Effect of Annealing Treatment on Microstructure and Properties of Cr-Coatings Deposited on AISI 5140 Steel by Brush-Plating
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Characterization
3. Results and Discussion
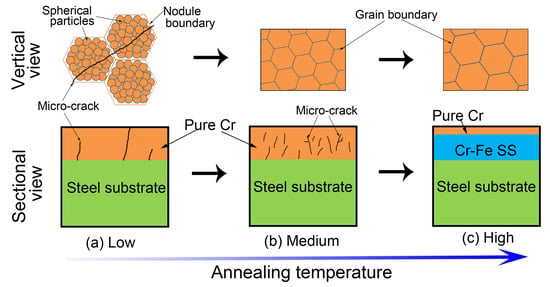
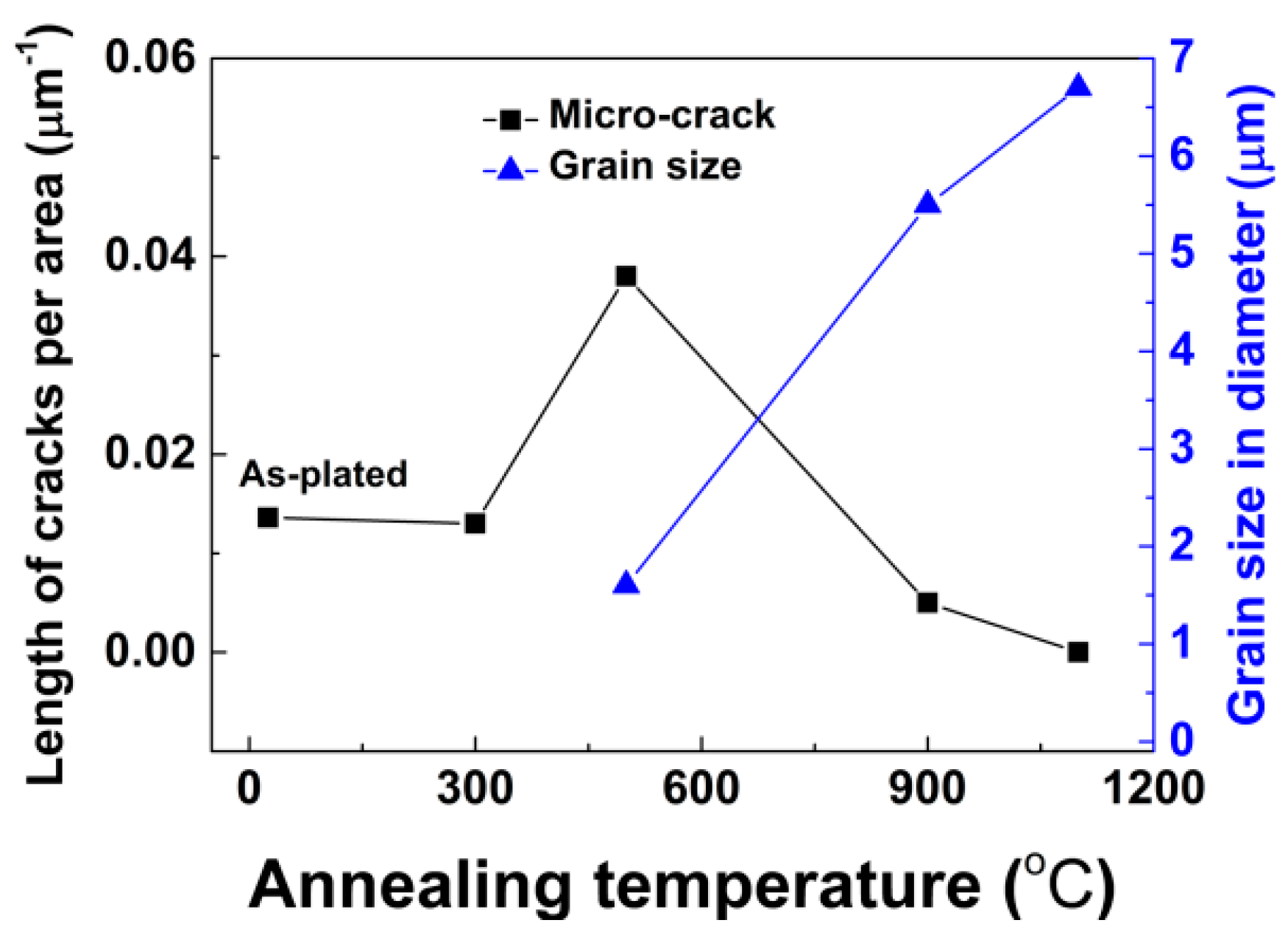
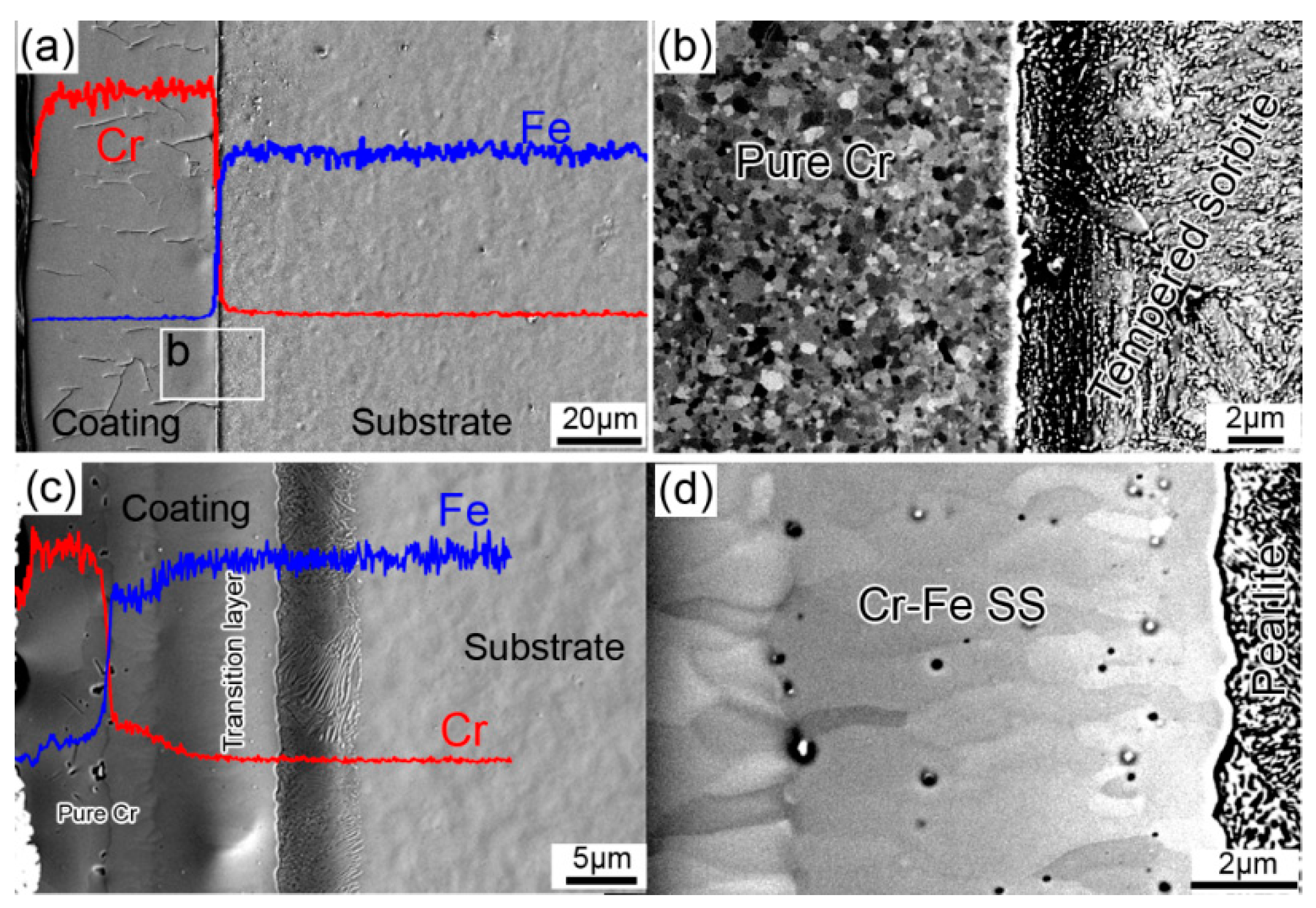
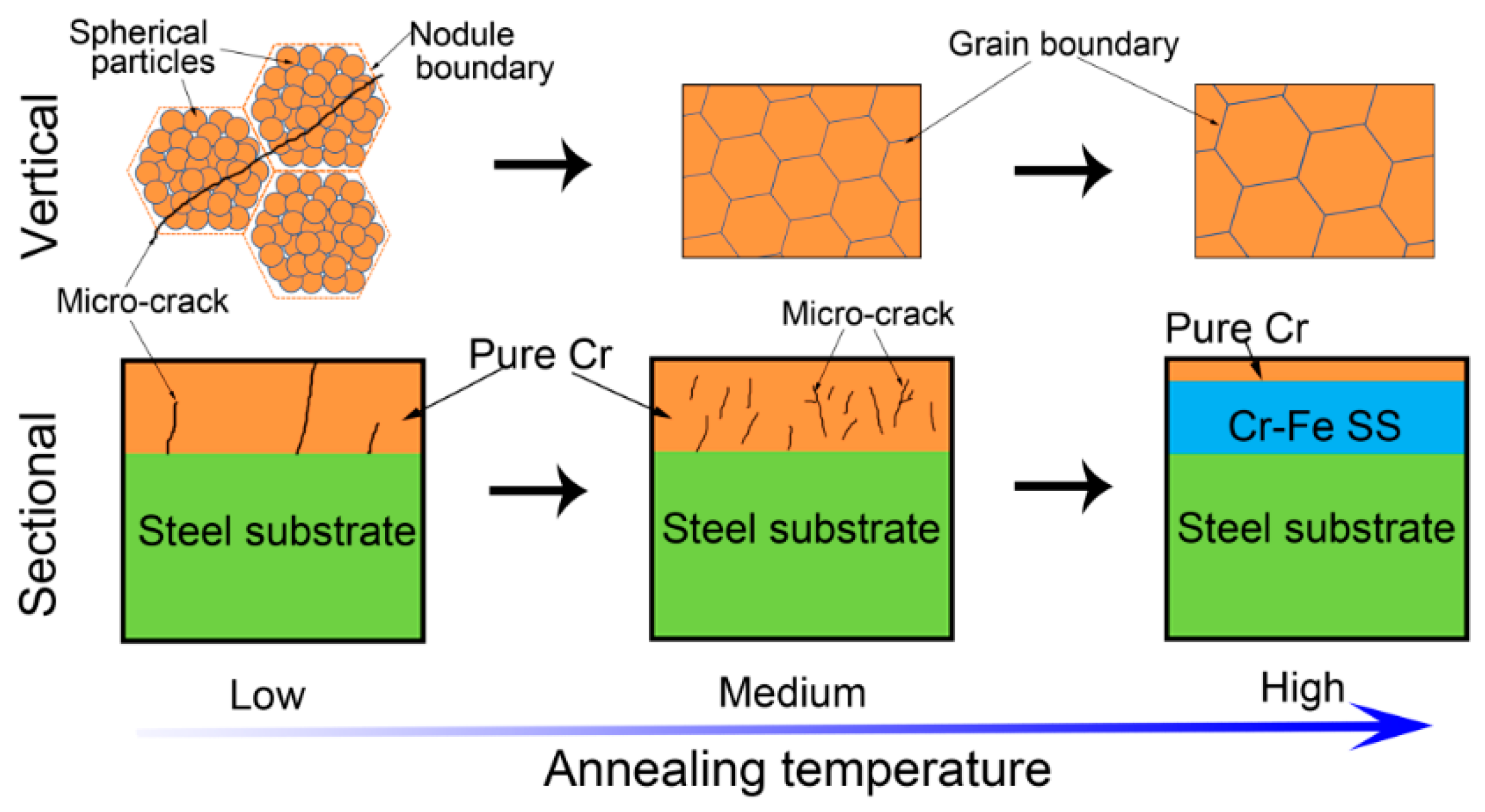
3.1. Microstructure Evolution
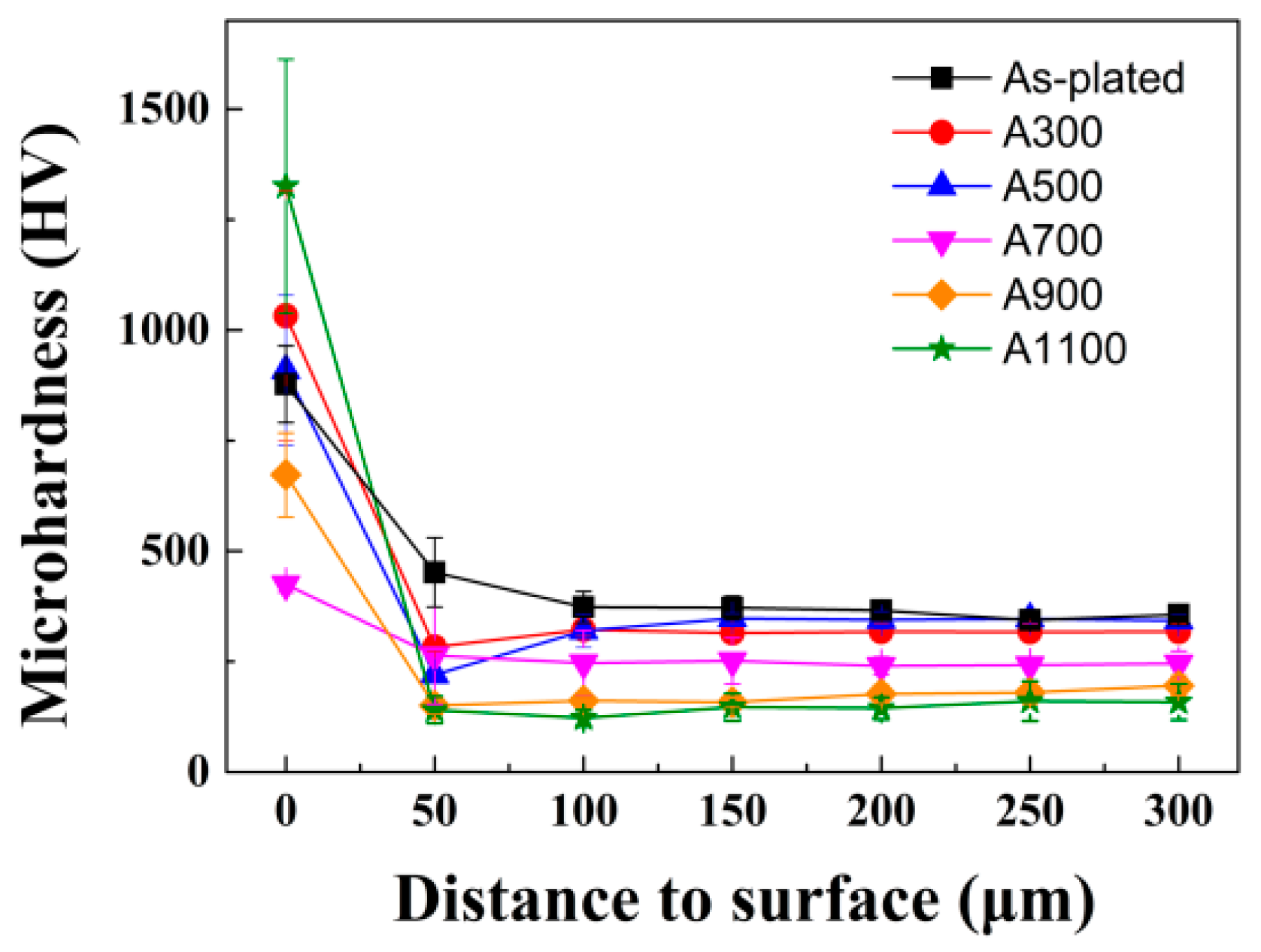
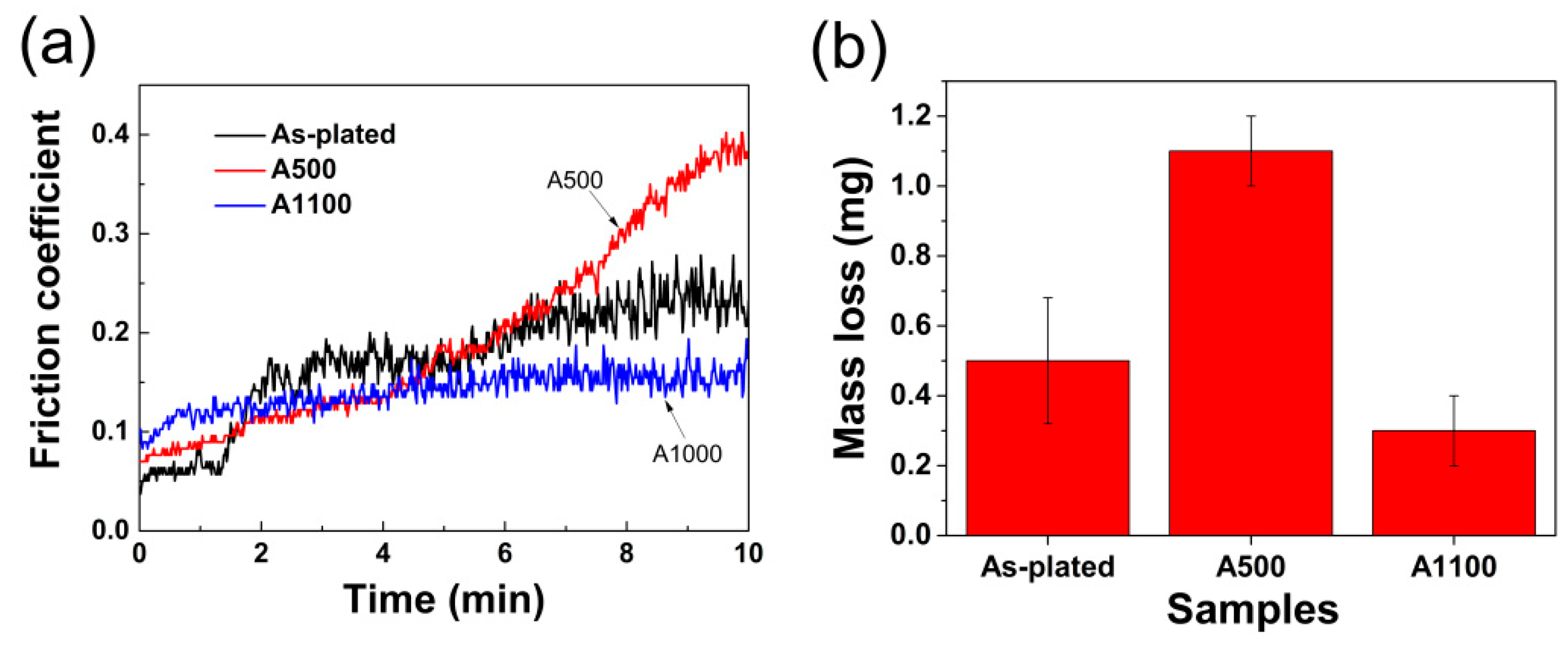
3.2. Mechanical Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dengo, C.; Meneghetti, G.; Dabalà, M. Experimental analysis of bending fatigue strength of plain and notched case-hardened gear steels. Int. J. Fatigue 2015, 80, 145–161. [Google Scholar] [CrossRef]
- Tobie, T.; Hippenstiel, F.; Mohrbacher, H. Optimizing gear performance by alloy modification of carburizing steels. Metals 2017, 7, 415. [Google Scholar] [CrossRef]
- Qiu, Z.-K.; Zhang, P.-Z.; Wei, D.-B.; Wei, X.-F.; Chen, X.-H. A study on tribological behavior of double-glow plasma surface alloying W-Mo coating on gear steel. Surf. Coat. Technol. 2015, 278, 92–98. [Google Scholar] [CrossRef]
