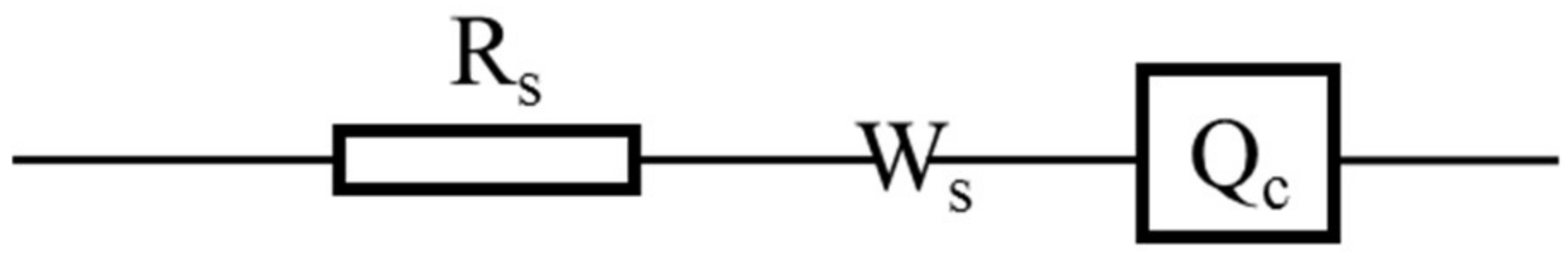3.1. Phase Compositions and Microstructures
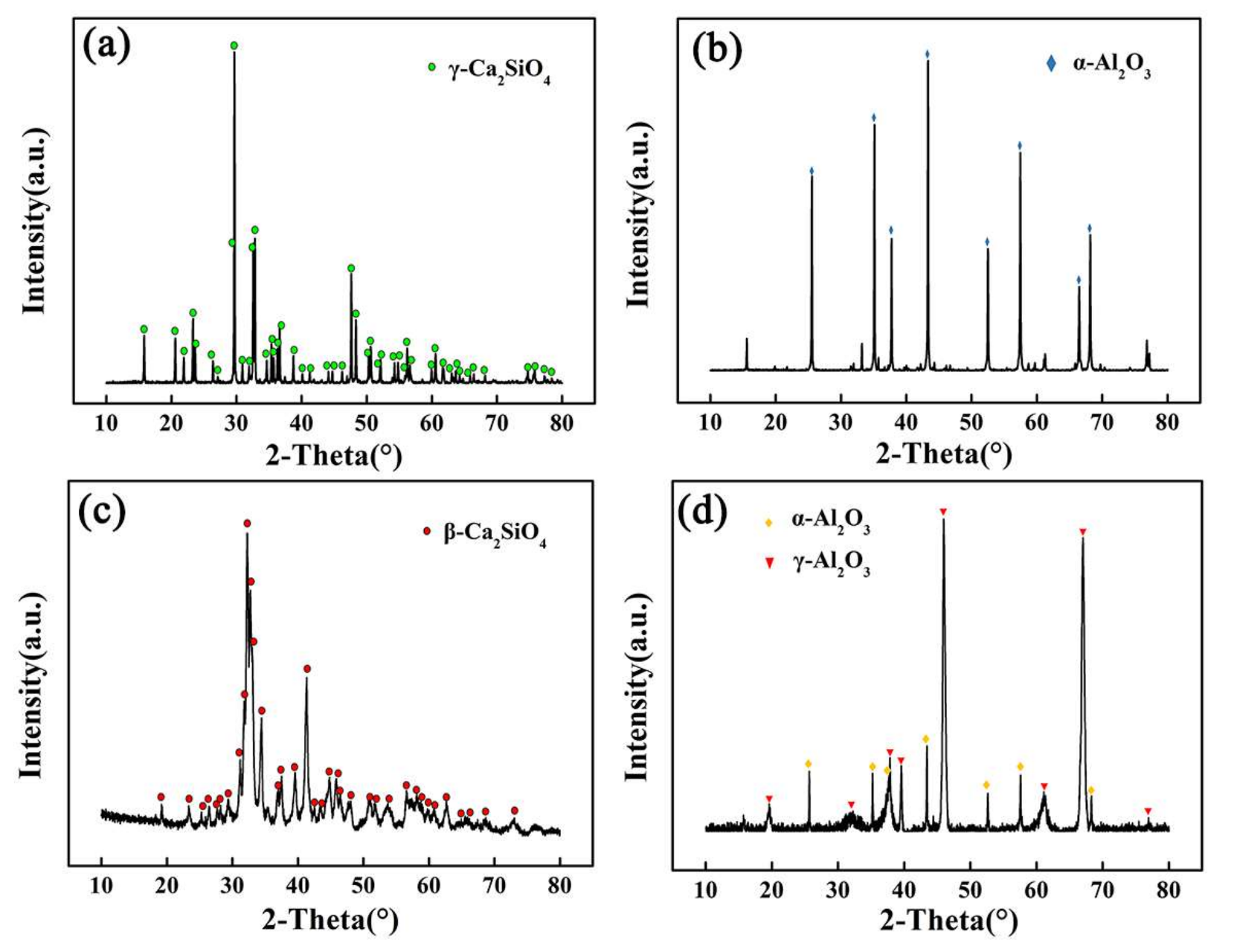
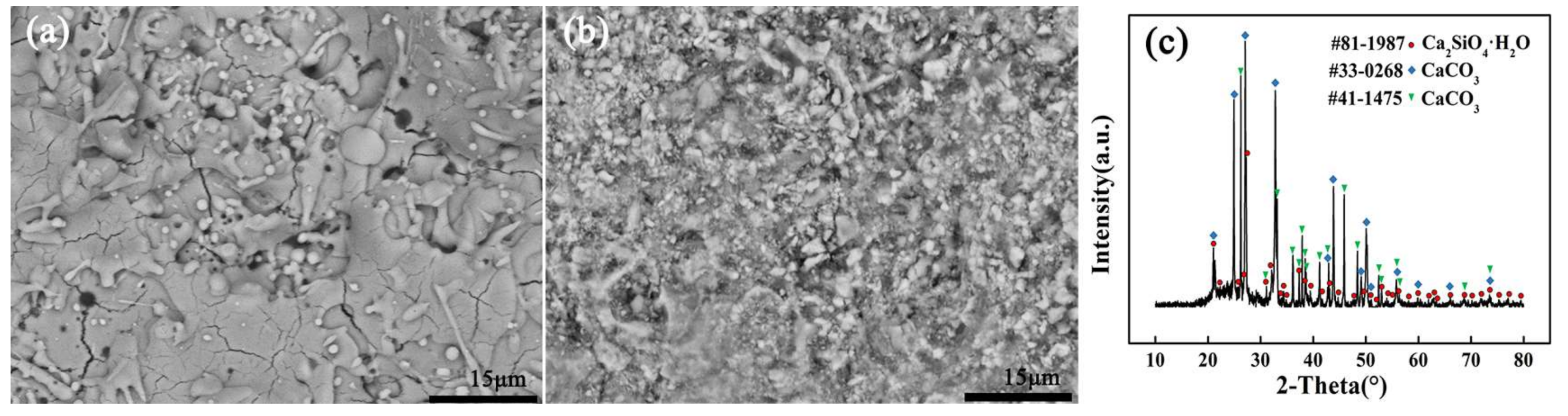
The XRD patterns of the powders and as-sprayed coatings are shown in
Figure 1. Ca
2SiO
4 powders were composed of γ-Ca
2SiO
4, while the phase in as-sprayed Ca
2SiO
4 coatings was β-Ca
2SiO
4 with a part of glassy phase. In
Figure 1c, the increase in the background area under the peaks indicated the glassy phase [
13]. In the high-temperature plasma flame flow, γ-Ca
2SiO
4 powders were rapidly melted into droplets, and the droplets hit/impacted the substrate, then quickly deposited on the surface to form coating. β-Ca
2SiO
4 was generated during the rapid cooling process, some of the droplets did not crystallize in time and were converted into glass phase. The rate of change in heat evolution leads to the formation of different types of polymorphs (α, α
H’, α
L’, β and γ) of Ca
2SiO
4 [
15], as shown in
Figure 2.
γ-Ca
2SiO
