Tachyporinae Revisited: Phylogeny, Evolution, and Higher Classification Based on Morphology, with Recognition of a New Rove Beetle Subfamily (Coleoptera: Staphylinidae)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. General Outline of Study
2.2. Taxon Sampling and Deposition of Material
2.3. Microscopy, Imaging, Measurements, and Terminology
2.4. Selection of Characters
2.5. Phylogenetic Analyses
3. Results
3.1. Phylogenetic Analyses
3.1.1. Maximum Parsimony Analysis
3.1.2. Character Optimization
3.2. Systematic Part
3.2.1. Subfamily Tachyporinae MacLeay sensu nov.
- Tribe Tachyporini MacLeay, 1825 [40] sensu nov.
- Tribe Vatesini Seevers, 1958 [13] sensu nov.
- Tribe Deropini Smetana, 1983 [23]
- Tribe Tachinusini Fleming, 1821 [45] stat. rev., sensu nov.
3.2.2. Tribe Tachyporini MacLeay, 1825 sensu nov.
- Subtribe Tachyporina MacLeay, 1825 [40] stat. nov., sensu nov.
- Subtribe Euconosomatina Cameron, 1918 [44] stat. rev., sensu nov.
3.2.3. Subtribe Tachyporina MacLeay, 1825 stat. nov., sensu nov.
- Lamprinodes Luze, 1901 [57]: 181.Type species: Tachyporus saginatus Gravenhorst, 1806 [58]: 6.
- Lamprinus Heer, 1839 [59]: 286.
- Palporus Campbell, 1979 [61]: 11 stat. nov.Type species: Staphylinus nitidulus Fabricius, 1781 [62]: 337.
- Symmixus Bernhauer, 1915 [63]: 56.Type species: Symmixus sikkimensis Bernhauer, 1915 [63]: 57.
- Type species: Staphylinus chrysomelinus Linnaeus, 1758 [38]: 423.
3.2.4. Genus Tachyporus Gravenhorst, 1802 sensu nov.
3.2.5. Genus Palporus Campbell, 1979 stat nov.
- nitidulus (Fabricius, 1781 [62]: 337), comb. nov. (Staphylinus). Distribution: Palaearctic, Oriental, and Afrotropical Regions; intro. Nearctic and Australian Regions.
- neomexicanus (Campbell, 1979 [61]: 14), comb. nov. (Tachyporus). Distribution: USA (New Mexico, Colorado).
3.2.6. Subtribe Euconosomatina Cameron, 1918 stat. rev., sensu nov.
- Euconosoma Cameron, 1918 [44]: 215.
- †Palaeosepedophilus Paśnik & Kubisz, 2002 [82]: 357.Type species: †Palaeosepedophilus succinicus Paśnik & Kubisz, 2002 [82]: 357.
3.2.7. Tribe Vatesini Seevers, 1958 sensu nov.
- Cilea Jacquelin du Val, 1856 [93]: 25.Type species: Staphylinus silphoides Linnaeus, 1767 [94]: 684.
- Cileoporus Campbell, 1994 [91]: 126.Type species: Tachinus politus Bernhauer, 1923 [95]: 59.
- Coproporus Kraatz, 1857 [96]: 399.Type species: Coproporus colchicus Kraatz, 1858 [97]: cxc.
- Coprotachinus Cameron, 1933a [98]: 44.
- Mimocyptus Cameron, 1919 [101]: 241.Type species: Mimocyptus globulus Cameron, 1919 [101]: 241.
- Tachinoporus Cameron, 1928 [11]: 447.Type species: Tachinoporus basalis Cameron, 1928 [11]: 448.
- Tachinoproporus Cameron, 1928 [11]: 449.Type species: Tachinoproporus ferrugineus Cameron, 1928 [11]: 449.
- Termitoplus Silvestri, 1946 [12]: 547.Type species: Termitoplus grandis Silvestri, 1946 [12]: 551.
- Vatesus Sharp, 1876 [92]: 201.Type species: Vatesus latitans Sharp, 1876 [92]: 202.
- †Procileoporus Yamamoto, 2016 [46]: 165.Type species: †Procileoporus burmiticus Yamamoto, 2016 [46]: 167.
3.2.8. Genus Tachinoporus Cameron, 1928
3.2.9. Tachinoporus basalis Cameron, 1928
3.2.10. Genus Tachinoproporus Cameron, 1928
3.2.11. Tachinoproporus ferrugineus Cameron, 1928
3.2.12. Tribe Deropini Smetana, 1983
- Derops Sharp, 1889 [105]: 418
3.2.13. Tribe Tachinusini Fleming, 1821 stat. rev., sensu nov.
- Austrotachinus Steel, 1956 [110]: 13.Type species: Austrotachinus fuscipes Steel, 1956 [110]: 14.
- Lacvietina Herman, 2004 [25]: 39.Type species: Lacvietina cuprina Herman, 2004 [25]: 44.
- Leucotachinus Coiffait & Sáiz, 1968 [111]: 411.Type species: Tachinus luteonitens Fairmaire & Germain, 1862 [112]: 425.
- Megarthropsis Cameron, 1919 [101]: 231.Type species: Megarthropsis decorata Cameron, 1919 [101]: 232.
- Nepaliodes Coiffait, 1977 [113]: 272.Type species: Nepaliodes variolosa Coiffait, 1977 [113]: 272.
- Nitidotachinus Campbell, 1993 [109]: 522.Type species: Tachinus tachyporoides Horn, 1877 [114]: 94.
- Olophrinus Fauvel, 1895 [115]: 280.Type species: Olophrinus striatus Fauvel, 1895: 281.
- Peitawopsis Smetana, 1992 [116]: 199.Type species: Peitawopsis monticola Smetana, 1992 [116]: 204.
- Pseudotachinus Cameron, 1932 [117]: 398.Type species: Pseudotachinus niger Cameron, 1932 [117]: 398.
- Tachinomorphus Kraatz, 1859 [118]: 54.Type species: Tachinus fulvipes Erichson, 1840 [119]: 921.
- Tachinoplesius Bernhauer, 1936 [120]: 326.Type species: Tachinoplesius turneri Bernhauer, 1936 [120]: 327.
- Tachinus Gravenhorst, 1802 [41]: 134.Type species: Staphylinus rufipes Linnaeus, 1758 [38]: 423.
- -
- Subgenus Latotachinus Ullrich, 1975 [121]: 282.Type species: Tachinus punctiventris Sharp, 1888 [122]: 385.
- -
- Subgenus Tachinoderus Motschulsky, 1858 [123]: 217.Type species: Tachinoderus longicornis Motschulsky, 1858 [123]: 218.
- -
- Subgenus Tachinus Gravenhorst, 1802 [41]: 134.Type species: Staphylinus rufipes Linnaeus, 1758 [38]: 423.
- †Hesterniasca Zhang, Wang & Xu, 1992 [124]: 279.Type species: Hesterniasca obesa Zhang, Wang & Xu, 1992 [124]: 279.
- †Mesotachinus Tikhomirova, 1968 [50]: 148.Type species: †Mesotachinus major Tikhomirova, 1968 [50]: 148.
- †Protachinus Cai, Yan, Beattie, Wang & Huang, 2013 [52]: 651.Type species: Protachinus minor Cai, Yan, Beattie, Wang & Huang, 2013 [52]: 652.
3.2.14. Tachyporinae, incertae sedis: List of Genera with Unknown Systematic Placements within Tachyporinae sensu nov.
- †Abscondus Tikhomirova, 1968 [50]: 151.Type species: Abscondus regularis Tikhomirova, 1968 [50]: 152.
- †Tachyporoides Tikhomirova, 1968 [50]: 150.Type species: Tachyporoides villosus Tikhomirova, 1968 [50]: 151.
3.2.15. Subfamily Mycetoporinae Thomson, 1859 stat. nov.
- Bobitobus Tottenham, 1939 [140]: 225 stat. rev.Type species: Staphylinus lunulatus Linnaeus, 1767 [94]: 684.
- Bolitobius Leach, 1819 [141]: 176.Type species: Megacronus castaneus Stephens, 1832 [142]: 166.
- Bolitopunctus Campbell, 1993 [33]: 29.Type species: Bryoporus muricatulus Hatch, 1957 [143]: 127.
- Bryophacis Reitter, 1909 [144]: 102.Type species: Bolitobius rufus Erichson, 1839 [145]: 407.
- Bryoporus Kraatz, 1857 [96]: 452.Type species: Tachinus cernuus Gravenhorst, 1806 [58]: 31.
- Canariobolitobius Schülke, 2004 [146]: 1024.Type species: Bolitobius filicornis Wollaston, 1864 [147]: 560.
- Carphacis Gozis, 1886 [148]: 14.Type species: Staphylinus striatus Olivier, 1795 [149]: (42): 28.
- Ischnosoma Stephens, 1829 [150]: 22.Type species: Tachinus splendidus Gravenhorst, 1806 [58]: 24.
- Lordithon Thomson, 1859 [136]: 47 sensu nov.
- Mycetoporus Mannerheim, 1831 [137]: 476.Type species: Tachinus punctus Gravenhorst, 1806 [58]: 27.
- Neobolitobius Campbell, 1993 [33]: 35.Type species: Lordithon varians Hatch, 1957 [143]: 128.
- Parabolitobius Li, Zhao & Sakai, 2000 [152]: 11.Type species: Megacronus prolongatus Sharp, 1888 [122]: 460.
- †Cuneocharis Ryvkin, 1990 [153]: 64.Type species: †Cuneocharis elongatus Ryvkin, 1990 [153]: 65.
- †Glabrimycetoporus Yue, Zhao & Ren, 2009 [154]: 64.Type species: †Glabrimycetoporus amoenus Yue, Zhao & Ren, 2009 [154]: 67.
- †Undiatina Ryvkin, 1990 [153]: 62.Type species: †Undiatina pilosa Ryvkin, 1990 [153]: 63.
3.2.16. Genus Bobitobus Tottenham, 1939 stat. rev.
- arcuatus (Solsky, 1871 [159]: 238), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Russia (East & West Siberia, Far East), Japan (Hokkaidô), South Korea, China (Jilin).
- femoralis (Cameron, 1932 [117]: 339), comb. nov. (Bolitobius). Distribution: India (Sikkim, Darjeeling).
- imitator (Luze, 1901 [166]: 735), comb. nov. (Bolitobius). Distribution: Russia (Siberia), Japan (Honshû).
- kelleyi (Malkin, 1944 [170]: 26), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Canada, USA.
- longiceps (LeConte, 1863 [161]: 32), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Canada, USA.
- lunulatus (Linnaeus, 1767 [94]: 684), comb. nov. (Staphylinus). Distribution: Europe, Russia, Caucasus.
- maacki (Solsky, 1871 [159]: 236), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Russia (East Siberia).
- nigricollis (J. Sahlberg, 1880 [178]: 104), comb. nov. (Bolitobius). Distribution: Russia (Siberia).
- pulchellus (Mannerheim, 1830 [137]: 64), comb. rev. (Bolitobius). Distribution: Europe, Ukraine, Russia (European, East Siberia, Far East).
- puncticeps (Luze, 1901 [166]: 733), comb. nov. (Bolitobius). Distribution: Russia (Siberia), China (Heilongjiang).
- rostratus (Motschulsky, 1860 [183]: 573), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Russia (Siberia), Armenia, Turkey.
- semirufus (Sharp, 1888 [122]: 457), comb. nov. (Bolitobius). Distribution: Japan, South Korea, China.
- speciosus (Erichson, 1839 [145]: 277), comb. nov. (Bolitobius, cited as Boletobius). Distribution: Europe, Russia (European, Siberia).
3.3. Identification Keys
3.3.1. Key to Subfamilies of Tachyporine Group of Subfamilies
- Body various, sometimes distinctive; antennae inserted to vertex, posterior to anterior margins of each eye ([189]: Figure 1.13.1); elytra with posterolateral margins frequently sinuate (cf. Figures 25A and 30C: 83-1, 83-2); elytral epipleural keel absent; male paramere with velum [190,191]; female without gonocoxites (except Gymnusini) … Aleocharinae
- -
- Body narrowly elongate, fusiform, or sublimuloid; antennae inserted to anterior margins of each eye; elytra with posterolateral margins rarely sinuate; elytral epipleural keel present OR absent; male paramere simple, lacking velum; female with well-sclerotized gonocoxites … 2
- 2.
- Pronotal postcoxal process separated basally by a suture ([16]: Figure 63.22) … Olisthaerinae
- -
- Pronotal postcoxal process not separated basally by a suture … 3
- 3.
- -
- 4.
- Neck with strong constriction, distinct all around; elytral epipleural keel absent … Phloeocharinae
- -
- Neck without constriction (sometimes with narrowing); elytral epipleural keel present (Figure 52C: 80-0) … 5
- 5.
- Metacoxa uniformly covered with extended ventral lamella (or plates, vlmtc; Figure S3B) … 6
- -
- Metacoxa without OR with small ventral lamella (see Supplementary Figures S2 and S3A) … 7
- 6.
- 7.
- Body fusiform, slender (Figures 62 and 63); head with distinct and complete ridge below eye (Figure 67A, C: 13-2); elytron with longitudinally raised sutural edge (Figures 65C and 66A: 78-1); metacoxae extremely large (Figure 70A); male parameres widely separated from each other, setose (Figure 72D–F) … Mycetoporinae stat. nov.
- -
- Body usually sublimuloid, except Derops (Figure 43A,B); head without ridge below eye (Figure 26A: 13-0) (rarely with short, incomplete ridge: Leucotachinus and Nepaliodes; Figures 53A and 58D: 13-1); elytron without longitudinally raised sutural edge (Figure 7D: 78-0); metacoxae medium to small (Figure 52B); male parameres very closely appressed to median lobes (Figure 33E,G), glabrous (Tachinomorphus with minute setulae; Figure 56C: 148-2) … Tachyporinae sensu. nov.
3.3.2. Key to Tribes of Tachyporinae
- Habitus distinctive (Figure 43A,B); pronotum strongly constricted in posterior half (Figure 44A: 49-1) … Deropini
- -
- Habitus usually sublimuloid; pronotum not constricted in posterior half… 2
- 2.
- Elytra pubescent (Figures 7B and 15C: 74-2); antenna basally without clear borderline of dense and fine recumbent setae (Figure 9D: 21-0); maxillary palpomere 3 setose, longer than palpomere 4 (Figures 10A–D and 16E: 32-1) … Tachyporini sensu nov.
- -
- Forebody glabrous (not covered with long ground setae); antenna basally with clear borderline of dense and fine recumbent setae (Figures 26D and 59C: 21-1, 21-3); maxillary palpomere 3 glabrous (except Mimocyptus, Leucotachinus), shorter than palpomere 4 (except Mimocyptus, Vatesus) (Figures 27A, 45D, 53C and 59A: 32-2, 32-3, 34-2) … 3
- 3.
- Antennae longer than width of pronotum; elytron much longer than wide (length/width: >1.3×); pronotal hypomeron with long and projecting postcoxal process (Figures 54G and 59D,E: 61-5); male sternite VII (s7) with posterior margin emarginate, bearing peg-like setae (most) or short and stout setae (Figures 52D, 55A and 61A: 131-1, 131-2) … Tachinusini stat. rev., sensu nov.
- -
- Antennae usually shorter than width of pronotum (except Cilea, Cileoporus, and Tachinoporus) (Figure 23B: 20-1); elytron usually slightly longer than wide (length/width: <1.3×, except Cilea, Cileoporus, and Tachinoporus) (Figure 30A,B: 70-2); pronotal hypomeron with short and rounded postcoxal process (Figure 27E–G: 61-1); male sternite VII with posterior margin usually truncate (occasionally emarginate, i.e., Cilea), without peg-like setae or stout setae (Figure 25C: 131-0) … Vatesini sensu nov.
3.3.3. Key to Subtribes and Extant Genera of Tachyporini
- Forebody densely pubescent (Figure 15B, C); protibia with a row of comb-like close-spaced spines along outer margin (Figure 17B,C: 97-1); pronotum without arranged macrosetae; metatarsus very long, much longer than whole length of metatibia (Figure 15D: 103-1); abdomen without paratergites (Figure 15D: 124-2) … 2 (Euconosomatina stat. rev., sensu nov.)
- -
- Forebody not entirely pubescent, usually glabrous on head and pronotum (Figure 7A–D); protibia without a row of comb-like close-spaced spines along outer margin; pronotum with arranged macrosetae (Figures 7A and 8A); metatarsus long, but clearly shorter than whole length of metatibia (Figure 7E: 103-0); abdomen with single pair of paratergites (Figure 7E: 124-0) … 3 (Tachyporina stat. nov., sensu nov.)
- 2.
- Labial palpomere 3 distinctly expanded, crescent-shaped ([44]: Figure 1) … Euconosoma
- -
- Labial palpomere 3 not expanded, conical (Figure 16F) … Sepedophilus
- 3.
- Maxillary palpomere 3 widest at or near apex (Figure 10C,D: 30-0) … 4
- -
- Maxillary palpomere 3 widest around middle, not at or near apex (Figure 10A,B: 30-1) … 5
- 4.
- Body strongly convex in cross-section (Figure 6B); antenna modified, distinctly short, as long as head width (Figures 7C and 9C: 19-2, 20-1) … Lamprinus
- -
- Body not strongly convex in cross-section (Figures 6A and 7D); antenna less modified, much longer than head width (Figure 6A) … Lamprinodes
- 5.
- Metatarsomere 3 with ventrally modified projection ([22]: Figure 18); pronotum widest between basal 1/5 and basal 1/4 (Figure 8A: 50-2); elytra with long and thick macrosetae (Figure 8A,D) …Symmixus
- -
- Metatarsomere 3 normal, without ventrally modified projection; pronotum not widest between basal 1/5 and basal 1/4; elytra with inconspicuous macrosetae (Figure 7B) … 6
- 6.
- Body small (length: <1.4 mm [61]), narrow, very slender (Figure 6C); maxillary palpomere 4 short, but very thick, widened at base (Figure 10B and Supplementary Figure S1A); pronotum widest between basal 1/4 to middle (Figure 7B: 50-3); elytron without epipleural gutter along outer margin (Figure 7B: 79-0) … Palporus stat. nov.
- -
- Body small to medium (length: ≥1.4 mm), somewhat slender (Figure 6F); maxillary palpomere 4 narrowly elongate, much narrower (Figure 10A); pronotum widest between base and basal 1/5; elytron with very narrow and inconspicuous epipleural gutter along outer margin (cf. Figure 7D: 79-1) … Tachyporus sensu nov.
3.3.4. Key to Extant Genera of Vatesini
- Elytron moderately elongate (length/width: 1.3–1.8×, if dissected) (Figure 21B); pronotum widest between basal 1/4 to middle (Figure 37B) … 2
- -
- Elytron only slightly elongate (length/width: <1.3×, if dissected) (Figure 30A,B); pronotum widest between base and basal 1/5 … 4
- 2.
- Body very slender, streamlined (Figures 21B and 36C); abdominal terminalia with extremely long macrosetae (Figures 21B and 39A–E); tergite VIII and sternite VIII narrowly elongate, with short and indistinct lobes (if present, see Figure 39A,D,E); male tergite IX with each apex elongate, not modified to two lobes ([91]: Figure 62) … 3
- -
- Body not slender, sublimuloid (Figure 21A); abdominal terminalia without extremely long macrosetae (Figures 31A and 32A,C); tergite VIII and sternite VIII only weakly elongate to transverse, with long and distinct lobes (Figures 31A, 32A, C); male tergite IX with each apex modified to two lobes (Figure 33A: 122-3) … Cilea
- 3.
- Pronotum and elytra with erect macrosetae (Figures 37B and 38A–C); pronotal disc with shallow and very narrow longitudinal sulcus along midline (Figure 37B, lsp); distributed in Oriental region… Tachinoporus
- -
- Pronotum and elytra without erect macrosetae (Figure 21B); pronotal disc without longitudinal sulcus along midline; distributed in Central and South America … Cileoporus
- 4.
- Antennae and legs with distinct modifications (Figures 26G and 29B,D: 19-2, 93-1) … Vatesus
- -
- Antennae and legs without distinct modifications … 5
- 5.
- Body minute (length: ca. 1.2 mm) (Figure 21G); midcranial suture absent (Figure 23A: 11-0); male tegite IX with basal 1/3 dorsally contiguous (Figure 33C: 120-0) … Mimocyptus
- -
- Body small to large, not minute; midcranial suture present (Figure 23B: 11-1) … 6
- 6.
- Forebody matte, not glossy (Figures 40C–E and 41D,E); mesoventrite with extremely large plate-like keel (Figure 40E,F, pkmsv); abdomen in basal half without blackish macrosetae (Figure 40C); male aedeagus with conspicuous spines (Figure 42B, dsp) … Tachinoproporus
- -
- Forebody glossy; mesoventrite without plate-like keel; abdomen in basal half with blackish macrosetae (Figure 21C–F,H); male aedeagus without spines … 7
- 7.
- Body strongly convex dorsally (Figure 21H); head much smaller than pronotum; antennae shorter than width of head (Figure 23B) … Termitoplus
- -
- Body not strongly convex dorsally (Figure 21C–E); head moderately smaller than pronotum; antennae longer than width of head (Figure 21C–F) … 8
- 8.
- Body rather flattened dorsally (Figure 21E); forebody with bluish metallic sheen; mesoventrite without any trace of longitudinal carina (Figure 24B: 85-0); distributed only in Africa … Coprotachinus
- -
- Body moderately convex dorsally (Figure 21C,D); forebody without bluish metallic sheen; mesoventrite with weakly raised longitudinal carina (Figures 24C, 28B: 85-1); distribution cosmopolitan … Coproporus
3.3.5. Key to Extant Genera of Tachinusini
- Head and pronotum with deep, pit-like punctation (Figure 58A–C); clypeal anterior margin weakly to strongly reflexed upward (Figure 58A–C: 7-1, 7-2) … 2 (members of the former Megarthropsini)
- -
- Head and pronotum without deep, pit-like punctation (Figure 51); clypeal anterior margin not reflexed upward (Figure 50: 7-0) … 5
- 2.
- Habitus distinctive (Figure 57D, E), with highly and very strongly extended pronotal and elytral margins (Figure 58); antenna with apically tapered scape (Figure 58B,C) … Nepaliodes
- -
- Habitus less distinctive (Figure 57A,C,F), with slightly to strongly extended pronotal and elytral margins (Figure 58A); antenna with subparallel-sided scape (Figure 59C) … 3
- 3.
- Metaventrite with intermesocoxal pit adjacent to apex of mesosternal process (Figure 60A, B: 89-1) … Lacvietina
- -
- Metaventrite without intermesocoxal pit … 4
- 4.
- Head with postocular vertical carina (Figure 58D: 13-1); antennae long, very slender, reaching beyond posterior margin of elytra (Figure 57C) … Megarthropsis
- -
- Head without postocular vertical carina; antennae short, slender, reaching only to near middle of elytra (Figure 57F) … Peitawopsis
- 5.
- Abdomen setose with macrosetae on tergites IV–VII (Figure 48D); head without any type of neck or narrowing (Figure 51A: 12-0) … Nitidotachinus
- -
- Abdomen usually without macrosetae on tergites IV–VII; head with neck or narrowing (Figures 50 and 51B,D: 12-3) … 6
- 6.
- Elytron with distinct rows of punctures (Figures 48F, 51D); male tergite IX with each apex distinctly modified to two lobes (Figure 55F: 122-3) … Olophrinus
- -
- Elytron usually without distinct rows of punctures (Figure 51C); male tergite IX with each apex not modified to two separate lobes (Figure 55E, G) … 7
- 7.
- Elytron long, concealing more than half of abdomen (Figure 48B,C); head with a pair of ridges basolaterally along underside of eyes (Figure 53A: 13-1); mesoventrite with transverse ridge, lying near base of intercoxal process (Figure 52A and 54E: 86-1) … Leucotachinus
- -
- Elytron moderately long, not concealing half of abdomen; head without a pair of ridges basolaterally along underside of eyes; mesoventrite without transverse ridge … 8
- 8.
- Elytra covered with reduced, modified setae (Figure 48A); ligula with pair of medial lobes ([110]: Figure 2); distributed in Australia … Austrotachinus
- -
- Elytra not covered with reduced, modified setae; ligula without pair of medial lobes; distribution nearly all outside Australia … 9
- 9.
- Pronotum widest at base (Figures 49C and 51C); main part of mesoventrite with strongly raised longitudinal carina (Figures 52C and 54C: 85-1); male parameres densely covered with minute sensilla or filiform setulae (Figure 56C: 148-2) … Tachinomorphus
- -
- Pronotum widest between basal 1/4 to middle (Figure 51B: 50-3); main part of mesoventrite without strongly raised longitudinal carina (Figures 52B and 54F: 85-0); male parameres without minute sensilla … 10
- 10.
- Head with narrow furrows along inner edge of eyes (Figures 50B and 51B); pronotum with furrows near anterolateral margins (Figure 51B: 52-1); mesoventrite with longitudinal carina on mesoventral process and its basal area only ([193]: Figure 2E,F) … Pseudotachinus
- -
- Head without narrow furrows along inner edge of eyes; pronotum usually without furrows near anterolateral margins (shallow and inconspicuous, if present) … 11
- 11.
- Pronotum with rather strongly sinuate posterior margin (Figure 49F); scutellum markedly enlarged, but with exceptions [194] (Figure 49F); mesoventrite with deep longitudinal furrow along mesoventral process (Figure 52B); male sternite VII with posterior margin very broadly weakly emarginate, bearing inconspicuous modified setae ([194]: Figure 3A,B); distributed in Africa … Tachinoplesius
- -
- Pronotum with truncate, rounded, or only weakly sinuate posterior margin; scutellum small to medium (Figure 49D,E); mesoventrite without deep longitudinal furrow along mesoventral process; male sternite VII with posterior margin usually somewhat narrowly strongly emarginate, bearing conspicuous modified setae, frequently furnished with dozens of strong peg-like setae; widespread, but not in Africa … Tachinus
3.3.6. Key to Extant Genera of Mycetoporinae
- Head distinctly elongate, with long tempora, lacking ocular setae (Figures 62A and 64D: 6-0); antenna basally distinctly separated from base of mandible by anterior lobe of gena (Figure 64D); antennomere 1 very long and slender (Figure 64D); maxillary palpomeres 3 and 4 combined long and slender (Figure 68D) … Bobitobus stat. rev.
- -
- Head not distinctly elongate, usually only weakly to moderately elongate (Figure 67A,B), with short tempora, mostly having ocular setae (Figure 64A–C: 6-2); antenna basally contiguous with base of mandible (Figure 64A); antennomere 1 less elongate, thicker (Figure 68F,G); maxillary palpomeres 3 and 4 combined usually somewhat shorter and wider (Figure 68A,B,D) … 2
- 2.
- Maxillary palpomere 4 narrower and slender, not beyond more than 1/2 as wide as penultimate palpomere (Figure 68C) … 3
- -
- Maxillary palpomere 4 thicker, more than 1/2 as wide as penultimate palpomere (Figure 68A,B,D) … 4
- 3.
- Meso- and metatibial apices bordered by ctenidium of evenly arranged dense equal length spines with two larger and one smaller apical spur ([32]: Figures 103–105); antennae long, even longer than head and pronotum combined (Figure 63D); maxillary palpomeres 2 and 3 densely finely pubescent ([32]: figures 38 and 39); scutellum with basal carina linear ([32]: Figures 76, 77) … Ischnosoma
- -
- Meso- and metatibial apices bordered by more irregularly arranged numerous unequal spines ([32]: Figures 99–102); maxillary palpomeres 2 and 3 sparsely, coarsely pubescent ([32]: Figures 31–37); antennae shorter than head and pronotum combined (Figure 63F); scutellum with basal carina acutely pointed medially ([32]: Figures 74 and 75) … Mycetoporus
- 4.
- Meso- and metatibial apices with ctenidium of equal length spines, plus 2–3 long spurs, forming a straight edge ([33]: Figures 103–105) … 5
- -
- Meso- and metatibial apices with numerous unequal spines, forming a jagged edge ([33]: Figures 99–102) … 6
- 5.
- Elytron with 5–6 irregular longitudinal rows of setigerous punctures (Figure 65D); male sternite VIII with patch of 2 or 3 pairs of oblique setae near middle of posterior margin (Figure 71F: 134-1); abdomen rather strongly tapered posteriorly (Figure 62F) … Bryoporus
- -
- 6.
- Elytron evenly densely punctate (Figure 62C); labial palpomere 1 short, strongly transverse ([33]: Figure 91) … Bolitopunctus
- -
- Elytron not evenly densely punctate; labial palpomere 1 weakly transverse to elongate … 7
- 7.
- Labial palpomere 3 longer than labial palpomeres 1 and 2 combined (Figure 67F) … 8
- -
- Labial palpomere 3 shorter than labial palpomeres 1 and 2 combined (cf. Figure 67E) … 10
- 8.
- Antenna with preapical antennomeres broadly transverse ([138]: Figure 8); abdomen sub-parallel sided in basal 2/3 (Figures 63A,C and 66A) … Carphacis
- -
- Antenna rather slender, with preapical antennomeres not broadly transverse; abdomen tapering posteriorly … 9
- 9.
- Body large (length: 5.6–10.8 mm [16]); antenna without clear micropubescence borderline, density increasing apically; maxillary palpomere 3 pubescent; pronotum and elytra with dense waves of microsculapture visible only under high magnification (above 100x) [16,33]; elytron usually with more than 6 irregular longitudinal rows of setigerous punctures; male sternite VII with distinct patterns of modified setae (Figure 71G: 134-2); distributed in Holarctic region … Bolitobius
- -
- Body medium to relatively large (length: <5.6 mm [16,33]); antenna with clear micropubescence borderline, lacking dense and fine recumbent setae on basal four antennomeres ([33]: Figure 104); maxillary palpomere 3 nearly glabrous; pronotum and elytra with distinct waves of coarse, transverse microsculapture easily visible with low magnification (24x) [16,33]; male sternite VII without distinct patterns of modified setae; distributed in North America only … Neobolitobius
- 10.
- Maxillary palpomere 3 moderately coarsely pubescent ([33]: Figures 99 and 100); maxillary palpomere 4 shorter than penultimate palpomere ([33]: Figures 99 and 100); labial palpomere 1 very large, even larger than labial palpomeres 2 and 3 combined ([33]: Figures 87 and 88); scutellum with basal carina distinctly divided medially ([33]: Figure 130) … Bryophacis
- -
- Maxillary palpomere 3 with sparse setae only; maxillary palpomere 4 equal in length or longer than penultimate palpomere (Figure 68D); labial palpomere 1 large, but shorter and smaller than labial palpomeres 2 and 3 combined; scutellum with basal carina obtusely convex in middle ([157]: Figure 59) … 11
- 11.
- Head with long, strong ocular setae; elytra with rather conspicuous microreticulation; abdomen sub-parallel sided in basal 2/3; distributed in Canary Islands … Canariobolitobius
- -
- Head with rather short, inconspicuous ocular setae (rarely absent, e.g., L. lewisi [158]); elytra without microreticulation; abdomen strongly tapering posteriorly; distribution cosmopolitan … Lordithon sensu nov.
4. Discussion
4.1. Non-Monophyly of Tachyporinae and Tachyporini
4.2. Revised Status of Mycetoporini as a Subfamily
4.3. New Higher Classification of Tachyporinae
4.4. Sister Group of Tachyporinae
4.5. Sister Group of Mycetoporinae
4.6. Evolutionary Origins and Fossil Records
5. Conclusions
- Non-monophyly of current Tachyporinae and Tachyporini is again supported. To improve the higher classification of this subfamily, Mycetoporini is removed from Tachyporinae and considered as a newly recognized rove beetle subfamily Mycetoporinae. I reclassify Tachyporinae into four monophyletic tribes: Tachyporini, Vatesini, Deropini, and Tachinusini. The tribe Megarthropsini is synonymized under Tachinusini. Within Tachyporini, two subtribes are newly established: Tachyporina and Euconosomatina. The resulting topology is as follows: Tachyporini + (Vatesini + (Deropini + Tachinusini)).
- Most tachyporine and mycetoporine genera, including disjunctly distributed Leucotachinus, are found to be monophyletic, except Lordithon, Sepedophilus, and Tachyporus. With some taxonomic changes, all genera of both subfamilies appear to be monophyletic for now. After direct examination of the two enigmatic extant genera Tachinoporus and Tachinoproporus, both are indeed true tachyporines and should be considered valid genera. A total of 36 (†7) genera of Tachyporinae and 16 (†4) genera of Mycetoporinae are recognized here.
- The carrion beetle family Silphidae (Silpha) and Tachyporinae might be sister groups, whereas Mycetoporinae forms a sister group relationship with Olisthaerinae. These results support the polyphyly of the Tachyporine Group of subfamilies consistent with several previous studies (e.g., [6,7,196]). Nevertheless, further study is needed to corroborate these hypotheses.
- Re-examination of fossil records of both Tachyporinae and Mycetoporinae dramatically changes the known evolutionary history of each taxon: Tachinusini (~ Upper Jurassic, but likely Middle Jurassic [51]), Vatesini (~ mid-Cretaceous), and Tachyporini (~ Upper Cretaceous). Deropini so far has no fossil record, but must have originated in the Late Jurassic, or earlier, in accordance with its sister tribe Tachinusini. In contrast, the origin of Mycetoporinae can be reliably traced back only to the Early Cretaceous.
- My backbone phylogeny of Tachyporinae may play an important role for evolutionary ecology or inventory surveys in the future. For example, semi-aquatic taxa are found only in the Deropini + Tachinusini clade, while myrmecophily and termitophily occur only in Vatesini and Tachyporini. Mycetoporinae species are frequently associated with fresh mushrooms. The clarification of morphological adaptations of these taxa would be an interesting and valuable subject for further study.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Additional Note
Appendix B. List of Characters
- Head:
- Antennae.
- Mouthparts.
- Thorax and legs.
- Abdomen.
- Genitalia.
Appendix C. Table
| New Classification | Former Tribal Assignment | Described Species | Distribution |
|---|---|---|---|
| Tachyporini MacLeay, 1825 [40] sensu nov. | 510 (†6) | AUS, ETH, MAD, NEA, NEO, OCE, ORI, PAL | |
| -Subtribe Tachyporina MacLeay, 1825 [40] stat. nov., sensu nov. | 144 (†4) | NEA, NEO, ORI, PAL | |
| 1. Lamprinodes Luze, 1901 [57] | Tachyporini | 3 | PAL |
| 2. Lamprinus Heer, 1839 [59] | Tachyporini | 1 | PAL |
| 3. Palporus Campbell, 1979 [61] stat. nov. | Tachyporini | 2 | PAL; intro. AUS, NEA, OCE |
| 4. Symmixus Bernhauer, 1915 [63] | Tachyporini | 2 | ORI, PAL |
| 5. Tachyporus Gravenhorst, 1802 [41] sensu nov. [= †Mesotachyporus Gusarov, 2000 [56] syn. nov.] | Tachyporini | 136 (†4) | NEA, NEO, ORI, PAL |
| -Subtribe Euconosomatina Cameron, 1918 [44] stat. rev., sensu nov. | 366 (†2) | AUS, ETH, MAD, NEA, NEO, OCE, ORI, PAL | |
| 6. Euconosoma Cameron, 1918 [44] | Tachyporini | 4 | ORI, PAL |
| 7. Sepedophilus Gistel, 1856 [84] [= Urolitus Silvestri, 1947 [54] syn. nov.] | Tachyporini | 361 (†1) | AUS, ETH, MAD, NEA, NEO, OCE, ORI, PAL |
| 8. †Palaeosepedophilus Paśnik & Kubisz, 2002 [82] | Tachyporini | †1 | Europe (Baltic Sea Coast) [mid-Eocene Baltic amber] |
| Vatesini Seevers, 1958 [13] sensu nov. | 290 (†2) | AUS, ETH, MAD, NEA, NEO, OCE, ORI, PAL | |
| 1. Cilea Jacquelin du Val, 1856 [93] | Tachyporini | 19 | ETH, MAD, NEA, NEO, ORI, PAL |
| 2. Cileoporus Campbell, 1994 [91] | Tachyporini | 2 | NEO |
| 3. Coproporus Kraatz, 1857 [96] | Tachyporini | 229 (†1) | AUS, ETH, MAD, NEA, NEO, OCE, ORI, PAL |
| 4. Coprotachinus Cameron, 1933 [98] | Tachyporini | 7 | ETH |
| 5. Mimocyptus Cameron, 1919 [101] | Tachyporini | 2 | AUS, ORI |
| 6. Tachinoporus Cameron, 1928 [11] | Tachyporini | 1 | ORI |
| 7. Tachinoproporus Cameron, 1928 [11] | Tachyporini | 1 | ORI |
| 8. Termitoplus Silvestri, 1946 [12] | Tachyporini | 1 | NEO |
| 9. Vatesus Sharp, 1876 [92] | Vatesini | 27 | NEO |
| 10. †Procileoporus Yamamoto, 2016 [46] | Tachyporini | †1 | Myanmar (mid-Cretaceous Kachin amber) |
| Deropini Smetana, 1983 [23] | 20 | NEA, ORI, PAL | |
| 1. Derops Sharp, 1889 [105] | Deropini | 20 | NEA, ORI, PAL |
| Tachinusini Fleming, 1821 [45] stat. rev., sensu nov. | 371 (†7) | AUS, ETH, MAD, NEA, NEO, ORI, PAL | |
| 1. Austrotachinus Steel, 1956 [110] | Tachyporini | 1 | AUS |
| 2. Lacvietina Herman, 2004 [25] | Megarthropsini | 9 | ORI, PAL |
| 3. Leucotachinus Coiffait & Sáiz, 1968 [111] | Tachyporini | 3 | AUS, NEO |
| 4. Megarthropsis Cameron, 1919 [101] | Megarthropsini | 8 | ORI |
| 5. Nepaliodes Coiffait, 1977 [113] | Megarthropsini | 2 | ORI, PAL |
| 6. Nitidotachinus Campbell, 1993 [109] | Tachyporini | 16 | NEA, ORI, PAL |
| 7. Olophrinus Fauvel, 1895 [115] | Tachyporini | 15 | ORI, PAL |
| 8. Peitawopsis Smetana, 1992 [116] | Megarthropsini | 3 | PAL |
| 9. Pseudotachinus Cameron, 1932 [117] | Tachyporini | 5 | ORI, PAL |
| 10. Tachinomorphus Kraatz, 1859 [118] | Tachyporini | 21 | AUS, ETH, MAD, NEA, ORI, PAL |
| 11. Tachinoplesius Bernhauer, 1936 [120] | Tachyporini | 9 | ETH |
| 12. Tachinus Gravenhorst, 1802 [41] | Tachyporini | 273 (†1) | NEA, NEO, ORI, PAL |
| - Subgenus Latotachinus Ullrich, 1975 [121] | Tachyporini | 7 | PAL |
| - Subgenus Tachinoderus Motschulsky, 1858 [123] | Tachyporini | 60 | NEA, ORI, PAL |
| - Subgenus Tachinus Gravenhorst, 1802 [41] | Tachyporini | 204 (†1) | NEA, NEO, ORI, PAL |
| 13. †Hesterniasca Zhang, Wang & Xu, 1992 [124] | N/A | †2 | China (Lower Cretaceous) * |
| 14. †Mesotachinus Tikhomirova, 1968 [50] | N/A | †3 | Kazakhstan (Middle–Upper Jurassic) * |
| 15. †Protachinus Cai, Yan, Beattie, Wang & Huang, 2013 [52] | Tachyporini | †1 | Australia (Upper Jurassic) * |
| Tachyporinae, incertae sedis | †3 | ||
| 1. †Abscondus Tikhomirova, 1968 [50] | N/A | †2 | Kazakhstan (Middle–Upper Jurassic) * |
| 2. †Tachyporoides Tikhomirova, 1968 [50] | N/A | †1 | Kazakhstan (Middle–Upper Jurassic) * |
| TOTAL: 36 (†7) genera | 1194 (†18) | ||
| New Classification | Former Tribal Assignment | Described Species | Distribution |
|---|---|---|---|
| 1. Bobitobus Tottenham, 1939 [140] stat. rev. | Mycetoporini | 34 | NEA, ORI, PAL |
| 2. Bolitobius Leach, 1819 [141] | Mycetoporini | 21 | PAL; intro.? NEA |
| 3. Bolitopunctus Campbell, 1993 [33] | Mycetoporini | 2 | NEA |
| 4. Bryophacis Reitter, 1909 [144] | Mycetoporini | 12 | NEA, PAL |
| 5. Bryoporus Kraatz, 1857 [96] | Mycetoporini | 37 | ETH, NEA, NEO, ORI, PAL |
| 6. Canariobolitobius Schülke, 2004 [146] | Mycetoporini | 1 | PAL |
| 7. Carphacis Gozis, 1886 [148] | Mycetoporini | 16 | NEA, PAL |
| 8. Ischnosoma Stephens, 1829 [150] | Mycetoporini | 118 | AUS, ETH, NEA, NEO, ORI, PAL |
| 9. Lordithon Thomson, 1859 [136] sensu nov. | Mycetoporini | 97 (†4) | AUS, NEA, NEO, ORI, PAL |
| 10. Mycetoporus Mannerheim, 1831 [137] | Mycetoporini | 88 (†1) | NEA, PAL |
| 11. Neobolitobius Campbell, 1993 [33] | Mycetoporini | 1 | NEA |
| 12. Parabolitobius Li, Zhao & Sakai, 2000 [152] | Mycetoporini | 13 (†1) | PAL |
| 13. †Cuneocharis Ryvkin, 1990 [153] | N/A | †1 | Russia (Upper Jurassic) * |
| 14. †Glabrimycetoporus Yue, Zhao & Ren, 2009 [154] | Mycetoporini | †1 | China (Lower Cretaceous) * |
| 15. †Ryvkinius Herman, 2001 [67] | N/A | †1 | Russia (Upper Jurassic) * |
| 16. †Undiatina Ryvkin, 1990 [153] | N/A | †1 | Russia (Upper Jurassic) * |
| TOTAL: 16 (†4) genera | 444 (†10) | ||
References
- Tihelka, E.; Thayer, M.K.; Newton, A.F.; Cai, C. New data, old story: Molecular data illuminate the tribal relationships among rove beetles of the subfamily Staphylininae (Coleoptera: Staphylinidae). Insects 2020, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Grebennikov, V.V.; Newton, A.F. Detecting the basal dichotomies in the monophylum of carrion and rove beetles (Insecta: Coleoptera: Silphidae and Staphylinidae) with emphasis on the Oxyteline Group of subfamilies. Arthropod Syst. Phylogeny 2012, 70, 133–165. [Google Scholar]
- Thayer, M.K. Staphylinidae Latreille, 1802. In Handbook of Zoology; Arthropoda: Insecta, Coleoptera, Beetles. Morphology and Systematics (Archostemata, Adephaga, Myxophaga, Polyphaga Partim); Beutel, R.G., Leschen, R.A.B., Eds.; De Gruyter: Berlin/Heidelberg, Germany, 2016; Volume 1, pp. 394–442. [Google Scholar]
- Lawrence, J.F.; Newton, A.F., Jr. Evolution and classification of beetles. Annu. Rev. Ecol. Syst. 1982, 13, 261–290. [Google Scholar] [CrossRef]
- Lawrence, J.F.; Newton, A.F., Jr. Families and subfamilies of Coleoptera (with selected genera, notes, references and data on family-group names). In Biology, Phylogeny, and Classification of Coleoptera: Papers Celebrating the 80th Birthday of Roy A. Crowson; Pakaluk, J., Ślipiński, A., Eds.; Muzeum i Instytut Zoologii PAN: Warszawa, Poland, 1995; pp. 779–1006. [Google Scholar]
- McKenna, D.D.; Farrell, B.D.; Caterino, M.S.; Farnum, C.W.; Hawks, D.C.; Maddison, D.R.; Seago, A.E.; Short, A.E.Z.; Newton, A.F.; Thayer, M.K. Phylogeny and evolution of Staphyliniformia and Scarabaeiformia: Forest litter as a stepping stone for diversification of nonphytophagous beetles. Syst. Entomol. 2015, 40, 35–60. [Google Scholar] [CrossRef]
- Lü, L.; Cai, C.-Y.; Zhang, X.; Newton, A.F.; Thayer, M.K.; Zhou, H.-Z. Linking evolutionary mode to palaeoclimate change reveals rapid radiations of staphylinoid beetles in low-energy conditions. Curr. Zool. 2020, 66, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Thayer, M.K. Proglypholoma aenigma gen. et. sp. nov., Glypholoma spp. nov. and new records, and a phylogenetic analysis of Glypholomatinae (Coleoptera: Staphylinidae). Ann. Zool. 1997, 47, 157–174. [Google Scholar]
- Kim, S.I.; Farrell, B.D. Phylogeny of world stag beetles (Coleoptera: Lucanidae) reveals a Gondwanan origin of Darwin’s stag beetle. Mol. Phylogenet. Evol. 2015, 86, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Damgaard, J.; Burckhardt, D.; Gibbs, G.; Yuan, J.; Yang, H.; Bu, W. Phylogeny and historical biogeography of Gondwanan moss-bugs (Insecta: Hemiptera: Coleorrhyncha: Peloridiidae). Cladistics 2019, 35, 135–149. [Google Scholar] [CrossRef]
- Cameron, M. New species of Staphylinidae from Borneo. Sarawak Mus. J. 1928, 3, 423–451. [Google Scholar]
- Silvestri, F. Primo contributo alla conoscenza dei termitofili viventi con specie di Syntermes. Comment. Della Pontif. Acad. Sci. 1946, 9, 515–559. [Google Scholar]
- Seevers, C.H. A revision of the Vatesini, a tribe of Neotropical myrmecophiles (Coleoptera, Staphylinidae). Rev. Bras. Entomol. 1958, 8, 181–202. [Google Scholar]
- von Beeren, C.; Maruyama, M.; Kronauer, D.J. Cryptic diversity, high host specificity and reproductive synchronization in army ant-associated Vatesus beetles. Mol. Ecol. 2016, 25, 990–1005. [Google Scholar] [CrossRef] [PubMed]
- Parker, J. Myrmecophily in beetles (Coleoptera): Evolutionary patterns and biological mechanisms. Myrmecol. News 2016, 22, 65–108. [Google Scholar]
- Newton, A.F.; Thayer, M.K.; Ashe, J.S.; Chandler, D.S. Staphylinidae Latreille, 1802. In American Beetles (Archostemata, Myxophaga, Adephaga, Polyphaga: Staphyliniformia); Arnett, R.H., Jr., Thomas, M.C., Eds.; CRC Press: Boca Raton, FL, USA, 2000; Volume 1, pp. 272–418. [Google Scholar]
- Newton, A.F., Jr. Mycophagy in Staphylinoidea (Coleoptera). In Fungus-Insect Relationships: Perspectives in Ecology and Evolution; Wheeler, Q., Blackwell, M., Eds.; Columbia University Press: New York, NY, USA, 1984; pp. 302–353. [Google Scholar]
- Leschen, R.A.B.; Beutel, R.G. Pseudotracheal tubes, larval head, and mycophagy in Sepedophilus (Coleoptera: Staphylinidae: Tachyporinae). J. Zool. Syst. Evol. Res. 2001, 39, 25–35. [Google Scholar] [CrossRef]
