Energy Transfer between Er3+ and Pr3+ for 2.7 μm Fiber Laser Material
Abstract
:1. Introduction
2. Experimental
3. Results and Discussions
3.1. Absorption Spectra and Infrared Transmittance Spectrum
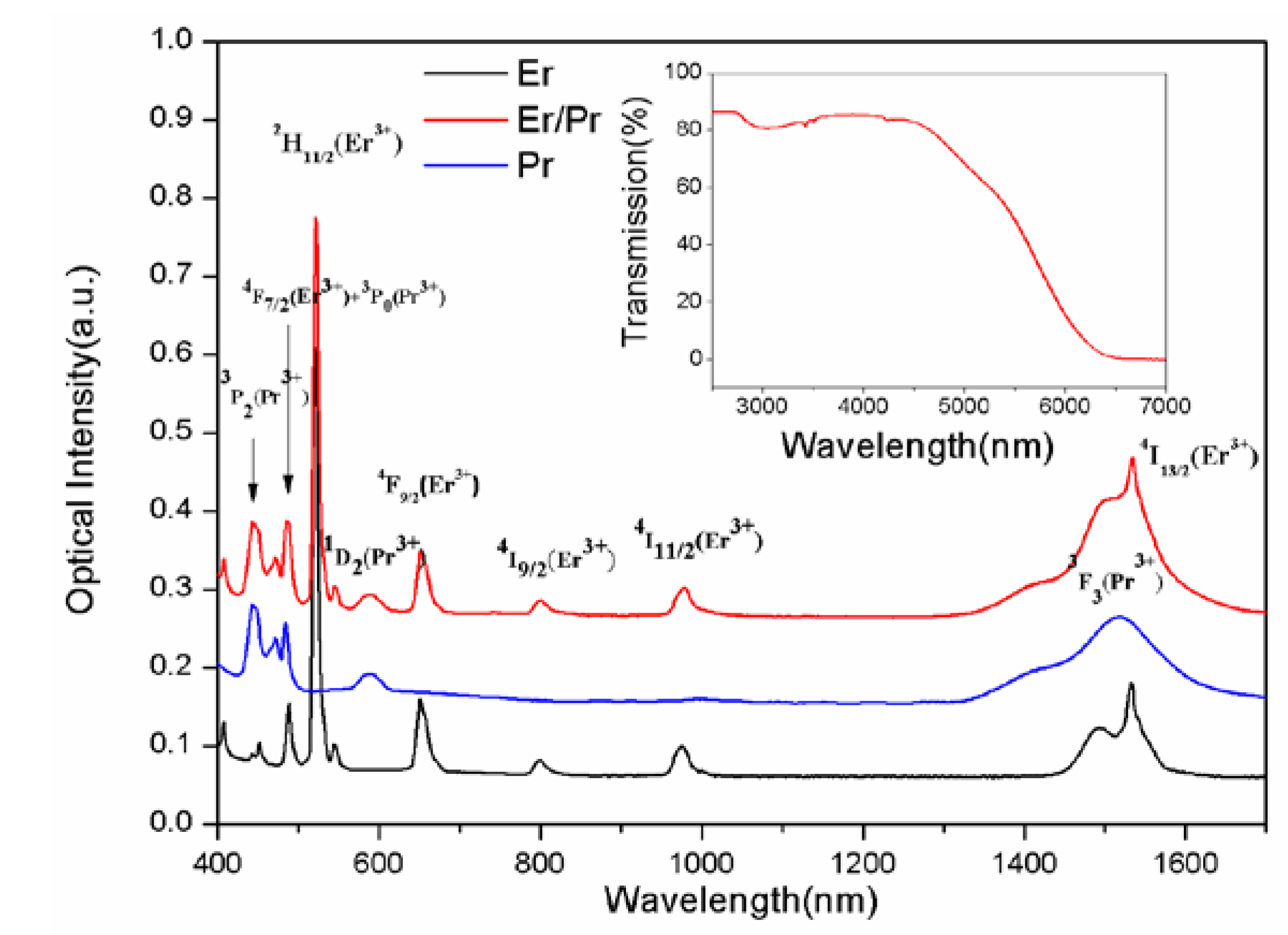
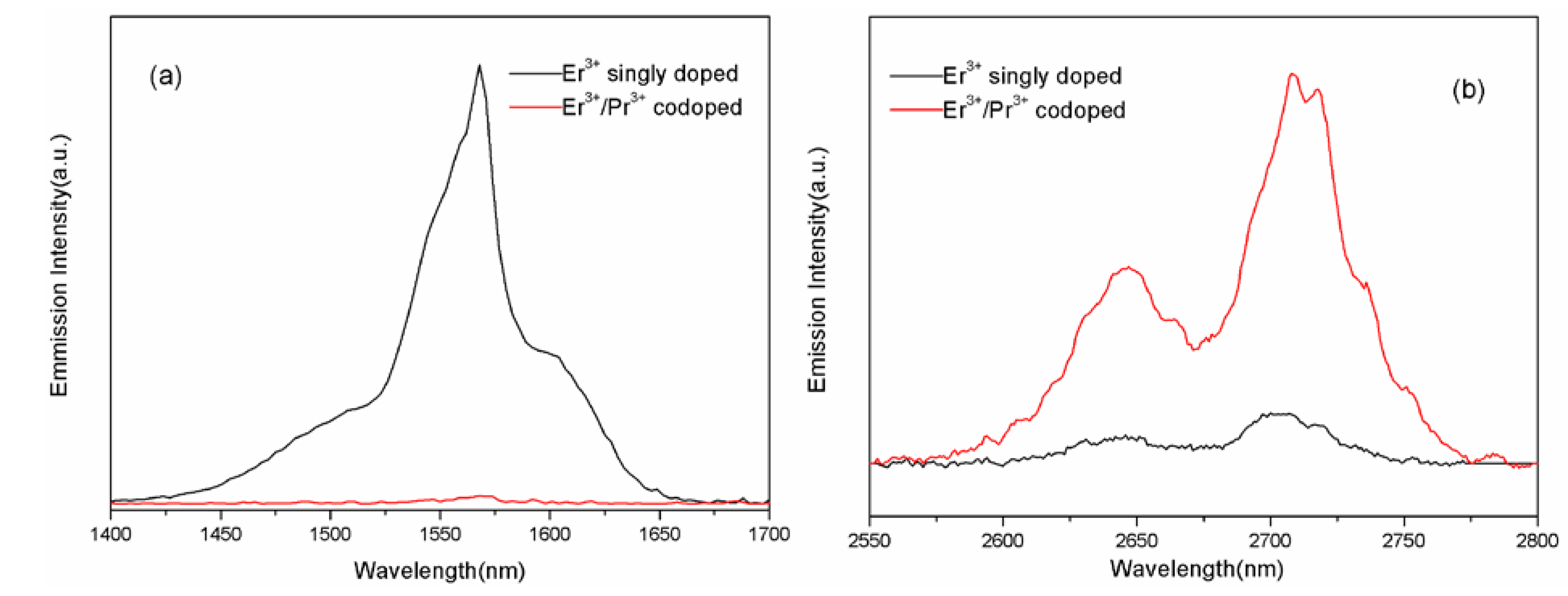
3.2. Fluorescence Spectra and Judd-Ofelt Analysis
| Parameters | Germanate | Fluorophosphate | Fluoride | Silicate | Fluorotellurite | |
|---|---|---|---|---|---|---|
| Er3+ | Er3+/Pr3+ | |||||
| Ω2 | 6.60 | 13.23 | 5.14 ± 0.10 | 2.98 | 4.23 | 4.38 |
| Ω4 | 1.75 | 2.81 | 1.02 ± 0.08 | 1.40 | 1.04 | 3.05 |
| Ω6 | 0.99 | 2.26 | 0.91 ± 0.06 | 1.04 | 0.61 | 1.04 |
| Reference | this work | [18] | [19] | [20] | [4] | |
| InitialLevel | EndLevel | Er3+ | Er3+/Pr3+ | ||||
|---|---|---|---|---|---|---|---|
| Arad | β | τ (ms) | Arad | β | τ (ms) | ||
| 4I13/2 | 4I15/2 | 183.17 | 100.00% | 5.46 | 334.21 | 100.00% | 2.99 |
| 4I11/2 | 4I15/2 | 182.92 | 83.29% | 4.55 | 402.18 | 86.84% | 2.16 |
| 4I13/2 | 36.69 | 16.71% | 60.93 | 13.16% | |||
| 4I9/2 | 4I15/2 | 183.28 | 77.57% | 4.23 | 297.32 | 71.91% | 2.42 |
| 4I13/2 | 49.82 | 21.08% | 112.95 | 27.32% | |||
| 4I11/2 | 3.19 | 1.35% | 3.19 | 0.77% | |||
