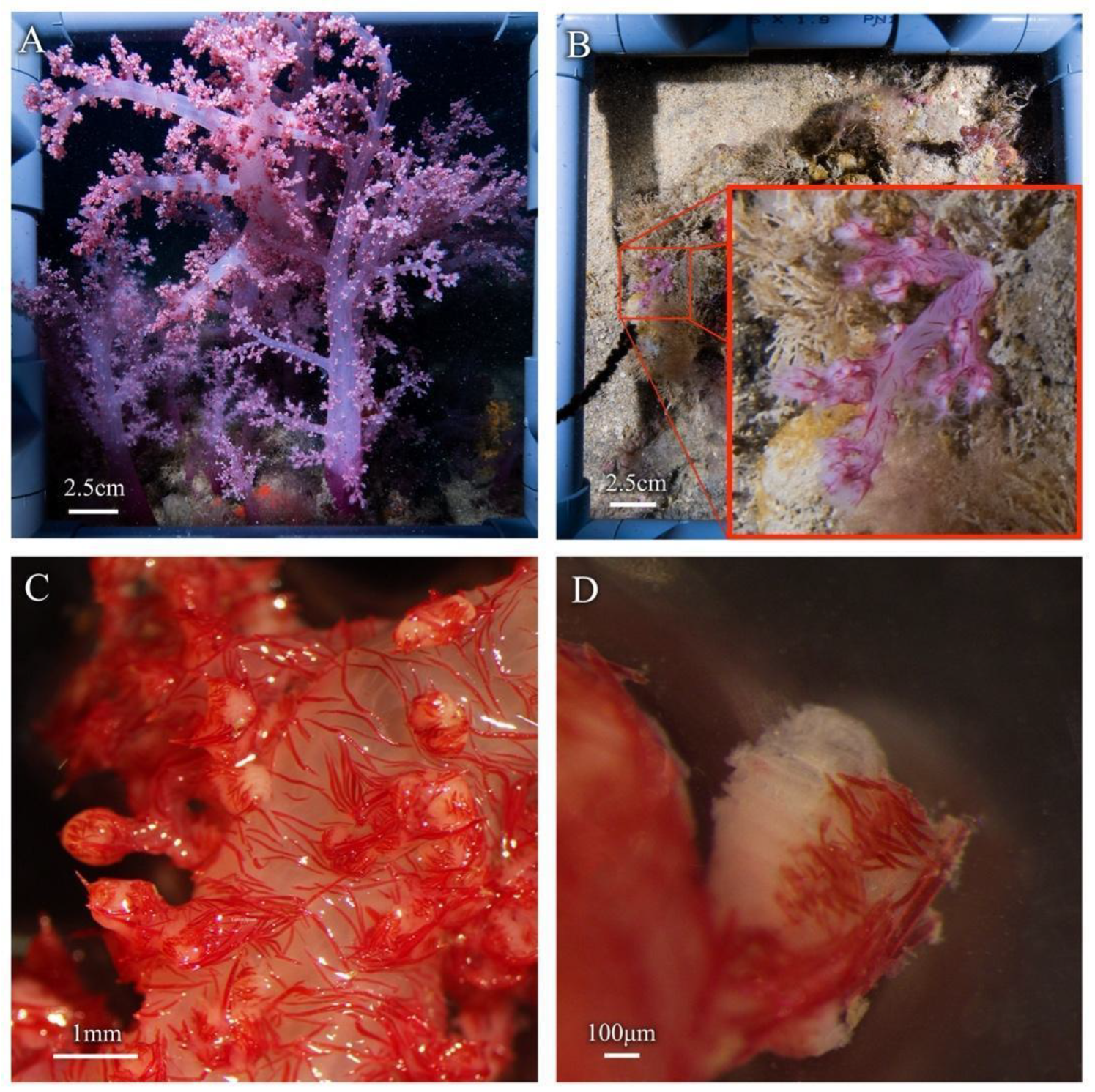
3.1. Macro Morphological Analysis
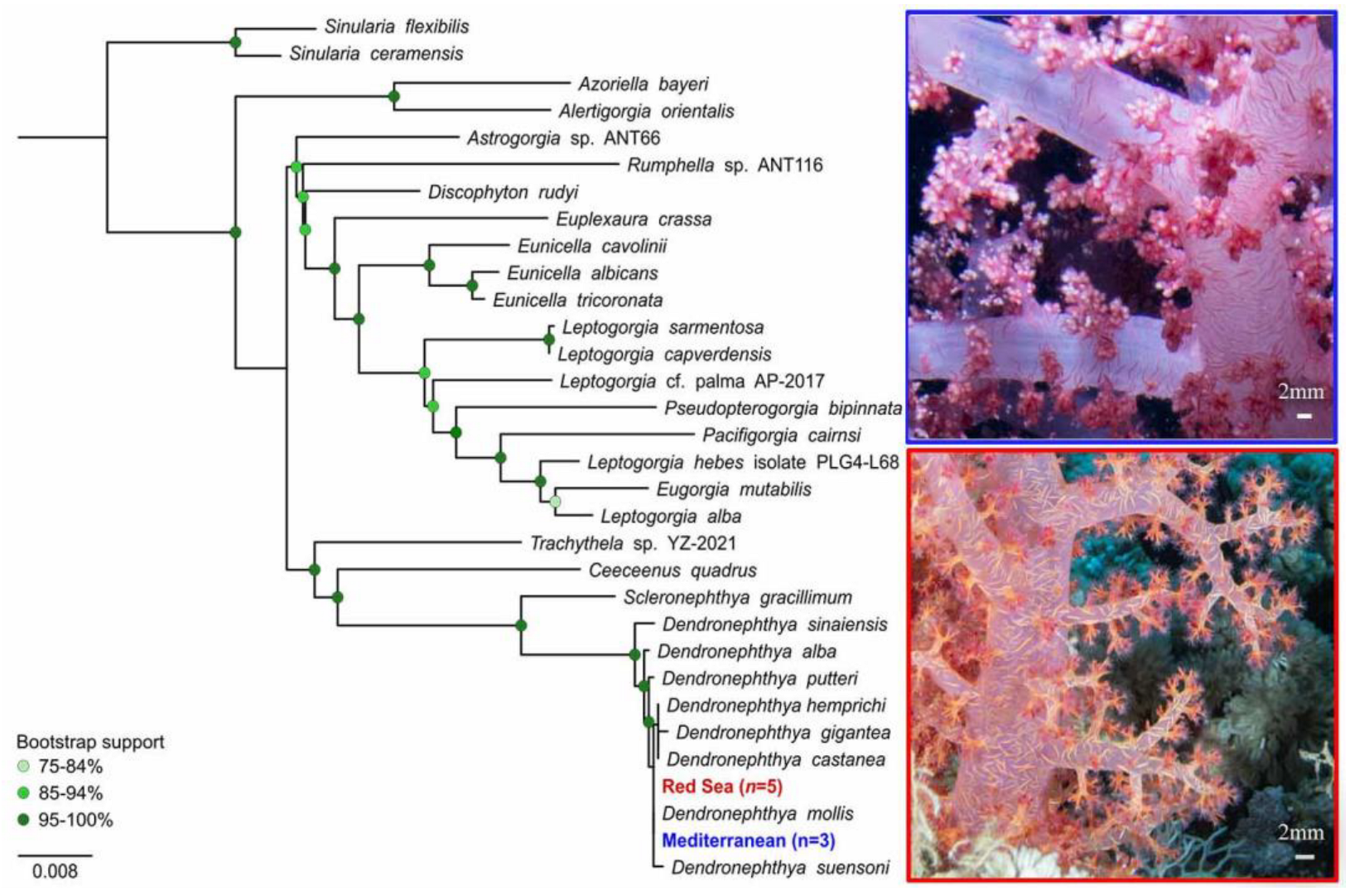
3.2. Molecular Analysis
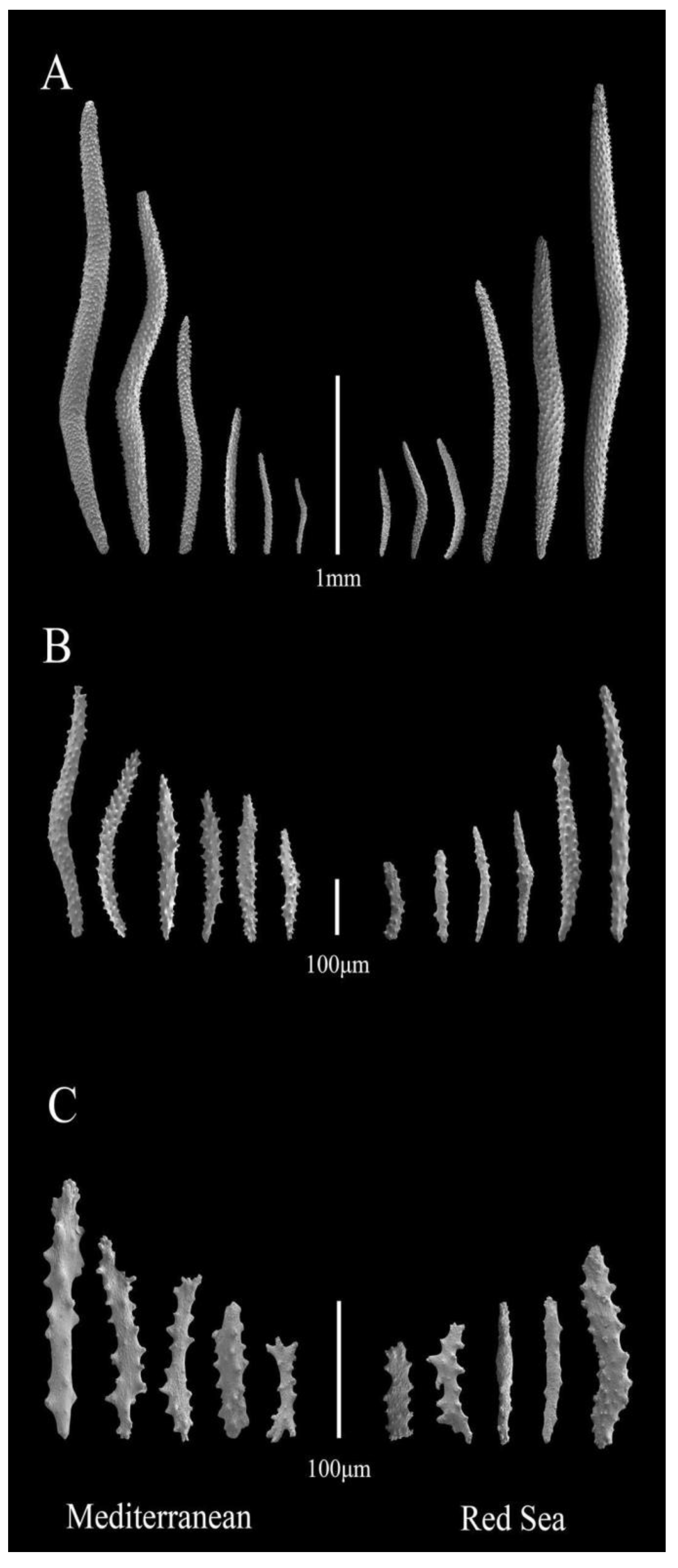
3.3. Micro-Morphological Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Conversi, A.; Dakos, V.; Gårdmark, A.; Ling, S.; Folke, C.; Mumby, P.J.; Greene, C.; Edwards, M.; Blenckner, T.; Casini, M.; et al. A Holistic View of Marine Regime Shifts. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20130279. [Google Scholar] [CrossRef]
- Ehrenfeld, J.G. Ecosystem Consequences of Biological Invasions. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 59–80. [Google Scholar] [CrossRef]
- Theoharides, K.A.; Dukes, J.S. Plant Invasion across Space and Time: Factors Affecting Nonindigenous Species Success during Four Stages of Invasion. New Phytol. 2007, 176, 256–273. [Google Scholar] [CrossRef] [PubMed]
- Capel, K.C.C.; Toonen, R.J.; Rachid, C.T.C.C.; Creed, J.C.; Kitahara, M.V.; Forsman, Z.; Zilberberg, C. Clone Wars: Asexual Reproduction Dominates in the Invasive Range of Tubastraea spp. (Anthozoa: Scleractinia) in the South-Atlantic Ocean. PeerJ 2017, 5, e3873. [Google Scholar] [CrossRef] [PubMed]
- Hoeksema, B.W.; Benzoni, F. Multispecies Aggregations of Mushroom Corals in the Gambier Islands, French Polynesia. Coral Reefs 2013, 32, 1041. [Google Scholar] [CrossRef]
- Hoeksema, B.W. Impact of Budding on Free-Living Corals at East Kalimantan, Indonesia. Coral Reefs 2004, 23, 492. [Google Scholar] [CrossRef]
- Hoeksema, B.W.; Gittenberger, A. High Densities of Mushroom Coral Fragments at West Halmahera, Indonesia. Coral Reefs 2010, 29, 691. [Google Scholar] [CrossRef][Green Version]
- Hoeksema, B.W.; Giyanto; Suharsono. The Role of Maximum Shelf Depth versus Distance from Shore in Explaining a Diversity Gradient of Mushroom Corals (Fungiidae) off Jakarta. Diversity 2019, 11, 46. [Google Scholar] [CrossRef]
- Bailey, S.A. An Overview of Thirty Years of Research on Ballast Water as a Vector for Aquatic Invasive Species to Freshwater and Marine Environments. Aquat. Ecosyst. Health Manag. 2015, 18, 261–268. [Google Scholar] [CrossRef]
- Hewitt, C.L.; Gollasch, S.; Minchin, D. The Vessel as a Vector—Biofouling, Ballast Water and Sediments. In Biological Invasions in Marine Ecosystems: Ecological, Management, and Geographic Perspectives; Rilov, G., Crooks, J.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 117–131. ISBN 9783540792369. [Google Scholar]
- Padilla, D.K.; Williams, S.L. Beyond Ballast Water: Aquarium and Ornamental Trades as Sources of Invasive Species in Aquatic Ecosystems. Front. Ecol. Environ. 2004, 2, 131–138. [Google Scholar] [CrossRef]
