Epidermal Gland Inspired Self-Repairing Slippery Lubricant-Infused Porous Coatings with Durable Low Ice Adhesion
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Preparation of Hybrid Precursors
2.3. Preparation of Icephobic SLIPS Coatings
2.4. Characterization
3. Results and Discussion
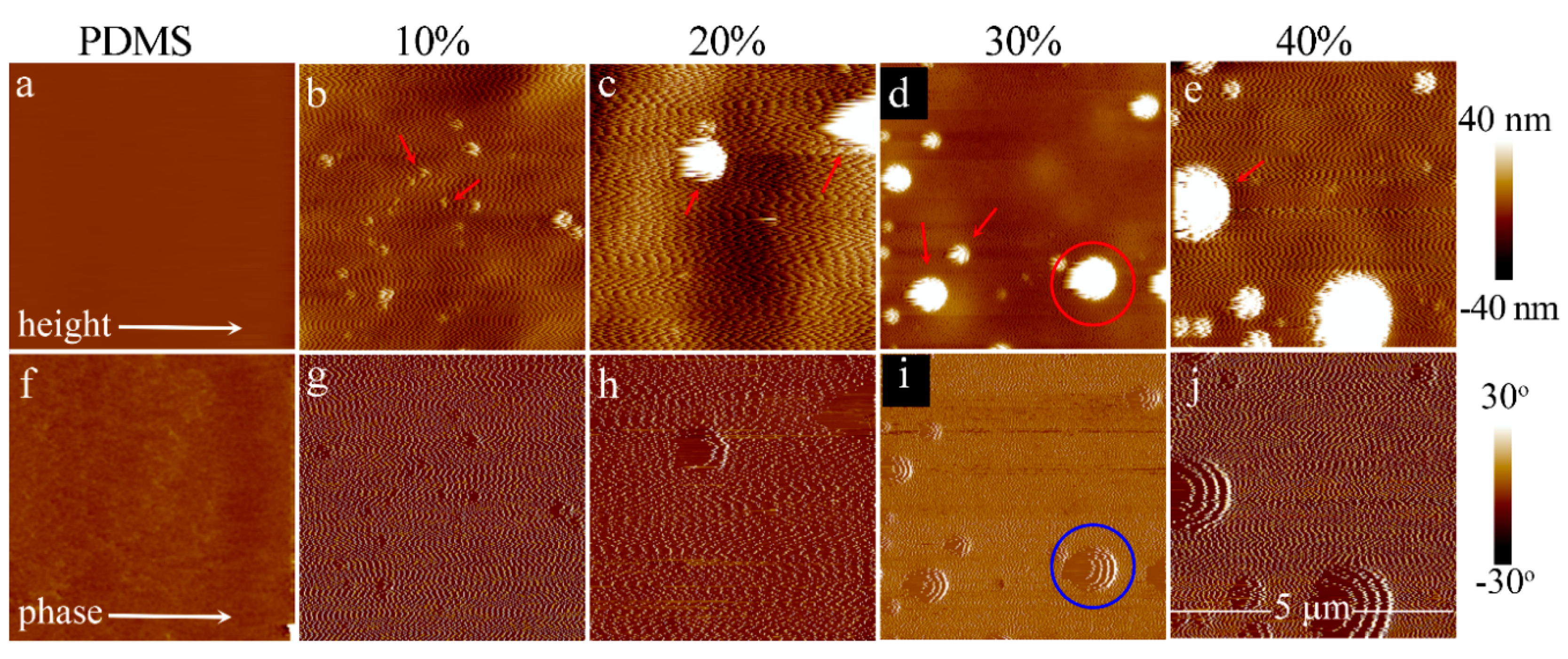
3.1. Morphology and Structure
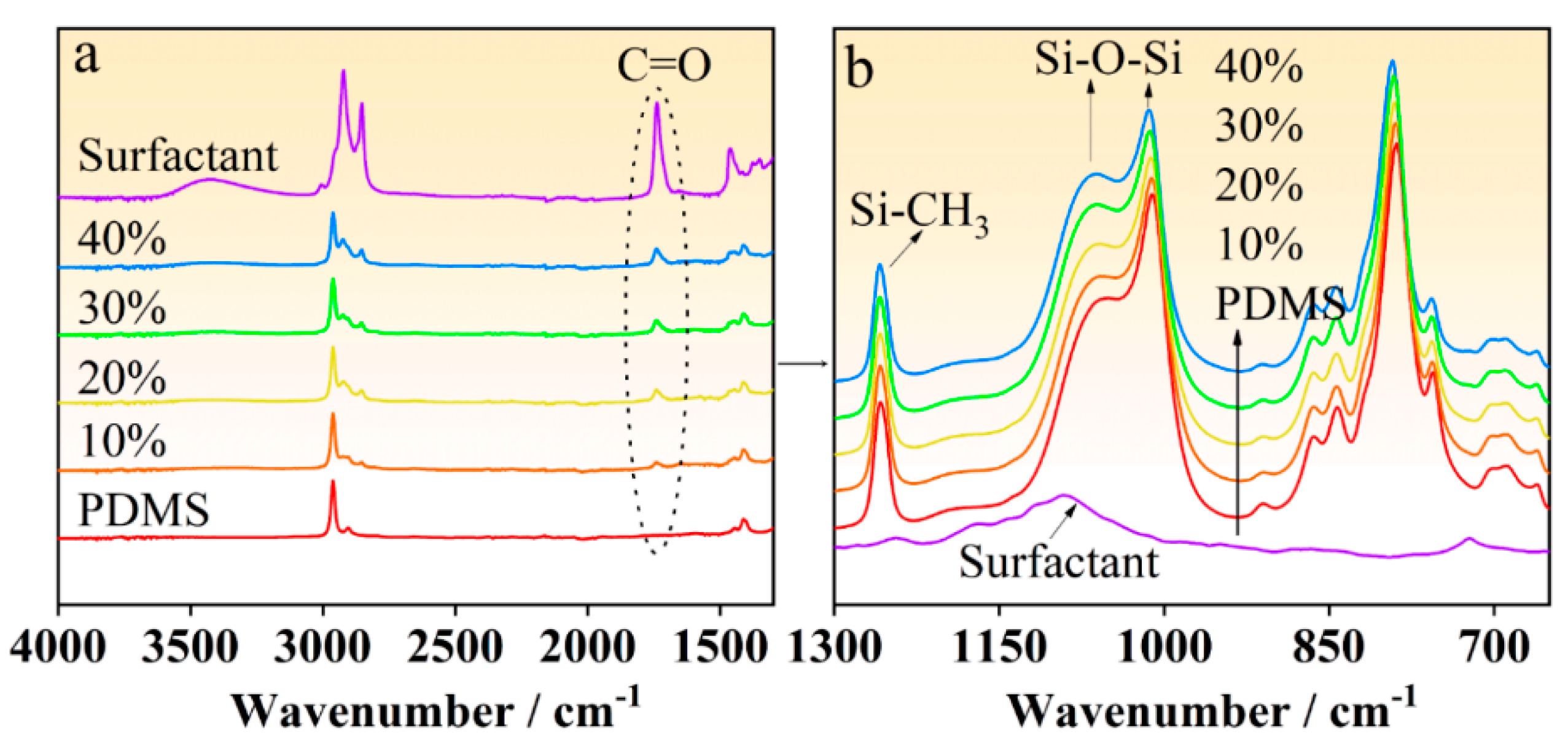
3.2. Chemical Composition
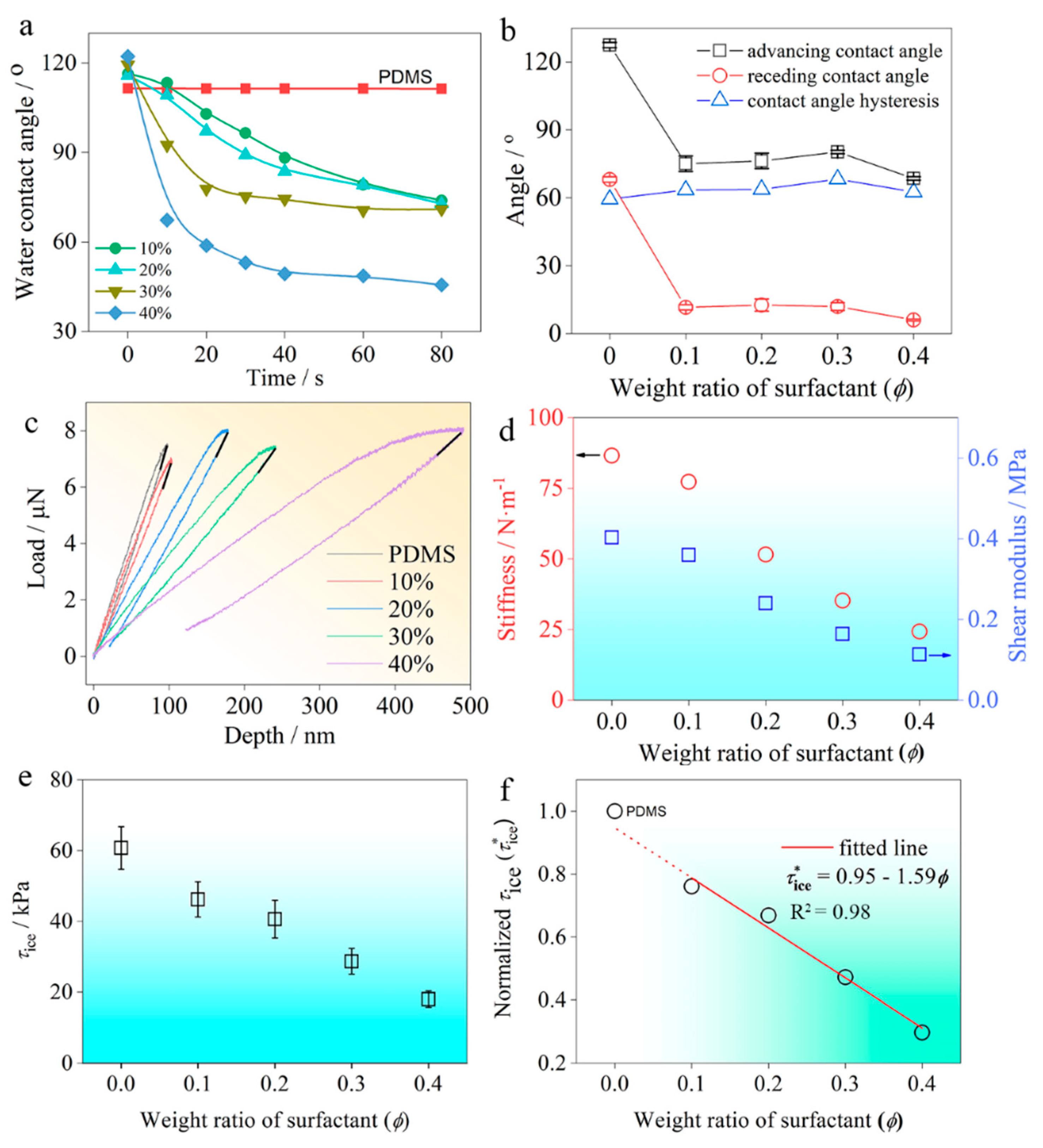
3.3. Self-Repairing Property
3.4. Wettability, Mechanical Properties and Ice Adhesion Strength
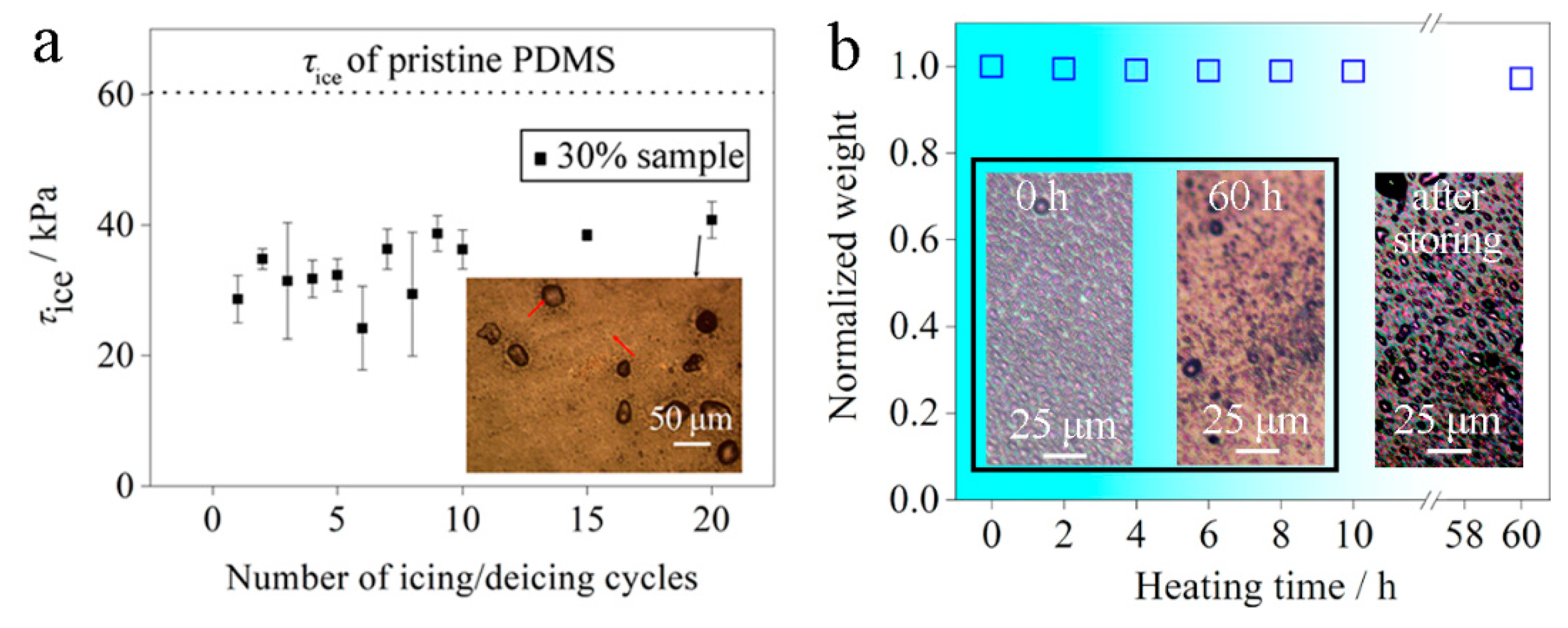
3.5. Icephobic Durability
3.6. Thermal Durability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- He, Z.; Vagenes, E.T.; Delabahan, C.; He, J.; Zhang, Z. Room Temperature Characteristics of Polymer-Based Low Ice Adhesion Surfaces. Sci. Rep. 2017, 7, 42181. [Google Scholar] [CrossRef] [PubMed]
- Kreder, M.J.; Alvarenga, J.; Kim, P.; Aizenberg, J. Design of anti-icing surfaces: Smooth, textured or slippery? Nat. Rev. Mater. 2016, 1, 15003. [Google Scholar] [CrossRef]
- Shen, Y.Z.; Wu, X.H.; Tao, J.; Zhu, C.L.; Lai, Y.K.; Chen, Z. Icephobic materials: Fundamentals, performance evaluation, and applications. Prog. Mater. Sci. 2019, 103, 509–557. [Google Scholar] [CrossRef]
- Rønneberg, S.; He, J.; Zhang, Z. The Need for Standards in Low Ice Adhesion Surface Research: A Critical Review. J. Adhes. Sci. Technol. 2019. accepted. [Google Scholar]
