The Tribocorrosion and Corrosion Properties of Thermally Oxidized Ti6Al4V Alloy in 0.9 wt.% NaCl Physiological Saline
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Sample Preparation
2.2. Surface Characterization
2.3. Electrochemical Corrosion Test
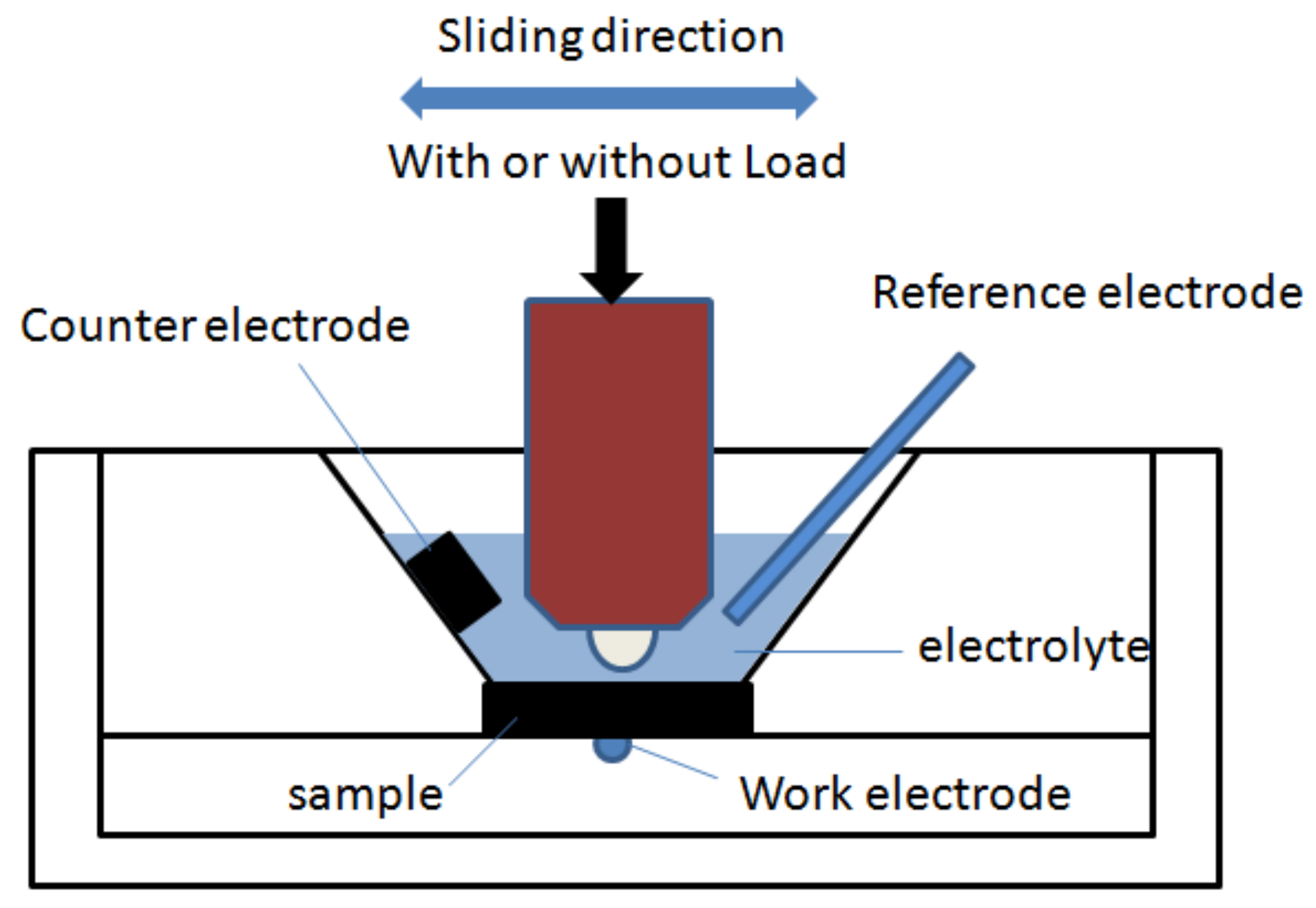
2.4. Tribocorrosion Test
3. Results and Discussion
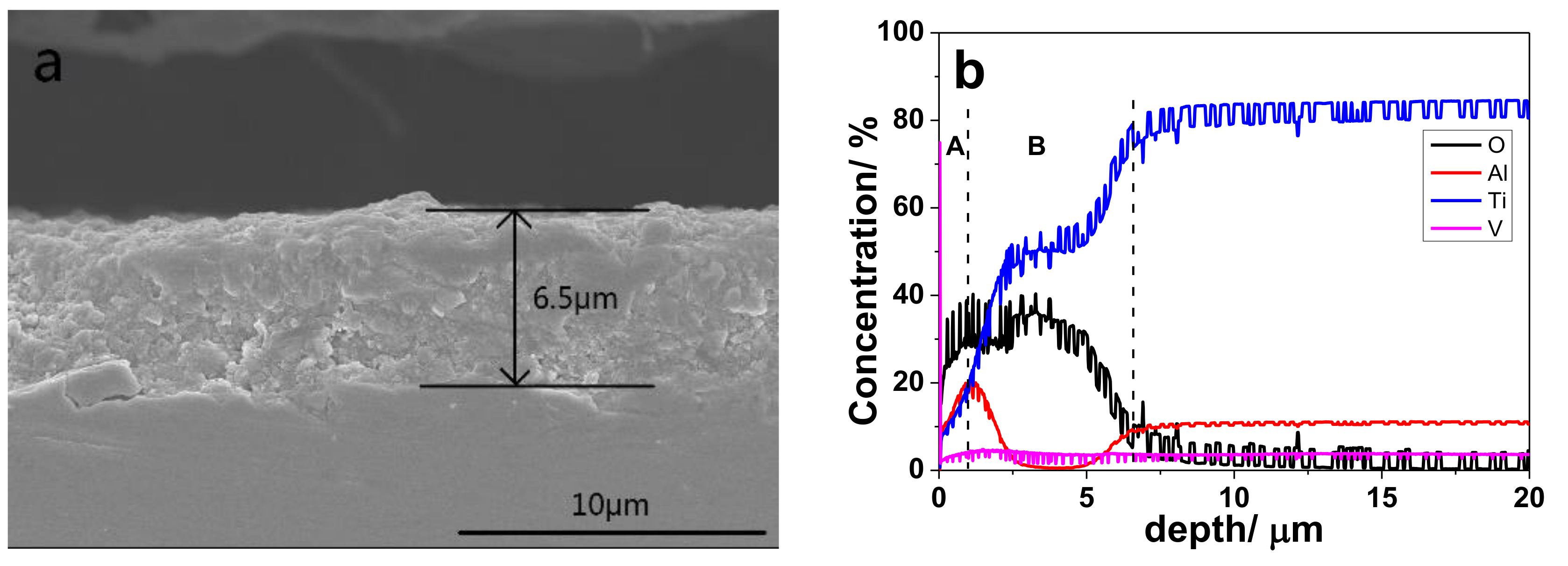
3.1. Characterization of Thermally Oxidized Samples
3.2. Corrosion Behavior
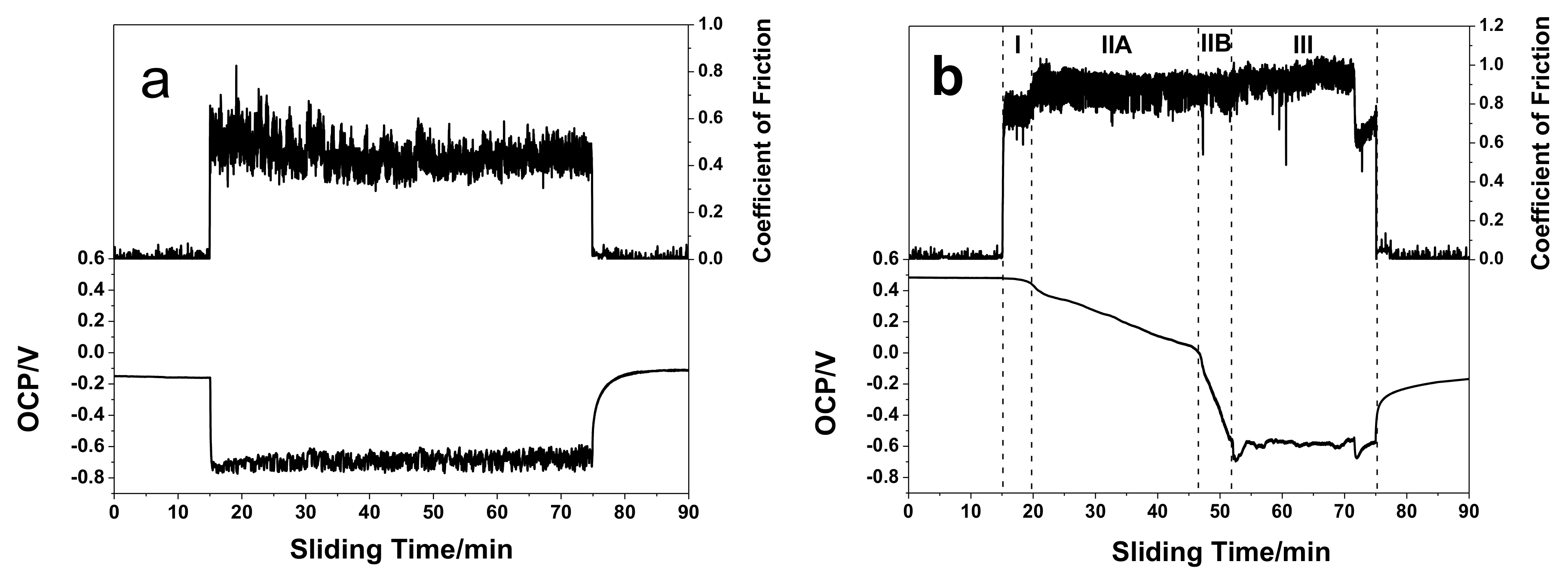
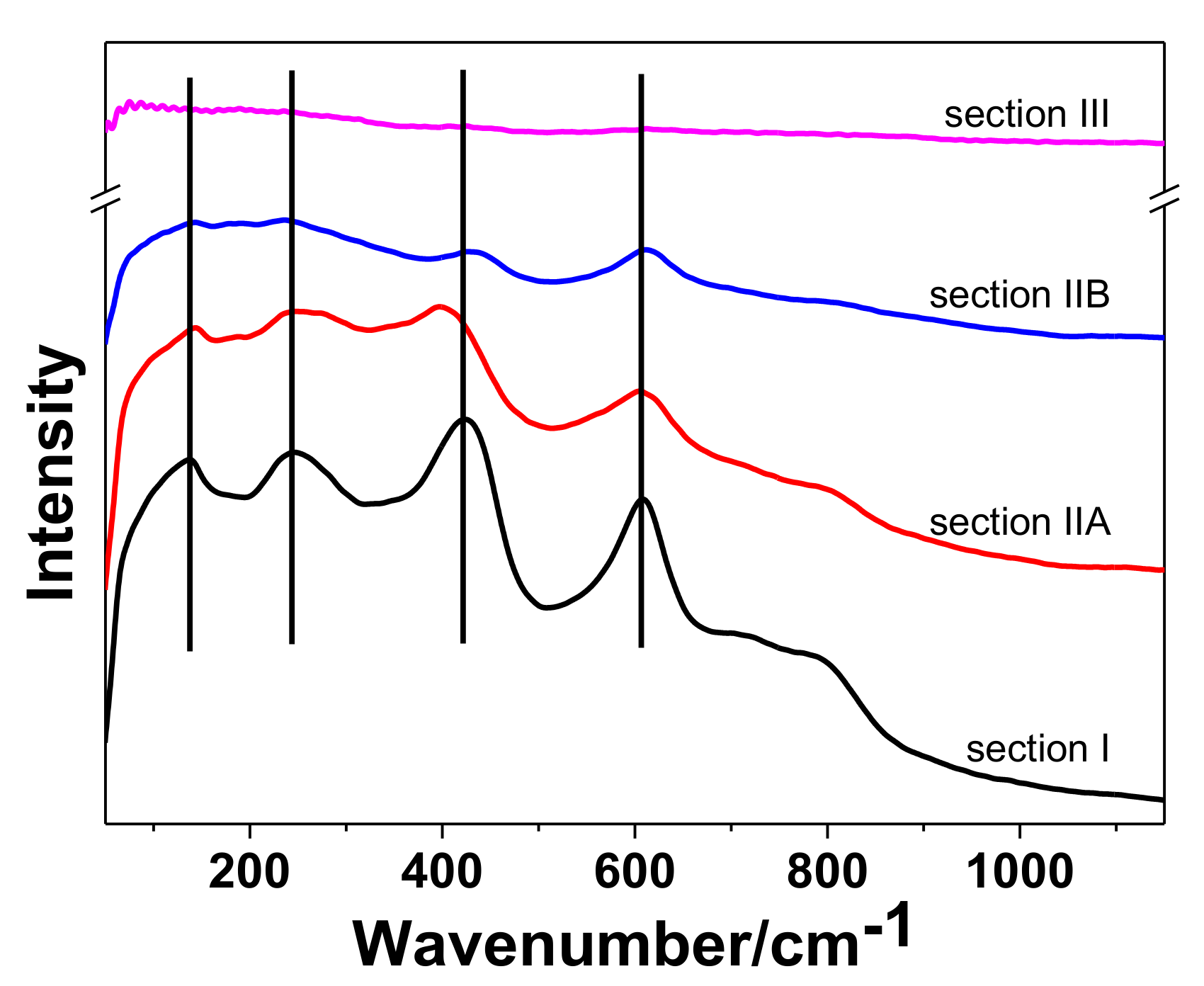
3.3. Tribocorrosion Behavior
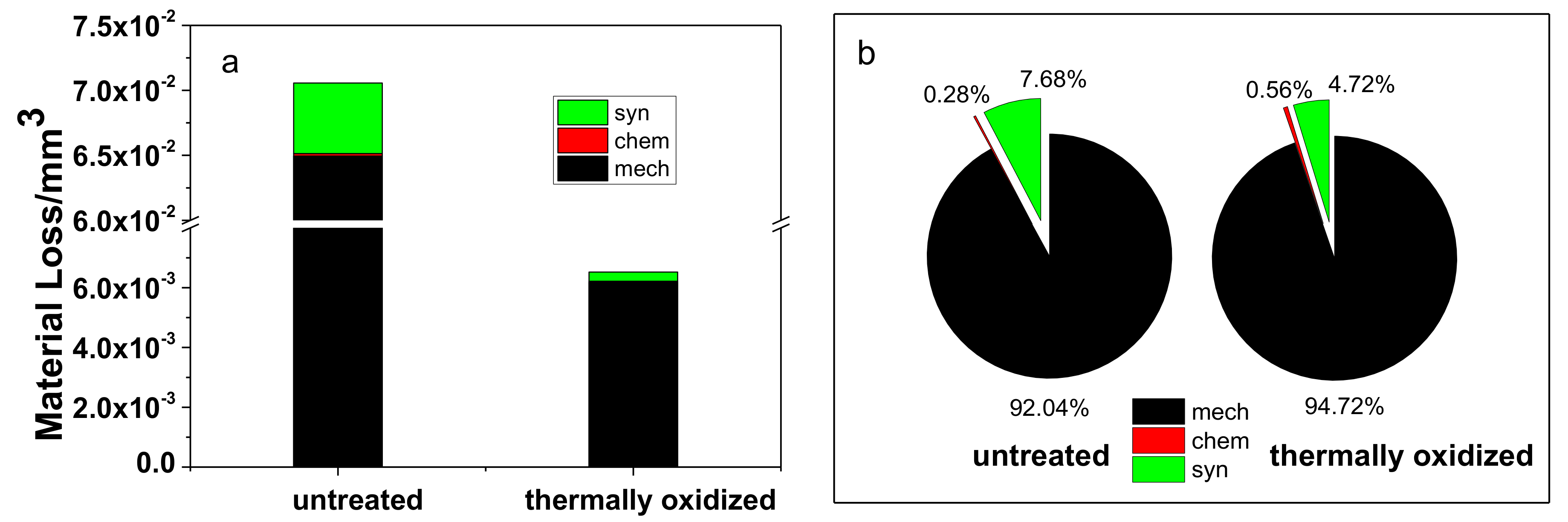
3.4. Material Loss Volume
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barao, V.A.; Mathew, M.T.; Assuncao, W.G.; Yuan, J.C.C.; Wimmer, M.A.; Sukotjo, C. Stability of cpTi and Ti6Al4V alloy for dental implants as a function of saliva pH—An electrochemical study. Clin. Oral Implants Res. 2012, 23, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.C.L.; Rodrigues, R.C.S.; Claro, A.P.R.A.; de Mattos, M.D.G.C.; Ribeiro, R.F. Wear resistance of experimental titanium alloys for dental applications. J. Mech. Behav. Biomed. 2011, 4, 1873–1879. [Google Scholar] [CrossRef] [PubMed]
- Licausi, M.P.; Muñoz, A.I.; Borrás, V.A. Influence of the fabrication process and fluoride content on the tribocorrosion behaviour of Ti6Al4V biomedical alloy in artificial saliva. J. Mech. Behav. Biomed. 2013, 20, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Rack, H.J.; Qazi, J.I. Titanium alloys for biomedical applications. Mat. Sci. Eng. C 2006, 26, 1269–1277. [Google Scholar] [CrossRef]