- Naylor, R.L.; Williams, S.L.; Strong, D.R. Ecology. Aquaculture—A Gateway for Exotic Species. Science 2001, 294, 1655–1656. [Google Scholar] [CrossRef] [PubMed]
- Gollasch, S.; Galil, B.S.; Cohen, A.N. Bridging Divides: Maritime Canals as Invasion Corridors; Springer: Berlin/Heidelberg, Germany, 2006; ISBN 9781402050466. [Google Scholar]
- Carlton, J.T. Transoceanic and Interoceanic Dispersal of Coastal Marine Organisms: The Biology of Ballast Water. Oceanogr. Mar. Biol. Annu. Rev. 1985, 23, 313–371. [Google Scholar]
- Galil, B.S.; Marchini, A.; Occhipinti-Ambrogi, A. East Is East and West Is West? Management of Marine Bioinvasions in the Mediterranean Sea. Estuar. Coast. Shelf Sci. 2018, 201, 7–16. [Google Scholar] [CrossRef]
- Por, F.D. Lessepsian Migration: The Influx of Red Sea Biota into the Mediterranean by Way of the Suez Canal; Springer: Berlin/Heidelberg, Germany, 2012; ISBN 9783642667282. [Google Scholar]
- Krom, M.D.; Herut, B.; Mantoura, R.F.C. Nutrient Budget for the Eastern Mediterranean: Implications for Phosphorus Limitation. Limnol. Oceanogr. 2004, 49, 1582–1592. [Google Scholar] [CrossRef]
- Soukissian, T.H.; Denaxa, D.; Karathanasi, F.; Prospathopoulos, A.; Sarantakos, K.; Iona, A.; Georgantas, K.; Mavrakos, S. Marine Renewable Energy in the Mediterranean Sea: Status and Perspectives. Energies 2017, 10, 1512. [Google Scholar] [CrossRef]
- Shaltout, M.; Omstedt, A. Recent Sea Surface Temperature Trends and Future Scenarios for the Mediterranean Sea. Oceanologia 2014, 56, 411–443. [Google Scholar] [CrossRef]
- Saraçoğlu, F.A.; Aydoğan, B.; Ayat, B.; Saraçoğlu, K.E. Spatial and Seasonal Variability of Long-Term Sea Surface Temperature Trends in Aegean and Levantine Basins. Pure Appl. Geophys. 2021, 178, 3769–3791. [Google Scholar] [CrossRef]
- Sisma-Ventura, G.; Yam, R.; Shemesh, A. Recent Unprecedented Warming and Oligotrophy of the Eastern Mediterranean Sea within the Last Millennium. Geophys. Res. Lett. 2014, 41, 5158–5166. [Google Scholar] [CrossRef]
- El-Geziry, T.M. Long-Term Changes in Sea Surface Temperature (SST) within the Southern Levantine Basin. Acta Oceanol. Sin. 2021, 40, 27–33. [Google Scholar] [CrossRef]
- Ozer, T.; Gertman, I.; Kress, N.; Silverman, J.; Herut, B. Interannual Thermohaline (1979–2014) and Nutrient (2002–2014) Dynamics in the Levantine Surface and Intermediate Water Masses, SE Mediterranean Sea. Glob. Planet. Change 2017, 151, 60–67. [Google Scholar] [CrossRef]
- Spanier, E.; Galil, B.S. Lessepsian Migration: A Continuous Biogeographical Process. Endeavour 1991, 15, 102–106. [Google Scholar] [CrossRef]
- Golani, D. Distribution of Lessepsian Migrant Fish in the Mediterranean. Ital. J. Zool. 1998, 65, 95–99. [Google Scholar] [CrossRef]