- Hu, J.; Ma, C.; Xu, H.; Guo, N.; Hou, T. Development of a composite technique for preconditioning of 41Cr4 steel used as gear material: Examination of its microstructural characteristics and properties. Sci. Technol. Nucl. Install. 2016, 2016, 6. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, M.; Pang, X.; Volinsky, A.; Chen, M.; Gao, K. Applications and thermodynamic analysis of equilibrium solution for secondary phases in Ti–N–C gear steel system with nano-particles. Metals 2017, 7, 110. [Google Scholar] [CrossRef]
- Tian, L.; Feng, Z.; Xiong, W. Microstructure, microhardness, and wear resistance of AlCoCrFeNiTi/Ni60 coating by plasma spraying. Coatings 2018, 8, 112. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Khonsari, M. Effect of untampered plasma coating and surface texturing on friction and running-in behavior of piston rings. Coatings 2018, 8, 110. [Google Scholar] [CrossRef]
- Paiva, J.; Fox-Rabinovich, G.; Locks Junior, E.; Stolf, P.; Seid Ahmed, Y.; Matos Martins, M.; Bork, C.; Veldhuis, S. Tribological and wear performance of nanocomposite PVD hard coatings deposited on aluminum die casting tool. Materials 2018, 11, 358. [Google Scholar] [CrossRef] [PubMed]
- Damm, D.; Contin, A.; Barbieri, F.; Trava-Airoldi, V.; Barquete, D.; Corat, E. Interlayers applied to CVD diamond deposition on steel substrate: A review. Coatings 2017, 7, 141. [Google Scholar] [CrossRef]