- Sojoudi, H.; Wang, M.; Boscher, N.D.; McKinley, G.H.; Gleason, K.K. Durable and scalable icephobic surfaces: Similarities and distinctions from superhydrophobic surfaces. Soft Matter 2016, 12, 1938–1963. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.Y.; Zhang, Y.; Wang, D.A.; Liu, Z.L.; Liu, Y.P.; Pei, X.W.; Yu, B.; Zhou, F. Integration of Self-Lubrication and Near-Infrared Photothermogenesis for Excellent Anti-Icing/Deicing Performance. Adv. Funct. Mater. 2015, 25, 4237–4245. [Google Scholar] [CrossRef]
- Zhang, S.; Huang, J.; Cheng, Y.; Yang, H.; Chen, Z.; Lai, Y. Bioinspired Surfaces with Superwettability for Anti-Icing and Ice-Phobic Application: Concept, Mechanism, and Design. Small 2017, 13, 701867. [Google Scholar] [CrossRef] [PubMed]
- Guadarrama-Cetina, J.; Mongruel, A.; González-Viñas, W.; Beysens, D. Frost formation with salt. EPL (Europhys. Lett.) 2015, 110, 56002. [Google Scholar] [CrossRef]
- Sun, X.; Damle, V.G.; Uppal, A.; Linder, R.; Chandrashekar, S.; Mohan, A.R.; Rykaczewski, K. Inhibition of Condensation Frosting by Arrays of Hygroscopic Antifreeze Drops. Langmuir 2015, 31, 13743–13752. [Google Scholar] [CrossRef]
- Li, T.; Zhuo, Y.; Håkonsen, V.; He, J.; Zhang, Z. Durable low ice adhesion foams modulated by submicron pores. Ind. Eng. Chem. Res. 2019. [Google Scholar] [CrossRef]
- Golovin, K.; Dhyani, A.; Thouless, M.D.; Tuteja, A. Low-interfacial toughness materials for effective large-scale deicing. Science 2019, 364, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, R.; Beysens, D.; Anand, S. Delaying Ice and Frost Formation Using Phase-Switching Liquids. Adv. Mater. 2019, 31, 1807812. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Liu, B.; Peng, J.; Zhu, K.; Zhao, Y.; Li, X.; Yuan, X. Icephobic Durability of Branched PDMS Slippage Coatings Co-Cross-Linked by Functionalized POSS. ACS Appl. Mater. Interfaces 2019, 11, 4654–4666. [Google Scholar] [CrossRef] [PubMed]