- Goren, M.; Galil, B.S. A Review of Changes in the Fish Assemblages of Levantine Inland and Marine Ecosystems Following the Introduction of Non-Native Fishes. J. Appl. Ichthyol. 2005, 21, 364–370. [Google Scholar] [CrossRef]
- Galil, B. Seeing Red: Alien Species along the Mediterranean Coast of Israel. Aquat. Invasions 2007, 2, 281–312. [Google Scholar] [CrossRef]
- Galil, B.S.; Marchini, A.; Occhipinti-Ambrogi, A.; Minchin, D.; Narščius, A.; Ojaveer, H.; Olenin, S. International Arrivals: Widespread Bioinvasions in European Seas. Ethol. Ecol. Evol. 2014, 26, 152–171. [Google Scholar] [CrossRef]
- Rilov, G.; Crooks, J.A. Marine Bioinvasions: Conservation Hazardsand Vehicles for Ecological Understanding. In Biological Invasions in Marine Ecosystems: Ecological, Management, and Geographic Perspectives; Rilov, G., Crooks, J.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 3–12. ISBN 9783540792369. [Google Scholar]
- Castellanos-Galindo, G.A.; Robertson, D.R.; Sharpe, D.M.T.; Torchin, M.E. A New Wave of Marine Fish Invasions through the Panama and Suez Canals. Nat. Ecol. Evol. 2020, 4, 1444–1446. [Google Scholar] [CrossRef]
- Kletou, D.; Hall-Spencer, J.M.; Kleitou, P. A Lionfish (Pterois Miles) Invasion Has Begun in the Mediterranean Sea. Mar. Biodivers. Rec. 2016, 9, 1–7. [Google Scholar] [CrossRef]
- Karachle, P.K.; Triantaphyllidis, C.; Stergiou, K.I. Bluespotted Cornetfish, Fistularia commersonii Rüppell, 1838: A Lessepsian Sprinter. AIeP 2004, 34, 103–108. [Google Scholar] [CrossRef]
- Hoffman, R.; Sternberg, M.; Serio, D. First Report of Laurencia chondrioides (Ceramiales, Rhodophyta) and Its Potential to Be an Invasive in the Eastern Mediterranean Sea. Bot. Mar. 2014, 57, 449–457. [Google Scholar] [CrossRef]
- Hamad, N.; Millot, C.; Taupier-Letage, I. The Surface Circulation in the Eastern Basin of the Mediterranean Sea. Sci. Mar. 2006, 70, 457–503. [Google Scholar]
- Fine, M.; Aluma, Y.; Meroz-Fine, E.; Abelson, A.; Loya, Y. Acabaria erythraea (Octocorallia: Gorgonacea) a Successful Invader to the Mediterranean Sea? Coral Reefs 2005, 24, 161–164. [Google Scholar] [CrossRef]
- Grossowicz, M.; Shemesh, E.; Martinez, S.; Benayahu, Y.; Tchernov, D. New Evidence of Melithaea erythraea Colonization in the Mediterranean. Estuar. Coast. Shelf Sci. 2020, 236, 106652. [Google Scholar] [CrossRef]
- Fabricius, K.; Alderslade, P. Soft Corals and Sea Fans: A Comprehensive Guide to the Tropical Shallow Water Genera of the Central-West Pacific, the Indian Ocean and the Red Sea; Australian Institute of Marine Science: Townsville, Australia, 2001. [Google Scholar]
