Porous Zinc Oxide Thin Films: Synthesis Approaches and Applications
Abstract
:1. Introduction
2. Physical Vapor Deposition of Porous ZnO Thin Films
2.1. Sputtering
2.2. Pulsed Laser Deposition
3. Chemical Deposition of Porous ZnO Thin Films
3.1. Spray Pyrolysis
3.2. Electrodeposition
3.3. Sol-Gel Assisted Methods
4. Template-Assisted Methods
5. Others
6. Conclusions and Future Outlooks
Author Contributions
Conflicts of Interest
References
- Zhu, P.; Weng, Z.Y.; Li, X.; Liu, X.M.; Wu, S.L.; Yeung, K.W.K.; Wang, X.B.; Cui, Z.D.; Yang, X.J.; Chu, P.K. Biomedical applications of functionalized ZnO nanomaterials: From biosensors to bioimaging. Adv. Mater. Interfaces 2016, 3, 1500494. [Google Scholar] [CrossRef]
- Laurenti, M.; Cauda, V. ZnO nanostructures for tissue engineering applications. Nanomaterials 2017, 7, 374. [Google Scholar] [CrossRef] [PubMed]
- Janotti, A.; Van de Walle, C.G. Fundamentals of zinc oxide as a semiconductor. Rep. Prog. Phys. 2009, 72, 126501. [Google Scholar] [CrossRef]
- Djurisic, A.B.; Leung, Y.H. Optical properties of ZnO nanostructures. Small 2006, 2, 944–961. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.G.; Yu, X.X. Hydrothermal synthesis and photocatalytic activity of zinc oxide hollow spheres. Environ. Sci. Technol. 2008, 42, 4902–4907. [Google Scholar] [CrossRef] [PubMed]
- Laurenti, M.; Porro, S.; Pirri, C.F.; Ricciardi, C.; Chiolerio, A. Zinc oxide thin films for memristive devices: A review. Crit. Rev. Solid State Mater. Sci. 2017, 42, 153–172. [Google Scholar] [CrossRef]
- Wang, Z.L. Zinc oxide nanostructures: Growth, properties and applications. J. Phys. Condens. Matter 2004, 16, R829–R858. [Google Scholar] [CrossRef]
- Laurenti, M.; Stassi, S.; Lorenzoni, M.; Fontana, M.; Canavese, G.; Cauda, V.; Pirri, C.F. Evaluation of the piezoelectric properties and voltage generation of flexible zinc oxide thin films. Nanotechnology 2015, 26, 215704. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.B. Zinc oxide nanowires. Mater. Charact. 2012, 64, 43–52. [Google Scholar] [CrossRef]
- Li, J.Y.; Chen, X.L.; Li, H.; He, M.; Qiao, Z.Y. Fabrication of zinc oxide nanorods. J. Cryst. Growth 2001, 233, 5–7. [Google Scholar] [CrossRef]
- Dumontel, B.; Canta, M.; Engelke, H.; Chiodoni, A.; Racca, L.; Ancona, A.; Limongi, T.; Canavese, G.; Cauda, V. Enhanced biostability and cellular uptake of zinc oxide nanocrystals shielded with a phospholipid bilayer. J. Mater. Chem. B 2017, 5, 8799–8813. [Google Scholar] [CrossRef]
- Cauda, V.; Pugliese, D.; Garino, N.; Sacco, A.; Bianco, S.; Bella, F.; Lamberti, A.; Gerbaldi, C. Multi-functional energy conversion and storage electrodes using flower-like zinc oxide nanostructures. Energy 2014, 65, 639–646. [Google Scholar] [CrossRef]
- Soci, C.; Zhang, A.; Xiang, B.; Dayeh, S.A.; Aplin, D.P.R.; Park, J.; Bao, X.Y.; Lo, Y.H.; Wang, D. ZnO nanowire UV photodetectors with high internal gain. Nano Lett. 2007, 7, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.Y.; Sun, X.W.; Lo, G.Q.; Kwong, D.L.; Wang, J.X. Improved dye-sensitized solar cells with a ZnO-nanoflower photoanode. Appl. Phys. Lett. 2007, 90, 263501. [Google Scholar] [CrossRef]
- Wang, C.X.; Yin, L.W.; Zhang, L.Y.; Xiang, D.; Gao, R. Metal oxide gas sensors: Sensitivity and influencing factors. Sensors 2010, 10, 2088–2106. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L.; Song, J.H. Piezoelectric nanogenerators based on zinc oxide nanowire arrays. Science 2006, 312, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Ozgur, U.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Dogan, S.; Avrutin, V.; Cho, S.J.; Morkoc, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar] [CrossRef]
- Fu, Y.Q.; Luo, J.K.; Du, X.Y.; Flewitt, A.J.; Li, Y.; Markx, G.H.; Walton, A.J.; Milne, W.I. Recent developments on ZnO films for acoustic wave based bio-sensing and microfluidic applications: A review. Sens. Actuators B Chem. 2010, 143, 606–619. [Google Scholar] [CrossRef]