- Matsumoto, T.K.; Miyazaki, Y.; Sueyoshi, M.; Senda, Y.; Yamada, K.; Hirobe, M. Pre-pollination barriers between two sympatric Arisaema species in northern Shikoku Island, Japan. Am. J. Bot. 2019, 106, 1612–1621. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, P.; Bousquet, Y.; Davies, A.E.; Alonso-Zarazaga, M.A.; Lawrence, J.F.; Lyal, C.H.C.; Newton, A.F.; Reid, C.A.M.; Schmitt, M.; Ślipiński, A.; et al. Family-group names in Coleoptera (Insecta). ZooKeys 2011, 88, 1–972. [Google Scholar] [CrossRef] [PubMed]
- Herman, L.H. Catalog of the Staphylinidae (Insecta: Coleoptera). 1758 to the end of the second millennium. Parts I–VII. Bull. Am. Mus. Nat. Hist. 2001, 265, 1–4218. [Google Scholar] [CrossRef]
- Schülke, M. Eine neue art der Gattung Symmixus Bernhauer, mit Bemerkungen zur Tribus Symmixini Bernhauer (Coleoptera, Staphylinidae, Tachyporinae). Linzer Biol. Beitr. 2003, 35, 443–452. [Google Scholar]
- Smetana, A. The status of the staphylinid genera Derops Sharp and Rimulincola Sanderson (Coleoptera). Entomol. Scand. 1983, 14, 269–279. [Google Scholar] [CrossRef]
- Smetana, A. The tribe Megarthropsini Cameron (Insecta: Coleoptera: Staphylinidae). Senckenberg. Biol. 1983, 64, 141–155. [Google Scholar]
- Herman, L.H. Revision of the Asian tribe Megarthropsini (Coleoptera: Staphylinidae: Tachyporinae). Am. Mus. Novit. 2004, 3430, 1–72. [Google Scholar] [CrossRef]
- Hammond, P.M. The phylogeny of a remarkable new genus and species of gymnusine staphylinid (Coleoptera) from the Auckland Islands. J. Entomol. B 1975, 44, 153–173. [Google Scholar] [CrossRef]
- Ashe, J.S.; Newton, A.F., Jr. Larvae of Trichophya and phylogeny of the Tachyporine Group of subfamilies (Coleoptera: Staphylinidae) with a review, new species and characterization of the Trichophyinae. Syst. Entomol. 1993, 18, 267–286. [Google Scholar] [CrossRef]
- Ashe, J.S. Phylogeny of the Tachyporine Group subfamilies and ‘basal’ lineages of the Aleocharinae (Coleoptera: Staphylinidae) based on larval and adult characteristics. Syst. Entomol. 2005, 30, 3–37. [Google Scholar] [CrossRef]
- Grebennikov, V.V.; Newton, A.F. Good-bye Scydmaenidae, or why the ant-like stone beetles should become megadiverse Staphylinidae sensu latissimo (Coleoptera). Eur. J. Entomol. 2009, 106, 275–301. [Google Scholar] [CrossRef]
- Blackwelder, R.E. Morphology of the coleopterous family Staphylinidae. Smithson. Misc. Collect. 1936, 94, 1–102. [Google Scholar]
- Wappler, T. The age of Baltic amber: Could eckfeld resolve this problem? In Fossils X3, 3rd International Congress of Palaeoentomology with 2nd International Meeting on Palaeoarthropodology and 2nd World Congress on Amber and its Inclusions. Programme and Abstracts; Brothers, D., Mostovski, M., Eds.; 7th to 11th February 2005; University of Kwazulu-Natal, South African National Biodiversity Institute: Pretoria, South Africa, 2005; p. 53. [Google Scholar]
- Campbell, J.M. A revision of the genera Mycetoporus Mannerheim and Ischnosoma Stephens (Coleoptera: Staphylinidae: Tachyporinae) of North and Central America. Mem. Entomol. Soc. Can. 1991, 156, 1–169. [Google Scholar] [CrossRef]
- Campbell, J.M. A revision of the genera Bryoporus Kraatz and Bryophacis Reitter and two new related genera from America North of Mexico (Coleoptera: Staphylinidae: Tachyporinae). Mem. Entomol. Soc. Can. 1993, 166, 1–85. [Google Scholar] [CrossRef]
- Maddison, W.P.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis. Version 3.61. 2019. Available online: http://mesquiteproject.org (accessed on 9 March 2020).
- Fauvel, A. Études sur les Staphylinides de l’Amérique centrale, principalement du Mexique. Not. Entomol. 1864, 2, 3–62. [Google Scholar]
- Goloboff, P.A.; Catalano, S.A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 2016, 32, 221–238. [Google Scholar] [CrossRef]
- Nixon, K.C. WinClada Version 1.00.08 (Program and Documentation); Published by the Author; Cornell University: Ithaca, NY, USA, 2002. [Google Scholar]
- Linnaeus, C. Systema Naturae per Regna Tria Naturae: Secundum Classes, Ordines, Genera, Species, Cum Characteribus, Differentiis, Synonymis, Locis. Editio Decima, Reformata; Laurentius Salvius: Stockholm, Sweden, 1758; Volume I, pp. 1–823. [Google Scholar]
- Latreille, P.A. Histoire Naturelle, Générale et Particulière Des Crustacés et Des Insectes. Tome troisième. Familles Naturelles et Genres; F. Dufart: Paris, France, 1802; pp. xii + 13–468. [Google Scholar]
- MacLeay, W.S. Annulosa Javanica, an Attempt to Illustrate the Natural Affinities and Analogies of the Insects Collected in Java by Thomas Horsfield, M. D. F. L. & G. S. and Deposited by Him in the Museum of the Honourable East-India Company. Number 1; Kingsbury, Parbury, and Allen: London, UK, 1825; pp. xii + 1–50. [Google Scholar]
- Gravenhorst, J.L.C. Coleoptera Microptera Brunsvicensia Nec Non Exoticorum Quotquot Exstant in Collectionibus Entomologorum Brunsvicensium in Genera Familias et Species Distribuit; Carolus Reichard: Brunsuigae, Germany, 1802; pp. lxvi + 1–206. [Google Scholar]
- Campbell, J.M. A revision of the genus Tachinus (Coleoptera: Staphylinidae) of North and Central America. Mem. Entomol. Soc. Can. 1973, 90, 1–137. [Google Scholar] [CrossRef]
- Naomi, S.-I. On the genus Derops Sharp (Staphylinidae) and its systematic position. Nat. Insects Tokyo 1986, 21, 17–22, (In Japanese, with English Title). [Google Scholar]
- Cameron, M. New Oriental Staphylinidae (3). Entomol. Mon. Mag. 1918, 54, 214–219. [Google Scholar]
- Fleming, J. Insecta. In Supplement to the Fourth, Fifth, and Sixth Editions of the Encyclopedia Britannica, with Preliminary Dissertations on the History of the Sciences; Archibald Constable and Company: Edinburgh, UK, 1821; Volume 5, pp. 41–56. [Google Scholar]
- Yamamoto, S. The oldest tachyporine rove beetle in amber (Coleoptera, Staphylinidae): A new genus and species from Upper Cretaceous Burmese amber. Cretaceous Res. 2016, 65, 163–171. [Google Scholar] [CrossRef]
- Chatzimanolis, S. A review of the fossil history of Staphylinoidea. In Biology of Rove Beetles (Staphylinidae); Betz, O., Irmler, U., Klimaszewski, J., Eds.; Springer Nature: Cham, Switzerland, 2018; pp. 27–45. [Google Scholar] [CrossRef]
- Chatzimanolis, S.; Grimaldi, D.A.; Engel, M.S.; Fraser, N.C. Leehermania prorova, the earliest staphyliniform beetle, from the Late Triassic of Virginia (Coleoptera: Staphylinidae). Am. Mus. Novit. 2012, 3761, 1–28. [Google Scholar] [CrossRef]
- Fikáček, M.; Beutel, R.G.; Cai, C.; Lawrence, J.F.; Newton, A.F.; Solodovnikov, A.; Ślipiński, A.; Thayer, M.K.; Yamamoto, S. Reliable placement of beetle fossils via phylogenetic analyses—Triassic Leehermania as a case study (Staphylinidae or Myxophaga?). Syst. Entomol. 2020, 45, 175–187. [Google Scholar] [CrossRef]
- Tikhomirova, A.L. Staphylinid beetles from Jurassic of Karatau (Coleoptera: Staphylinidae). In Jurassic Insects of Karatau; Rohdendorf, B.B., Ed.; Akademiya Nauk SSSR Otdelenie Obshchej Biologii: Moscow, Russia, 1968; pp. 139–154. (In Russian) [Google Scholar]
- Cai, C.; Huang, D. Current knowledge on Jurassic staphylinids of China (Insecta, Coleoptera). Earth Sci. Front. 2010, 17, 151–153. [Google Scholar]
- Cai, C.-Y.; Yan, E.V.; Beattie, R.; Wang, B.; Huang, D.-Y. First rove beetles from the Jurassic Talbragar Fish Bed of Australia (Coleoptera, Staphylinidae). J. Paleontol. 2013, 87, 650–656. [Google Scholar] [CrossRef]
- ICZN (International Commission on Zoological Nomenclature). Opinion 1743. Tachinidae Fleming, 1821 (Insecta, Coleoptera): Spelling emended to Tachinusidae to remove homonymy with Tachinidae Robineau-Desvoidy, 1830 (Insecta, Diptera), and Tachyporidae MacLeay, 1825 (Insecta, Coleoptera): Given precedence. Bull. Zool. Nomencl. 1993, 50, 248–250. [Google Scholar]
- Silvestri, F. Contributo alla conoscenza dei Termitodiscinae e Cephaloplectinae (Staphylinidae, Coleoptera) termitofili. Arch. Zool. Ital. 1947, 31, 123–149. [Google Scholar]
- Campbell, J.M. A review of the Tachyporinae genus Euconosoma Cameron (Coleoptera: Staphylinidae) with a description of a new species from Nepal. Coleopt. Bull. 1976, 30, 139–145. [Google Scholar]
- Gusarov, V.I. Mesotachyporus puer, a new genus and species of Cretaceous Tachyporinae (Coleoptera, Staphylinidae) from New Jersey amber. In Studies on Fossils in Amber, with Particular Reference to the Cretaceous of New Jersey; Grimaldi, D., Ed.; Backhuys Publishers: Leiden, The Netherlands, 2000; pp. 255–258. [Google Scholar]
- Luze, G. Revision der europäischen und sibirischen Arten der Staphyliniden-Gattung Tachyporus Grav. und Lamprinus Heer. Verh. K.K. Zool.-Bot. Ges. Wien 1901, 51, 146–185. [Google Scholar]
- Gravenhorst, J.L.C. Monographia Coleopterorum Micropterorum; Henricus Dieterich: Gottingae, Germany, 1806; pp. 1–248. [Google Scholar]
- Heer, O. Fascicule II. In Fauna Coleopterorum Helvetica. Pars I; Orelii, Fuesslini et Sociorum: Turici, Switzerland, 1839; pp. 145–366. [Google Scholar]
- Panzer, G.W.F. Faunae Insectorum Germanicae Initia Oder Deutschlands Insecten. Dritter Iahrgang; Felssecker: Nürnberg, Germany, 1796; Volume 27, pp. 1–20. [Google Scholar]
- Campbell, J.M. A revision of the genus Tachyporus Gravenhorst (Coleoptera: Staphylinidae) of North and Central America. Mem. Entomol. Soc. Can. 1979, 109, 1–95. [Google Scholar] [CrossRef]
- Fabricius, J.C. Species Insectorvm Exhibentes Eorvm Differentias Specificas, Synonyma Avctorvm, Loca Natalia, Metamorphosin Adiectis Observationibvs, Descriptionibvs; C.E. Bohn: Hamburgi & Kilonii, Germany, 1781; Volume 1, pp. viii + 1–552. [Google Scholar]
- Bernhauer, M. Zur Staphylinidenfauna des indo-malayischen Gebietes, insbesonders der Himalaya. X. Coleopterol. Rundsch. 1915, 4, 49–60. [Google Scholar]
- Klebs, R. Über Bernsteineinschlüsse in allgemeinen und die Coleopteren meiner Bernsteinsammlung. Schr. Phys. Okon. Ges. Königsberg 1910, 51, 217–242. [Google Scholar]
- Larsson, S.G. Baltic Amber—A Palaeobiological Study. Entomonograph; Scandinavian Science Press: Klampenborg, Denmark, 1978; Volume 1, pp. 1–192. [Google Scholar]
- Paśnik, J. Fossils of Staphylinidae from Baltic amber: A new genus and three new species (Insecta, Coleoptera, Staphylinidae). Senckenberg. Biol. 2005, 85, 97–100. [Google Scholar]
- Herman, L.H. Nomenclatural changes in the Staphylinidae (Insecta: Coleoptera). Bull. Am. Mus. Nat. Hist. 2001, 264, 1–83. [Google Scholar] [CrossRef]
- Scudder, S.H. Adephagous and clavicorn Coleoptera from the Tertiary deposits at Florissant, Colorado with descriptions of a few other forms and a systematic list of the non-rhynchophorous Tertiary Coleoptera of North America. Monogr. U. S. Geol. Surv. 1900, 40, 1–148. [Google Scholar]
- Poinar, G., Jr.; Poinar, R. The Amber Forest: A Reconstruction of a Vanished World; Princeton University Press: Princeton, NJ, USA, 1999; pp. xiii + 1–239. [Google Scholar]
- Schülke, M.; Smetana, A. Staphylinidae. In Catalogue of Palaearctic Coleoptera. Hydrophiloidea—Staphylinoidea; Revised and Updated Edition; Löbl, I., Löbl, D., Eds.; Brill: Leiden, The Netherlands, 2015; Volume 2, pp. 304–1134. [Google Scholar]
- Grimaldi, D.A.; Nascimbene, P.C.; Penney, D. Chapter 10 Raritan (New Jersey) amber. In Biodiversity of Fossils in Amber from the Major World Deposits; Siri Scientific Press: Manchester, UK, 2010; pp. 167–191. [Google Scholar]
- Yamamoto, S.; Solodovnikov, A. The first fossil Megalopsidiinae (Coleoptera: Staphylinidae) from Upper Cretaceous Burmese amber and its potential for understanding basal relationships of rove beetles. Cretaceous Res. 2016, 59, 140–146. [Google Scholar] [CrossRef]
- Simpson, G. Tempo and Mode in Evolution; Columbia University Press: New York, NY, USA, 1944; pp. 1–237. [Google Scholar]
- Clarke, D.J.; Chatzimanolis, S. Antiquity and long-term morphological stasis in a group of rove beetles (Coleoptera: Staphylinidae): Description of the oldest Octavius species from Cretaceous Burmese amber and a review of the “Euaesthetine subgroup” fossil record. Cretaceous Res. 2009, 30, 1426–1434. [Google Scholar] [CrossRef]
- Yamamoto, S.; Takahashi, Y.; Parker, J. Evolutionary stasis in enigmatic jacobsoniid beetles. Gondwana Res. 2017, 45, 275–281. [Google Scholar] [CrossRef]
- Yamamoto, S.; Takahashi, Y. First and oldest Leptochirini rove beetles illuminate diverse cephalic structures in the Cretaceous (Coleoptera: Staphylinidae: Osoriinae). Syst. Entomol. 2019, 44, 588–611. [Google Scholar] [CrossRef]
- Cai, C.; Lawrence, J.F.; Yamamoto, S.; Leschen, R.A.B.; Newton, A.F.; Ślipiński, A.; Yin, Z.; Huang, D.; Engel, M.S. Basal polyphagan beetles in mid-Cretaceous amber from Myanmar: Biogeographic implications and long-term morphological stasis. Proc. R. Soc. B 2019, 286, 20182175. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Maruyama, M.; Parker, J. Evidence for social parasitism of early insect societies by Cretaceous rove beetles. Nat. Commun. 2016, 7, 13658. [Google Scholar] [CrossRef] [PubMed]
- Makranczy, G.; Yamamoto, S.; Engel, M.S. Description of a Cretaceous amber fossil putatively of the tribe Coprophilini (Coleoptera, Staphylinidae, Oxytelinae). ZooKeys 2018, 782, 81–94. [Google Scholar] [CrossRef]
- Yin, Z.-W.; Lü, L.; Yamamoto, S.; Thayer, M.K.; Newton, A.F.; Cai, C.-Y. Dasycerine rove beetles: Cretaceous diversification, phylogeny and historical biogeography (Coleoptera: Staphylinidae: Dasycerinae). Cladistics 2021, 37, 185–210. [Google Scholar] [CrossRef]
- Yin, Z.; Cai, C.; Newton, A.F. Beetle with long ‘nose’—A bizarre stem scydmaenine in amber from Myanmar (Coleoptera: Staphylinidae: Scydmaeninae). Cretaceous Res. 2018, 89, 98–106. [Google Scholar] [CrossRef]
- Paśnik, J.; Kubisz, D. A new genus and new species of Staphylinidae (Coleoptera) from Baltic amber. Eur. J. Entomol. 2002, 99, 353–362. [Google Scholar] [CrossRef]
- Bernhauer, M. Beitrag Zur Staphylinidenfauna von Ostindien und den Sundainseln. Stettin. Entomol. Ztg. 1903, 64, 21–36. [Google Scholar]
- Gistel, J.N.F.X. Die Mysterien Der Europäischen Insectenwelt. Ein Geheimer Schlüssel Für Sammler Aller Insecten-Ordnungen Und Stände, Behufs Des Fangs, Des Aufenhalts-Orts, Der Wohnung, Tag- Und Jahreszeit u.s.w., Oder Autoptische Darstellung Des Insectenstaats in Seinem Zusammenhange Zum Bestehen Des Naturhaushaltes Überhaupt Und Insbesondere in Seinem Einflusse Auf Die Phanerogamische Und Cryptogamische Pflanzenberöltzerrung Europa’s. Zum Ersten Male Nach 25 Jährigen Eigenen Erfahrungen Zusammengestellt Und Herausgegeben; T. Dannheimer: Kempten, Germany, 1856; pp. xii+ 1–530 + [2]. [Google Scholar]
- de Paykull, G. Monographia Caraborum Sueciae; J. Edman: Upsaliae, Sweden, 1790; pp. 1–138. [Google Scholar]
- Ádám, L. Systematic content. In Adatok a Zempléni-Hegység, a Hernád-Völgy, a Bodrogköz, a Rédköz és a Taktaköz Holyfaunájához (Coleoptera); Ádám, L., Hegyessy, G., Eds.; A Sátoraljaújhelyi Kazinczy Ferenc Múzeum Füzetei V: Sátoraljaújhely, Hungary, 2001; pp. 1–249. [Google Scholar]
- Żyła, D.; Solodovnikov, A. Multilocus phylogeny defines a new classification of Staphylininae (Coleoptera, Staphylinidae), a rove beetle group with high lineage diversity. Syst. Entomol. 2020, 45, 114–127. [Google Scholar] [CrossRef]
- Campbell, J.M. A revision of the genus Sepedophilus Gistel (Coleoptera: Staphylinidae) of America North of Mexico. Mem. Entomol. Soc. Can. 1976, 99, 1–89. [Google Scholar] [CrossRef]
- Naomi, S.-I.; Maruyama, M. Revision of the genus Sepedophilus Gistel (Coleoptera: Staphylinidae: Tachyporinae) from Japan: Species groups of S. glabratus and S. exiguous. Entomol. Rev. Jpn. 1997, 52, 61–71. [Google Scholar]
- Naomi, S.-I.; Maruyama, M. A revision of the genus Sepedophilus Gistel (Coleoptera, Staphylinidae, Tachyporinae) from Japan: Species group of S. pedicularius. Jpn. J. Syst. Entomol. 1998, 4, 51–75. [Google Scholar]
- Campbell, J.M. Cileoporus, a new genus of rove beetles from Central and South America (Coleoptera, Staphylinidae, Tachyporinae). Stud. Neotrop. Fauna Environ. 1994, 29, 125–144. [Google Scholar] [CrossRef]
- Sharp, D. On a new genus and species of the family Staphylinidae. Entomol. Mon. Mag. 1876, 12, 199–202. [Google Scholar]
- Jacquelin du Val, P.N.C. Famille Des Staphylinides. In Manuel Entomologique. Genera Des Coléoptères d’ Europe Comprenant Leur Classification En Familles Naturelles, La Description de Tous Les Genres, Des Tableaux Synoptiques Destinés à Faciliter l’ Étude, Le Catalogue de Toutes Les Espèces de Nombreux Au Trait de Caractères Tome Deuxième; A. Deyrolle: Paris, France, 1856; pp. 1–40. [Google Scholar]
- Linnaeus, C. Systema Naturae, per Regna Tria Naturae, Secundum Classes, Ordines, Genera, Species, Cum Characteribus, Differentiis, Synonymis, Locis. Editio Duodecima Reformata; Laurentii Salvii: Holmiae, Sweden, 1767; Volume I, Pars II, pp. 533–1327 + [37]. [Google Scholar]
- Bernhauer, M. Neue Staphyliniden aus Südamerika. Wien. Entomol. Ztg. 1923, 40, 49–60. [Google Scholar] [CrossRef]
- Kraatz, G. Naturgeschichte der Insecten Deutschlands. Erste Abtheilung Coleoptera; Nicolai: Berlin, Germany, 1857; Volume Zweiter, Lieferung 3–6, pp. 377–1080. [Google Scholar]
- Kraatz, G. Description de quelques nouvelles espèces de Coléoptères. Bull. Société Entomol. Fr. 1858, 6, clxxxviii–cxcii. [Google Scholar]
- Cameron, M. New species of Staphylinidae (Col.) from the Belgian Congo. Bull. Ann. Soc. Entomol. Belg. 1933, 73, 35–53. [Google Scholar]
- Cameron, M. New species of Staphylinidae from the Belgian Congo. Bull. Ann. Soc. Entomol. Belg. 1926, 66, 281–284. [Google Scholar]
- Fauvel, A. Staphylinides nouveaux d’ Afrique tropicale. Rev. D’Entomol. 1905, 24, 194–198. [Google Scholar]
- Cameron, M. New species of Staphylinidae from Singapore. Trans. Entomol. Soc. Lond. 1919, 1919, 231–246. [Google Scholar]
- Yamamoto, S.; Takahashi, Y. Coproporus electron sp. nov., the first tachyporine rove beetle in Dominican amber (Coleoptera, Staphylinidae). PalZ 2016, 90, 629–635. [Google Scholar] [CrossRef]
- Campbell, J.M. A revision of the genera Coproporus and Cilea (Coleoptera: Staphylinidae) of America North of Mexico. Can. Entomol. 1975, 107, 175–216. [Google Scholar] [CrossRef]
- Blackwelder, R.E. The generic names of the beetle family Staphylinidae, with an essay on genotypy. U. S. Natl. Mus. Bull. 1952, 200, 1–483. [Google Scholar]
- Sharp, D. The Staphylinidae of Japan. Ann. Mag. Nat. Hist. 1889, 3, 28–44, 108–121, 249–267, 319–334, 406–419, 463–476. [Google Scholar] [CrossRef]
- Zhao, J.-Q.; Li, L.-Z. New data on the genus Derops Sharp (Coleoptera, Staphylinidae, Tachyporinae) from China with description of two new species. ZooKeys 2013, 317, 53–67. [Google Scholar] [CrossRef]
- Sanderson, M.W. A new genus of Nearctic Staphylinidae (Coleoptera). J. Kans. Entomol. Soc. 1947, 19, 130–133. [Google Scholar]
- Campbell, J.M. A review of the genus Tachinus Gravenhorst (Coleoptera: Staphylinidae: Tachyporinae) of Taiwan. Bull. Natl. Mus. Nat. Sci. 1993, 4, 33–46. [Google Scholar]
- Campbell, J.M. A review of the species of Nitidotachinus new genus (Coleoptera: Staphylinidae: Tachyporinae). Can. Entomol. 1993, 125, 521–548. [Google Scholar] [CrossRef]
- Steel, W.O. A new genus and species of Tachyporinae from Australia (Coleoptera: Staphylinidae). Proc. R. Entomol. Soc. Lond. B 1956, 25, 13–16. [Google Scholar] [CrossRef]
- Coiffait, H.; Saiz, F. Les Staphylinidae (sensu lato) du Chile. In Biologie de Amérique Australe. Études sur la Faune du sol. 4; Deboutteville, C., Rapoport, E., Eds.; Centre National de la Recherche Scientifique: Paris, France, 1968; pp. 339–468. [Google Scholar]
- Fairmaire, L.; Germain, P. Révision des Coléoptères du Chili. Ann. Soc. Entomol. Fr. 1862, 4, 405–456. [Google Scholar] [CrossRef]
- Coiffait, H. Staphylinides récoltés au Népal par le Professeur Franz (2e partie). Bull. Société D’Histoire Nat. Toulouse 1977, 112, 243–275. [Google Scholar]
- Horn, G. Synopsis of the genera and species of the staphylinid tribe Tachyporini of the United States. Trans. Am. Entomol. Soc. 1877, 6, 81–128. [Google Scholar]
- Fauvel, A. Staphylinides nouveaux de l’Inde et de La Malaisie. Rev. D’Entomol. 1895, 14, 180–286. [Google Scholar]
- Smetana, A. Peitawopsis monticola, a new genus and species of the tribe Megarthropsini from southern Taiwan (Coleoptera: Staphylinidae: Tachyporinae). Bull. Natl. Mus. Nat. Sci. 1992, 3, 199–208. [Google Scholar]
- Cameron, M. The Fauna of British India Including Ceylon and Burma. Coleoptera. Staphylinidae; Taylor & Francis: London, UK, 1932; Volume 3, pp. xiii + 1–443. [Google Scholar]
- Kraatz, G. Die Staphylinen-Fauna von Ostindien, insbesondere der Insel Ceylan. Arch. Naturgeschichte 1859, 25, 1–196. [Google Scholar]
- Erichson, W.F. Zweiter Band. In Genera et Species Staphylinorum Insectorum Coleopterorum Familiae; F.H. Morin: Berlin, Germany, 1840; pp. 401–954. [Google Scholar]
- Bernhauer, M. Neuheiten der ostafrikanischen Staphylinidenfauna (Coleoptera). Ann. Mag. Nat. Hist. 1936, 10, 321–336. [Google Scholar] [CrossRef]
- Ullrich, W.G. Monographie Der Gattung Tachinus Gravenhorst (Coleoptera: Staphylinidae), Mit Bemerkungen Zur Phylogenie Und Verbreitung Der Arten; Dissertation zur Erlangung des Doktorgrades der Mathematisch-Naturwissenschaftlichen Fakultät der Christian-Albrechts-Universität zu Kiel: Kiel, Germany, 1975; pp. 1–365. [Google Scholar]
- Sharp, D. The Staphylinidae of Japan. Ann. Mag. Nat. Hist. 1888, 2, 369–387, 451–464. [Google Scholar] [CrossRef]
- de Motschulsky, V. Énumeration des nouvelles espèces de coléopterès rapportés de ses voyages. Bull. Soc. Imp. Nat. Moscou 1858, 31, 204–264. [Google Scholar]
- Zhang, J.-F.; Wang, X.; Xu, G. A new genus and two new species of fossil Staphylinidae (Coleoptera) from Laiyang, Shandong Province, China. Entomotaxonomia 1992, 14, 277–281, (In Chinese, with English Abstract). [Google Scholar]
- Cai, C.; Huang, D.; Solodovnikov, A. A new species of Hesterniasca (Coleoptera, Staphylinidae, Tachyporinae) from Early Cretaceous of China with discussion of its systematic position. Insect Syst. Evol. 2011, 42, 213–220. [Google Scholar]
- Campbell, J.M. A review of the genus Olophrinus Fauvel (Coleoptera: Staphylinidae: Tachyporinae) with descriptions of three new species. Bull. Natl. Mus. Nat. Sci. 1993, 4, 47–70. [Google Scholar]
- Chang, Y.; Yin, Z.-W.; Li, L.-Z.; Schülke, M. A review of the genus Olophrinus from China (Coleoptera: Staphylinidae: Tachyporinae). Acta Entomol. Mus. Natl. Pragae 2019, 59, 307–324. [Google Scholar] [CrossRef]
- Lin, Q.-B. The Jurassic fossil insects from Western Liaoning. Acta Palaeontol. Sin. 1976, 15, 97–116, (In Chinese, with English Abstract). [Google Scholar]
- Hong, Y.-C. Middle Jurassic Fossil Insects in North China; Geological Publishing House: Beijing, China, 1983; pp. 1–223, (In Chinese, with English Abstract). [Google Scholar]
- Newton, A.F.; Thayer, M.K.; Sabrosky, C.W. Case 2786. Tachinidae Fleming, 1821 (Insecta, Coleoptera) and Tachinidae Robineau-Desvoidy, 1830 (Insecta, Diptera): Proposed removal of homonymy and Tachyporidae MacLeay, 1825 (Insecta, Coleoptera): Proposed precedence over Tachinusidae Fleming, 1821. Bull. Zool. Nomencl. 1992, 49, 122–126. [Google Scholar]
- Chang, Y.; Li, L.-Z.; Yin, Z.-W.; Schülke, M. A review of the Tachinus longicornis-group of the subgenus Tachinoderus Motschulsky (Coleoptera: Staphylinidae: Tachyporinae) from China. Zootaxa 2019, 4545, 478–494. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Li, L.-Z.; Yin, Z.-W.; Schülke, M. Eleven new species and new records of the Tachinus nepalensis Ullrich group of the subgenus Tachinoderus Motschulsky from China, Vietnam and Laos (Coleoptera: Staphylinidae: Tachyporinae). Zootaxa 2019, 4686, 1–52. [Google Scholar] [CrossRef]
- Chang, Y.; Li, L.-Z.; Yin, Z.-W. A contribution to the knowledge of the genus Lacvietina Herman (Coleoptera: Staphylinidae: Tachyporinae) from China. Zootaxa 2019, 4664, 574–580. [Google Scholar] [CrossRef]
- Hayashi, Y. Notes on Peitawopsis (Coleoptera: Staphylinidae: Tachyporinae), with a description of a new species from Japan. Entomol. Rev. Jpn. 2003, 58, 113–119. [Google Scholar]
- Li, L.-Z. Two new species of the genus Tachinus (Coleoptera, Staphylinidae) from the Ryukyu Islands, southwest Japan. Jpn. J. Entomol. 1994, 62, 661–666. [Google Scholar]
- Thomson, C.G. Skandinaviens Coleoptera, Synoptiskt Bearbetade; Berlingska Boktryckeriet: Lund, Sweden, 1859; Volume I, pp. [5] + 1–290. [Google Scholar]
- von Mannerheim, C.G. Précis d’un Nouvel Arrangement de La Famille Des Brachélytres de l’Ordre Des Insectes Coléoptères; St. Petersbourg, Russia, 1830 [1831]; pp. 1–87. [Google Scholar]
- Campbell, J.M. A revision of the genus Carphacis des Gozis (Coleoptera: Staphylinidae) of North America. Can. Entomol. 1980, 112, 935–953. [Google Scholar] [CrossRef]
- ICZN (International Commission on Zoological Nomenclature). Opinion 1726. Mycetoporus Mannerheim, 1831 (Insecta, Coleoptera): Tachinus punctus Gravenhorst, 1806 designated as the type species; Ischnosoma Stephens, 1829 conserved; and Mycetoporus given precedence over Ischnosoma. Bull. Zool. Nomencl. 1993, 50, 171–173. [Google Scholar]
- Tottenham, C.E. Some notes on the nomenclature of the Staphylinidae (Coleoptera). Proc. R. Entomol. Soc. Lond. B 1939, 8, 224–237. [Google Scholar] [CrossRef]
- Leach, W.E. New genera. In The Entomologist’s Useful Compendium; or an Introduction to the Knowledge of British Insects, Comprising the Best Means of Obtaining and Preserving Them, and a Description of the Apparatus Generally Used; Together with the Genera of Linne, and the Modern Method of Arranging the Classes Crustacea, Myriapoda, Spiders, Mites, and Insects from Their Affinities and Structure, According to the Views of Dr. Leach. Also, an Explanation of the Terms Used in Entomology; a Calendar of the Times of Appearance, and Usual Situations of Near 3000 Species of British Insects; with Instructions for Collecting and Fitting up Objects for the Microscope; Thomas Boys: London, UK, 1819; pp. 1–496. [Google Scholar]
- Stephens, J.F. Illustrations of British Entomology; or, a Synopsis of Indigenous Insects; Containing Their Generic and Specific Distinctions; with an Account of Their Metamorphoses, Times of Appearance, Localities, Food, and Economy, as Far as Practicable. Mandibulata. 5; Baldwin and Cradock: London, UK, 1832; pp. 1–448. [Google Scholar]
- Hatch, M.H. The Beetles of the Pacific Northwest. Staphyliniformia; University of Washington Press: Seattle, WA, USA, 1957; Part II, pp. ix + 1–384. [Google Scholar]
- Reitter, E. Fauna Germanica. Die Käfer Des Deutschen Reiches. Nach Der Analytischen Methode Bearbeitet. Schriften Des Deutschen Lehrervereins Für Naturkunde 24; K.G. Lutz: Stuttgart, Germany, 1909; Volume II, pp. 1–392. [Google Scholar]
- Erichson, W.F. Die Käfer Der Mark Brandenburg; Morin: Berlin, Germany, 1839; Volume Erster. Zweite Abtheilung, pp. 385–740. [Google Scholar]
- Schülke, M. Revision der paläarktischen Arten der Gattung Bryophacis Reitter, mit Bemerkungen Zu Bolitobius biseriatus Mannerheim und Bolitobius filicornis Wollaston (Coleoptera, Staphylinidae, Tachyporinae). Linzer Biol. Beitr. 2004, 36, 1001–1054. [Google Scholar]
- Wollaston, T.V. Catalogue of the Coleopterous Insects of the Canaries in the Collection of the British Museum; Taylor & Francis: London, UK, 1864; pp. xiii + 1–648. [Google Scholar]
- des Gozis, M. Recherche de l’ Espece Typique de Quelques Anciens Genres. Rectifications Synonymiques et Notes Diverses; Herbin: Montluçon, France, 1886; pp. 1–36. [Google Scholar]
- Olivier, A.G. Entomologie, Ou Histoire Naturelle Des Insectes, Avec Leur Caractères Génériques et Spécifiques, Leur Description, Leur Synonymie, et Leur Figure Illuminée. Coléoptères; Lanneau: Paris, France, 1795; Volume Troisième, pp. 1–557. [Google Scholar]
- Stephens, J.F. The Nomenclature of British Insects; Being a Compendious List of Such Species as Are Contained in the Systematic Catalogue of British Insects, and Forming a Guide to Their Classification, &c. &c; Baldwin and Cradock: London, UK, 1829; pp. 1–68. [Google Scholar]
- Fabricius, J.C. Genera Insectorum Eorumque Characteres Naturales Secundum Numerum, Figuram, Situm et Proportionem Omnium Partium Oris Adiecta Mantissa Specierum Nuper Detectarum; M.F. Bartsch: Chilonii, Germany, 1777; pp. 14 + 1–310. [Google Scholar]
- Li, L.-Z.; Zhao, M.-J.; Sakai, M. A new genus of the subfamily Tachyporinae (Coleoptera, Staphylinidae) with description of a new species from Japan. Jpn. J. Syst. Entomol. 2000, 6, 11–18. [Google Scholar]
- Ryvkin, A.B. Semeistvo Staphylinidae Latreille, 1802. Tr. Paleontol. Inst. Akad. Nauk SSSR 1990, 239, 52–66. (In Russian) [Google Scholar]
- Yue, Y.; Zhao, Y.; Ren, D. Glabrimycetoporus amoenus, a new Tachyporine genus and species of Mesozoic Staphylinidae (Coleoptera) from Liaoning, China. Zootaxa 2009, 2225, 63–68. [Google Scholar] [CrossRef]
- Cameron, M. New species of Staphylinidae (Col.) from Angola (IV). Publicações Cult. Co. Diam. Angola 1959, 48, 109–121. [Google Scholar]
- Schülke, M. Eine neue Art der Gattung Bolitobius Leach in Samouelle aus dem Beltischen Bernstein (Col., Staphylinidae, Tachyporinae). Entomol. Nachrichten Berichte 2000, 44, 101–103. [Google Scholar]
- Campbell, J.M. A revision of the genus Lordithon Thomson of North and Central America (Coleoptera: Staphylinidae). Mem. Entomol. Soc. Can. 1982, 119, 1–116. [Google Scholar] [CrossRef]
- Li, L.-Z.; Zhao, M.-J.; Ohbayashi, N. A revision of the genus Lordithon Thomson (Coleoptera, Staphylinidae) of Japan. Jpn. J. Syst. Entomol. 1999, 5, 217–254. [Google Scholar]
- Solsky, S.M. Coléoptères de la Sibérie orientale. Horae Soc. Entomol. Ross. 1871, 8, 232–272. [Google Scholar]
- Say, T. Descriptions of coleopterous insects collected in the late expedition to the Rocky Mountains, performed by order of Mr. Calhoun, Secretary of War, under the command of Major Long. J. Acad. Nat. Sci. Phila. 1823, 3, 139–216. [Google Scholar]
- LeConte, J.L. New Species of North American Coleoptera. Part I. Smithson. Misc. Collect. 1863, 6, 1–92. [Google Scholar]
- Luze, G. Eine neue Art der Staphyliniden-Gattung Bolitobius Mannh. aus Siberien. Verh. K.K. Zool.-Bot. Ges. Wien 1902, 52, 110–111. [Google Scholar]
- Bernhauer, M. Neue Kurzflügler des Paläarktischen Gebietes. Koleopterol. Rundsch. 1929, 14, 177–195. [Google Scholar]
- Li, L.-Z.; Sakai, M. Two new species of the genus Lordithon (Coleoptera, Staphylinidae) form [sic] Japan. Jpn. J. Syst. Entomol. 1996, 2, 251–256. [Google Scholar]
- Schubert, K. Einige neue paläalrktische Staphyliniden aus Kashmir. Dtsch. Entomol. Z. 1906, 1906, 378–383. [Google Scholar]
- Luze, G. Bolitobiini. Revision der paläarktischen Arten der Staphyliniden-Gattung Bryocharis Boisd. et Lac., Bolitobius Mannh., Bryoporus Kraatz und Mycetoporus Mannh. Verh. K.K. Zool. Bot. Ges. Wien 1901, 51, 662–746. [Google Scholar]
- Katayama, Y.; Ito, T. Two new species of the genus Lordithon (Coleoptera: Staphylinidae: Tachyporinae) from Japan. Entomol. Rev. Jpn. 2010, 65, 293–298. [Google Scholar]
- Weise, J. Japanische Staphilinidae [sic] und Pselaphidae. In: Beiträge zur Käferfauna von Japan, meist auf R. Hiller’s Sammulungen basirt. Dtsch. Entomol. Z. 1877, 21, 88–100. [Google Scholar]
- Bernhauer, M. Beiträge zur Kenntnis der paläarktischen Staphyliniden-Fauna. Münchener Koleopterol. Z. 1915, 4, 262–270. [Google Scholar]
- Malkin, B. New species of Boletobius [sic] from the Northwest with a distributional note on Coproporus (Coleoptera, Staphylinidae). Pan-Pac. Entomol. 1944, 20, 23–30. [Google Scholar]
- Sachse, C.T. Neue Käfer. Entomol. Ztg. Stettin 1852, 13, 115–127, 142–149, 454. [Google Scholar]
- Couper, W. Descriptions of new species of Canadian Coleoptera. Can. Nat. Geol. N. Ser. 1865, 2, 60–63. [Google Scholar]
- von Paula Schrank, F. Fauna Boica. Durchgedachte Geschichte der in Baiern Einheimischen und Zahmen Thiere; Stein’schen Buchhandlung: Nürnberg, Germany, 1798; Volume Erster, pp. xii + 1–720. [Google Scholar]
- Bernhauer, M. Zur Staphylinidenfauna von Nordamerika. V. Beitrag. Pomona Coll. J. Entomol. 1912, 4, 678–683. [Google Scholar]
- Fabricius, J.C. Systema Entomologiae, Sistens Insectorum Classes, Ordines, Genera, Species, Adiectis Synonymis, Locis, Descriptionibus, Observationibus; Libraria Kortii: Flensburgi et Lipsiae, Germany, 1775; pp. [32] + 1–832. [Google Scholar]
- von Paula Schrank, F. Enumeratio insectorum Austriae Indigenorum; Klett et Franck: Augustae Vindelicorum, Germany, 1781; pp. 22 + 1–548 + [2]. [Google Scholar]
- Zetterstedt, J.W. Fauna Insectorum Lapponica; Libraria Schulziana: Hammone, Germany, 1828; Pars I, pp. xx + 1–563. [Google Scholar]
- Sahlberg, J.R. Bidrag till Nordvestra Sibiriens Insektfauna. Coloeptera. Insamlade under Expeditionerna till obi och Jenessej 1876 och 1877. I. Cicindelidae, Carabidae, Dytiscidae, Hydrophilidae, Gyrinidae, Dryopidae, Georyssidae, Limnichidae, Heteroceridae, Staphylinidae och Micropeplidae. Kongliga Sven. Vetensk. Akad. Handl. 1880, 17, 1–115. [Google Scholar]
- Bernhauer, M. Neue Staphyliniden der Palaearktischen Fauna. Koleopterol. Rundsch. 1928, 14, 8–23. [Google Scholar]
- Cameron, M. New species of Staphylinidae (Col.) from Japan. Entomol. Mon. Mag. 1933, 69, 168–175, 208–219. [Google Scholar]
- Say, T. Untitled continuation of Say, 1830. In Descriptions of New Species of North American Insects, and Observations on Some Already Described; T. Say: New Harmony, IN, USA, 1832; pp. 50–57. [Google Scholar]
- Kraatz, G. Neue Käfer vom Amur. Dtsch. Entomol. Z. 1879, 23, 121–144. [Google Scholar] [CrossRef]
- de Motschulsky, V. Énumération des nouvelles especes de coléopteres rapportées de ses voyages. 3-ième article. Bull. Soc. Imp. Nat. Moscou 1860, 33, 539–588. [Google Scholar]
- Bernhauer, M. Neuheiten der Staphylinidenfauna der Mandschurei. Koleopterol. Rundsch. 1938, 24, 20–29. [Google Scholar]
- Dethlefsen, E.S. A new species of Bolitobius from the Pacific Coast. Pan-Pac. Entomol. 1946, 22, 71–72. [Google Scholar]
- Bernhauer, M. Elfte Folge neuer Staphyliniden des paläarktischen Fauna, nebst Bemerkungen. Verh. K.K. Zool.-Bot. Ges. Wien 1902, 52, 695–705. [Google Scholar]
- Smetana, A. Staphylinidae except Pselaphinae and Scaphidiinae. In Catalogue of Palaearctic Coleoptera. Hydrophiloidea-Histeroidea-Staphylinoidea; Löbl, I., Smetana, A., Eds.; Apollo Books: Stenstrup, Denmark, 2004; Volume 2, pp. 237–698. [Google Scholar]
- Schülke, M. Zur Taxonomie und Faunistik westpaläarktischer Staphylinidae (Coleoptera: Staphylinidae: Omaliinae, Oxytelinae et Tachyporinae). Linzer Biol. Beitr. 2009, 41, 803–844. [Google Scholar]
- Brunke, A.; Newton, A.; Klimaszewski, J.; Majka, C.; Marshall, S. Staphylinidae of Eastern Canada and adjacent United States. Key to subfamilies: Staphylininae: Tribes and subtribes, and species of Staphylinina. Can. J. Arthropod Identif. 2011, 12, 1–110. [Google Scholar]
- Ashe, J.S. Evolution of aedeagal parameres of aleocharine staphylinids (Coleoptera: Staphylinidae: Aleocharinae). Can. Entomol. 1994, 126, 475–491. [Google Scholar] [CrossRef]
- Klimaszewski, J.; Webster, R.P.; Langor, D.W.; Brunke, A.; Davies, A.; Bourdon, C.; Labrecque, M.; Newton, A.F.; Dorval, J.-A.; Frank, J.H. Aleocharine Rove Beetles of Eastern Canada (Coleoptera, Staphylinidae, Aleocharinae): A Glimpse of Megadiversity; Springer Nature: Cham, Switzerland, 2018; pp. xvi + 1–902. [Google Scholar] [CrossRef]
- Assing, V.; Wunderle, P. A revision of the species of the subfamily Habrocerinae (Coleoptera: Staphylinidae) of the world. Rev. Suisse Zool. 1995, 102, 307–359. [Google Scholar]
- Yin, Z.-W.; Li, L.-Z. A new species and an additional record of Pseudotachinus Cameron from China (Coleoptera: Staphylinidae: Tachyporinae). Zootaxa 2018, 4425, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Schülke, M. Zur Kenntnis der Gattung Tachinoplesius Bernhauer (Coleoptera, Staphylinidae, Tachyporinae). Linzer Biol. Beitr. 2006, 38, 889–901. [Google Scholar]
- Yin, Z.-W.; Li, L.-Z. A new species of Parabolitobius Li, Zhao & Sakai, 2000 from central China (Coleoptera: Staphylinidae: Tachyporinae). Zootaxa 2019, 4695, 492–496. [Google Scholar] [CrossRef]
- Gusarov, V.I. Phylogeny of the family Staphylinidae based on molecular data: A review. In Biology of Rove Beetles (Staphylinidae): Life History, Evolution, Ecology and Distribution; Betz, O., Irmler, U., Klimaszewski, J., Eds.; Springer Nature: Cham, Switzerland, 2018; pp. 7–25. [Google Scholar] [CrossRef]
- Hunt, T.; Bergsten, J.; Levkanicova, Z.; Papadopoulou, A.; John, O.S.; Wild, R.; Hammond, P.M.; Ahrens, D.; Balke, M.; Caterino, M.S.; et al. A comprehensive phylogeny of beetles reveals the evolutionary origins of a superradiation. Science 2007, 318, 1913–1916. [Google Scholar] [CrossRef]
- Zhang, S.-Q.; Che, L.-H.; Li, Y.; Liang, D.; Pang, H.; Ślipiński, A.; Zhang, P. Evolutionary history of Coleoptera revealed by extensive sampling of genes and species. Nat. Commun. 2018, 9, 205. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-Y.; Li, L.-Z.; Yin, Z.-W. Three new species of the subgenus Latotachinus Ullrich, 1975 (Coleoptera: Staphylinidae: Tachyporinae) from China. Ann. Soc. Entomol. Fr. (N. S.) 2021, 1–10. [Google Scholar] [CrossRef]
- Naomi, S.-I. Comparative morphology of the Staphylinidae and the allied groups (Coleoptera, Staphylinoidea). I. Introduction, head sutures, eyes and ocelli. Kontyû Tokyo 1987, 55, 450–458. [Google Scholar]
- Naomi, S.-I. Comparative morphology of the Staphylinidae and the allied groups (Coleoptera, Staphylinoidea). II. Cranial structure and tentorium. Kontyû Tokyo 1987, 55, 666–675. [Google Scholar]
- Lawrence, J.F.; Ślipiński, A.; Seago, A.E.; Thayer, M.K.; Newton, A.F.; Marvaldi, A.E. Phylogeny of the Coleoptera based on morphological characters of adults and larvae. Ann. Zool. 2011, 61, 1–217. [Google Scholar] [CrossRef]
- Lawrence, J.F.; Ślipiński, A. Australian Beetles: Morphology, Classification and Keys; CSIRO Publishing: Collingwood, Victoria, Australia; Volume 1, pp. viii + 1–561.
- Clarke, D.J. Testing the phylogenetic utility of morphological character systems, with a revision of Creophilus Leach (Coleoptera: Staphylinidae). Zool. J. Linn. Soc. 2011, 163, 723–812. [Google Scholar] [CrossRef]
- Hansen, M. Phylogeny and classification of the Staphyliniform beetle families (Coleoptera). Biol. Skr. 1997, 48, 1–339. [Google Scholar]
- Schülke, M. Zwei neue Arten der Gattung Pseudotachinus Cameron aus dem Himalaja-Gebiet (Coleoptera, Staphylinidae, Tachyporinae). Linzer Biol. Beitr. 2005, 37, 1609–1624. [Google Scholar]
- Maruyama, M. Revision of the Palearctic species of the myrmecophilous genus Pella (Coleoptera, Staphylinidae, Aleocharinae). Natl. Sci. Mus. Monogr. 2006, 32, 1–207. [Google Scholar]
- Naomi, S.-I. Comparative morphology of the Staphylinidae and the allied groups (Coleoptera, Staphylinoidea). VII. Metendosternite and wings. Jpn. J. Entomol. 1989, 57, 82–90. [Google Scholar]
- Cai, C.; Beattie, R.; Huang, D. Jurassic olisthaerine rove beetles (Coleoptera, Staphylinidae): 165 million years of morphological and probably behavioral stasis. Gondwana Res. 2015, 28, 425–431. [Google Scholar] [CrossRef]
- Yamamoto, S.; Maruyama, M. Revision of the seashore-dwelling subgenera Emplenota Casey and Triochara Bernhauer (Coleoptera: Staphylinidae: Genus Aleochara) from Japan. Zootaxa 2012, 3517, 1–52. [Google Scholar] [CrossRef]
- Hammond, P.M. The micro-structure, distribution and possible function of peg-like setae in male Coleoptera. Insect Syst. Evol. 1972, 3, 40–54. [Google Scholar] [CrossRef]
- Jenkins Shaw, J.; Żyła, D.; Solodovnikov, A. A spectacular new genus of Staphylinini rove beetle from the tropical Andes and its phylogenetic assessment (Coleoptera: Staphylinidae). Invertebr. Syst. 2017, 31, 713–722. [Google Scholar] [CrossRef]
Short Biography of Author

