Horizontal Ridge Augmentation: A Comparison between Khoury and Urban Technique
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Eligibility Criteria
2.2.1. Inclusion Criteria
- Including at least 10 adult patients without contraindication to oral surgery who presented horizontal alveolar ridge defects requiring reconstructive procedures aimed at augmenting the alveolar ridge to allow proper placement of dental implants;
- Treated by either the Khoury or the Urban technique;
- Studies reporting an observation period of at least 4 months, analyzing volumetric changes to the alveolar ridge.
2.2.2. Exclusion Criteria
- Editorials, reviews, preclinical studies, animal studies, in vitro studies;
- Studies reporting simultaneous implant placement;
- Studies evaluating onlay block grafts placed in direct contact to the alveolar ridge or studies describing the additional use of other biomaterials or bioactive substances in combination with the above defined techniques.
2.3. Search
2.4. Data Collection
- General study, characteristics, and demographic data of subjects (author, year of publication, number of groups studied, number of subjects in each group, method of measurement of variables, age and gender distribution of subjects, history of smoking habits);
- Surgical procedures (defect localization, graft material and membrane, donor site, method to obtain the graft, post-surgical pharmacological treatment);
- Qualitative data for the assessment of possible risk of bias;
- Outcome variables of interest.
2.5. Quality Assessment
2.6. Descriptive Analysis
3. Results
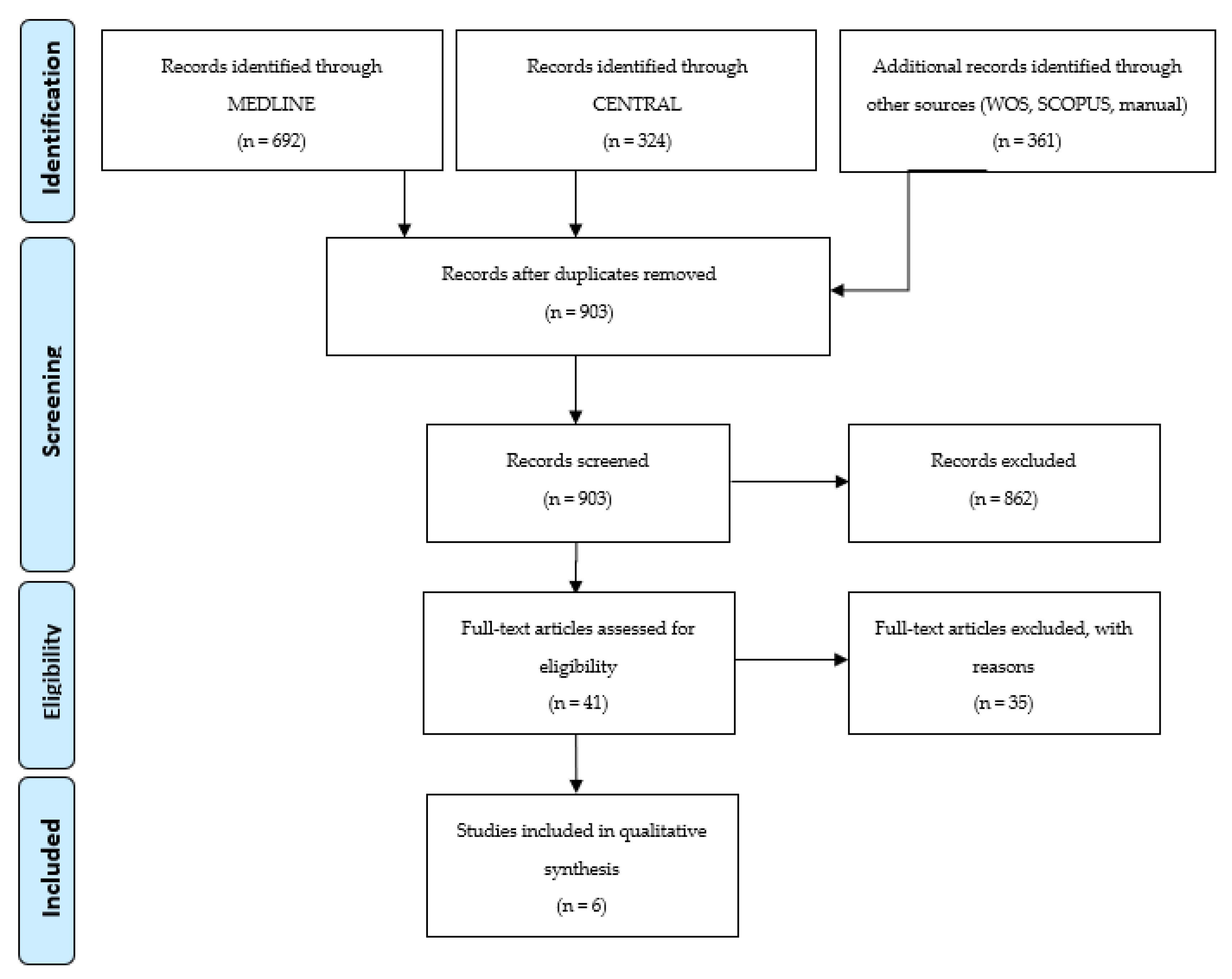
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias Assessment
3.4. Outcome Variables
3.4.1. Clinical Bone Gain (CBG)
3.4.2. Graft Resorption (R)
3.4.3. Complications (COM)
3.4.4. Histomorphometric Analysis (H)
3.4.5. Implant Survival (IS)
3.4.6. Marginal Bone Loss (MBL)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schropp, L.; Wenzel, A.; Kostopoulos, L.; Karring, T. Bone healing and soft tissue contour changes following single-tooth extraction: A clinical and radiographic 12-month prospective study. Int. J. Periodontics Restor. Dent. 2003, 23, 313–323. [Google Scholar]
- Cawood, J.I.; Howell, R.A. A classification of the edentulous jaws. Int. J. Oral Maxillofac. Surg. 1988, 17, 232–236. [Google Scholar] [CrossRef]
- Tan, W.L.; Wong, T.L.; Wong, M.C.; Lang, N.P. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin. Oral Implants Res. 2012, 23 (Suppl. 5), 1–21. [Google Scholar] [CrossRef]
- Buser, D.; Sennerby, L.; De Bruyn, H. Modern implant dentistry based on osseointegration: 50 years of progress, current trends and open questions. Periodontology 2000 2017, 73, 7–21. [Google Scholar] [CrossRef]
- Liu, J.; Kerns, D.G. Mechanisms of guided bone regeneration: A review. Open Dent. J. 2014, 8, 56–65. [Google Scholar] [CrossRef] [Green Version]
