In Vivo Monitoring of Acetylcholine Release from Nerve Endings in Salivary Gland
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Chemicals
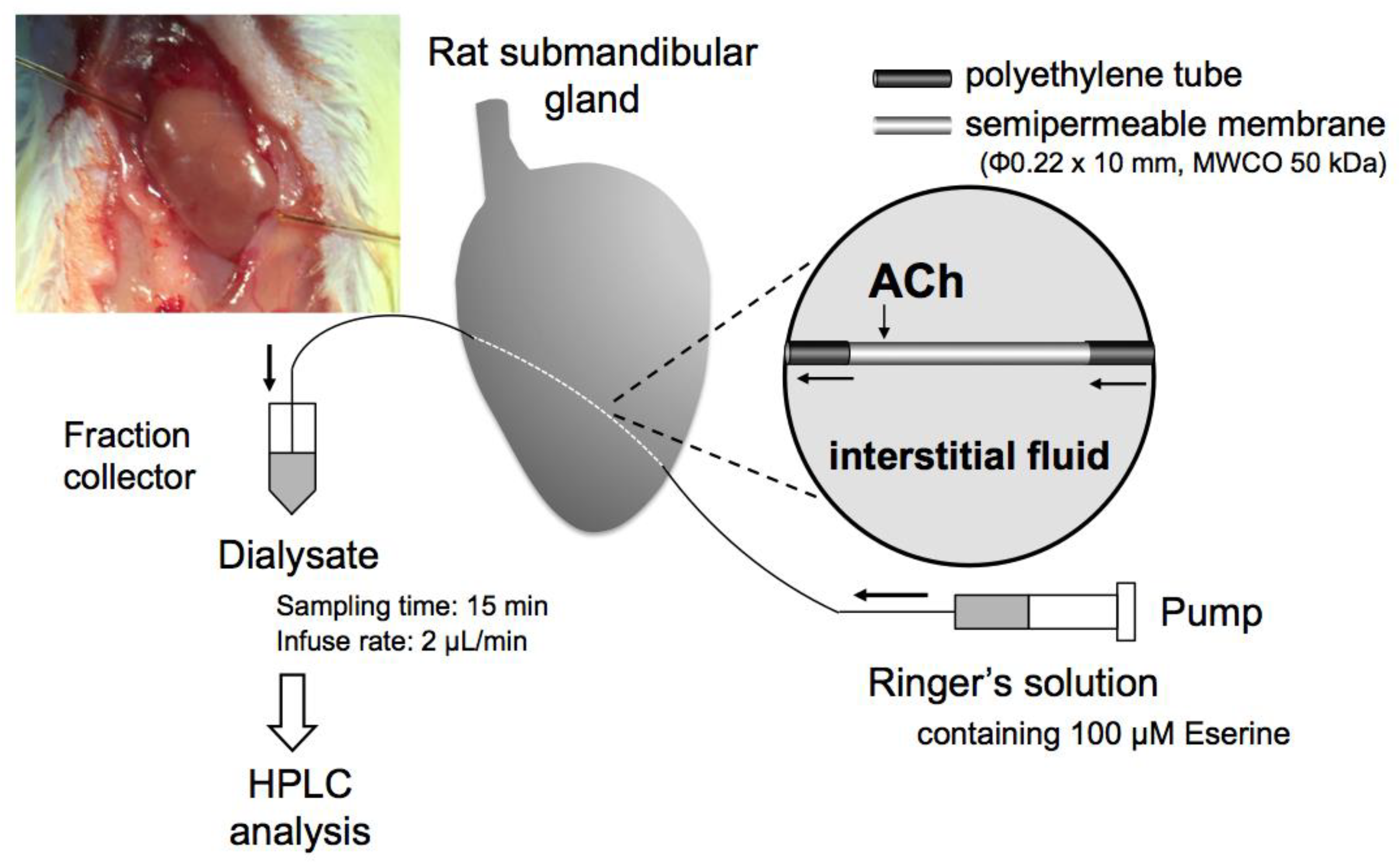
2.3. Microdialysis in Anesthetized Rats
2.4. Acethylcholine Determination
2.5. Electrical Stimulation of Parasympathetic Nerve
2.6. Statistical Analyses
3. Results
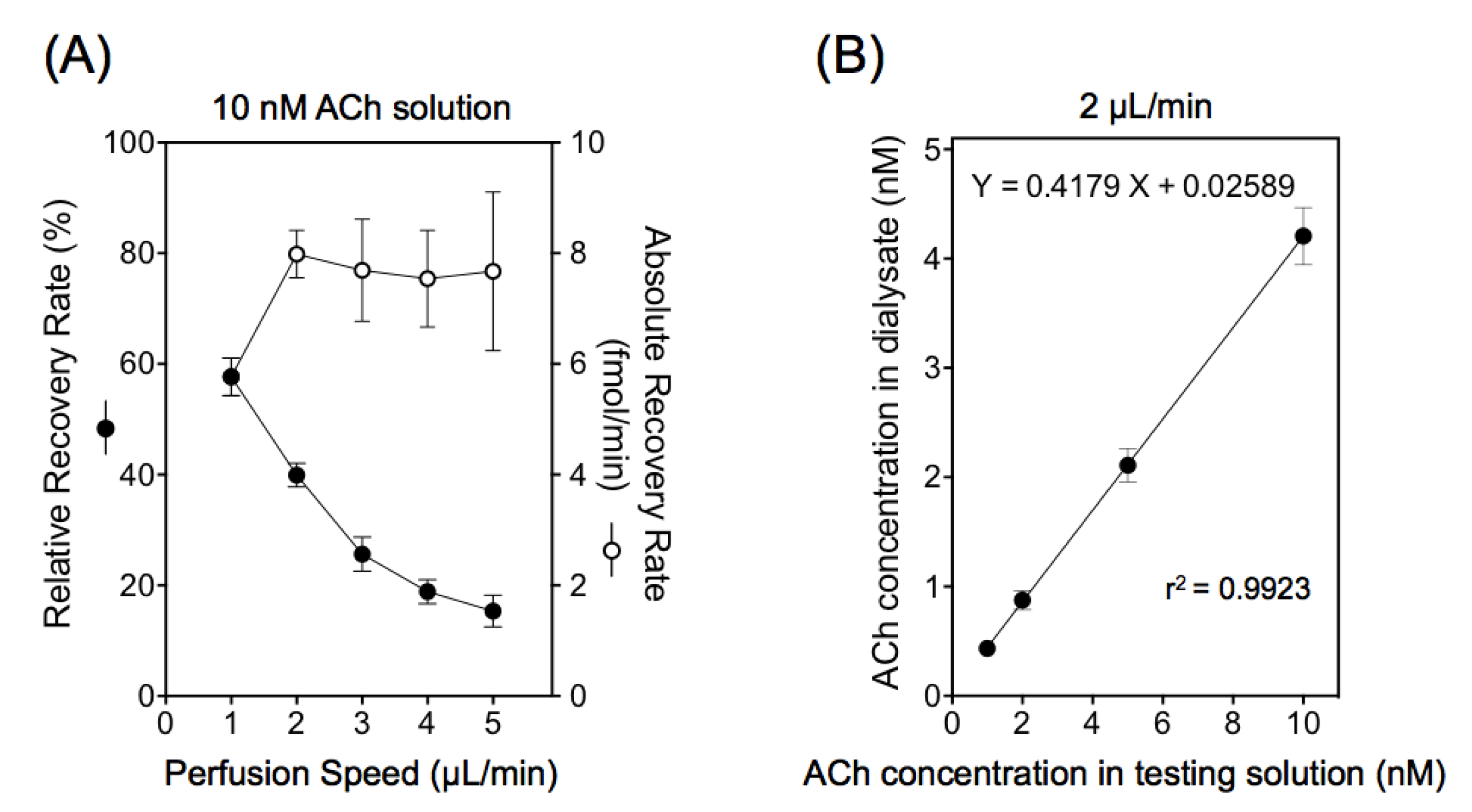
3.1. Microdialysis Condition
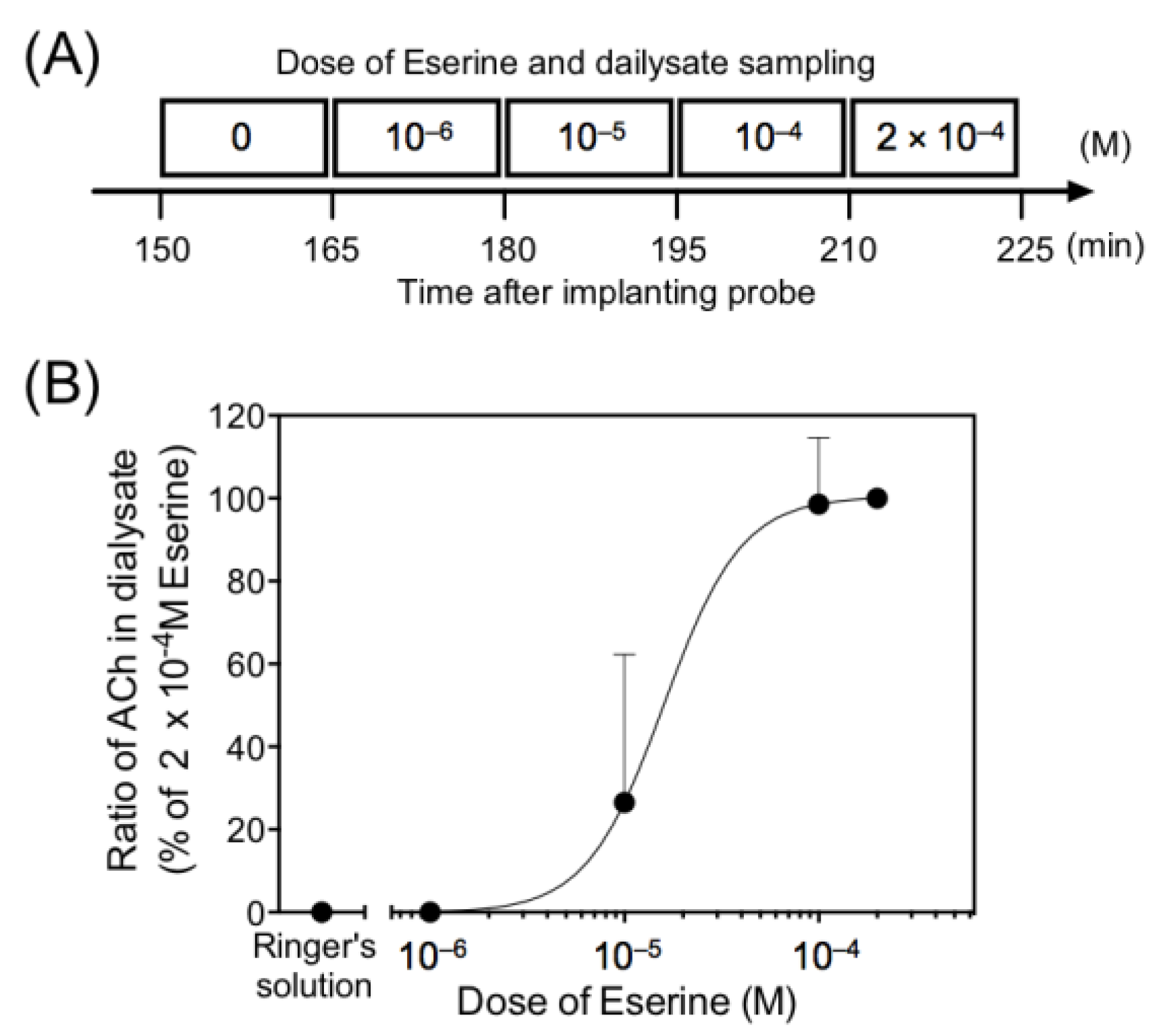
3.2. Effect of Eserine on Basal Level of Acetylcholine
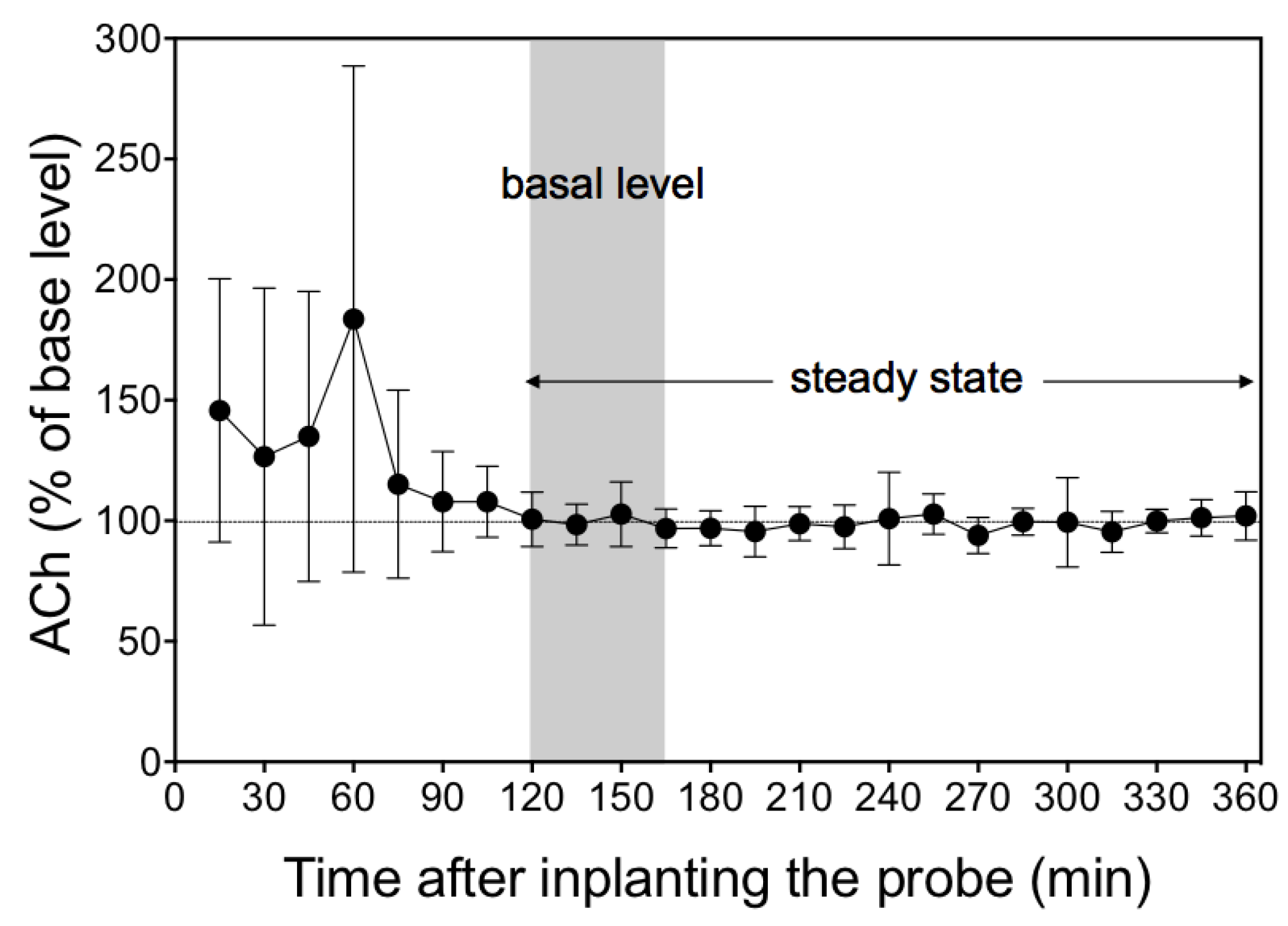
3.3. Time Course of Dialysate Acetylcholine Levels after Implantation of Probe
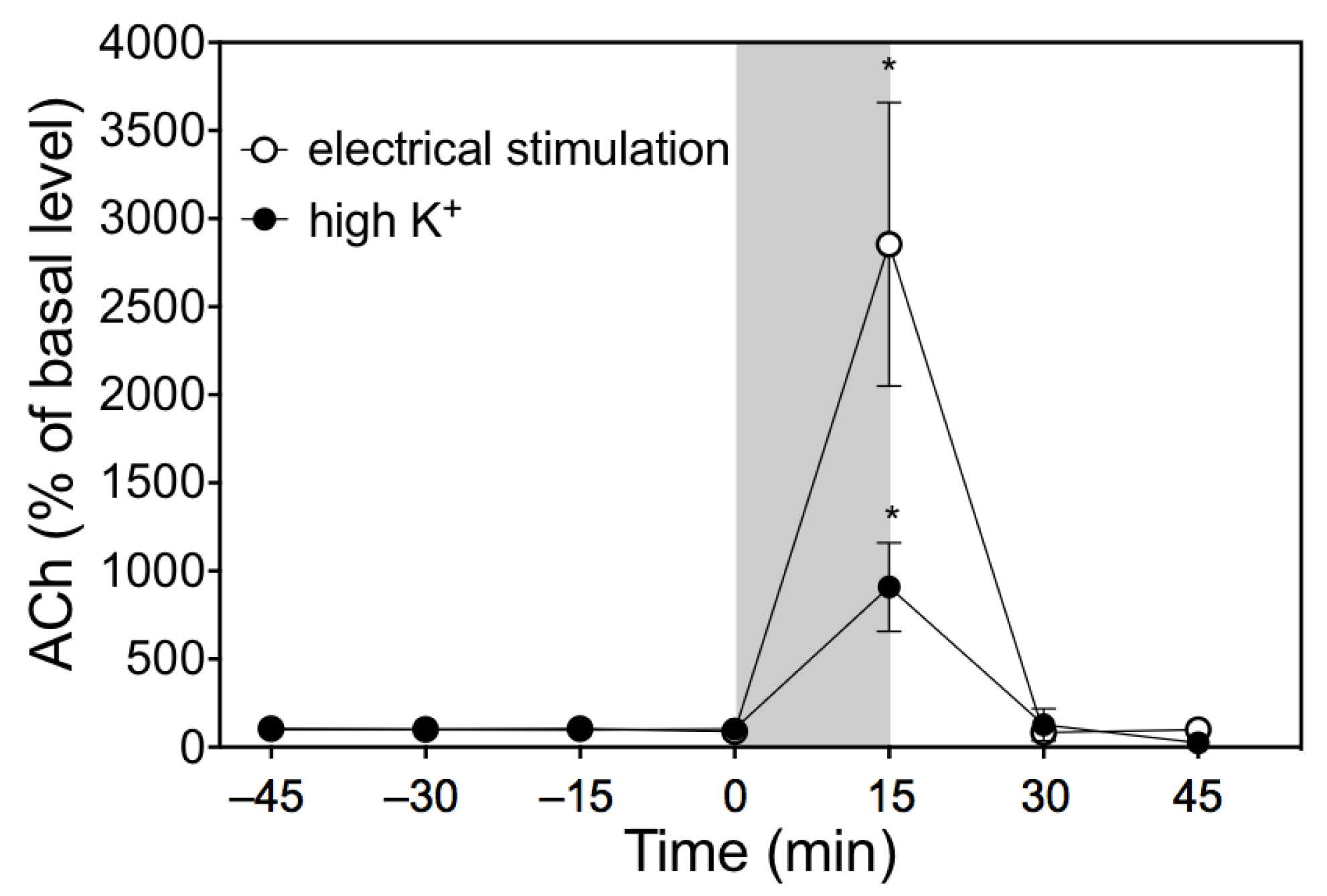
3.4. Response to Electrical Stimulation of Chorda Tympani Nerve
3.5. Response to High-K+ Ringer’s Solution
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matsui, M.; Motomura, D.; Karasawa, H.; Fujikawa, T.; Jiang, J.; Komiya, Y.; Takahashi, S.; Taketo, M.M. Multiple functional defects in peripheral autonomic organs in mice lacking muscarinic acetylcholine receptor gene for the M3 subtype. Proc. Natl. Acad. Sci. USA 2000, 97, 9579–9584. [Google Scholar] [CrossRef]
