MaNmrA, a Negative Transcription Regulator in Nitrogen Catabolite Repression Pathway, Contributes to Nutrient Utilization, Stress Resistance, and Virulence in Entomopathogenic Fungus Metarhizium acridum
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
2.2. Bioinformatics Analysis
2.3. Creation of MaNmrA Mutants
2.4. Growth Characteristic Assays
2.5. Stress Tolerance Analysis
2.6. Virulence Assays
2.7. qRT-PCR Analysis
2.8. Data Analysis
3. Results
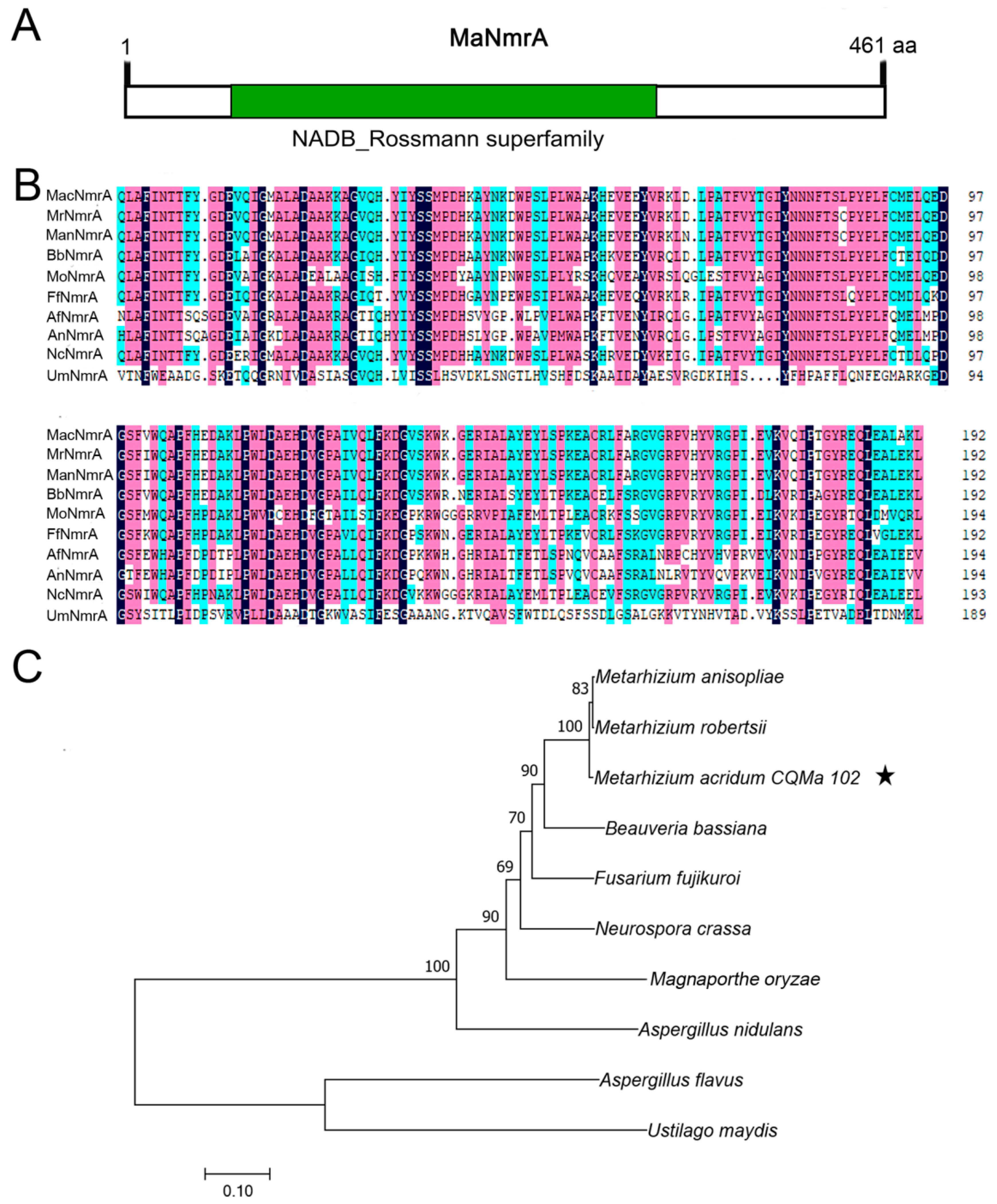
3.1. Identification and Sequence Features of NmrA Ortholog in M. acridum
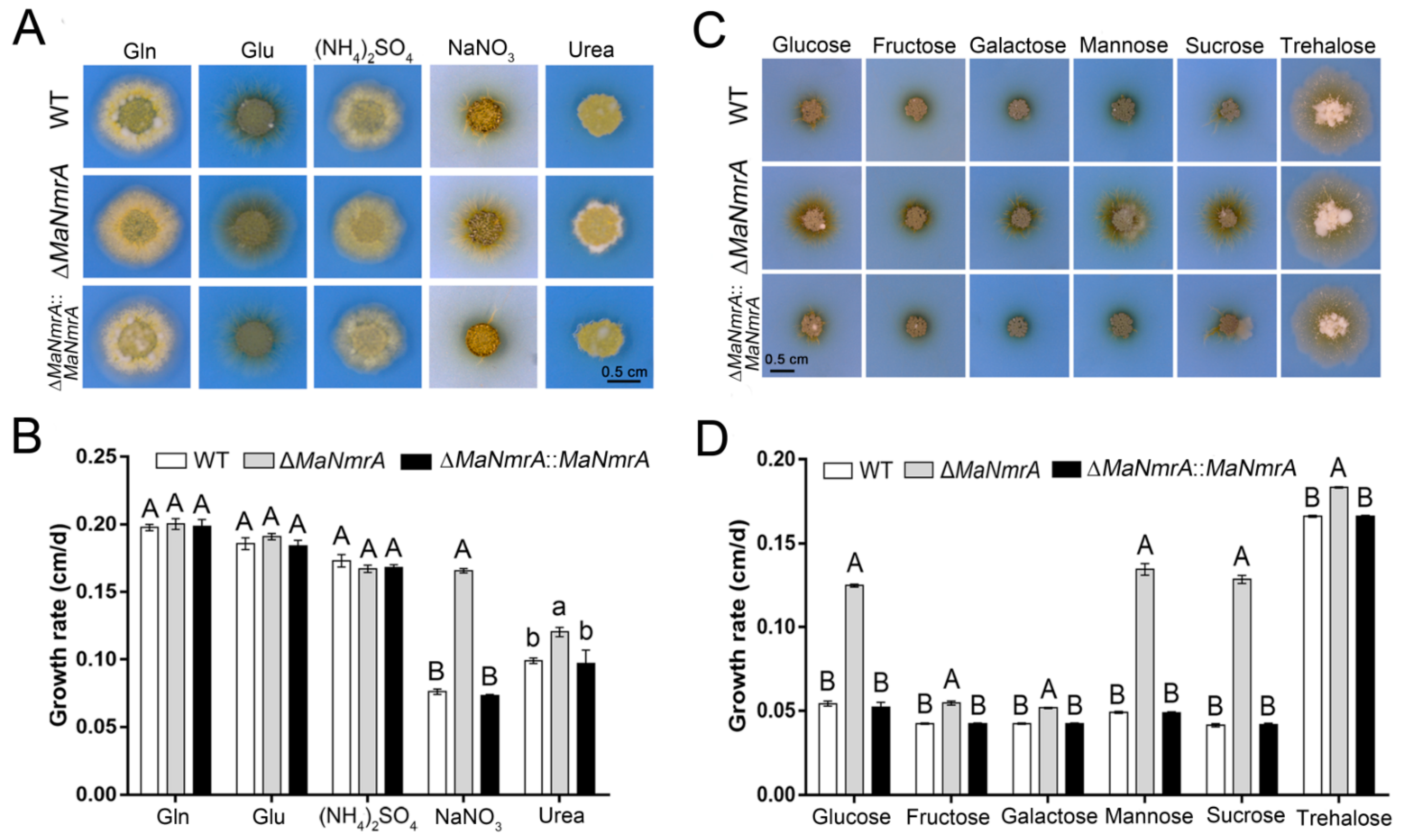
3.2. Deletion of MaNmrA Affected the Nitrogen and Carbon Utilization
3.3. Disruption of MaNmrA Affected Conidial Germination and Conidial Yield
3.4. Disruption of MaNmrA Affected the Fungal Stress Tolerances
3.5. Deletion of MaNmrA Decreased Virulence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hyde, K.D.; Xu, J.; Rapior, S.; Jeewon, R.; Lumyong, S.; Niego, A.G.T.; Abeywickrama, P.D.; Aluthmuhandiram, J.V.S.; Brahamanage, R.S.; Brooks, S.; et al. The amazing potential of fungi: 50 ways we can exploit fungi industrially. Fungal Divers. 2019, 97, 1–136. [Google Scholar] [CrossRef] [Green Version]
- Faria, M.; Wraight, S.P. Mycoinsecticides and Mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control. 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Goettel, M.S.; St Leger, R.J.; Rizzo, N.W.; Staples, R.C.; Roberts, D.W. Ultrastructural localization of a cuticledegrading protease produced by the entomopathogenic fungus Metarhizium anisopliae during penetration of host (Manduca sexta) cuticle. Microbiology 1989, 135, 2233–2239. [Google Scholar] [CrossRef] [Green Version]
- Vega, F.E.; Meyling, N.V.; Luangsa-ard, J.J.; Blackwell, M. Fungal entomopathogens. In Insect Pathology, 2nd ed.; Vega, F., Kaya, H.K., Eds.; Academic Press: San Diego, CA, USA, 2012; pp. 171–220. [Google Scholar]
- Brivio, M.F.; Mastore, M. When appearance misleads: The role of the entomopathogen surface in the relationship with its host. Insects 2020, 11, 387. [Google Scholar] [CrossRef] [PubMed]
- Pinos, D.; Andrés-Garrido, A.; Ferré, J.; Hernández-Martínez, P. Response mechanisms of invertebrates to Bacillus thuringiensis and its pesticidal proteins. Microbiol. Mol. Biol. Rev. 2021, 85, e00007-20. [Google Scholar] [CrossRef]