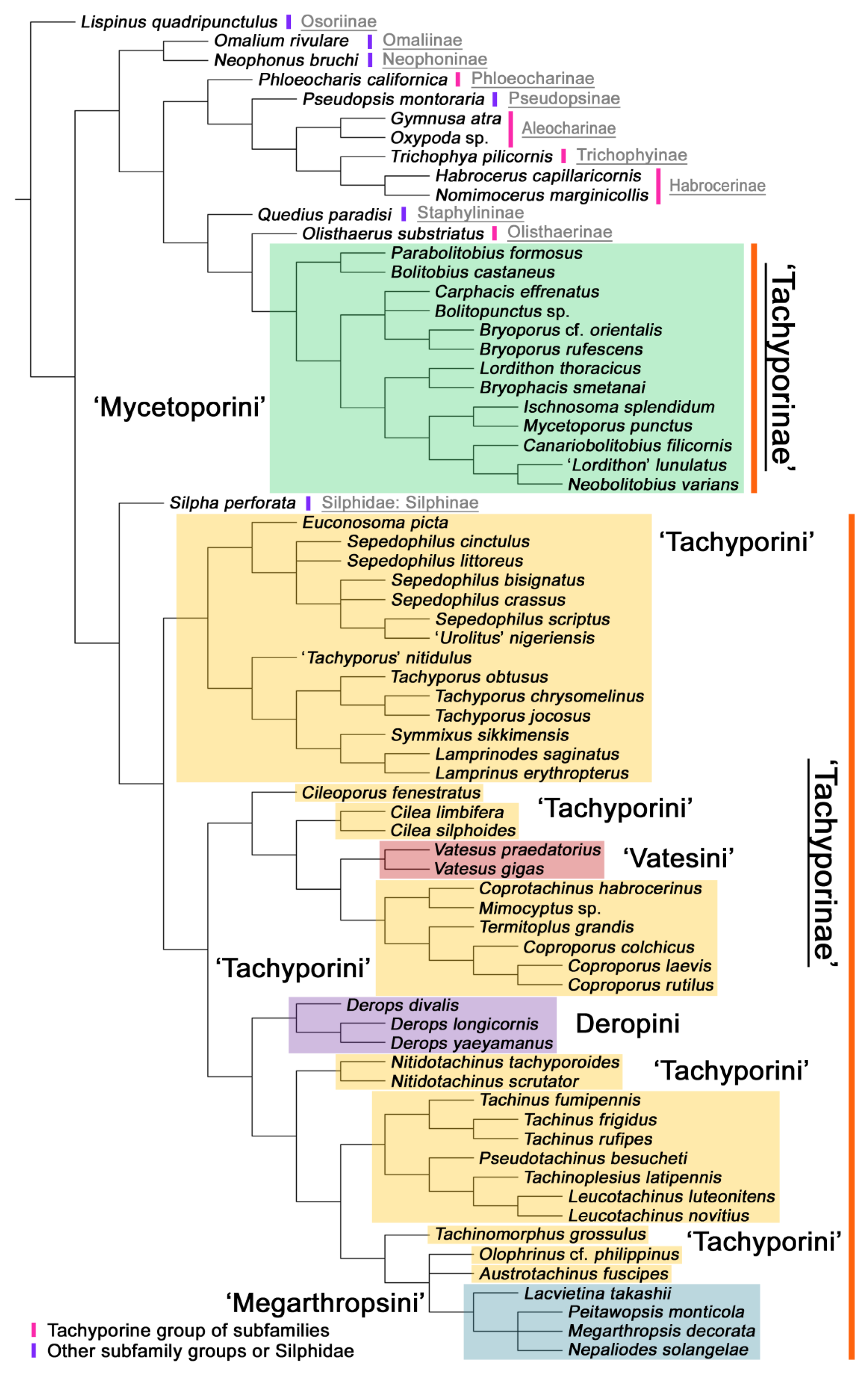
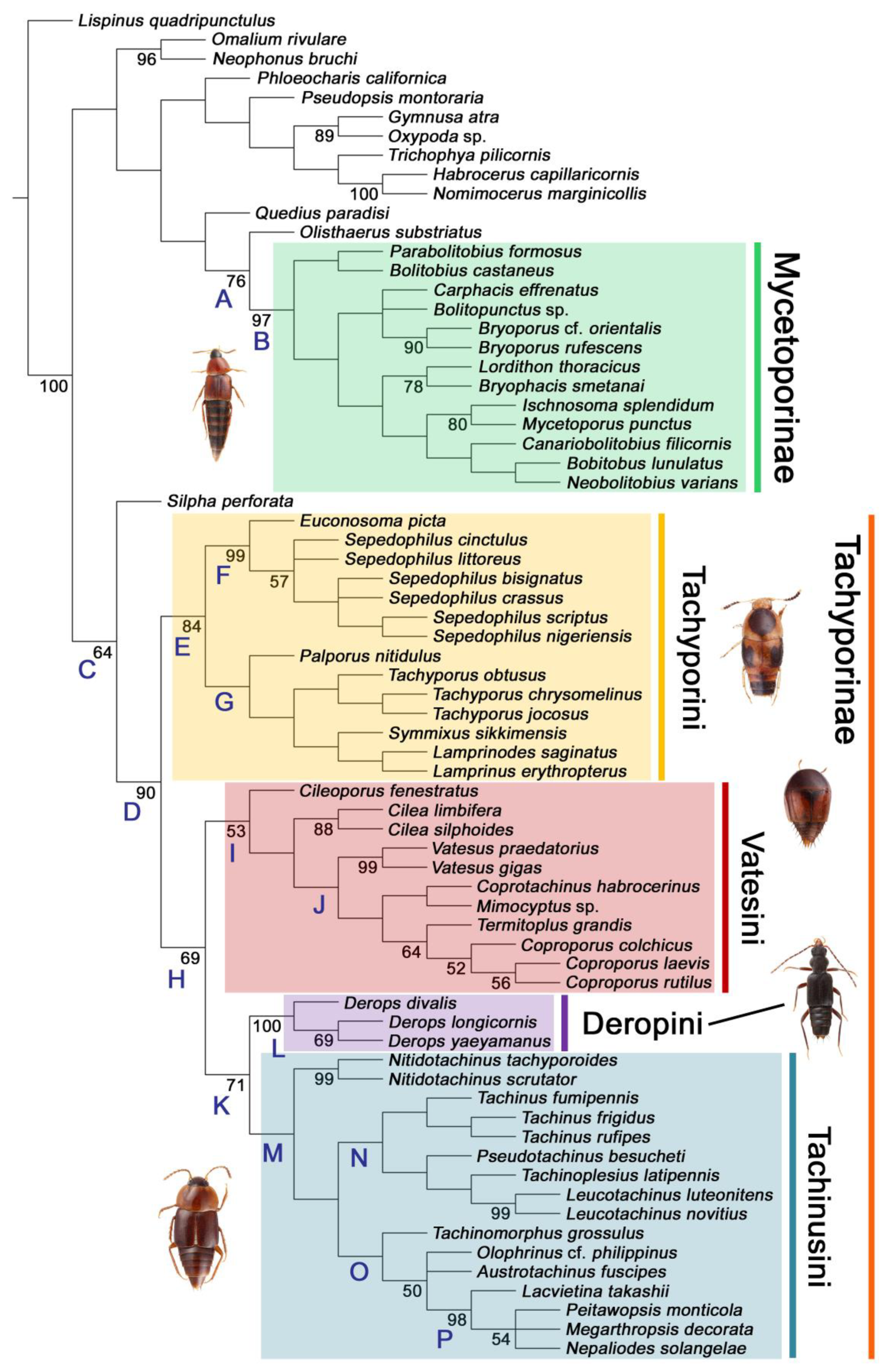
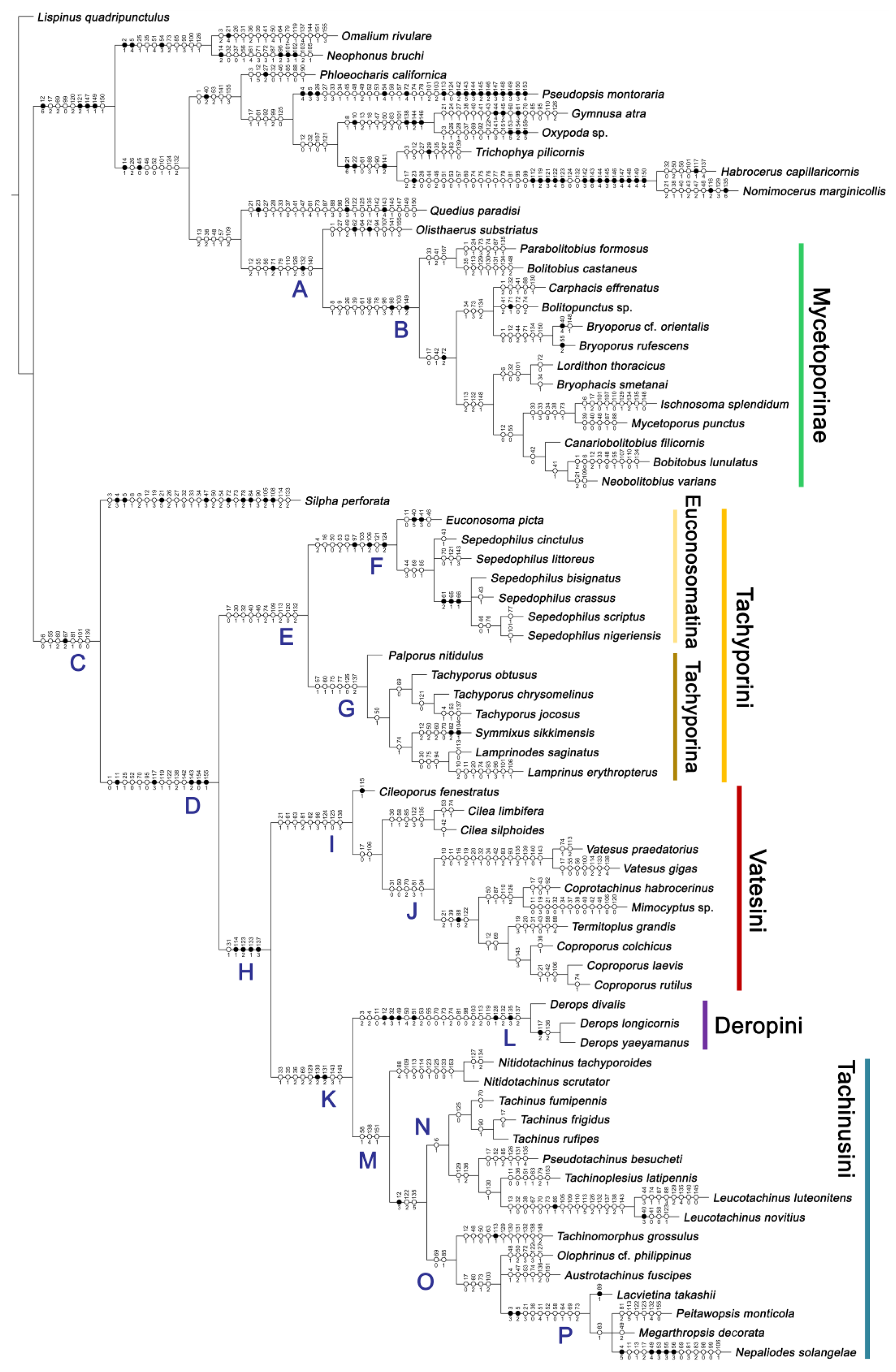
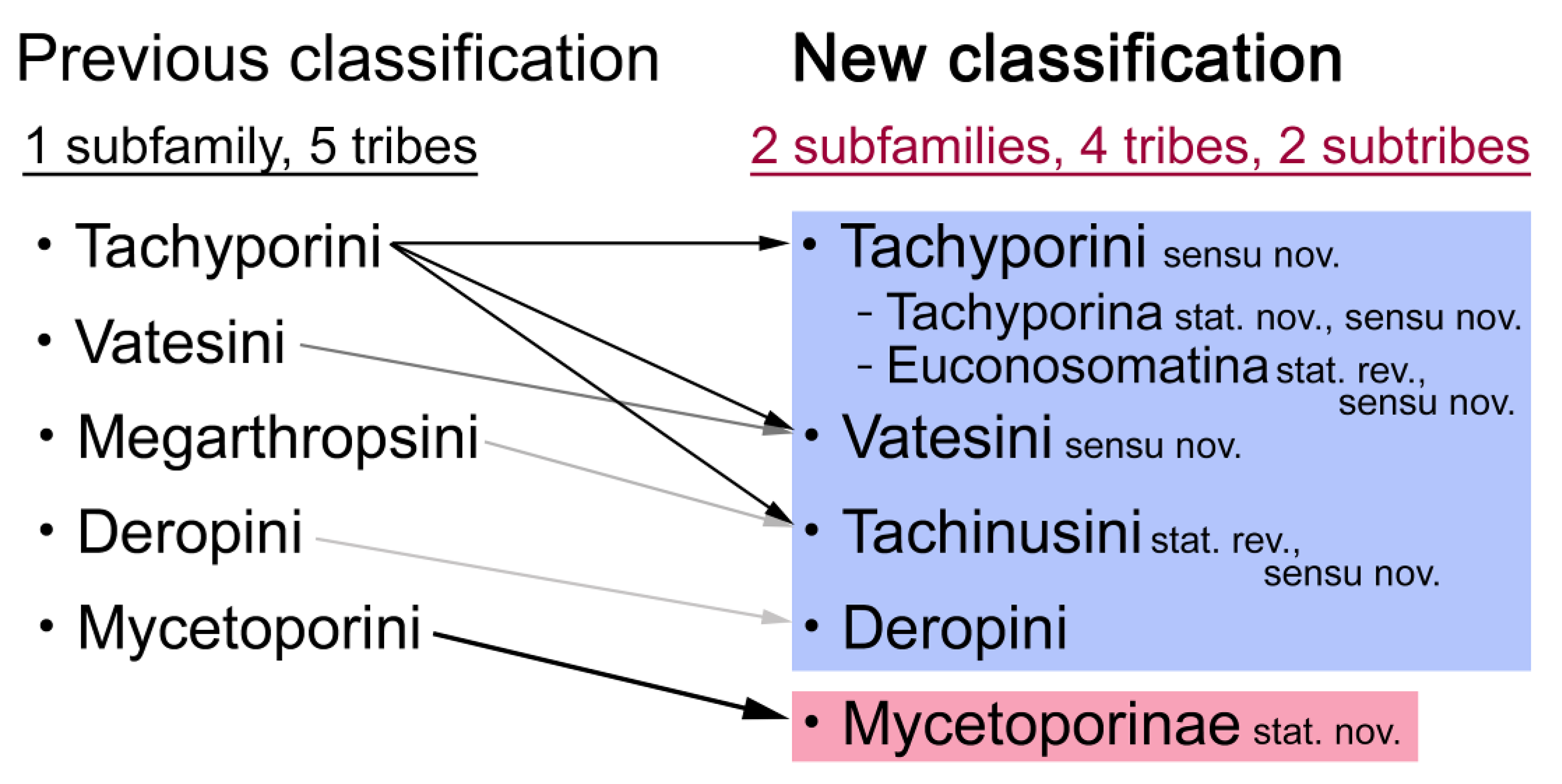