- Yadroitsev, I.; Krakhmalev, P.; Yadroitsava, I. Selective laser melting of Ti6Al4V alloy for biomedical applications: Temperature monitoring and microstructural evolution. J. Alloy. Compd. 2014, 583, 404–409. [Google Scholar] [CrossRef]
- Dearnley, P.A.; Dahm, K.L.; Çimenoǧlu, H. The corrosion-wear behaviour of thermally oxidised CP-Ti and Ti6Al4V. Wear 2004, 256, 469–479. [Google Scholar] [CrossRef]
- Guleryuz, H.; Cimenoglu, H. Effect of thermal oxidation on corrosion and corrosion-wear behaviour of a Ti6Al4V alloy. Biomaterials 2004, 25, 3325–3333. [Google Scholar] [CrossRef] [PubMed]
- Guleryuz, H.; Cimenoglu, H. Oxidation of Ti6Al4V alloy. J. Alloy. Compd. 2009, 472, 241–246. [Google Scholar] [CrossRef]
- Kumar, S.; Narayanan, T.S.; Raman, S.G.S.; Seshadri, S.K. Thermal oxidation of Ti6Al4V alloy: Microstructural and electrochemical characterization. Mater. Chem. Phys. 2010, 119, 337–346. [Google Scholar] [CrossRef]
- Azadeh, A.; Ashrafizadeh, F. Structural features and corrosion analysis of thermally oxidized titanium. J. Alloy. Compd. 2009, 480, 849–852. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Y.; Zhang, C.; Liao, Z.; Liu, W. The improvement of wettability, biotribological behavior and corrosion resistance of titanium alloy pretreated by thermal oxidation. Tribol. Int. 2014, 79, 174–182. [Google Scholar] [CrossRef]
