Molecular Characterization of Blastocystis from Animals and Their Caregivers at the Gdańsk Zoo (Poland) and the Assessment of Zoonotic Transmission
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. DNA Extraction and Amplification
2.3. Nucleotide Sequencing and Phylogenetic Analysis
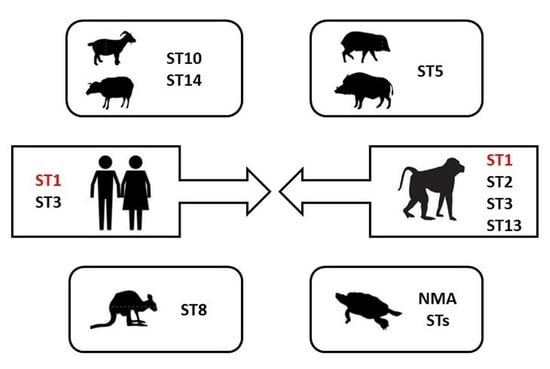
3. Results and Discussion
3.1. Infection Rate of Blastocystis
3.2. Detected Subtypes of Blastocystis
3.3. Genetic Diversity of Detected Blastocystis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stensvold, C.R.; Clark, C.G. Current status of Blastocystis: A personal view. Parasitol. Int. 2016, 65, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.; Stark, D.; Harkness, J.; Ellis, J. Update on the pathogenic potential and treatment options for Blastocystis sp. Gut Pathog. 2014, 6, 17–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahrami, F.; Babaei, E.; Badirzadeh, A.; Riabi, T.R.; Abdoli, A. Blastocystis, urticaria, and skin disorders: Review of the current evidences. Eur. J. Clin. Microbiol. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Scanlan, P.D.; Stensvold, C.R.; Rajilić-Stojanović, M.; Heilig, H.G.H.J.; De Vos, W.M.; O’Toole, P.W.; Cotter, P.D. The microbial eukaryote Blastocystis is a prevalent and diverse member of the healthy human gut microbiota. FEMS Microbiol. Ecol. 2014, 90, 326–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Audebert, C.; Even, G.; Cian, A.; Loywick, A.; Merlin, S.; Viscogliosi, E.; Chabe, M. Colonization with the enteric protozoa Blastocystis is associated with increased diversity of human gut bacterial microbiota. Sci. Rep. 2016, 6, 25255. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.O.B.; Stensvold, C.R. Blastocystis in Health and Disease: Are We Moving from a Clinical to a Public Health Perspective? J. Clin. Microbiol. 2016, 54, 524–528. [Google Scholar] [CrossRef] [Green Version]
- Laforest-Lapointe, I.; Arrieta, M.-C. Microbial Eukaryotes: A Missing Link in Gut Microbiome Studies. mSystems 2018, 3, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Clark, C.G.; van der Giezen, M.; Alfellani, M.A.; Stensvold, C.R. Recent developments in Blastocystis research. Adv. Parasitol. 2013, 82, 1–32. [Google Scholar] [CrossRef] [Green Version]
- Stensvold, C.R.; Clark, C.G. Pre-empting Pandora’s Box: Blastocystis Subtypes Revisited. Trends Parasitol. 2020, 36, 229–232. [Google Scholar] [CrossRef]
- Maloney, J.G.; Santin, M. Mind the gap: New full-length sequences of Blastocystis subtypes generated via oxford nanopore minion sequencing allow for comparisons between full-length and partial sequences of the small subunit of the ribosomal rna gene. Microorganisms 2021, 9, 997. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Abe, N.; Wu, Z. PCR-based identification of zoonotic isolates of Blastocystis from mammals and birds. Microbiology 2004, 150, 1147–1151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stensvold, C.R.; Alfellani, M.A.; Nørskov-Lauritsen, S.; Prip, K.; Victory, E.L.; Maddox, C.; Nielsen, H.V.; Clark, C.G. Subtype distribution of Blastocystis isolates from synanthropic and zoo animals and identification of a new subtype. Int. J. Parasitol. 2009, 39, 473–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alfellani, M.A.; Taner-Mulla, D.; Jacob, A.S.; Imeede, C.A.; Yoshikawa, H.; Stensvold, C.R.; Clark, C.G. Genetic Diversity of Blastocystis in Livestock and Zoo Animals. Protist 2013, 164, 497–509. [Google Scholar] [CrossRef] [Green Version]
- Roberts, T.; Stark, D.; Harkness, J.; Ellis, J. Subtype distribution of Blastocystis isolates from a variety of animals from New South Wales, Australia. Vet. Parasitol. 2013, 196, 85–89. [Google Scholar] [CrossRef]