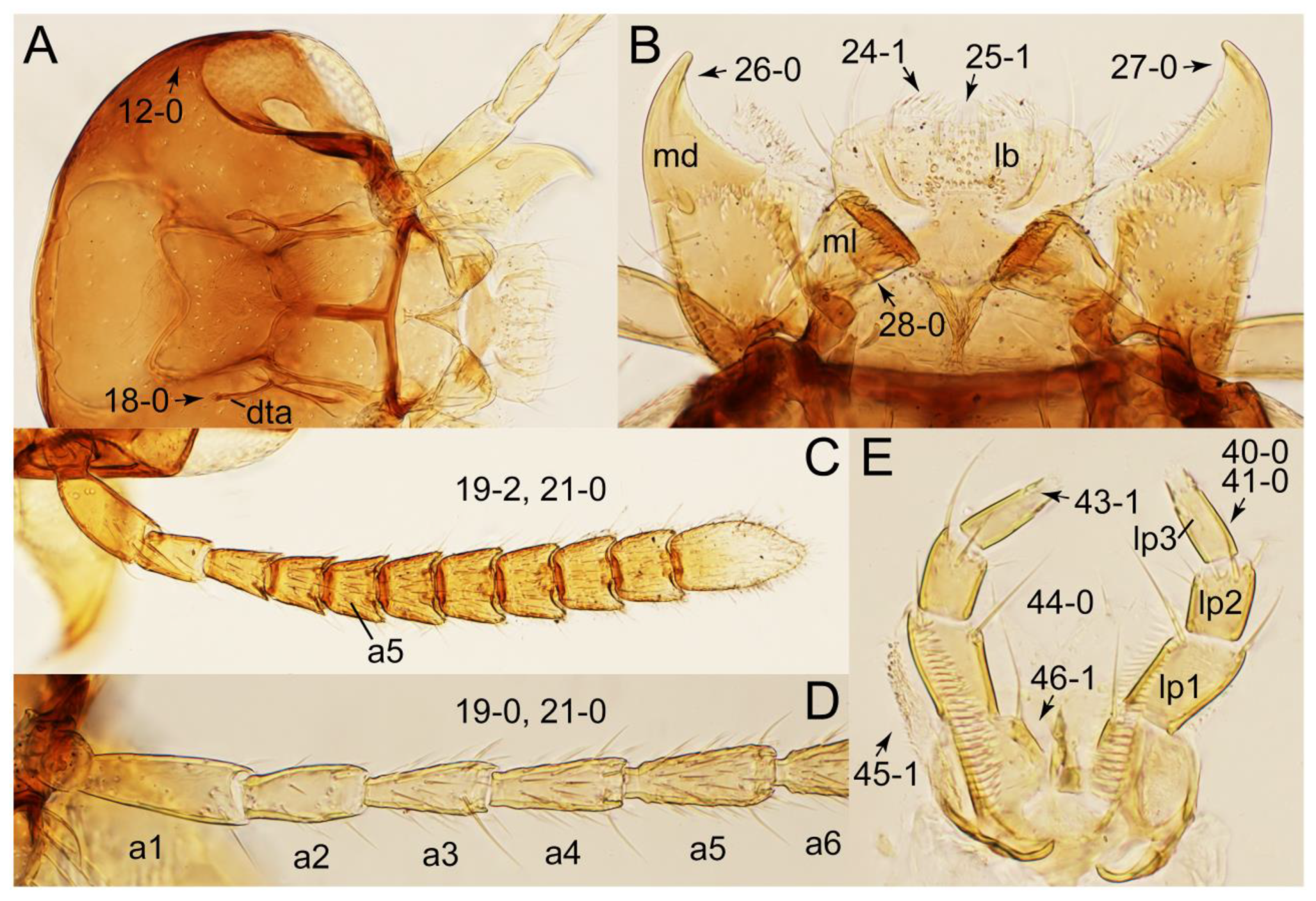
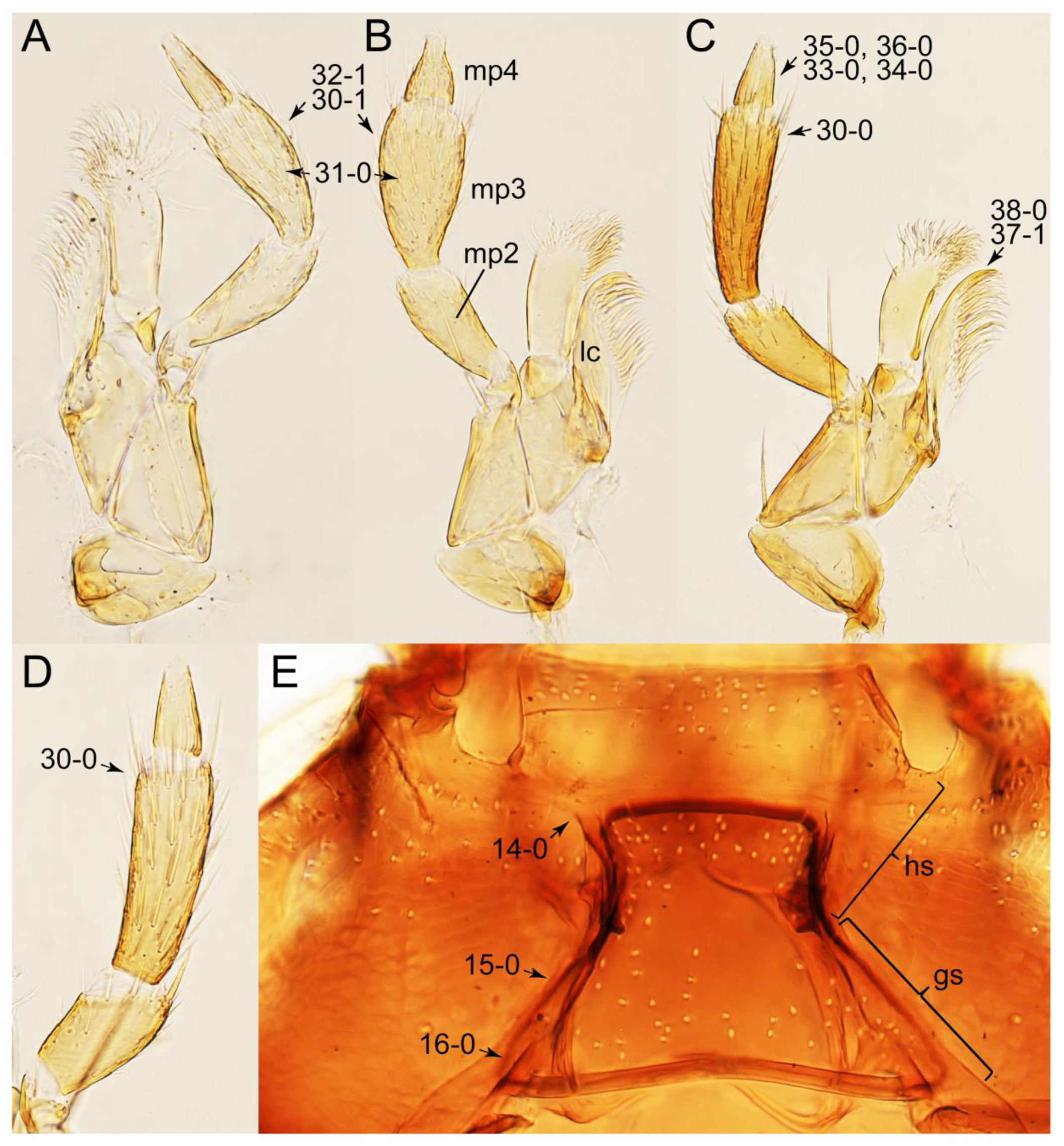
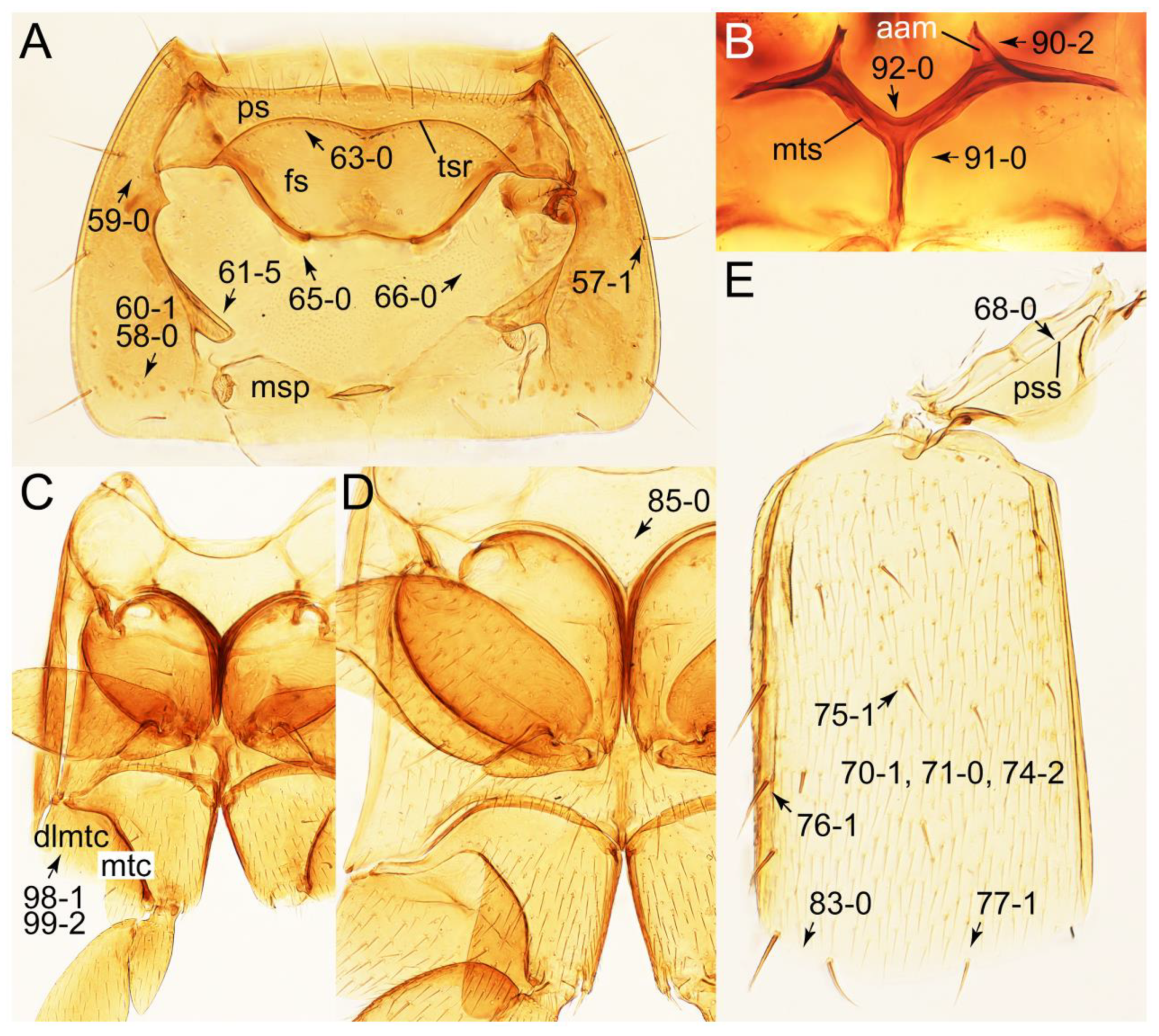