- Benayahu, Y.; Loya, Y. Competition for Space among Coral-Reef Senssile Organisms at Eilat, Red Sea. Bull. Mar. Sci. 1981, 31, 514–522. [Google Scholar]
- Dai, C.-F. Interspecific Competition in Taiwanese Corals with Special Reference to Interactions between Alcyonaceans and Scleractinians. Mar. Ecol. Prog. Ser. 1990, 60, 291–297. [Google Scholar] [CrossRef]
- Barış Özalp, H.; Suat Ateş, A. New Records of Anthozoan Species (Cnidaria, Octocorallia, Hexacorallia) for the Turkish Straits System. Oceanol. Hydrobiol. Stud. 2015, 44, 193–205. [Google Scholar] [CrossRef]
- Vafidis, D.; Koukouras, A.; Voultisiadou-Koukoura, E. With a Check List of the Mediterranean Species: New Information, Faunal Comparisons. Inst Ocean. 1994, 70, 217–229. [Google Scholar]
- Cordeiro, R.T.S.; Carpinelli, Á.N.; Francini-Filho, R.B.; de Moura Neves, B.; Pérez, C.D.; de Oliveira, U.; Sumida, P.; Maranhão, H.; Monteiro, L.H.U.; Carneiro, P.; et al. Neospongodes Atlantica, a Potential Case of an Early Biological Introduction in the Southwestern Atlantic. PeerJ 2022, 10, e14347. [Google Scholar] [CrossRef] [PubMed]
- Williamson, J.E.; Gillings, M.R.; Nevatte, R.J.; Harasti, D.; Raoult, V.; Ghaly, T.M.; Stow, A.J.; Smith, T.M.; Gaston, T.F. Genetic Differentiation in the Threatened Soft Coral Dendronephthya australis in Temperate Eastern Australia. Austral Ecol. 2022, 47, 804–817. [Google Scholar] [CrossRef]
- Figueroa, D.F.; Baco, A.R. Octocoral Mitochondrial Genomes Provide Insights into the Phylogenetic History of Gene Order Rearrangements, Order Reversals, and Cnidarian Phylogenetics. Genome Biol. Evol. 2014, 7, 391–409. [Google Scholar] [CrossRef]
- Uda, K.; Komeda, Y.; Fujita, T.; Iwasaki, N.; Bavestrello, G.; Giovine, M.; Cattaneo-Vietti, R.; Suzuki, T. Complete Mitochondrial Genomes of the Japanese Pink Coral (Corallium elatius) and the Mediterranean Red Coral (Corallium rubrum): A Reevaluation of the Phylogeny of the Family Coralliidae Based on Molecular Data. Comp. Biochem. Physiol. Part D Genom. Proteom. 2013, 8, 209–219. [Google Scholar] [CrossRef]
- Grossowicz, M.; Benayahu, Y. Differential Morphological Features of Two Dendronephthya Soft Coral Species Suggest Differences in Feeding Niches. Mar. Biodivers. 2012, 42, 65–72. [Google Scholar] [CrossRef]
- Fabricius, K.E.; Genin, A.; Benayahu, Y. Flow-Dependent Herbivory and Growth in Zooxanthellae-Free Soft Corals. Limnol. Oceanogr. 1995, 40, 1290–1301. [Google Scholar] [CrossRef]
- Perkol-Finkel, S.; Benayahu, Y. Community Structure of Stony and Soft Corals on Vertical Unplanned Artificial Reefs in Eilat (Red Sea): Comparison to Natural Reefs. Coral Reefs 2004, 23, 195–205. [Google Scholar] [CrossRef]