- Cian, A.; El Safadi, D.; Osman, M.; Moriniere, R.; Gantois, N.; Benamrouz-Vanneste, S.; Delgado-Viscogliosi, P.; Guyot, K.; Li, L.L.; Monchy, S.; et al. Molecular epidemiology of Blastocystis sp. in various animal groups from two French zoos and evaluation of potential zoonotic risk. PLoS ONE 2017, 12, e0169659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Betts, E.L.; Gentekaki, E.; Thomasz, A.; Breakell, V.; Carpenter, A.I.; Tsaousis, A.D. Genetic diversity of Blastocystis in non- primate animals. Parasitology 2017, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Greige, S.; El Safadi, D.; Bécu, N.; Gantois, N.; Pereira, B.; Chabé, M.; Benamrouz-Vanneste, S.; Certad, G.; El Hage, R.; Chemaly, M.; et al. Prevalence and subtype distribution of Blastocystis sp. isolates from poultry in Lebanon and evidence of zoonotic potential. Parasit. Vectors 2018, 11, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Weining, Z.; Tao, W.; Gong, B.; Yang, H.; Li, Y.; Song, M.; Lu, Y.; Li, W. First report of Blastocystis infections in cattle in China. Vet. Parasitol. 2017, 246, 38–42. [Google Scholar] [CrossRef]
- Wang, J.; Gong, B.; Yang, F.; Zhang, W.; Zheng, Y.; Liu, A. Subtype distribution and genetic characterizations of Blastocystis in pigs, cattle, sheep and goats in northeastern China’s Heilongjiang Province. Infect. Genet. Evolut. 2018, 57, 171–176. [Google Scholar] [CrossRef]
- Wawrzyniak, I.; Poirier, P.; Texier, C.; Delbac, F.; Viscogliosi, E.; Dionigia, M.; Alaoui, H.E. Blastocystis, an unrecognized parasite: An overview of pathogenesis and diagnosis. Ther. Adv. Infect. Dis. 2013, 1, 167–178. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Owen, H.; Traub, R.J.; Cuttell, L.; Inpankaew, T.; Bielefeldt-Ohmann, H. Molecular epidemiology of Blastocystis in pigs and their in-contact humans in Southeast Queensland, Australia, and Cambodia. Vet. Parasitol. 2014, 203, 264–269. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Wu, Z.; Pandey, K.; Pandey, B.D.; Sherchand, J.B.; Yanagi, T.; Kanbara, H. Molecular characterization of Blastocystis isolates from children and rhesus monkeys in Kathmandu, Nepal. Vet. Parasitol. 2009, 160, 295–300. [Google Scholar] [CrossRef]
- Ruaux, C.G.; Stang, B.V. Prevalence of Blastocystis in shelter-resident and client-owned companion animals in the US Pacific Northwest. PLoS ONE 2014, 9, e0107496. [Google Scholar] [CrossRef]
- Pakandl, M.; Koudela, B.; Vitovec, J. An experimental infection of conventional and gnotobiotic piglets with human and porcine strains of Blastocystis. Folia Parasitol 1993, 40, 319–320. [Google Scholar]
- Iguchi, A.; Ebisu, A.; Nagata, S.; Saitou, Y.; Yoshikawa, H.; Iwatani, S.; Kimata, I. Infectivity of different genotypes of human Blastocystis hominis isolates in chickens and rats. Parasitol. Int. 2007, 56, 107–112. [Google Scholar] [CrossRef]
- Scicluna, S.M.; Tawari, B.; Clark, C.G. DNA barcoding of Blastocystis. Protist 2006, 157, 77–85. [Google Scholar] [CrossRef]
- Rudzińska, M.; Kowalewska, B.; Szostakowska, B.; Grzybek, M.; Sikorska, K.; Świątalska, A. First report on the occurrence and subtypes of Blastocystis in pigs in Poland using sequence-tagged-site pcr and barcode region sequencing. Pathogens 2020, 9, 595. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasegawa, M.; Kishino, H.; Yano, T. Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J. Mo. Evol. 1985, 22, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Posada, D.; Crandall, K.A. A note on chaotic unimodal maps and applications. Bioinformatics 1998, 14, 817–818. [Google Scholar] [CrossRef] [Green Version]
