The Role of Zinc and Copper in Platelet Activation and Pathophysiological Thrombus Formation in Patients with Pulmonary Embolism in the Course of SARS-CoV-2 Infection
Abstract
Simple Summary
Abstract
1. Introduction
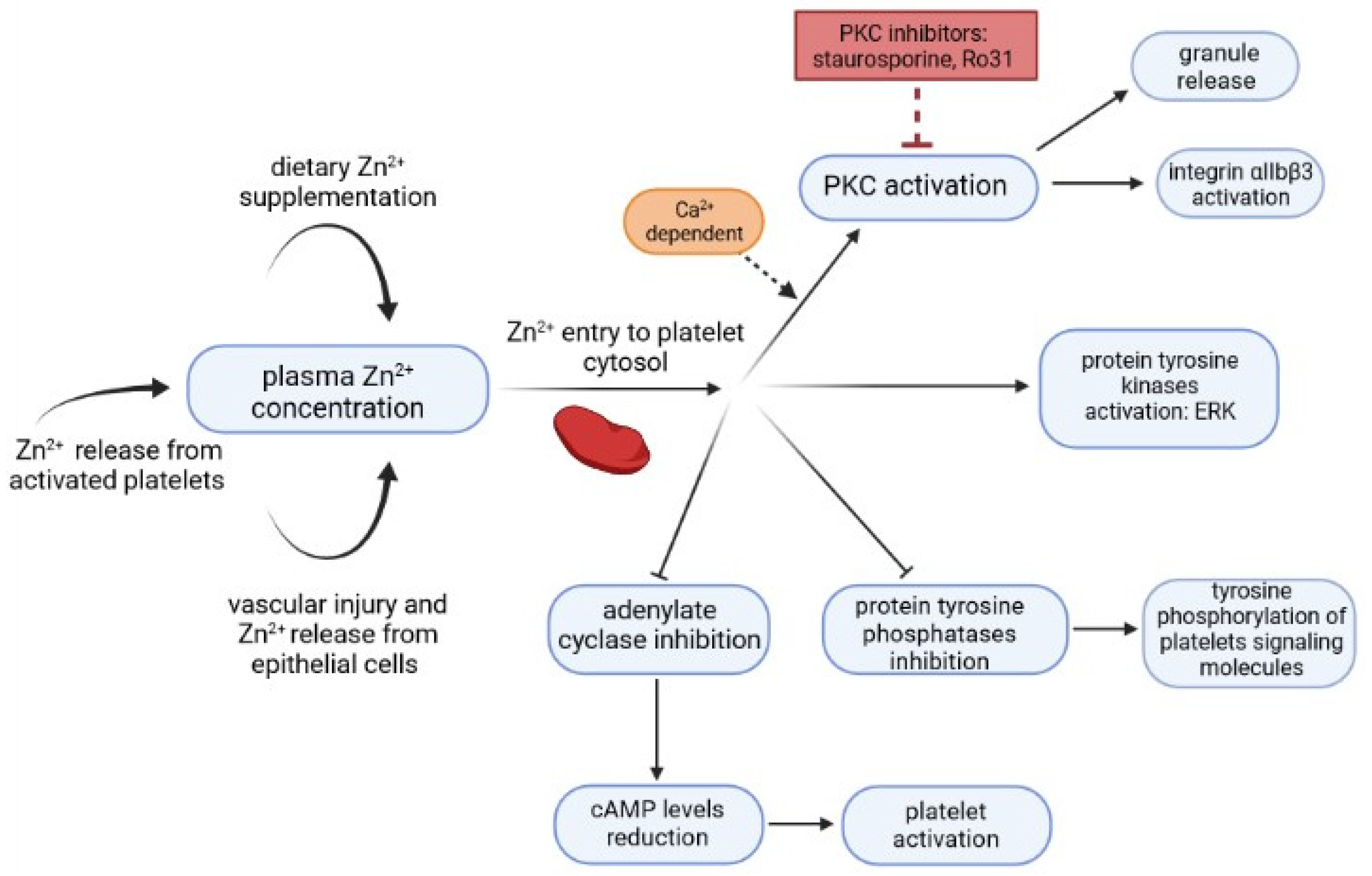
2. Relationship between Zn2+/Cu2+ and CRP Levels and Platelet Activation
3. Vascular Changes versus Metals and Metalloproteins
4. Oxidative Stress and Inflammation
- -
- Reactions involving compounds that quench excited molecules (carotenoids, vitamin E)
- -
- Non-enzymatic mechanisms—uric acid, bilirubin, glutathione, pyruvate, ubiquinone (coenzyme Q), transferrin, polyamides, transition metal ions, metalloproteins
- -
- Enzymatic mechanisms: superoxide dismutase (SOD)—catalyzes the superoxide anion radical dismutation reaction, catalase (CT)—catalyzes the hydrogen peroxide dismutation reaction, glutathione peroxidase (GPx), ceruloplasmin, glutathione S-transferase (GST), secretory phospholipase group A2 (sPLA2)
- -
- Heat shock proteins (Hsps)—a large family of molecular chaperones that can be divided into two groups: first—small, ATP-independent Hsps with molecular weights from 8 to 28 dKA e.g., ubiquitin, and second—large, ATP-dependent Hsps with molecular weights from 40 to 105 kDA; a group of proteins whose expression increases when cells are exposed to stress factors (e.g., osmotic stress, heavy metals).
- -
- It is well known that viral infections can alter the redox system increasing oxidant species and reducing antioxidant molecules. Varga et al. reported that there is a link between ROS, endothelial damage, and inflammation, and this above mechanism also occurs during the course of COVID-19 [47].