- Wang, S.; Liao, Z.; Liu, Y.; Liu, W. Influence of thermal oxidation temperature on themicrostructural and tribological behavior of Ti6Al4V alloy. Surf. Coat. Technol. 2014, 240, 470–477. [Google Scholar] [CrossRef]
- Marin, E.; Offoiach, R.; Regis, M.; Fusi, S.; Lanzutti, A.; Fedrizzi, L. Diffusive thermal treatments combined with PVD coatings for tribological protection of titanium alloys. Mater. Des. 2016, 89, 314–322. [Google Scholar] [CrossRef]
- Rahmati, B.; Sarhan, A.A.; Zalnezhad, E.; Kamiab, Z.; Dabbagh, A.; Choudhury, D.; Abas, W.A.B.W. Development of tantalum oxide (TaO) thin film coating on biomedical Ti-6Al-4V alloy to enhance mechanical properties and biocompatibility. Ceram. Int. 2016, 42, 466–480. [Google Scholar] [CrossRef]
- Oliveira, V.M.C.A.; Aguiar, C.; Vazquez, A.M.; Robin, A.; Barboza, M.J.R. Improving corrosion resistance of Ti6Al4V alloy through plasma-assisted PVD deposited nitride coatings. Corros. Sci. 2014, 88, 317–327. [Google Scholar] [CrossRef]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R.; Hamouda, A.M.; Goh, B.T.; Yoon, G.H. Ti/TiN/HA coating on Ti–6Al–4V for biomedical applications. Ceram. Int. 2015, 41, 14447–14457. [Google Scholar] [CrossRef]
- Kessler, O.; Surm, H.; Hoffmann, F.; Mayr, P. Enhancing surface hardness of titanium alloy Ti6Al4V by combined nitriding and CVD coating. Surf. Eng. 2002, 18, 299–304. [Google Scholar] [CrossRef]
- Sathish, S.; Geetha, M.; Pandey, N.D.; Richard, C.; Asokamani, R. Studies on the corrosion and wear behavior of the laser nitrided biomedical titanium and its alloys. Mater. Sci. Eng. C 2010, 30, 376–382. [Google Scholar] [CrossRef]