- Golovin, K.; Kobaku, S.P.; Lee, D.H.; DiLoreto, E.T.; Mabry, J.M.; Tuteja, A. Designing durable icephobic surfaces. Sci. Adv. 2016, 2, e1501496. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Yao, X.; Zheng, Y.; Wang, J.; Jiang, L. Antiadhesion Organogel Materials: From Liquid to Solid. Adv. Mater. 2017, 29, 1703032. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Håkonsen, V.; He, Z.; Xiao, S.; He, J.; Zhang, Z. Enhancing the mechanical durability of icephobic surfaces by introducing autonomous self-healing function. ACS Appl. Mater. Interfaces 2018, 10, 11972–11978. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, K.; Li, K.; Gutowski, V.; Yin, Y.; Wang, J. Fabrication of Anti-Icing Surfaces by Short α-Helical Peptides. ACS Appl. Mater. Interfaces 2018, 10, 1957–1962. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Xiao, S.; Gao, H.; He, J.; Zhang, Z. Multiscale crack initiator promoted super-low ice adhesion surfaces. Soft Matter 2017, 13, 6562–6568. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhuo, Y.; He, J.; Zhang, Z. Design and preparation of sandwich-like polydimethylsiloxane (PDMS) sponges with super-low ice adhesion. Soft Matter 2018, 14, 4846–4851. [Google Scholar] [CrossRef] [PubMed]
- Bahadur, V.; Mishchenko, L.; Hatton, B.; Taylor, J.A.; Aizenberg, J.; Krupenkin, T. Predictive model for ice formation on superhydrophobic surfaces. Langmuir 2011, 27, 14143–14150. [Google Scholar] [CrossRef]