4.1. The Role of Zn2+/Cu2+ in the Development of Oxidative Stress
4.2. Vascular Complications and Their Relationship with CRP Levels
5. The Role of Inflammation in Relation to COVID-19 and Potential Complications
6. Therapeutic Modifications of Zn2+/Cu2 Levels in Relation to Pulmonary Embolism and Patients with COVID-19
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gammoh, N.Z.; Rink, L. Zinc in Infection and Inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Linder, M.C.; Hazegh-Azam, M. Copper biochemistry and molecular biology. Am. J. Clin. Nutr. 1996, 63, 797S–811S. [Google Scholar] [CrossRef] [PubMed]
- A Read, S.; Obeid, S.; Ahlenstiel, C.; Ahlenstiel, G. The Role of Zinc in Antiviral Immunity. Adv. Nutr. Int. Rev. J. 2019, 10, 696–710. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, J.; Hannigan, B.; Strain, J.; Bonham, M. The immune system as a physiological indicator ofmarginal copper status? Br. J. Nutr. 2002, 87, 393–403. [Google Scholar] [CrossRef]
- Zischka, H.; Kroemer, G. Copper—A novel stimulator of autophagy. Cell Stress 2020, 4, 92–94. [Google Scholar] [CrossRef]
- Johnstone, J.; Roth, D.E.; Guyatt, G.; Loeb, M. Zinc for the treatment of the common cold: A systematic review and meta-analysis of randomized controlled trials. Can. Med. Assoc. J. 2012, 184, E551–E561. [Google Scholar] [CrossRef]
- Taylor, K.A.; Pugh, N. The contribution of zinc to platelet behaviour during haemostasis and thrombosis. Metallomics 2016, 8, 144–155. [Google Scholar] [CrossRef]
- Henderson, S.J.; Xia, J.; Wu, H.; Stafford, A.R.; Leslie, B.A.; Fredenburgh, J.C.; Weitz, D.A.; Weitz, J.I. Zinc promotes clot stability by accelerating clot formation and modifying fibrin structure. Thromb. Haemost. 2016, 115, 533–542. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- World Health Organization. Coronavirus Disease (COVID-19) Situation Reports. 2020. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports (accessed on 13 July 2020).
- Lodigiani, C.; Iapichino, G.; Carenzo, L.; Cecconi, M.; Ferrazzi, P.; Sebastian, T.; Kucher, N.; Studt, J.-D.; Sacco, C.; Bertuzzi, A.; et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020, 191, 9–14. [Google Scholar] [CrossRef]
- Cui, S.; Chen, S.; Li, X.; Liu, S.; Wang, F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Tavazzi, G.; Civardi, L.; Caneva, L.; Mongodi, S.; Mojoli, F. Thrombotic events in SARS-CoV-2 patients: An urgent call for ultrasound screening. Intensive Care Med. 2020, 46, 1121–1123. [Google Scholar] [CrossRef]
- Helms, J.; Tacquard, C.; Severac, F.; Leonard-Lorant, I.; Ohana, M.; Delabranche, X.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Gandet, F.F.; et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study. Intensive Care Med. 2020, 46, 1089–1098. [Google Scholar] [CrossRef]
- Poissy, J.; Goutay, J.; Caplan, M.; Parmentier-Decrucq, E.; Duburcq, T.; Lassalle, F.; Jeanpierre, E.; Rauch, A.; Labreuche, J.; Susen, S.; et al. Pulmonary Embolism in Patients with COVID-19: Awareness of an increased prevalence. Circulation 2020, 142, 184–186. [Google Scholar] [CrossRef] [PubMed]
- Léonard-Lorant, I.; Delabranche, X.; Séverac, F.; Helms, J.; Pauzet, C.; Collange, O.; Schneider, F.; Labani, A.; Bilbault, P.; Molière, S.; et al. Acute Pulmonary Embolism in Patients with COVID-19 at CT Angiography and Relationship to d-Dimer Levels. Radiology 2020, 296, E189–E191. [Google Scholar] [CrossRef]
- Grillet, F.; Behr, J.; Calame, P.; Aubry, S.; Delabrousse, E. Acute Pulmonary Embolism Associated with COVID-19 Pneumonia Detected with Pulmonary CT Angiography. Radiology 2020, 296, E186–E188. [Google Scholar] [CrossRef] [PubMed]
- Avila, J.; Long, B.; Holladay, D.; Gottlieb, M. Thrombotic complications of COVID-19. Am. J. Emerg. Med. 2020, 39, 213–218. [Google Scholar] [CrossRef]
- Shibeeb, S.; Ahmad, M.N. Thrombotic and Hypercoagulability Complications of COVID-19: An Update. J. Blood Med. 2021, 12, 785–793. [Google Scholar] [CrossRef]
- Hippensteel, J.A.; Burnham, E.L.; Jolley, S.E. Prevalence of venous thromboembolism in critically ill patients with COVID-19. Br. J. Haematol. 2020, 190, e134–e137. [Google Scholar] [CrossRef]
- Middeldorp, S.; Coppens, M.; Van Haaps, T.F.; Foppen, M.; Vlaar, A.P.; Müller, M.C.A.; Bouman, C.C.S.; Beenen, L.F.M.; Kootte, R.S.; Heijmans, J.; et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 2020, 18, 1995–2002. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb. Res. 2020, 191, 148–150. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal Coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Malas, M.B.; Naazie, I.N.; Elsayed, N.; Mathlouthi, A.; Marmor, R.; Clary, B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. eClinicalMedicine 2020, 29, 100639. [Google Scholar] [CrossRef]
- Lax, S.F.; Skok, K.; Zechner, P.; Kessler, H.H.; Kaufmann, N.; Koelblinger, C.; Vander, K.; Bargfrieder, U.; Trauner, M. Pulmonary Arterial Thrombosis in COVID-19 with Fatal Outcome: Results from a Prospective, Single-Center, Clinicopathologic Case Series. Ann. Intern. Med. 2020, 173, 350–361. [Google Scholar] [CrossRef]