- Vega, L.; Herrera, G.; Munoz, M.; Patarroyo, M.A.; Maloney, J.G.; Santin, M.; Ramirez, J.D. Gut microbiota profiles in diarrheic patients with co-occurrence of Clostridioides difficile and Blastocystis. PLoS ONE 2021, 16, 1–23. [Google Scholar] [CrossRef]
- Dogruman-Al, F.; Simsek, Z.; Boorom, K.; Ekici, E.; Sahin, M.; Tuncer, C.; Kustimur, S.; Altinbas, A. Comparison of methods for detection of Blastocystis infection in routinely submitted stool samples, and also in IBS/IBD Patients in Ankara, Turkey. PLoS ONE 2010, 5, e15484. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.; Barratt, J.; Harkness, J.; Ellis, J.; Stark, D. Comparison of microscopy, culture, and conventional polymerase chain reaction for detection of Blastocystis sp. in clinical stool samples. Am. J. Trop. Med. Hyg. 2011, 84, 308–312. [Google Scholar] [CrossRef]
- Solaymani-Mohammadi, S.; Rezaian, M.; Hooshyar, H.; Mowlavi, G.R.; Babaei, Z.; Anwar, M.A. Intestinal protozoa in wild boars (Sus scrofa) in western Iran. J. Wildl. Dis. 2004, 40, 801–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yaghoobi, K.; Sarkari, B.; Mansouri, M.; Motazedian, M.H. Zoonotic intestinal protozoan of the wild boars, Sus scrofa, in Persian Gulf’s coastal area (Bushehr province), Southwestern Iran. Vet. World 2016, 9, 1047–1050. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.; Seo, M.-G.; Oem, J.-K.; Kim, Y.-S.; Lee, S.-Y.; Kim, J.; Jeong, H.; Jheong, W.-H.; Kim, Y.; Lee, W.-J.; et al. Molecular Detection and Subtyping of Blastocystis Detected in Wild Boars (Sus scrofa) in South Korea. J. Wildl. Dis. 2020, 56, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Russini, V.; Di Filippo, M.M.; Fanelli, R.; Polidori, M.; Berrilli, F.; Di Cave, D.; Novelletto, A.; Calderini, P. Characterization of prevalence and genetic subtypes of Blastocystis sp. in wild and domestic Suidae of central Italy aided by amplicon NGS. Vet. Parasitol. Reg. Stud. Rep. 2020, 22, 100472. [Google Scholar] [CrossRef] [PubMed]
- Valença-Barbosa, C.; Do Bomfim, T.C.B.; Teixeira, B.R.; Gentile, R.; Da Costa Neto, S.F.; Magalhães, B.S.N.; De Almeida Balthazar, D.; Da Silva, F.A.; Biot, R.; D’Avila Levy, C.M.; et al. Molecular epidemiology of Blastocystis isolated from animals in the state of Rio de Janeiro, Brazil. PLoS ONE 2019, 14, e0210740. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Su, S.; Ye, J.; Lai, X.; Lai, R.; Liao, H.; Chen, G.; Zhang, R.; Hou, Z.; Luo, X. Blastocystis sp. subtype 5: A possibly zoonotic genotype. Parasit. Res. 2007, 101, 1527–1532. [Google Scholar] [CrossRef]
- Chandrasekaran, H.; Chandrawathani, P.; Bathmanaban, P. The diagnosis of Blastocystis sp. from animals - an emerging zoonosis. Malays. J. Vet. Res. 2014, 5, 15–22. [Google Scholar]
- Song, J.K.; Hu, R.S.; Fan, X.C.; Wang, S.S.; Zhang, H.J.; Zhao, G.H. Molecular characterization of Blastocystis from pigs in Shaanxi province of China. Acta Trop. 2017, 173, 130–135. [Google Scholar] [CrossRef]
- Moura, R.G.F.; de Oliveira-Silva, M.B.; Pedrosa, A.L.; Nascentes, G.A.N.; Cabrine-Santos, M. Occurrence of Blastocystis spp. In domestic animals in triângulo mineiro area of Brazil. Rev. Soc. Bras. Med. Trop. 2018, 51, 240–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rivero-Juarez, A.; Dashti, A.; López-López, P.; Muadica, A.S.; Risalde, M.D.L.A.; Risalde, M.D.L.A.; Köster, P.C.; Machuca, I.; Bailo, B.; De Mingo, M.H.; et al. Protist enteroparasites in wild boar (Sus scrofa ferus) and black Iberian pig (Sus scrofa domesticus) in southern Spain: A protective effect on hepatitis e acquisition? Parasit. Vectors 2020, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.C.; Tan, P.; Sharma, R.; Sugnaseelan, S.; Suresh, K. Genetic diversity of