Antireflection Coatings for Strongly Curved Glass Lenses by Atomic Layer Deposition
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Characterization of ALD Thin Films
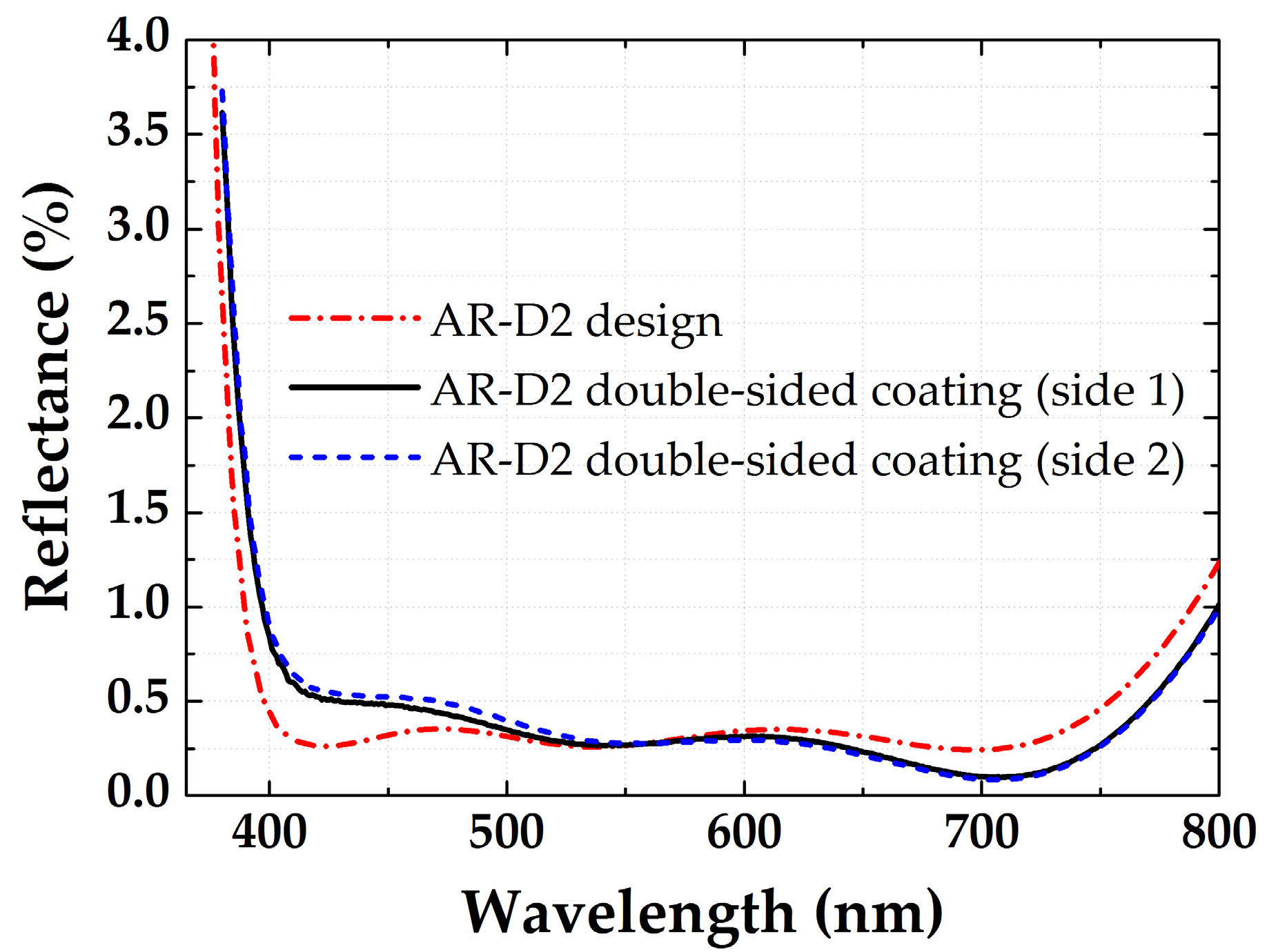
3.2. Antireflection Coatings on Plane Glass Substrates
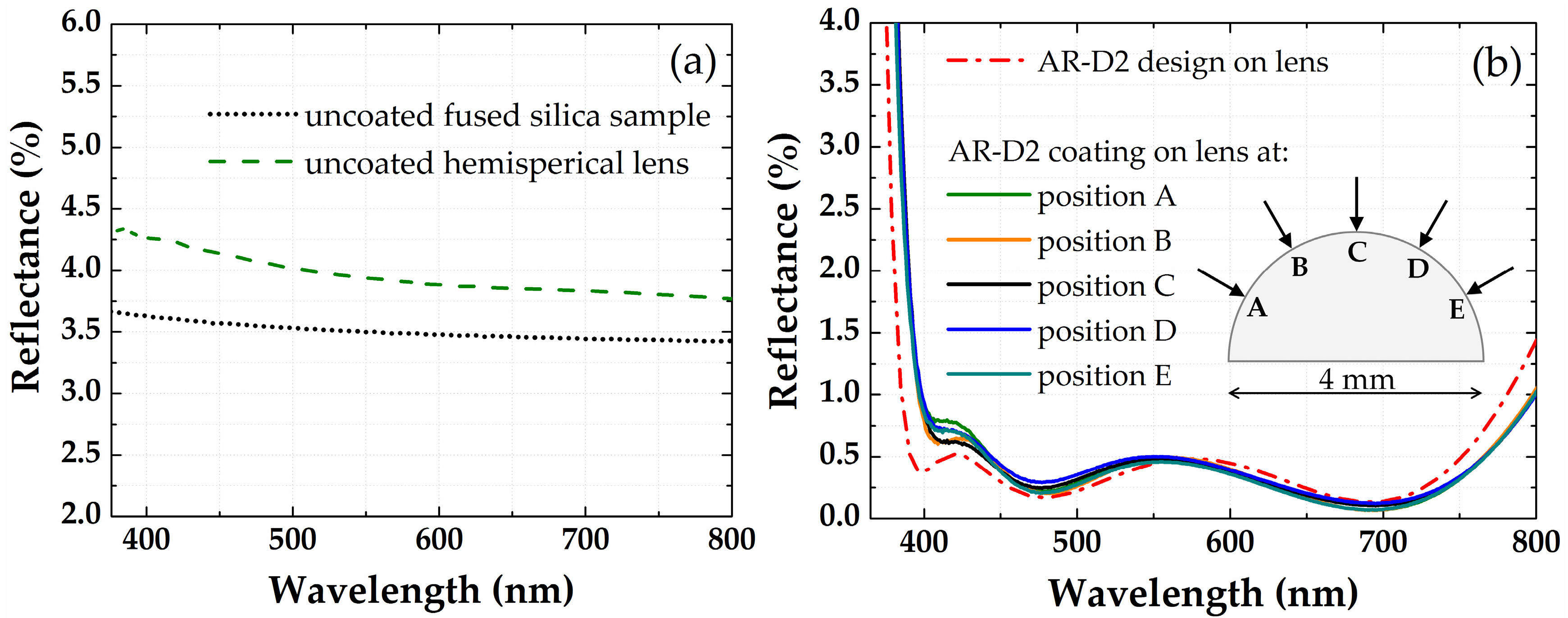
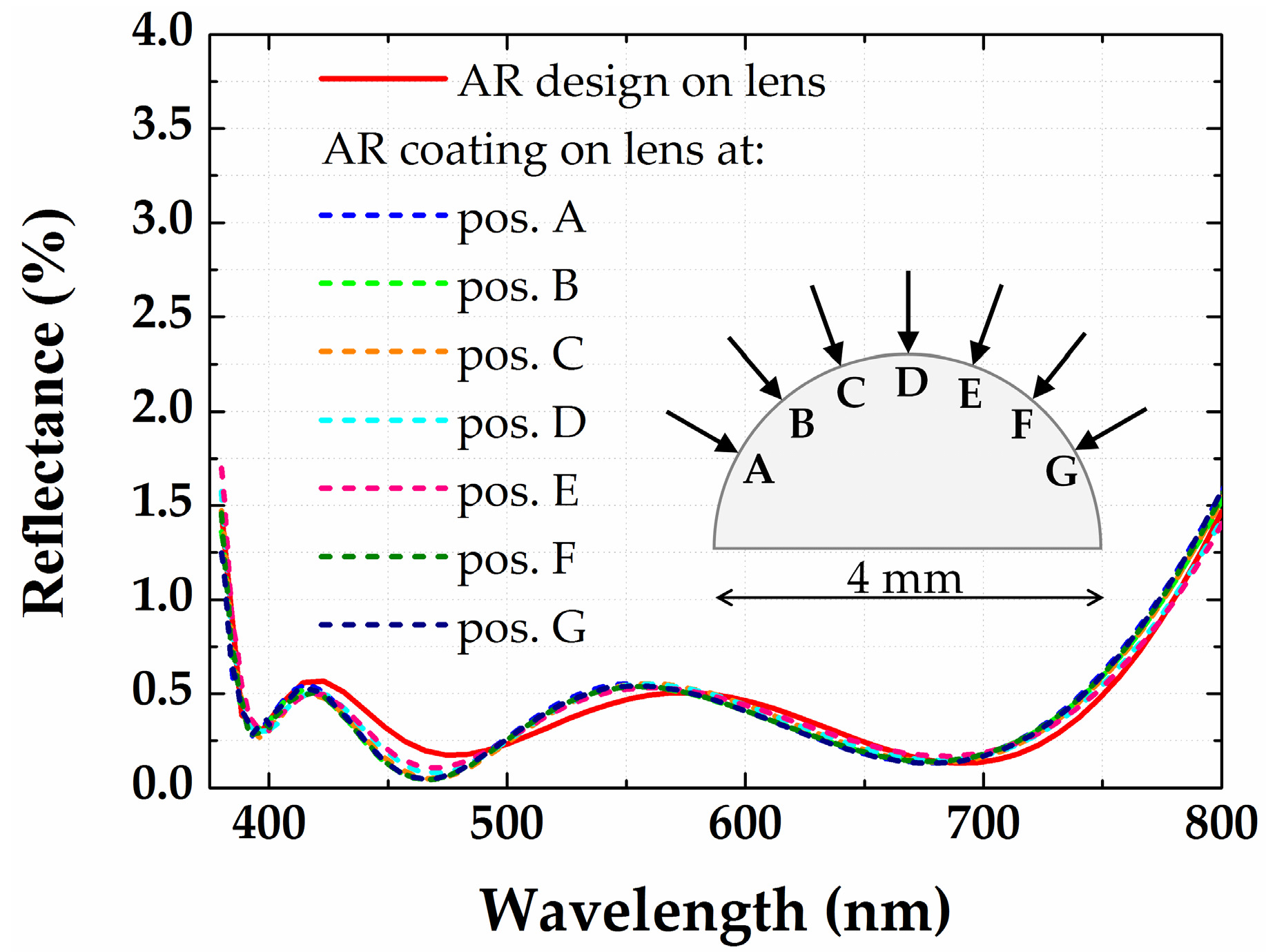
3.3. Antireflection Coatings on a Half-Ball Lens
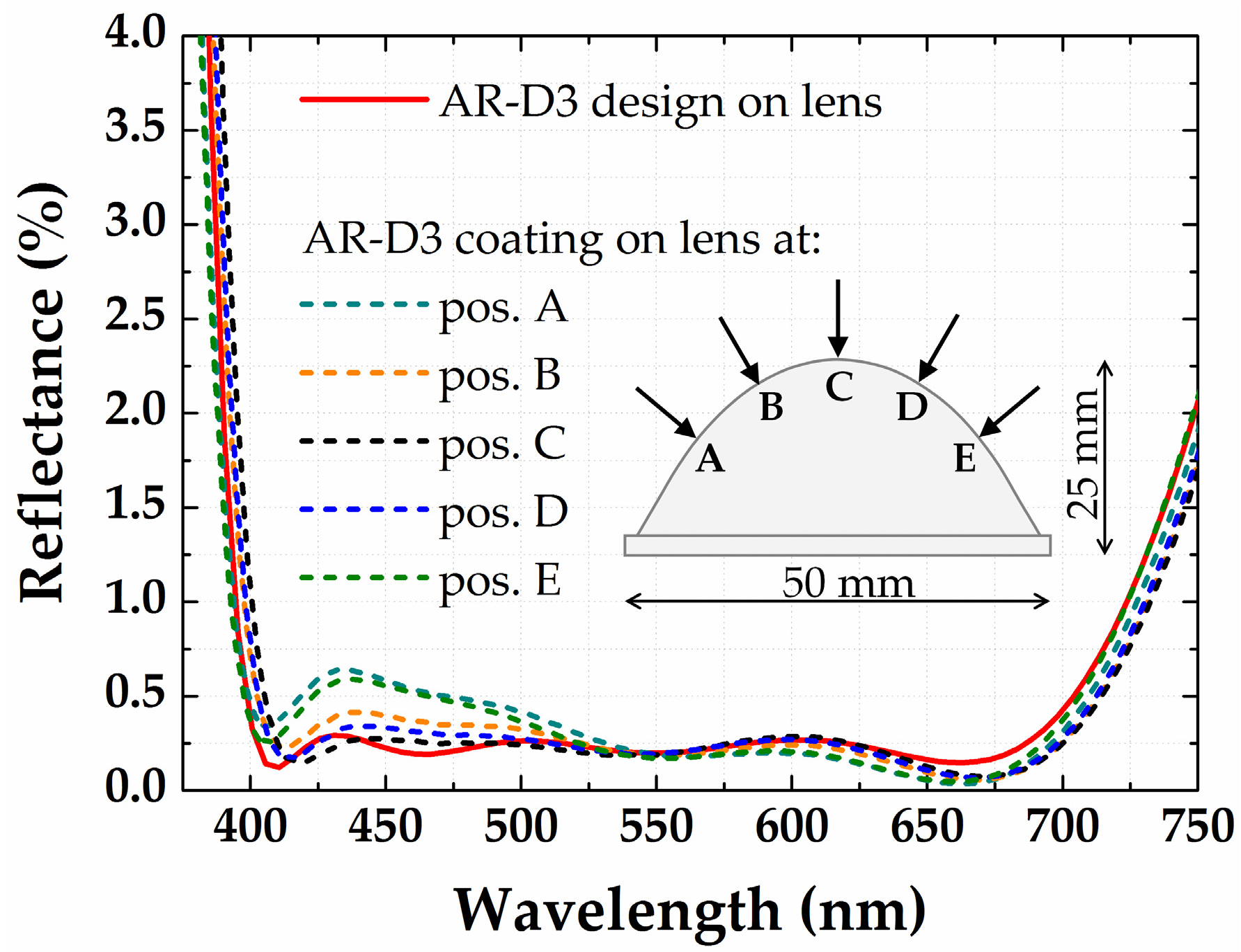
3.4. Antireflection Coating for an Aspheric Lens
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Raut, H.K.; Ganesh, V.A.; Nair, A.S.; Ramakrishna, S. Anti-reflective coatings: A critical, in-depth review. Energy Environ. Sci. 2011, 4, 3779. [Google Scholar] [CrossRef]
- Buskens, P.; Burghoorn, M.; Mourad, M.C.D.; Vroon, Z. Antireflective coatings for glass and transparent polymers. Langmuir 2016, 32, 6781–6793. [Google Scholar] [CrossRef] [PubMed]
- Hedayati, K.M.; Elbahri, M. Antireflective coatings: Conventional stacking layers and ultrathin plasmonic metasurfaces, a mini-review. Materials 2016, 9, 497. [Google Scholar] [CrossRef] [PubMed]
- Das, N.; Islam, S. Design and Analysis of nano-structured gratings for conversion efficiency improvement in GaAs solar cells. Energies 2016, 9, 690. [Google Scholar] [CrossRef]
- Pulker, H.K. Optical coatings deposited by ion and plasma pvd processes. Surf. Coat. Technol. 1999, 112, 250–256. [Google Scholar] [CrossRef]
- Thielsch, R.; Gatto, A.; Heber, J.; Kaiser, N. A comparative study of the UV optical and structural properties of SiO2, Al2O3, and HfO2 single layers deposited by reactive evaporation, ion-assisted deposition and plasma ion-assisted deposition. Thin Solid Films 2002, 410, 86–93. [Google Scholar] [CrossRef]