- Fortunato, E.; Barquinha, P.; Martins, R. Oxide semiconductor thin-film transistors: A review of recent advances. Adv. Mater. 2012, 24, 2945–2986. [Google Scholar] [CrossRef] [PubMed]
- Eranna, G.; Joshi, B.C.; Runthala, D.P.; Gupta, R.P. Oxide materials for development of integrated gas sensors—A comprehensive review. Crit. Rev. Solid State Mater. Sci. 2004, 29, 111–188. [Google Scholar] [CrossRef]
- Tanev, P.T.; Pinnavaia, T.J. Mesoporous silica molecular sieves prepared by ionic and neutral surfactant templating: A comparison of physical properties. Chem. Mater. 1996, 8, 2068–2079. [Google Scholar] [CrossRef]
- Romero-Gomez, P.; Toudert, J.; Sanchez-Valencia, J.R.; Borras, A.; Barranco, A.; Gonzalez-Elipe, A.R. Tunable nanostructure and photoluminescence of columnar ZnO films grown by plasma deposition. J. Phys. Chem. C 2010, 114, 20932–20940. [Google Scholar] [CrossRef]
- Sanchez-Valencia, J.R.; Alcaire, M.; Romero-Gomez, P.; Macias-Montero, M.; Aparicio, F.J.; Borras, A.; Gonzalez-Elipe, A.R.; Barranco, A. Oxygen optical sensing in gas and liquids with nanostructured ZnO thin films based on exciton emission detection. J. Phys. Chem. C 2014, 118, 9852–9859. [Google Scholar] [CrossRef]
- Ramos, F.J.; Lopez-Santos, M.C.; Guillen, E.; Nazeeruddin, M.K.; Gratzel, M.; Gonzalez-Elipe, A.R.; Ahmad, S. Perovskite solar cells based on nanocolumnar plasma–deposited ZnO thin films. Chemphyschem 2014, 15, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Simon, Q.; Barreca, D.; Gasparotto, A.; Maccato, C.; Montini, T.; Gombac, V.; Fornasiero, P.; Lebedev, O.I.; Turner, S.; Van Tendeloo, G. Vertically oriented CuO/ZnO nanorod arrays: From plasma-assisted synthesis to photocatalytic H-2 production. J. Mater. Chem. 2012, 22, 11739–11747. [Google Scholar] [CrossRef]
- Bakrudeen, H.B.; Tsibouklis, J.; Reddy, B.S.R. Facile fabrication of mesoporous ZnO nanospheres for the controlled delivery of captopril. J. Nanoparticle Res. 2013, 15, 1505. [Google Scholar] [CrossRef]
- Zeng, H.B.; Cai, W.P.; Liu, P.S.; Xu, X.X.; Zhou, H.J.; Klingshirn, C.; Kalt, H. ZnO-based hollow nanoparticles by selective etching: Elimination and reconstruction of metal-semiconductor interface, improvement of blue emission and photocatalysis. ACS Nano 2008, 2, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.L.; Luo, Y.N.; Zhang, W.Y.; Du, D.; Lin, Y.H. pH-sensitive ZnO quantum dots-doxorubicin nanoparticles for lung cancer targeted drug delivery. ACS Appl. Mater. Inter. 2016, 8, 22442–22450. [Google Scholar] [CrossRef] [PubMed]
- Park, W.I.; Yi, G.C.; Kim, M.; Pennycook, S.J. Quantum confinement observed in ZnO/ZnMgO nanorod heterostructures. Adv. Mater. 2003, 15, 526–529. [Google Scholar] [CrossRef]
- Wang, Z.L. Splendid one-dimensional nanostructures of zinc oxide: A new nanomaterial family for nanotechnology. ACS Nano 2008, 2, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Gu, Y.; Kuskovsky, I.L.; Andelman, T.; Zhu, Y.; Neumark, G.F.; O’Brien, S. Zinc oxide quantum rods. J. Am. Chem. Soc. 2004, 126, 6206–6207. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Arya, S.K.; Pandey, P.; Malhotra, B.D.; Saha, S.; Sreenivas, K.; Gupta, V. Cholesterol biosensor based on rf sputtered zinc oxide nanoporous thin film. Appl. Phys. Lett. 2007, 91, 063901. [Google Scholar] [CrossRef]
- Lai, Y.F.; Huang, J.H.; Chen, Y.C.; Liu, C.P.; Yang, Y.W. Growth of large-area non-polar ZnO film without constraint to substrate using oblique-angle sputtering deposition. J. Eur. Ceram. Soc. 2013, 33, 1809–1814. [Google Scholar] [CrossRef]
- Sharma, P.; Mansingh, A.; Sreenivas, K. Ultraviolet photoresponse of porous ZnO thin films prepared by unbalanced magnetron sputtering. Appl. Phys. Lett. 2002, 80, 553–555. [Google Scholar] [CrossRef]
- Li, L.; Gao, W.; Reeves, R.J. Zinc oxide films by thermal oxidation of zinc thin films. Surf. Coat. Technol. 2005, 198, 319–323. [Google Scholar] [CrossRef]
- Gazia, R.; Chiodoni, A.; Bianco, S.; Lamberti, A.; Quaglio, M.; Sacco, A.; Tresso, E.; Mandracci, P.; Pirri, C.F. An easy method for the room-temperature growth of spongelike nanostructured Zn films as initial step for the fabrication of nanostructured ZnO. Thin Solid Films 2012, 524, 107–112. [Google Scholar] [CrossRef]