caprine Blastocystis from Peninsular Malaysia. Parasitol. Res. 2013, 112, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Song, J.K.; Yin, Y.L.; Yuan, Y.J.; Tang, H.; Ren, G.J.; Zhang, H.J.; Li, Z.X.; Zhang, Y.M.; Zhao, G.H. First genotyping of Blastocystis sp. in dairy, meat, and cashmere goats in northwestern China. Acta Trop. 2017, 176, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Li, W.C.; Wang, K.; Gu, Y. Occurrence of Blastocystis sp. and Pentatrichomonas hominis in sheep and goats in China. Parasit. Vectors 2018, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Udonsom, R.; Prasertbun, R.; Mahittikorn, A.; Mori, H.; Changbunjong, T.; Komalamisra, C.; Pintong, A.R.; Sukthana, Y.; Popruk, S. Blastocystis infection and subtype distribution in humans, cattle, goats, and pigs in central and western Thailand. Infect. Genet. Evol. 2018, 65, 107–111. [Google Scholar] [CrossRef]
- Wang, J.; Gong, B.; Liu, X.; Zhao, W.; Bu, T.; Zhang, W.; Liu, A.; Yang, F. Distribution and genetic diversity of Blastocystis subtypes in various mammal and bird species in northeastern China. Parasit. Vectors 2018, 11, 1–7. [Google Scholar] [CrossRef] [Green Version]
- AbuOdeh, R.; Ezzedine, S.; Madkour, M.; Stensvold, C.R.; Samie, A.; Nasrallah, G.; AlAbsi, E.; ElBakri, A. Molecular Subtyping of Blastocystis from Diverse Animals in the United Arab Emirates. Protist 2019, 170, 125679. [Google Scholar] [CrossRef]
- Petrášová, J.; Uzlíková, M.; Kostka, M.; Petrželková, K.J.; Huffman, M.A.; Modrý, D. Diversity and host specificity of Blastocystis in syntopic primates on Rubondo Island, Tanzania. Int. J. Parasitol. 2011, 41, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Helenbrook, W.D.; Shields, W.M.; Whipps, C.M. Characterization of Blastocystis species infection in humans and mantled howler monkeys, Alouatta palliata aequatorialis, living in close proximity to one another. Parasitol. Res. 2015, 114, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Zanzani, S.A.; Gazzonis, A.L.; Epis, S.; Manfredi, M.T. Study of the gastrointestinal parasitic fauna of captive non-human primates (Macaca fascicularis). Parasitol. Res. 2016, 115, 307–312. [Google Scholar] [CrossRef]
- Zhao, G.H.; Hu, X.F.; Liu, T.L.; Hu, R.S.; Yu, Z.Q.; Yang, W.B.; Wu, Y.L.; Yu, S.K.; Song, J.K. Molecular characterization of Blastocystis sp. in captive wild animals in Qinling Mountains. Parasitol. Res. 2017, 116, 2327–2333. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Arbex, A.P.; David, É.B.; Tenório, M.d.S.; Cicchi, P.J.P.; Patti, M.; Coradi, S.T.; Lucheis, S.B.; Jim, J.; Guimarães, S. Diversity of Blastocystis subtypes in wild mammals from a zoo and two conservation units in southeastern Brazil. Infect. Genet. Evol. 2020, 78. [Google Scholar] [CrossRef] [PubMed]
- Parkar, U.; Traub, R.J.; Vitali, S.; Elliot, A.; Levecke, B.; Robertson, I.; Geurden, T.; Steele, J.; Drake, B.; Thompson, R.C.A. Molecular characterization of Blastocystis isolates from zoo animals and their animal-keepers. Vet. Pparasitol. 2010, 169, 8–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Karim, M.R.; Li, D.; Rahaman Sumon, S.M.M.; Siddiki, S.H.M.F.; Rume, F.I.; Sun, R.; Jia, Y.; Zhang, L. Molecular characterization of Blastocystis sp. in captive wildlife in Bangladesh National Zoo: Non-human primates with high prevalence and zoonotic significance. Int. J. Parasitol. Parasit. Wildl. 2019, 10, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Alfellani, M.A.; Jacob, A.S.; Perea, N.O.; Krecek, R.C.; Taner-Mulla, D.; Verveij, J.J.; Levecke, B.; Tannich, E.; Clarc, C.G. Rune Stensvold Diversity and distribution of Blastocystis sp. subtypes in non-human primates. Parasitology 2013, 140, 966–971. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Zhang, Y.; Qiao, H.; Li, S.; Wang, H.; Zhang, N.; Zhang, X. Cockroach as a vector of Blastocystis sp. Is risk for golden monkeys in zoo. Korean J. Parasitol. 2020, 58, 583–587. [Google Scholar] [CrossRef]