- Fernandes, A.; Vaz, F.; Ariza, E.; Rocha, L.; Ribeiro, A.; Vieira, A.; Rivière, J.; Pichon, L. Tribocorrosion behaviour of plasma nitrided and plasma nitrided + oxidised Ti6Al4V alloy. Surf. Coat. Technol. 2006, 200, 6218–6224. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Giovanardi, R.; Veronesi, P. Modification of Ti6Al4V implant surfaces by biocompatible TiO2/PCL hybrid layers prepared via sol-gel dip coating: Structural characterization, mechanical and corrosion behavior. Mater. Sci. Eng. C 2017, 74, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.S.; Chen, C.Z.; Li, S.T.; Huo, Q.H. Research progress on laser surface modification of titanium alloys. Appl. Surf. Sci. 2005, 242, 177–184. [Google Scholar] [CrossRef]
- Vázquez Martínez, J.M.; Salguero Gómez, J.; Batista Ponce, M.; Botana Pedemonte, F.J. Effects of Laser Processing Parameters on Texturized Layer Development and Surface Features of Ti6Al4V Alloy Samples. Coatings 2018, 8, 6. [Google Scholar] [CrossRef]
- Aniołek, K.; Kupka, M.; Barylski, A. Sliding wear resistance of oxide layers formed on a titanium surface during thermal oxidation. Wear 2016, 356, 23–29. [Google Scholar] [CrossRef]
- Bailey, R.; Sun, Y. Unlubricated sliding friction and wear characteristics of thermally oxidized commercially pure titanium. Wear 2013, 308, 61–70. [Google Scholar] [CrossRef]