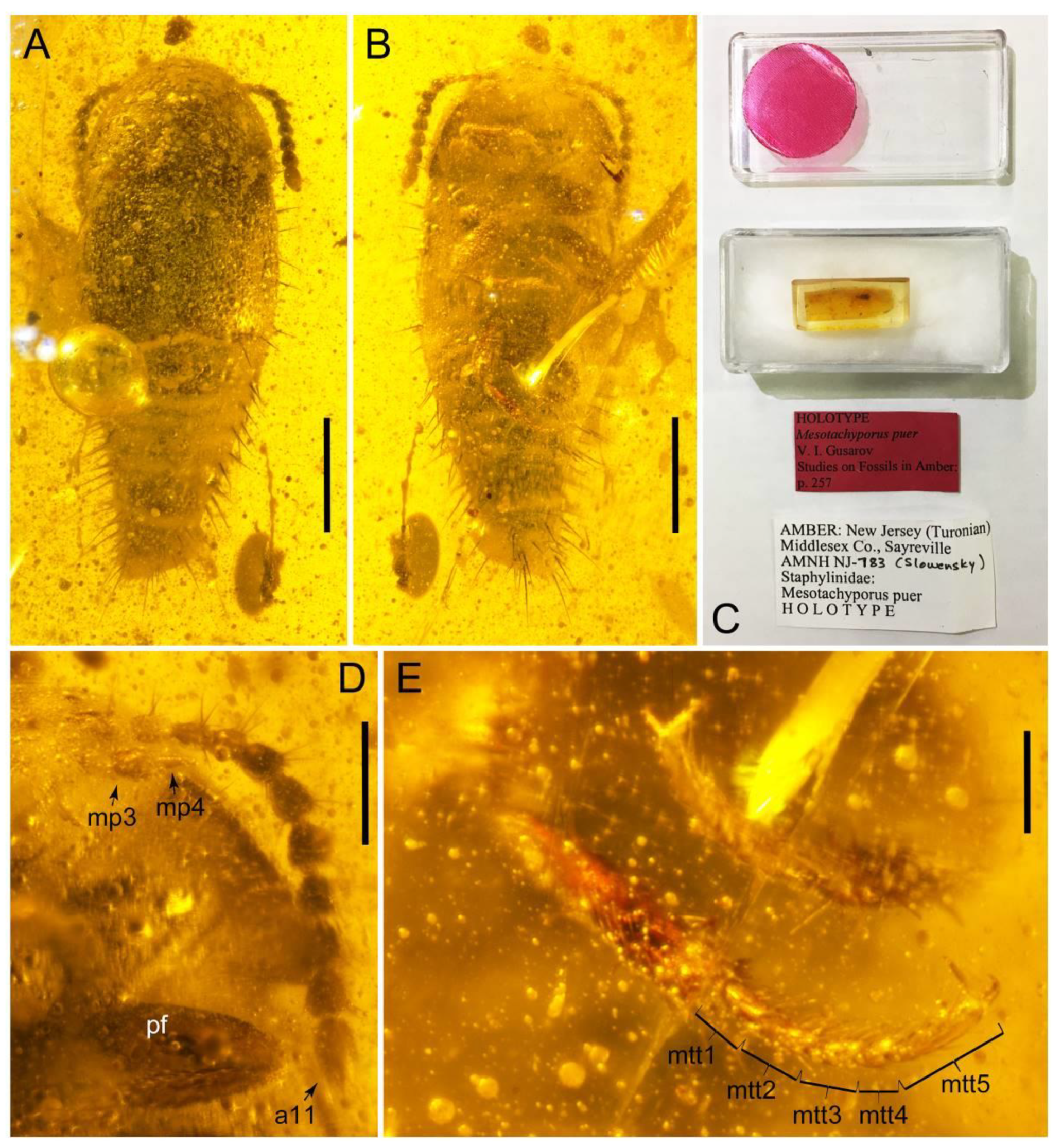

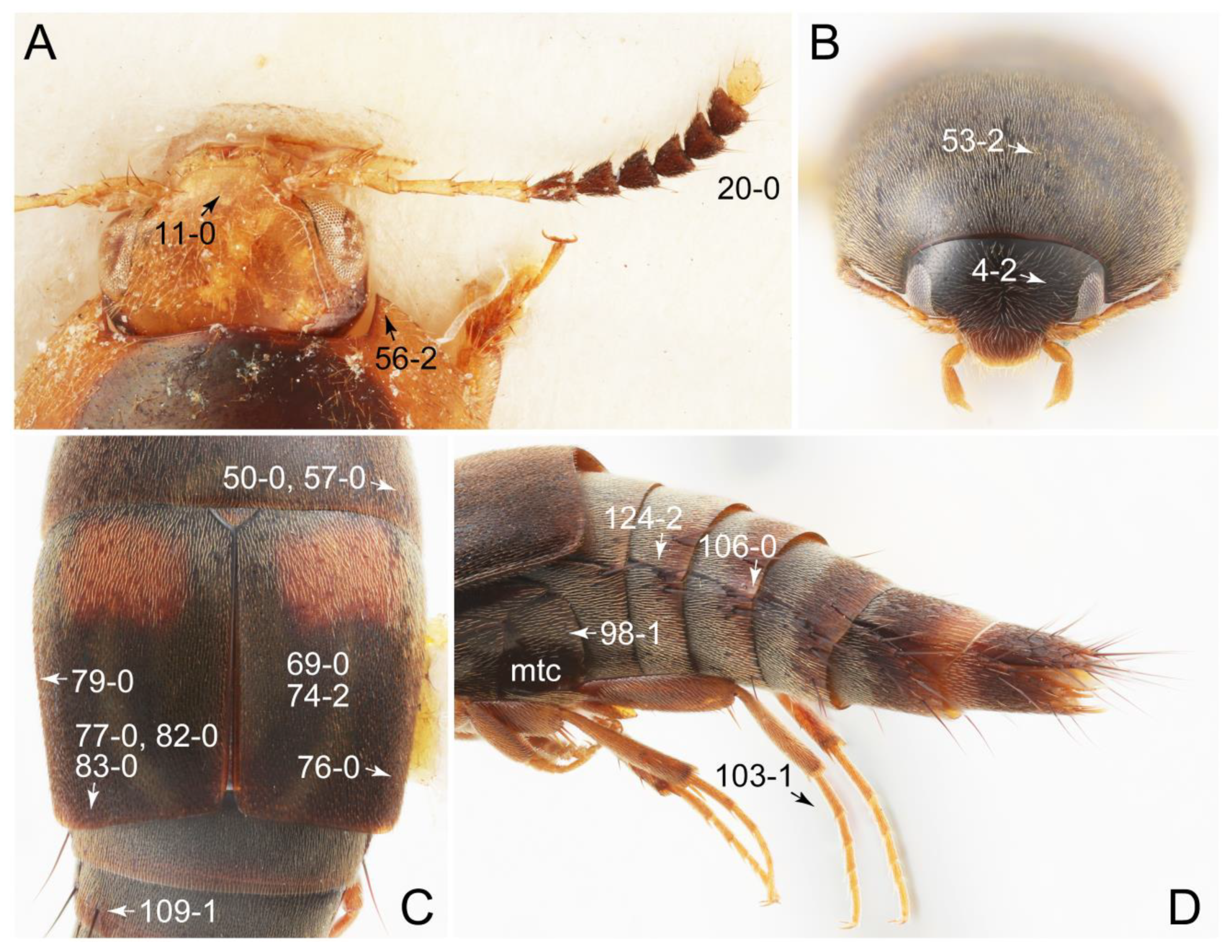



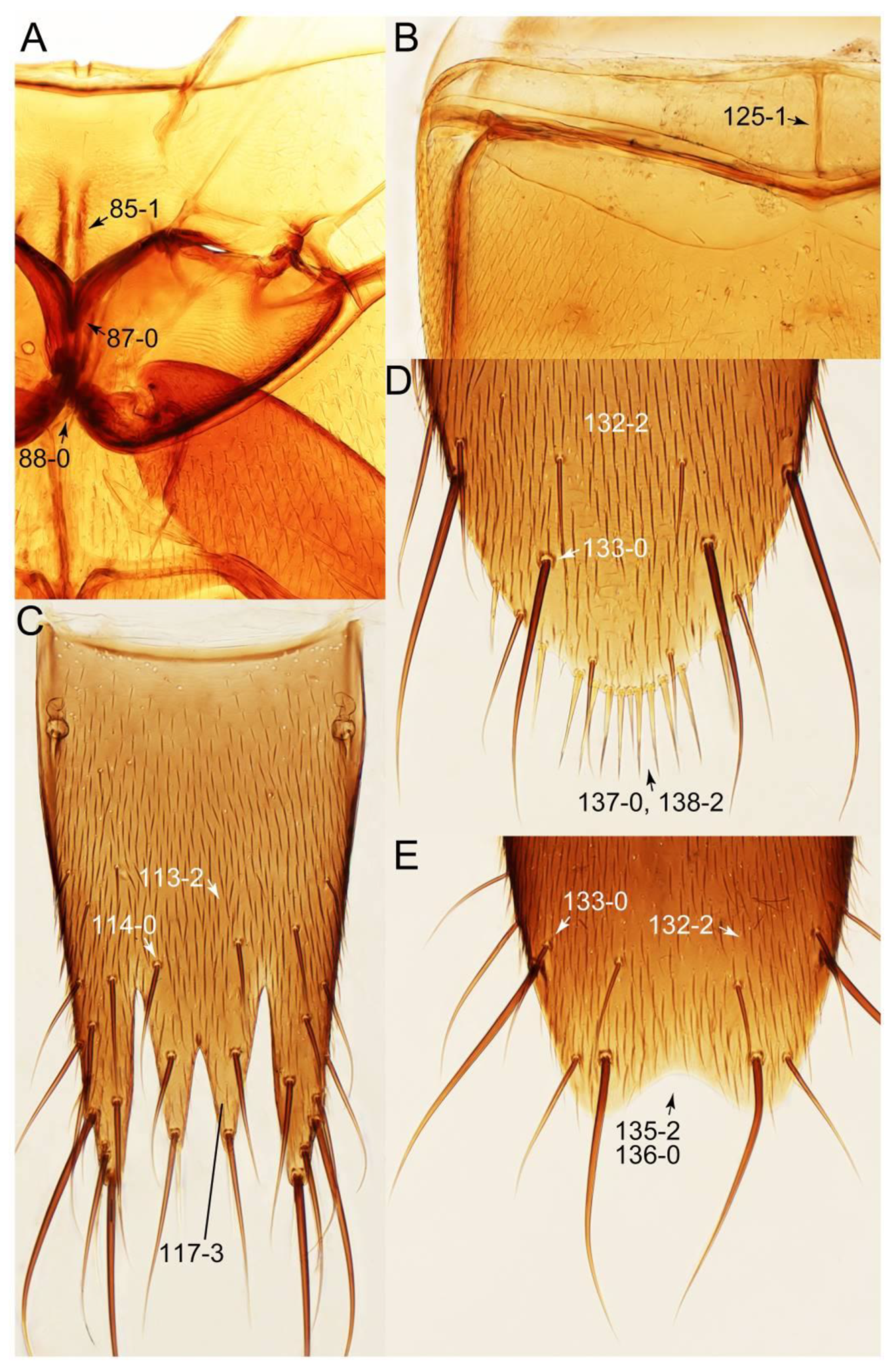
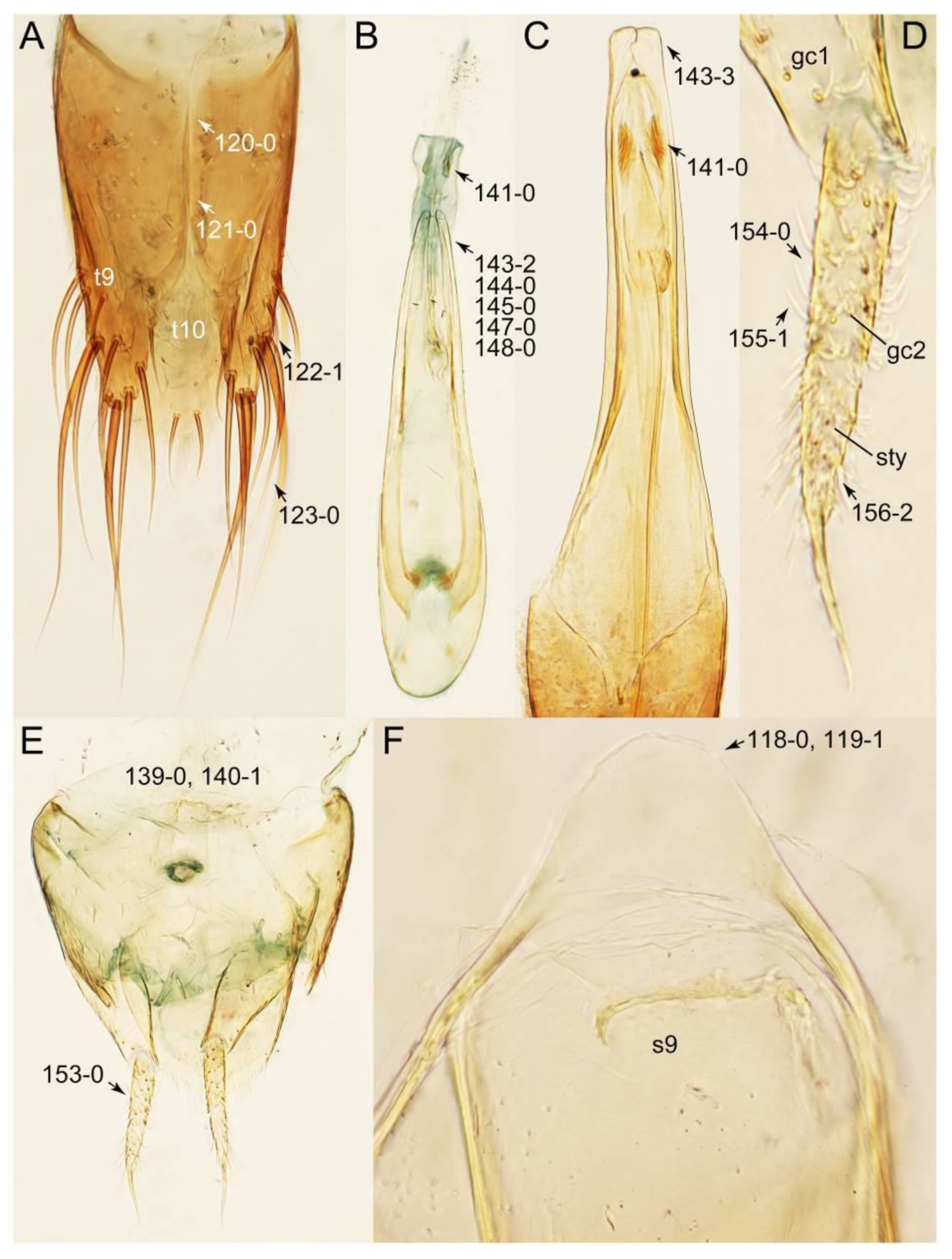





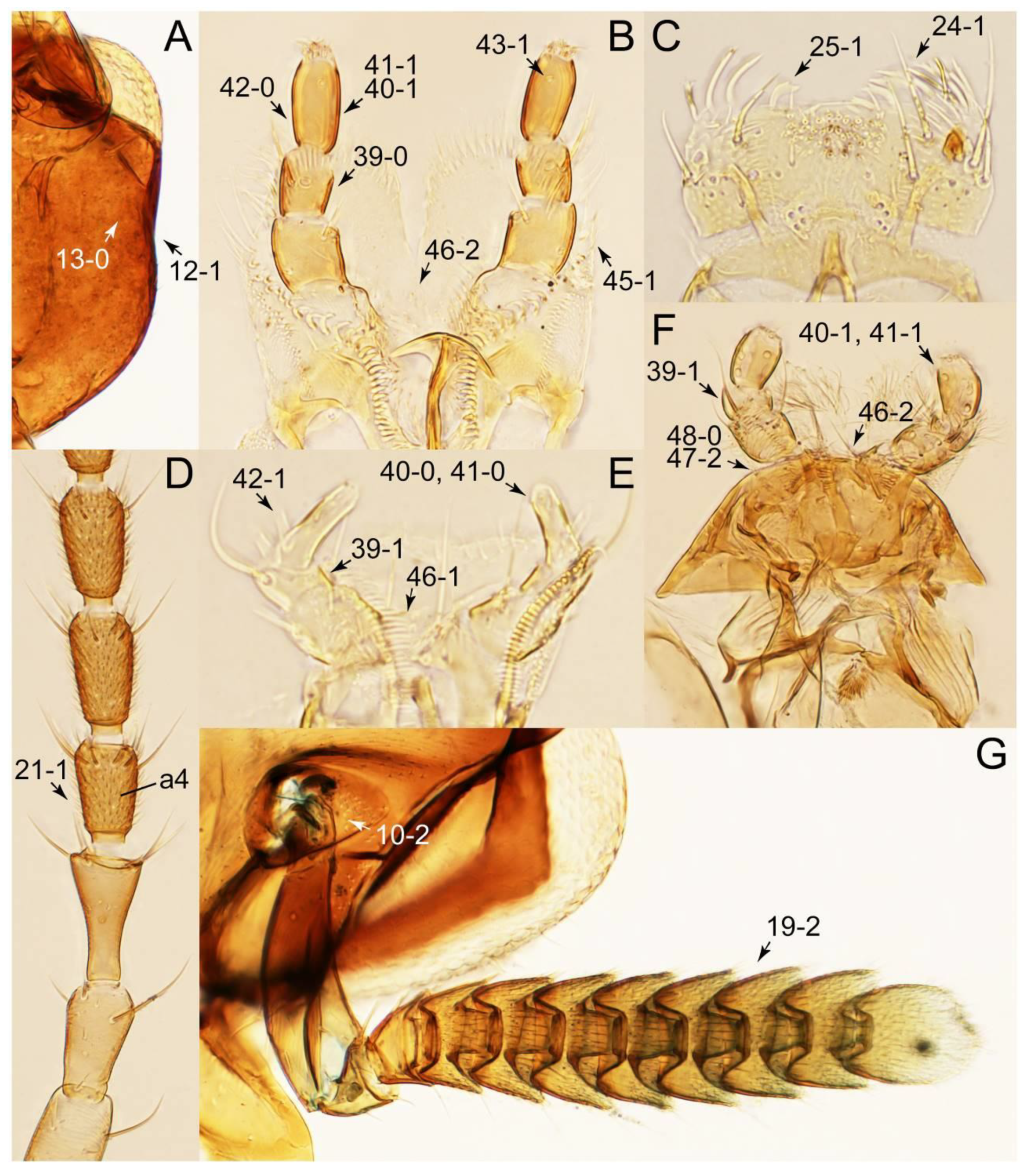
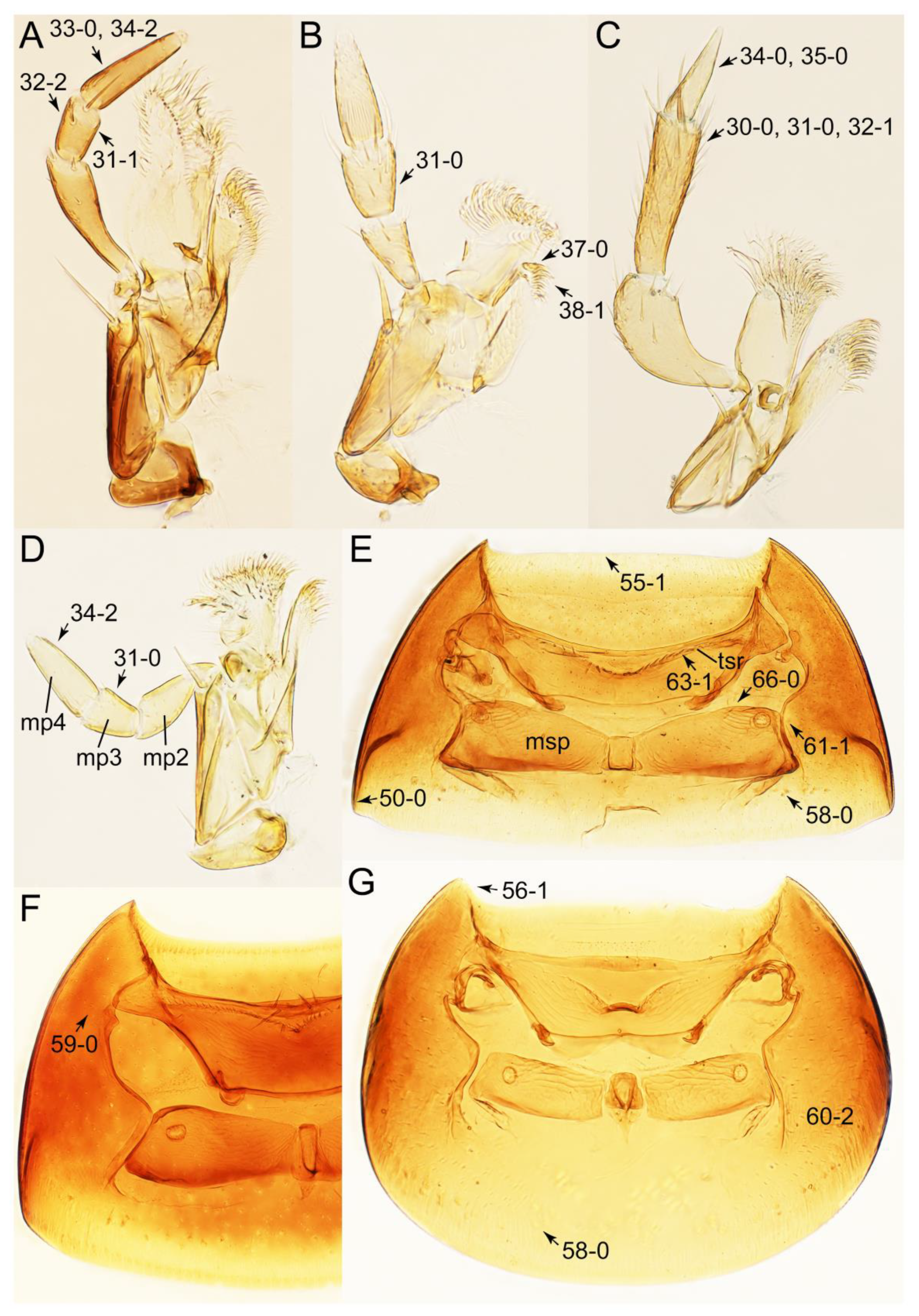