| 4F9/2 | 4I15/2 | 1882.77 | 90.80% | 0.48 | 3428.00 | 90.09% | 0.26 |
| 4I13/2 | 104.40 | 5.03% | 188.15 | 4.94% | |||
| 4I11/2 | 80.91 | 3.90% | 178.36 | 4.69% | |||
| 4I9/2 | 5.36 | 0.26% | 10.75 | 0.28% | |||
| 4S3/2 | 4I15/2 | 1249.87 | 62.83% | 0.50 | 2853.25 | 63.07% | 0.22 |
| 4I13/2 | 632.44 | 31.79% | 1443.76 | 31.91% | |||
| 4I11/2 | 39.90 | 2.01% | 89.01 | 1.97% | |||
| 4I9/2 | 67.14 | 3.37% | 138.05 | 3.05% | |||
| 2H11/2 | 4I15/2 | 11581.49 | 22658.36 | ||||
| 4F7/2 | 4I15/2 | 3390.03 | 7067.49 | ||||
| 4F5/2 | 4I15/2 | 779.07 | 1778.47 | ||||
| 2H9/2 | 4I15/2 | 1631.01 | 33.35% | 0.20 | 3228.73 | 33.05% | 0.10 |
| 4I13/2 | 2325.23 | 47.54% | 4722.89 | 48.35% | |||
| 4I11/2 | 844.79 | 17.27% | 1640.00 | 16.79% | |||
| 4I9/2 | 39.18 | 0.80% | 76.81 | 0.79% | |||
| 4F9/2 | 50.44 | 1.03% | 99.76 | 1.02% | |||
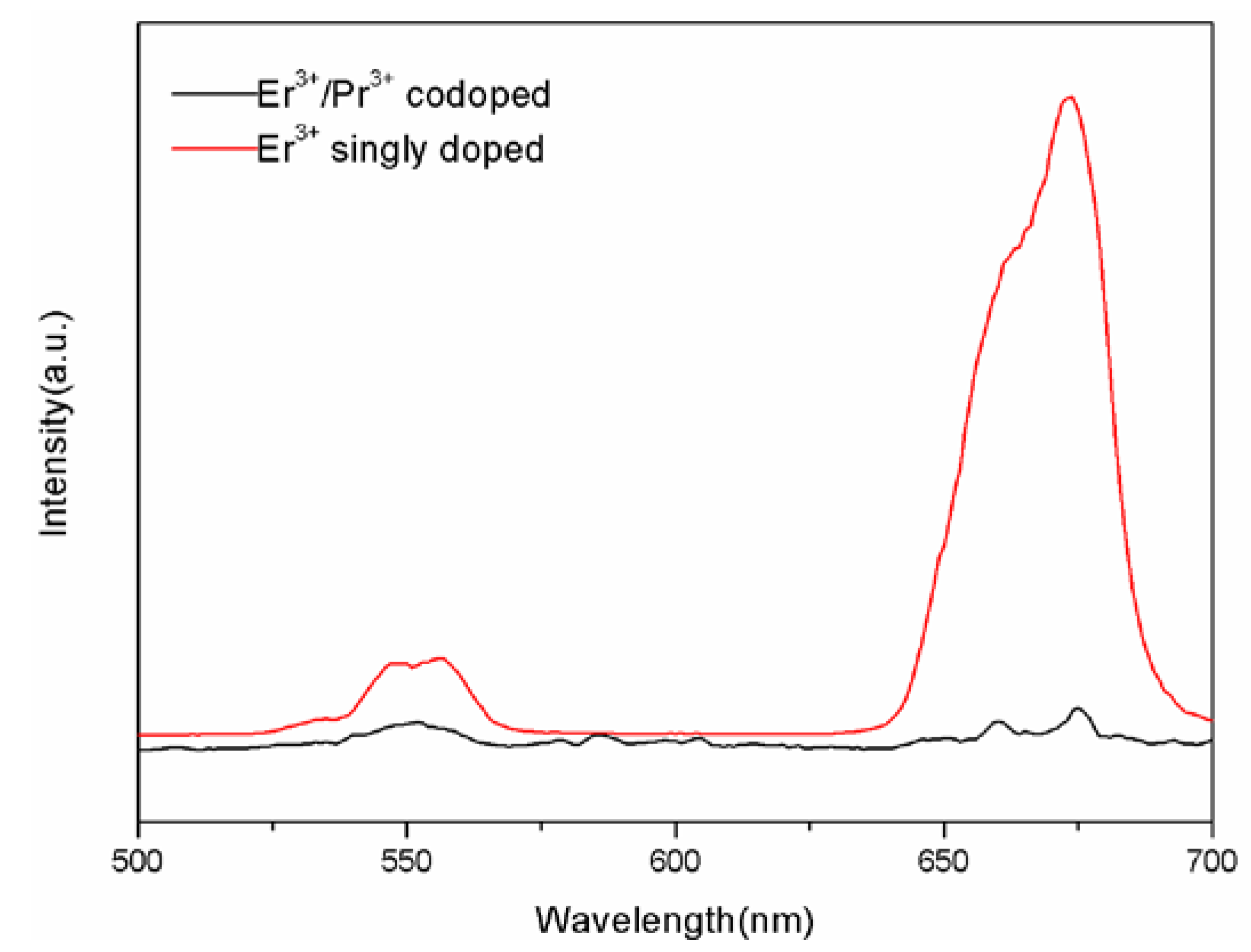
3.3. Energy Transfer Mechanisms between Er3+and Pr3+
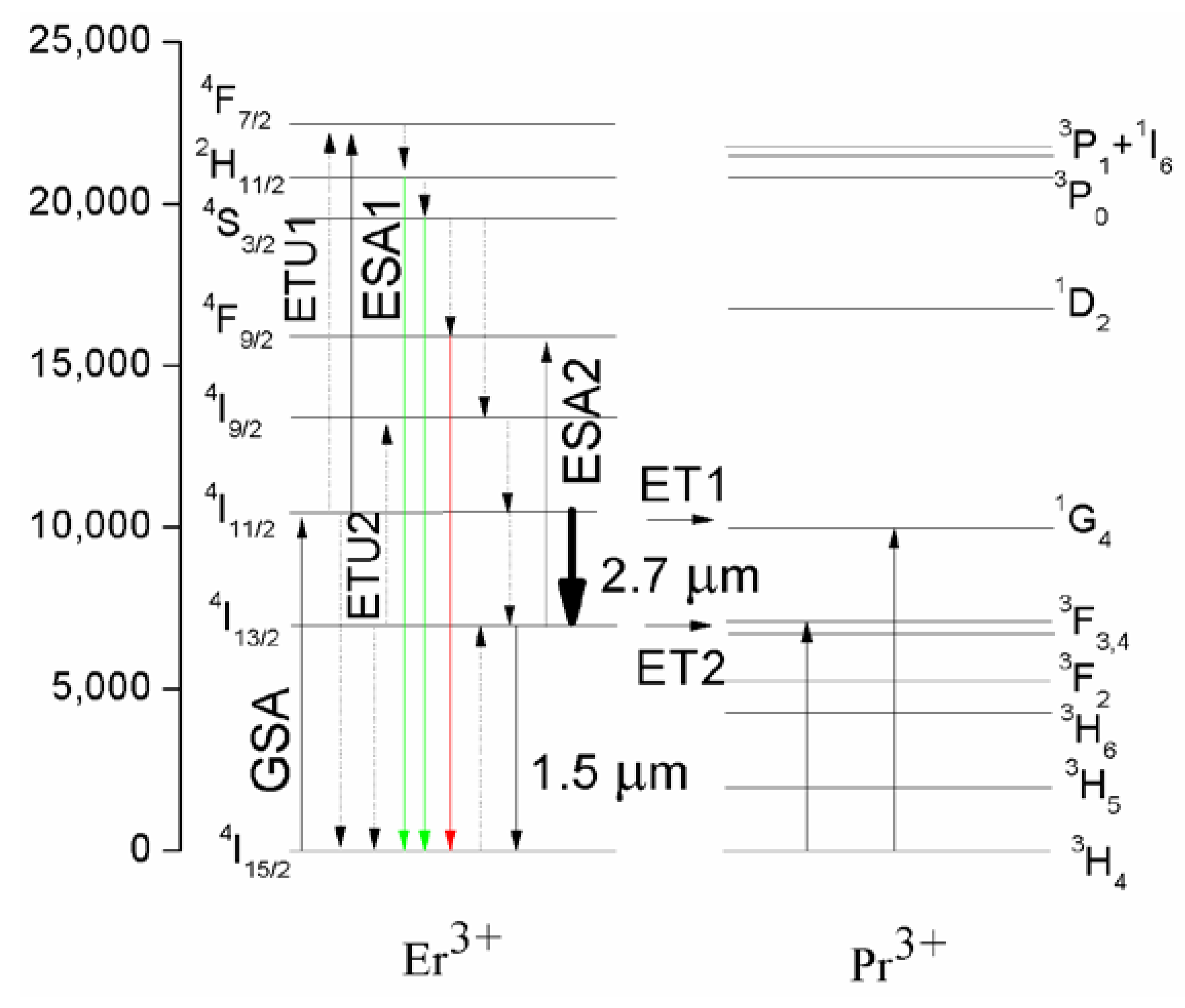









| Energy Transfer | N (% Phonon Assisted) | Transfer Coefficient (10−40 cm6/s) | |||
|---|---|---|---|---|---|
| Er3+: 4I11/2→Pr3+: 1G4 | 0 | 1 | 2 | 3 | 3.67 |
| 55 | 32 | 12 | 1% | ||
| Er3+: 4I13/2→Pr3+: 3F3, 4 | 0 | 1 | 2 | 42.25 | |