- García-Ortega, A.; Oscullo, G.; Calvillo, P.; López-Reyes, R.; Méndez, R.; Gómez-Olivas, J.D.; Bekki, A.; Fonfría, C.; Trilles-Olaso, L.; Zaldívar, E.; et al. Incidence, risk factors, and thrombotic load of pulmonary embolism in patients hospitalized for COVID-19 infection. J. Infect. 2021, 82, 261–269. [Google Scholar] [CrossRef]
- Yuan, Y.; Long, P.; Liu, K.; Xiao, Y.; He, S.; Li, J.; Mo, T.; Liu, Y.; Yu, Y.; Wang, H.; et al. Multiple plasma metals, genetic risk and serum C-reactive protein: A metal-metal and gene-metal interaction study. Redox Biol. 2019, 29, 101404. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Djafarian, K.; Mojtahed, A.; Varkaneh, H.K.; Shab-Bidar, S. The effect of zinc supplementation on plasma C-reactive protein concentrations: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Pharmacol. 2018, 834, 10–16. [Google Scholar] [CrossRef]
- Wacewicz, M.; Socha, K.; Soroczyńska, J.; Niczyporuk, M.; Aleksiejczuk, P.; Ostrowska, J.; Borawska, M.H. Concentration of selenium, zinc, copper, Cu/Zn ratio, total antioxidant status and c-reactive protein in the serum of patients with psoriasis treated by narrow-band ultraviolet B phototherapy: A case-control study. J. Trace Elements Med. Biol. 2017, 44, 109–114. [Google Scholar] [CrossRef]
- Saad, A.A.; Doka, Y.A.; Osman, S.M.; Magzoub, M.; Ali, N.I.; Adam, I. Zinc, Copper and C-reactive Protein in Children with Severe Plasmodium falciparum Malaria in an Area of Unstable Malaria Transmission in Eastern Sudan. J. Trop. Pediatr. 2012, 59, 150–153. [Google Scholar] [CrossRef]
- Strang, F.; Schunkert, H. C-Reactive Protein and Coronary Heart Disease: All Said—Is Not It? Mediat. Inflamm. 2014, 2014, 757123. [Google Scholar] [CrossRef] [PubMed]
- Emerging Risk Factors Collaboration; Kaptoge, S.; Di Angelantonio, E.; Pennells, L.; Wood, A.M.; White, I.R.; Gao, P.; Walker, M.; Thompson, A.; Sarwar, N.; et al. C-Reactive Protein, Fibrinogen, and Cardiovascular Disease Prediction. N. Engl. J. Med. 2012, 367, 1310–1320. [Google Scholar] [CrossRef] [PubMed]
- Yousuf, O.; Mohanty, B.D.; Martin, S.S.; Joshi, P.H.; Blaha, M.J.; Nasir, K.; Blumenthal, R.S.; Budoff, M.J. High-Sensitivity C-Reactive Protein and Cardiovascular Disease: A resolute belief or an elusive link? J. Am. Coll. Cardiol. 2013, 62, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Molins, B.; Peña, E.; Vilahur, G.; Mendieta, C.; Slevin, M.; Badimon, L. C-Reactive Protein Isoforms Differ in Their Effects on Thrombus Growth. Arter. Thromb. Vasc. Biol. 2008, 28, 2239–2246. [Google Scholar] [CrossRef]
- De La Torre, R.; Pena, E.; Vilahur, G.; Slevin, M.; Badimon, L. Monomerization of C-reactive protein requires glycoprotein IIb-IIIa activation: Pentraxins and platelet deposition. J. Thromb. Haemost. 2013, 11, 2048–2058. [Google Scholar] [CrossRef]
- Yasojima, K.; Schwab, C.; McGeer, E.G.; McGeer, P.L. Generation of C-Reactive Protein and Complement Components in Atherosclerotic Plaques. Am. J. Pathol. 2001, 158, 1039–1051. [Google Scholar] [CrossRef]
- Krupinski, J.; Turu, M.M.; Martinez-Gonzalez, J.; Carvajal, A.; Juan-Babot, J.O.; Iborra, E.; Slevin, M.; Rubio, F.; Badimon, L. Endogenous Expression of C-Reactive Protein Is Increased in Active (Ulcerated Noncomplicated) Human Carotid Artery Plaques. Stroke 2006, 37, 1200–1204. [Google Scholar] [CrossRef]
- Badimon, L.; Chesebro, J.H.; Badimon, J.J. Thrombus formation on ruptured atherosclerotic plaques and re-thrombosis on evolving thrombi. Circulation 1992, 86, III74–III85. [Google Scholar]
- Yokoyama, S.; Ikeda, H.; Haramaki, N.; Yasukawa, H.; Murohara, T.; Imaizumi, T. Platelet P-selectin plays an important role in arterial thrombogenesis by forming large stable platelet-leukocyte aggregates. J. Am. Coll. Cardiol. 2005, 45, 1280–1286. [Google Scholar] [CrossRef]
- Boncler, M.; Rywaniak, J.; Szymański, J.; Potempa, L.A.; Rychlik, B.; Watała, C. Modified C-reactive protein interacts with platelet glycoprotein Ibα. Pharmacol. Rep. 2011, 63, 464–475. [Google Scholar] [CrossRef]
- Tzima, E.; Walker, J.H. Platelet annexin V: The ins and outs. Platelets 2000, 11, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Gordon, P.R.; Woodruff, C.W.; Anderson, H.L.; O’Dell, B.L. Effect of acute zinc deprivation on plasma zinc and platelet aggregation in adult males. Am. J. Clin. Nutr. 1982, 35, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Tautz, L.; Senis, Y.A.; Oury, C.; Rahmouni, S. Perspective: Tyrosine phosphatases as novel targets for antiplatelet therapy. Bioorg. Med. Chem. 2015, 23, 2786–2797. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Heyduk, T.; Sunahara, R.K. Zinc inhibition of adenylyl cyclase correlates with conformational changes in the enzyme. Cell. Signal. 2004, 16, 1177–1185. [Google Scholar] [CrossRef]
- Harper, M.T.; Poole, A.W. Diverse functions of protein kinase C isoforms in platelet activation and thrombus formation. J. Thromb. Haemost. 2009, 8, 454–462. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Levi, M.; Thachil, J.; Iba, T.; Levy, J.H. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020, 7, e438–e440. [Google Scholar] [CrossRef]
- Wessels, I.; Rolles, B.; Rink, L. The Potential Impact of Zinc Supplementation on COVID-19 Pathogenesis. Front. Immunol. 2020, 11, 1712. [Google Scholar] [CrossRef]