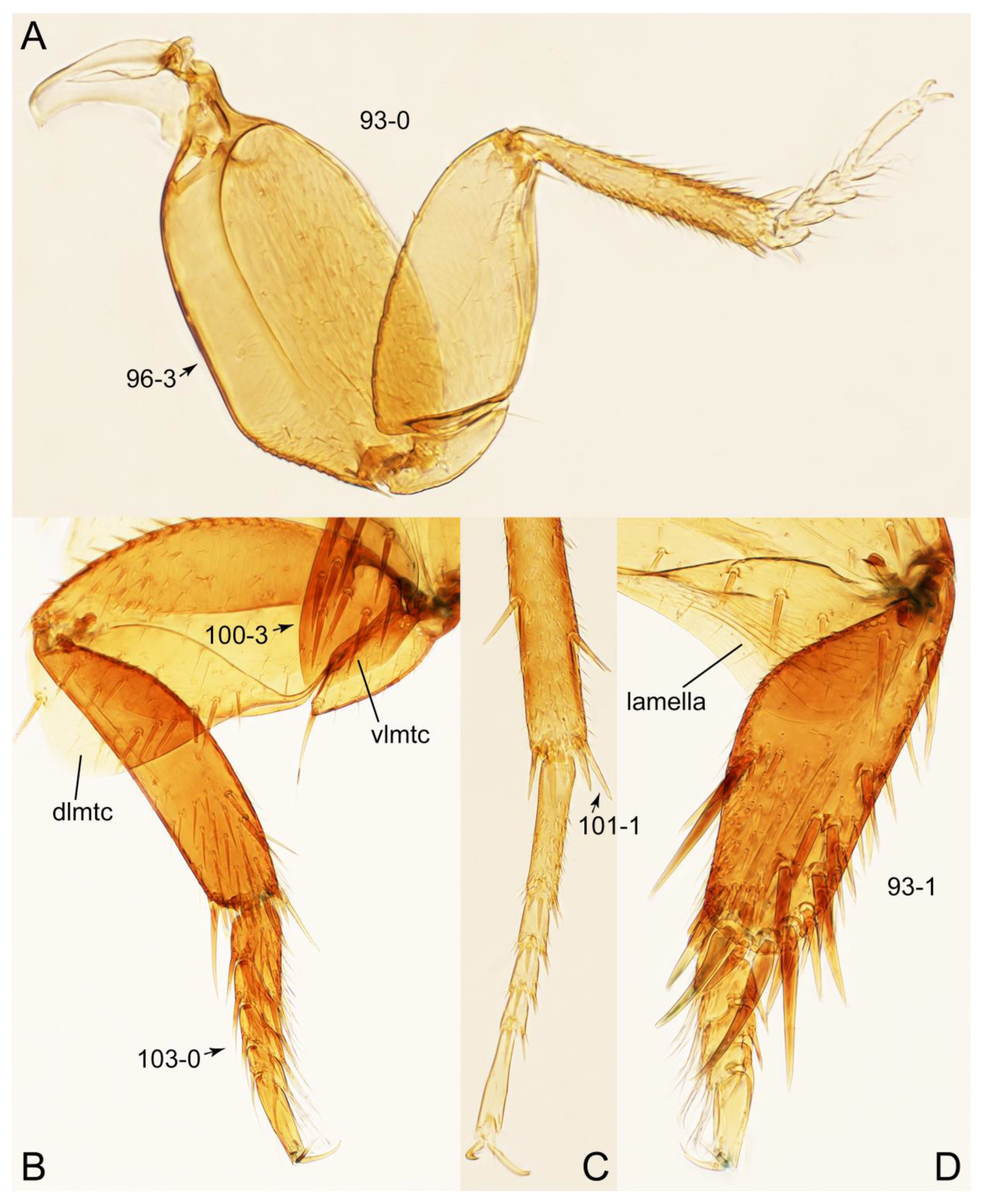

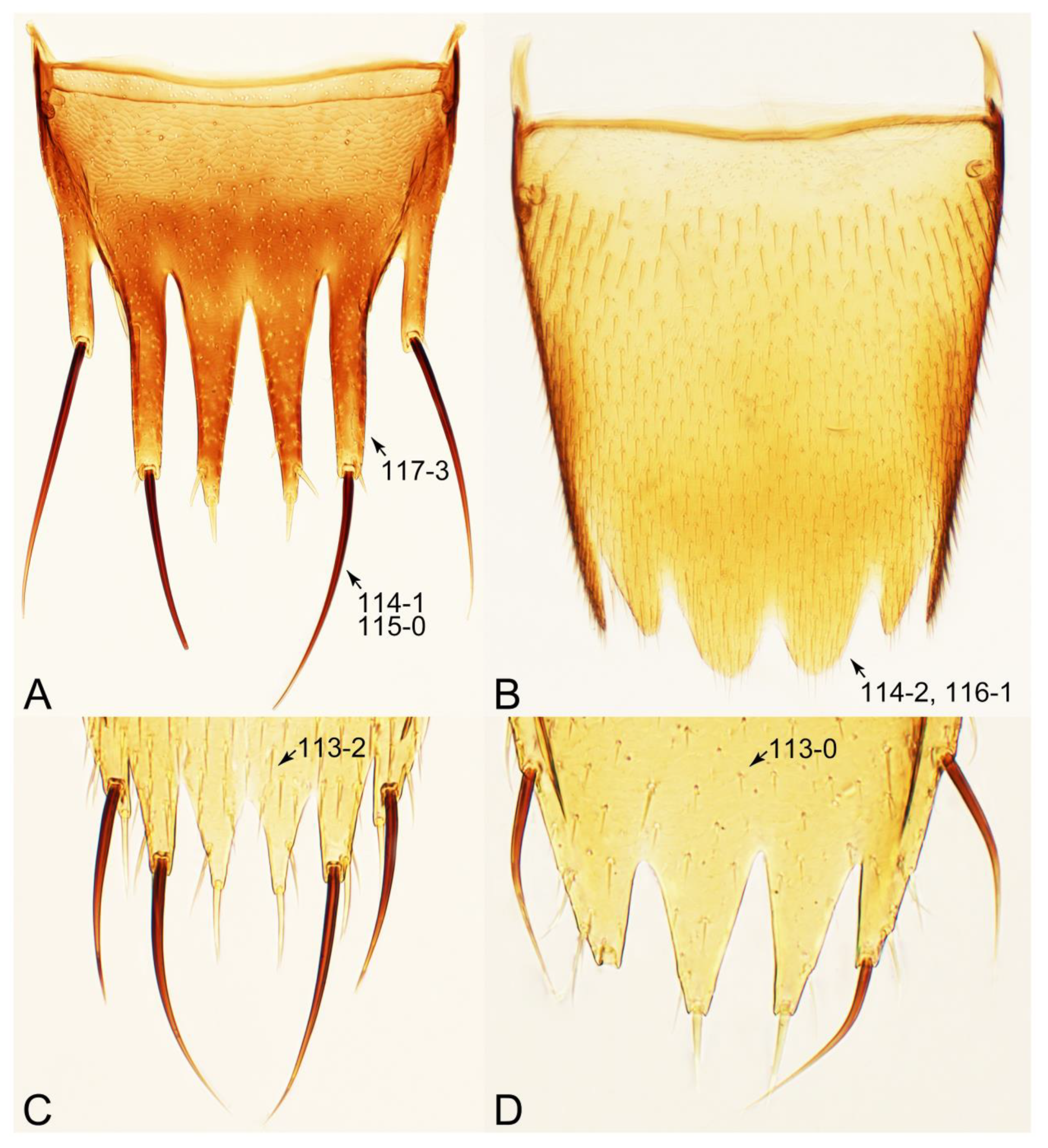
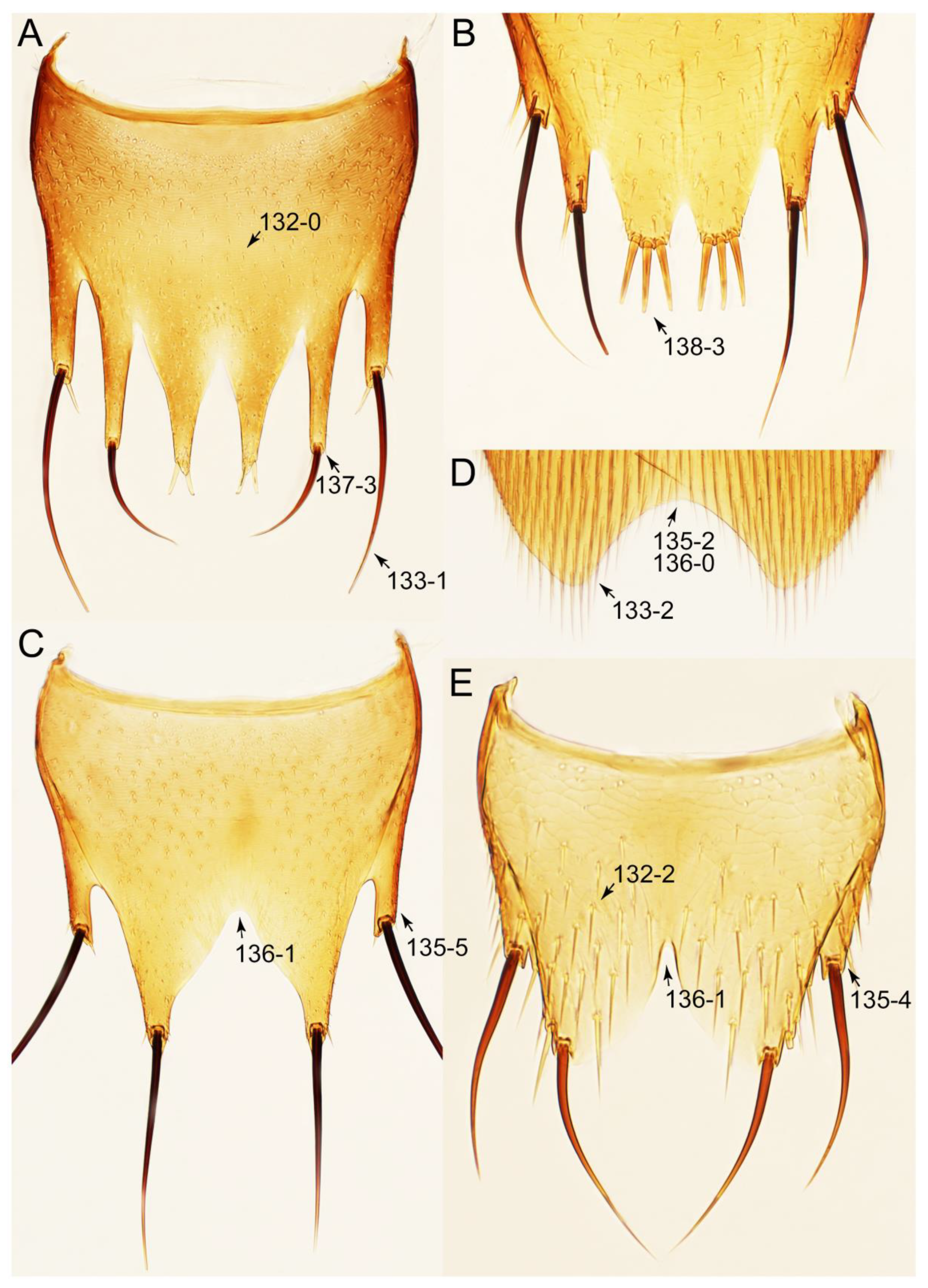
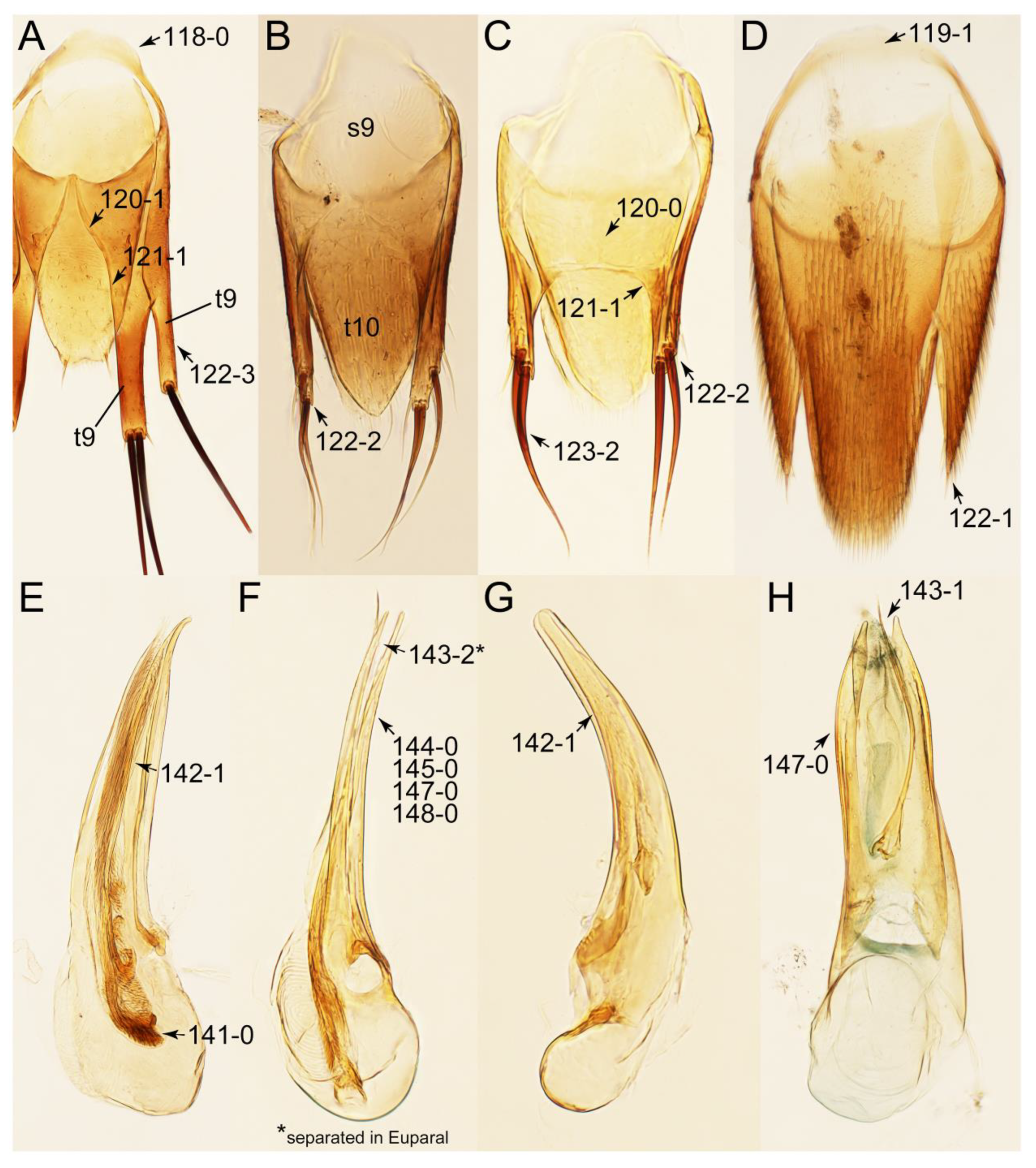




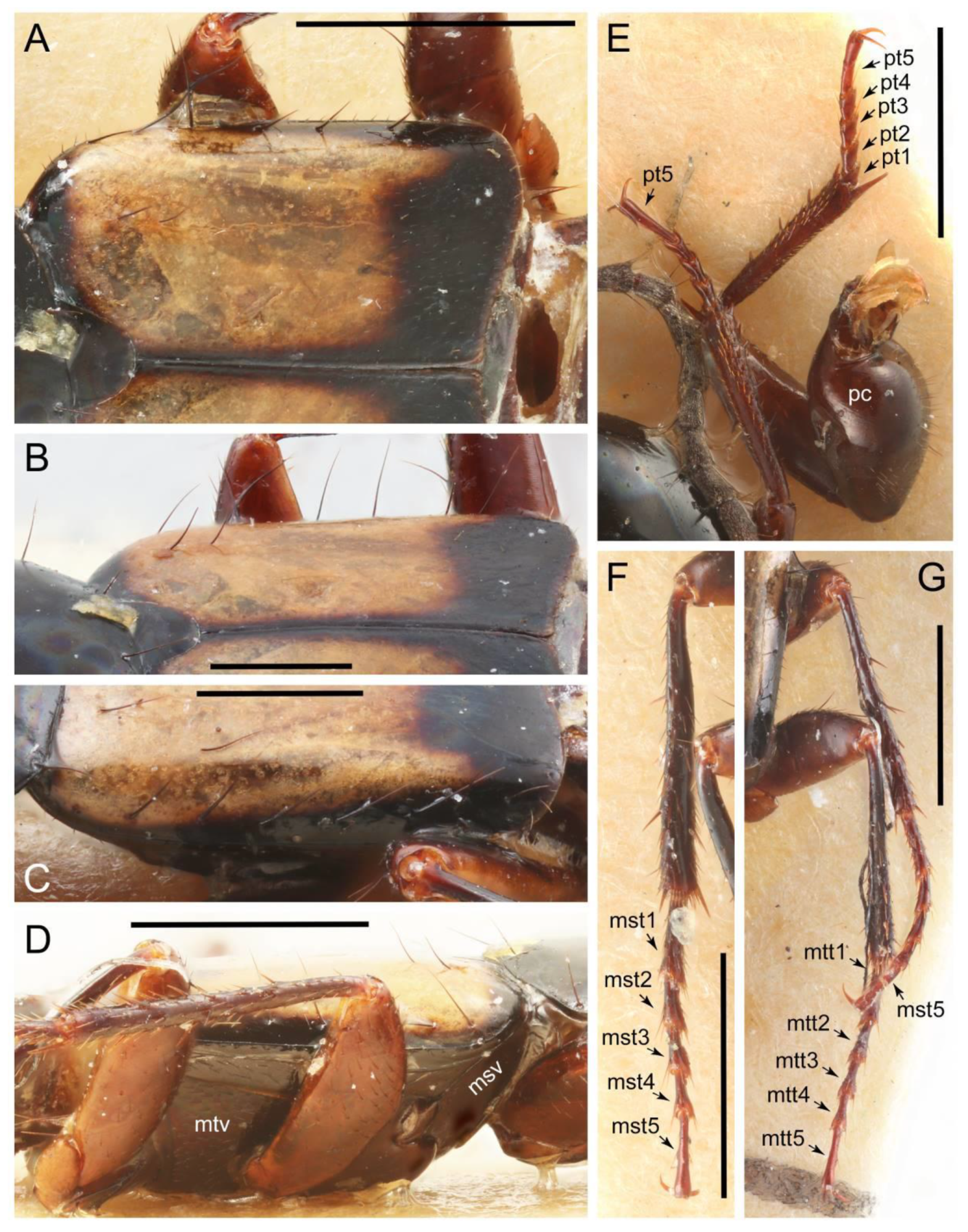
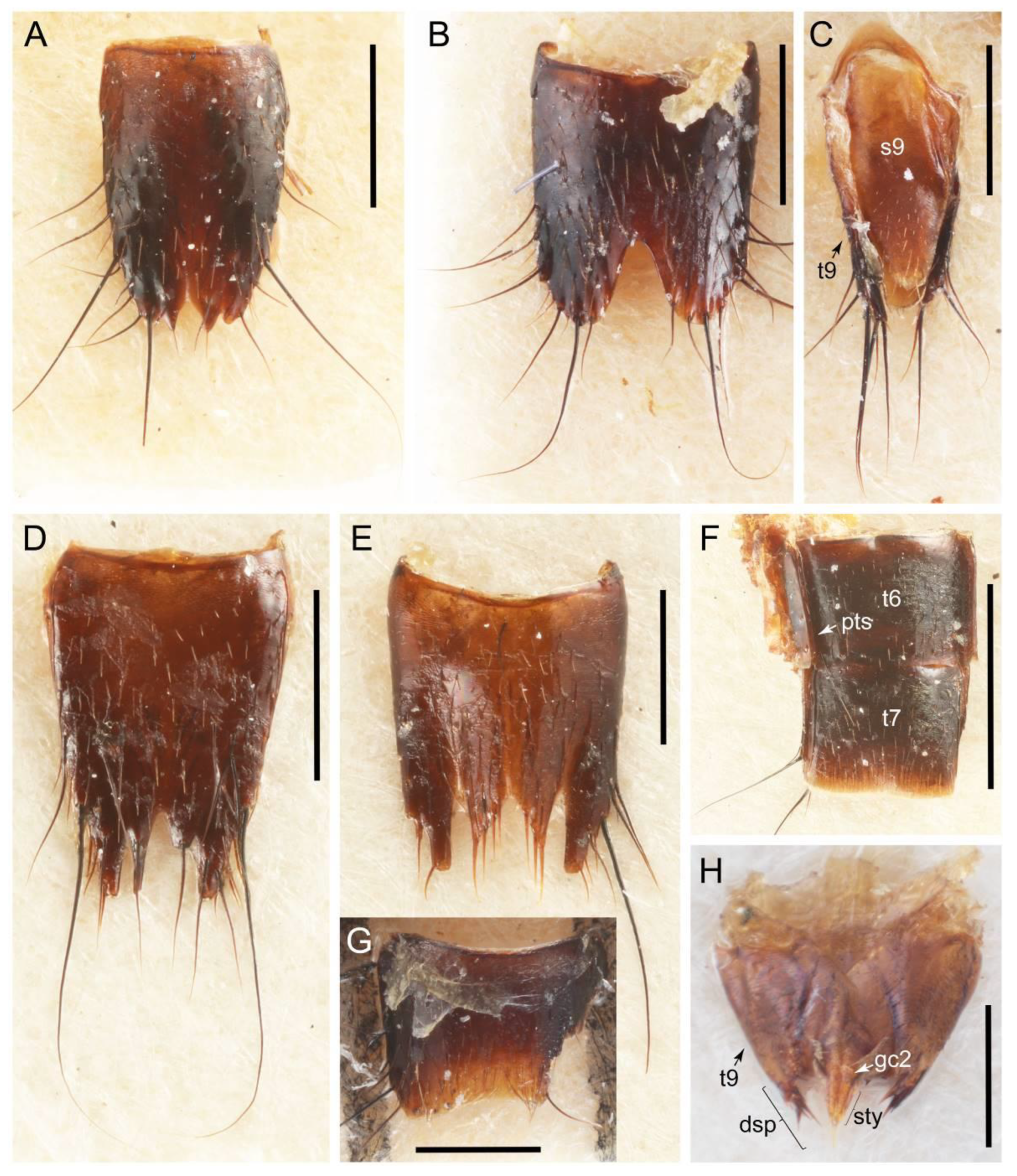