- Lamberti, A.; Sacco, A.; Laurenti, M.; Fontana, M.; Pirri, C.F.; Bianco, S. Sponge-like ZnO nanostructures by low temperature water vapor-oxidation method as dye-sensitized solar cell photoanodes. J. Alloy Comp. 2014, 615, S487–S490. [Google Scholar] [CrossRef]
- Lamberti, A.; Gazia, R.; Sacco, A.; Bianco, S.; Quaglio, M.; Chiodoni, A.; Tresso, E.; Pirri, C.F. Coral-shaped ZnO nanostructures for dye-sensitized solar cell photoanodes. Prog. Photovolt. 2014, 22, 189–197. [Google Scholar] [CrossRef]
- Garino, N.; Lamberti, A.; Gazia, R.; Chiodoni, A.; Gerbaldi, C. Cycling behaviour of sponge-like nanostructured ZnO as thin-film Li-ion battery anodes. J. Alloy Comp. 2014, 615, S454–S458. [Google Scholar] [CrossRef]
- Gazia, R.; Motto, P.; Stassi, S.; Sacco, A.; Virga, A.; Lamberti, A.; Canavese, G. Photodetection and piezoelectric response from hard and flexible sponge-like ZnO-based structures. Nano Energy 2013, 2, 1294–1302. [Google Scholar] [CrossRef]
- Gazia, R.; Canavese, G.; Chiodoni, A.; Lamberti, A.; Stassi, S.; Sacco, A.; Bianco, S.; Virga, A.; Tresso, E.; Pirri, C.F. Novel spongelike nanostructured ZnO films: Properties and applications. J. Alloy Comp. 2014, 586, S331–S335. [Google Scholar] [CrossRef]
- Laurenti, M.; Canavese, G.; Stassi, S.; Fontana, M.; Castellino, M.; Pirri, C.F.; Cauda, V. A porous nanobranched structure: An effective way to improve piezoelectricity in sputtered ZnO thin films. RSC Adv. 2016, 6, 76996. [Google Scholar] [CrossRef]
- Laurenti, M.; Canavese, G.; Sacco, A.; Fontana, M.; Bejtka, K.; Castellino, M.; Pirri, C.F.; Cauda, V. Nanobranched ZnO structure: p-type doping induces piezoelectric voltage generation and ferroelectric-photovoltaic effect. Adv. Mater. 2015, 27, 4218–4223. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Sharma, A.K. Highly c-axis oriented growth and optical characterization of ZnO pore-like structures surrounded by craters via pulsed laser deposition. Silicon 2017, 5, 1–6. [Google Scholar] [CrossRef]
- Hartanto, A.B.; Ning, X.; Nakata, Y.; Okada, T. Growth mechanism of ZnO nanorods from nanoparticles formed in a laser ablation plume. Appl. Phys. A 2004, 78, 299–301. [Google Scholar] [CrossRef]
- Sun, Y.W.; Gospodyn, J.; Kursa, P.; Sit, J.; DeCorby, R.G.; Tsui, Y.Y. Dense and porous ZnO thin films produced by pulsed laser deposition. Appl. Surf. Sci. 2005, 248, 392–396. [Google Scholar] [CrossRef]
- Vinodkumar, R.; Navas, I.; Porsezian, K.; Ganesan, V.; Unnikrishnan, N.V.; Pillai, V.P.M. Structural, spectroscopic and electrical studies of nanostructured porous ZnO thin films prepared by pulsed laser deposition. Spectrochim. Acta A 2014, 118, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Sharma, A.K. Pulsed laser deposition and characterization of ZnO nanopores. Appl. Phys. A 2016, 122, 357. [Google Scholar] [CrossRef]
- Sun, Y.W.; Tsui, Y.Y. Production of porous nanostructured zinc oxide thin films by pulsed laser deposition. Opt. Mater. 2007, 29, 1111–1114. [Google Scholar] [CrossRef]
- Wolcott, A.; Smith, W.A.; Kuykendall, T.R.; Zhao, Y.P.; Zhang, J.Z. Photoelectrochemical study of nanostructured ZnO thin films for hydrogen generation from water splitting. Adv. Funct. Mater. 2009, 19, 1849–1856. [Google Scholar] [CrossRef]
- Labis, J.P.; Hezam, M.; Al-Anazi, A.; Al-Brithen, H.; Ansari, A.A.; El-Toni, A.M.; Enriquez, R.; Jacopin, G.; Al-Hoshan, M. Pulsed laser deposition growth of 3D ZnO nanowall network in nest-like structures by two-step approach. Sol. Energy Mater. Sol. Cells 2015, 143, 539–545. [Google Scholar] [CrossRef]
- El Zein, B.; Boulfrad, S.; Jabbour, G.E.; Dogheche, E. Parametric study of self-forming ZnO nanowall network with honeycomb structure by pulsed laser deposition. Appl. Surf. Sci. 2014, 292, 598–607. [Google Scholar] [CrossRef]