- Yamazaki, T.; Tenjimbayashi, M.; Manabe, K.; Moriya, T.; Nakamura, H.; Nakamura, T.; Matsubayashi, T.; Tsuge, Y.; Shiratori, S. Antifreeze Liquid-Infused Surface with High Transparency, Low Ice Adhesion Strength, and Antifrosting Properties Fabricated through a Spray Layer-by-Layer Method. Ind. Eng. Chem. Res. 2019, 58, 2225–2234. [Google Scholar] [CrossRef]
- Cao, M.; Guo, D.; Yu, C.; Li, K.; Liu, M.; Jiang, L. Water-Repellent Properties of Superhydrophobic and Lubricant-Infused “Slippery” Surfaces: A Brief Study on the Functions and Applications. ACS Appl. Mater. Interfaces 2016, 8, 3615–3623. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Gong, Q.H.; Zhan, S.H.; Jiang, L.; Zheng, Y.M. Robust Anti-Icing Performance of a Flexible Superhydrophobic Surface. Adv. Mater. 2016, 28, 7729–7735. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.Z.; Wang, G.Y.; Tao, J.; Zhu, C.L.; Liu, S.Y.; Jin, M.M.; Xie, Y.H.; Chen, Z. Anti-Icing Performance of Superhydrophobic Texture Surfaces Depending on Reference Environments. Adv. Mater. Interfaces 2017, 4, 1700836. [Google Scholar] [CrossRef]
- Xing, Y.; Du, X.; Li, X.; Huang, H.; Li, J.; Wen, Y.; Zhang, X. Tunable dendrimer-like porous silica nanospheres: Effects of structures and stacking manners on surface wettability. J. Alloy. Compd. 2018, 732, 70–79. [Google Scholar] [CrossRef]
- Varanasi, K.K.; Deng, T.; Smith, J.D.; Hsu, M.; Bhate, N. Frost formation and ice adhesion on superhydrophobic surfaces. Appl. Phys. Lett. 2010, 97, 234102. [Google Scholar] [CrossRef]
