Electrophoretic Coating of Octahedral Molybdenum Metal Clusters for UV/NIR Light Screening
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the EPD Suspension
2.2. Preparation of the Mo6 Film Incorporated with PDMS
2.2.1. Hydrophilization of the ITO Glass
2.2.2. PDMS Coating on the Mo6 Film
2.2.3. Characterization of the Mo6 Film
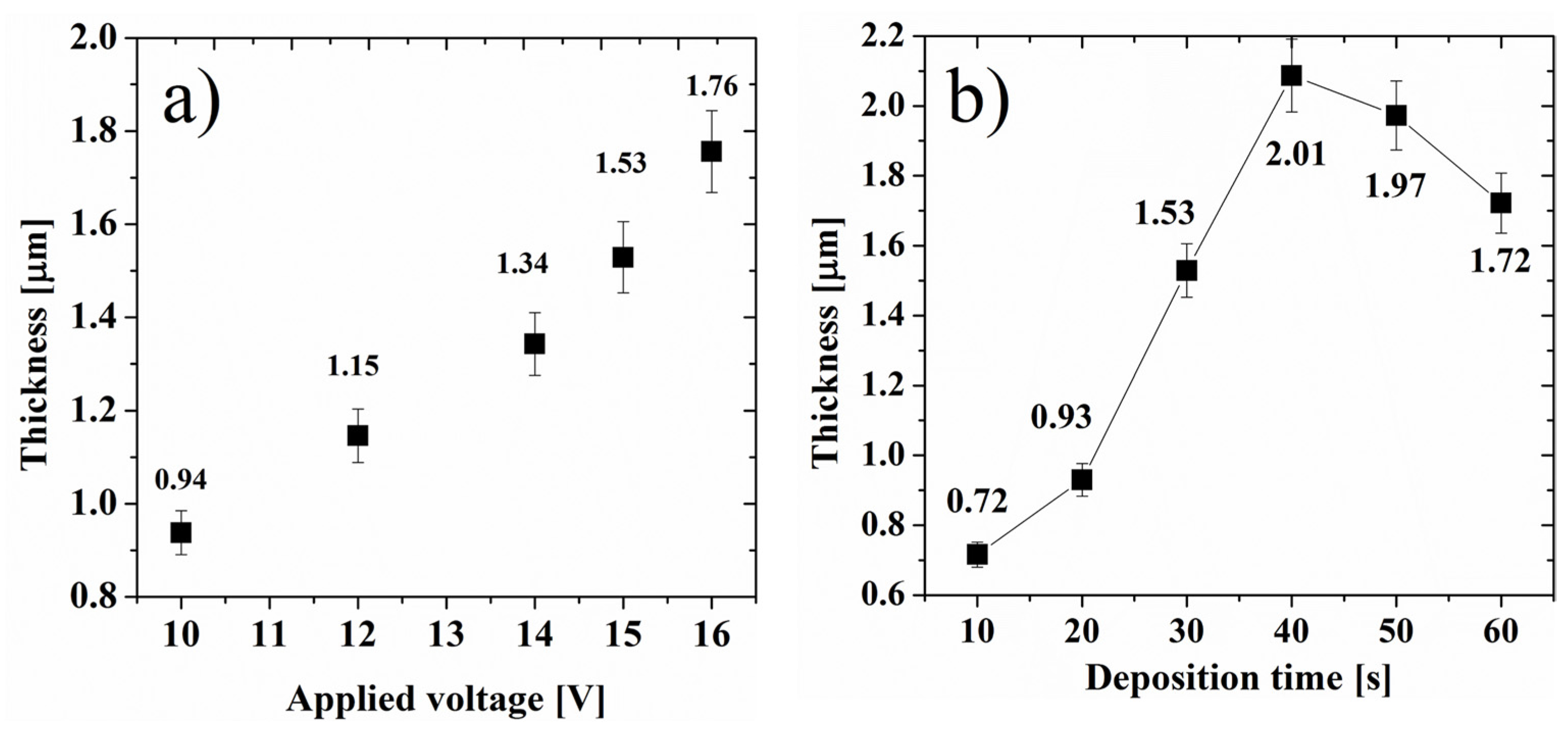