- Monje, A.; Chappuis, V.; Monje, F.; Muñoz, F.; Wang, H.L.; Urban, I.A.; Buser, D. The Critical Peri-implant Buccal Bone Wall Thickness Revisited: An Experimental Study in the Beagle Dog. Int. J. Oral Maxillofac. Implants 2019, 34, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Araújo, M.G.; Lindhe, J. Ridge alterations following tooth extraction with and without flap elevation: An experimental study in the dog. Clin. Oral Implants Res. 2009, 20, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Bengazi, F.; Botticelli, D.; Favero, V.; Perini, A.; Urbizo Velez, J.; Lang, N.P. Influence of presence or absence of keratinized mucosa on the alveolar bony crest level as it relates to different buccal marginal bone thicknesses. An experimental study in dogs. Clin. Oral Implants Res. 2014, 25, 1065–1071. [Google Scholar] [CrossRef]
- Rocchietta, I.; Fontana, F.; Simion, M. Clinical outcomes of vertical bone augmentation to enable dental implant placement: A systematic review. J. Clin. Periodontol. 2008, 35 (Suppl. 8), 203–215. [Google Scholar] [CrossRef] [PubMed]
- Darby, I.; Chen, S.; De Poi, R. Ridge preservation: What is it and when should it be considered. Aust. Dent. J. 2008, 53, 11–21. [Google Scholar] [CrossRef]
- Lucaciu, O.; Apostu, D.; Mester, A.; Campian, R.S.; Gheban, D.; Miron, R.J. Atelo-collagen type I bovine bone substitute and membrane in guided bone regeneration: A series of clinical cases and histopathological assessments. Histol. Histopathol. 2019, 34, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Apostu, D.; Lucaciu, O.; Mester, A.; Oltean-Dan, D.; Gheban, D.; Rares Ciprian Benea, H. Tibolone, alendronate, and simvastatin enhance implant osseointegration in a preclinical in vivo model. Clin. Oral Implants Res. 2020, 31, 655–668. [Google Scholar] [CrossRef]
- Wang, H.L.; Boyapati, L. “PASS” principles for predictable bone regeneration. Implant Dent. 2006, 15, 8–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khoury, F.; Hanser, T. Mandibular bone block harvesting from the retromolar region: A 10-year prospective clinical study. Int. J. Oral Maxillofac. Implants 2015, 30, 688–697. [Google Scholar] [CrossRef] [Green Version]
- Urban, I.A.; Lozada, J.L.; Jovanovic, S.A.; Nagy, K. Horizontal guided bone regeneration in the posterior maxilla using recombinant human platelet-derived growth factor: A case report. Int. J. Periodontics Restor. Dent. 2013, 33, 421–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urban, I.A.; Nagursky, H.; Lozada, J.L.; Nagy, K. Horizontal ridge augmentation with a collagen membrane and a combination of particulated autogenous bone and anorganic bovine bone-derived mineral: A prospective case series in 25 patients. Int. J. Periodontics Restor. Dent. 2013, 33, 299–307. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, 332–336. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.; Savović, J.; Page, M.J.; Elbers, R.G.; Sterne, J.A. Assessing risk of bias in a randomized trial. In Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Wiley Online Library: Hoboken, NJ, USA, 2019; pp. 205–227. [Google Scholar]