- Wessels, I.; Maywald, M.; Rink, L. Zinc as a Gatekeeper of Immune Function. Nutrients 2017, 9, 1286. [Google Scholar] [CrossRef]
- Prasad, A.S.; Beck, F.W.; Bao, B.; Fitzgerald, J.T.; Snell, D.C.; Steinberg, J.D.; Cardozo, L.J. Zinc supplementation decreases incidence of infections in the elderly: Effect of zinc on generation of cytokines and oxidative stress. Am. J. Clin. Nutr. 2007, 85, 837–844. [Google Scholar] [CrossRef]
- Lin, P.-H.; Sermersheim, M.; Li, H.; Lee, P.H.U.; Steinberg, S.M.; Ma, J. Zinc in Wound Healing Modulation. Nutrients 2017, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Pires, M.; Ahmed, N.S.; Vara, D.; Gibbins, J.M.; Pula, G.; Pugh, N. Zinc regulates reactive oxygen species generation in platelets. Platelets 2020, 32, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Raptis, C.A.; Hammer, M.; Short, R.G.; Shah, A.; Bhalla, S.; Bierhals, A.J.; Filev, P.D.; Hope, M.D.; Jeudy, J.; Kligerman, S.J.; et al. Chest CT and Coronavirus Disease (COVID-19): A Critical Review of the Literature to Date. Am. J. Roentgenol. 2020, 215, 839–842. [Google Scholar] [CrossRef]
- Liu, P.P.; Blet, A.; Smyth, D.; Li, H. The Science Underlying COVID-19: Implications for the Cardiovascular System. Circulation 2020, 142, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Abrishami, M.; Emamverdian, Z.; Shoeibi, N.; Omidtabrizi, A.; Daneshvar, R.; Rezvani, T.S.; Saeedian, N.; Eslami, S.; Mazloumi, M.; Sadda, S.; et al. Optical coherence tomography angiography analysis of the retina in patients recovered from COVID-19: A case-control study. Can. J. Ophthalmol. 2020, 56, 24–30. [Google Scholar] [CrossRef]
- Jidigam, V.K.; Singh, R.; Batoki, J.C.; Milliner, C.; Sawant, O.B.; Bonilha, V.L.; Rao, S. Histopathological assessments reveal retinal vascular changes, inflammation, and gliosis in patients with lethal COVID-19. Inflamm. Disord. 2021, 260, 1275–1288. [Google Scholar] [CrossRef]
- Oudkerk, M.; Kuijpers, D.; Oudkerk, S.F.; Van Beek, E.J. The vascular nature of COVID-19. Br. J. Radiol. 2020, 93, 20200718. [Google Scholar] [CrossRef] [PubMed]
- Kulbacka, J.; Saczko, J.; Chwiłkowska, A. Stres oksydacyjny w procesach uszkodzenia komórek. Pol. Merk. Lek. 2009, 157, 44–47. [Google Scholar]
- Sies, H. Oxidative stress: From basic research to clinical application. Am. J. Med. 1991, 91, S31–S38. [Google Scholar] [CrossRef]
- Lushchak, V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014, 224, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Sikora, N.; Znajewska, Z.; Dąbrowska, G. Wybrane mechanizmy obronne organizmów przed stresem oksydacyjnym ze szczególnym uwzględnieniem dysmutaz ponadtlenkowych. Edukacja Biologiczna i Środowiskowa 2017, 4, 19–31. [Google Scholar]
- Kołodziejczyk, J.; Saluk, J.; Wachowicz, B. Stres oksydacyjny—Reaktywne formy tlenu i azotu w patogenezie zaburzeń układu krążenia. Ann. Acad. Med. Sil. 2011, 65, 63–69. [Google Scholar]
- Krzyściak, W.; Kózka, M.; Kowalska, J.; Kwiatek, W.M. Rola Zn i Cu—Pierwiastków śladowych oraz dysmutazy ponadtlenkowej (SOD) w rozwoju stresu oksydacyjnego w przewlekłej chorobie żylnej (PChŻ). Przegląd Lekarski 2010, 67, 446–449. [Google Scholar]
- I Aruoma, O.; Halliwell, B.; Gajewski, E.; Dizdaroglu, M. Copper-ion-dependent damage to the bases in DNA in the presence of hydrogen peroxide. Biochem. J. 1991, 273, 601–604. [Google Scholar] [CrossRef]
- Marreiro, D.D.N.; Cruz, K.J.C.; Morais, J.B.S.; Beserra, J.B.; Severo, J.S.; De Oliveira, A.R.S. Zinc and oxidative stress: Current mechanisms. Antioxidants 2017, 6, 24. [Google Scholar] [CrossRef]
- Cruz, K.J.C. Antioxidant role of zinc in diabetes mellitus. World J. Diabetes 2015, 6, 333–337. [Google Scholar] [CrossRef]
- Eide, D.J. The oxidative stress of zinc deficiency. Metallomics 2011, 3, 1124–1129. [Google Scholar] [CrossRef]
- Özcelik, D.; Nazıroglu, M.; Tunçdemir, M.; Çelik, Ö.; Öztürk, M.; Flores-Arce, M.F. Zinc Supplementation Attenuates Metallothionein and Oxidative Stress Changes in Kidney of Streptozotocin-Induced Diabetic Rats. Biol. Trace Element Res. 2012, 150, 342–349. [Google Scholar] [CrossRef]
- Pepys, M.B.; Hirschfield, G. C-reactive protein: A critical update. J. Clin. Investig. 2003, 111, 1805–1812. [Google Scholar] [CrossRef]
- Pepys, M.B. The acute phase response and C-reactive protein. In Oxford Textbook of Medicine; Oxford University Press: Oxford, UK, 1995; Volume 2, pp. 1527–1533. [Google Scholar]
- Thompson, D.; Pepys, M.B.; Wood, S.P. The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure 1999, 7, 169–177. [Google Scholar] [CrossRef]
- Eklund, C.M. Chapter 5 Proinflammatory cytokines in CRP baseline regulation. Adv. Clin. Chem. 2009, 48, 111–136. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, S.; Jialal, I. C-Reactive Protein Polarizes Human Macrophages to an M1 Phenotype and Inhibits Transformation to the M2 Phenotype. Arter. Thromb. Vasc. Biol. 2011, 31, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Inflammation in Atherosclerosis. Arter. Thromb. Vasc. Biol. 2012, 32, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Calabrò, P.; Chang, D.W.; Willerson, J.T.; Yeh, E.T. Release of C-Reactive Protein in Response to Inflammatory Cytokines by Human Adipocytes: Linking Obesity to Vascular Inflammation. J. Am. Coll. Cardiol. 2005, 46, 1112–1113. [Google Scholar] [CrossRef]