- Han, B.S.; Caliskan, S.; Sohn, W.; Kim, M.; Lee, J.K.; Jang, H.W. Room temperature deposition of crystalline nanoporous ZnO nanostructures for direct use as flexible DSSC photoanode. Nanoscale Res. Lett. 2016, 11, 221. [Google Scholar] [CrossRef] [PubMed]
- Achkar, A.; Jedrzejowski, P.; Pignolet, A.; Sarkissian, A. Porous and dense ZnO films produced by femtosecond and picosecond pulsed laser deposition. J. Optoelectron. Adv. Mater. 2010, 12, 646–649. [Google Scholar]
- Dhamodharan, P.; Manoharan, C.; Dhanapandian, S.; Venkatachalam, P. Dye-sensitized solar cell using sprayed ZnO nanocrystalline thin films on ITO as photoanode. Spectrochim. Acta A 2015, 136, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Studenikin, S.A.; Golego, N.; Cocivera, M. Fabrication of green and orange photoluminescent, undoped ZnO films using spray pyrolysis. J. Appl. Phys. 1998, 84, 2287–2294. [Google Scholar] [CrossRef]
- Dobrozhan, O.; Kurbatov, D.; Opanasyuk, A.; Cheong, H.; Cabot, A. Influence of substrate temperature on the structural and optical properties of crystalline ZnO films obtained by pulsed spray pyrolysis. Surf. Interface Anal. 2015, 47, 601–606. [Google Scholar] [CrossRef]
- Zahedi, F.; Dariani, R.S.; Rozati, S.M. Structural, optical and electrical properties of ZnO thin films prepared by spray pyrolysis: Effect of precursor concentration. Bull. Mater. Sci. 2014, 37, 433–439. [Google Scholar] [CrossRef]
- Benramache, S.; Rahal, A.; Benhaoua, B. The effects of solvent nature on spray-deposited ZnO thin film prepared from Zn(CH3COO)2, 2H2O. Optik 2014, 125, 663–666. [Google Scholar] [CrossRef]
- Mani, G.K.; Rayappan, J.B.B. Impact of annealing duration on spray pyrolysis deposited nanostructured zinc oxide thin films. Superlattice Microstruct. 2014, 67, 82–87. [Google Scholar] [CrossRef]
- Shaban, Z.; Ara, M.H.M.; Falahatdoost, S.; Ghazyani, N. Optimization of ZnO thin film through spray pyrolysis technique and its application as a blocking layer to improving dye sensitized solar cell efficiency. Curr. Appl. Phys. 2016, 16, 131–134. [Google Scholar] [CrossRef]
- Mani, G.K.; Rayappan, J.B.B. Novel and facile synthesis of randomly interconnected ZnO nanoplatelets using spray pyrolysis and their room temperature sensing characteristics. Sens. Actuators B Chem. 2014, 198, 125–133. [Google Scholar] [CrossRef]
- Mani, G.K.; Rayappan, J.B.B. A highly selective room temperature ammonia sensor using spray deposited zinc oxide thin film. Sens. Actuators B Chem. 2013, 183, 459–466. [Google Scholar] [CrossRef]
- Mariappan, R.; Ponnuswamy, V.; Suresh, P.; Ashok, N.; Jayamurugan, P.; Bose, A.C. Influence of film thickness on the properties of sprayed ZnO thin films for gas sensor applications. Superlattice Microstruct. 2014, 71, 238–249. [Google Scholar] [CrossRef]
- Shewale, P.S.; Agawane, G.L.; Shin, S.W.; Moholkar, A.V.; Lee, J.Y.; Kim, J.H.; Uplane, M.D. Thickness dependent H2S sensing properties of nanocrystalline ZnO thin films derived by advanced spray pyrolysis. Sens. Actuators B Chem. 2013, 177, 695–702. [Google Scholar] [CrossRef]
- Tarwal, N.L.; Rajgure, A.V.; Inamdar, A.I.; Devan, R.S.; Kim, I.Y.; Suryavanshi, S.S.; Ma, Y.R.; Kim, J.H.; Patil, P.S. Growth of multifunctional ZnO thin films by spray pyrolysis technique. Sens. Actuators A Phys. 2013, 199, 67–73. [Google Scholar] [CrossRef]
- Ammaih, Y.; Lfakir, A.; Hartiti, B.; Ridah, A.; Thevenin, P.; Siadat, M. Structural, optical and electrical properties of ZnO:Al thin films for optoelectronic applications. Opt. Quant. Electron. 2014, 46, 229–234. [Google Scholar] [CrossRef]
- Mahadik, M.A.; Hunge, Y.M.; Shinde, S.S.; Rajpure, K.Y.; Bhosale, C.H. Semiconducting properties of aluminum-doped ZnO thin films grown by spray pyrolysis technique. J. Semicond. 2015, 36, 033002. [Google Scholar] [CrossRef]
- Allah, F.K.; Abe, S.Y.; Nunez, C.M.; Khelil, A.; Cattin, L.; Morsli, M.; Bernede, J.C.; Bougrine, A.; del Valle, M.A.; Diaz, F.R. Characterisation of porous doped ZnO thin films deposited by spray pyrolysis technique. Appl. Surf. Sci. 2007, 253, 9241–9247. [Google Scholar] [CrossRef]