- Tudzynski, B. Nitrogen regulation of fungal secondary metabolism in fungi. Front. Microbiol. 2014, 5, 656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caddick, M.X. Nitrogen metabolite repression. Prog. Ind. Microbiol. 1994, 29, 323–353. [Google Scholar]
- Marzluf, G.A. Genetic regulation of nitrogen metabolism in fungi. Mierobiol. Mol. Biol. Rev. 1997, 61, 17–32. [Google Scholar] [CrossRef]
- Wong, K.H.; Hynes, M.J.; Todd, R.B.; Davis, M.A. Deletion and overexpression of the Aspergillus nidulans GATA factor AreB reveals unexpected pleiotropy. Microbiology 2009, 155, 3868–3880. [Google Scholar] [CrossRef] [Green Version]
- Andrianopoulos, A.; Kourambas, S.; Sharp, J.A.; Davis, M.A.; Hynes, M.J. Characterization of the Aspergillus nidulans nmrA gene involved in nitrogen metabolite repression. J. Bact. 1998, 180, 1973–1977. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.A.; Arst, H.N. Mutational analysis of AREA, a transcriptional activator mediating nitrogen metabolite repression in Aspergillus nidulans and a member of the ‘‘streetwise’’ GATA family of transcription factors. Microbiol. Mol. Biol. Rev. 1998, 62, 586–596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiemann, P.; Tudzynski, B. The nitrogen regulation network and its impact on secondary metabolism and pathogenicity. In Fusarium: Genomics, Molecular and Cellular Biology; Brown, D.W., Proctor, R.H., Eds.; Caister Academic Press: Norwich, UK, 2013; pp. 111–146. [Google Scholar]
- Narendja, F.; Goller, S.P.; Wolschuk, M.; Strauss, J. Nitrate and the GATA factor AreA are necessary for in vivo binding of NirA, the pathway-specific transcriptional activator of Aspergillus nidulans. Mol. Microbiol. 2002, 44, 573–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conlon, H.; Zadra, I.; Haas, H.; Arst, H.N.; Jr Jones, M.G.; Caddick, M.X. The Aspergillus nidulans GATA transcription factor gene AreB encodes at least three proteins and features three classes of mutation. Mol. Microbiol. 2001, 40, 361–375. [Google Scholar] [CrossRef]