- Molins, B.; Peña, E.; de la Torre, R.; Badimon, L. Monomeric C-reactive protein is prothrombotic and dissociates from circulating pentameric C-reactive protein on adhered activated platelets under flow. Cardiovasc. Res. 2011, 92, 328–337. [Google Scholar] [CrossRef]
- Lutsey, P.L.; Astor, B.C.; Cushman, M.; Folsom, A.R. C-reactive protein and venous thromboembolism. A prospective investigation in the ARIC cohort. Thromb. Haemost. 2009, 102, 615–619. [Google Scholar] [CrossRef]
- Goldhaber, S.Z.; Bounameaux, H. Pulmonary embolism and deep vein thrombosis. Lancet 2012, 379, 1835–1846. [Google Scholar] [CrossRef]
- Keller, K.; Hobohm, L.; Münzel, T.; Ostad, M.A. Impact of symptomatic atherosclerosis in patients with pulmonary embolism. Int. J. Cardiol. 2018, 278, 225–231. [Google Scholar] [CrossRef]
- Prandoni, P.; Bilora, F.; Marchiori, A.; Bernardi, E.; Petrobelli, F.; Lensing, A.W.A.; Prins, M.H.; Girolami, A. An Association between Atherosclerosis and Venous Thrombosis. N. Engl. J. Med. 2003, 348, 1435–1441. [Google Scholar] [CrossRef]
- Badimon, L.; Suades, R.; Crespo, J.; Padro, T.; Chiva-Blanch, G. Diet microparticles and atherothrombosis. Front. Biosci. 2018, 23, 432–457. [Google Scholar] [CrossRef] [PubMed]
- Daigo, K.; Inforzato, A.; Barajon, I.; Garlanda, C.; Bottazzi, B.; Meri, S.; Mantovani, A. Pentraxins in the activation and regulation of innate immunity. Immunol. Rev. 2016, 274, 202–217. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Nahrendorf, M.; Pittet, M.J.; Swirski, F. Diversity of Denizens of the Atherosclerotic Plaque. Circulation 2008, 117, 3168–3170. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.-K.; Binder, C.J.; Torzewski, M.; Witztum, J.L. C-reactive protein binds to both oxidized LDL and apoptotic cells through recognition of a common ligand: Phosphorylcholine of oxidized phospholipids. Proc. Natl. Acad. Sci. USA 2002, 99, 13043–13048. [Google Scholar] [CrossRef]
- Kones, R. Primary prevention of coronary heart disease: Integration of new data, evolving views, revised goals, and role of rosuvastatin in management. A comprehensive survey. Drug Des. Dev. Ther. 2011, 5, 325–380. [Google Scholar] [CrossRef]
- Hattori, Y.; Matsumura, M.; Kasai, K. Vascular smooth muscle cell activation by C-reactive protein. Cardiovasc. Res. 2003, 58, 186–195. [Google Scholar] [CrossRef]
- Kawanami, D.; Maemura, K.; Takeda, N.; Harada, T.; Nojiri, T.; Saito, T.; Manabe, I.; Imai, Y.; Nagai, R. C-reactive protein induces VCAM-1 gene expression through NF-κB activation in vascular endothelial cells. Atherosclerosis 2006, 185, 39–46. [Google Scholar] [CrossRef]
- Zouki, C.; Haas, B.; Chan, J.S.D.; Potempa, L.A.; Filep, J.G. Loss of Pentameric Symmetry of C-Reactive Protein Is Associated with Promotion of Neutrophil-Endothelial Cell Adhesion. J. Immunol. 2001, 167, 5355–5361. [Google Scholar] [CrossRef]
- Khreiss, T.; József, L.; Potempa, L.A.; Filep, J.G. Loss of pentameric symmetry in C-reactive protein induces interleukin-8 secretion through peroxynitrite signalling in human neutrophils. Circ. Res. 2005, 97, 690–697. [Google Scholar] [CrossRef]
- Doronzo, G.; Russo, I.; Mattiello, L.; Trovati, M.; Anfossi, G. C-reactive protein increases matrix metalloproteinase-2 expression and activity in cultured human vascular smooth muscle cells. J. Lab. Clin. Med. 2005, 146, 287–298. [Google Scholar] [CrossRef]
- Cimmino, G.; Ragni, M.; Cirillo, P.; Petrillo, G.; Loffredo, F.; Chiariello, M.; Gresele, P.; Falcinelli, E.; Golino, P. C-reactive protein induces expression of matrix metalloproteinase-9: A possible link between inflammation and plaque rupture. Int. J. Cardiol. 2013, 168, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Slevin, M.; Rovira, N.; Turu, M.; Luque, A.; Badimon, L.; Gaffney, J.; Potempa, L.M.; Krupinski, J. Modified C-Reactive Protein is Expressed in Adventitia and Intimal Neovessels from Complicated Regions of Unstable Carotid Plaques. Open Circ. Vasc. J. 2009, 2, 23–29. [Google Scholar] [CrossRef][Green Version]
- Chen, J.; Gu, Z.; Wu, M.; Yang, Y.; Zhang, J.; Ou, J.; Zuo, Z.; Wang, J.; Chen, Y. C-reactive protein can upregulate VEGF expression to promote ADSC-induced angiogenesis by activating HIF-1α via CD64/PI3k/Akt and MAPK/ERK signaling pathways. Stem Cell Res. Ther. 2016, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; Wang, P.; Hui, R.; Edin, M.L.; Zeldin, D.C.; Wang, D.W. Adeno-Associated Virus–Mediated Human C-Reactive Protein Gene Delivery Causes Endothelial Dysfunction and Hypertension in Rats. Clin. Chem. 2009, 55, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Jialal, I.; Verma, S.; Devaraj, S. Inhibition of Endothelial Nitric Oxide Synthase by C-Reactive Protein: Clinical Relevance. Clin. Chem. 2009, 55, 206–208. [Google Scholar] [CrossRef]
- General Office of National Health Commission; General Office of National Administration of Traditional Chinese Medicine. Diagnostic and Treatment Protocol for Novel Coronavirus Pneumonia; (Trial Version 6). Available online: http://www.nhc.gov.cn/yzygj/s7653p/202002/8334a8326dd94d329df351d7da8aefc2.shtml (accessed on 21 January 2022).