- Liu, C.; Kong, M.; Chun, G.; Gao, W.; Li, B. Theoretical design of shadowing masks for uniform coatings on spherical substrates in planetary rotation systems. Opt. Express 2012, 20, 23790. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; West, P.R.; Meng, X.; Kinsey, N.; Shalaev, V.M.; Boltasseva, A. Angled physical vapor deposition techniques for non-conformal thin films and three-dimensional structures. MRS Commun. 2016, 6, 17–22. [Google Scholar] [CrossRef]
- Gross, M.; Dligatch, S.; Chtanov, A.C. Optimization of coating uniformity in an ion beam sputtering system using a modified planetary rotation method. Appl. Opt. 2011, 50, C316–C320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.C.; Cai, X.K. Uniformity masks design method based on the shadow matrix for coating materials with different condensation characteristics. Sci. World J. 2013, 10, 160792. [Google Scholar] [CrossRef] [PubMed]
- Diao, Z.; Kraus, M.; Brunner, R.; Dirks, J.H.; Spatz, J.P. Nanostructured stealth surfaces for visible and near-infrared light. Nano. Lett. 2016, 16, 6610–6616. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.; Lehr, D.; Helgert, M.; Kley, E.B.; Tünnermann, A. Transmission enhanced optical lenses with self-organized antireflective subwavelength structures for the uv range. Opt. Lett. 2011, 36, 3924–3926. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.D.; Busse, L.E.; Frantz, J.; Sanghera, J.S.; Aggarwal, I.D.; Poutous, M.K. Angle-of-incidence performance of random anti-reflection structures on curved surfaces. Appl. Opt. 2016, 55, 2203–2213. [Google Scholar] [CrossRef] [PubMed]
- Riihelä, D.; Ritala, M.; Matero, R.; Leskelä, M. Introduction atomic layer epitaxy for the deposition of optical thin films. Thin Solid Films 1996, 289, 250–255. [Google Scholar] [CrossRef]
