1. Introduction
Pure zirconia (ZrO
2) is a polymorphic material which may exist in three crystalline forms: Monoclinic (m-ZrO
2), tetragonal (t-ZrO
2), and cubic (c-ZrO
2). The m-ZrO
2 is stable at temperatures up to 1170 °C, t-ZrO
2 in the range of 1170–2370 °C, and c-ZrO
2 at over 2370 °C [
1]. A noticeable ~4% increase in the volume of sintered pure zirconia ceramic during the cooling process associated with t-ZrO
2 → m-ZrO
2 transformation was accompanied by the material microcracking [
2]. Therefore, m-ZrO
2 ceramic with poor mechanical properties is hardly ever used for applications requiring mechanical strength. To stabilize t-ZrO
2 and c-ZrO
2 at room temperatures and to prevent ceramic volume expansion during the cooling process, zirconia was doped by different oxides (Y
2O
3, CeO
2, CaO, MgO, and others). An important feature of zirconia ceramics is that varying t-ZrO
2 and c-ZrO
2 content by changing the dopant concentration it is possible to produce materials for different applications. Herein, c-ZrO
2 content increased with the rise in dopant concentration. For example, introduction of 3 mol% Y
2O
3 in zirconia allows the preparation of engineering ceramics with high values of bending strength and fracture toughness [
1]. Such characteristics as thermal expansion coefficient close to that of super alloys, low thermal conductivity, and high erosion resistance make zirconia ceramics with Y
2O
3 content in the range of 6–8 mol% very suitable materials for thermal barrier coatings [
3]. Ceramics containing 8–10 mol% of Y
2O
3 are used in solid fuel cells as electrolyte material due to the high oxygen ion conductivity [
4].
Stabilized zirconia ceramics can be used not only as bulk materials or coatings, but also in the form of fibers, too, for example, for composites’ reinforcement. Microfibers of yttria-stabilized tetragonal zirconia with tensile strength up to 2.6 GPa were fabricated in [
5]. Due to the size effects, nanofibers show remarkable properties compared to microfibers, namely, high surface-to-mass ratio, flexibility in surface functionalization, and high mechanical strength [
6]. In [
7], electrospun alumina nanofibers with the tensile strength average value ca. 11 GPa were obtained, while commercial alumina microfibers only reached a strength value of 3.3 GPa (Nextel™ 610, 3M, Saint Paul, MN, USA).
Among a number of nanofibers’ fabrication techniques, electrospinning is the most commonly used method due to its simplicity, adaptability, cost-efficiency, versatility, and capability to control the diameter, composition, and morphology of nanofibers. Besides, electrospinning is the only method applicable for the large-scale fabrication of nanofibers [
6].
Previously, 8 mol% Y
2O
3—stabilized zirconia nanofibers were prepared by electrospinning and calcination at elevated temperatures with zirconium acetate hydroxide and zirconium propoxide as zirconia precursors [
8,
9]. These nanofibers of c-ZrO
2 can be used as a part of an electrode of solid oxide fuel cells for improved electrode performance, as was shown in [
10]. In [
11], 3 mol% Y
2O
3—stabilized zirconia nanofibers composed of t-ZrO
2 grains were obtained. The electrospun macroporous membranes of t-ZrO
2 nanofibers providing efficient gas transport are prospective for catalytic applications, for example, in hydrogenation [
12] and oxidation [
13] processes. Besides, nanofibers of t-ZrO
2 are more preferred for reinforcement than similar microfibers, mentioned above, due to their expected higher strength and better matrix-fiber adhesion which improves load transfer from the matrix to the reinforcing fibers.
It is well known that porosity reduces strength in nanofibers. In [
9,
14], it was revealed that a rise in calcination temperature led to zirconia nanofibers’ densification. In this study we examined the opportunity for controlling dense zirconia nanofibers’ phase composition to fabricate non-porous t-ZrO
2 nanofibers, which can be considered as a promising material for composite reinforcement via varying dopant content on an example of a commonly used stabilizer, Y
2O
3. As far as we know, there is no information about the range of Y
2O
3 concentrations to obtain t-ZrO
2 nanofibers.