| 82 | 15 | 3% | |||
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chai, G.; Dong, G.; Qiu, J.; Zhang, Q.; Yang, Z. 2.7 μm emission from transparent Er3+,Tm3+ codoped yttrium aluminum Garnet (Y3Al5O12) nanocrystals–tellurate glass composites by novel comelting technology. J. Phys. Chemi. C 2012, 116, 19941–19950. [Google Scholar] [CrossRef]
- Tsang, Y.H.; El-Taher, A.E.; King, T.A.; Jackson, S.D. Efficient 2.96 µm dysprosium-doped fluoride fibre laser pumped with a Nd:YAG laser operating at 1.3 µm. Opt. Express 2006, 14, 678–685. [Google Scholar] [CrossRef]
- Jackson, S.D. Single-transverse-mode 2.5-W holmium-doped fluoride fiber laser operating at 2.86 µm. Opt. Lett. 2004, 29, 334–336. [Google Scholar] [CrossRef]
- Guo, Y.; Li, M.; Hu, L.; Zhang, J. Effect of fluorine ions on 2.7 μm emission in Er3+/Nd3+-codoped fluorotellurite glass. J. Phys. Chem. A 2012, 116, 5571–5576. [Google Scholar] [CrossRef]
- Wen, L.; Wang, J.; Hu, L.; Zhang, L. Sensitizing effect of Nd3+ on the Er3+:2.7 μm emission in fluorophosphate glass. Chin. Opt. Lett. 2011, 9. [Google Scholar] [CrossRef]