- Grotberg, J.; Hamlekhan, A.; Butt, A.; Patel, S.; Royhman, D.; Shokuhfar, T.; Sukotjob, C.; Takoudisa, C.; Mathew, M.T. Thermally oxidized titania nanotubes enhance the corrosion resistance of Ti6Al4V. Mater. Sci. Eng. C 2016, 59, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Basiaga, M.; Kajzer, W.; Walke, W.; Kajzer, A.; Kaczmarek, M. Evaluation of physicochemical properties of surface modified Ti6Al4V and Ti6Al7Nb alloys used for orthopedic implants. Mater. Sci. Eng. C 2016, 68, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Basiaga, M.; Szewczenko, J.; Walke, W.; Zbigniew, P.; Magdalena, A.; Agnieszka, H. Electrochemical properties of TiO2 oxide layer deposited on Ti6Al7Nb alloy. Innov. Biomed. Eng. 2017, 526, 3–10. [Google Scholar] [CrossRef]
- Jamesh, M.; Kumar, S.; Narayanan, T.S.N.; Chu, P.K. Effect of thermal oxidation on the corrosion resistance of Ti6Al4V alloy in hydrochloric and nitric acid medium. Mater. Corros. 2013, 64, 902–907. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, W.; Tian, M.; Teng, S.H. Thermal oxidation of Ti6Al4V alloy and its biotribological properties under serum lubrication. Tribol. Int. 2015, 89, 67–71. [Google Scholar] [CrossRef]
- Lin, N.; Li, D.; Zou, J.; Xie, R.; Wang, Z.; Tang, B. Surface Texture-Based Surface Treatments on Ti6Al4V Titanium Alloys for Tribological and Biological Applications: A Mini Review. Materials 2018, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.M.V.; Pedemonte, F.J.B.; Galvin, M.B.; Gomez, J.S.; Barcena, M.M. Sliding Wear Behavior of UNS R56400 Titanium Alloy Samples Thermally Oxidized by Laser. Materials 2017, 10, 830. [Google Scholar] [CrossRef] [PubMed]