- Bymaster, F.P.; Carter, P.A.; Yamada, M.; Gomeza, J.; Wess, J.; Hamilton, S.E.; Nathanson, N.M.; McKinzie, D.L.; Felder, C.C. Role of specific muscarinic receptor subtypes in cholinergic parasympathomimetic responses, in vivo phosphoinositide hydrolysis, and pilocarpine-induced seizure activity. Eur. J. Neurosci. 2003, 17, 1403–1410. [Google Scholar]
- Nakamura, T.; Matsui, M.; Uchida, K.; Futatsugi, A.; Kusakawa, S.; Matsumoto, N.; Nakamura, K.; Manabe, T.; Taketo, M.M.; Mikoshiba, K. M(3) muscarinic acetylcholine receptor plays a critical role in parasympathetic control of salivation in mice. J. Physiol. 2004, 558, 561–575. [Google Scholar] [CrossRef] [PubMed]
- Baum, B.J. Neurotransmitter control of secretion. J. Dent. Res. 1987, 66, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Melvin, J.E.; Yule, D.; Shuttleworth, T.; Begenisich, T. Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Ann. Rev. Physiol. 2005, 67, 445–469. [Google Scholar] [CrossRef] [PubMed]
- Proctor, G.; Carpenter, G. Regulation of salivary gland function by autonomic nerves. Auton. Neurosci. 2007, 133, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Proctor, G.B. Muscarinic receptors and salivary secretion. J. Appl. Physiol. 2006, 100, 1103–1104. [Google Scholar] [CrossRef]