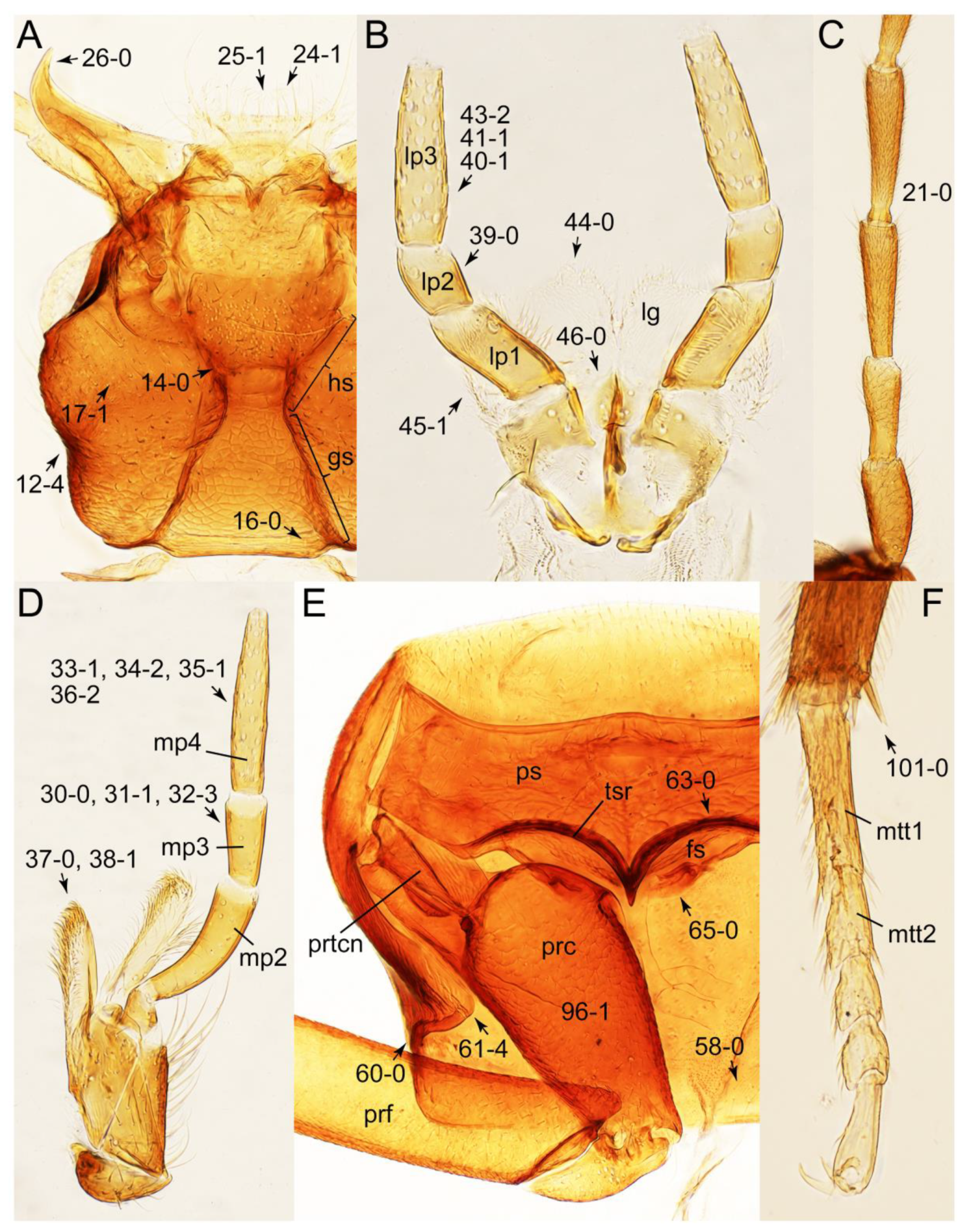
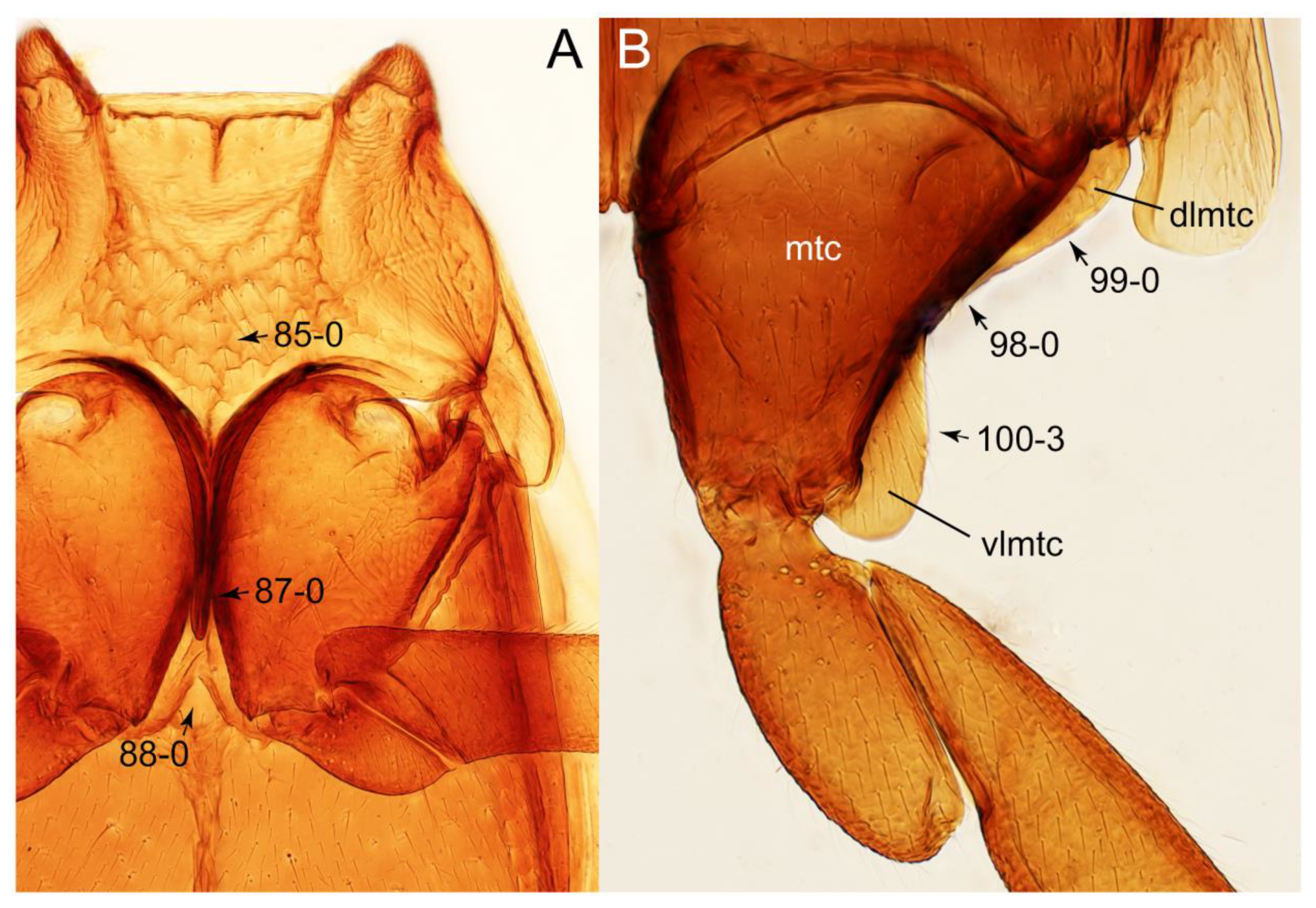
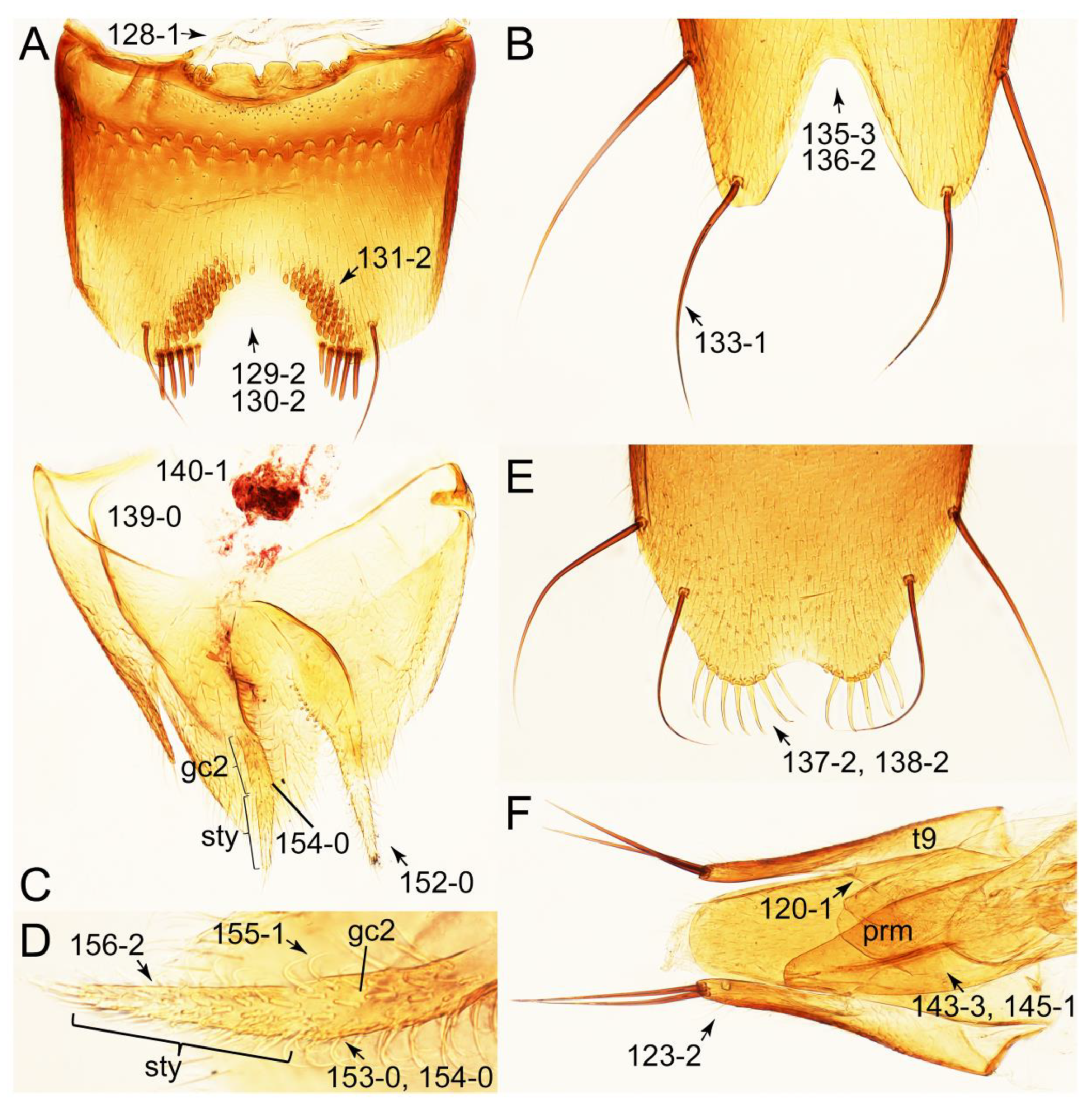





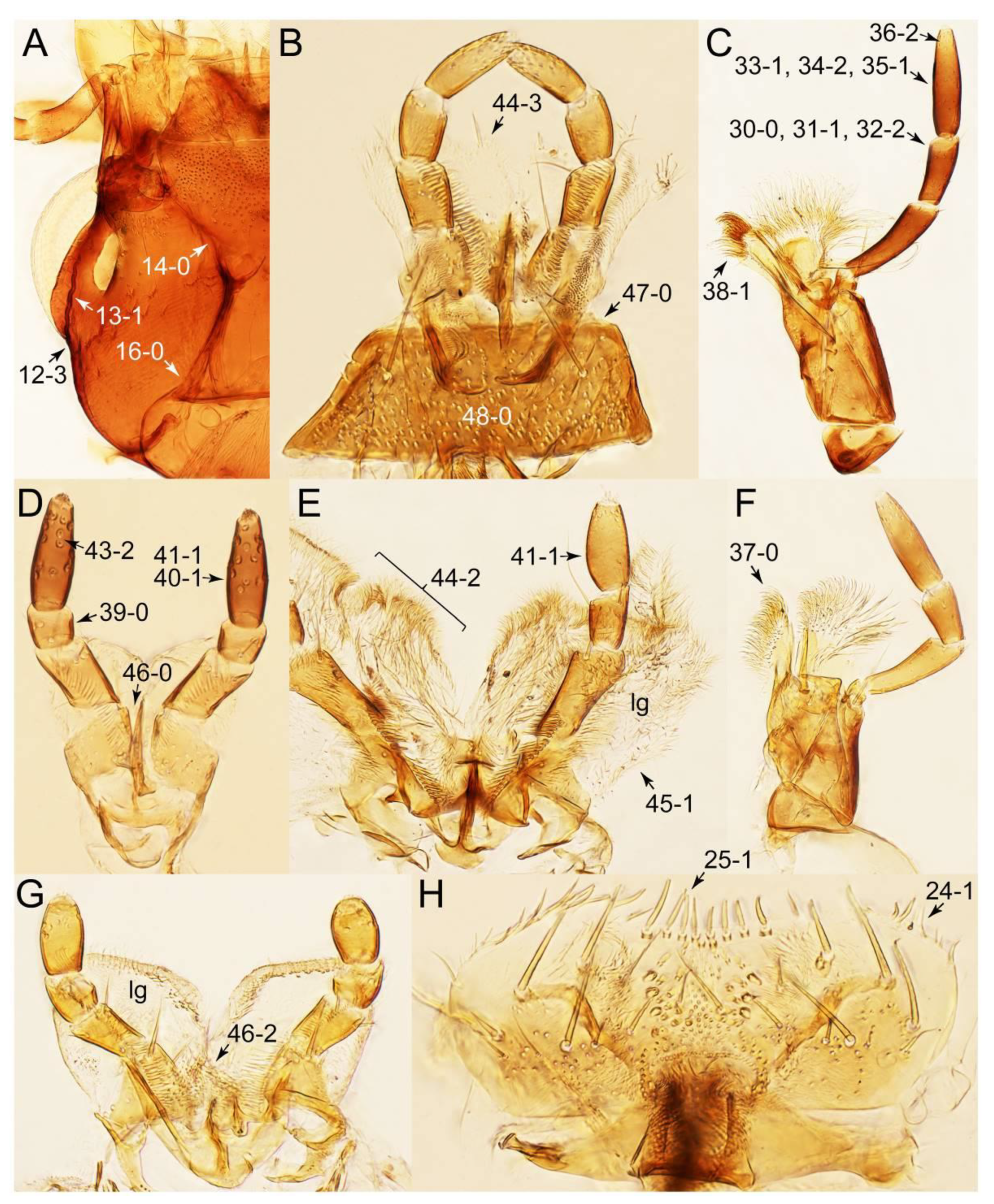
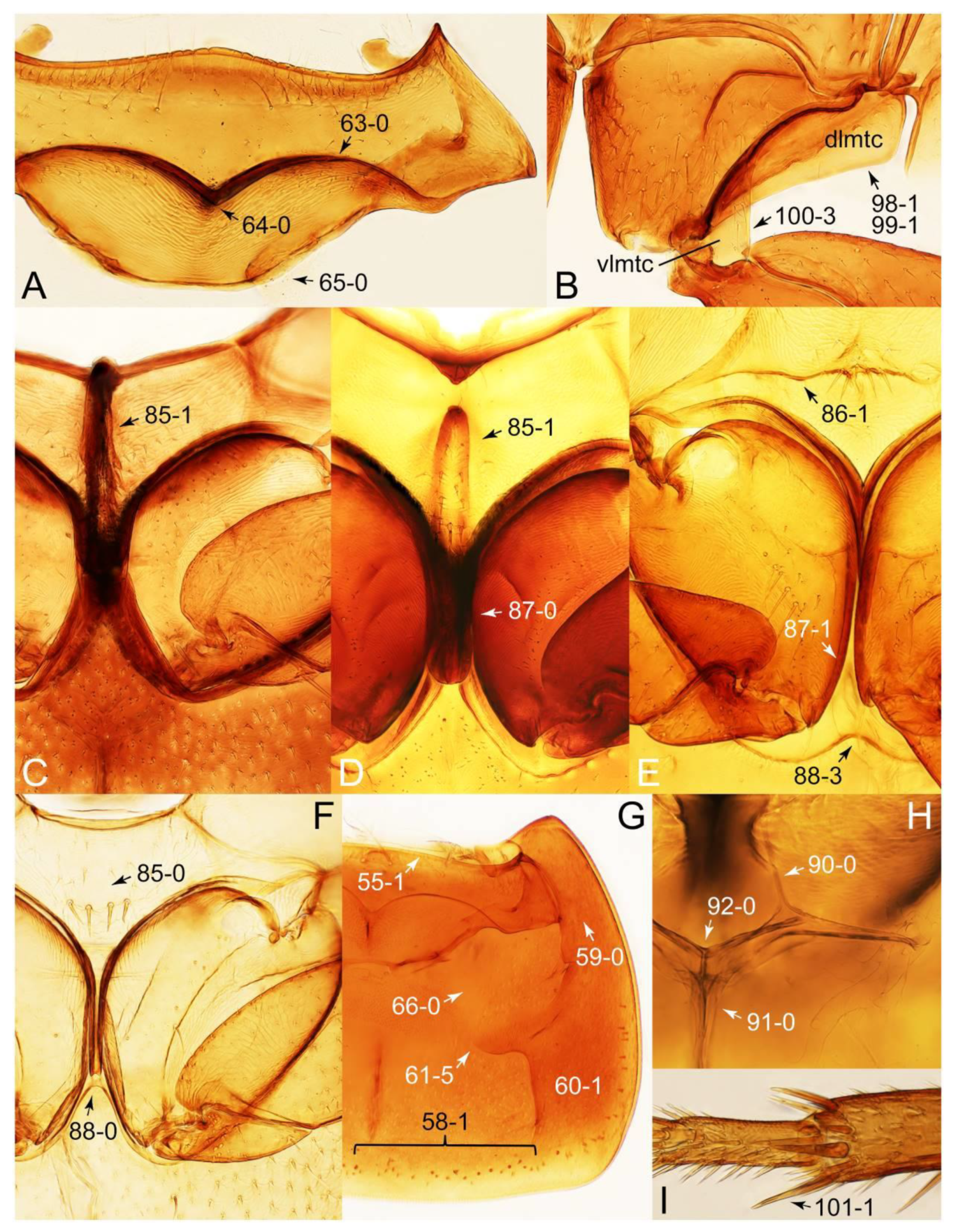
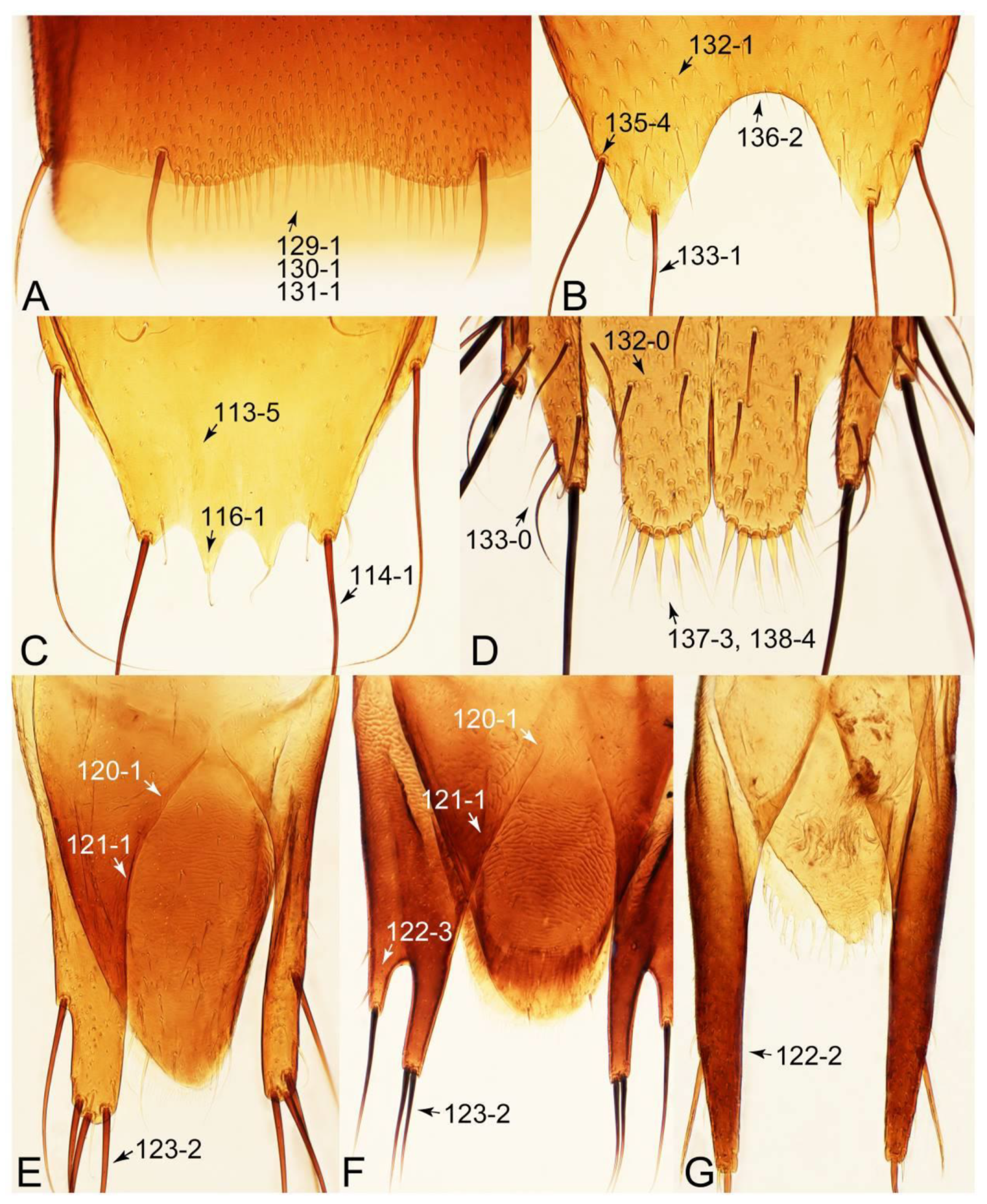
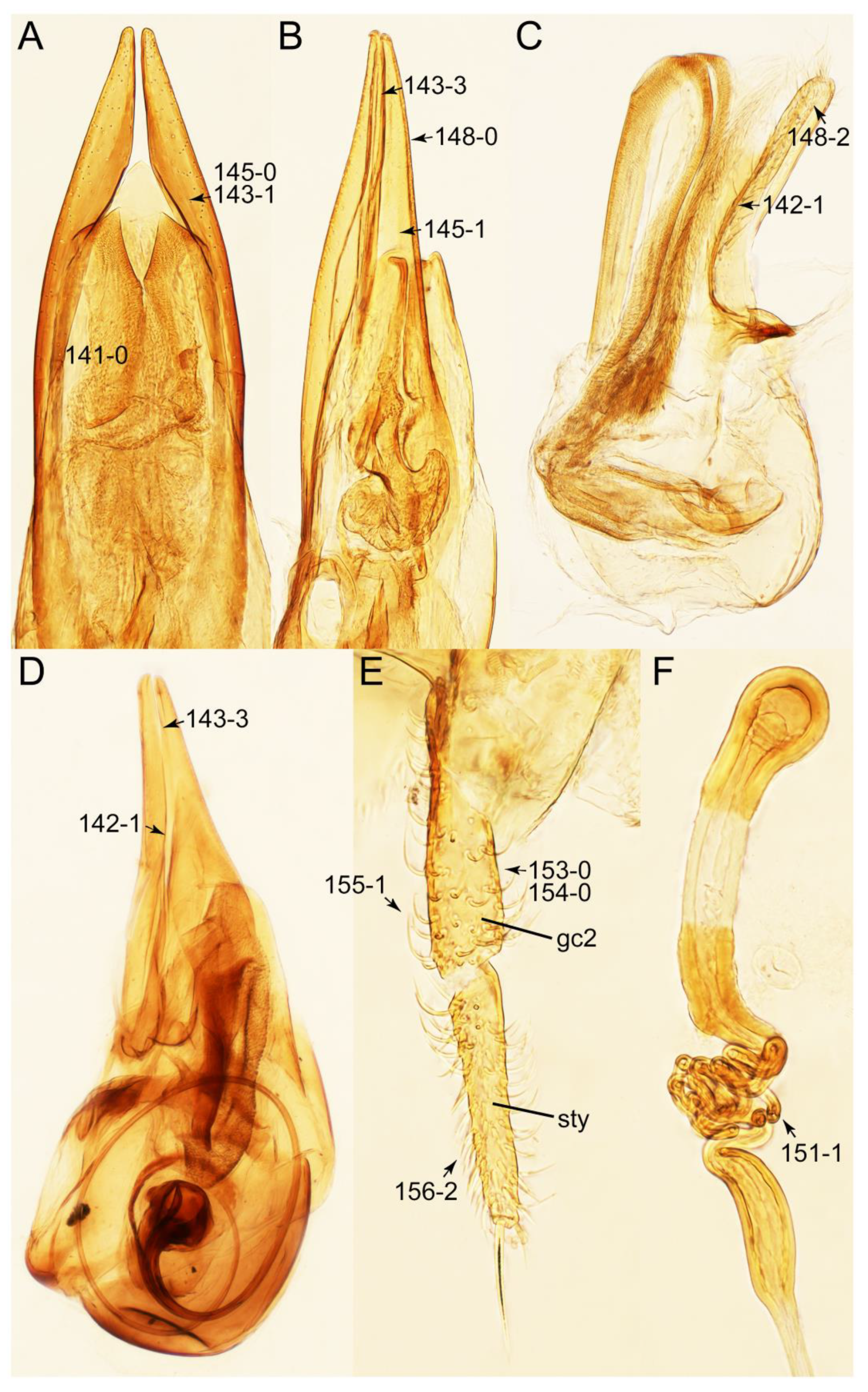