- Sousa, D.F.d.; Sampaio, J.A.; Nunes, L.A.; Baesso, M.L.; Bento, A.C.; Miranda, L.C. Energy transfer and the 2.8-μm emission of Er3+ and Yb3+ doped low silica content calcium aluminate glasses. Phys. Rev. B 2000, 62, 3176–3180. [Google Scholar]
- Bai, G.; Ding, J.; Tao, L.; Li, K.; Hu, L.; Tsang, Y.H. Efficient 2.7 μm emission from Er3+/Pr3+ codoped oxyfluorotellurite glass. J. Non-Cryst. Solids 2012, 358, 3403–3406. [Google Scholar] [CrossRef]
- Bai, G.; Tao, L.; Li, K.; Hu, L.; Tsang, Y.H. Enhanced light emission near 2.7 μm from Er–Nd co-doped germanate glass. Opt. Mater. 2013, 35, 1247–1250. [Google Scholar] [CrossRef]
- Schneider, J.; Hauschild, D.; Frerichs, C.; Wetenkamp, L. Highly efficient Er3+: Pr3+ codoped CW fluorozirconate fiber laser operating at 2.7 µm. Int. J. Infrared Millim. Waves 1994, 15, 1907–1922. [Google Scholar] [CrossRef]
- Coleman, D.J.; King, T.A.; Ko, D.-K.; Lee, J. Q-switched operation of a 2.7 μm cladding-pumped Er3+/Pr3+ codoped ZBLAN fibre laser. Opt. Commun. 2004, 236, 379–385. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Z.; Tian, Y.; Zhang, J.; Hu, L. Comparative investigation on the 2.7 μm emission in Er3+/Ho3+ codoped fluorophosphate glass. J. Appl. Phys. 2011, 110, 093106. [Google Scholar] [CrossRef]