- Kumagai, H.; Toyoda, K.; Kobayashi, K.; Obara, M.; Iimura, Y. Titanium oxide aluminum oxide multilayer reflectors for ”water-window” wavelengths. Appl. Phys. Lett. 1997, 70, 2338–2340. [Google Scholar] [CrossRef]
- Maula, J. Atomic layer deposition for industrial optical coatings. Chin. Opt. Lett. 2010, 8, 53–58. [Google Scholar] [CrossRef]
- Li, Y.H.; Shen, W.D.; Zhang, Y.G.; Hao, X.; Fan, H.H.; Liu, X. Precise broad-band anti-refection coating fabricated by atomic layer deposition. Opt. Commun. 2013, 292, 31–35. [Google Scholar] [CrossRef]
- Li, Y.H.; Shen, W.D.; Hao, X.; Lang, T.T.; Jin, S.Z.; Liu, X. Rugate notch filter fabricated by atomic layer deposition. Appl. Opt. 2014, 53, A270–A275. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, K.; Shestaeva, S.; Bingel, A.; Munzert, P.; Ghazaryan, L.; van Helvoirt, C.; Kessels, W.M.M.; Sanli, U.T.; Grévent, C.; Schütz, G.; et al. Comparative study of ALD SiO2 thin films for optical applications. Opt. Mater. Express 2016, 6, 660–670. [Google Scholar] [CrossRef]
- Szeghalmi, A.; Helgert, M.; Brunner, R.; Heyroth, F.; Gosele, U.; Knez, M. Atomic layer deposition of Al2O3 and TiO2 multilayers for applications as bandpass filters and antireflection coatings. Appl. Opt. 2009, 48, 1727–1732. [Google Scholar] [CrossRef]
- Shestaeva, S.; Bingel, A.; Munzert, P.; Ghazaryan, L.; Patzig, C.; Tünnermann, A.; Szeghalmi, A. Mechanical, structural, and optical properties of PEALD metallic oxides for optical applications. Appl. Opt. 2017, 56, C47–C59. [Google Scholar] [CrossRef] [PubMed]
- Ritala, M.; Niinistö, J. Atomic layer deposition. In Chemical Vapour Deposition: Precursors, Processes and Applications; Jones, A.C., Hitchman, M.L., Eds.; Royal Society of Chemistry: Cambridge, UK, 2009; pp. 158–206. [Google Scholar]