- Arslan, E.; Totik, Y.; Demirci, E.; Alsaran, A. Influence of surface roughness on corrosion and tribological behavior of CP-Ti after thermal oxidation treatment. J. Mater. Eng. Perform. 2010, 19, 428–433. [Google Scholar] [CrossRef]
- Bailey, R.; Sun, Y. Corrosion and tribocorrosion performance of thermally oxidized commercially pure titanium in a 0.9% NaCl solution. J. Mater. Eng. Perform. 2015, 24, 1669–1678. [Google Scholar] [CrossRef]
- Cao, L.; Liu, J.; Wan, Y.; Yang, S.; Gao, J.; Pu, J. Low-friction carbon-based tribofilm from poly-alpha-olefin oil on thermally oxidized Ti6Al4V. Surf. Coat. Technol. 2018, 337, 471–477. [Google Scholar] [CrossRef]
- De Assis, S.L.; Wolynec, S.; Costa, I. Corrosion characterization of titanium alloys by electrochemical techniques. Electrochim. Acta 2006, 51, 1815–1819. [Google Scholar] [CrossRef]
- Du, H.L.; Datta, P.K.; Lewis, D.B.; Burnell-Gray, J.S. Air oxidation behaviour of Ti6Al4V alloy between 650 and 850 °C. Corros. Sci. 1994, 36, 631–642. [Google Scholar] [CrossRef]
- Manhabosco, T.M.; Tamborim, S.M.; dos Santos, C.B.; Müller, I.L. Tribological, electrochemical and tribo-electrochemical characterization of bare and nitrided Ti6Al4V in simulated body fluid solution. Corros. Sci. 2011, 53, 1786–1793. [Google Scholar] [CrossRef]
- Mareci, D.; Chelariu, R.; Ciurescu, G.; Sutiman, D.; Gordin, D.M.; Gloriant, T. Corrosion behaviour of some titanium dental alloys synthesized by cold crucible levitation melting. J. Optoelectron. Adv. Mater. 2010, 12, 1590–1596. [Google Scholar]