- The Joanna Briggs Institute. The Joanna Briggs Institute Reviewers’ Manual: Methodology for JBI Umbrella Reviews; The Joanna Briggs Institute: Adelaide, Australia, 2014; pp. 1–34. [Google Scholar]
- Saravanan, P.; Ramakrishnan, T.; Ambalavanan, N.; Emmadi, P.; John, T.L. Efficacy of guided bone regeneration using composite bone graft and resorbable collagen membrane in Seibert’s Class I ridge defects: Radiological evaluation. J. Oral Implantol. 2013, 39, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Meloni, S.M.; Jovanovic, S.A.; Urban, I.; Baldoni, E.; Pisano, M.; Tallarico, M. Horizontal ridge augmentation using GBR with a native collagen membrane and 1:1 ratio of particulate xenograft and autologous bone: A 3-year after final loading prospective clinical study. Clin. Implant Dent. Relat. Res. 2019, 21, 669–677. [Google Scholar] [CrossRef]
- Atef, M.; Tarek, A.; Shaheen, M.; Alarawi, R.M.; Askar, N. Horizontal ridge augmentation using native collagen membrane vs titanium mesh in atrophic maxillary ridges: Randomized clinical trial. Clin. Implant Dent. Relat. Res. 2020, 22, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Atef, M.; Osman, A.H.; Hakam, M. Autogenous interpositional block graft vs onlay graft for horizontal ridge augmentation in the mandible. Clin. Implant Dent. Relat. Res. 2019, 21, 678–685. [Google Scholar] [CrossRef]
- Bartols, A.; Kasprzyk, S.; Walther, W.; Korsch, M. Lateral alveolar ridge augmentation with autogenous block grafts fixed at a distance versus resorbable Poly-D-L-Lactide foil fixed at a distance: A single-blind, randomized, controlled trial. Clin. Oral Implants Res. 2018, 29, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Khoury, F.; Antoun, A.M.P. Bone Augmentation in Oral Implantology; Quintessence: London, UK; Berlin, Germany, 2007; pp. 115–213. [Google Scholar]
- Banodkar, A.B.; Gaikwad, R.P.; Gunjikar, T.U.; Lobo, T.A. Evaluation of accuracy of cone beam computed tomography for measurement of periodontal defects: A clinical study. J. Indian Soc. Periodontol. 2015, 19, 285–289. [Google Scholar] [CrossRef]
- Mehra, A.; Pai, K.M. Evaluation of dimensional accuracy of panoramic cross-sectional tomography, its ability to identify the inferior alveolar canal, and its impact on estimation of appropriate implant dimensions in the mandibular posterior region. Clin. Implant Dent. Relat. Res. 2012, 14, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Von Arx, T.; Buser, D. Horizontal ridge augmentation using autogenous block grafts and the guided bone regeneration technique with collagen membranes: A clinical study with 42 patients. Clin. Oral Implants Res. 2006, 17, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Mordenfeld, A.; Johansson, C.B.; Albrektsson, T.; Hallman, M. A randomized and controlled clinical trial of two different compositions of deproteinized bovine bone and autogenous bone used for lateral ridge augmentation. Clin. Oral Implants Res. 2014, 25, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.L. The functional matrix hypothesis revisited. 1. The role of mechanotransduction. Am. J. Orthod. Dentofac. Orthop. Off. Publ. Am. Assoc. Orthod. Const. Soc. Am. Board Orthod. 1997, 112, 8–11. [Google Scholar] [CrossRef]