- Hameed, M.S.; Princice, J.J.; Babu, N.R.; Arunachalam, A. Effect of silver doping on optical properties of nanoflower ZnO thin films prepared by spray pyrolysis technique. J. Mater. Sci. Mater. Electron. 2017, 28, 8675–8683. [Google Scholar] [CrossRef]
- Mariappan, R.; Ponnuswamy, V.; Suresh, R.; Suresh, P.; Bose, A.C.; Ragavendar, M. Role of substrate temperature on the properties of Na-doped ZnO thin film nanorods and performance of ammonia gas sensors using nebulizer spray pyrolysis technique. J. Alloy Comp. 2014, 582, 387–391. [Google Scholar] [CrossRef]
- Kurtaran, S.; Aldag, S.; Ofofoglu, G.; Akyuz, I.; Atay, F. Transparent conductive ZnO thin films grown by chemical spray pyrolysis: The effect of Mg. J. Mater. Sci. Mater. Electron. 2016, 27, 8478–8485. [Google Scholar] [CrossRef]
- Dhasmana, H.; Shital, S.; Dutta, V. ZnO nanostructure thin films by continuous spray pyrolysis using doped precursor for Si solar cell application. J. Mater. Sci. Mater. Electron. 2014, 25, 4095–4102. [Google Scholar] [CrossRef]
- Shewale, P.S.; Yu, Y.S. H2S gas sensing properties of undoped and Ti doped ZnO thin films deposited by chemical spray pyrolysis. J. Alloy Comp. 2016, 684, 428–437. [Google Scholar] [CrossRef]
- Mani, G.K.; Rayappan, J.B.B. Facile synthesis of ZnO nanostructures by spray pyrolysis technique and its application as highly selective H2S sensor. Mater. Lett. 2015, 158, 373–376. [Google Scholar] [CrossRef]
- Shewale, P.S.; Patil, V.B.; Shin, S.W.; Kim, J.H.; Uplane, M.D. H2S gas sensing properties of nanocrystalline Cu-doped ZnO thin films prepared by advanced spray pyrolysis. Sens. Actuators B Chem. 2013, 186, 226–234. [Google Scholar] [CrossRef]
- Mani, G.K.; Rayappan, J.B.B. Selective detection of ammonia using spray pyrolysis deposited pure and nickel doped ZnO thin films. Appl. Surf. Sci. 2014, 311, 405–412. [Google Scholar] [CrossRef]
- Muniyandi, I.; Mani, G.K.; Shankar, P.; Rayappan, J.B.B. Effect of nickel doping on structural, optical, electrical and ethanol sensing properties of spray deposited nanostructured ZnO thin films. Ceram. Int. 2014, 40, 7993–8001. [Google Scholar] [CrossRef]
- Zhang, X.M.; Dong, Z.J.; Liu, S.; Yan, S.; Dong, Y.H.; Wei, F. Maize straw-templated hierarchical porous ZnO:Ni with enhanced acetone gas sensing properties. Sens. Actuators B Chem. 2017, 243, 1224–1230. [Google Scholar] [CrossRef]
- El khalidi, Z.; Comini, E.; Hartiti, B.; Moumen, A.; Munasinghe Arachchige, H.M.M.; Fadili, S.; Thevenin, P.; Kamal, A. Effect of vanadium doping on ZnO sensing properties synthesized by spray pyrolysis. Mater. Des. 2018, 139, 56–64. [Google Scholar] [CrossRef]
- Kulandaisamy, A.J.; Reddy, J.R.; Srinivasan, P.; Babu, K.J.; Mani, G.K.; Shankar, P.; Rayappan, J.B.B. Room temperature ammonia sensing properties of ZnO thin films grown by spray pyrolysis: Effect of Mg doping. J. Alloy Comp. 2016, 688, 422–429. [Google Scholar] [CrossRef]
- Duta, M.; Perniu, D.; Duta, A. Photocatalytic zinc oxide thin films obtained by surfactant assisted spray pyrolysis deposition. Appl. Surf. Sci. 2014, 306, 80–88. [Google Scholar] [CrossRef]
- Ravichandran, K.; Mohan, R.; Sakthivel, B.; Varadharajaperumal, S.; Devendran, P.; Alagesan, T.; Pandian, K. Enhancing the photocatalytic efficiency of sprayed ZnO thin films through double doping (Sn plus F) and annealing under different ambiences. Appl. Surf. Sci. 2014, 321, 310–317. [Google Scholar] [CrossRef]
- Dhamodharan, P.; Manoharan, C.; Dhanapandian, S.; Bououdina, M.; Ramalingam, S. Preparation and characterization of spray deposited Sn-doped ZnO thin films onto ITO subtracts as photoanode in dye sensitized solar cell. J. Mater. Sci. Mater. Electron. 2015, 26, 4830–4839. [Google Scholar] [CrossRef]
- Manoharan, C.; Pavithra, G.; Dhanapandian, S.; Dhamodharan, P. Effect of in doping on the properties and antibacterial activity of ZnO films prepared by spray pyrolysis. Spectrochim. Acta A 2015, 149, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Vasanthi, M.; Ravichandran, K.; Begum, N.J.; Muruganantham, G.; Snega, S.; Panneerselvam, A.; Kavitha, P. Influence of Sn doping level on antibacterial activity and certain physical properties of ZnO films deposited using a simplified spray pyrolysis technique. Superlattice Microstruct. 2013, 55, 180–190. [Google Scholar] [CrossRef]