- Nosonovsky, M.; Hejazi, V. Why superhydrophobic surfaces are not always icephobic. ACS Nano 2012, 6, 8488–8491. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Jeon, J.; Heu, C.S.; Kim, D.R. Three-Dimensionally Programmed Slippery Wrinkles with High Stretchability for Tunable Functionality of Icephobicity and Effective Water Harvesting. Adv. Mater. Interfaces 2018, 5, 1800980. [Google Scholar] [CrossRef]
- Niemela-Anttonen, H.; Koivuluoto, H.; Tuominen, M.; Teisala, H.; Juuti, P.; Haapanen, J.; Harra, J.; Stenroos, C.; Lahti, J.; Kuusipalo, J.; et al. Icephobicity of Slippery Liquid Infused Porous Surfaces under Multiple Freeze-Thaw and Ice Accretion-Detachment Cycles. Adv. Mater. Interfaces 2018, 5, 1800828. [Google Scholar] [CrossRef]
- Wong, T.-S.; Kang, S.H.; Tang, S.K.Y.; Smythe, E.J.; Hatton, B.D.; Grinthal, A.; Aizenberg, J. Bioinspired self-repairing slippery surfaces with pressure-stable omniphobicity. Nature 2011, 477, 443. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.D.; Dhiman, R.; Anand, S.; Reza-Garduno, E.; Cohen, R.E.; McKinley, G.H.; Varanasi, K.K. Droplet mobility on lubricant-impregnated surfaces. Soft Matter 2013, 9, 1772–1780. [Google Scholar] [CrossRef]
- Coady, M.J.; Wood, M.; Wallace, G.Q.; Nielsen, K.E.; Kietzig, A.M.; Lagugne-Labarthet, F.; Ragogna, P.J. Icephobic behavior of UV-Cured polymer networks incorporated into slippery lubricant-infused porous surfaces: Improving SLIPS durability. ACS Appl. Mater. Interfaces 2018, 10, 2890–2896. [Google Scholar] [CrossRef] [PubMed]
- Wu, L. Modelling and simulation of the lubricant depletion process induced by laser heating in heat-assisted magnetic recording system. Nanotechnology 2007, 18, 215702. [Google Scholar] [CrossRef]