- Gautam, D.; Heard, T.S.; Cui, Y.; Miller, G.; Bloodworth, L.; Wess, J. Cholinergic stimulation of salivary secretion studied with M1 and M3 muscarinic receptor single- and double-knockout mice. Mol. Pharmacol. 2004, 66, 260–267. [Google Scholar] [CrossRef]
- Murai, S.; Saito, H.; Masuda, Y.; Itoh, T. Sex-dependent differences in the concentrations of the principal neurotransmitters, noradrenaline and acetylcholine, in the three major salivary glands of mice. Arch. Oral Biol. 1998, 43, 9–14. [Google Scholar] [CrossRef]
- Murai, S.; Saito, H.; Masuda, Y.; Itsukaichi, O.; Itoh, T. Basal levels of noradrenaline, dopamine, 5-hydroxytryptamine, and acetylcholine in the submandibular, parotid, and sublingual glands of mice and rats. Arch. Oral Biol. 1995, 40, 663–668. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Murai, S.; Saito, H.; Itoh, T. Changes in the noradrenaline and acetylcholine content of three major salivary glands and in the salivation and protein component patterns of whole saliva in chronically isoprenaline-administered mice. Arch. Oral Biol. 1997, 42, 225–234. [Google Scholar] [CrossRef]
- Murai, S.; Saito, H.; Masuda, Y.; Nakamura, K.; Michijiri, S.; Itoh, T. Effects of short-term (2 weeks) streptozotocin-induced diabetes on acetylcholine and noradrenaline in the salivary glands and secretory responses to cholinergic and adrenergic sialogogues in mice. Arch. Oral Biol. 1996, 41, 673–677. [Google Scholar] [CrossRef]