2. Materials and Methods
Ten identical 10 wt.% polymer solutions were prepared by adding 1 g polyacrylonitrile (PAN) (molecular weight Mw = 150,000, Sigma-Aldrich, Saint Louis, MO, USA) into 9 g dimethylformamide (DMF) with periodic stirring by hand for 2 h at 50 °C. To synthesize nanofibers of yttria-stabilized zirconia, zirconium acetylacetonate Zr(C
5H
7O
2)
4 (hereinafter referred to as ZrAA) (Sigma-Aldrich, Saint Louis, MO, USA) as zirconia precursor and yttrium nitrate hexahydrate Y(NO
3)
3·6H
2O (Sigma-Aldrich, Saint Louis, MO, USA) as yttria precursor were added into polymer solutions under stirring in the ultrasonic bath for 6 min at 42 kHz at room temperature to prepare ten transparent electrospinning solutions. The ZrAA/PAN mass ratio in all solutions was 0.3 to 1. Yttrium nitrate hexahydrate was added to solutions in amounts to obtain 0–9 mol% Y
2O
3 concentration in zirconia nanofibers. Herein, insignificant change of solutions’ viscosity was observed. It was important since viscosity of the electrospinning solution significantly affects the diameter and morphology of the obtained nanofibers [
15]. The dynamic viscosity of all electrospinning solutions was measured using the rotational viscometer DV2T (AMETEK Brookfield, Middleborough, MA, USA).
Each prepared composite solution was transferred into a 10 mL plastic syringe and electrospun using the electrospinning machine NANON-01A (MECC, Fukuoka, Japan) at a feeding rate of 1.2 mL/h through a 21 G blunt-tip needle. The distances between the needle tip and the flat collector of 21 cm and the accelerating voltage of 23 kV were chosen as operative parameters to fabricate bead-free and smooth composite fibers. The flat collector was covered by aluminum foil. The fibers were collected on aluminum foil as non-woven mats.
As-spun mats were calcined at temperatures in the range of 1100–1300 °C for 1 h using two-stage heating: Heating to 500 °C with a heating rate of 1 °C/min, and then further heating to the target temperature with a heating rate of 5 °C/min. According to the thermogravimetric (TG) analysis, composite fibers transformed to ZrO
2 fibers at 500 °C (
Figure 1a) and there was no further weight loss of filaments observed at higher temperature. To prevent fiber destruction, a low heating rate at the first annealing stage was chosen to ensure delicate removal of the decomposition products of the ceramic precursor, the stabilizer precursor, and binding polymer.
The fibers’ surface texture was analyzed using a scanning electron microscope (SEM) Merlin (Carl Zeiss, Oberkochen, Germany). The average diameter of the fibers and the average grain size were determined from the SEM images. X-ray diffraction (XRD) patterns were registered in the 20–80° 2 θ range by an X-ray diffractometer D2 Phaser (Bruker AXS, Karlsruhe, Germany) using CuKα1 monochromatic radiation. XRD patterns were identified using the PDF-2 Diffraction Database File of the International Centre for Diffraction. The TOPAS software (Bruker AXS, Karlsruhe, Germany) was used to determine nanofibers’ quantitative phase composition by the Rietveld method. SEM and XRD measurements were carried out at room temperature. Nitrogen adsorption-desorption isotherms at 77 K were registered with a gas sorption analyzer, Autosorb iQ-C (Quantachrome Instruments, Boynton Beach, FL, USA). Before nitrogen adsorption measurements, the samples were evacuated at 300 °C for 3 h under vacuum to remove contaminants. Specific surface areas of the nanofibers were determined with the Brunauer–Emmett–Teller (BET) method. Total pore volume was calculated through the quantity of adsorbed N2 at the relative pressure of 0.995. The TG analysis was performed on the thermal analyzer EXSTAR TG/DTA7200 (SII Nano Technology, Tokyo, Japan) in air atmosphere with a heating rate of 10 °C/min.
3. Results and Discussion
As shown in
Figure 1b, as-spun composite fibers were uniform in shape with a smooth surface and the average diameter of 544 ± 38 nm. Randomly distributed, cylindrical, bead-free fibers formed a macroporous non-woven mat, typical for electrospinning. The transformation of composite fibers to ZrO
2 ones was accompanied with the decrease in their thickness due to residual solvent removal and ZrAA and PAN decomposition (
Figure 1a).