- Montemor, M.F.; Ferreira, M.G.S. Electrochemical study of modified bis-[triethoxysilylpropyl] tetrasulfide silane films applied on the AZ31 Mg alloy. Electrochim. Acta 2007, 52, 7486–7495. [Google Scholar] [CrossRef]
- Fazel, M.; Salimijazi, H.R.; Golozar, M.A. A comparison of corrosion, tribocorrosion and electrochemical impedance properties of pure Ti and Ti6Al4V alloy treated by micro-arc oxidation process. Appl. Surf. Sci. 2015, 324, 751–756. [Google Scholar] [CrossRef]
- González, J.E.G.; Mirza-Rosca, J.C. Study of the corrosion behaviour of titanium and some of its alloys for biomedical and dental implant applications. J. Electroanal. Chem. 1999, 471, 109–115. [Google Scholar] [CrossRef]




| Material | Composition (wt.%) | ||||||
|---|---|---|---|---|---|---|---|
| Ti | Al | V | Zr | Mo | Fe | Ni | |
| Ti6Al4V | 89.740 | 6.188 | 4.067 | <0.100 | <0.100 | <0.100 | 0.005 |
| Parameter | Samples | |
|---|---|---|
| Untreated | Thermally Oxidized | |
| Ecorr (V) | −0.3052 | 0.2226 |
| Icorr (A·cm−2) | 1.68 × 10−6 | 3.86 × 10−7 |
| Sample | Rs (Ω cm2) | Qo (Ssn/cm2) | no | Ro (Ω cm2) | QODZ (Ssn/cm2) | nODZ | RODZ (Ω cm2) |
|---|---|---|---|---|---|---|---|
| Untreated | 514.6 | 8.54 × 10−5 | 0.906 | 333.1 | 8.46 × 10−6 | 0.896 | 5.51 × 105 |
| Thermally oxidized | 388.8 | 8.42 × 10−7 | 0.813 | 1.6 × 104 | 5.02 × 10−6 | 0.864 | 3.57 × 106 |
| Samples | Region 1 | Element (at.%) | |||
|---|---|---|---|---|---|
| Ti | Al | V | O | ||
| Untreated | 1 | 90.31 | 6.01 | 1.16 | 2.52 |
| 2 | 90.13 | 6.53 | 1.32 | 2.02 | |
