Poly (Lactic Acid)/Thermoplastic Starch Films: Effect of Cardoon Seed Epoxidized Oil on Their Chemicophysical, Mechanical, and Barrier Properties
Abstract
:1. Introduction
2. Experimental Section
2.1. Material and Chemicals
2.2. Epoxidation Process
2.3. Preparation of PLA-TPS-ECO Based Films
2.4. Attenuated Total Reflection Fourier Transform Infrared Spectroscopy (FTIR-ATR)
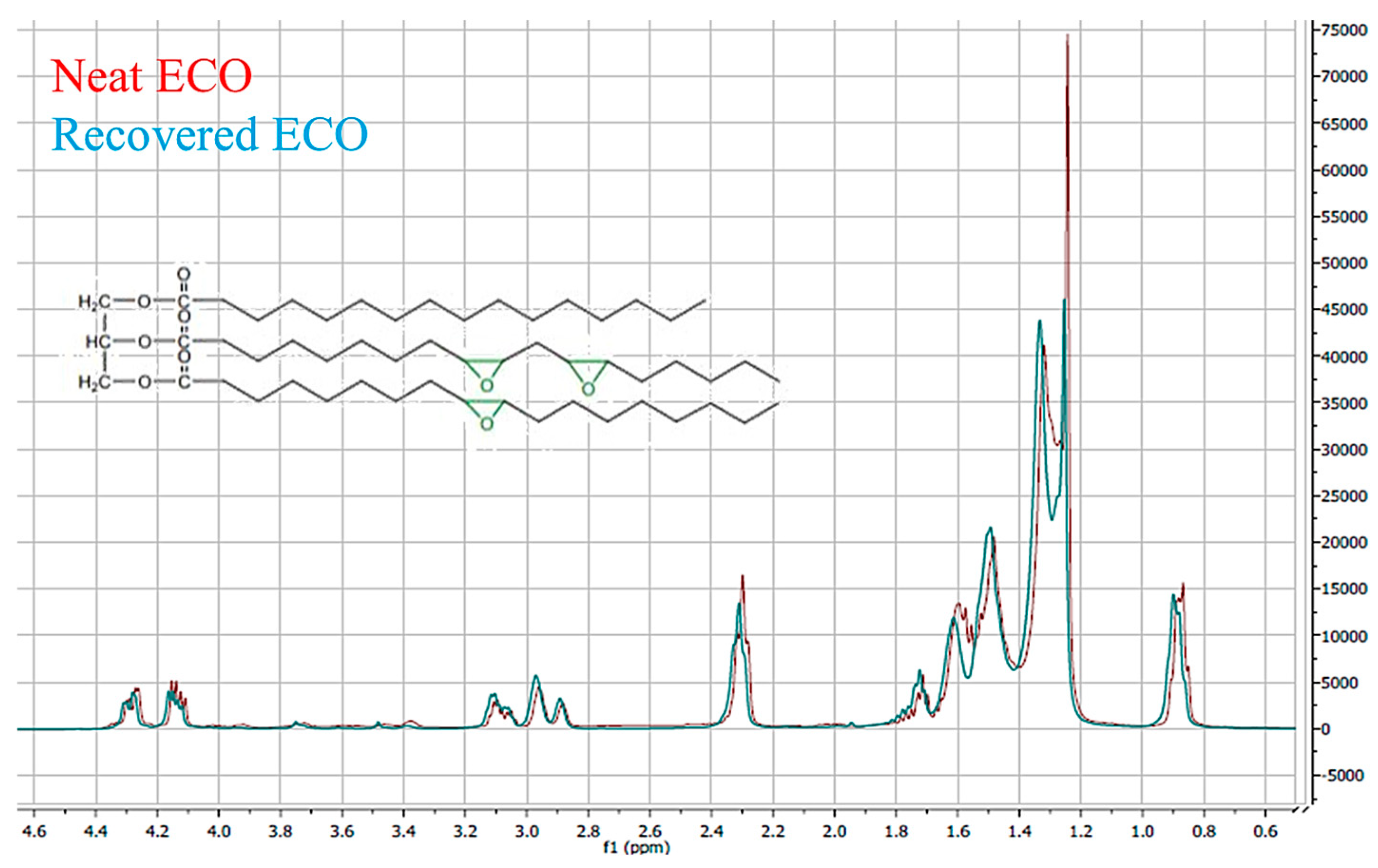
2.5. ECO Mass Evaluation by Solvent Extraction of Oily Phase from Films and 1H-NMR (Nuclear Magnetic Resonance) Analysis
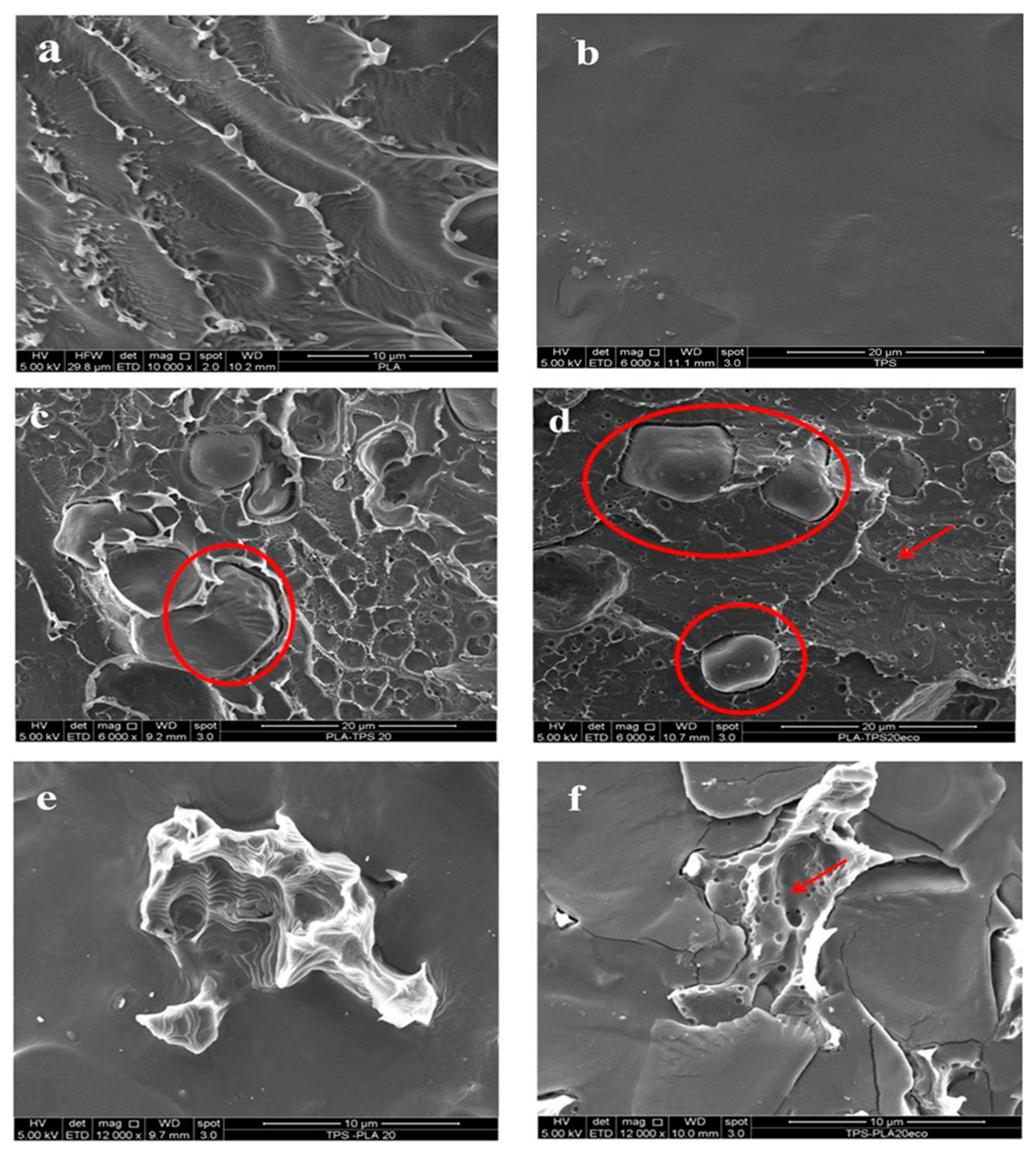
2.6. Scanning Electron Microscope (SEM)
2.7. Differential Scanning Calorimetry (DSC)
2.8. Thermogravimetric Analysis (TGA)
2.9. Film Thickness
2.10. Mechanical Properties
2.11. Permeability Tests
2.12. Surface Wettability: Water Contact Angle Measurements
2.13. UV/VIS Spectral Characterization of the Films
3. Results and Discussion
3.1. Epoxidation Process
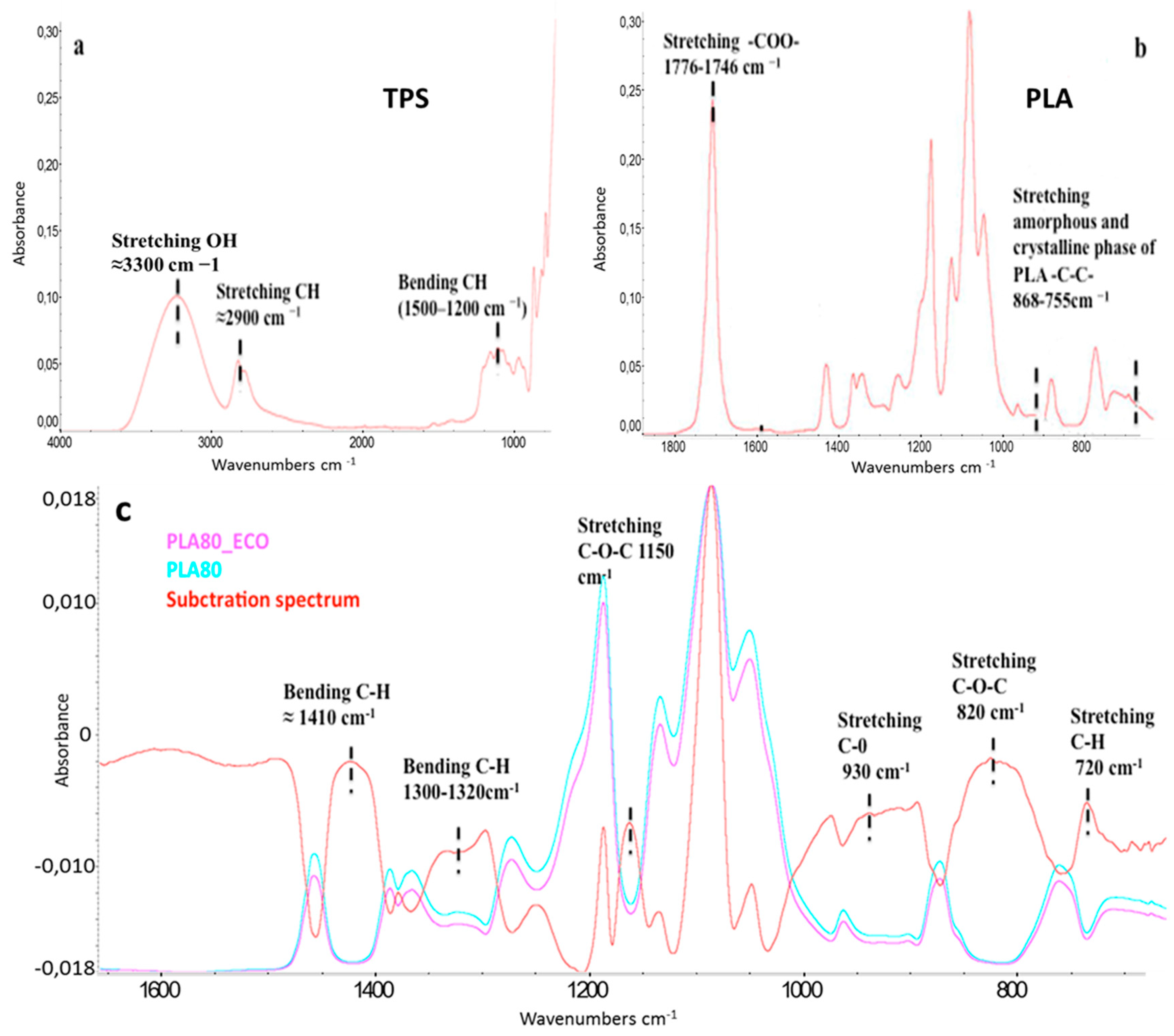
3.2. Structural, Quantitative and Morphological Analyses: Attenuated Total Reflection Fourier Transform Infrared Spectroscopy (FTIR-ATR), ECO Mass Evaluation, Nuclear Magnetic Resonance Spectroscopy (1H-NMR), Scanning Electron Microscopy (SEM)
3.3. Thermogravimetric Analysis (TGA)
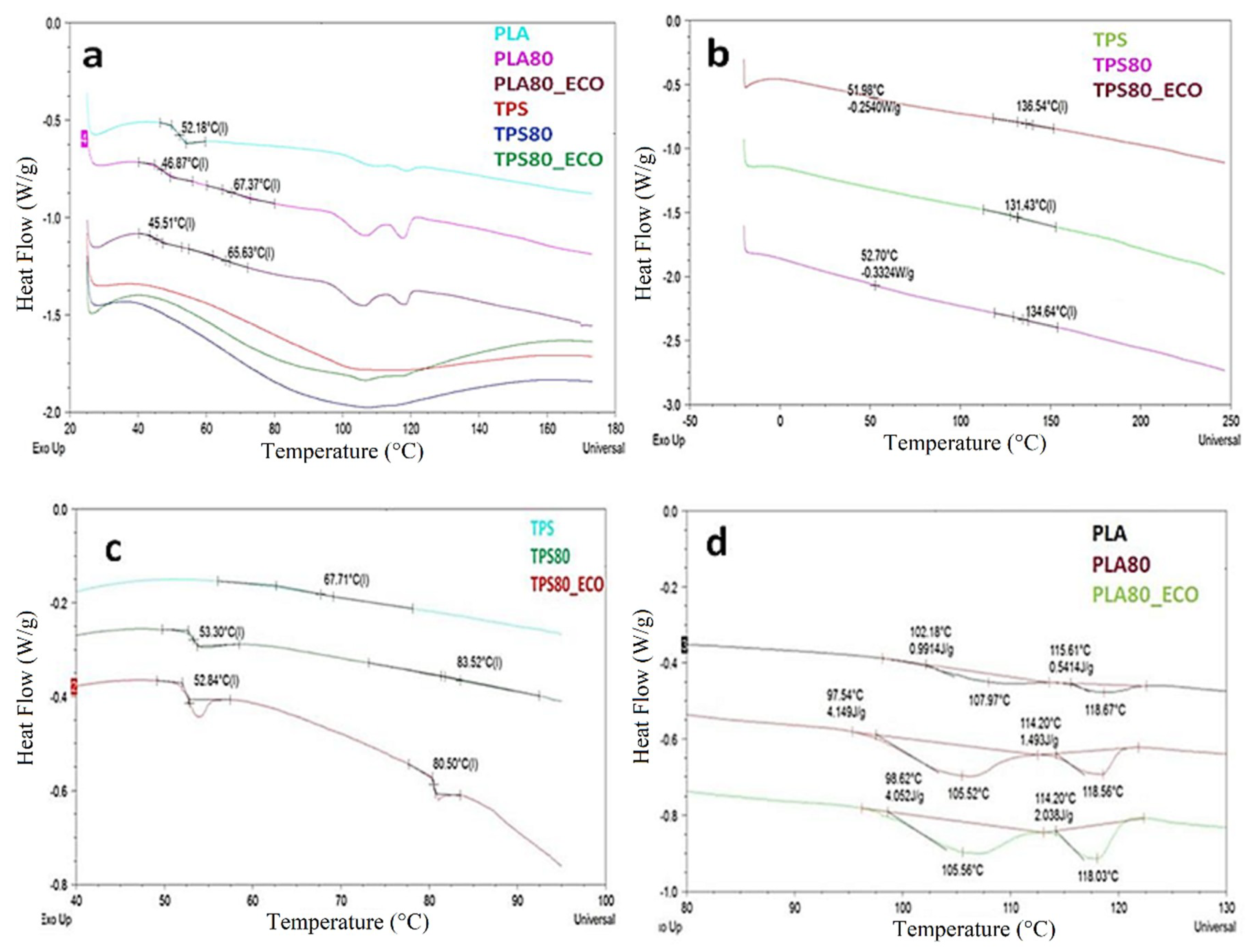
3.4. Differential Scanning Calorimetry (DSC)
3.5. Mechanical Properties (Tensile Test)
3.6. Water Vapor and Oxygen Permeability
3.7. Surface Wettability: Water Contact Angle Measurements
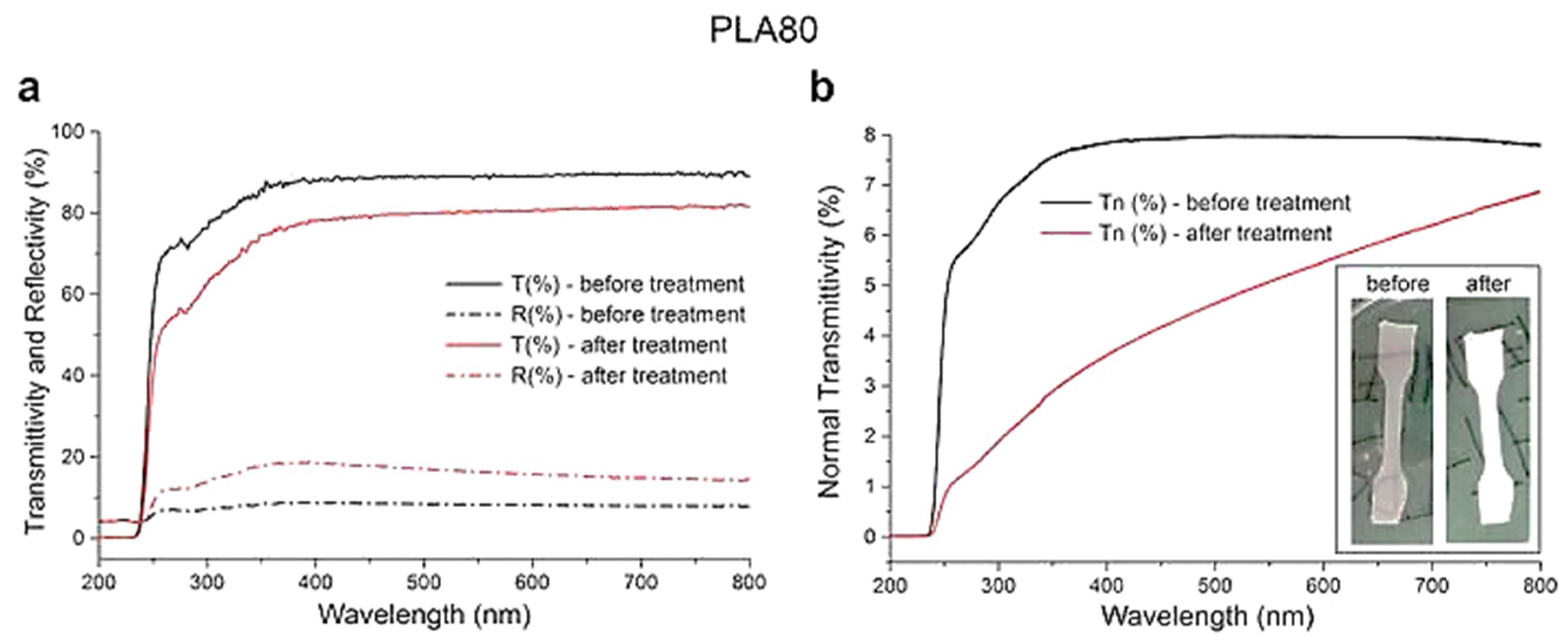
3.8. Spectral Characterization of the Films
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Europe, P. Plastics—The Facts 2016. An Analysis of European Plastics Production, Demand and Waste Data. 2016. Available online: https://www.plasticseurope.org/application/files/4315/1310/4805/plastic-the-fact-2016.pdf (accessed on 3 June 2019).
- Gregory, M.R. Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2013–2025. [Google Scholar] [CrossRef] [PubMed]
- Ren, X. Biodegradable plastics: A solution or a challenge? J. Clean. Prod. 2003, 11, 27–40. [Google Scholar] [CrossRef]
- Mormile, P.; Petti, L.; Rippa, M.; Immirzi, B.; Malinconico, M. Monitoring of the degradation dynamics of agricultural films by IR thermography. Polym. Degrad. Stab. 2006, 92, 777–784. [Google Scholar] [CrossRef]