After calcination at 1100 °C, the average diameter of fibers decreased distinctly to ca. 140 nm (
Figure 1c). The obtained nanofibers had rough surfaces and were characterized by grain structure. Zirconia nanofibers with 2–3 mol% of Y
2O
3 possessed the smallest grain size of ca. 60 nm among all the ceramic fibers prepared at 1100 °C. With a rise in the calcination temperature from 1100 °C to 1300 °C, fine-grain structure of 2–3 mol% yttria-stabilized zirconia nanofibers (
Figure 1c) transformed to coarse-grain ones (
Figure 1d) and an insignificant decrease within the measurement error was detected in the nanofibers’ average diameter. At a calcination temperature of 1300 °C, a chain-like structure was formed when the grains with the average size of ca. 130 nm reached the size equal to the nanofiber diameter. The observed grain growth was associated with the recrystallization rate increase induced by temperature rising. As in [
9], it was noted that, at calcination temperature of 1300 °C, the grain size of 8 mol% Y
2O
3–ZrO
2 nanofibers exceeded 200 nm (
Figure 1e). The grain growth of 8 mol% yttria-stabilized zirconia being fully cubic zirconia [
9] is well known to be accelerated during sintering above 1100 °C, and grains reached micrometer size [
16]. Comparing SEM images both of 2 mol% Y
2O
3–ZrO
2 nanofibers and undoped ZrO
2 nanofibers calcined at 1300 °C (
Figure 1d,f) it can be concluded that the addition of a small amount of yttria to zirconia inhibited ZrO
2 grain growth at high calcination temperatures. Creating a diffusion barrier prevented nanofibers’ destruction. Cracking along the grain boundaries due to t-ZrO
2 → m-ZrO
2 transition upon cooling in undoped zirconia nanofibers may also have contributed to their destruction, presented in
Figure 1f.
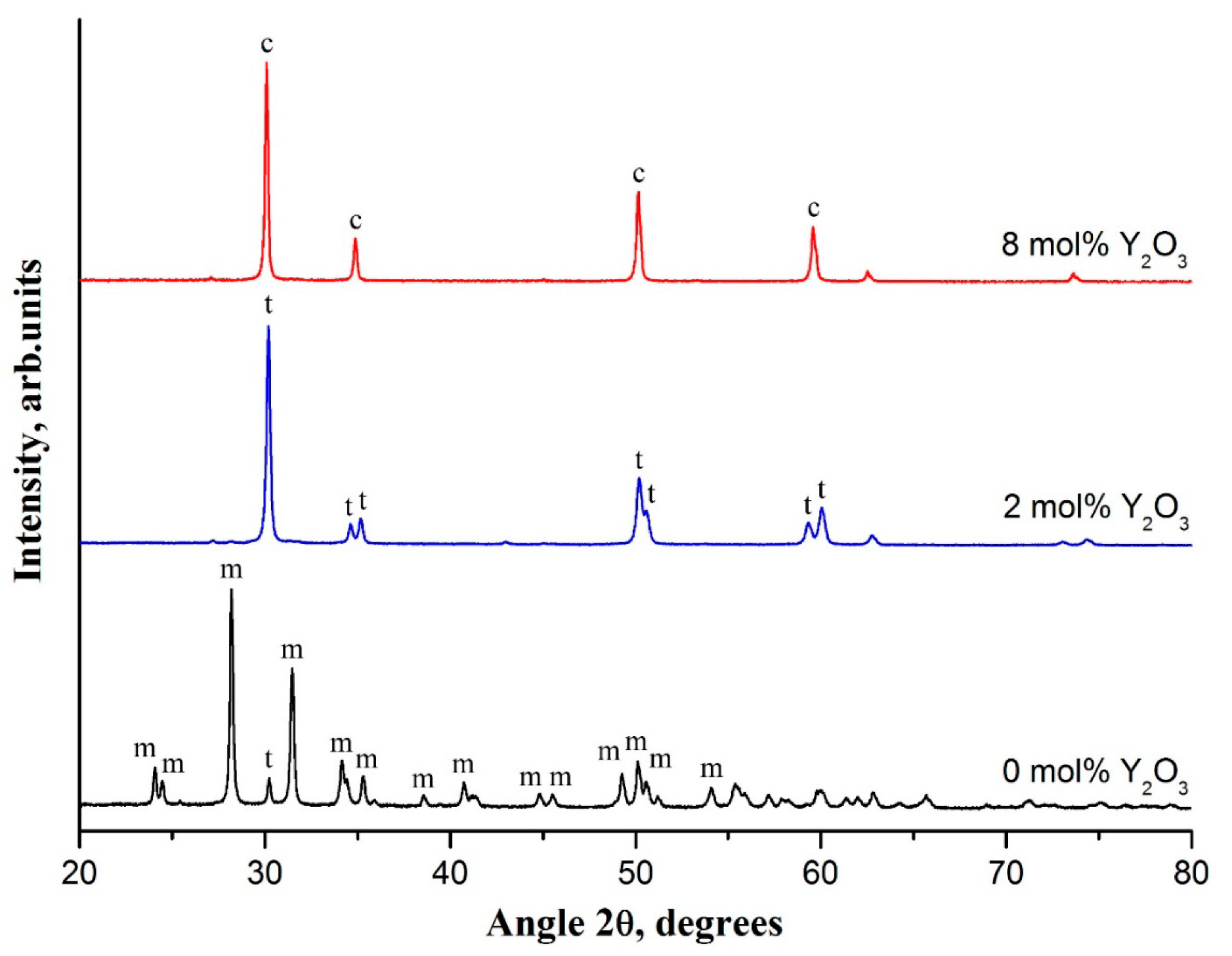
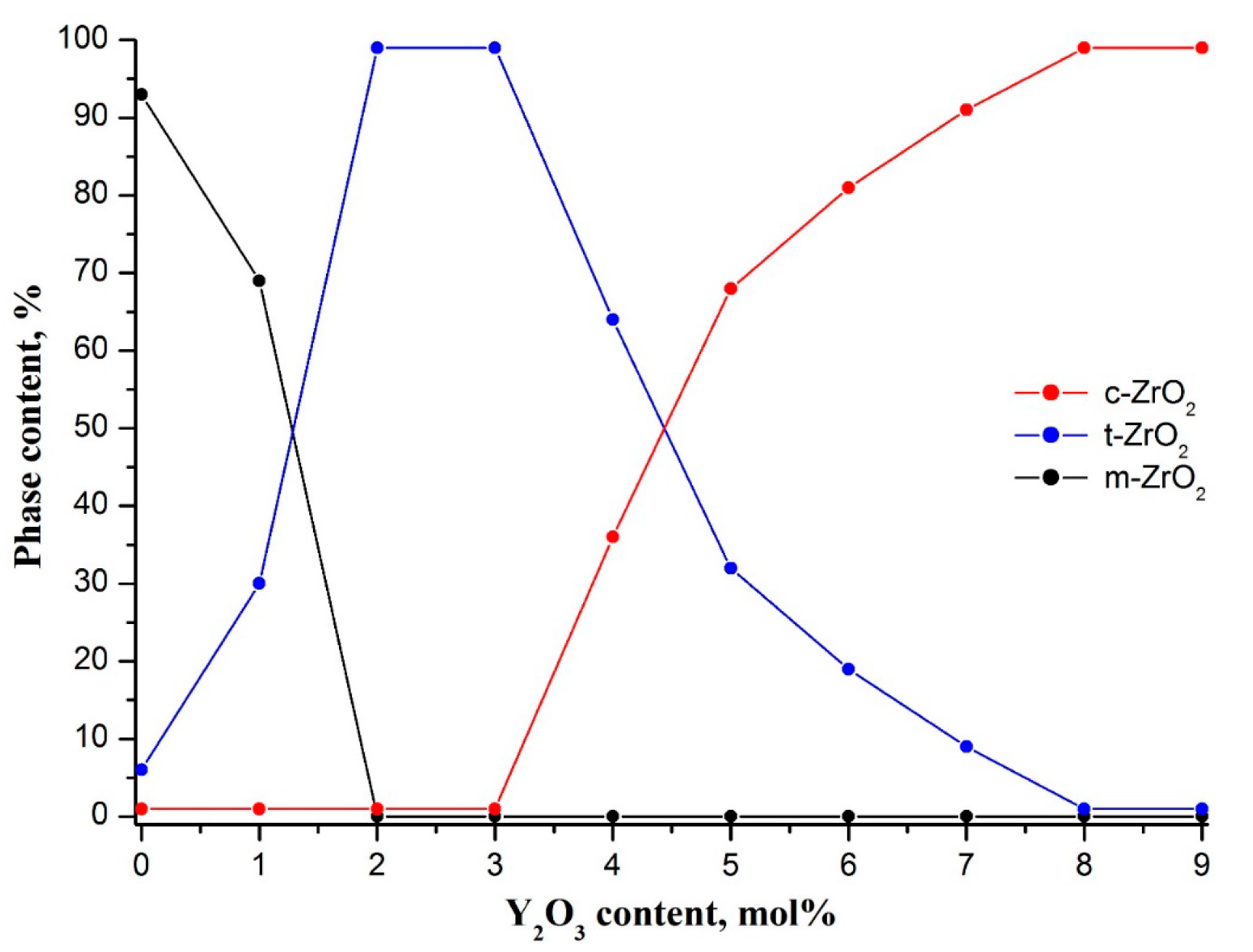
The evolution of the crystalline structure of zirconia nanofibers doped with various amounts of yttrium oxide is shown in
Figure 2 and the phase analysis results in the whole range of studied Y
2O
3 concentrations are presented in
Figure 3.