- Guo, Y.; Li, M.; Tian, Y.; Xu, R.; Hu, L.; Zhang, J. Enhanced 2.7 μm emission and energy transfer mechanism of Nd3+/Er3+ co-doped sodium tellurite glasses. J. Appl. Phys. 2011, 110, 013512:1–013512:5. [Google Scholar]
- Lin, H.; Chen, D.; Yu, Y.; Yang, A.; Wang, Y. Enhanced mid-infrared emissions of Er3+ at 2.7 μm via Nd3+ sensitization in chalcohalide glass. Opt. Lett. 2011, 36, 1815–1817. [Google Scholar] [CrossRef]
- Song, J.H.; Heo, J.; Park, S.H. Emission properties of PbO-Bi2O3-Ga2O3-GeO2 glasses doped with Tm3+ and Ho3+. J. Appl. Phys. 2003, 93, 9441–9445. [Google Scholar] [CrossRef]
- Richards, B.D.O.; Teddy-Fernandez, T.; Jose, G.; Binks, D.; Jha, A. Mid-IR (3–4 μm) fluorescence and ASE studies in Dy3+ doped tellurite and germanate glasses and a fs laser inscribed waveguide. Laser Phys. Lett. 2013, 10. [Google Scholar] [CrossRef]
- Golding, P.S.; Jackson, S.D.; King, T.A.; Pollnau, M. Energy transfer processes in Er3+-doped and Er3+,Pr3+-codoped ZBLAN glasses. Phys. Rev. B 2000, 62, 856–864. [Google Scholar] [CrossRef]
- Feng, X.; Tanabe, S.; Hanada, T. Spectroscopic properties and thermal stability of Er3+-doped germanotellurite glasses for broadband fiber amplifiers. J. Am. Ceram. Soc. 2001, 84, 165–171. [Google Scholar] [CrossRef]