- Michielse, C.B.; Pfannmuller, A.; Macios, M.; Rengers, P.; Dzikowska, A.; Tudzynski, B. The interplay between the GATA transcription factors AreA, the global nitrogen regulator and AreB in Fusarium fujikuroi. Mol. Microbiol. 2014, 91, 472–493. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.J.; Shi, Y. Metabolic enzymes and coenzymes in transcription—A direct link between metabolism and transcription? Trends. Genet. 2004, 20, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, M.T.; Chaudhry, R. Molecular modeling and in silico characterization of nitrogen metabolite repressor NmrA of opportunistic human pathogen Aspergillus fumigatus. J. Microbiol. Biotech. Food. Sci. 2019, 8, 1169–1173. [Google Scholar] [CrossRef]
- Huberman, L.B.; Wu, V.W.; Kowbel, D.J.; Lee, J.; Daum, C.; Grigoriev, I.V.; O’Malley, R.C.; Glass, N.L. DNA affinity purification sequencing and transcriptional profiling reveal new aspects of nitrogen regulation in a filamentous fungus. Proc. Natl. Acad. Sci. USA 2021, 118, e2009501118. [Google Scholar] [CrossRef] [PubMed]
- Schönig, B.; Brown, D.W.; Oeser, B.; Tudzynski, B. Cross-species hybridization with Fusarium verticillioides microarrays reveals new insights into Fusarium fujikuroi nitrogenre gelation and the role of AreA and NMR. Eukaryot. Cell 2008, 7, 1831–1846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, D.; Schmeinck, A.; Mos, M.; Morozov, I.Y.; Caddick, M.X.; Tudzynski, B. The bZIP transcription factor MeaB mediates nitrogen metabolite repression at specific loci. Eukaryot. Cell 2010, 9, 1588–1601. [Google Scholar] [CrossRef] [Green Version]
- Wong, K.H.; Hynes, M.J.; Todd, R.B.; Davis, M.A. Transcriptional control of nmrA by the bZIP transcription factor MeaB reveals a new level of nitrogen regulation in Aspergillus nidulans. Mol. Microbiol. 2007, 66, 534–551. [Google Scholar] [CrossRef]
- Han, X.; Qiu, M.; Wang, B.; Yin, W.B.; Nie, X.Y.; Qin, Q.P.; Ren, S.L.; Yang, K.L.; Zhang, F.; Zhuang, Z.H.; et al. Functional analysis of the nitrogen metabolite repression regulator gene nmrA in Aspergillus flavus. Front. Microbiol. 2016, 7, 1794. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, J.; Wright, J.D.; Hartline, D.; Quispe, C.F.; Madayiputhiya, N.; Wilson, R.A. Principles of carbon catabolite repression in the rice blast fungus: Tps1, Nmr1-3, and a MATE-family pump regulate glucose metabolism during infection. PLoS Genet. 2012, 8, e1002673. [Google Scholar] [CrossRef] [Green Version]