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
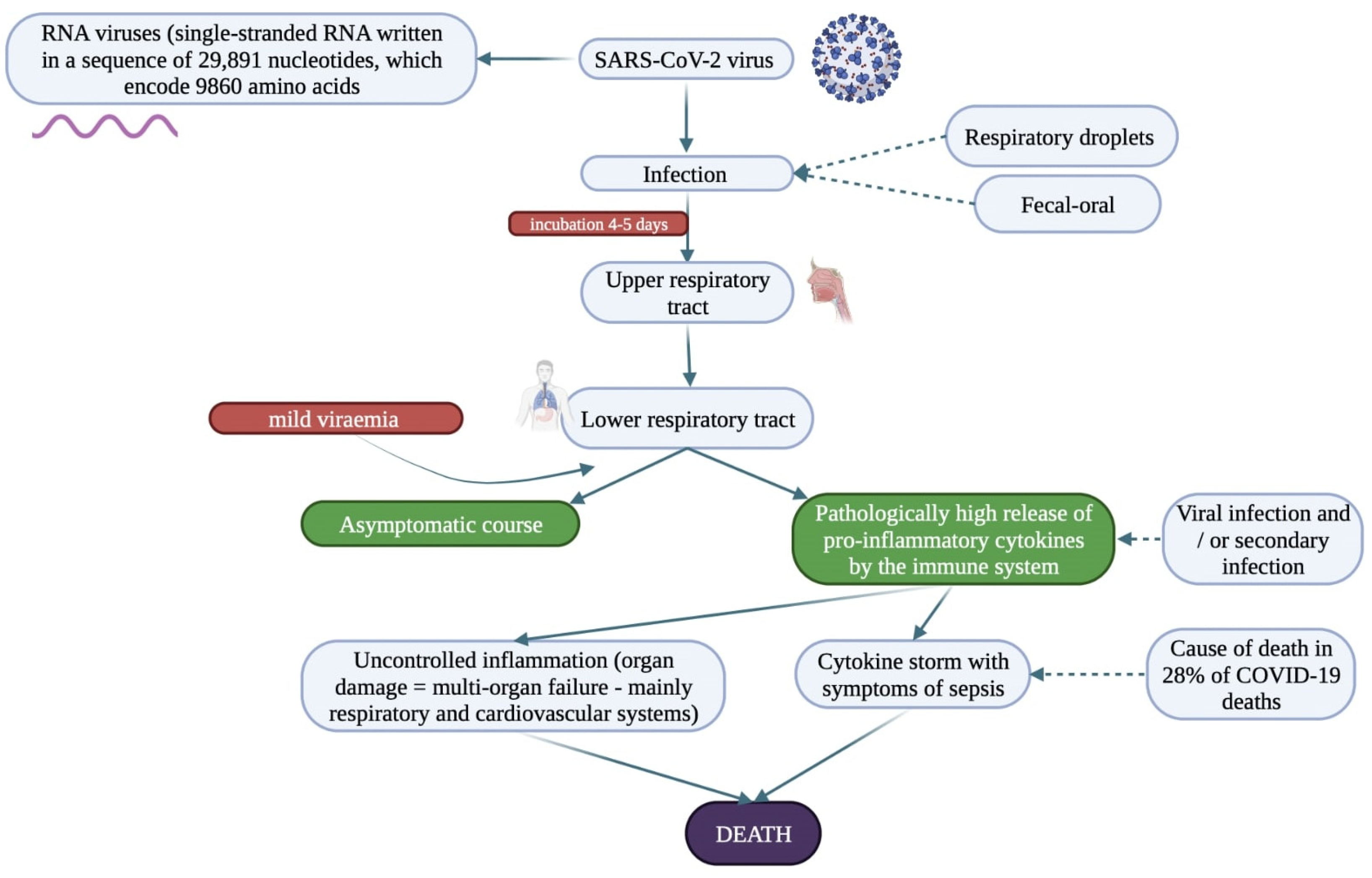
- Jin, Y.; Yang, H.; Ji, W.; Wu, W.; Chen, S.; Zhang, W.; Duan, G. Virology, Epidemiology, Pathogenesis, and Control of COVID-19. Viruses 2020, 12, 372. [Google Scholar] [CrossRef]
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Li, X.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833.e3. [Google Scholar] [CrossRef]
- Zawilska, J.; Swaczyna, T.; Masiarek, P.; Waligórska, A.; Dominiak, Z. COVID-19: Epidemiology, pathogenesis, diagnosis and clinical symptoms. Farm. Pol. 2021, 77, 166–177. [Google Scholar] [CrossRef]
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) from Publicly Reported Confirmed Cases: Estimation and Application. Ann. Intern. Med. 2020, 172, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Nile, A.; Qiu, J.; Li, L.; Jia, X.; Kai, G. COVID-19: Pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020, 53, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Darif, D.; Hammi, I.; Kihel, A.; Saik, I.E.I.; Guessous, F.; Akarid, K. The pro-inflammatory cytokines in COVID-19 pathogenesis: What goes wrong? Microb. Pathog. 2021, 153, 104799. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Geng, M.; Peng, Y.; Meng, L.; Lu, S. Molecular immune pathogenesis, and diagnosis of COVID-19. J. Pharm. Anal. 2020, 10, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhou, X.; Qiu, Y.; Song, Y.; Feng, F.; Feng, J.; Song, Q.; Jia, Q.; Wang, J. Clinical characteristics of 82 cases of death from COVID-19. PLoS ONE 2020, 15, e0235458. [Google Scholar] [CrossRef]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; Macary, P.A.; Ng, L.F.P. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020, 20, 363–374. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, J.; Liu, C.; Su, L.; Zhang, D.; Fan, J.; Yang, Y.; Xiao, M.; Xie, J.; Xu, Y.; et al. IP-10 and MCP-1 as biomarkers associated with disease severity of COVID-19. Mol. Med. 2020, 26, 97. [Google Scholar] [CrossRef]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef]
- García, L.F. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front. Immunol. 2020, 11, 1441. [Google Scholar] [CrossRef]
- Sakr, Y.; Giovini, M.; Leone, M.; Pizzilli, G.; Kortgen, A.; Bauer, M.; Tonetti, T.; Duclos, G.; Zieleskiewicz, L.; Buschbeck, S.; et al. Pulmonary embolism in patients with coronavirus disease-2019 (COVID-19) pneumonia: A narrative review. Ann. Intensiv. Care 2020, 10, 124. [Google Scholar] [CrossRef]
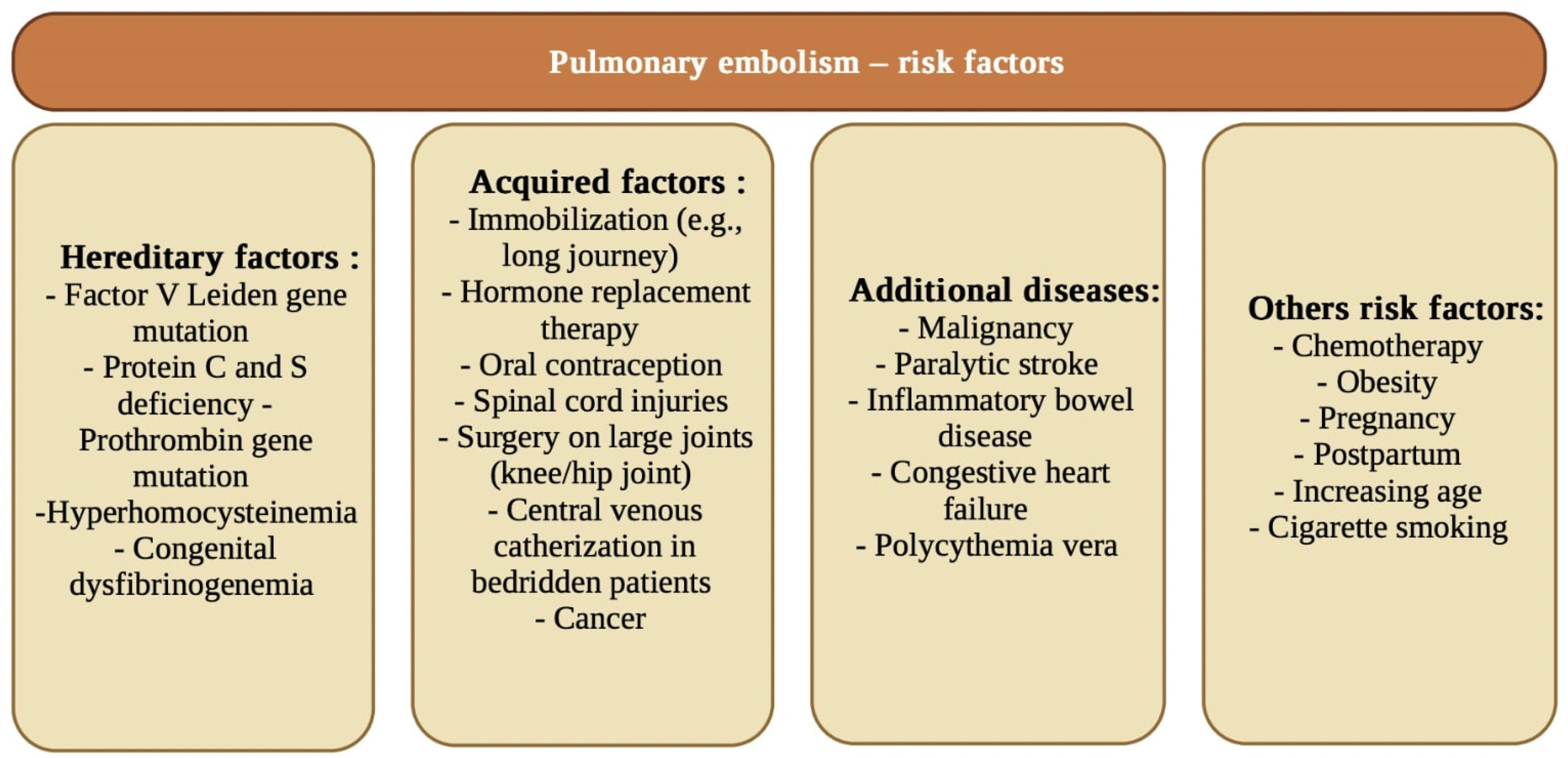