- Michaelis, E.; Wohrle, D.; Rathousky, J.; Wark, M. Electrodeposition of porous zinc oxide electrodes in the presence of sodium laurylsulfate. Thin Solid Films 2006, 497, 163–169. [Google Scholar] [CrossRef]
- Kim, Y.K.; Hwang, S.H.; Kim, S.; Park, H.; Lim, S.K. ZnO nanostructure electrodeposited on flexible conductive fabric: A flexible photo-sensor. Sens. Actuators B Chem. 2017, 240, 1106–1113. [Google Scholar] [CrossRef]
- Lv, J.G.; Wang, W.H.; Zhao, M.; Zhu, W.L.; Cheng, Y.B.; He, G.; Zhang, M.; Chen, X.S.; Sun, Z.Q. Enhanced visible light response of ZnO porous thin film by post-annealing treatment. J. Mater. Sci. Mater. Electron. 2017, 28, 4051–4057. [Google Scholar] [CrossRef]
- Reemts, J.; Kittel, A. Persistent photoconductivity in highly porous ZnO films. J. Appl. Phys. 2007, 101, 013709. [Google Scholar] [CrossRef]
- Chen, Z.G.; Tang, Y.W.; Zhang, L.S.; Luo, L.J. Electrodeposited nanoporous ZnO films exhibiting enhanced performance in dye-sensitized solar cells. Electrochim. Acta 2006, 51, 5870–5875. [Google Scholar] [CrossRef]
- Venditti, L.; Barbero, N.; Russo, M.V.; Di Carlo, A.; Decker, F.; Fratoddi, I.; Barolo, C.; Dini, D. Electrodeposited ZnO with squaraine sentisizers as photoactive anode of DSCs. Mater. Res. Express 2014, 1, 015040. [Google Scholar] [CrossRef]
- Dunkel, C.; Wark, M.; Oekermann, T.; Ostermann, R.; Smarsly, B.M. Electrodeposition of zinc oxide on transparent conducting metal oxide nanofibers and its performance in dye sensitized solar cells. Electrochim. Acta 2013, 90, 375–381. [Google Scholar] [CrossRef]
- Pauporte, T.; Rathousky, J. Electrodeposited mesoporous ZnO thin films as efficient photocatalysts for the degradation of dye pollutants. J. Phys. Chem. C 2007, 111, 7639–7644. [Google Scholar] [CrossRef]
- Dikici, T. Temperature-dependent growth of ZnO structures by thermal oxidation of Zn coatings electrodeposited on steel substrates and their photocatalytic activities. Ceram. Int. 2017, 43, 8289–8293. [Google Scholar] [CrossRef]
- Wanotayan, T.; Panpranot, J.; Qin, J.; Boonyongmaneerat, Y. Microstructures and photocatalytic properties of ZnO films fabricated by Zn electrodeposition and heat treatment. Mater. Sci. Semicond. Proc. 2018, 74, 232–237. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, M.; Guo, M. Controllable electrodeposition of ZnO nanorod arrays on flexible stainless steel mesh substrate for photocatalytic degradation of Rhodamine B. Appl. Surf. Sci. 2014, 317, 672–681. [Google Scholar] [CrossRef]
- Yang, J.; Wang, Y.Q.; Kong, J.H.; Jia, H.X.; Wang, Z.S. Synthesis of ZnO nanosheets via electrodeposition method and their optical properties, growth mechanism. Opt. Mater. 2015, 46, 179–185. [Google Scholar] [CrossRef]
- Li, H.; Yu, S.R.; Han, X.X. Preparation of a biomimetic superhydrophobic ZnO coating on an X90 pipeline steel surface. New J. Chem. 2015, 39, 4860–4868. [Google Scholar]
- Li, M.; Zhai, J.; Liu, H.; Song, Y.L.; Jiang, L.; Zhu, D.B. Electrochemical deposition of conductive superhydrophobic zinc oxide thin films. J. Phys. Chem. B 2003, 107, 9954–9957. [Google Scholar] [CrossRef]
- Pal, B.; Sharon, M. Enhanced photocatalytic activity of highly porous ZnO thin films prepared by sol-gel process. Mater. Chem. Phys. 2002, 76, 82–87. [Google Scholar] [CrossRef]
- Wang, C.F.; Tzeng, F.S.; Chen, H.G.; Chang, C.J. Ultraviolet-durable superhydrophobic zinc oxide-coated mesh films for surface and underwater-oil capture and transportation. Langmuir 2012, 28, 10015–10019. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, S.R.; Xu, M.J.; Wang, Y.; Zhu, B.L.; Zhang, S.M.; Huang, W.P.; Wu, S.H. Hierarchically porous ZnO architectures for gas sensor application. Cryst. Growth Des. 2009, 9, 3532–3537. [Google Scholar] [CrossRef]