4 is the stable phase at room temperature. It is generally believed that the higher the formation temperature, the higher the hydration rate, so the hydration rate rank of the different phase is α > α
H’ > α
L’ > β > γ [
16]. Previous investigations indicated that γ-Ca
2SiO
4 almost cannot react with water [
17], while β-Ca
2SiO
4 can react with water to generate gel of hydrated calcium silicate. The alumina powders were α-Al
2O
3, but as-sprayed coating contained α-Al
2O
3 and γ-Al
2O
3. A part of α-Al
2O
3 transformed into γ-Al
2O
3 during the plasma spraying process. The phase of Al
2O
3 coatings produced by plasma spraying mainly existed in γ-Al
2O
3 phase [
18].
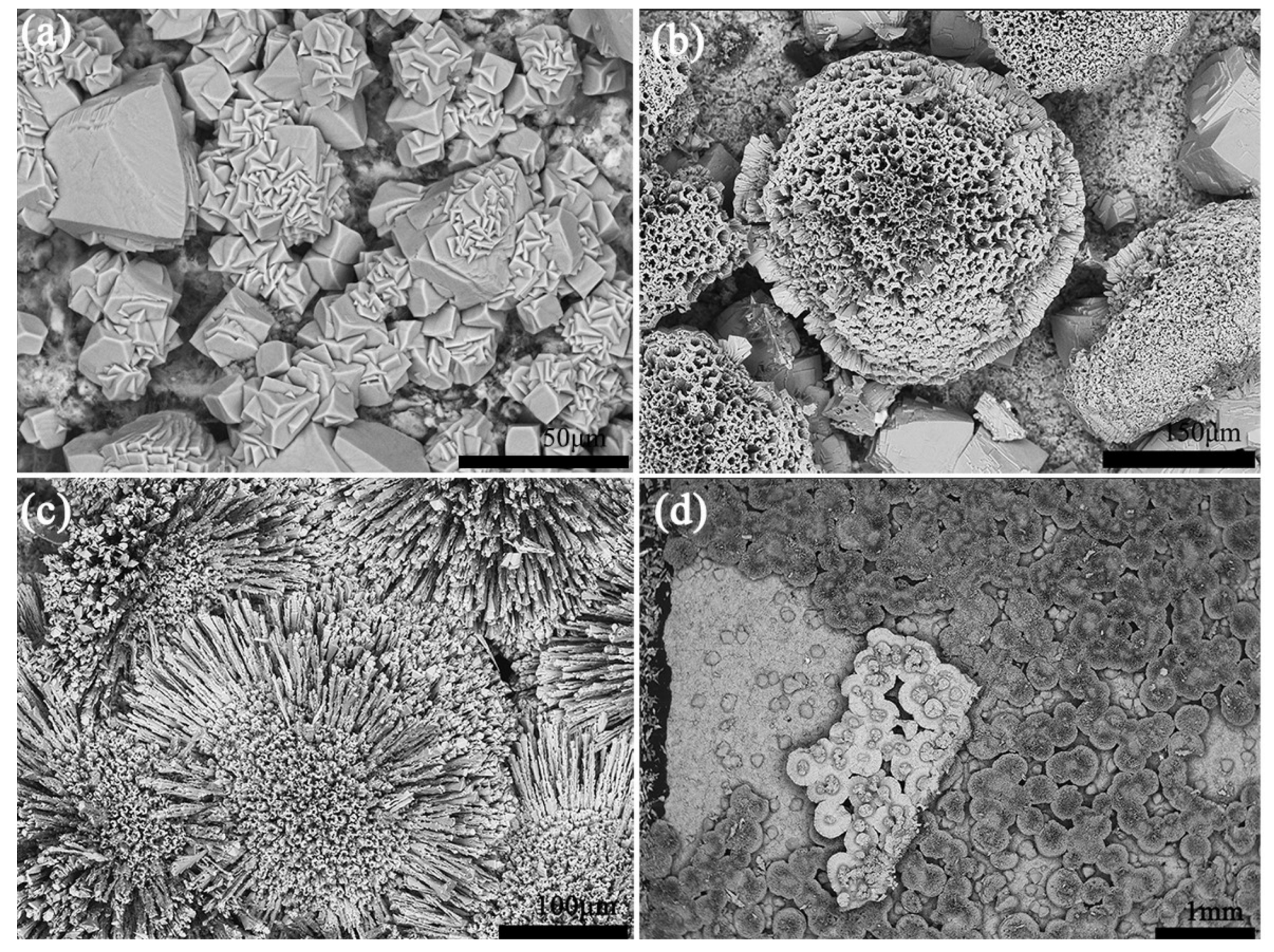
The surface and cross-sectional morphologies of as-sprayed coatings are shown in
Figure 3. The surface morphology of coatings revealed that the coating was built up from melted droplets. The cross-sectional view of the coating showed obvious pores and micro cracks. The porosity of Ca
2SiO
4 coating and Al
2O
3 coating were 6.50% and 8.85%, respectively. It showed that Ca
2SiO
4 coating was a little denser than Al
2O
3 coating. The bond strength of Ca