- Chai, L.; Wang, S.; Wu, H.; Yang, Z.; Pan, H.; Song, B.; Guo, N. Bimodal plate structures induced by pulsed laser in duplex-phase Zr alloy. Sci. China Technol. Sci. 2017, 60, 587–592. [Google Scholar] [CrossRef]
- Chen, Y.; Guo, Y.; Lu, B.; Xu, M.; Xu, J. Microstructure and properties of the interface area in the laser cladded Ni based coatings on the 1Cr10Mo1niwvnbn steel. Metals 2017, 7, 175. [Google Scholar] [CrossRef]
- Sen, S.; Sen, U. Sliding wear behavior of niobium carbide coated AISI 1040 steel. Wear 2008, 264, 219–225. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, Y.; Yang, X.; Li, H.; Xu, H.; Ma, C.; Dong, Q.; Guo, N.; Yao, Z. Effect of pack-chromizing temperature on microstructure and performance of AISI 5140 steel with Cr-coatings. Surf. Coat. Technol. 2018, 344, 656–663. [Google Scholar] [CrossRef]
- Subramanian, B.; Mohan, S.; Jayakrishnan, S. Selective area deposition of tin–nickel alloy coating—An alternative for decorative chromium plating. J. Appl. Electrochem. 2007, 37, 219–224. [Google Scholar] [CrossRef]