- Abe, N.; Wu, Z.; Yoshikawa, H. Molecular characterization of Blastocystis isolates from primates. Vet. Parasitol. 2003, 113, 321–325. [Google Scholar] [CrossRef]
- Deng, L.; Yao, J.; Chen, S.; He, T.; Chai, Y.; Zhou, Z.; Shi, X.; Liu, H.; Zhong, Z.; Fu, H.; et al. First identification and molecular subtyping of Blastocystis sp. in zoo animals in southwestern China. Parasit. Vectors 2021, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wei, Z.; Li, Q.; Lin, Y.; Yang, H.; Li, W. Prevalence and subtype diversity of Blastocystis in human and nonhuman primates in North China. Parasitol. Res. 2020, 119, 2719–2725. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Alfellani, M.; Clark, C.G. Levels of genetic diversity vary dramatically between Blastocystis subtypes. Infect. Genet. Evol. 2012, 12, 263–273. [Google Scholar] [CrossRef] [Green Version]
- Lim, Y.A.L.; Ngui, R.; Shukri, J.; Rohela, M.; Mat Naim, H.R. Intestinal parasites in various animals at a zoo in Malaysia. Vet. Parasitol. 2008, 157, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Calero-Bernal, R.; Santín, M.; Maloney, J.G.; Martín-Pérez, M.; Habela, M.A.; Fernández-García, J.L.; Figueiredo, A.; Nájera, F.; Palacios, M.J.; Mateo, M.; et al. Blastocystis sp. Subtype Diversity in Wild Carnivore Species from Spain. J. Eukaryot. Microbiol. 2019, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Li, X.D.; Zou, Y.; Pan, J.; Liang, Q.L.; Zeng, Z.; Meng, Y.M.; Wang, X.L.; Wang, H.N.; Zhu, X.Q. Prevalence and subtypes of Blastocystis sp. infection in zoo animals in three cities in China. Parasitol. Res. 2020, 119, 465–471. [Google Scholar] [CrossRef]
- Deng, L.; Yao, J.X.; Liu, H.F.; Zhou, Z.Y.; Chai, Y.J.; Wang, W.Y.; Zhong, Z.J.; Deng, J.L.; Ren, Z.H.; Fu, H.L.; et al. First report of Blastocystis in giant pandas, red pandas, and various bird species in Sichuan province, southwestern China. Int. J. Parasitol. Parasit. Wildl. 2019, 9, 298–304. [Google Scholar] [CrossRef]
- Farah Haziqah, M.T.; Chandrawathani, P.; Douadi, B.; Suresh, K.; Wilson, J.J.; Mohd Khalid, M.K.N.; Rajamanikam, A.; Lewis, J.W.; Mohd Zain, S.N. Impact of pH on the viability and morphology of Blastocystis isolates. Trop. Biomed. 2018, 35, 501–510. [Google Scholar]
- Nagel, R.; Cuttell, L.; Stensvold, C.R.; Mills, P.C.; Bielefeldt-Ohmann, H.; Traub, R.J. Blastocystis subtypes in symptomatic and asymptomatic family members and pets and response to therapy. Intern. Med. J. 2012, 42, 1187–1195. [Google Scholar] [CrossRef]
- Belleza, M.L.B.; Reyes, J.C.B.; Tongol-Rivera, P.N.; Rivera, W.L. Subtype analysis of Blastocystis sp. isolates from human and canine hosts in an urban community in the Philippines. Parasitol. Int. 2016, 65, 291–294. [Google Scholar] [CrossRef]
- Liao, S.; Lin, X.; Sun, Y.; Qi, N.; Lv, M.; Wu, C.; Li, J.; Hu, J.; Yu, L.; Cai, H.; et al. Occurrence and genotypes of Cryptosporidium spp., Giardia duodenalis, and Blastocystis sp. in household, shelter, breeding, and pet market dogs in Guangzhou, southern China. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Mohammadpour, I.; Bozorg-Ghalati, F.; Gazzonis, A.L.; Manfredi, M.T.; Motazedian, M.H.; Mohammadpour, N. First molecular subtyping and phylogeny of Blastocystis sp. isolated from domestic and synanthropic animals (dogs, cats and brown rats) in southern Iran. Parasit. Vectors 2020, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, J.D.; Sánchez, L.V.; Bautista, D.C.; Corredor, A.F.; Flórez, A.C.; Stensvold, C.R. Blastocystis subtypes detected in humans and animals from Colombia. Infect. Genet. Evol. 2014, 22, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Asghari, A.; Sadrae, I.J.; Pirestani, M.M.I. First molecular identification and subtype distribution of Blastocystis sp. isolated from hooded crows (Corvus cornix) and pigeons (Columba livia) in Tehran Province, Iran. Comp. Immunol. Microbiol. Infect. Dis. 2019, 62, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Maloney, J.G.; Molokin, A.; da Cunha, M.J.R.; Cury, M.C.; Santin, M. Blastocystis subtype distribution in domestic and captive wild bird species from Brazil using next generation amplicon sequencing. Parasit. Epidemiol. Control. 2020, 9, e00138. [Google Scholar] [CrossRef]
- Hublin, J.S.Y.; Maloney, J.G.; Santin, M. Blastocystis in domesticated and wild mammals and birds. Res. Vet. Sci. 2021, 135, 260–282. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Stensvold, C.R.; Vidal-Lapiedra, A.; Onuoha, E.S.U.; Fagbenro-Beyioku, A.F.; Clark, C.G. Variable geographic distribution of Blastocystis subtypes and its potential implications. Acta Trop. 2013, 126, 11–18. [Google Scholar] [CrossRef] [Green Version]
- Tanizaki, A.; Yoshikawa, H.; Iwatani, S.; Kimata, I. Infectivity of Blastocystis isolates from chickens, quails and geese in chickens. Parasitol. Res. 2005, 96, 57–61. [Google Scholar] [CrossRef]
- Teow, W.L.; Ng, G.C.; Chan, P.P.; Chan, Y.C.; Yap, E.H.; Zaman, V.; Singh, M. A survey of Blastocystis in reptiles. Parasitol. Res. 1992, 78, 453–455. [Google Scholar] [CrossRef]
- Noël, C.; Dufernez, F.; Gerbod, D.; Edgcomb, V.P.; Delgado-Viscogliosi, P.; Ho, L.C.; Singh, M.; Wintjens, R.; Sogin, M.L.; Capron, M.; et al. Molecular phylogenies of Blastocystis isolates from different hosts: Implications for genetic diversity, identification of species, and zoonosis. J. Clin. Microbiol. 2005, 43, 348–355. [Google Scholar] [CrossRef] [Green Version]
- Yoshikawa, H.; Koyama, Y.; Tsuchiya, E.; Takami, K. Blastocystis phylogeny among various isolates from humans to insects. Parasitol. Int. 2016, 65, 750–759. [Google Scholar] [CrossRef]
- Lintner, M.; Weissenbacher, A.; Heiss, E. The Oropharyngeal Morphology in the Semiaquatic Giant Asian Pond Turtle, Heosemys grandis, and Its Evolutionary Implications. PLoS ONE 2012, 7, e46344. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Host Name | Scientific Name | No. Examined | No. of Positives | % Positives (±CL95) |
|---|---|---|---|---|
| Human | Homo sapiens | 35 | 6 | 17.14 (8.2–31.3) |
| NHPs total | 39 | 23 | 58.97 (43.0–73.7) | |
| Rhesus macaque | Macaca mulatta | 1 | 1 | |
| Bornean orangutan | Pongo pygmaeus | 2 | 0 | |
| Black howler | Alouatta caraya | 4 | 0 | |
| Chimpanzee | Pan troglodytes | 5 | 3 | |
| Patas monkey | Erythrocebus patas | 5 | 4 | |
| Mandrill | Mandrillus sphinx | 8 | 4 | |
| Javan lutung | Trachypithecus auratus | 5 | 5 | |
| Guereza | Colobus guereza kikuyuensis | 4 | 4 | |
| Buff-cheeked gibbon | Nomascus gabriellae | 1 | 1 | |
| White-cheeked gibbon | Nomascus leucogenys | 1 | 1 | |
| Emperor tamarin | Saguinus imperator subgrisescens | 3 | 0 | |
| Carnivora total | 20 | 0 | ||
| Canidae | ||||
| European grey wolf | Canis lupus | 2 | 0 | |
| Maned wolf | Chrysocyon brachyurus | 2 | 0 | |
| Fennec fox | Vulpes zerda | 1 | 0 | |
| Ursidae | ||||
| Brown bear | Ursus arctos | 1 | 0 | |
| Felidae | ||||
| African lion | Panthera leo bleyenberghi | 6 | 0 | |
| Amur tiger | Panthera tigris altaica | 2 | 0 | |
| Persian leopard | Panthera pardus saxicolor | 2 | 0 | |
| Serval | Leptailurus serval | 2 | 0 | |
| Procyonidae | ||||