- Compton, J.; Martinez, J.R.; Martinez, A.M.; Young, J.A. Fluid and electrolyte secretion from the isolated, perfused submandibular and sublingual glands of the rat. Arch. Oral Biol. 1981, 26, 555–561. [Google Scholar] [CrossRef]
- Murakami, M.; Sugiya, H.; Puxeddu, R.; Loffredo, F.; Riva, A. Dose-dependent morphological changes of intercellular canaliculi during stimulation with carbachol and isoproterenol in the isolated rat submandibular gland. Eur. J. Morphol. 2003, 41, 89–92. [Google Scholar] [CrossRef]
- Kanno, T.; Asada, N.; Yanase, H.; Iwanaga, T.; Ozaki, T.; Nishikawa, Y.; Iguchi, K.; Mochizuki, T.; Hoshino, M.; Yanaihara, N. Salivary Secretion of Highly Concentrated Chromogranin a in Response to Noradrenaline and Acetylcholine in Isolated and Perfused Rat Submandibular Glands. Exp. Physiol. 1999, 84, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Okubo, M.; Kawaguchi, M. Rat submandibular gland perfusion method for clarifying inhibitory regulation of GABAA receptor. J. Pharmacol. Sci. 2013, 122, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, R.; Kobashi, M.; Fujita, M. Electrophysiological study on sensory nerve activity from the submandibular salivary gland in rats. Brain Res. 2018, 1680, 137–142. [Google Scholar] [CrossRef]