- Luckachan, G.E.; Pillai, C.K.S. Biodegradable polymers—A review on recent trends and emerging perspectives. J. Polym. Environ. 2011, 19, 637–676. [Google Scholar] [CrossRef]
- Shen, K.; Haufe, J.; Patel, M.K. Product Overview and Market Projection of Emerging Bio-Based Plastics; Final Report; Utrecht University: Utrecht, The Netherlands, 2009. [Google Scholar]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Erdogan Orhan, I.; Sener, B.; Choudhary, M.I.; Khalid, A. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of some Turkish medicinal plants. J. Ethnopharmacol. 2004, 91, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.S.; Jardini, A.L.; Maciel Filho, R.J.P.E. Poly (Lactic Acid) production for tissue engineering applications. In Proceedings of the 20th International Congress of Chemical and Process Engineering CHISA, Prague, Czech Republic, 25–29 August 2012; pp. 1402–1413. [Google Scholar]
- Farah, S.; Anderson, D.G.; Langer, R. Physical and mechanical properties of PLA, and their functions in widespread applications, A comprehensive review. Adv. Drug Deliv. Rev. 2016, 107, 367–392. [Google Scholar] [CrossRef]
- Arrieta, M.; Dominici, F.; López, J.; Kenny, J. Bionanocomposite films based on plasticized PLA-PHB/cellulose nanocrystal blends. Carbohydr. Polym. 2015, 121, 265–275. [Google Scholar] [CrossRef]
- Lim, M.H.; Chin, J.E.; Ko, H.M.; Kim, I.C.; Lee, H.B.; Im, S.Y.; Bai, S. Simultaneous degradation of phytic acid and starch by an industrial strain of Saccharomyces cerevisiae producing phytase and alpha-amylase. Biotechnol. Lett. 2008, 30, 2125–2130. [Google Scholar] [CrossRef]
- Rasal, R.; Janorkar, A.; Hirt, D. Poly (lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Zhang, Y.; Rempel, C.; Liu, Q. Thermoplastic starch processing and characteristics—A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1353–1370. [Google Scholar] [CrossRef] [PubMed]
- Pushpadass, H.A.; Marx, D.B.; Hanna, M.A. Effects of extrusion temperature and plasticizers on the physical and functional properties of starch films. Starch Stärke 2008, 60, 527–538. [Google Scholar] [CrossRef]
- Ortega-Toro, R.; Santagata, G.; d’Ayala, G.; Cerruti, P.; Talens Oliag, P.; Amparo Chiralt Boix, M.; Malinconico, M. Enhancement of interfacial adhesion between starch and grafted poly (ε-caprolactone). Carbohydr. Polym. 2016, 147, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Collazo-Bigliardi, S.; Cuello, R.E.; Ortega-Toro, R. Epoxidised soybean oil addition into starch- and PLA-based biocomposites. Contemp. Eng. Sci. 2018, 11, 1953–1960. [Google Scholar] [CrossRef]