| Brown-nosed coati | Nasua nasua | 2 | 0 | |
| Artiodactyla total | 77 | 25 | 32.46 (22.1–44.6) | |
| Suidae | ||||
| Wild boar | Sus scrofa | 10 | 8 | |
| Tayassuidae | ||||
| Collared peccary | Pecari tajacu | 10 | 9 | |
| Hippopotamidae | ||||
| Pigmy hippopotamus | Choeropsis liberiensis | 2 | 0 | |
| Hippopotamus | Hippopotamus amphibius | 1 | 0 | |
| Bovidae | 23 | 8 | 34.78 (17.8–56.7) | |
| European wisent | Bison bonasus bonasus | 5 | 0 | |
| Yak | Bos grunniens | 5 | 0 | |
| Mishmi takin | Budorcas taxicolor | 3 | 0 * | |
| Domestic goat | Capra hircus | 8 | 7 | |
| Polish heath sheep | Ovis aries polish_heath | 2 | 1 | |
| Camelidae total | 29 | 0 | ||
| Alpaca | Vicugna pacos | 8 | 0 | |
| Llama | Lama glama | 12 | 0 | |
| Dromedary | Camelus dromedarius | 5 | 0 | |
| Bactrian camel | Camelus bactrianus | 4 | 0 | |
| Perissodactyla | ||||
| South American tapir | Tapirus terrestris | 2 | 0 | |
| Metatheria total | 15 | 1 | 6.66 (0,4–30.2) | |
| Red-necked wallaby | Macropus rufogriseus | 10 | 0 | |
| Red kangaroo | Macropus rufus | 5 | 1 | |
| Leporidae European rabbit Rodentia | Oryctolagus cuniculus | 3 | 0 | |
| Capybara | Hydrochoerus hydrochaeris | 6 | 0 | |
| Common gundi | Ctenodactylus gundi | 1 | 0 | |
| Aves total | 25 | 0 | ||
| Eurasian eagle owl | Bubo bubo | 1 | 0 | |
| Great hornet owl | Bubo virginianus | 1 | 0 | |
| Greater rhea | Rhea Americana | 5 | 0 * | |
| Violet turaco | Musophaga violacea | 1 | 0 | |
| Salmon-crested cockatoo | Cacatua moluccensis | 1 | 0 | |
| Military macaw | Ara militaris mexicana | 2 | 0 | |
| Yellow-crowned amazon | Amazona ochrocephala ochrocephala | 1 | 0 | |
| Greater flamingo | Phoenicopterus roseus | 5 | 0 | |
| Great curassow | Crax rubra rubra | 2 | 0 | |
| Southern ground hornbill | Bucorvus leadbeateri | 3 | 0 | |
| Andean condor | Vultur gryphus | 3 | 0 * | |
| Reptilia total | 12 | 5 | 41.66 (18.1–70.6) | |
| Nile crocodile | Crocodylus niloticus | 1 | 0 | |
| Reticulate Gila monster | Heloderma suspectum suspectum | 1 | 0 | |
| African rock python | Python sebae | 1 | 0 | |
| Cuban tree boa | Chilabothrus angulifer | 1 | 0 | |
| Boa constrictor | Boa constrictor | 1 | 0 | |
| Yellow anaconda | Eunectes notaeus | 1 | 0 | |
| Leopard tortoise | Stigmochelys pardalis | 1 | 1 | |
| Spur-thighed tortoise (Greek tortoise) | Testudo graeca | 1 | 1 | |
| Giant Asian pond turtle | Heosemys grandis | 1 | 1 | |
| Radiated tortoise | Astrochelys radiata | 1 | 1 | |
| African spurred tortoise | Centrochelys sulcata | 1 | 1 | |
| Malaysian giant pond turtle | Orlitia borneensis | 1 | 0 | |
| Animals total | 201 | 54 | 26.86 (23.1–31.0) |
| Sample id | Host | No of Identified Sequences | ST1 (n) | ST2 (n) | ST3 (n) | ST5 (n) | ST8 (n) | ST10 (n) | ST13 (n) | ST14 (n) | NMA ST (n) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 6CZ, 7CZ, 10CZ, 22CZ, 23CZ, 33CZ | Human | 6 | 3 | - | 3 | - | - | - | - | - | - |
| NHPs total | 23 | 4 | 7 | 6 | - | - | - | 6 | - | - | |
| 135R | Rhesus macaque | 1 | - | - | 1 | - | - | - | - | - | - |
| 2S, 4S, 5S | Chimpanzee | 3 | - | 3 | - | - | - | - | - | - | - |
| 1P, 3P, 4P, 34P | Patas monkey | 4 | 1 | 1 | 2 | - | - | - | - | - | - |
| 3M, 5M, 25M, 27M | Mandrill | 4 | 3 | - | 1 | - | - | - | - | - | - |
| 1L-5L | Javan lutung | 5 | - | - | - | - | - | - | 5 | - | - |
| 14GE-17GE | Guereza | 4 | - | 3 | - | - | - | - | 1 | - | - |
| 18GI | Buff-cheeked gibbon | 1 | - | - | 1 | - | - | - | - | - | - |
| 19GI | White-cheeked gibbon | 1 | - | - | 1 | - | - | - | - | - | - |
| Suidae | |||||||||||
| 171DZ-173DZ 175DZ-179DZ | Wild boar | 8 | - | - | - | 8 | - | - | - | - | - |
| Tayassuidae | |||||||||||