- Zhu, X.; Lu, J.; Li, X.; Wang, B.; Song, Y.; Miao, X.; Wang, Z.; Ren, G. Simple Way to a Slippery Lubricant Impregnated Coating with Ultrastability and Self-Replenishment Property. Ind. Eng. Chem. Res. 2019, 58, 8148–8153. [Google Scholar] [CrossRef]
- Zheng, S.; Bellido-Aguilar, D.A.; Wu, X.; Zhan, X.; Huang, Y.; Zeng, X.; Zhang, Q.; Chen, Z. Durable Waterborne Hydrophobic Bio-Epoxy Coating with Improved Anti-Icing and Self-Cleaning Performance. ACS Sustain. Chem. Eng. 2019, 7, 641–649. [Google Scholar] [CrossRef]
- Zhang, K.Q.; Li, X.H.; Zhao, Y.H.; Zhu, K.Y.; Li, Y.C.; Tao, C.; Yuan, X.Y. UV-curable POSS-fluorinated methacrylate diblock copolymers for icephobic coatings. Prog. Org. Coat. 2016, 93, 87–96. [Google Scholar] [CrossRef]
- Yeong, Y.H.; Wang, C.; Wynne, K.J.; Gupta, M.C. Oil-Infused Superhydrophobic Silicone Material for Low Ice Adhesion with Long-Term Infusion Stability. ACS Appl. Mater. Interfaces 2016, 8, 32050–32059. [Google Scholar] [CrossRef]
- Zhuo, Y.; Wang, F.; Xiao, S.; Zhang, Z.; He, J. One-Step Fabrication of Bioinspired Lubricant-Regenerable Icephobic Slippery Liquid-Infused Porous Surfaces (SLIPS). ACS Omega 2018, 3, 10139–10144. [Google Scholar] [CrossRef]
- Zhao, H.; Sun, Q.; Deng, X.; Cui, J. Earthworm-Inspired Rough Polymer Coatings with Self-Replenishing Lubrication for Adaptive Friction-Reduction and Antifouling Surfaces. Adv. Mater. 2018, 30, e1802141. [Google Scholar] [CrossRef]
- Federle, W.; Barnes, W.J.P.; Baumgartner, W.; Drechsler, P.; Smith, J.M. Wet but not slippery: Boundary friction in tree frog adhesive toe pads. J. R. Soc. Interface 2006, 3, 689–697. [Google Scholar] [CrossRef]