- Liminana, P.; Garcia-Sanoguera, D.; Quiles-Carrillo, L.; Balart, R.; Montanes, N. Optimization of maleinized linseed oil loading as a biobased compatibilizer in poly (butylene succinate) composites with almond shell flour. Materials 2019, 12, 685. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Liu, P.; Ji, N.; Hou, H.; Dong, H. Effects of various cross-linking agents on the physicochemical properties of starch/PHA composite films produced by extrusion blowing. Food Hydrocoll. 2017, 77, 964–975. [Google Scholar] [CrossRef]
- Ortega-Toro, R.; Collazo-Bigliardi, S.; Talens, P.; Chiralt, A. Thermoplastic starch: Improving their barrier properties. Agron. Colomb. 2016, 34, S73. [Google Scholar] [CrossRef]
- Ortega-Toro, R.; Bonilla, J.; Talens, P.; Chiralt, A. Future of starch-based materials in food packaging. In Starch-Based Materials in Food Packaging; Academic Press: Cambridge, UK, 2017; pp. 257–305. [Google Scholar] [CrossRef]
- Xiong, Z.; Li, C.; Ma, S.; Feng, J.; Yang, Y.; Zhang, R.; Zhu, J. The properties of poly (lactic acid)/starch blends with a functionalized plant oil: Tung oil anhydride. Carbohydr. Polym. 2013, 95, 77–84. [Google Scholar] [CrossRef]
- Phetwarotai, W.; Potiyaraj, P.; Aht-Ong, D. Characteristics of biodegradable polylactide/gelatinized starch films: Effects of starch, plasticizer, and compatibilizer. J. Appl. Polym. Sci. 2012, 126, e162–e172. [Google Scholar] [CrossRef]
- Orozco, V.; Brostow, W.; Chonkaew, W.; Lopez, B. Preparation and characterization of poly (lactic acid)-g-maleic anhydride + starch blends. Macromol. Symp. 2009, 277, 69–80. [Google Scholar] [CrossRef]
- Feng Zuo, Y.; Gu, J.; Qiao, Z.; Tan, H.; Cao, J.; Zhang, Y. Effects of dry method esterification of starch on the degradation characteristics of starch/polylactic acid composites. Int. J. Biol. Macromol. 2014, 72, 391–402. [Google Scholar] [CrossRef]
- Przybytek, A.; Sienkiewicz, M.; Kucińska-Lipka, J.; Janik, H. Preparation and characterization of biodegradable and compostable PLA/TPS/ESO compositions. Ind. Crop. Prod. 2018, 122, 375–383. [Google Scholar] [CrossRef]