- Macios, M.; Caddick, M.X.; Weglenski, P.; Scazzocchio, C.; Dzikowska, A. The GATA factors AREA and AREB together with the co-repressor NMRA, negatively regulate arginine catabolism in Aspergillus nidulans in response to nitrogen and carbon source. Fungal. Genet. Biol. 2012, 49, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Jin, K.; Xia, Y. Involvement of MaSom1, a downstream transcriptional factor of cAMP/PKA pathway, in conidial yield, stress tolerances, and virulence in Metarhizium acridum. Appl. Microbiol. Biot. 2018, 102, 5611–5623. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cao, Y.Q.; Xia, Y.X. Mmc, a gene involved in microcycle conidiation of the entomopathogenic fungus Metarhizium anisopliae. J. Invertebr. Pathol. 2010, 105, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Z.; Peng, G.X.; Xia, Y.X. Microcycle conidiation and the conidial properties in the entomopathogenic fungus Metarhizium acridum on agar medium. Biocontrol. Sci. Technol. 2010, 20, 809–819. [Google Scholar] [CrossRef]
- Peng, G.X.; Xia, Y.X. The mechanism of themycoinsecticide diluent on the efficacy of the oil formulation of insecticidal fungus. BioControl 2011, 56, 893–902. [Google Scholar] [CrossRef]
- Guo, H.Y.; Wang, H.J.; Keyhani, N.O.; Xia, Y.X.; Peng, G.X. Disruption of an adenylate-forming reductase required for conidiation, increases virulence of the insect pathogenic fungus Metarhizium acridum by enhancing cuticle invasion. Pest. Manag. Sci. 2020, 76, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Wang, C.; Wang, S. Insect pathogenic fungi: Genomics, molecular interactions, and genetic improvements. Annu. Rev. Entomol. 2017, 62, 73–90. [Google Scholar] [CrossRef]
- Becker, A.; Schlöder, P.; Steele, J.E.; Wegener, G. The regulation of trehalose metabolism in insects. Experientia 1996, 52, 433–439. [Google Scholar] [CrossRef]
- Elbein, A.D.; Pan, Y.T.; Pastuszak, I.; Carroll, D. New insights on trehalose: A multifunctional molecule. Glycobiology 2003, 13, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.A.; Gibson, R.P.; Quispe, C.F.; Littlechild, J.A.; Talbot, N.J. An NADPH-dependent genetic switch regulates plant infection by the rice blast fungus. Proc. Natl. Acad. Sci. USA 2010, 107, 21902–21907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Platt, A.; Langdon, T.; Arst, H.N.; Kirk, D.; Tollervey, D.; Sanchez, J.M.M.; Caddick, M.X. Nitrogen metabolite signalling involves the C-terminus and the GATA domain of the Aspergillus transcription factor AREA and the 3′ untranslated region of its mRNA. EMBO J. 1996, 15, 2791–2801. [Google Scholar] [CrossRef]