2SiO
4 coatings and Al
2O
3 coating were 35.0 ± 3.5 MPa and 26.0 ± 3.5 MPa, which revealed that the bonding strength between Ca
2SiO
4 coating and metal substrate was better than that for Al
2O
3 coating.
3.2. Immersion Test
Figure 4 shows the surface morphologies of Ca
2SiO
4 coating after being immersed in still 3.5 wt % NaCl solution at (35 ± 2) °C for 5, 10, 20 and 30 days.
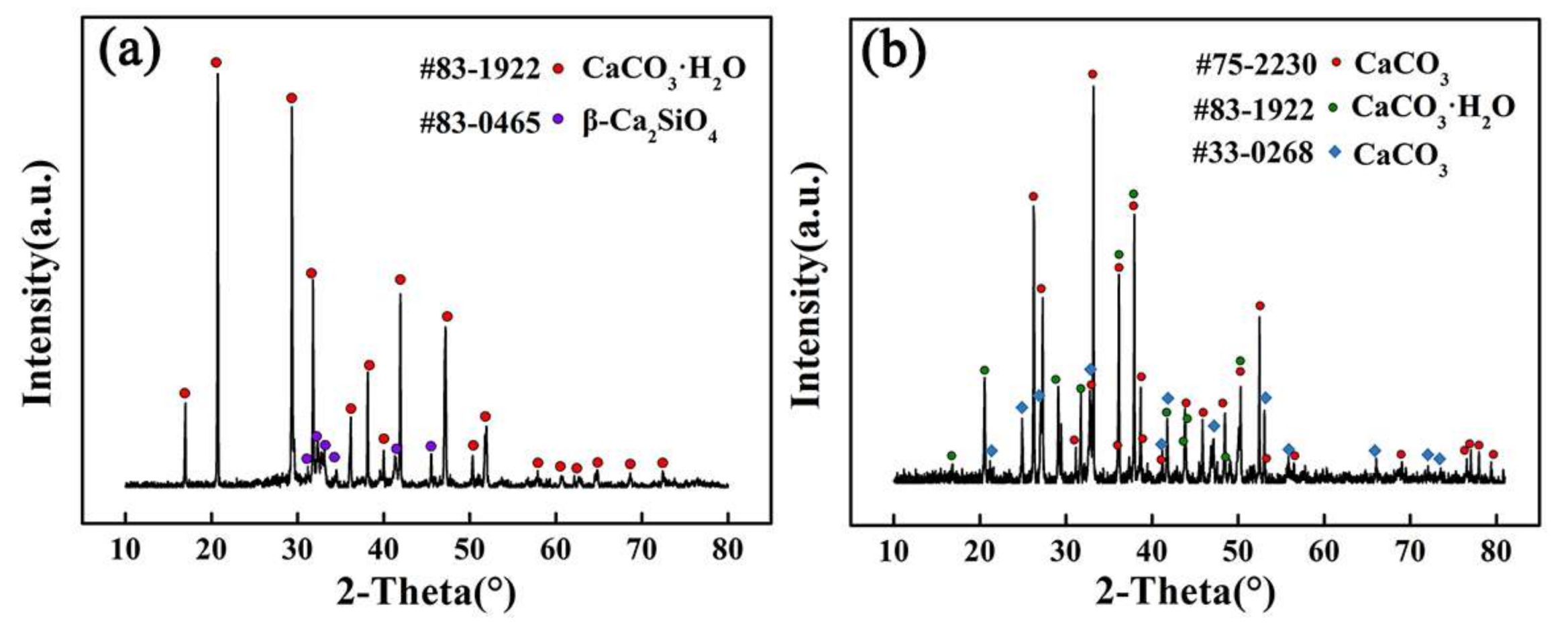
As shown in
Figure 4, after being immersed in 3.5 wt % NaCl solution for 5 days, there were some rhombic solid particles on the surface of Ca
2SiO
4 coating. XRD patterns in
Figure 5a show that the particles are rhombic calcite (CaCO
3). More and more calcium carbonate crystals were formed on the surface of Ca
2SiO
4 coating as the immersed time increased. When immersed for 20 days, chrysanthemum-like or spindle-like [
19] aragonite (CaCO
3) and spherical vaterite (CaCO
3) appeared. After 30 days, calcium carbonate crystals spread all over the coating surface. It was easy for calcium carbonate crystals to fall from the coating surface.