- Cui, J.; Daniel, D.; Grinthal, A.; Lin, K.; Aizenberg, J. Dynamic polymer systems with self-regulated secretion for the control of surface properties and material healing. Nat. Mater. 2015, 14, 790. [Google Scholar] [CrossRef] [PubMed]
- Li, J.S.; Ueda, E.; Paulssen, D.; Levkin, P.A. Slippery Lubricant-Infused Surfaces: Properties and Emerging Applications. Adv. Funct. Mater. 2019, 29, 1802317. [Google Scholar] [CrossRef]
- Sun, X.; Damle, V.G.; Liu, S.; Rykaczewski, K. Bioinspired Stimuli-Responsive and Antifreeze-Secreting Anti-Icing Coatings. Adv. Mater. Interfaces 2015, 2, 1400479. [Google Scholar] [CrossRef]
- Tenjimbayashi, M.; Nishioka, S.; Kobayashi, Y.; Kawase, K.; Li, J.; Abe, J.; Shiratori, S. A Lubricant-Sandwiched Coating with Long-Term Stable Anticorrosion Performance. Langmuir 2018, 34, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Urata, C.; Dunderdale, G.J.; England, M.W.; Hozumi, A. Self-lubricating organogels (SLUGs) with exceptional syneresis-induced anti-sticking properties against viscous emulsions and ices. J. Mater. Chem. A 2015, 3, 12626–12630. [Google Scholar] [CrossRef]
- Flory, P.J. Thermodynamics of High Polymer Solutions. J. Chem. Phys. 1942, 10, 51–61. [Google Scholar] [CrossRef]
- Rønneberg, S.; Laforte, C.; Volat, C.; He, J.; Zhang, Z. The effect of ice type on ice adhesion. AIP Adv. 2019, 9, 055304. [Google Scholar] [CrossRef]
- Beemer, D.L.; Wang, W.; Kota, A.K. Durable gels with ultra-low adhesion to ice. J. Mater. Chem. A 2016, 4, 18253–18258. [Google Scholar] [CrossRef]
- Subramanyam, S.B.; Rykaczewski, K.; Varanasi, K.K. Ice adhesion on lubricant-impregnated textured surfaces. Langmuir 2013, 29, 13414–13418. [Google Scholar] [CrossRef]