- Kotaka, M.; Johnson, C.; Lamb, H.K.; Hawkins, A.R.; Ren, J.; Stammers, D.K. Structural analysis of the recognition of the negative regulator NmrA and DNA by the zinc finger from the GATA-type transcription factor AreA. J. Mol. Biol. 2008, 381, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.A.; Heale, J.B.; Hall, R.A. Traits associated with virulence to the aphid Macrosiphoniella sanborni in 18 isolates of Verticillium lecanii. Ann. Appl. Biol. 1985, 106, 39–48. [Google Scholar] [CrossRef]
- Lane, B.S.; Trinci, A.P.J.; Gillespie, A.T. Influence of cultural conditions on virulence of conidia and blastospores of Beauveria bassiana to the green leafhopper, Nephotettix virescens. Mycol. Res. 1991, 95, 829–833. [Google Scholar] [CrossRef]
- Chandler, D.; Heale, J.B.; Gillespie, A.T. Germination of the entomopathogenic fungus Verticillium lecanii on scales of the glasshouse whitefly Trialeurodes vaporariorum. Biocontrol Sci. Technol. 1993, 3, 161–164. [Google Scholar] [CrossRef]
- Altre, J.A.; Vandenberg, J.D.; Cantone, F.A. Pathogenicity of Paecilomyces fumosoroseus isolates to diamondback moth, Plutella xylostella: Correllation with spore size, germination speed, and attachment to cuticle. J. Invertebr. Pathol. 1999, 73, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Urquiza, A.; Luo, Z.; Keyhani, N.O. Improving mycoinsecticides for insect biological control. Appl. Microbiol. Biot. 2015, 99, 1057–1068. [Google Scholar] [CrossRef]
- Lovett, B.; St Leger, R.J. Stress is the rule rather than the exception for Metarhizium. Curr. Genet. 2015, 61, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Chaurasia, A.; Lone, Y.; Wani, O.; Gupta, U.S. Effect of certain entomopathogenic fungi on oxidative stress and mortality of Periplaneta americana. Pestic. Biochem. Phys. 2016, 127, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Chen, X.R.; Liu, Z.J.; Meng, R.Z.; Zhao, X.C.; Liu, Z.H.; Guo, N. Oleuropein protects L-02 cells against H2O2-induced oxidative stress by increasing SOD1, GPx1 and CAT expression. Biomed. Pharmacother. 2017, 85, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Pichersky, E.; Gang, D.R. Genetics and biochemistry of secondary metabolites in plants: An evolutionary perspective. Trends. Plant. Sci. 2000, 5, 439–445. [Google Scholar] [CrossRef]