- Hu, J.J.; Chai, L.J.; Xu, H.B.; Ma, C.P.; Deng, S.B. Microstructural modification of brush-plated nanocrystalline Cr by high current pulsed electron beam irradiation. J. Nano Res. SW 2016, 41, 87–95. [Google Scholar] [CrossRef]
- Dey, S.; Chatterjee, S.; Singh, B.P.; Bhattacharjee, S.; Rout, T.K.; Sengupta, D.K.; Besra, L. Development of superhydrophobic corrosion resistance coating on mild steel by electrophoretic deposition. Surf. Coat. Technol. 2018, 341, 24–30. [Google Scholar] [CrossRef]
- Chen, T.; Ge, S.; Liu, H.; Sun, Q.; Zhu, W.; Yan, W.; Qi, J. Fabrication of low adhesive superhydrophobic surfaces using nano Cu/Al2O3 Ni–Cr composited electro-brush plating. Appl. Surf. Sci. 2015, 356, 81–90. [Google Scholar] [CrossRef]
- Kosugi, D.; Hagio, T.; Kamimoto, Y.; Ichino, R. Effect of the addition of molybdenum on the structure and corrosion resistance of zinc–iron plating. Coatings 2017, 7, 235. [Google Scholar] [CrossRef]
- Ma, G.; Xu, B.; Wang, H.; Wang, X.; Li, G.; Zhang, S. Research on the microstructure and space tribology properties of electric-brush plated Ni/MoS2–C composite coating. Surf. Coat. Technol. 2013, 221, 142–149. [Google Scholar] [CrossRef]
- Xu, B.; Wang, H.; Dong, S.; Jiang, B. Fretting wear-resistance of Ni-base electro-brush plating coating reinforced by nano-alumina grains. Mater. Lett. 2006, 60, 710–713. [Google Scholar]