- Rigoussen, A.; Raquez, J.-M.; Dubois, P.; Verge, P. A dual approach to compatibilize PLA/ABS immiscible blends with epoxidized cardanol derivatives. Eur. Polym. J. 2019, 114, 118–126. [Google Scholar] [CrossRef]
- Carbonell, A.; Samper, M.; Garcia, D.; Sanchez-Nacher, L.; Balart, R. Plasticization effect of epoxidized cottonseed oil (ECSO) on poly (lactic acid). Ind. Crop. Prod. 2017, 104, 278–286. [Google Scholar] [CrossRef]
- Belhassen, R.; Vilaseca, F.; Mutjé, P.; Boufi, S. Thermoplasticized starch modified by reactive blending with epoxidized soybean oil. Ind. Crop. Prod. 2014, 53, 261–267. [Google Scholar] [CrossRef]
- Masuda, T.; Goldsmith, P. World soybean production: Area harvested yield, and long-term projections. Int. Food Agribus. Man. Rev. 2009, 12, 143–162. [Google Scholar]
- Turco, R.; Pischetola, C.; Di Serio, M.; Vitiello, R.; Tesser, R.; Santacesaria, E. Selective epoxidation of soybean oil in the presence of H-Y zeolite. Ind. Eng. Chem. Res. 2017, 56, 7930–7936. [Google Scholar] [CrossRef]
- Turco, R.; Tesser, R.; Cucciolito, M.E.; Fagnano, M.; Ottaiano, L.; Mallardo, S.; Malinconico, M.; Santagata, G.; Di Serio, M. Cynara cardunculus biomass recovery: An eco-sustainable, nonedible resource of vegetable oil for the production of poly (lactic acid) bioplasticizers. ACS Sustain. Chem. Eng. 2019, 7, 4069–4077. [Google Scholar] [CrossRef]
- Gominho, J.; Curt, M.; Lourenço, A.; Fernández, J.; Pereira, H. Cynara cardunculus L. as a biomass and multi-purpose crop: A review of 30 years of research. Biomass Bioenergy 2018, 109, 257–275. [Google Scholar] [CrossRef]
- Toscano, V.; Sollima, L.; Genovese, C.; Melilli, M.G.; Raccuia, S.A. Pilot plant system for biodiesel and pellet production from cardoon: Technical and economic feasibility. Acta Hortic. 2016, 2016, 429–442. [Google Scholar] [CrossRef]
- Fernando, A.; Costa, J.; Barbosa, B.; Monti, A.; Rettenmaier, N. Environmental impact assessment of perennial crops cultivation on marginal soils in the Mediterranean Region. Biomass Bioenergy 2017. [Google Scholar] [CrossRef]
- Turco, R.; Vitiello, R.; Tesser, R.; Vergara, A.; Andini, S.; Di Serio, M. Niobium based catalysts for methyl oleate epoxidation reaction. Top. Catal. 2017, 60, 1054–1061. [Google Scholar] [CrossRef]
- ASTM-E96. American standard test methods for water vapour transmission of materials. Am. Soc. Test. Mater. 1993. [Google Scholar]