Filaments prepared at 1100 °C were selected for investigation of phase composition variation in zirconia nanofibers, since undoped zirconia nanofibers cease to be fibers at a calcination temperature of 1300 °C (
Figure 1f). The XRD pattern of undoped zirconia nanofibers contained two intensive peaks at 28.2° and 31.5° and a large number of low-intensity peaks in the 2 θ range of 20–80° which belong to m-ZrO
2 (
Figure 2). A low-intensity peak of t-ZrO
2 at 30.2° was observed, too. The content of m-ZrO
2 was determined as 93%. So, undoped zirconia nanofibers calcined at 1100 °C mostly consisted of m-ZrO
2 grains. Previously, nanofibers of m-ZrO
2 were also detected when intermediate composite fibers with zirconium oxychloride [
17] and zirconium acetate [
18] as zirconia precursors and containing no stabilizing agent were heat-treated at elevated temperatures. From the reflection at 28.2° via the Scherrer equation, the average m-ZrO
2 grain size was estimated to be about 70 nm. According to Garvie’s theory, stabilizer-free t-ZrO
2 grains with size not exceeding 30 nm may be stable at room temperature [
19]. Otherwise, they are subject for t-ZrO
2 → m-ZrO
2 transition.
Introduction of yttria into zirconia nanofibers led to a dramatic decrease in m-ZrO
2 content to 0% at 2 mol% Y
2O
3 (
Figure 3). Fully tetragonal zirconia was observed in the yttrium oxide concentration range of 2–3 mol%. XRD patterns contained only characteristic reflections of t-ZrO
2. Main t-ZrO
2 peaks localized at 30.2°, 34.6°, 35.2°, 50.2°, 50.7°, 59.3°, and 60.2° (
Figure 2). Further increase in yttria concentration caused a monotonic decrease in volume fraction of t-ZrO
2 to 0% at 8 mol% Y
2O
3. A sharp drop in t-ZrO
2 content from 100% to 32% occurred in the interval of 3–5 mol% Y
2O
3. At the same time, a corresponding monotonic rise in c-ZrO
2 content attaining saturation at 8 mol% Y
2O
3 was observed. Yttria-stabilized zirconia nanofibers were composed of t-ZrO
2 and c-ZrO
2 grains in the interval of 3–8 mol% Y
2O
3. At 8 mol% Y
2O
3 XRD pattern contained only intensive reflections of c-ZrO
2 at 30.1°, 35.0°, 50.2°, and 59.7° that indicated fully cubic nanofibrous zirconia (
Figure 2).