- Du, L.; Xu, B.; Dong, S.; Yang, H.; Wu, Y. Preparation, microstructure and tribological properties of nano-Al2O3/Ni brush plated composite coatings. Surf. Coat. Technol. 2005, 192, 311–316. [Google Scholar] [CrossRef]
- Subramanian, B.; Mohan, S.; Jayakrishnan, S.; Jayachandran, M. Structural and electrochemical characterization of Ni nanostructure films on steels with brush plating and sputter deposition. Curr. Appl. Phys. 2007, 7, 305–313. [Google Scholar] [CrossRef]
- Lee, D.B. Oxidation of Cr–C electroplating between 400 and 900 °C in air. Mater. Corros. 2008, 59, 598–601. [Google Scholar] [CrossRef]
- Jin, G.; Lu, B.; Hou, D.; Cui, X.; Song, J.; Liu, E. Influence of rare earths addition on residual stress of Fe-based coating prepared by brush plating technology. J. Rare Earth 2016, 34, 336–340. [Google Scholar] [CrossRef]
- Ghaziof, S.; Golozar, M.A.; Raeissi, K. Characterization of as-deposited and annealed Cr–C alloy coatings produced from a trivalent chromium bath. J. Alloy. Compd. 2010, 496, 164–168. [Google Scholar] [CrossRef]
- Tan, J.; Yu, T.; Xu, B.; Yao, Q. Microstructure and wear resistance of nickel–carbon nanotube composite coating from brush plating technique. Tribol. Lett. 2006, 21, 107–111. [Google Scholar] [CrossRef]