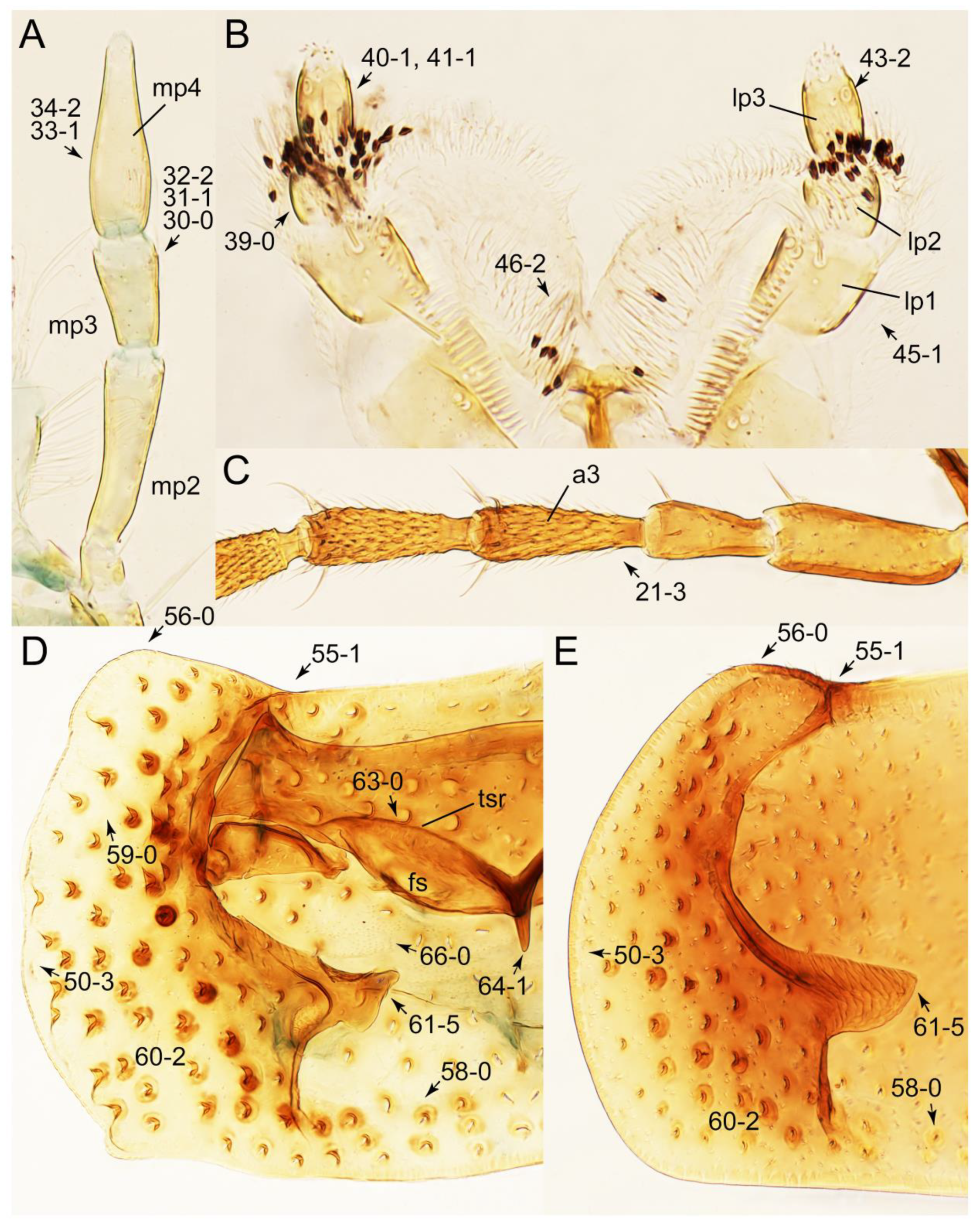

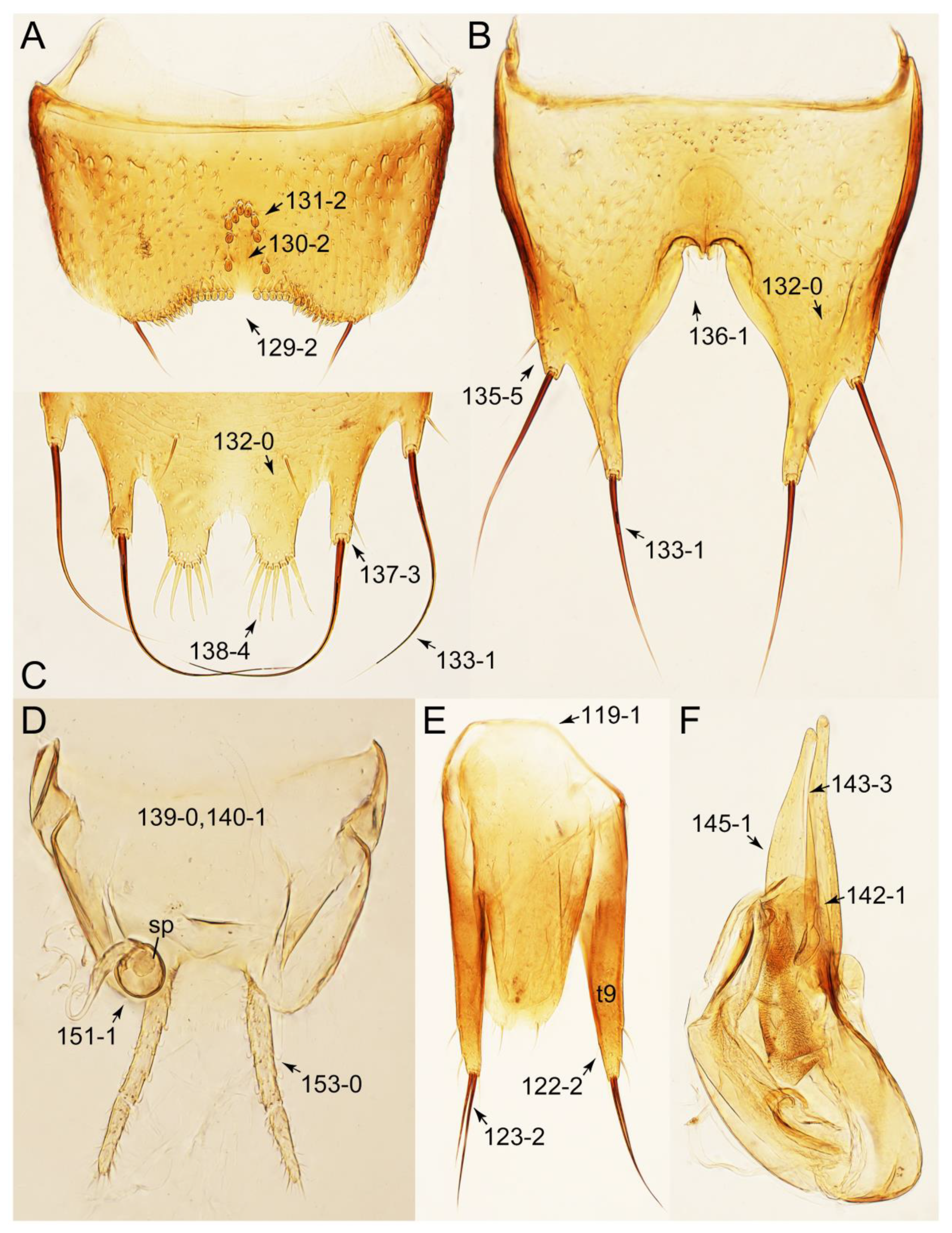



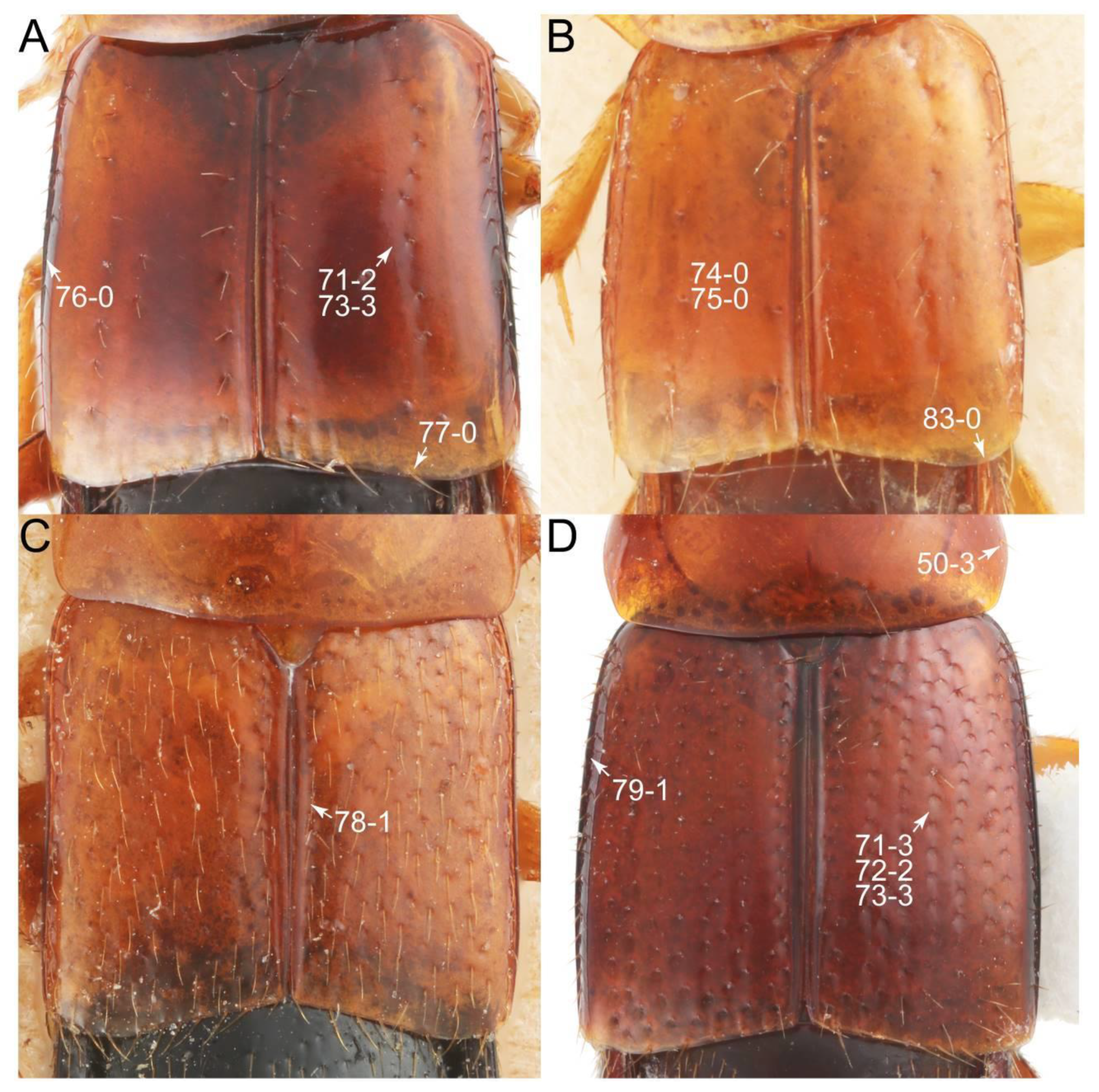

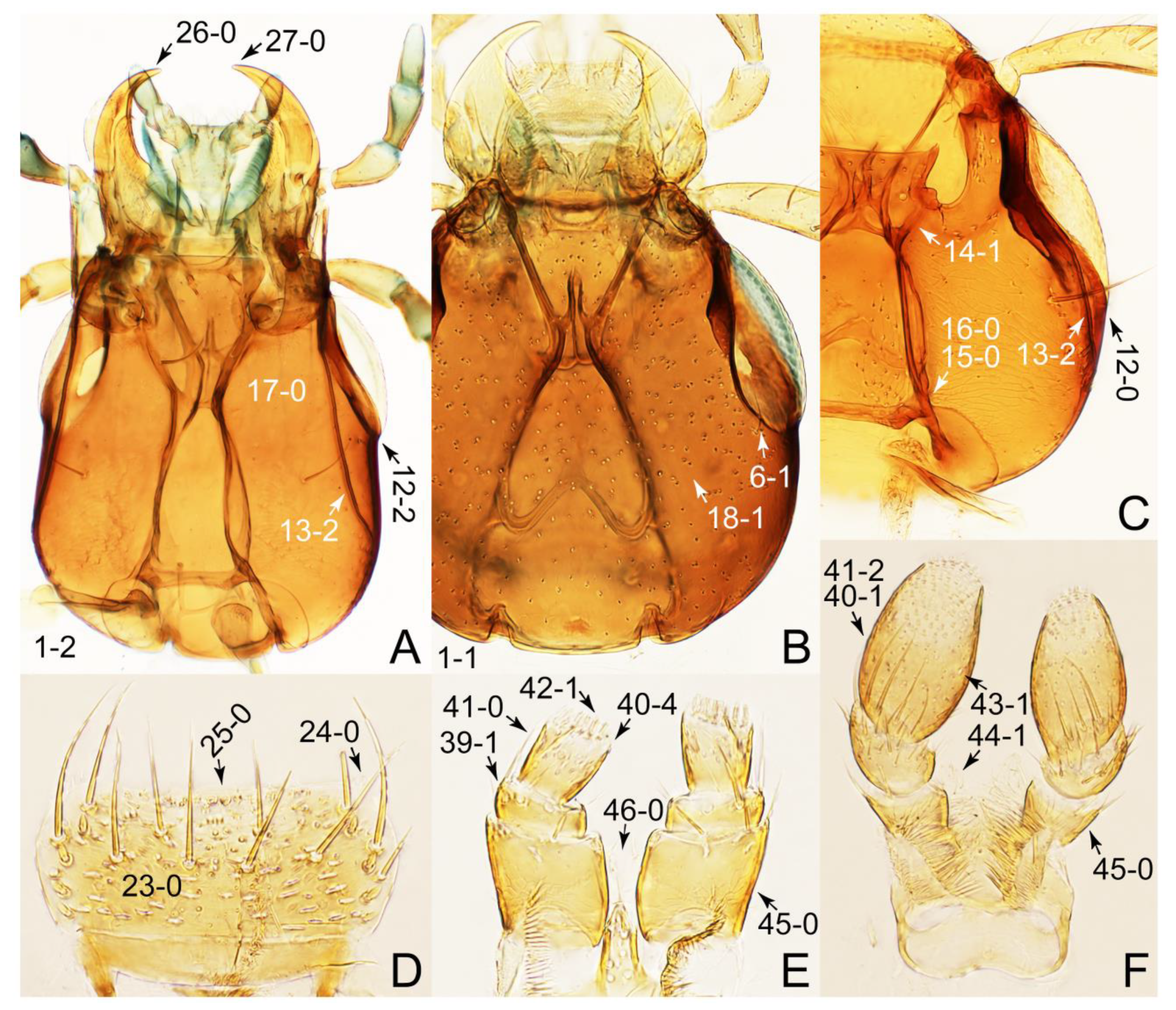
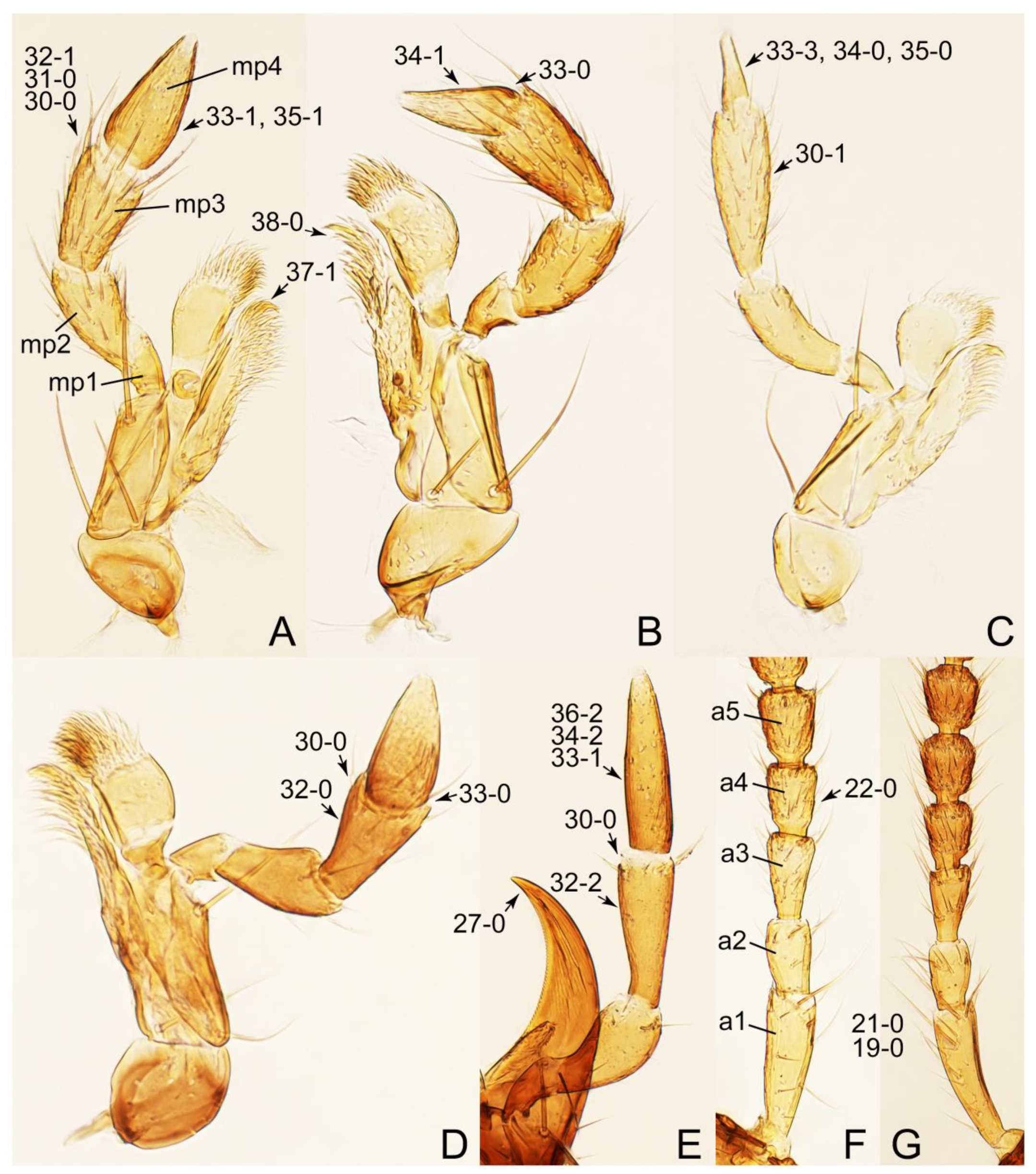
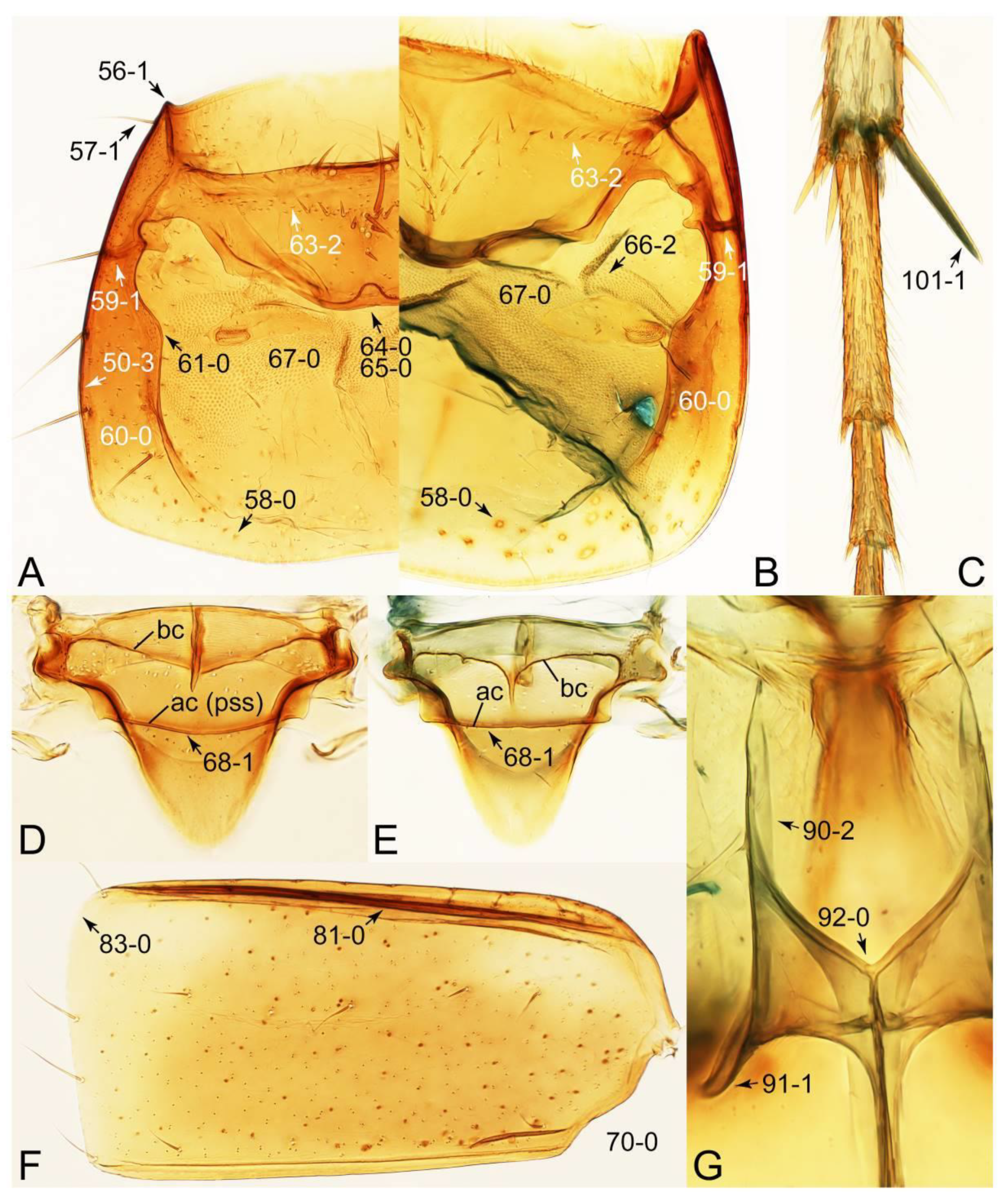
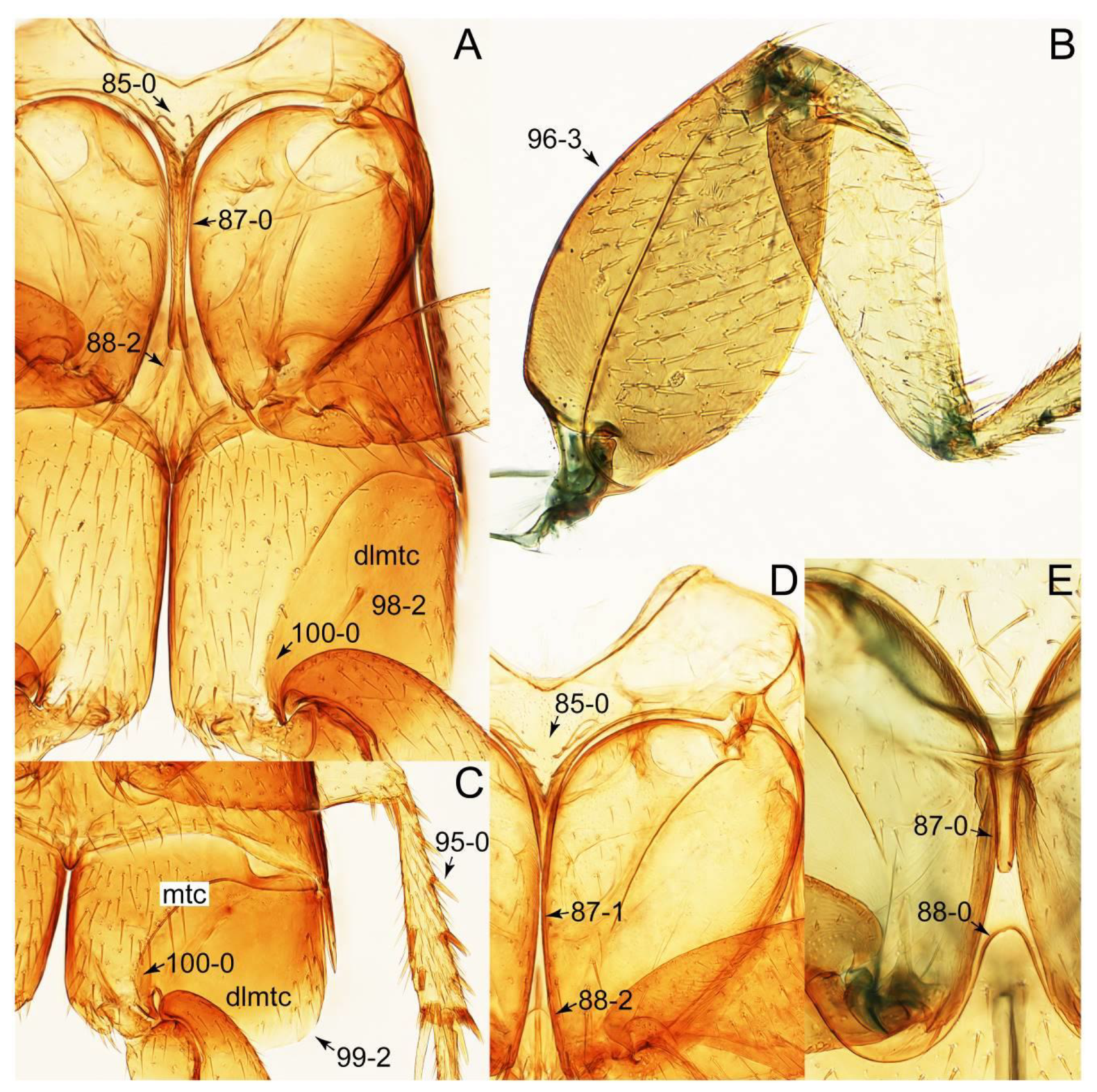
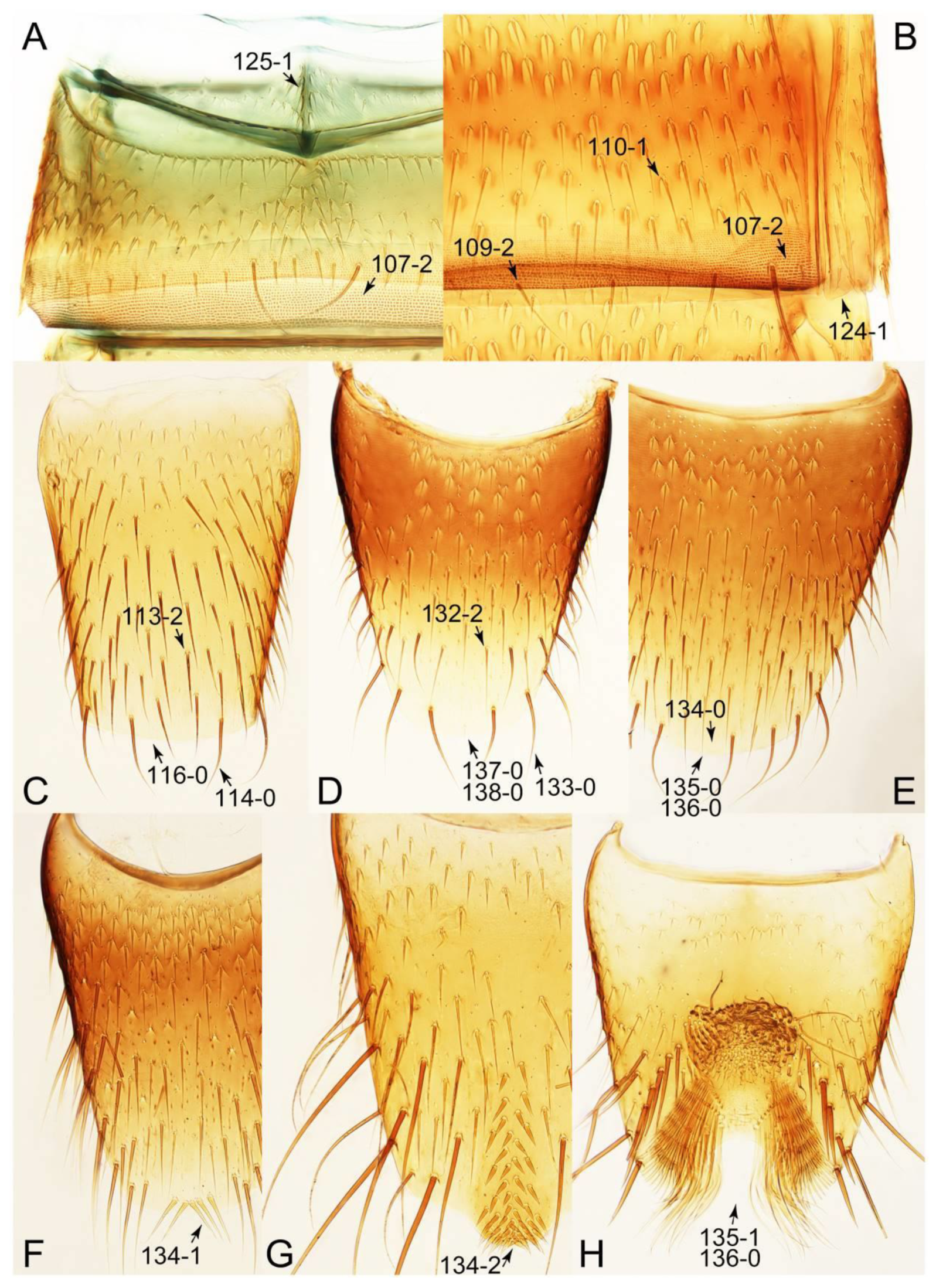

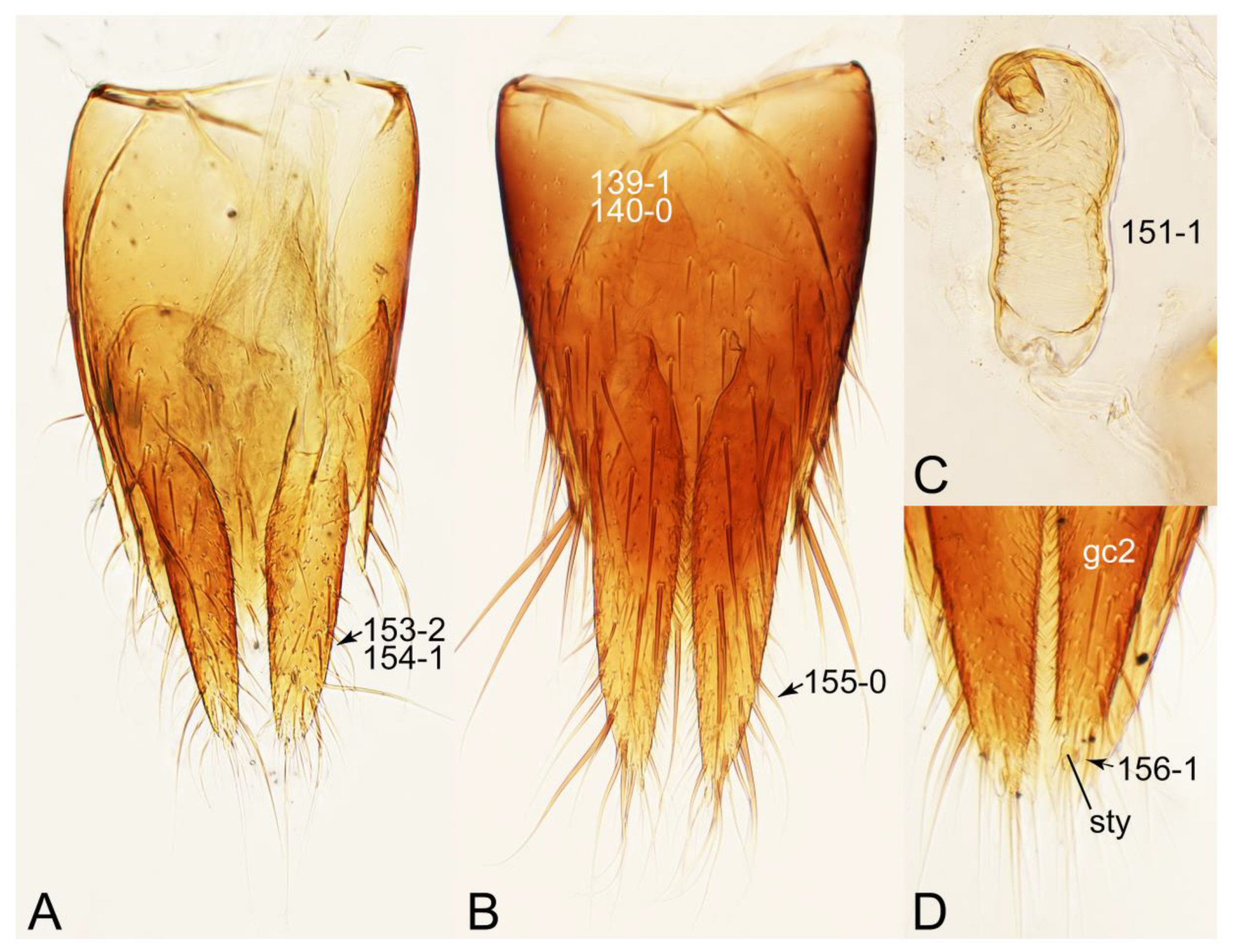
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamamoto, S. Tachyporinae Revisited: Phylogeny, Evolution, and Higher Classification Based on Morphology, with Recognition of a New Rove Beetle Subfamily (Coleoptera: Staphylinidae). Biology 2021, 10, 323. https://doi.org/10.3390/biology10040323
Yamamoto S. Tachyporinae Revisited: Phylogeny, Evolution, and Higher Classification Based on Morphology, with Recognition of a New Rove Beetle Subfamily (Coleoptera: Staphylinidae). Biology. 2021; 10(4):323. https://doi.org/10.3390/biology10040323
Chicago/Turabian StyleYamamoto, Shûhei. 2021. "Tachyporinae Revisited: Phylogeny, Evolution, and Higher Classification Based on Morphology, with Recognition of a New Rove Beetle Subfamily (Coleoptera: Staphylinidae)" Biology 10, no. 4: 323. https://doi.org/10.3390/biology10040323
APA StyleYamamoto, S. (2021). Tachyporinae Revisited: Phylogeny, Evolution, and Higher Classification Based on Morphology, with Recognition of a New Rove Beetle Subfamily (Coleoptera: Staphylinidae). Biology, 10(4), 323. https://doi.org/10.3390/biology10040323