- Cordaro, L.; Torsello, F.; Morcavallo, S.; di Torresanto, V.M. Effect of bovine bone and collagen membranes on healing of mandibular bone blocks: A prospective randomized controlled study. Clin. Oral Implants Res. 2011, 22, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- Benic, G.I.; Hämmerle, C.H. Horizontal bone augmentation by means of guided bone regeneration. Periodontology 2000 2014, 66, 13–40. [Google Scholar] [CrossRef] [PubMed]
- Dias, G.J.; Mahoney, P.; Hung, N.A.; Sharma, L.A.; Kalita, P.; Smith, R.A.; Kelly, R.J.; Ali, A. Osteoconduction in keratin-hydroxyapatite composite bone-graft substitutes. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 2034–2044. [Google Scholar] [CrossRef]
- Kübler, N. Osteoinduktion und -reparation. Mund Kiefer Gesichts Chir. 1997, 1, 2–25. [Google Scholar] [CrossRef] [PubMed]
- Buser, D.; Dula, K.; Hess, D.; Hirt, H.P.; Belser, U.C. Localized ridge augmentation with autografts and barrier membranes. Periodontology 2000 1999, 19, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Calciolari, E.; Ravanetti, F.; Strange, A.; Mardas, N.; Bozec, L.; Cacchioli, A.; Kostomitsopoulos, N.; Donos, N. Degradation pattern of a porcine collagen membrane in an in vivo model of guided bone regeneration. J. Periodontal Res. 2018, 53, 430–439. [Google Scholar] [CrossRef]
- Pérez-González, F.; Molinero-Mourelle, P.; Sánchez-Labrador, L.; Sáez-Alcaide, L.M.; Limones, A.; Cortés-Bretón Brinkmann, J.; López-Quiles, J. Assessment of clinical outcomes and histomorphometric findings in alveolar ridge augmentation procedures with allogeneic bone block grafts: A systematic review and meta-analysis. Med. Oral Patol. Oral y Cir. Bucal 2020, 25, e291–e298. [Google Scholar] [CrossRef]
- Weibull, L.; Widmark, G.; Ivanoff, C.J.; Borg, E.; Rasmusson, L. Morbidity after chin bone harvesting--a retrospective long-term follow-up study. Clin. Implant Dent. Relat. Res. 2009, 11, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Ersanli, S.; Arısan, V.; Bedeloğlu, E. Evaluation of the autogenous bone block transfer for dental implant placement: Symphysal or ramus harvesting? BMC Oral Health 2016, 16, 4. [Google Scholar] [CrossRef] [Green Version]
- Joshi, A. An investigation of post-operative morbidity following chin graft surgery. Br. Dent. J. 2004, 196, 215–218. [Google Scholar] [CrossRef] [Green Version]
- Clavero, J.; Lundgren, S. Ramus or chin grafts for maxillary sinus inlay and local onlay augmentation: Comparison of donor site morbidity and complications. Clin. Implant Dent. Relat. Res. 2003, 5, 154–160. [Google Scholar] [CrossRef]