- Fabricius, K.E.; Benayahu, Y.; Genin, A. Herbivory In Asymbiotic Soft Corals. Science 1995, 268, 90–92. [Google Scholar] [CrossRef]
- Dahan, M.; Benayahu, Y. Reproduction of Dendronephthya hemprichi (Cnidaria: Octocorallia): Year-Round Spawning in an Azooxanthellate Soft Coral. Mar. Biol. 1997, 129, 573–579. [Google Scholar] [CrossRef]
- Dahan, M.; Benayahu, Y. Clonal Propagation by the Azooxanthellate Octocoral Dendronephthya hemprichi. Coral Reefs 1997, 16, 5–12. [Google Scholar] [CrossRef]
- Pupier, C.A.; Bednarz, V.N.; Ferrier-Pagès, C. Studies with Soft Corals—Recommendations on Sample Processing and Normalization Metrics. Front. Mar. Sci. 2018, 5, 348. [Google Scholar] [CrossRef]
- Perkol-Finkel, S.; Benayahu, Y. Differential Recruitment of Benthic Communities on Neighboring Artificial and Natural Reefs. J. Exp. Mar. Bio. Ecol. 2007, 340, 25–39. [Google Scholar] [CrossRef]
- Perkol-Finkel, S.; Benayahu, Y. Recruitment of Benthic Organisms onto a Planned Artificial Reef: Shifts in Community Structure One Decade Post-Deployment. Mar. Environ. Res. 2005, 59, 79–99. [Google Scholar] [CrossRef]
- Beijbom, O.; Edmunds, P.J.; Roelfsema, C.; Smith, J.; Kline, D.I.; Neal, B.P.; Dunlap, M.J.; Moriarty, V.; Fan, T.-Y.; Tan, C.-J.; et al. Towards Automated Annotation of Benthic Survey Images: Variability of Human Experts and Operational Modes of Automation. PLoS ONE 2015, 10, e0130312. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Grossowicz, M.; Benayahu, Y. Occurrence and Survivorship of Azooxanthellate Octocorals Reflect Recruitment Preferences and Depth Distribution. Hydrobiologia 2015, 759, 85–93. [Google Scholar] [CrossRef]
- Ruiz-Allais, J.P.; Benayahu, Y.; Lasso-Alcalá, O.M. The invasive octocoral Unomia stolonifera (Alcyonacea, Xeniidae) is dominating the benthos in the Southeastern Caribbean Sea. Mem. Fund. Salle Cienc. Nat. 2021, 79, 63–80. [Google Scholar]
- Creed, J.C. Two Invasive Alien Azooxanthellate Corals, Tubastraea coccinea and Tubastraea tagusensis, Dominate the Native Zooxanthellate Mussismilia hispida in Brazil. Coral Reefs 2006, 25, 350. [Google Scholar] [CrossRef]
- Luz, B.L.P.; Kitahara, M.V. Could the Invasive Scleractinians Tubastraea coccinea and T. tagusensis Replace the Dominant Zoantharian Palythoa caribaeorum in the Brazilian Subtidal? Coral Reefs 2017, 36, 875. [Google Scholar] [CrossRef]
- Miranda, R.J.; Cruz, I.C.S.; Barros, F. Effects of the Alien Coral Tubastraea tagusensis on Native Coral Assemblages in a Southwestern Atlantic Coral Reef. Mar. Biol. 2016, 163, 45. [Google Scholar] [CrossRef]