- Kariniemi, M.; Niinisto, J.; Vehkamaki, M.; Kemell, M.; Ritala, M.; Leskela, M.; Putkonen, M. Conformality of remote plasma-enhanced atomic layer deposition processes: An experimental study. J. Vac. Sci. Technol. A 2012, 30, 01A115. [Google Scholar] [CrossRef]
- Siefke, T.; Kroker, S.; Pfeiffer, K.; Puffky, O.; Dietrich, K.; Franta, D.; Ohlidal, I.; Szeghalmi, A.; Kley, E.B.; Tünnermann, A. Materials pushing the application limits of wire grid polarizers further into the deep ultraviolet spectral range. Adv. Opt. Mater. 2016, 4, 1780–1786. [Google Scholar] [CrossRef]
- Stenzel, O.; Wilbrandt, S.; Friedrich, K.; Kaiser, N. Realistische Modellierung der NIR/VIS/UV-optischen Konstanten dünner optischer Schichten im Rahmen des Oszillatormodells. Vak. Forsch. Prax. 2009, 21, 15–23. [Google Scholar] [CrossRef]
- Groner, M.D.; Fabreguette, F.H.; Elam, J.W.; George, S.M. Low-temperature Al2O3 atomic layer deposition. Chem. Mat. 2004, 16, 639–645. [Google Scholar] [CrossRef]
- Puurunen, R.L. Surface chemistry of atomic layer deposition: A case study for the trimethylaluminum/water process. J. Appl. Phys. 2005, 97, 121301. [Google Scholar] [CrossRef]
- Pfeiffer, K.; Schulz, U.; Tünnermann, A.; Szeghalmi, A. Ta2O5/Al2O3/SiO2—Antireflective coating for non-planar optical surfaces by atomic layer deposition. Proc. SPIE 2017, 10115, 1011513. [Google Scholar]
- Ghazaryan, L.; Kley, E.B.; Tünnermann, A.; Szeghalmi, A. Nanoporous SiO2 thin films made by atomic layer deposition and atomic etching. Nanotechnology 2016, 27, 255603. [Google Scholar] [CrossRef] [PubMed]
- Dingemans, G.; van Helvoirt, C.A.A.; Pierreux, D.; Keuning, W.; Kessels, W.M.M. Plasma-assisted ALD for the conformal deposition of SiO2: Process, material and electronic properties. J. Electrochem. Soc. 2012, 159, H277–H285. [Google Scholar] [CrossRef]