- Zhou, W.D.; Zeng, Y.; Liu, B.; Yu, S.K.; Hua, W.; Huang, X.Y. Evaporation of Polydisperse Perfluoropolyether Lubricants in Heat-Assisted Magnetic Recording. Appl. Phys. Express 2011, 4, 095201. [Google Scholar] [CrossRef]


| Samples | Stiffness (N/m) | Reduced Modulus (MPa) | Young’s Modulus (MPa) | Apparent Shear Modulus (MPa) |
|---|---|---|---|---|
| PDMS: 0% | 86.6 | 1.613 | 1.210 | 0.403 |
| 10% | 77.3 | 1.440 | 1.080 | 0.360 |
| 20% | 51.6 | 0.960 | 0.720 | 0.240 |
| 30% | 35.2 | 0.656 | 0.492 | 0.164 |
| 40% | 24.3 | 0.453 | 0.340 | 0.113 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, T.; Zhuo, Y.; Håkonsen, V.; Rønneberg, S.; He, J.; Zhang, Z. Epidermal Gland Inspired Self-Repairing Slippery Lubricant-Infused Porous Coatings with Durable Low Ice Adhesion. Coatings 2019, 9, 602. https://doi.org/10.3390/coatings9100602
Li T, Zhuo Y, Håkonsen V, Rønneberg S, He J, Zhang Z. Epidermal Gland Inspired Self-Repairing Slippery Lubricant-Infused Porous Coatings with Durable Low Ice Adhesion. Coatings. 2019; 9(10):602. https://doi.org/10.3390/coatings9100602
Chicago/Turabian StyleLi, Tong, Yizhi Zhuo, Verner Håkonsen, Sigrid Rønneberg, Jianying He, and Zhiliang Zhang. 2019. "Epidermal Gland Inspired Self-Repairing Slippery Lubricant-Infused Porous Coatings with Durable Low Ice Adhesion" Coatings 9, no. 10: 602. https://doi.org/10.3390/coatings9100602
APA StyleLi, T., Zhuo, Y., Håkonsen, V., Rønneberg, S., He, J., & Zhang, Z. (2019). Epidermal Gland Inspired Self-Repairing Slippery Lubricant-Infused Porous Coatings with Durable Low Ice Adhesion. Coatings, 9(10), 602. https://doi.org/10.3390/coatings9100602