- Giakoumi, S.; Katsanevakis, S.; Albano, P.G.; Azzurro, E.; Cardoso, A.C.; Cebrian, E.; Deidun, A.; Edelist, D.; Francour, P.; Jimenez, C.; et al. Management Priorities for Marine Invasive Species. Sci. Total Environ. 2019, 688, 976–982. [Google Scholar] [CrossRef]
- Otero, M.; Cebrian, E.; Francour, P.; Galil, B.; Savini, D. Monitoring marine invasive species in Mediterranean marine protected areas (MPAs): A strategy and practical guide for managers. IUCN Malaga 2013, 1–136. [Google Scholar]
- Gissi, E.; Manea, E.; Mazaris, A.D.; Fraschetti, S.; Almpanidou, V.; Bevilacqua, S.; Coll, M.; Guarnieri, G.; Lloret-Lloret, E.; Pascual, M.; et al. A Review of the Combined Effects of Climate Change and Other Local Human Stressors on the Marine Environment. Sci. Total Environ. 2021, 755, 142564. [Google Scholar] [CrossRef]
- Edelist, D.; Rilov, G.; Golani, D.; Carlton, J.T.; Spanier, E. Restructuring the Sea: Profound Shifts in the World’s Most Invaded Marine Ecosystem. Divers. Distrib. 2013, 19, 69–77. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Morri, C. Global Sea Warming and “tropicalization” of the Mediterranean Sea: Biogeographic and Ecological Aspects. Biogeogr.—J. Integr. Biogeogr. 2003, 24, b6110129. [Google Scholar] [CrossRef]
- Bianchi, C.N. Biodiversity Issues for the Forthcoming Tropical Mediterranean Sea. Hydrobiologia 2007, 580, 7–21. [Google Scholar] [CrossRef]
- Martinez, S.; Bellworthy, J.; Ferrier-Pagès, C.; Mass, T. Selection of Mesophotic Habitats by Oculina patagonica in the Eastern Mediterranean Sea Following Global Warming. Sci. Rep. 2021, 11, 18134. [Google Scholar] [CrossRef] [PubMed]
- Canning-Clode, J.; Carlton, J.T. Refining and Expanding Global Climate Change Scenarios in the Sea: Poleward Creep Complexities, Range Termini, and Setbacks and Surges. Divers. Distrib. 2017, 23, 463–473. [Google Scholar] [CrossRef]
- Vergés, A.; Steinberg, P.D.; Hay, M.E.; Poore, A.G.B.; Campbell, A.H.; Ballesteros, E.; Heck, K.L., Jr.; Booth, D.J.; Coleman, M.A.; Feary, D.A.; et al. The Tropicalization of Temperate Marine Ecosystems: Climate-Mediated Changes in Herbivory and Community Phase Shifts. Proc. Biol. Sci. 2014, 281, 20140846. [Google Scholar] [CrossRef]
- Rilov, G.; Benayahu, Y.; Gasith, A. Prolonged Lag in Population Outbreak of an Invasive Mussel: A Shifting-Habitat Model. Biol. Invasions. 2004, 6, 347–364. [Google Scholar] [CrossRef]
- Por, F.D. Tethys Returns to the Mediterranean: Success and Limits of Tropical Re-Colonization. BioRisk 2009, 3, 5–19. [Google Scholar] [CrossRef]
| Species/Specimen | Family | Genus | Source | NCBI Accession Number | rRNA | MSH | COX1 |
|---|---|---|---|---|---|---|---|
| Dendronephthya mollis | Nephtheidae | Dendronephthya | NCBI, complete genome | NC_020456.1 | NC_020456.1 | NC_020456.1 | NC_020456.1 |