- Putkonen, M.; Bosund, M.; Ylivaara, O.M.E.; Puurunen, R.L.; Kilpi, L.; Ronkainen, H.; Sintonen, S.; Ali, S.; Lipsanen, H.; Liu, X.W.; et al. Thermal and plasma enhanced atomic layer deposition of SiO2 using commercial silicon precursors. Thin Solid Films 2014, 558, 93–98. [Google Scholar] [CrossRef]
- Ylivaara, O.M.E.; Liu, X.W.; Kilpi, L.; Lyytinen, J.; Schneider, D.; Laitinen, M.; Julin, J.; Ali, S.; Sintonen, S.; Berdova, M.; et al. Aluminum oxide from trimethylaluminum and water by atomic layer deposition: The temperature dependence of residual stress, elastic modulus, hardness and adhesion. Thin Solid Films 2014, 552, 124–135. [Google Scholar] [CrossRef]
- Kukli, K.; Ritala, M.; Leskelä, M. Atomic layer epitaxy growth of tantalum oxide thin films from Ta(OC2H5)5 and H2O. J. Electrochem. Soc. 1995, 142, 1670–1675. [Google Scholar] [CrossRef]
- Blanquart, T.; Longo, V.; Niinistö, J.; Heikkilä, M.; Kukli, K.; Ritala, M.; Leskelä, M. High-performance imido–amido precursor for the atomic layer deposition of Ta2O5. Semicond. Sci. Technol. 2012, 27, 074003. [Google Scholar] [CrossRef]
- Hausmann, D.M.; de Rouffignac, P.; Smith, A.; Gordon, R.; Monsma, D. Highly conformal atomic layer deposition of tantalum oxide using alkylamide precursors. Thin Solid Films 2003, 443, 1–4. [Google Scholar] [CrossRef]
- Zhao, C.; Hedhili, M.N.; Li, J.; Wang, Q.; Yang, Y.; Chen, L.; Li, L. Growth and characterization of titanium oxide by plasma enhanced atomic layer deposition. Thin Solid Films 2013, 542, 38–44. [Google Scholar] [CrossRef]
- Potts, S.E.; Kessels, W.M.M. Energy-enhanced atomic layer deposition for more process and precursor versatility. Coord. Chem. Rev. 2013, 257, 3254–3270. [Google Scholar] [CrossRef]