- Damsma, G.; Westerink, B.H.C.; de Vries, J.B.; Van den Berg, C.J.; Horn, A.S. Measurement of Acetylcholine Release in Freely Moving Rats by Means of Automated Intracerebral Dialysis. J. Neurochem. 1987, 48, 1523–1528. [Google Scholar] [CrossRef]
- Imperato, A.; Di Chiara, G. Trans-striatal dialysis coupled to reverse phase high performance liquid chromatography with electrochemical detection: A new method for the study of the in vivo release of endogenous dopamine and metabolites. J. Neurosci. 1984, 4, 966–977. [Google Scholar] [CrossRef]
- Akiyama, T.; Yamazaki, T. Adrenergic inhibition of endogenous acetylcholine release on postganglionic cardiac vagal nerve terminals. Cardiovas. Res. 2000, 46, 531–538. [Google Scholar] [CrossRef]
- Akiyama, T.; Yamazaki, T.; Ninomiya, I. In vivo detection of endogenous acetylcholine release in cat ventricles. Am. J. Physiol. Heart Circ. Physiol. 1994, 266, H854–H860. [Google Scholar] [CrossRef]
- Ungerstedt, J.; Nowak, G.; Ericzon, B.G.; Ungerstedt, U. Intraperitoneal microdialysis (IPM): A new technique for monitoring intestinal ischemia studied in a porcine model. Shock 2003, 20, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Matthiessen, P.; Strand, I.; Jansson, K.; Tnquist, C.; Andersson, M.; Rutegerd, J.; Norgren, L. Is early detection of anastomotic leakage possible by intraperitoneal microdialysis and intraperitoneal cytokines after anterior resection of the rectum for cancer? Dis. Colon Rectum 2007, 50, 1918–1927. [Google Scholar] [CrossRef] [PubMed]
- Sugisawa, M.; Takai, N. The Role of Substance P in Parasympathetic Nerve-induced Secretion in the Rat Submandibular Gland. J. Osaka Dent. Univ. 1991, 25, 51–62. [Google Scholar] [PubMed]
- Fujii, T.; Yamada, S.; Yamaguchi, N.; Fujimoto, K.; Suzuki, T.; Kawashima, K. Species differences in the concentration of acetylcholine, a neurotransmitter, in whole blood and plasma. Neurosci. Lett. 1995, 201, 207–210. [Google Scholar] [CrossRef]
- Liu, J.K.; Kato, T. Effect of physostigmine on relative acetylcholine output induced by systemic treatment with scopolamine in in vivo microdialysis of rat frontal cortex. Neurochem. Int. 1994, 24, 589–596. [Google Scholar] [CrossRef]
- Topilko, A.; Caillou, B. Fine structural localization of acetylcholinesterase activity in rat submandibular gland. J. Histochem. Cytochem. 1985, 33, 439–445. [Google Scholar] [CrossRef]