| Thermally oxidized | 1 | 21.47 | 8.66 | 1.71 | 68.16 |
| 2 | 53.99 | 6.64 | 2.86 | 36.51 | |
| 3 | 86.77 | 8.24 | 2.65 | 2.34 | |
| Contribution | Untreated | Thermally Oxidized Sample | ||
|---|---|---|---|---|
| Material Loss Volume (10−3 mm3) | Proportion of Total Material Loss Volume (%) | Material Loss Volume (10−3 mm3) | Proportion of Total Material Loss Volume (%) | |
| mech | 64.95 ± 1.26 | 92.04 | 6.18 ± 0.16 | 94.72 |
| chem | 0.20 ± 0.02 | 0.28 | 0.04 ± 0.01 | 0.56 |
| syn | 5.42 ± 0.33 | 7.68 | 0.31 ± 0.04 | 4.72 |
| total | 70.57 ± 1.38 | 100 | 6.53 ± 0.19 | 100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, L.; Wan, Y.; Yang, S.; Pu, J. The Tribocorrosion and Corrosion Properties of Thermally Oxidized Ti6Al4V Alloy in 0.9 wt.% NaCl Physiological Saline. Coatings 2018, 8, 285. https://doi.org/10.3390/coatings8080285
Cao L, Wan Y, Yang S, Pu J. The Tribocorrosion and Corrosion Properties of Thermally Oxidized Ti6Al4V Alloy in 0.9 wt.% NaCl Physiological Saline. Coatings. 2018; 8(8):285. https://doi.org/10.3390/coatings8080285
Chicago/Turabian StyleCao, Lei, Yong Wan, Shuyan Yang, and Jibin Pu. 2018. "The Tribocorrosion and Corrosion Properties of Thermally Oxidized Ti6Al4V Alloy in 0.9 wt.% NaCl Physiological Saline" Coatings 8, no. 8: 285. https://doi.org/10.3390/coatings8080285
APA StyleCao, L., Wan, Y., Yang, S., & Pu, J. (2018). The Tribocorrosion and Corrosion Properties of Thermally Oxidized Ti6Al4V Alloy in 0.9 wt.% NaCl Physiological Saline. Coatings, 8(8), 285. https://doi.org/10.3390/coatings8080285