- Xie, Q.; Musschoot, J.; Deduytsche, D.; Vanmeirhaeghe, R.; Detavernier, C.; Van Den Berghe, S. Growth kinetics and crystallization behavior of TiO2 films prepared by plasma enhanced atomic layer deposition. J. Electrochem. Soc. 2008, 155, H688–H692. [Google Scholar] [CrossRef]
- Elers, K.E.; Blomberg, T.; Peussa, M.; Aitchison, B.; Haukka, S.; Marcus, S. Film uniformity in atomic layer deposition. Chem. Vap. Depos. 2006, 12, 13–24. [Google Scholar] [CrossRef]
- Miikkulainen, V.; Leskela, M.; Ritala, M.; Puurunen, R.L. Crystallinity of inorganic films grown by atomic layer deposition: Overview and general trends. J. Appl. Phys. 2013, 113, 021301. [Google Scholar] [CrossRef]
- Ratzsch, S.; Kley, E.B.; Tunnermann, A.; Szeghalmi, A. Influence of the oxygen plasma parameters on the atomic layer deposition of titanium dioxide. Nanotechnology 2015, 26, 024003. [Google Scholar] [CrossRef] [PubMed]
- Ylivaara, O.M.E.; Kilpi, L.; Liu, X.W.; Sintonen, S.; Ali, S.; Laitinen, M.; Julin, J.; Haimi, E.; Sajavaara, T.; Lipsanen, H.; et al. Aluminum oxide/titanium dioxide nanolaminates grown by atomic layer deposition: Growth and mechanical properties. J. Vac. Sci. Technol. A 2017, 35, 01B105. [Google Scholar] [CrossRef]
- Hoffmann, L.; Theirich, D.; Pack, S.; Kocak, F.; Schlamm, D.; Hasselmann, T.; Fahl, H.; Raupke, A.; Gargouri, H.; Riedl, T. Gas diffusion barriers prepared by spatial atmospheric pressure plasma enhanced ALD. ACS Appl. Mater. Interfaces 2017, 9, 4171–4176. [Google Scholar] [CrossRef] [PubMed]
- Szeghalmi, A.; Senz, S.; Bretschneider, M.; Gösele, U.; Knez, M. All dielectric hard X-ray mirror by atomic layer deposition. Appl. Phys. Lett. 2009, 94, 133111. [Google Scholar] [CrossRef]
- Mayer, M.; Grévent, C.; Szeghalmi, A.; Knez, M.; Weigand, M.; Rehbein, S.; Schneider, G.; Baretzky, B.; Schütz, G. Multilayer Fresnel zone plate for soft X-ray microscopy resolves sub-39 nm structures. Ultramicrospcopy 2011, 111, 1706–17011. [Google Scholar] [CrossRef] [PubMed]