- Di Chiara, G. In-vivo brain dialysis of neurotransmitters. Trends Pharmacol. Sci. 1990, 11, 116–121. [Google Scholar] [CrossRef]
- Benveniste, H.; Hansen, A.J.; Ottosen, N.S. Determination of Brain Interstitial Concentrations by Microdialysis. J. Neurochem. 1989, 52, 1741–1750. [Google Scholar] [CrossRef]
- Joukhadar, C.; Muller, M. Microdialysis. Clin. Pharmacokinet. 2005, 44, 895–913. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshikawa, M.; Kawaguchi, M. In Vivo Monitoring of Acetylcholine Release from Nerve Endings in Salivary Gland. Biology 2021, 10, 351. https://doi.org/10.3390/biology10050351
Yoshikawa M, Kawaguchi M. In Vivo Monitoring of Acetylcholine Release from Nerve Endings in Salivary Gland. Biology. 2021; 10(5):351. https://doi.org/10.3390/biology10050351
Chicago/Turabian StyleYoshikawa, Masanobu, and Mitsuru Kawaguchi. 2021. "In Vivo Monitoring of Acetylcholine Release from Nerve Endings in Salivary Gland" Biology 10, no. 5: 351. https://doi.org/10.3390/biology10050351
APA StyleYoshikawa, M., & Kawaguchi, M. (2021). In Vivo Monitoring of Acetylcholine Release from Nerve Endings in Salivary Gland. Biology, 10(5), 351. https://doi.org/10.3390/biology10050351