The chemical processes that took place on the surface of Ca
2SiO
4 during immersion of specimens in 3.5% NaCl solution are as follows [
20]:
And the Na+ and Cl− ions have no effect on the hydration process of Ca2SiO4 coatings.
Han et al. studied the factors affecting the phase and morphology of CaCO
3 prepared by a bubbling method. They found that low Ca
2+ concentration or sufficient supply of CO
2 was in favor of the formation of spherical vaterite [
21]. At the beginning of the immersion in 3.5 wt % NaCl solution, Ca
2SiO
4 coating reacted with water to release Ca(OH)
2. High Ca
2+ concentration promoted to form rhombic calcite as shown in
Figure 5a. As the slow dissolution of CO
2 gas, Ca
2+ concentration decreased, spherical vaterite formed on the coating surface as shown in
Figure 5b. In reference [
21], the size of calcium carbonate particle was below 10 μm, but the particle size of calcite and vaterite formed on Ca
2SiO
4 coating were more than 50 μm. During the immersion, the release of Ca(OH)
2 and dissolution of CO
2 gas were very slow, and the supersaturation of solution remained at a low level. In addition, supersaturation played an important role on the rate of crystal nucleus formation and crystal growth, and the supersaturation influenced the size and distribution of the formed particles. When the supersaturation was low, the ratio of crystal growth rate and crystal nucleus formation rate is bigger, so the crystal is bigger with relatively complete crystal shape, but the crystallization rate was slow [
22]. When the supersaturation of solution was kept low for a long time, the crystal growth was predominant in the solution. Therefore, the size of calcium carbonate particles on Ca
2SiO
4 coating was larger than that in other studies.
Figure 6 shows the surface morphologies of Al
2O
3 coating after being immersed in still 3.5 wt % NaCl solution at (35 ± 2) °C for 5, 10, 20 and 30 days. When immersed in 3.5 wt % NaCl solution for 5 days, there were more pores on the Al
2O
3 coating surface, thus the corrosion medium could permeate into the coating more easily than as-sprayed Al
2O
3 coating. Rust staining was discovered on the 10th day, and then rust staining became larger. Because of the dissolution of CO
2 gas in air, the pH value of the solution was below 7, and the swell was caused by the generation of hydrogen during the corrosion process.
Figure 7 shows the surface morphologies of coatings after being immersed 30 days, the Al
2O
3 coating appeared obvious rusty spot, but there was no rusty spot on Ca
2SiO
4 coating.
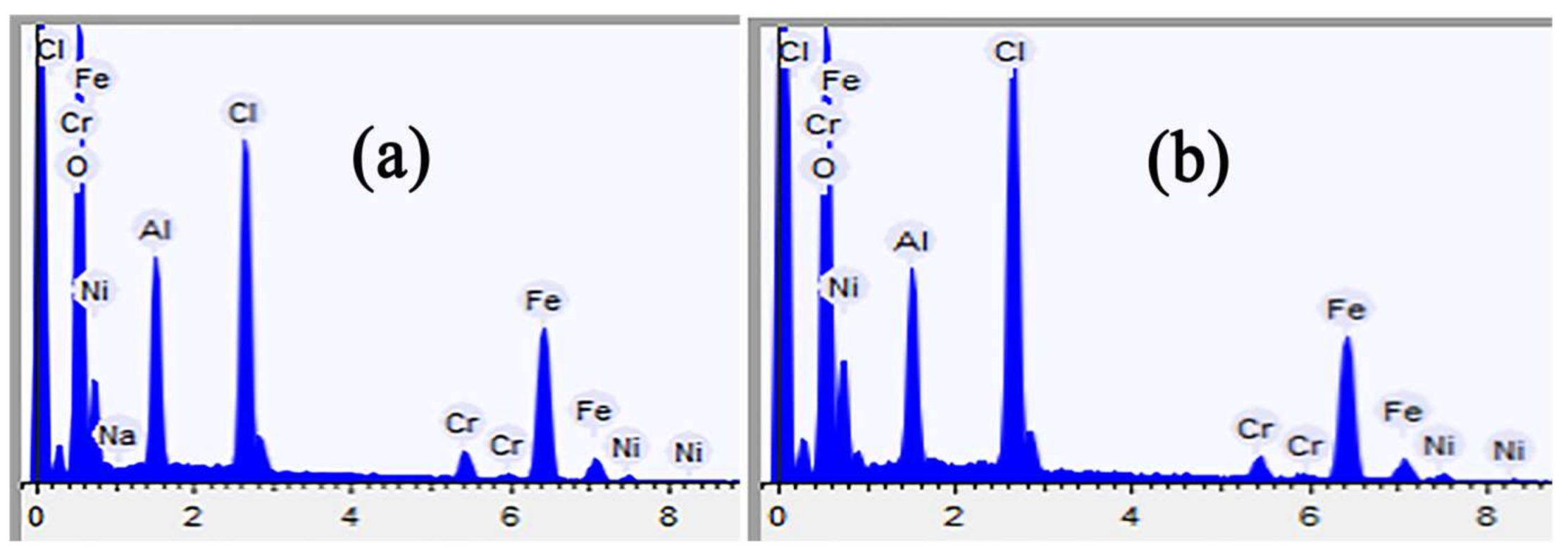
Figure 8 shows the EDS analysis of the region marked with a yellow rectangle in
Figure 6, the rust staining contains Fe, Ni, Cr and Cl elements. It is indicated that the corrosive medium penetrated into the metal substrate and the substrate suffered from corrosion.