- Chappuis, V.; Cavusoglu, Y.; Buser, D.; von Arx, T. Lateral Ridge Augmentation Using Autogenous Block Grafts and Guided Bone Regeneration: A 10-Year Prospective Case Series Study. Clin. Implant Dent. Relat. Res. 2017, 19, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Meijndert, C.M.; Raghoebar, G.M.; Meijndert, L.; Stellingsma, K.; Vissink, A.; Meijer, H.J. Single implants in the aesthetic region preceded by local ridge augmentation; a 10-year randomized controlled trial. Clin. Oral Implants Res. 2017, 28, 388–395. [Google Scholar] [CrossRef]
- Pistilli, R.; Felice, P.; Piatelli, M.; Nisii, A.; Barausse, C.; Esposito, M. Blocks of autogenous bone versus xenografts for the rehabilitation of atrophic jaws with dental implants: Preliminary data from a pilot randomised controlled trial. Eur. J. Oral Implantol. 2014, 7, 153–171. [Google Scholar] [PubMed]
- Sánchez-Labrador, L.; Molinero-Mourelle, P.; Pérez-González, F.; Saez-Alcaide, L.M.; Brinkmann, J.C.; Martínez, J.L.; Martínez-González, J.M. Clinical performance of alveolar ridge augmentation with xenogeneic bone block grafts versus autogenous bone block grafts. A systematic review. J. Stomatol. Oral Maxillofac. Surg. 2021, 122, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Nevins, M.; Mellonig, J.T.; Clem, D.S., 3rd; Reiser, G.M.; Buser, D.A. Implants in regenerated bone: Long-term survival. Int. J. Periodontics Restor. Dent. 1998, 18, 34–45. [Google Scholar]
- Moussa, N.T.; Dym, H. Maxillofacial Bone Grafting Materials. Dent. Clin. N. Am. 2020, 64, 473–490. [Google Scholar] [CrossRef] [PubMed]
- Mehl, C.; Kern, M.; Meinke, N.; Açil, Y.; Bähr, T.; Wiltfang, J.; Gaßling, V. Can one-wall bone defects be augmented with xenogenic bone grafting material alone? J. Cranio-Maxillo-Facial Surg. Off. Publ. Eur. Assoc. Cranio-Maxillo-Facial Surg. 2016, 44, 1137–1142. [Google Scholar] [CrossRef]
- Khojasteh, A.; Esmaeelinejad, M.; Aghdashi, F. Regenerative Techniques in Oral and Maxillofacial Bone Grafting. In A Textbook of Advanced Oral and Maxillofacial Surgery; IntechOpen: London, UK, 2015; Volume 2. [Google Scholar] [CrossRef]
| PICOS | Description |
|---|---|
| Patient (P) | Patiens with horizontal bone deficiences in the alveolar crest |
| Intervention (I) | Horizontal bone regeneration with the Split bone block technique described by Khoury. |
| Comparation (C) | Horizontal bone regeneration with the Sausage technique desbribed by Urban. |
| Outcome (O) | Clinical and histological efficacy of both techniques
|
| Study type (S) |
| Authors | Year | Study Design | Intervention Test (GBR/SBB) | Intervention Control | Donor Site | Recipient Site | Xenograft | Membrane | Graft Harvesting Technique | Age Range/Mean ± SD | Man/Woman | Tobacco (No Smokers/Smokers) | Antibiotics | NSAIDs |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Saravanan P et al. | 2013 | Prospective case series | GBR | n/a | Symphysis | Max | BioOss | BioMend | Scraper | 20–50 | n/a | No smokers | Amox 500 mg | Ibuprofen 400 mg |