- Schulz, U.; Schallenberg, U.B.; Kaiser, N. Symmetrical periods in antireflective coatings for plastic optics. Appl. Opt. 2003, 42, 1346–1351. [Google Scholar] [CrossRef] [PubMed]
- Poodt, P.; Cameron, D.C.; Dickey, E.; George, S.M.; Kuznetsov, V.; Parsons, G.N.; Roozeboom, F.; Sundaram, G.; Vermeer, A. Spatial atomic layer deposition: A route towards further industrialization of atomic layer deposition. J. Vac. Sci. Technol. A 2012, 30, 010802. [Google Scholar] [CrossRef]
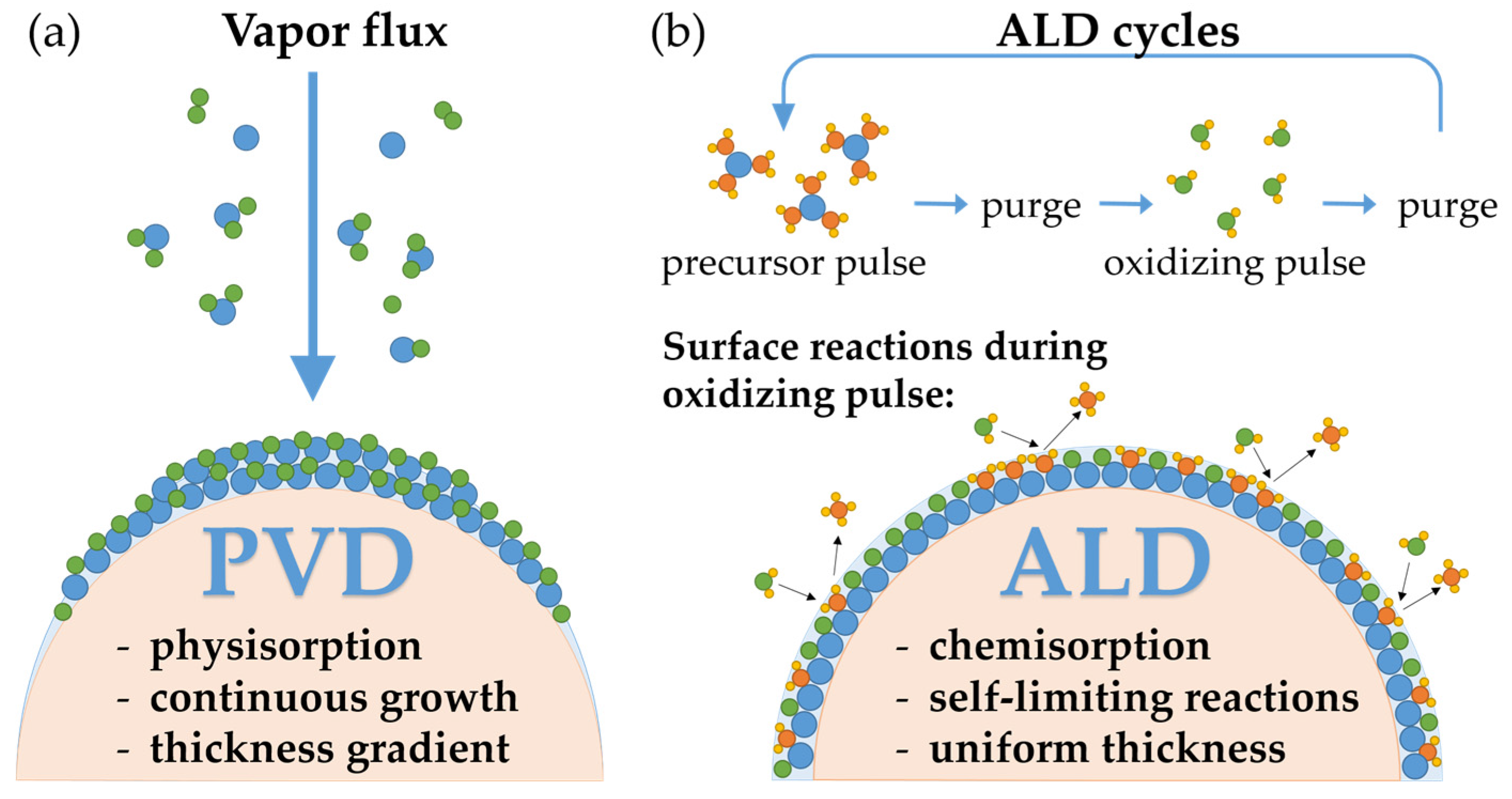
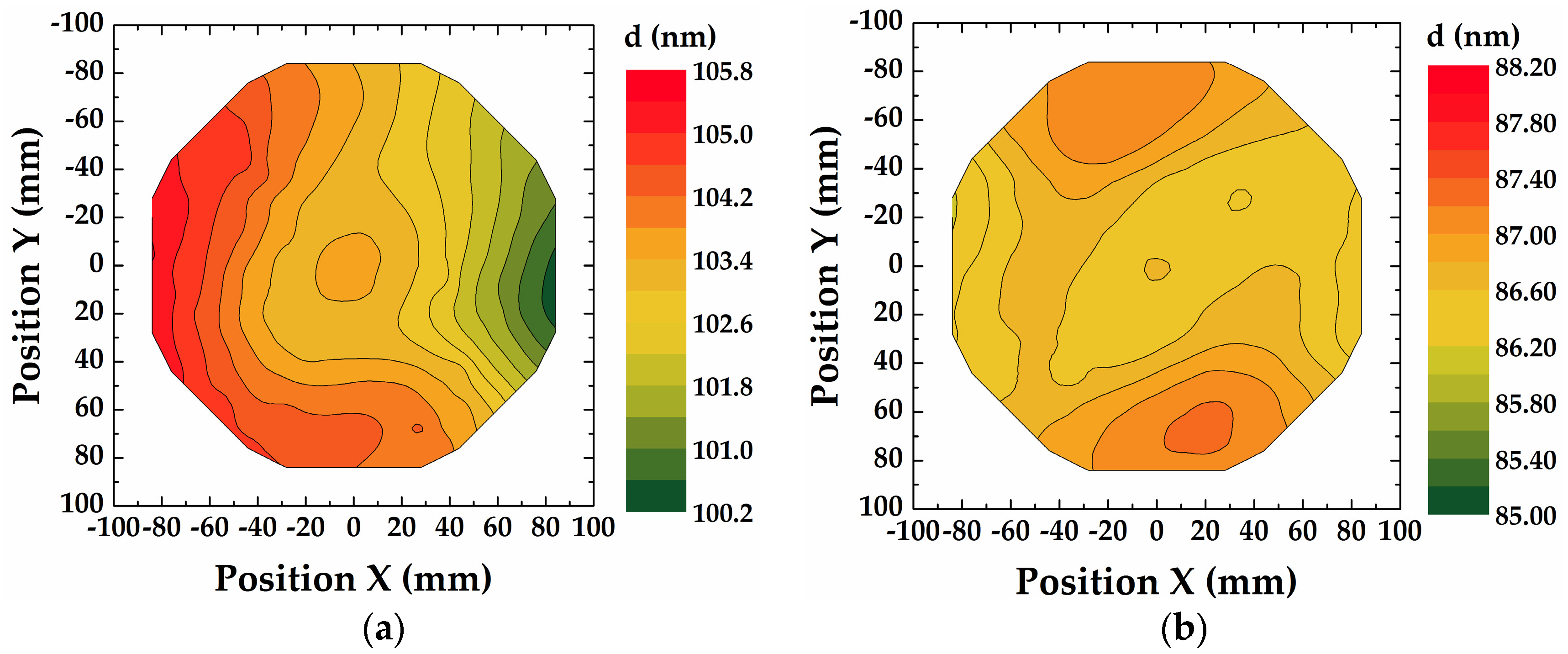
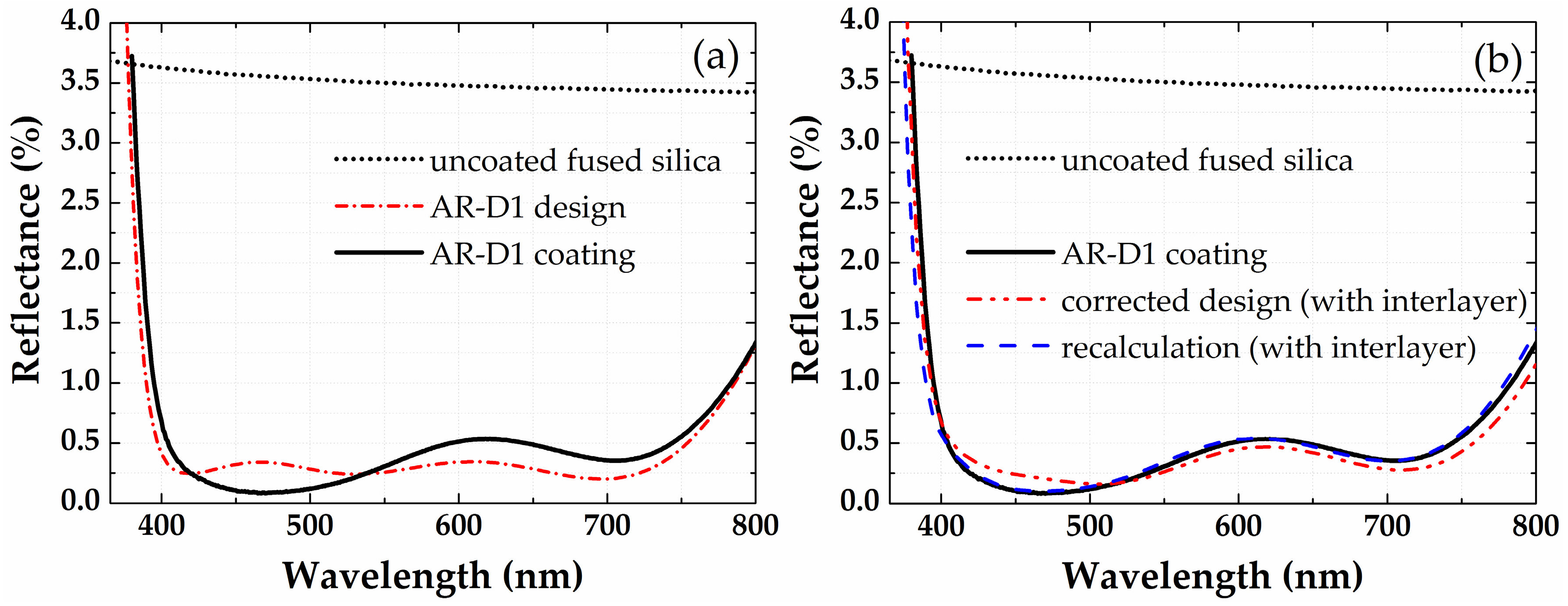
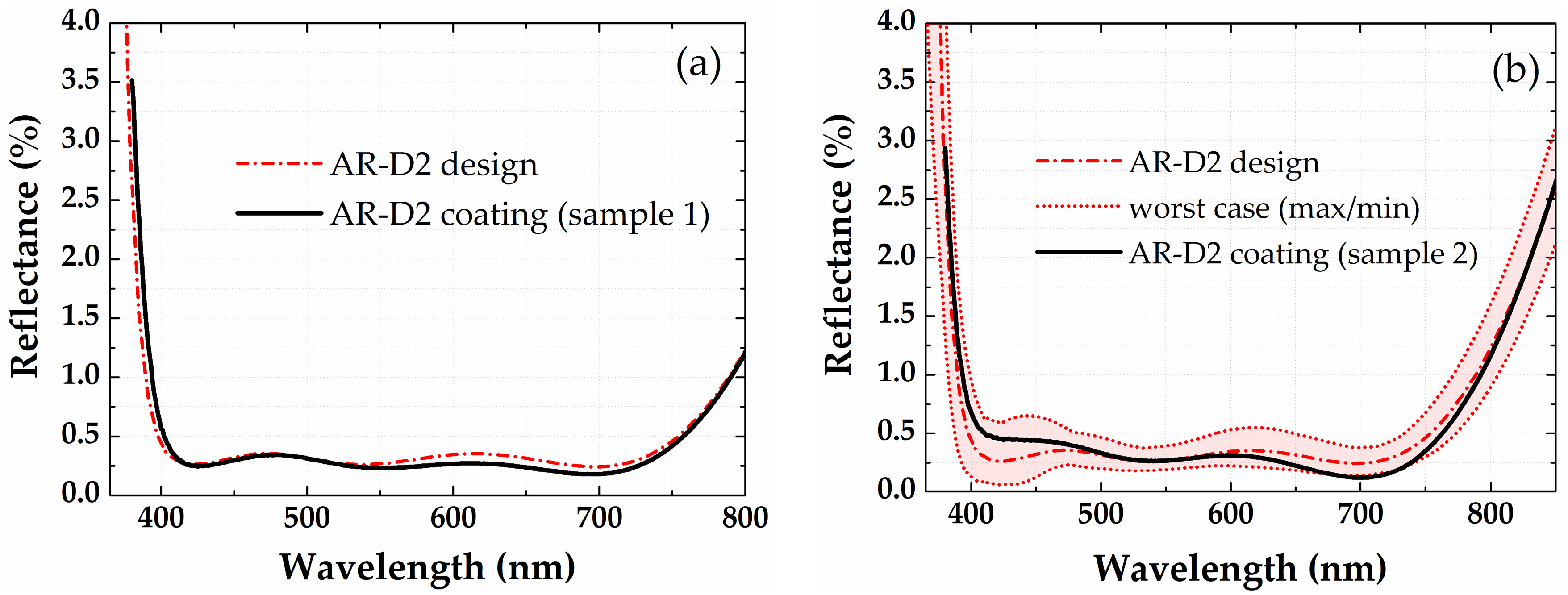
| Material | Precursor, Source Temperature, Delivery Method | Oxidizing Agent | ALD Tool | ALD Cycle [Pulse/Purge/Gas Stabilization/Oxidizing Pulse/Purge] (in s) |
|---|---|---|---|---|
| SiO2 | 3DMAS, 30 °C, vapor draw | O2-plasma | OpAL | [0.4 + 4 (hold)/–/5/3/4] |
| Al2O3 | TMA, 20 °C, vapor draw | O2-plasma | OpAL | [0.04/3.5/2.5/5/3.5] |
| Al2O3 | TMA, 20 °C, vapor draw | H2O2 | Sunale | [0.1/4.0/–/0.2/4.0] |
| TiO2 | TTIP, 50 °C, bubbling | O2-plasma | OpAL | [1.5/7.0/3.0/6.0/4.0] |
| Ta2O5 | Ta(OEt)5, 185 °C, pressure boost | H2O2 | Sunale | [1.6/6.0/–/2.0/10] |
| Material/Properties | SiO2 (100 °C) | Al2O3 (100 °C) | Al2O3 (300 °C) | Ta2O5 (300 °C) | TiO2 (100 °C) |
|---|---|---|---|---|---|
| Tool | OpAL | OpAL | Sunale | Sunale | OpAL |
| GPC on Si | 1.20 | 1.21 | 0.89 | 0.49 | 0.29 |
| n @ 550 nm | 1.46 | 1.62 | 1.66 | 2.21 | 2.44 |
| NU% 1 | ±1.5% | ±1.5% | ±2.1% | ±4.0% | ±2.0% |
| Material | AR-D1 | AR-D2 | |||||
|---|---|---|---|---|---|---|---|
| Experimental | Recalculation | Experimental | |||||