Figure 9 shows the cross-section images of the Ca
2SiO
4 coating after being immersed for different times. In order to present the hydration area of the Ca
2SiO
4 coating, the morphology of the yellow box in
Figure 9c is shown in
Figure 9f. The bottom figures,
Figure 9d–f, are the same as the top ones with higher magnification. From
Figure 9, the porosity of as-sprayed coating was 6.05%, C
2S-10 was 3.05%, and C
2S-30 was 1.10%. The porosity of C
2S coating was reduced after being immersed.
Figure 10 also shows the change of coating surface. It was evident that the pore became smaller and the cracks became less after immersion for 30 days.
Figure 10 shows that hydration products C–S–H gel formed during the immersion test, and the pores and micro cracks were filled with C–S–H gel.
In
Figure 11, it is obvious that hydration phenomenon occurred at the top of 80–110 μm thickness of the coating after being immersed for 30 days. The CO
2 gas in air dissolved in NaCl solution to form CO
32− or HCO
3−. They reacted with Ca(OH)
2 and CaCO
3 crystal was formed at the coating surface. When the corrosion medium containing CO
32− or HCO
3− penetrated into the internal of the coating, C
2S-30 showed carbon element enrichment at the top of 80–110 μm thickness.
As shown in
Figure 12, during the immersion, the internal of Al
2O
3 coatings becomes looser and more porous. The porosity of as-sprayed coatings was 8.85%, which increased to 10.14% after immersion for 10 days and then to 12.36% after 30 days. Corrosion products were observed in Al
2O
3-30 coatings. The bond strength of Ca
2SiO
4 coating and Al
2O
3 coating after being immersed in 3.5 wt % NaCl solution for 12 days were 25.0 ± 3.5 MPa and 20.5 ± 3.5 MPa, respectively. The bond strength of C
2S-12 and Al
2O
3-12 were lower than that in as-sprayed Ca
2SiO
4 coatings and Al
2O
3 coating.
From
Figure 9 and
Figure 12, it is concluded that Ca
2SiO
4 coating becomes denser, the micro cracks and pores are filled with hydration products, such as C–S–H gel and CaCO
3. The dense internal structure is beneficial in isolating the metal substrate from the corrosive medium. Al
2O
3 coating becomes loose and porous after being immersed for 30 days. Corrosion medium attacked the metal substrate under Al
2O
3 coating.
3.3. Potentiodynamic Polarization
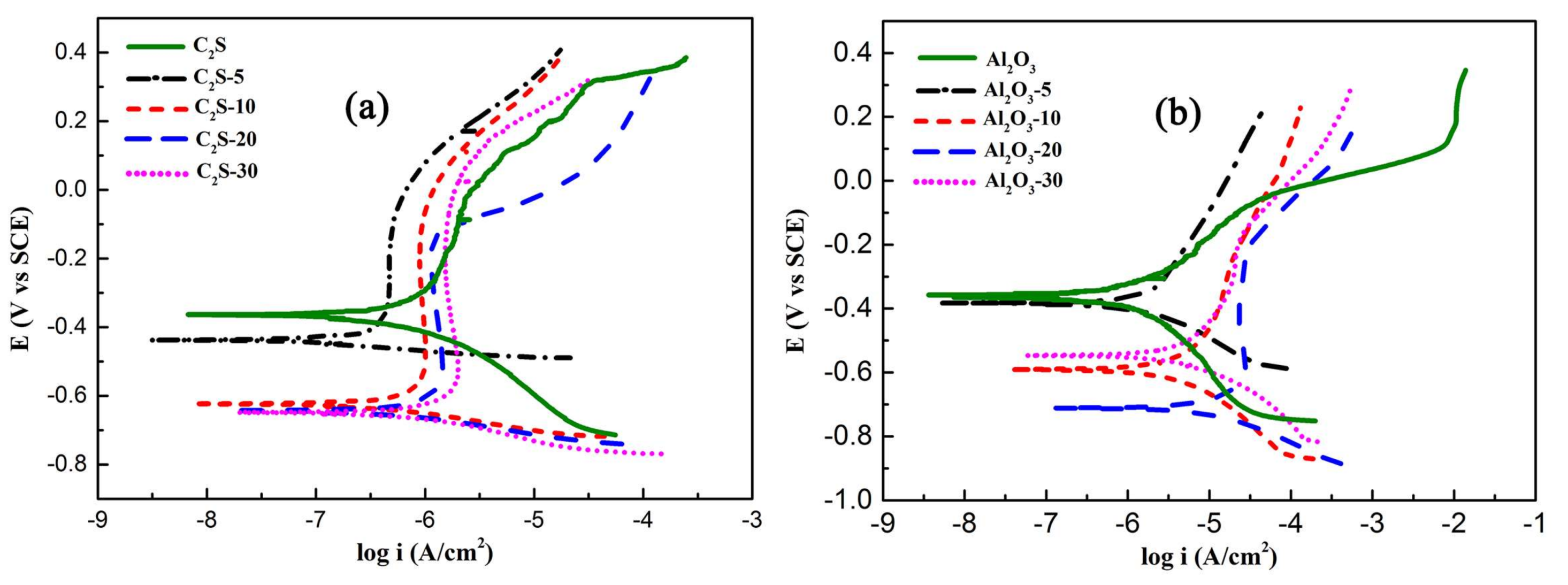
Figure 13 shows the potentiodynamic polarization curves of Ca
2SiO
4 coatings and Al
2O
3 coatings after being immersed in still 3.5 wt % NaCl solution at (35 ± 2) °C for 0, 5, 10, 20 and 30 days. The corrosion parameters for the tested electrodes are given in
Table 2.
Icorr was a key parameter to evaluate the kinetics of the corrosion reaction, which was inversely proportional to the corrosion protection. The polarization resistance (
Rp) is calculated as the slope of the
E vs.