- Kizil, R.; Irudayaraj, J.; Seetharaman, K. Characterization of irradiated starches by using FT-Raman and FTIR spectroscopy. J. Agric. Food Chem. 2002, 50, 3912–3918. [Google Scholar] [CrossRef] [PubMed]
- Nibler, J.W. Infrared absorption spectroscopy. J. Chem. Educ. 1978, 55, A316. [Google Scholar] [CrossRef]
- Molinaro, S.; Boaro, M.; Sensidoni, A.; Lagazio, C.; Morris, M.; Kerry, J. Effect of nanoclay-type and PLA optical purity on the characteristics of PLA-based nanocomposite films. J. Food Eng. 2013, 117, 113–123. [Google Scholar] [CrossRef]
- Santagata, G.; Valerio, F.; Cimmino, A.; Dal Poggetto, G.; Masi, M.; Di Biase, M.; Malinconico, M.; Lavermicocca, P.; Evidente, A. Chemico-Physical and antifungal properties of poly (butylene succinate)/cavoxin blend: Study of a novel bioactive polymeric based system. Eur. Polym. J. 2017, 94, 230–247. [Google Scholar] [CrossRef]
- Tığlı Aydın, R.S.; Hazer, B.; Acar, M.; Gümüşderelioğlu, M. Osteogenic activities of polymeric soybean oil-g-polystyrene membranes. Polym. Bull. 2013, 70, 2065–2082. [Google Scholar] [CrossRef]
- Balart, J.F.; Fenollar, V.F.; Boronat, T.; Sánchez-Nacher, L. Processing and characterization of high environmental efficiency composites based on PLA and hazelnut shell flour (HSF) with biobased plasticizers derived from epoxidized linseed oil (ELO). Compos. Part B Eng. 2016, 86, 168–177. [Google Scholar] [CrossRef]
- Shin, B.Y.; Jang, S.H.; Kim, B.S. Thermal, morphological, and mechanical properties of biobased and biodegradable blends of poly (lactic acid) and chemically modified thermoplastic starch. Polym. Eng. Sci. 2011, 51, 826–834. [Google Scholar] [CrossRef]
- Ke, T.; Sun, S.X.; Seib, P. Blending of poly (lactic acid) and starches containing varying amylose content. J. Appl. Polym. Sci. 2003, 89, 3639–3646. [Google Scholar] [CrossRef]
- Martin, O.; Avérous, L. Poly (lactic acid): Plasticization and properties of biodegradable multiphase systems. Polymer 2001, 42, 6209–6219. [Google Scholar] [CrossRef]
- Akrami, M.; Ghasemi, I.; Azizi, H.; Karrabi, M.; Seyedabadi, M. A new approach in compatibilization of the poly(lacticacid)/thermoplastic starch (PLA/TPS) blends. Carbohydr. Polym. 2016, 144, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Huneault, M.; Li, H. Morphology and properties of compatibilized polylactide/thermoplastic starch blends. Polymer 2007, 48, 270–280. [Google Scholar] [CrossRef]