- Chatterjee, A.P.; Mitra, P.; Mukhopadhyay, A.K. Chemically deposited zinc oxide thin film gas sensor. J. Mater. Sci. 1999, 34, 4225–4231. [Google Scholar] [CrossRef]
- Hosono, E.; Fujihara, S.; Kimura, T.; Imai, H. Growth of layered basic zinc acetate in methanolic solutions and its pyrolytic transformation into porous zinc oxide films. J. Colloid Interface Sci. 2004, 272, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.H.; Xie, C.S. Controlled fabrication of nanostructured ZnO particles and porous thin films via a modified chemical bath deposition method. J. Cryst. Growth 2006, 291, 187–195. [Google Scholar] [CrossRef]
- Wang, H.H.; Dong, S.J.; Chang, Y.; Zhou, X.P.; Hu, X.B. Microstructures and photocatalytic properties of porous ZnO films synthesized by chemical bath deposition method. Appl. Surf. Sci. 2012, 258, 4288–4293. [Google Scholar] [CrossRef]
- Ottone, C.; Bejtka, K.; Chiodoni, A.; Farias, V.; Roppolo, I.; Canavese, G.; Stassi, S.; Cauda, V. Comprehensive study of the templating effect on the ZnO nanostructure formation within porous hard membranes. New J. Chem. 2014, 38, 2058–2065. [Google Scholar] [CrossRef]
- Ottone, C.; Rivera, V.F.; Fontana, M.; Bejtka, K.; Onida, B.; Cauda, V. Ultralong and mesoporous ZnO and gamma-Al2O3 oriented nanowires obtained by template-assisted hydrothermal approach. J. Mater. Sci. Technol. 2014, 30, 1167–1173. [Google Scholar] [CrossRef]
- Ryu, H.W.; Park, B.S.; Akbar, S.A.; Lee, W.S.; Hong, K.J.; Seo, Y.J.; Shin, D.C.; Park, J.S.; Choi, G.P. ZnO sol-gel derived porous film for CO gas sensing. Sens. Actuators B Chem. 2003, 96, 717–722. [Google Scholar] [CrossRef]
- Leone, F.; Gignone, A.; Ronchetti, S.; Cavalli, R.; Manna, L.; Banchero, M.; Onida, B. A green organic-solvent-free route to prepare nanostructured zinc oxide carriers of clotrimazole for pharmaceutical applications. J. Clean. Prod. 2018, 172, 1433–1439. [Google Scholar] [CrossRef]
- Liu, Z.F.; Jin, Z.G.; Li, W.; Qiu, J.J.; Zhao, J.; Liu, X.X. Synthesis of PS colloidal crystal templates and ordered ZnO porous thin films by dip-drawing method. Appl. Surf. Sci. 2006, 252, 5002–5009. [Google Scholar] [CrossRef]
- Yan, H.W.; Yang, Y.L.; Fu, Z.P.; Yang, B.F.; Xia, L.S.; Fu, S.Q.; Li, F.Q. Fabrication of 2D and 3D ordered porous ZnO films using 3D opal templates by electrodeposition. Electrochem. Commun. 2005, 7, 1117–1121. [Google Scholar] [CrossRef]
- Liu, Z.F.; Jin, Z.G.; Li, W.; Qiu, J.J. Preparation of ZnO porous thin films by sol-gel method using PEG template. Mater. Lett. 2005, 59, 3620–3625. [Google Scholar] [CrossRef]
- Liu, Z.F.; Jin, Z.G.; Qiu, J.J.; Liu, X.X.; Wu, W.B.; Li, W. Preparation and characteristics of ordered porous ZnO films by a electrodeposition method using PS array templates. Semicond. Sci. Technol. 2006, 21, 60–66. [Google Scholar] [CrossRef]
- Liu, Z.F.; Jin, Z.G.; Li, W.; Liu, X.X. Ordered porous ZnO thin films formed by dip-coating method using PS templates. J. Sol-Gel Sci. Technol. 2006, 40, 25–30. [Google Scholar] [CrossRef]
- Cao, B.Q.; Cai, W.P.; Sun, F.Q.; Li, Y.; Lei, Y.; Zhang, L.D. Fabrication of large-scale zinc oxide ordered pore arrays with controllable morphology. Chem. Commun. 2004, 1604–1605. [Google Scholar] [CrossRef] [PubMed]
- Eltayeb, A.; Daniels, S.; McGlynn, E. Enhanced optical properties of ZnO and CeO2-coated ZnO nanostructures achieved via spherical nanoshells growth on a polystyrene template. Sci. Rep. 2017, 7, 3737. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.Y.; Kang, F.; Ke, Z.; Zhuo, S.Y.; Liang, C.F.; Zhang, Z.X. Fabrication and characterization of three-dimensional core-shell structure ZnO photonic crystals by magnetron sputtering based on opal template. J. Cryst. Growth 2010, 312, 2484–2488. [Google Scholar]
- Lin, X.; Chen, M. Fabrication and photo-detecting performance of 2D ZnO inverse opal films. Appl. Sci. 2016, 6, 259. [Google Scholar] [CrossRef]
- Wang, Z.H.; Tian, Z.W.; Han, D.M.; Gu, F.B. Highly sensitive and selective ethanol sensor fabricated with In-doped 3DOM ZnO. ACS Appl. Mater. Inter. 2016, 8, 5466–5474. [Google Scholar] [CrossRef] [PubMed]