- Wu, B.; Xu, B.-S.; Zhang, B.; Jing, X.-D.; Liu, C.-L. Automatic brush plating: An update on brush plating. Mater. Lett. 2006, 60, 1673–1677. [Google Scholar] [CrossRef]
- Wu, B.; Xu, B.-S.; Zhang, B.; Lü, Y.-H. Preparation and properties of Ni/nano-Al2O3 composite coatings by automatic brush plating. Surf. Coat. Technol. 2007, 201, 6933–6939. [Google Scholar] [CrossRef]
- Li, W.H.; Zhou, X.Y.; Xu, Z.; Yan, M.J. Effect of bath temperature on nanocrystalline Ni-polytetrafluoroethylene composite coatings prepared by brush electroplating. Surf. Eng. 2009, 25, 353–360. [Google Scholar] [CrossRef]
- Li, Z.; Qian, S.; Wang, W.; Shen, H.; Long, H. High-temperature tribological properties of Ni–P alloy coatings deposited by electro-brush plating. Rare Met. 2011, 30, 669–675. [Google Scholar] [CrossRef]
- Pang, X.; Gao, K.; Luo, F.; Yang, H.; Qiao, L.; Wang, Y.; Volinsky, A.A. Annealing effects on microstructure and mechanical properties of chromium oxide coatings. Thin Solid Films 2008, 516, 4685–4689. [Google Scholar] [CrossRef]