I graph near the
Ecorr. The polarization resistance (
Rp) value of the electrodes was calculated according to the formula:
The Icorr value of as-sprayed Ca2SiO4 coating was 1.24 μA/cm2, then decreased to 0.227 μA/cm2 after being immersed for 5 days. The Icorr value of C2S-30 was 1.61 μA/cm2. The Rp value of as-sprayed Ca2SiO4 coating was 39.53 kΩ·cm2, after being immersed, Rp value of C2S-5 went up to 81.38 kΩ·cm2, the Rp value of C2S-30 was 23.11 kΩ·cm2. The Icorr value of as-sprayed Al2O3 coating was 0.219 μA/cm2. The Icorr of Al2O3 coating went up sharply after being immersed, the Icorr value of Al2O3-20 was 25.5 μA/cm2, the Icorr value of Al2O3-30 was 9.13 μA/cm2, and it was larger than that of C2S coating. The Rp value of as-sprayed Al2O3 coating was 91.22 kΩ·cm2, after being immersed Rp value of Al2O3-5 went down sharply to 23.08 kΩ·cm2, the Rp value of Al2O3-30 was 6.91 kΩ·cm2.
When immersed for 30 days, the corrosion current density of Ca2SiO4 coatings increased slightly. For Ca2SiO4 coating, at the first 20 days, the corrosive medium penetrated to the metal substrate to promote the formation of passive film, which suppressed the corrosion process, but the passive film did not cover the whole metal surface. With the increasing immersion time, more solution arrived at substrate to induce pitting; therefore, the corrosion resistance of C2S-30 reclined. Polarization curves show the shifting of corrosion potential (Ecorr) towards more negative potential with increasing immersed time, indicating that immersed coatings preferentially inhibit the cathodic process.
From