- Su, X.S.; Duan, G.T.; Xu, Z.K.; Zhou, F.; Cai, W.P. Structure and thickness-dependent gas sensing responses to NO2 under UV irradiation for the multilayered ZnO micro/nanostructured porous thin films. J. Colloid Interface Sci. 2017, 503, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Li, Q.Q.; Meng, J.; Han, X.; Wu, Y.Q.; Wang, S.J.; Qian, L.; Du, Z.L. Interface engineering for efficient charge collection in Cu2O/ZnO heterojunction solar cells with ordered ZnO cavity-like nanopatterns. Sol. Energy Mater. Sol. Cells 2013, 116, 120–125. [Google Scholar] [CrossRef]
- Kim, E.; Vaynzof, Y.; Sepe, A.; Guldin, S.; Scherer, M.; Cunha, P.; Roth, S.V.; Steiner, U. Gyroid-structured 3D ZnO networks made by atomic layer deposition. Adv. Funct. Mater. 2014, 24, 863–872. [Google Scholar] [CrossRef]
- Yong, J.L.; Chen, F.; Yang, Q.; Fang, Y.; Huo, J.L.; Hou, X. Femtosecond laser induced hierarchical ZnO superhydrophobic surfaces with switchable wettability. Chem. Commun. 2015, 51, 9813–9816. [Google Scholar] [CrossRef] [PubMed]
- Balela, M.D.L.; Pelicano, C.M.O.; Ty, J.D.; Yanagi, H. Formation of zinc oxide nanostructures by wet oxidation of vacuum deposited Zn thin film. Opt. Quant. Electron. 2017, 49, 3. [Google Scholar] [CrossRef]
- Alcaire, M.; Filippin, A.N.; Macias-Montero, M.; Sanchez-Valencia, J.R.; Rojas, T.C.; Mora-Boza, A.; Lopez-Santos, C.; Espinos, J.P.; Barranco, A.; Borras, A. Highly porous ZnO thin films and 1D nanostructures by remote plasma processing of Zn-phthalocyanine. Plasma. Process. Polym. 2016, 13, 287–297. [Google Scholar] [CrossRef]
- Wang, H.J.; Sun, Y.Y.; Cao, Y.; Yu, X.H.; Ji, X.M.; Yang, L. Porous zinc oxide films: Controlled synthesis, cytotoxicity and photocatalytic activity. Chem. Eng. J. 2011, 178, 8–14. [Google Scholar] [CrossRef]
- Sacco, A.; Lamberti, A.; Gazia, R.; Bianco, S.; Manfredi, D.; Shahzad, N.; Cappelluti, F.; Ma, S.; Tresso, E. High efficiency dye-sensitized solar cells exploiting sponge-like ZnO nanostructures. Phys. Chem. Chem. Phys. 2012, 14, 16203–16208. [Google Scholar] [CrossRef] [PubMed]



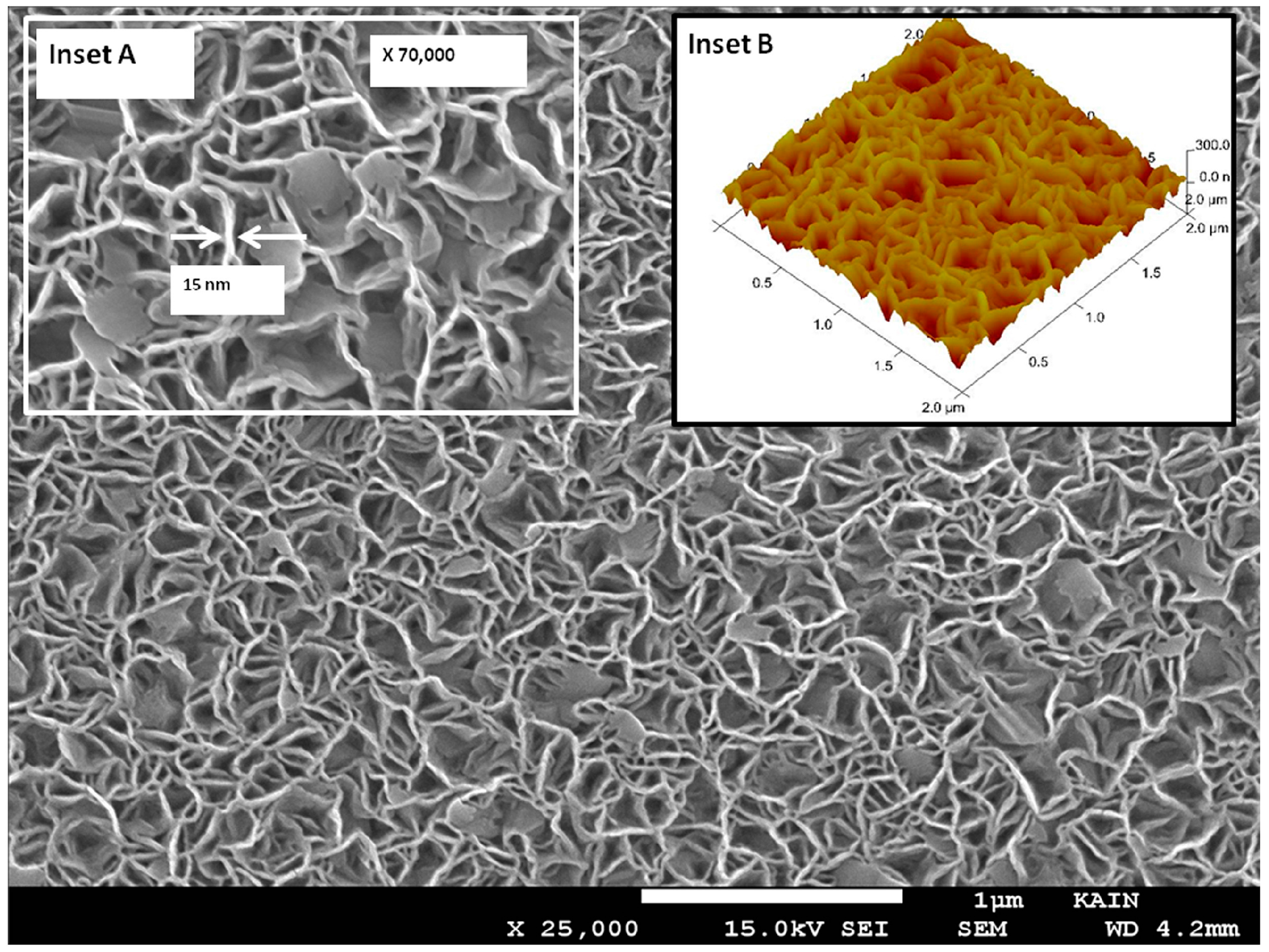


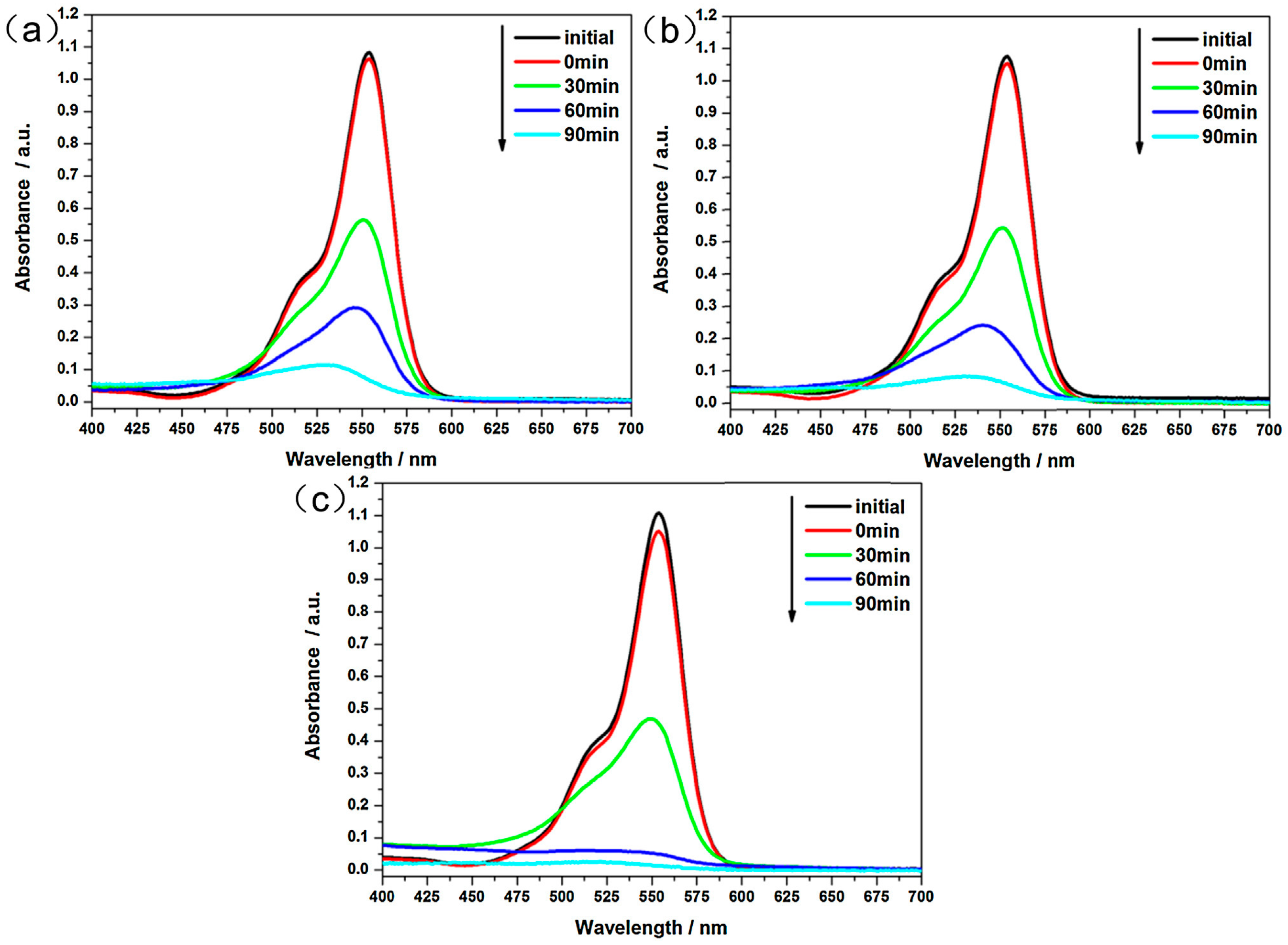



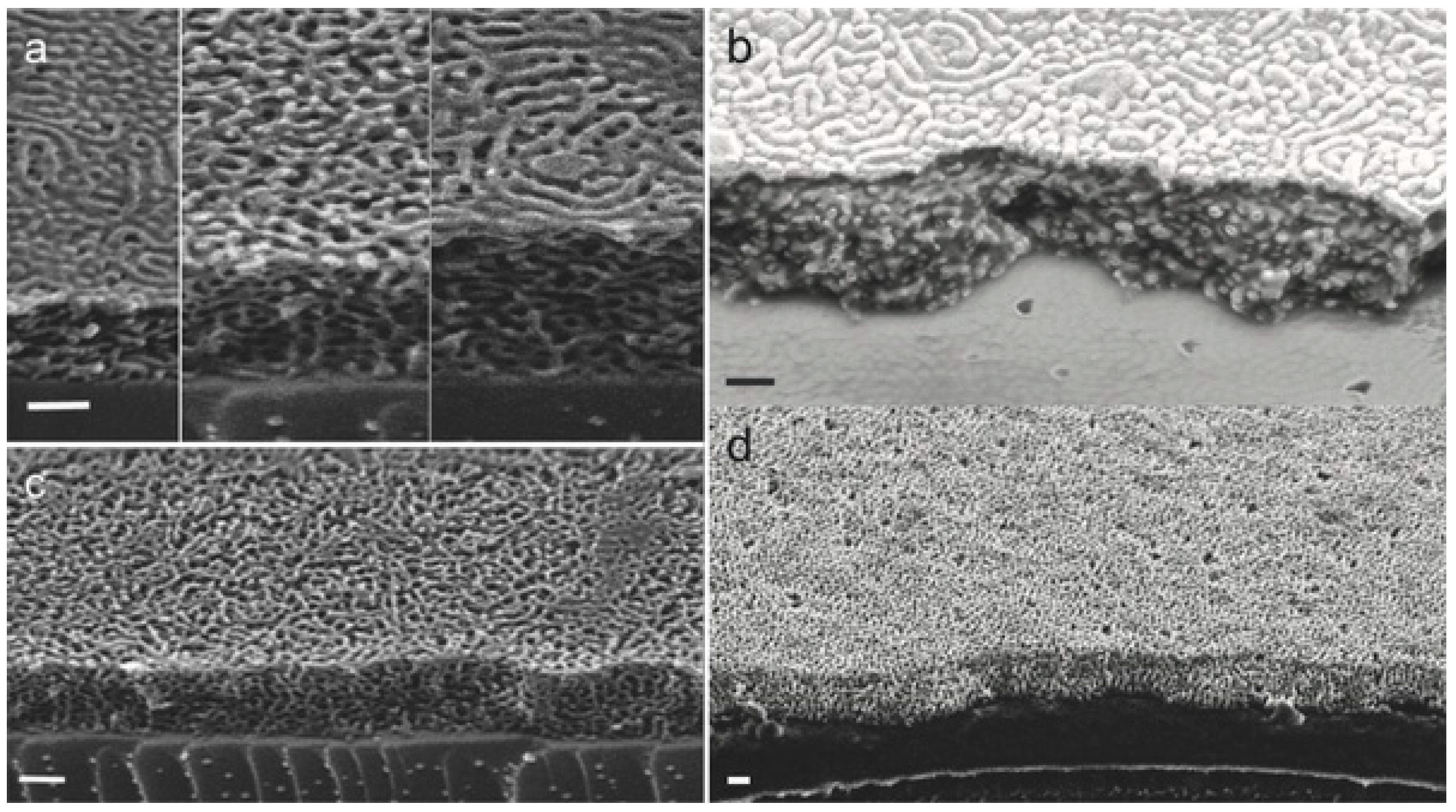
| Synthetic Approach | Porous Structure | Ref. | ||
|---|---|---|---|---|
| Type of Porosity 1 | Pore Size, Specific Surface Area (SSA) | Analyses Methods | ||
| Sputtering | Meso | 4 nm | AFM | [32] |
| Macro/Meso | <100 nm | FESEM, HRTEM | [33] | |
| Macro/Meso | 50–100 nm | FESEM | [35] | |
| Meso | ~27 nm, SSA 14 m2·g−1 | FESEM, N2 adsorption | [43,129] | |
| Pulsed laser deposition | Meso | <50 nm | FESEM, AFM | [44] |
| Macro | 113–184 nm | FESEM, AFM | [48] | |
| Macro | 50–140 nm | SEM | [52] | |
| Macro/Meso | 10–100 nm | FESEM | [53] | |
| Spray pyrolysis | Macro | ~100 nm | FESEM | [64] |
| Meso | 10–25 nm, SSA 28.88 m2·g−1 | N2 adsorption | [79] | |
| Electro deposition | Meso | 10–20 nm | FESEM | [92] |
| Meso | <50 nm | SEM, TEM | [93] | |
| Meso | <8 nm, SSA 20–140 m2·g−1 | Kr adsorption | [94] | |
| Sol-gel | Meso | ~7 nm, SSA 37.47 m2·g−1 | N2 adsorption | [103] |
| Macro | ~100 nm | FESEM | [105] | |
| Template-assisted | Macro | >1 μm | FESEM | [112,113,116,117] |
| Meso | 30 nm | FESEM | [124] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laurenti, M.; Cauda, V. Porous Zinc Oxide Thin Films: Synthesis Approaches and Applications. Coatings 2018, 8, 67. https://doi.org/10.3390/coatings8020067
Laurenti M, Cauda V. Porous Zinc Oxide Thin Films: Synthesis Approaches and Applications. Coatings. 2018; 8(2):67. https://doi.org/10.3390/coatings8020067
Chicago/Turabian StyleLaurenti, Marco, and Valentina Cauda. 2018. "Porous Zinc Oxide Thin Films: Synthesis Approaches and Applications" Coatings 8, no. 2: 67. https://doi.org/10.3390/coatings8020067
APA StyleLaurenti, M., & Cauda, V. (2018). Porous Zinc Oxide Thin Films: Synthesis Approaches and Applications. Coatings, 8(2), 67. https://doi.org/10.3390/coatings8020067