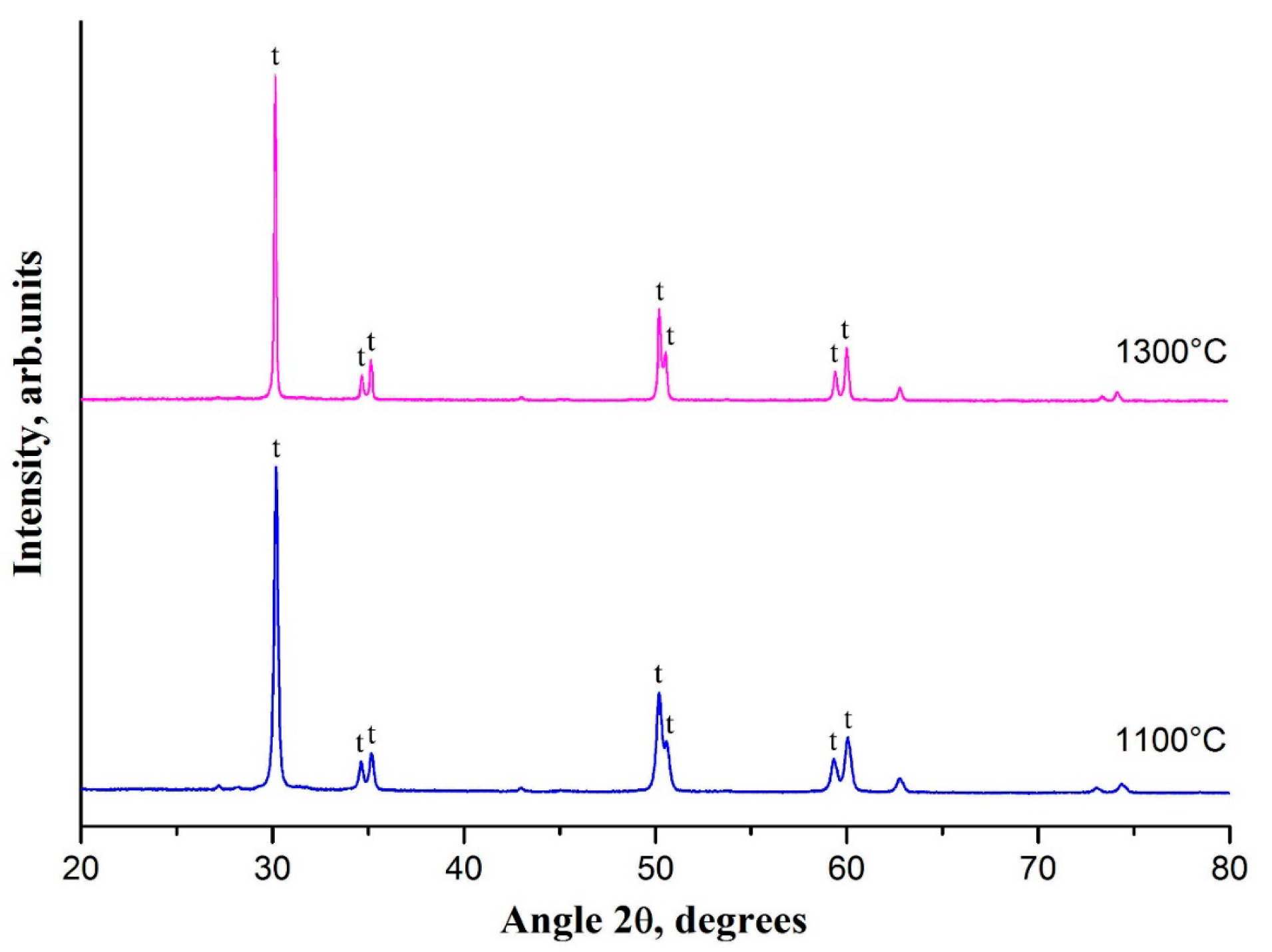
For t-ZrO
2 nanofibers of interest, the effect of calcination temperature on their phase composition was investigated. Comparison of the XRD patterns obtained for the samples calcined at 1100 °C and 1300 °C (
Figure 4) revealed that a rise in heat treatment temperature makes t-ZrO
2 peaks sharper and narrower, indicating grain size growth confirmed by SEM (
Figure 1c,d).
No additional reflections of another zirconia phase were detected in the XRD pattern of the sample calcined at 1300 °C. The observed essential increase in t-ZrO2 grain size from ca. 60 nm to ca. 130 nm unaccompanied by t-ZrO2 → m-ZrO2 transition indicated phase stability of the nanofibers of yttria-stabilized tetragonal zirconia at sufficiently high heat treatment temperatures and revealed that the grain size of 130 nm is not yet critical for t-ZrO2 retention in nanofibers of yttria-stabilized zirconia at room temperature.