Figure 13a, Ca
2SiO
4 coatings show obvious passivation phenomenon. Ca
2SiO
4 coatings released a lot of Ca(OH)
2 into NaCl aqueous solution. The solution was alkaline and the pH was above 8.0, although the dissolution of CO
2 gas in air may decrease the pH value. The alkaline environment was in favor of the formation of passivation film [
23]. Therefore, Ca
2SiO
4 coating promoted the formation of passivation film and passivation film could protect the metal from corrosion effectively. Only Al
2O
3-20 showed the passive behavior for Al
2O
3 coating. The passivation film might form partly. On the other hand, pitting corrosion occurred easily when the Al
2O
3 coating was loose. So, the corrosion current density was very large, the
Rp was very low. For Al
2O
3-30, the corrosion product possibly blocked up the crack and pores in the coating, the corrosion rate reclined and current became lower;
Rp increased to 6.36 kΩ·cm
2. From the potentiodynamic polarization curves and corrosion parameters of Ca
2SiO
4 and Al
2O
3 coatings, it could be concluded that Ca
2SiO
4 coating was more corrosion-resistant than Al
2O
3 coating.
3.4. Electrochemical Impedance Spectroscopy
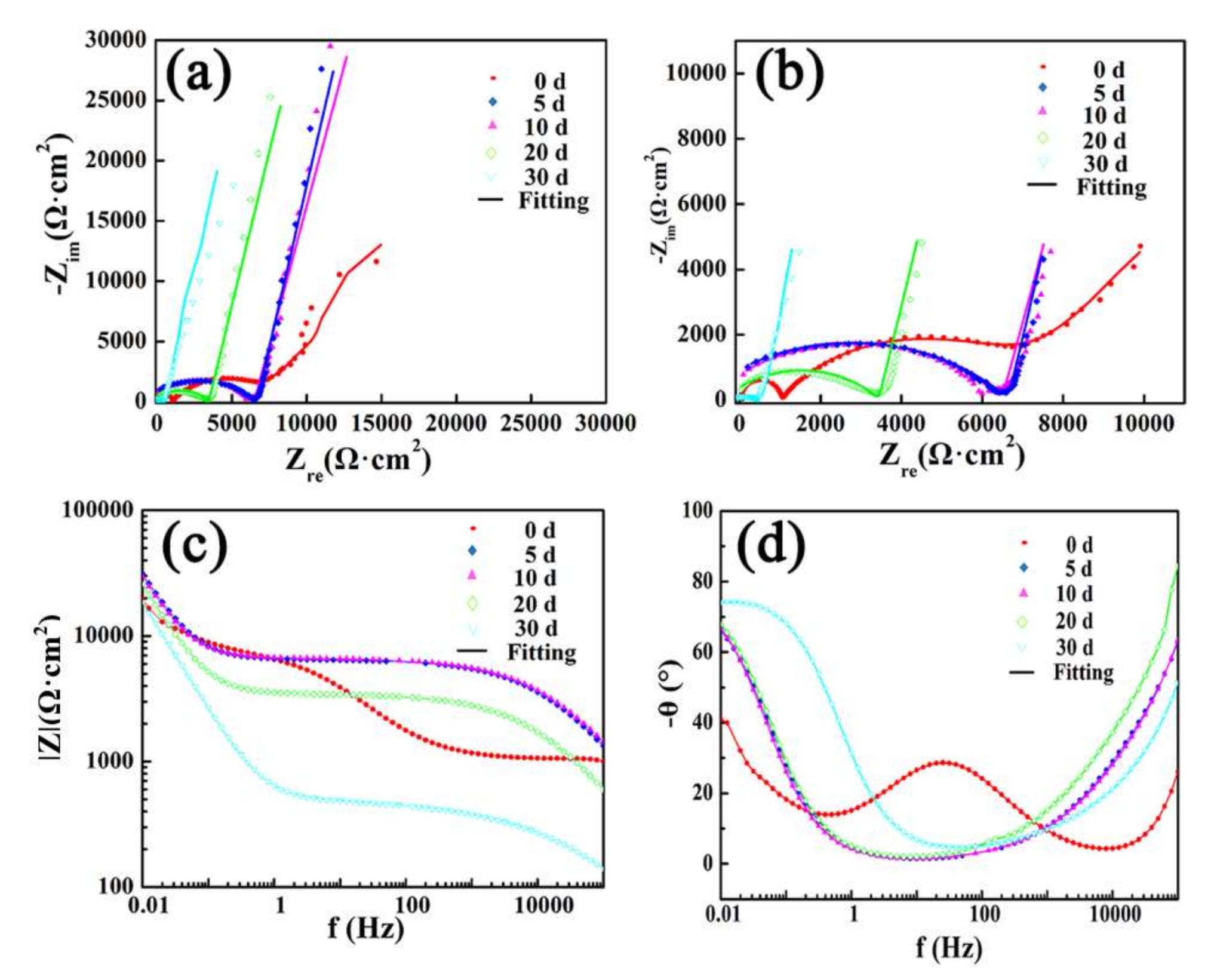
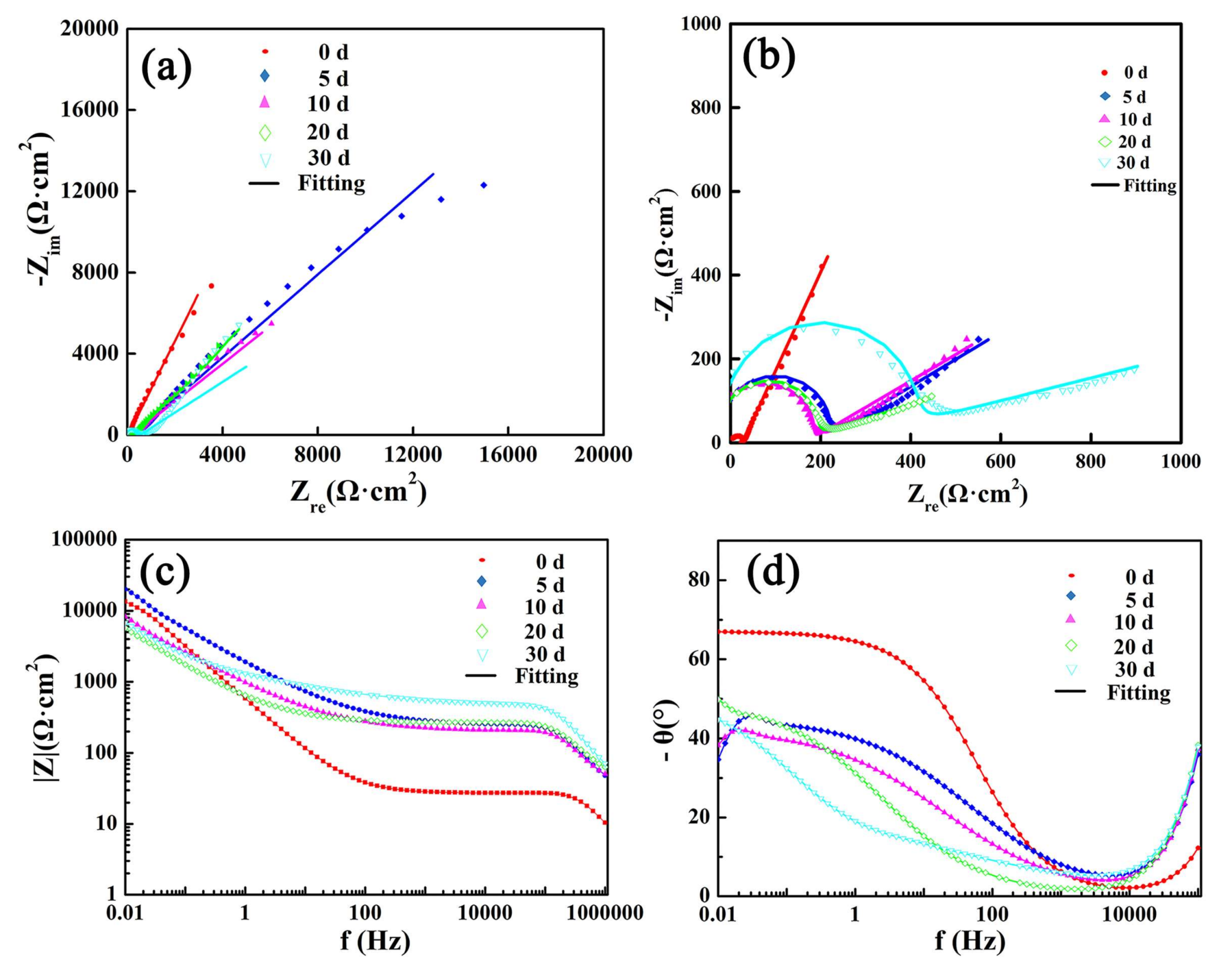
Figure 14 shows the EIS plots of Ca
2SiO
4 coatings after immersion. There was a depressed semi-circle at the high frequencies, it indicated that a CPE was placed in parallel to a resistor. The diameter of the semi-circle increased at the initial 10 days, then decreased with the increase of immersion time. At low frequencies, there was a line.