- Ferri, J.; Garcia, D.; Sanchez-Nacher, L.; Gimeno, O.; Balart, R. The effect of maleinized linseed oil (MLO) on mechanical performance of poly (lactic acid)-thermoplastic starch (PLA-TPS) blends. Carbohydr. Polym. 2016, 147, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Ferri, J.M.; Samper, M.D.; García-Sanoguera, D.; Reig, M.J.; Fenollar, O.; Balart, R. Plasticizing effect of biobased epoxidized fatty acid esters on mechanical and thermal properties of poly (lactic acid). J. Mater. Sci. 2016, 51, 5356–5366. [Google Scholar] [CrossRef]
- Ning, W.; Xingxiang, Z.; Na, H.; Jianming, F. Effects of water on the properties of thermoplastic starch poly(lactic acid) blend containing citric acid. J. Thermoplast. Compos. Mater. 2010, 23, 19–34. [Google Scholar] [CrossRef]
- Russo, R.; Malinconico, M.; Santagata, G. Effect of cross-linking with calcium ion on the physical properties of alginate films. Biomacromolecules 2007, 8, 3193–3197. [Google Scholar] [CrossRef]
- Bader, H.G.; Göritz, D. Investigations on high amylose corn starch films. Part 3: Stress strain behaviour. Starch Stärke 1994, 46, 435–439. [Google Scholar] [CrossRef]
- De Graaf, R.A.; Karman, A.P.; Janssen, L.P.B.M. Material properties and glass transition temperatures of different thermoplastic starches after extrusion processing. Starch Stärke 2003, 55, 80–86. [Google Scholar] [CrossRef]
- Santos, E.F.; Reiznautt, Q.B.; Samios, D.; Nachtigall, S.M.B. Sunflower-oil biodiesel oligoesters/polylactide blends: Plasticizing effect and ageing. Polym. Test. 2014, 39, 23–29. [Google Scholar] [CrossRef]
- Li, H.; Huneault, M.A. Effect of nucleation and plasticization on the crystallization of poly (lactic acid). Polymer 2007, 48, 6855–6866. [Google Scholar] [CrossRef]
- Wurzburg, O.B. Modified Starches: Properties and Uses; CRC Press: Boca Raton, FL, USA, 1986. [Google Scholar]
- Wang, N.; Yu, J.; Chang, P.R.; Ma, X. Influence of citric acid on the properties of glycerol-plasticized dry starch (DTPS) and DTPS/poly (lactic acid) blends. Starch Stärke 2007, 59, 409–417. [Google Scholar] [CrossRef]