- Tian, Y.; Xu, R.; Zhang, L.; Hu, L.; Zhang, J. Observation of 2.7 μm emission from diode-pumped Er3+/Pr3+-codoped fluorophosphate glass. Opt. Lett. 2011, 36, 109–111. [Google Scholar] [CrossRef]
- Ivanova, S.; Pellé, F. Strong 1.53 μm to NIR-VIS-UV upconversion in Er-doped fluoride glass for high-efficiency solar cells. J. Opt. Soc. Am. B 2009, 26, 1930–1938. [Google Scholar] [CrossRef]
- Zou, X.; Izumitani, T. Spectroscopic properties and mechanisms of excited state absorption and energy transfer upconversion for Er3+-doped glasses. J. Non-Cryst. Solids 1993, 162, 68–80. [Google Scholar] [CrossRef]
- Coleman, D.J.; Jackson, S.D.; Golding, P.; King, T.A. Measurements of the spectroscopic and energy transfer parameters for Er3+-doped and Er3+, Pr3+-codoped PbO-Bi2O3-Ga2O3 glasses. J. Opt. Soc. Am. B 2002, 19, 2927–2937. [Google Scholar] [CrossRef]
- Dexter, D.L. A theory of sensitized luminescence in solids. J. Chem. Phys. 1953, 21, 836–850. [Google Scholar] [CrossRef]
- Förster, T. Intermolecular energy migration and fluorescence (Ger). Ann. Phys.(Leipzig) 1948, 2, 55–75. [Google Scholar] [CrossRef]