In
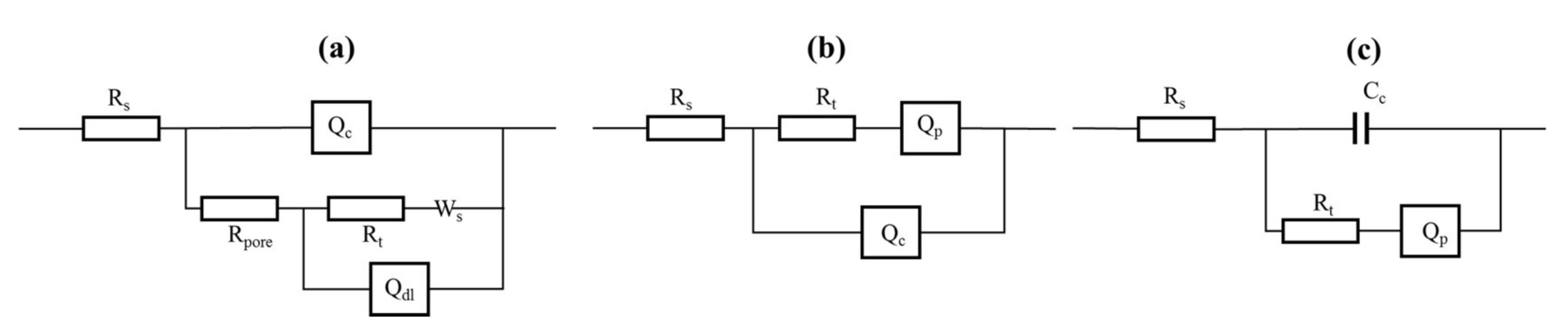
Figure 14, the points present the actual data recorded by the test system, and the solid lines represent fitting data. Furthermore, the lines fit the actual data very well. The equivalent circuits of Ca
2SiO
4 coating are given in
Figure 15. As shown in
Figure 15,
Rs is the resistance of solution;
Rt is the resistance of the charge transfer. The greater the
Rt, the better corrosion-resistance.
Rpore indicates the protective ability of coating, the higher porosity and larger pores would cause low
Rpore;
Qc represents the constant phase element of coating;
Qdl is the double-layer capacitance between substrate and coating interface;
Qp represents the constant phase element of passive film; a capacitor is more suitable for the actual Ca
2SiO
4 coating at the last 30 days, and
Cc is the capacitance of coating;
Ws is a closed-loop diffusion-related element of Warburg impedance, it is used to represent the finite layer diffusion resistance.
For as-sprayed Ca
2SiO
4 coating, the Nyquist plot could be divided into three parts, two depressed semi-circles at high frequencies and a line at low frequencies. The first small depressed semi-circle was produced as
Qc was placed in parallel to
Rpore [
24], and the second large depressed semi-circle was produced as
Qdl was placed in parallel to
Rt, the line is produced by
Ws. The equivalent circuits for as-sprayed Ca
2SiO
4 coating are shown in
Figure 15a. After immersion, passivation film was formed on the substrate, the equivalent circuit was changed, only one capacitive reactance arc occurred. For immersed Ca
2SiO
4 coating, as shown in
Figure 15b, the high-frequencies capacitive reactance arc presents
Rt in parallel with the
Qc; and at low frequencies, the plots show the character of passivation film [
24]. In
Figure 14b, the diameter of the high-frequencies capacitive reactance loop increased at the initial 10 days, then decreased with the increase of immersion time, it indicated that the
Rt became larger firstly and decreased as the corrosion medium penetrated to the internal of coatings.
The corrosion parameters of Ca
2SiO
4 and Al
2O
3 coatings after being immersed in 3.5 wt % NaCl solution for different times are shown in
Table 3. For Ca
2SiO
4 coating, the R
t value of C
2S was 6288 Ω, then it went up after being immersed,
Rt value of C
2S-5 was 7319 Ω,
Rt value of C
2S-10 was 7759 Ω, but it finally went down to 420.6 Ω. At the first 10 days, the corrosive medium penetrated to the metal substrate to promote the formation of passive film, which suppressed charge transfer, giving rise to the
Rt value. The passive film did not cover the whole metal surface, with the increasing immersion time, more solution arrived at substrate to induce pitting, the value of
Rt went down [
25]. At the last 30 days, the value of
Rt was just 420 Ω, which might mean the resistance of solution in the pit. The electrochemical impedance spectroscopies in
Figure 14a are lines with different slopes at low frequencies, which represents the great diffusion resistance of corrosive medium. The larger slope means better capacitive character. The slope is presented by the value of
Qp-p. When the value of
Qp-p was closer to 1.0, the passivation film was denser and thicker. From
Table 3, the value of
Qp-p went up and was closer to 1.0 after being immersed; it is concluded that the passivation film is more like a pure capacitance to hinder the pass of electronic, indicating better corrosion resistance [
25]. From
Figure 14c, it was shown that at 0.01 Hz, the values of impedance modulus increased after immersion, which was consistent with the lines length of EIS at low frequencies in
Figure 14a. If the constant phase element is a pure capacitance, its phase angle will be 90°, and in
Figure 13d, the phase angle went up and was more and more close to 90°, which presents the good capacitance of passive film. The change of phase angle was consistent with the value of
Qp-p.
The EIS results of Al
2O
3 coatings are presented in
Figure 16.
Figure 17 shows the equivalent circuits of Al
2O
3 coatings for different time.
Qc represents the constant phase element of coating; and
Ws is a closed-loop diffusion related element of Warburg impedance. For Al
2O
3 coating, the high-frequencies capacitive reactance arc represents the resistance of
Ws as shown in
Figure 17. In
Figure 16b, the diameter of the high-frequencies capacitive reactance loop was increased with the immersed time indicating that the resistance of
Ws enlarged. The
Ws value of as-sprayed Al
2O
3 coating was 32.45 Ω, and it went up after being immersed.
Ws value of Al
2O
3-5 was 253.4 Ω,
Ws value of Al
2O
3-30 was 414.0 Ω. It might be attributed to the fact that corrosion products blocked the diffusion channel in coatings [
26]. In
Figure 16b, at low frequencies, Nyquist plots of Al
2O
3 coatings also present lines indicating the diffusion resistance of corrosive medium. From
Table 3, the value of
Qc-p went down, the
Qc-p value of as-sprayed Al
2O
3 was 0.726,
Qc-p value of Al
2O
3-5 was 0.506, and it finally went down to 0.411. It was concluded that Al
2O
3 coating became loose and porous. The protection performance of Al
2O
3 coating is weakened. In
Figure 16d, the phase angle went down with the increasing immersed time, which was consistent with the change of the value of
Qc-p.