- Nešić, A.; Onjia, A.; Davidović, S.; Dimitrijević, S.; Errico, M.E.; Santagata, G.; Malinconico, M. Design of pectin-sodium alginate based films for potential healthcare application: Study of chemico-physical interactions between the components of films and assessment of their antimicrobial activity. Carbohydr. Polym. 2017, 157, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.O.; Pires, A.T.N.; Yamashita, F. Characterization of thermoplastic starch/poly (lactic acid) blends obtained by extrusion and thermopressing. J. Braz. Chem. Soc. 2012, 23, 426–434. [Google Scholar] [CrossRef]
- Cerruti, P.; Laurienzo, P.; Malinconico, M.; Carfagna, C. Thermal oxidative stability and effect of water on gas transport and mechanical properties in PA6-EVOH films. J. Polym. Sci. Part B Polym. Phys. 2007, 45, 840–849. [Google Scholar] [CrossRef]
- Hertlein, J.; Singh, R.; Weisser, H. Prediction of oxygen transport parameters of plastic packaging materials from transient state measurements. J. Food Eng. 1995, 24, 543–560. [Google Scholar] [CrossRef]
- Zhang, Z.; Lim, L.-T.; Tung, M.A. Limonene transport and mechanical properties of EVOH and nylon 6,6 films as influenced by RH. J. Appl. Polym. Sci. 2001, 79, 1949–1957. [Google Scholar] [CrossRef]
- Carlo, B. Sviluppo di Blend Polimerici per il Miglioramento delle Proprieta’ Barriera di Polimeri per il Packaging Alimentare; University of Padova: Padova, Italy, 2012. [Google Scholar]
- Shieh, Y.-T.; Liu, G.-L. Temperature-modulated differential scanning calorimetry studies on the origin of double melting peaks in isothermally melt-crystallized poly (L-lactic acid). J. Polym. Sci. Part B Polym. Phys. 2007, 45, 466–474. [Google Scholar] [CrossRef]
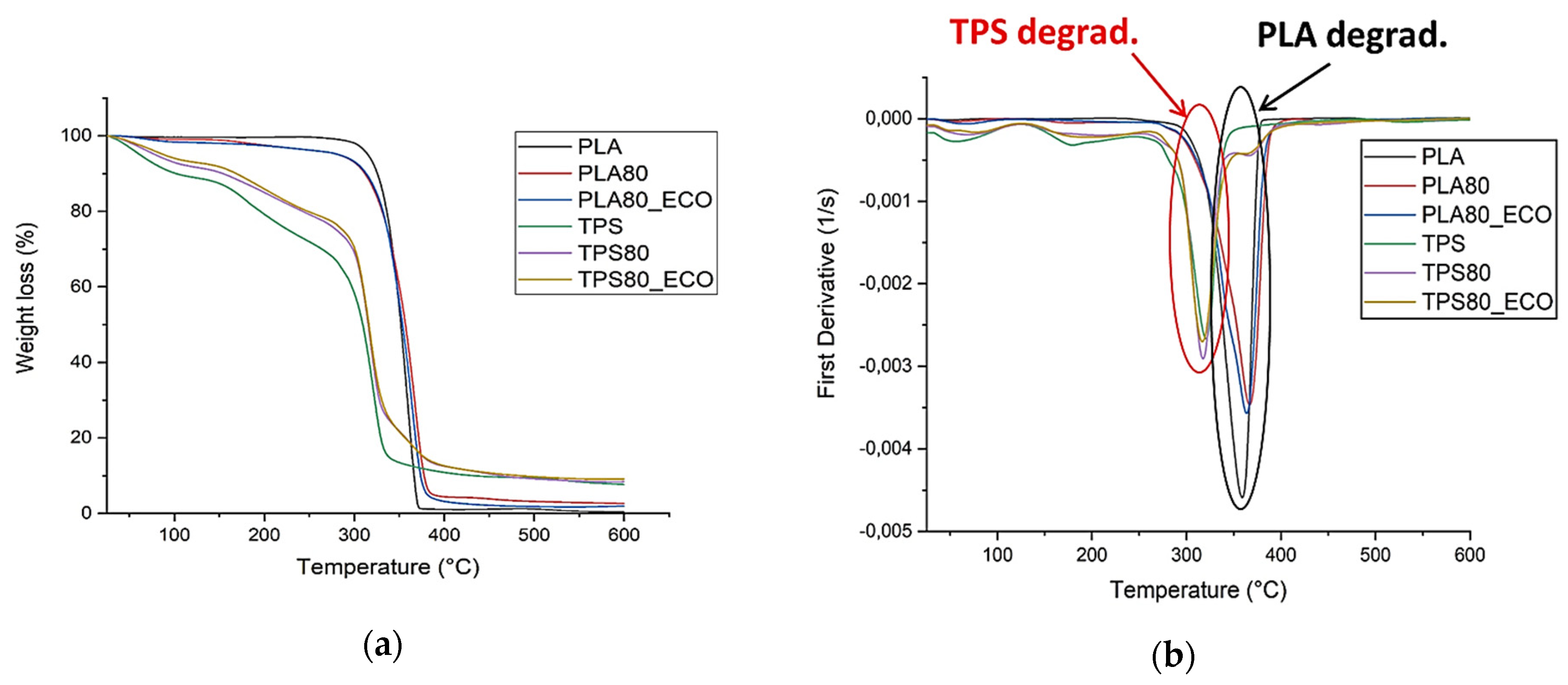
| Data/Samples | Tonset (°C) | TlossH2O (°C) Weakly Bound | TlossH2O (°C) Strongly Bound | Tdecomp (°C) Glycerol | Tpeak (°C) |
|---|---|---|---|---|---|
| PLA | 290 | – | – | – | 360 |
| PLA80 | 280 | – | – | – | 367 |
| PLA80_ECO | 275 | – | – | – | 365 |
| TPS | 260 | 25 < T < 80 | 80 < T < 180 | 200 < T < 260 | 320 |
| TPS80 | 280 | 25 < T < 80 | 80 < T < 180 | 200 < T < 260 | 318 |
| TPS80_ECO | 280 | 25 < T < 80 | 80 < T < 180 | 200 < T < 260 | 318 |
| Samples/Data | Tg PLA (°C) | Tg TPS (°C) | ΔHm (J/g) | Tmα (°C) | Tmα’ (°C) |
|---|---|---|---|---|---|
| PLA | 52 | – | 1.5 | 108 | 119 |
| PLA80 | 47 | 67 | 5.6 | 105 | 118 |
| PLA80_ECO | 45 | 65 | 6.1 | 105 | 118 |
| TPS | – | 67 | – | – | – |
| TPS80 | 53 | 83 | – | – | – |
| TPS80_ECO | 53 | 80 | – | – | – |
| Sample/Data | Young Modulus (MPa) | Stress at Break (MPa) | Strain at Break (%) |
|---|---|---|---|