- Hu, J.; Ma, C.; Yang, X.; Xu, H.; Guo, N.; Yu, H. Microstructure evolution during continuous cooling in AISI 5140 steel processed by induction heating chromizing. J. Mater. Eng. Perform. 2017, 26, 5530–5537. [Google Scholar] [CrossRef]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, J.; Jiang, J.; Li, H.; Yang, X.; Xu, H.; Jin, Y.; Ma, C.; Dong, Q.; Guo, N. Effect of Annealing Treatment on Microstructure and Properties of Cr-Coatings Deposited on AISI 5140 Steel by Brush-Plating. Coatings 2018, 8, 193. https://doi.org/10.3390/coatings8050193
Hu J, Jiang J, Li H, Yang X, Xu H, Jin Y, Ma C, Dong Q, Guo N. Effect of Annealing Treatment on Microstructure and Properties of Cr-Coatings Deposited on AISI 5140 Steel by Brush-Plating. Coatings. 2018; 8(5):193. https://doi.org/10.3390/coatings8050193
Chicago/Turabian StyleHu, Jianjun, Jie Jiang, Hui Li, Xian Yang, Hongbin Xu, Yan Jin, Chaoping Ma, Qingshan Dong, and Ning Guo. 2018. "Effect of Annealing Treatment on Microstructure and Properties of Cr-Coatings Deposited on AISI 5140 Steel by Brush-Plating" Coatings 8, no. 5: 193. https://doi.org/10.3390/coatings8050193
APA StyleHu, J., Jiang, J., Li, H., Yang, X., Xu, H., Jin, Y., Ma, C., Dong, Q., & Guo, N. (2018). Effect of Annealing Treatment on Microstructure and Properties of Cr-Coatings Deposited on AISI 5140 Steel by Brush-Plating. Coatings, 8(5), 193. https://doi.org/10.3390/coatings8050193