| Design (nm) | Coating (nm) | ALD Cycles | Actual Thickness (nm) | Actual GPC (Å/cycle) | Design and Coating (nm) | ALD Cycles | |
| Al2O3 | 75.1 | 75.1 | 621 | 75.4 | 1.21 | 76.8 | 635 |
| TiO2 | 16.1 | 16.1 | 556 | 16.1 | 0.29 | 16.1 | 555 |
| Al2O3 | 20.5 | 20.5 | 170 | 19.9 | 1.17 | 21.5 | 184 |
| TiO2 | 63.9 | 33.3 | 1150 | 32.5 | 0.28 | 37.5 | 1293 |
| Al2O3 | – | 1.5 | 12 | 1.4 | 1.17 | 1.5 | 13 |
| TiO2 | – | 30.6 | 1054 | 31.0 | 0.29 | 24.3 | 837 |
| Al2O3 | 13.2 | 13.2 | 109 | 12.8 | 1.17 | 14.1 | 120 |
| TiO2 | 25.00 | 25.00 | 862 | 25.3 | 0.29 | 24.2 | 834 |
| SiO2 | 92.3 | 92.3 | 769 | 90.2 | 1.17 | 92.7 | 792 |
| Material | AR-D3 | |
|---|---|---|
| Thickness (nm) | ALD Cycles | |
| Al2O3 | 101.6 | 1181 |
| Ta2O5 | 11.2 | 208 |
| Al2O3 | 186.9 | 2173 |
| Ta2O5 | 35.0 | 714 |
| Al2O3 | 21.8 | 253 |
| Ta2O5 | 43.6 | 891 |
| SiO2 | 93.7 | 787 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pfeiffer, K.; Schulz, U.; Tünnermann, A.; Szeghalmi, A. Antireflection Coatings for Strongly Curved Glass Lenses by Atomic Layer Deposition. Coatings 2017, 7, 118. https://doi.org/10.3390/coatings7080118
Pfeiffer K, Schulz U, Tünnermann A, Szeghalmi A. Antireflection Coatings for Strongly Curved Glass Lenses by Atomic Layer Deposition. Coatings. 2017; 7(8):118. https://doi.org/10.3390/coatings7080118
Chicago/Turabian StylePfeiffer, Kristin, Ulrike Schulz, Andreas Tünnermann, and Adriana Szeghalmi. 2017. "Antireflection Coatings for Strongly Curved Glass Lenses by Atomic Layer Deposition" Coatings 7, no. 8: 118. https://doi.org/10.3390/coatings7080118
APA StylePfeiffer, K., Schulz, U., Tünnermann, A., & Szeghalmi, A. (2017). Antireflection Coatings for Strongly Curved Glass Lenses by Atomic Layer Deposition. Coatings, 7(8), 118. https://doi.org/10.3390/coatings7080118