- Tian, Y.; Xu, R.; Hu, L.; Zhang, J. 2.7 μm fluorescence radiative dynamics and energy transfer between Er3+ and Tm3+ ions in fluoride glass under 800 nm and 980 nm excitation. J. Quant. Spectrosc. Radiat. Transf. 2012, 113, 87–95. [Google Scholar] [CrossRef]
- Miyakawa, T.; Dexter, D.L. Phonon sidebands, multiphonon relaxation of excited states, and phonon-assisted energy transfer between ions in solids. Phys. Rev. B 1970, 1, 2961–2969. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, X.; Yang, B.; Zhang, J.; Hu, L.; Zhang, L. Energy Transfer between Er3+ and Pr3+ for 2.7 μm Fiber Laser Material. Fibers 2014, 2, 24-33. https://doi.org/10.3390/fib2010024
Li X, Yang B, Zhang J, Hu L, Zhang L. Energy Transfer between Er3+ and Pr3+ for 2.7 μm Fiber Laser Material. Fibers. 2014; 2(1):24-33. https://doi.org/10.3390/fib2010024
Chicago/Turabian StyleLi, Xiangtan, Binhua Yang, Junjie Zhang, Lili Hu, and Liyan Zhang. 2014. "Energy Transfer between Er3+ and Pr3+ for 2.7 μm Fiber Laser Material" Fibers 2, no. 1: 24-33. https://doi.org/10.3390/fib2010024
APA StyleLi, X., Yang, B., Zhang, J., Hu, L., & Zhang, L. (2014). Energy Transfer between Er3+ and Pr3+ for 2.7 μm Fiber Laser Material. Fibers, 2(1), 24-33. https://doi.org/10.3390/fib2010024