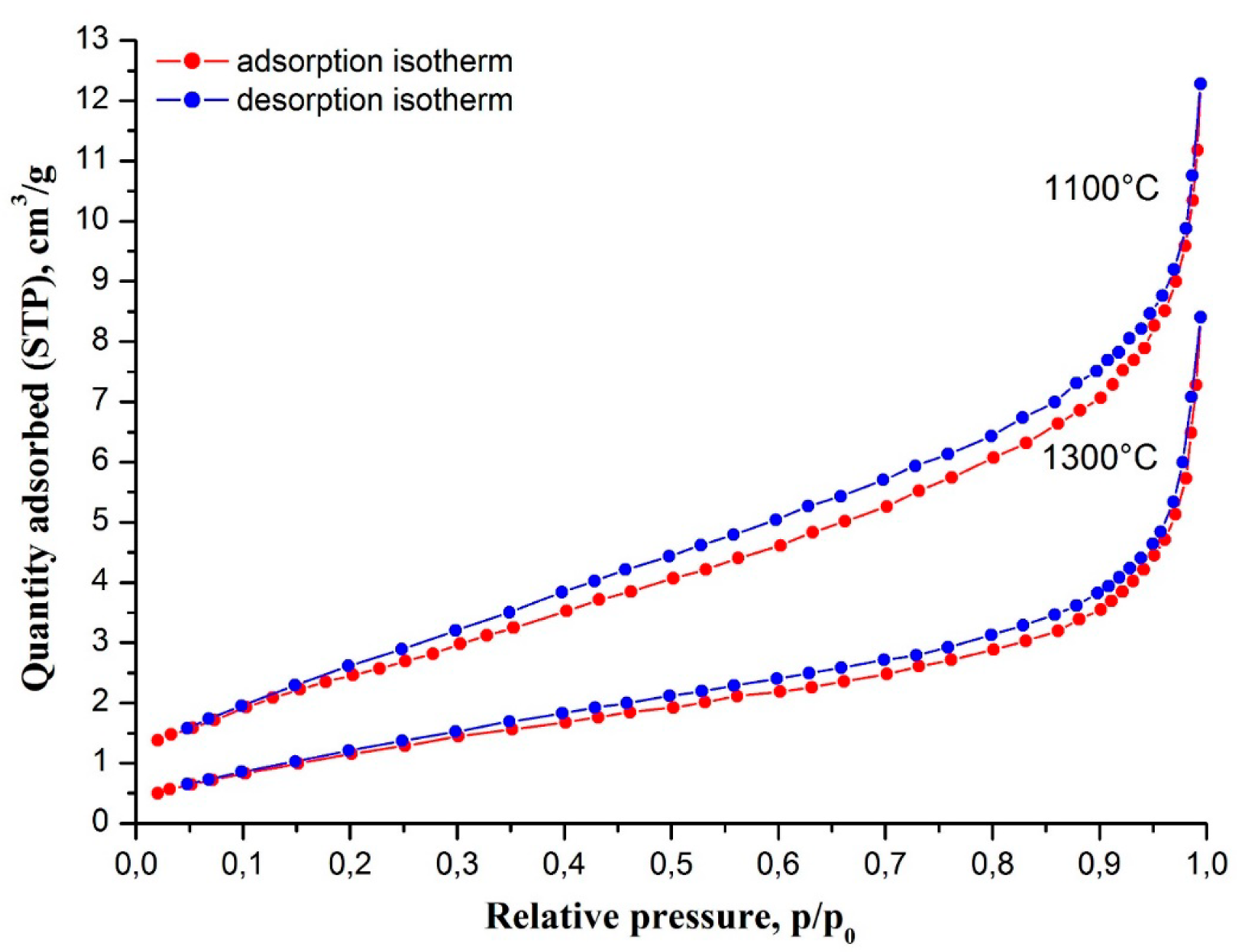
The porous structure of nanofibers of yttria-stabilized tetragonal zirconia calcined at 1100 °C and 1300 °C was examined by nitrogen adsorption measurements (
Figure 5). In both cases nanofibers showed type II isotherms. According to the International Union of Pure and Applied Chemistry (IUPAC), classification type II isotherms are inherent in non-porous materials. In our case, that was confirmed by small total pore volume values of 0.019 cm
3/g and 0.013 cm
3/g for zirconia nanofibers calcined at 1100 °C and 1300 °C, correspondingly. Feebly marked type H4 hysteresis loops by IUPAC classification are associated with narrow slit-like pores, few in number. In our case, the slit-like pores were most likely formed by the boundaries of adjacent ZrO
2 grains. An observed slight decrease of total pore volume (porosity reduction) is due to higher sintering efficiency at higher temperature. The rise in calcination temperature also led to a decrease in zirconia nanofibers’ specific surface area from 9.9 m
2/g at 1100 °C to 5.0 m
2/g at 1300 °C.