| 161PE-164PE 166PE-170PE | Collared peccary | 9 | - | - | - | 9 | - | - | - | - | - |
| Bovidae total | 8 | - | - | - | - | - | 2 | - | 6 | - | |
| 181-186KO 188KO | Domestic goat | 7 | - | - | - | - | - | 2 | - | 5 | - |
| 189OW | Polish heath sheep | 1 | - | - | - | - | - | - | - | 1 | - |
| Metatheria | |||||||||||
| 86KA | Red kangaroo | 1 | - | - | - | - | 1 | - | - | - | - |
| Reptilia total | 5 | - | - | - | - | - | - | - | - | 5 | |
| 2Z | Leopard tortoise | 1 | - | - | - | - | - | - | - | - | 1 |
| 3Z | Spur-thighed tortoise (Greek tortoise) | 1 | - | - | - | - | - | - | - | - | 1 |
| 5Z | Giant Asian pond turtle | 1 | - | - | - | - | - | - | - | - | 1 |
| 4Z | Radiated tortoise | 1 | - | - | - | - | - | - | - | - | 1 |
| 10Z | African spurred tortoise | 1 | - | - | - | - | - | - | - | - | 1 |
| Animals total | 54 | 4 | 7 | 6 | 17 | 1 | 2 | 6 | 6 | 5 |
| Host | Blastocystis STs | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Chimpanzee (A) Pan troglodytes | - | ST2 | - | - | - | - | - - - - - - - - - - - | - | This study |
| - - - ST1 - ST1 ST1 ST1 ST1 ST1 | ST2 ST2 ST2 - ST2 - - - - ST2 | ST3 - - - - - - - - ST3 | ST5 ST5 - - - ST5 - - - ST5 | - - - - - - - - - - | - - - ST11 - - - - - - | ST15 - - - - - - - - - | [57] [15] [38] [14] [53] [58] [59] [11] [50] [12] | ||
| Buff-cheecked gibbon (As) Nomascus gabriellae | - | - | ST3 | - | - | - | - - - - - - | - | This study |
| ST1 ST1 ST1 ST1 ST1 | - ST2 - - - | ST3 St3 - - ST3 | - ST5 - - | - ST8 - - | - - - - - | - ST15 - - - | [57] [16] [15] [58] [12] | ||
| White-cheecked gibbon (As) Nomascus leucogenys | - - ST1 | - ST2 - | ST3 ST3 - | - - - | - - - | - - - | - - - | - - - | This study [60] [57] |
| Mandrill (A) Mandrillus sphinx | ST1 | - | ST3 | - | - | - | - - - - - - | - | This study |
| ST1 ST1 - ST1 - | - - - - - | - - ST3 ST3 ST3 | - - - - - | - - - - - | - - - - - | - - - - - | [57] [15] [53] [58] [54] | ||
| Rhesus macaque (As) Macaca mulatta | - | - | ST3 | - | - | - | - - - - - - - - - | - | This study |
| ST1 ST1 ST1 ST1 - - - ST1 | ST2 - ST2 ST2 ST2 ST2 - ST2 | ST3 - ST3 ST3 ST3 ST3 - - | - - - - - - ST5 - | - - - - ST8 - - - | - - - - - - - - | - - - - - - - - | [53] [60] [61] [56] [38] [58] [11] [22] | ||
| Guereza (A) Colobus guereza | - | ST2 | - | - | - | - | ST13 - - ST13 | - | This study |
| - ST1 ST1 | - ST2 ST2 | ST3 - ST3 | - - - | - - - | - - - | - - - | [57] [15] [50] | ||
| Patas monkey (A) Erythrocebus patas | ST1 | ST2 | ST3 | - | - | - | - - | - | This study |
| ST1 | - | ST3 | - | - | - | - | [57] | ||
| Javan lutung (As) Trachypithecus auratus | - | - | - | - | - | - | ST13 - ST13 | - | This study |
| - ST1 | - - | ST3 - | ST5 - | - - | - - | - - | [57] [56] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rudzińska, M.; Kowalewska, B.; Waleron, M.; Kalicki, M.; Sikorska, K.; Szostakowska, B. Molecular Characterization of Blastocystis from Animals and Their Caregivers at the Gdańsk Zoo (Poland) and the Assessment of Zoonotic Transmission. Biology 2021, 10, 984. https://doi.org/10.3390/biology10100984
Rudzińska M, Kowalewska B, Waleron M, Kalicki M, Sikorska K, Szostakowska B. Molecular Characterization of Blastocystis from Animals and Their Caregivers at the Gdańsk Zoo (Poland) and the Assessment of Zoonotic Transmission. Biology. 2021; 10(10):984. https://doi.org/10.3390/biology10100984
Chicago/Turabian StyleRudzińska, Monika, Beata Kowalewska, Małgorzata Waleron, Mirosław Kalicki, Katarzyna Sikorska, and Beata Szostakowska. 2021. "Molecular Characterization of Blastocystis from Animals and Their Caregivers at the Gdańsk Zoo (Poland) and the Assessment of Zoonotic Transmission" Biology 10, no. 10: 984. https://doi.org/10.3390/biology10100984