| PLA | 1329 ± 25 | 50.5 ± 4.7 | 4 ± 0.5 |
| PLA80 | 1285 ± 33 | 27.4 ± 0.8 | 3.1 ± 0.7 |
| PLA80_ECO | 1135 ± 31 | 24.4 ± 0.5 | 4.6 ± 0.5 |
| TPS | 77 ± 15 | 5.4 ± 0.9 | 51 ± 3 |
| TPS80 | 150 ± 15 | 5.5 ± 0.7 | 19 ± 4 |
| TPS80_ECO | 158 ± 12 | 3.6 ± 0.5 | 7 ± 1.2 |
| Samples | WVP (ng/m·Pa·s) × 10−3 ± 10% | O2P (cm3/m·s·Pa) | D (m2/s) × 10−6 | S (cm3/m3·Pa) | CA (deg) | ST (dy/cm) |
|---|---|---|---|---|---|---|
| PLA | 0.025 | 23.3 ± 6.7 × 10−1 | 2.2 ± 2.6 × 10−7 | 0.12 ± 0.01 | 73 ± 2 | 45 ± 5 |
| PLA80 | 0.11 | 61.1 ± 1.7 | 2.2 ± 3.1 × 10−7 | 0.32 ± 0.01 | 64 ± 3 | 58 ± 3 |
| PLA80_ECO | 0.038 | 23.4 ± 6.7 × 10−1 | 4.0 ± 1.6 × 10−7 | 0.07 ± 0.0013 | 60 ± 2 | 74 ± 2 |
| TPS | 1.3 | – | – | – | – | – |
| TPS80 | 1.1 | – | – | – | – | – |
| TPS80_ECO | 1.0 | – | – | – | – | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turco, R.; Ortega-Toro, R.; Tesser, R.; Mallardo, S.; Collazo-Bigliardi, S.; Chiralt Boix, A.; Malinconico, M.; Rippa, M.; Di Serio, M.; Santagata, G. Poly (Lactic Acid)/Thermoplastic Starch Films: Effect of Cardoon Seed Epoxidized Oil on Their Chemicophysical, Mechanical, and Barrier Properties. Coatings 2019, 9, 574. https://doi.org/10.3390/coatings9090574
Turco R, Ortega-Toro R, Tesser R, Mallardo S, Collazo-Bigliardi S, Chiralt Boix A, Malinconico M, Rippa M, Di Serio M, Santagata G. Poly (Lactic Acid)/Thermoplastic Starch Films: Effect of Cardoon Seed Epoxidized Oil on Their Chemicophysical, Mechanical, and Barrier Properties. Coatings. 2019; 9(9):574. https://doi.org/10.3390/coatings9090574
Chicago/Turabian StyleTurco, Rosa, Rodrigo Ortega-Toro, Riccardo Tesser, Salvatore Mallardo, Sofia Collazo-Bigliardi, Amparo Chiralt Boix, Mario Malinconico, Massimo Rippa, Martino Di Serio, and Gabriella Santagata. 2019. "Poly (Lactic Acid)/Thermoplastic Starch Films: Effect of Cardoon Seed Epoxidized Oil on Their Chemicophysical, Mechanical, and Barrier Properties" Coatings 9, no. 9: 574. https://doi.org/10.3390/coatings9090574
APA StyleTurco, R., Ortega-Toro, R., Tesser, R., Mallardo, S., Collazo-Bigliardi, S., Chiralt Boix, A., Malinconico, M., Rippa, M., Di Serio, M., & Santagata, G. (2019). Poly (Lactic Acid)/Thermoplastic Starch Films: Effect of Cardoon Seed Epoxidized Oil on Their Chemicophysical, Mechanical, and Barrier Properties. Coatings, 9(9), 574. https://doi.org/10.3390/coatings9090574