- Duru, S.; Keleșoglu, A.; Ardıç, S. State of the art paper Clinical update on pulmonary embolism. Arch. Med. Sci. 2014, 3, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Tapson, V.F. Acute Pulmonary Embolism. N. Engl. J. Med. 2008, 358, 1037–1052. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; No, H.J.; Jang, S.Y.; Kim, N.; Choi, S.H.; Kim, H.; Kim, S.-H.; Kim, H.-J.; Kim, D.-K. Hereditary Thrombophilia in Korean Patients with Idiopathic Pulmonary Embolism. Yonsei Med. J. 2012, 53, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Fauvel, C.; Weizman, O.; Trimaille, A.; Mika, D.; Pommier, T.; Pace, N.; Douair, A.; Barbin, E.; Fraix, A.; Bouchot, O.; et al. Pulmonary embolism in COVID-19 patients: A French multicentre cohort study. Eur. Heart J. 2020, 41, 3058–3068. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (2019-nCoV) Infection Is Suspected; Interim Guidance; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Prasad, A.S. Discovery of Human Zinc Deficiency: Its Impact on Human Health and Disease. Adv. Nutr. 2013, 4, 176–190. [Google Scholar] [CrossRef]
- Vogel-González, M.; Talló-Parra, M.; Herrera-Fernández, V.; Pérez-Vilaró, G.; Chillón, M.; Nogués, X.; Gómez-Zorrilla, S.; López-Montesinos, I.; Arnau-Barrés, I.; Sorli-Redó, M.; et al. Low Zinc Levels at Admission Associates with Poor Clinical Outcomes in SARS-CoV-2 Infection. Nutrients 2021, 13, 562. [Google Scholar] [CrossRef]
- Te Velthuis, A.J.W.; van den Worm, S.H.E.; Sims, A.C.; Baric, R.S.; Snijder, E.J.; Van Hemert, M.J. Zn2+ inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010, 6, e1001176. [Google Scholar] [CrossRef]
- Xue, J.; Moyer, A.; Peng, B.; Wu, J.; Hannafon, B.N.; Ding, W.-Q. Chloroquine Is a Zinc Ionophore. PLoS ONE 2014, 9, e109180. [Google Scholar] [CrossRef]
- Myint, Z.W.; Oo, T.H.; Thein, K.Z.; Tun, A.M.; Saeed, H. Copper deficiency anemia: Review article. Ann. Hematol. 2018, 97, 1527–1534. [Google Scholar] [CrossRef]
- Kumar, N. Copper Deficiency Myelopathy (Human Swayback). Mayo Clin. Proc. 2006, 81, 1371–1384. [Google Scholar] [CrossRef]
- Bagi, Z.; Hamar, P.; Kardos, M.; Koller, A. Lack of flow-mediated dilation and enhanced angiotensin II-induced constriction in skeletal muscle arterioles of lupus-prone autoimmune mice. Lupus 2006, 15, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Gomez, N.N.; Ojeda, M.S.; Gimenez, M.S. Lung lipid composition in zinc-deficient rats. Lipids 2002, 37, 291–296. [Google Scholar] [CrossRef]
- Beatty, A.L.; Malloy, J.L.; Wright, J.R. Pseudomonas aeruginosa Degrades Pulmonary Surfactant and Increases Conversion In Vitro. Am. J. Respir. Cell Mol. Biol. 2005, 32, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T. Zinc(II) Induced Neurological Thrombolytic Activities for COVID-19 Thrombus Prevention, Inflammation, Fibrin Degradation, Fibrinolysis of Dissolving Blood Clots and Blood Flow Reperfusion after Thrombolysis. Arch. Surg. Clin. Case Rep. 2021, 4, 158. [Google Scholar] [CrossRef]
- Razzaque, M.S. COVID-19 pandemic: Can zinc supplementation provide an additional shield against the infection? Comput. Struct. Biotechnol. J. 2021, 19, 1371–1378. [Google Scholar] [CrossRef]