- Ugine, T.A.; Wraight, S.P.; Brownbridge, M.; Sanderson, J.P. Development of a novel bioassay for estimation of median lethal concentrations (LC50) and doses (LD50) of the entomopathogenic fungus Beauveria bassiana, against western flower thrips, Frankliniella occidentalis. J. Invertebr. Pathol. 2005, 89, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Sethiya, P.; Hu, X.; Guo, S.; Chen, Y.; Li, A.; Tan, K.; Wong, K.H. Transcription in fungal conidia before dormancy produces phenotypically variable conidia that maximize survival in different environments. Nat. Microbiol. 2021, 6, 1066–1081. [Google Scholar] [CrossRef] [PubMed]
- Vega, F.E.; Jackson, M.A.; Mercadier, G.; Poprawski, T.J. The impact of nutrition on spore yields for various fungal entomopathogens in liquid culture. World J. Microb. Biot. 2003, 19, 363–368. [Google Scholar] [CrossRef]
- St Leger, R.J.; Butt, T.M.; Goettel, M.S.; Staples, R.C.; Roberts, D.W. Production in vitro of appressoria by the entomopathogenic fungus Metarhizium anisopliae. Exp. Mycol. 1989, 13, 274–288. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.; Zhang, Q.; Xia, Y.; Jin, K. MaNmrA, a Negative Transcription Regulator in Nitrogen Catabolite Repression Pathway, Contributes to Nutrient Utilization, Stress Resistance, and Virulence in Entomopathogenic Fungus Metarhizium acridum. Biology 2021, 10, 1167. https://doi.org/10.3390/biology10111167
Li C, Zhang Q, Xia Y, Jin K. MaNmrA, a Negative Transcription Regulator in Nitrogen Catabolite Repression Pathway, Contributes to Nutrient Utilization, Stress Resistance, and Virulence in Entomopathogenic Fungus Metarhizium acridum. Biology. 2021; 10(11):1167. https://doi.org/10.3390/biology10111167
Chicago/Turabian StyleLi, Chaochuang, Qipei Zhang, Yuxian Xia, and Kai Jin. 2021. "MaNmrA, a Negative Transcription Regulator in Nitrogen Catabolite Repression Pathway, Contributes to Nutrient Utilization, Stress Resistance, and Virulence in Entomopathogenic Fungus Metarhizium acridum" Biology 10, no. 11: 1167. https://doi.org/10.3390/biology10111167
APA StyleLi, C., Zhang, Q., Xia, Y., & Jin, K. (2021). MaNmrA, a Negative Transcription Regulator in Nitrogen Catabolite Repression Pathway, Contributes to Nutrient Utilization, Stress Resistance, and Virulence in Entomopathogenic Fungus Metarhizium acridum. Biology, 10(11), 1167. https://doi.org/10.3390/biology10111167