| Dendronephthya suensoni | Nephtheidae | Dendronephthya | NCBI, complete genome | GU047878.1 | GU047878.1 | GU047878.1 | GU047878.1 |
| Dendronephthya putteri | Nephtheidae | Dendronephthya | NCBI, complete genome | JQ886185.1 | JQ886185.1 | JQ886185.1 | JQ886185.1 |
| Dendronephthya sinaiensis | Nephtheidae | Dendronephthya | NCBI, complete genome | NC_062008.1 | NC_062008.1 | NC_062008.1 | NC_062008.1 |
| Dendronephthya alba | Nephtheidae | Dendronephthya | NCBI, complete genome | MW423625.1 | MW423625.1 | MW423625.1 | MW423625.1 |
| Dendronephthya castanea | Nephtheidae | Dendronephthya | NCBI, complete genome | NC_023343.1 | NC_023343.1 | NC_023343.1 | NC_023343.1 |
| Dendronephthya gigantea | Nephtheidae | Dendronephthya | NCBI, complete genome | NC_013573.1 | NC_013573.1 | NC_013573.1 | NC_013573.1 |
| Dendronephthya hemprichi | Nephtheidae | Dendronephthya | NCBI | GU356019.1 | GU355996.1 | ||
| Scleronephthya gracillimum | Nephtheidae | Scleronephthya | NCBI, complete genome | NC_023344.1 | NC_023344.1 | NC_023344.1 | NC_023344.1 |
| Eunicella albicans | Eunicellidae | Eunicella | NCBI, complete genome | NC_035666.1 | NC_035666.1 | NC_035666.1 | NC_035666.1 |
| Eunicella tricoronata | Eunicellidae | Eunicella | NCBI, complete genome | NC_062012.1 | NC_062012.1 | NC_062012.1 | NC_062012.1 |
| Eunicella cavolinii | Eunicellidae | Eunicella | NCBI, complete genome | NC_035667.1 | NC_035667.1 | NC_035667.1 | NC_035667.1 |
| Trachythela sp. YZ-2021 | Eunicellidae | Trachyela | NCBI, complete genome | MW238423.1 | MW238423.1 | MW238423.1 | MW238423.1 |
| Eugorgia mutabilis | Gordoniidae | Eugorgia | NCBI, complete genome | NC_035665.1 | NC_035665.1 | NC_035665.1 | NC_035665.1 |
| Leptogorgia hebes isolate PLG4-L68 | Gordoniidae | Leptogorgia | NCBI, complete genome | MN052677.1 | MN052677.1 | MN052677.1 | MN052677.1 |
| Leptogorgia alba | Gordoniidae | Leptogorgia | NCBI, complete genome | NC_035669.1 | NC_035669.1 | NC_035669.1 | NC_035669.1 |
| Leptogorgia cf. palma AP-2017 | Gordoniidae | Leptogorgia | NCBI, complete genome | KY559406.1 | KY559406.1 | KY559406.1 | KY559406.1 |
| Leptogorgia capverdensis | Gordoniidae | Leptogorgia | NCBI, complete genome | NC_035663.1 | NC_035663.1 | NC_035663.1 | NC_035663.1 |
| Leptogorgia sarmentosa | Gordoniidae | Leptogorgia | NCBI, complete genome | NC_035670.1 | NC_035670.1 | NC_035670.1 | NC_035670.1 |
| Pseudopterogorgia bipinnata | Gordoniidae | Antillogorgia | NCBI, complete genome | NC_008157.1 | NC_008157.1 | NC_008157.1 | NC_008157.1 |
| Pacifigorgia cairnsi | Gordoniidae | Pacifigorgia | NCBI, complete genome | NC_035668.1 | NC_035668.1 | NC_035668.1 | NC_035668.1 |