- Szarpak, L.; Pruc, M.; Gasecka, A.; Jaguszewski, M.J.; Michalski, T.; Peacock, F.W.; Smereka, J.; Pytkowska, K.; Filipiak, K.J. Should we supplement zinc in COVID-19 patients? Evidence from meta-analysis. Pol. Arch. Intern. Med. 2021, 131, 802–807. [Google Scholar] [CrossRef]
- Percival, S.S. Copper and immunity. Am. J. Clin. Nutr. 1998, 67, 1064S–1068S. [Google Scholar] [CrossRef]
- Wazir, S.M.; Ghobrial, I. Copper deficiency, a new triad: Anemia, leucopenia, and myeloneuropathy. J. Community Hosp. Intern. Med. Perspect. 2017, 7, 265–268. [Google Scholar] [CrossRef]
- Lynch, S.M.; Klevay, L.M. Effects of a dietary copper deficiency on plasma coagulation factor activities in male and female mice. J. Nutr. Biochem. 1992, 3, 387–391. [Google Scholar] [CrossRef]
- Raha, S.; Mallick, R.; Basak, S.; Duttaroy, A.K. Is copper beneficial for COVID-19 patients? Med. Hypotheses 2020, 142, 109814. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.J.; Liu, Z.-G. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Lee, C. Therapeutic Modulation of Virus-Induced Oxidative Stress via the Nrf2-Dependent Antioxidative Pathway. Oxid. Med. Cell. Longev. 2018, 2018, e6208067. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szewc, M.; Markiewicz-Gospodarek, A.; Górska, A.; Chilimoniuk, Z.; Rahnama, M.; Radzikowska-Buchner, E.; Strzelec-Pawelczak, K.; Bakiera, J.; Maciejewski, R. The Role of Zinc and Copper in Platelet Activation and Pathophysiological Thrombus Formation in Patients with Pulmonary Embolism in the Course of SARS-CoV-2 Infection. Biology 2022, 11, 752. https://doi.org/10.3390/biology11050752
Szewc M, Markiewicz-Gospodarek A, Górska A, Chilimoniuk Z, Rahnama M, Radzikowska-Buchner E, Strzelec-Pawelczak K, Bakiera J, Maciejewski R. The Role of Zinc and Copper in Platelet Activation and Pathophysiological Thrombus Formation in Patients with Pulmonary Embolism in the Course of SARS-CoV-2 Infection. Biology. 2022; 11(5):752. https://doi.org/10.3390/biology11050752
Chicago/Turabian StyleSzewc, Monika, Agnieszka Markiewicz-Gospodarek, Aleksandra Górska, Zuzanna Chilimoniuk, Mansur Rahnama, Elżbieta Radzikowska-Buchner, Karolina Strzelec-Pawelczak, Jarosław Bakiera, and Ryszard Maciejewski. 2022. "The Role of Zinc and Copper in Platelet Activation and Pathophysiological Thrombus Formation in Patients with Pulmonary Embolism in the Course of SARS-CoV-2 Infection" Biology 11, no. 5: 752. https://doi.org/10.3390/biology11050752
APA StyleSzewc, M., Markiewicz-Gospodarek, A., Górska, A., Chilimoniuk, Z., Rahnama, M., Radzikowska-Buchner, E., Strzelec-Pawelczak, K., Bakiera, J., & Maciejewski, R. (2022). The Role of Zinc and Copper in Platelet Activation and Pathophysiological Thrombus Formation in Patients with Pulmonary Embolism in the Course of SARS-CoV-2 Infection. Biology, 11(5), 752. https://doi.org/10.3390/biology11050752