| Urban IA et al. | 2013 | Prospective case series | GBR | n/a | n/a | Post Max + Mand | ABBM | CM | n/a | 52.7 ± 11.4 | 10/15 | No smokers | n/a | n/a |
| Bartols A et al. | 2018 | Randomized clinical trial | SBB | GBR | External oblique line | Ant Max | n/a | n/a | Disc | 19–72/51.5 ± 17.3 | 7/8 | n/a | Amox 750 mg | Ibuprofen 400 mg |
| Meloni SM et al. | 2019 | Prospective case series | GBR | n/a | Retromolar | Post Max + Mand | ABBM | CM | Scraper | 24–78/56.8 | 7/11 | 10/8 | Amox 1 g | n/a |
| Atef et al. | 2019 | Randomized clinical trial | SBB | SBB onlay block | Symphysis | Post Mand | n/a | n/a | Disc | 29–54/42.1 | 9/11 | n/a | Amox/ácido clavulanico 1 g | ibuprofen 600 mg |
| Atef M et al. | 2020 | Randomized clinical trial | GBR | GBR with titanium mesh | External oblique line / Symphysis | Max | ABBM | CM | Trephine | n/a | n/a | n/a | Amox/clavulanic acid 1 g | Ibuprofen 600 mg |
| Study | Technique | Width in T = 0 | T Observation | Width in T = 1 | Horizontal Gain | Resorption |
|---|---|---|---|---|---|---|
| Saravanan 2013 | Urban | 2 mm: 3.63 ± 0.29 mm | 6 months | 2 mm: 5.07 ± 0.25 mm | 1.44 ± 0.09 mm | NA |
| 4 mm: 4.1 ± 0.18 mm | 4 mm: 5.51 ± 0.21 mm | 1.41 ± 0.08 mm | ||||
| 6 mm: 4.47 ± 0.25 mm | 6 mm: 5.88 ± 0.27 mm | 1.41 ± 0.08 mm | ||||
| Urban 2013 | Urban | 2 mm: 2.19 ± 0.64 mm | 8.9 ± 2.1 months | 2 mm: 7.87 ± 1.61 mm | 5.68 ± 1.42 mm | NA |
| Bartols 2018 | Khoury | Pre: 2.67 ± 0.61 mm | 12 months | 6.60 ± 1.18 mm | 3.93 mm | 2.33 mm |
| Post: 8.93 ± 1.05 mm | ||||||
| Meloni 2019 | Urban | 3.07 ± 0.64 mm | 7 months | 8.09 ± 2.16 mm | 5.03 ± 2.15 mm | NA |
| Atef 2019 | Khoury | Pre: 3.85 ± 0.6 mm | 4 months | 8.84 ± 0.54 mm | 5.02 ± 0.8 mm | 0.36 mm |
| Post: 9.22 ± 0.64 mm | ||||||
| Atef 2020 | Urban | Pre: 3.3 ± 0.4 mm | 6 months | 7.3 ± 0.9 mm | 3.9 ± 0.9 mm | 0.6 mm |
| Post: 7.9 ± 0.6 mm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Sánchez, J.; Pickert, F.N.; Sánchez-Labrador, L.; GF Tresguerres, F.; Martínez-González, J.M.; Meniz-García, C. Horizontal Ridge Augmentation: A Comparison between Khoury and Urban Technique. Biology 2021, 10, 749. https://doi.org/10.3390/biology10080749
Sánchez-Sánchez J, Pickert FN, Sánchez-Labrador L, GF Tresguerres F, Martínez-González JM, Meniz-García C. Horizontal Ridge Augmentation: A Comparison between Khoury and Urban Technique. Biology. 2021; 10(8):749. https://doi.org/10.3390/biology10080749
Chicago/Turabian StyleSánchez-Sánchez, Javier, Finn Niclas Pickert, Luis Sánchez-Labrador, Francisco GF Tresguerres, José María Martínez-González, and Cristina Meniz-García. 2021. "Horizontal Ridge Augmentation: A Comparison between Khoury and Urban Technique" Biology 10, no. 8: 749. https://doi.org/10.3390/biology10080749
APA StyleSánchez-Sánchez, J., Pickert, F. N., Sánchez-Labrador, L., GF Tresguerres, F., Martínez-González, J. M., & Meniz-García, C. (2021). Horizontal Ridge Augmentation: A Comparison between Khoury and Urban Technique. Biology, 10(8), 749. https://doi.org/10.3390/biology10080749