| Astrogorgia sp. ANT66 | Astrogorgiidae | Astrogorgia | NCBI, complete genome | OL616212.1 | OL616212.1 | OL616212.1 | OL616212.1 |
| Discophyton rudyi | Discophytidae | Discophyton | NCBI, complete genome | NC_061276.1 | NC_061276.1 | NC_061276.1 | NC_061276.1 |
| Alcyonium acaule | Alcyoniidae | Alcyonium | NCBI, complete genome | NC_061273.1 | NC_061273.1 | NC_061273.1 | NC_061273.1 |
| Alertigorgia orientalis | Alcyoniidae | Alertigorgia | NCBI, complete genome | NC_061994.1 | NC_061994.1 | NC_061994.1 | NC_061994.1 |
| Sinularia ceramensis | Sinulariidae | Sinularia | NCBI, complete genome | NC_044122.1 | NC_044122.1 | NC_044122.1 | NC_044122.1 |
| Sinularia flexibilis | Sinulariidae | Sinularia | NCBI, complete genome | NC_061282.1 | NC_061282.1 | NC_061282.1 | NC_061282.1 |
| Siphonogorgia godeffroyi | Nidaliidae | Siphonogorgia | NCBI, complete genome | NC_062032.1 | NC_062032.1 | NC_062032.1 | NC_062032.1 |
| Rumphella sp. ANT116 | Plexauridae | Rumphella | NCBI, complete genome | OL616268.1 | OL616268.1 | OL616268.1 | OL616268.1 |
| Ceeceenus quadrus | Paralcyoniidae | Ceeceenus | NCBI, complete genome | NC_062003.1 | NC_062003.1 | NC_062003.1 | NC_062003.1 |
| Azoriella bayeri | Cerveridae | Azoriella | NCBI, complete genome | NC_061999.1 | NC_061999.1 | NC_061999.1 | NC_061999.1 |
| Euplexaura crassa | Euplexauridae | Euplexaura | NCBI, complete genome | NC_020458.1 | NC_020458.1 | NC_020458.1 | NC_020458.1 |
| Red Sea 36 m | This study | OR458391 | OR526527 | OR520375 | |||
| Mediterranean Sea | This study | OR462245 | OR520989 | OR520376 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nativ, H.; Galili, O.; Almuly, R.; Einbinder, S.; Tchernov, D.; Mass, T. New Record of Dendronephthya sp. (Family: Nephtheidae) from Mediterranean Israel: Evidence for Tropicalization? Biology 2023, 12, 1220. https://doi.org/10.3390/biology12091220
Nativ H, Galili O, Almuly R, Einbinder S, Tchernov D, Mass T. New Record of Dendronephthya sp. (Family: Nephtheidae) from Mediterranean Israel: Evidence for Tropicalization? Biology. 2023; 12(9):1220. https://doi.org/10.3390/biology12091220
Chicago/Turabian StyleNativ, Hagai, Ori Galili, Ricardo Almuly, Shai Einbinder, Dan Tchernov, and Tali Mass. 2023. "New Record of Dendronephthya sp. (Family: Nephtheidae) from Mediterranean Israel: Evidence for Tropicalization?" Biology 12, no. 9: 1220. https://doi.org/10.3390/biology12091220
APA StyleNativ, H., Galili, O., Almuly, R., Einbinder, S., Tchernov, D., & Mass, T. (2023). New Record of Dendronephthya sp. (Family: Nephtheidae) from Mediterranean Israel: Evidence for Tropicalization? Biology, 12(9), 1220. https://doi.org/10.3390/biology12091220