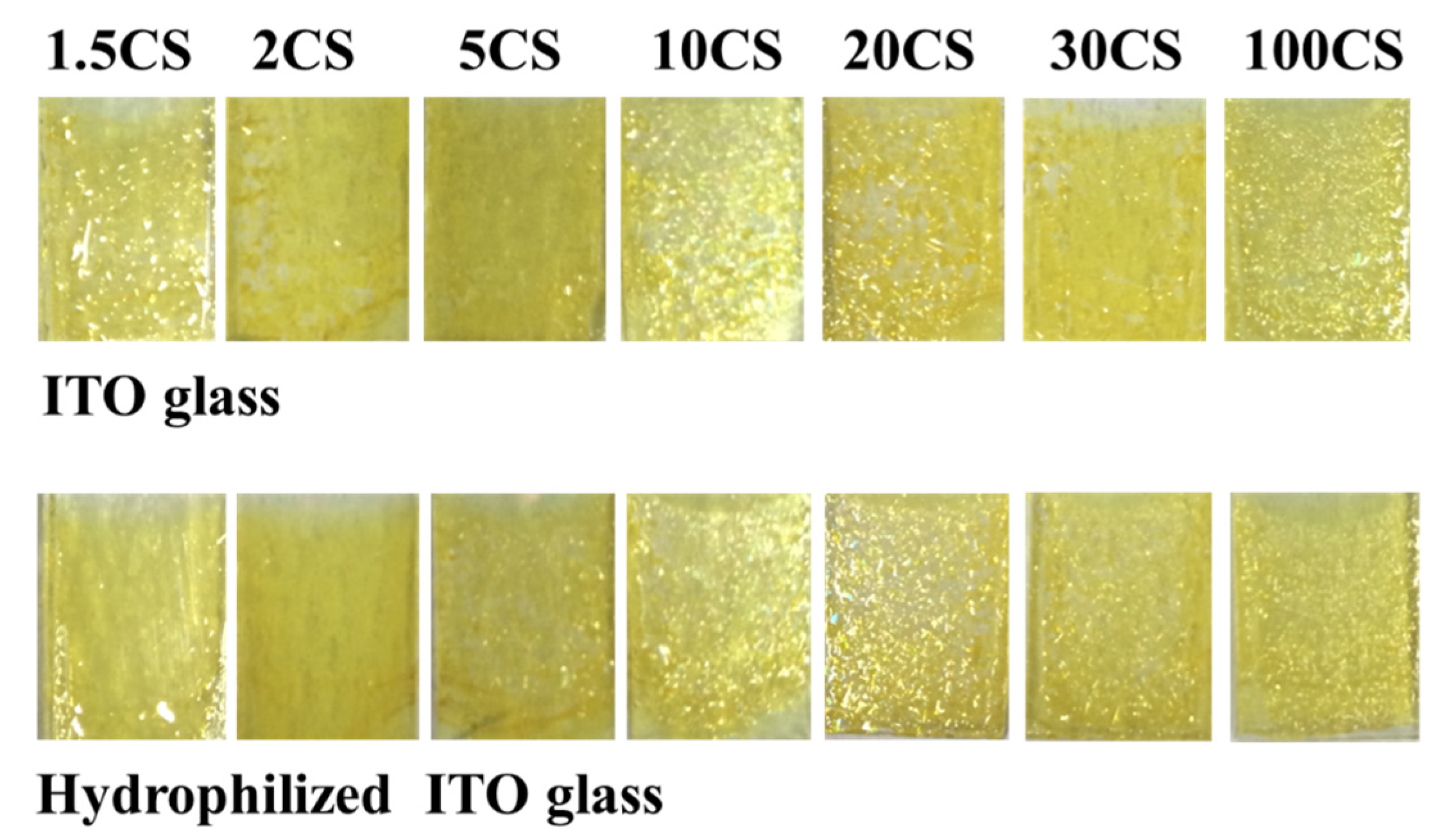
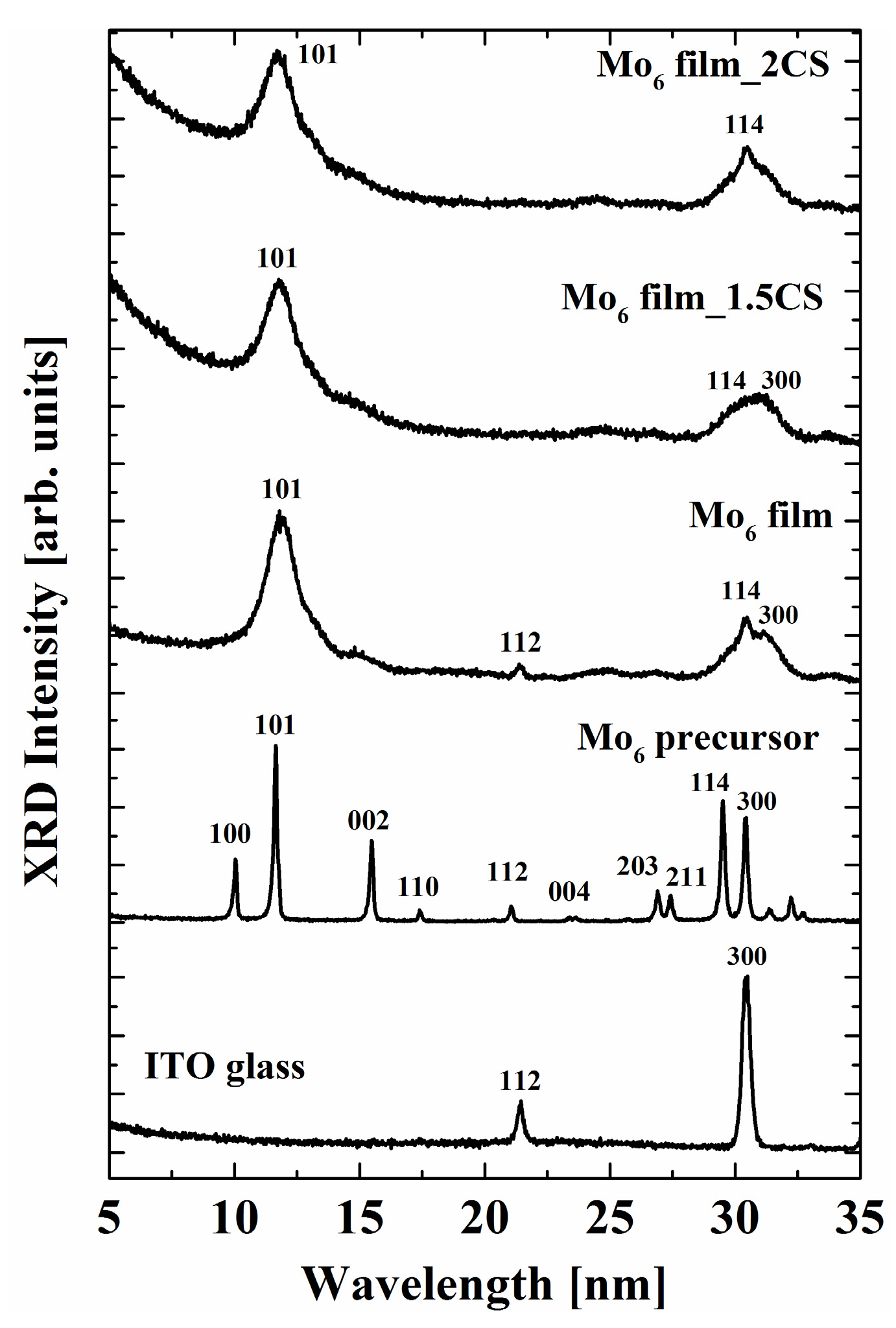
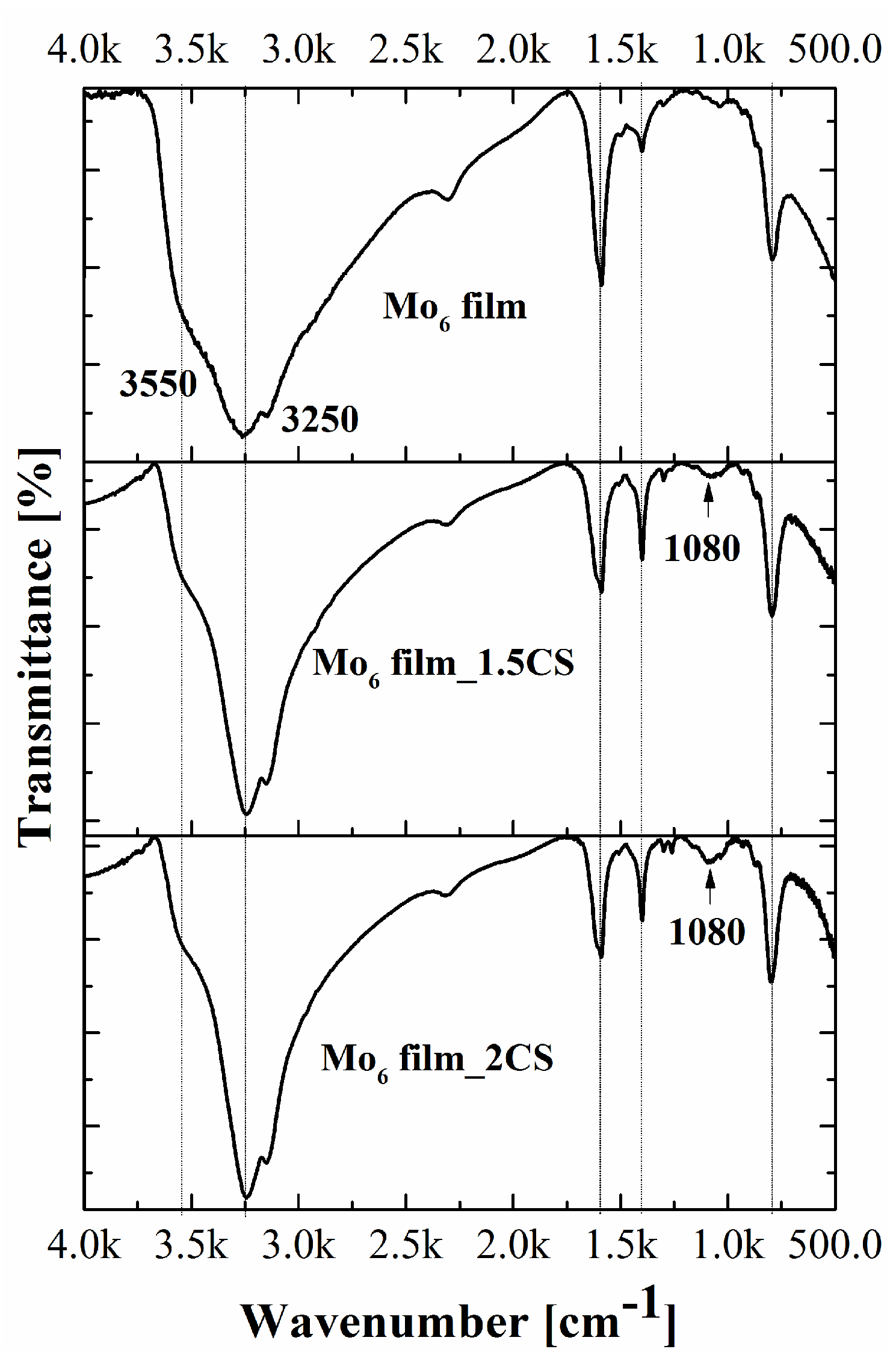
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shang, Q.; Zhou, Y. Fabrication of transparent superhydrophobic porous silica coating for self-cleaning and anti-fogging. Ceram. Int. 2016, 42, 8706–8712. [Google Scholar] [CrossRef]
- Liang, Z.; Zhao, L.; Meng, W.; Zhong, C.; Wei, S.; Dong, B.; Xu, Z.; Wan, L.; Wang, S. Tungsten-doped vanadium dioxide thin films as smart windows with self-cleaning and energy-saving functions. J. Alloy. Compd. 2017, 694, 124–131. [Google Scholar] [CrossRef]
- Granqvist, C. Electrochromics for smart windows: Oxide-based thin films and devices. Thin Solid Films 2014, 564, 1–38. [Google Scholar] [CrossRef]
- Feng, G.; Zou, L.; Gao, G.; Wua, G.; Shena, J.; Li, W. Gasochromic smart window: Optical and thermal properties, energy simulation and feasibility analysis. Sol. Energy Mater. Sol. Cells 2016, 144, 316–323. [Google Scholar] [CrossRef]
- Dai, J.; Gao, W.; Liu, B.; Cao, X.; Tao, T.; Xie, Z.; Zhao, H.; Chen, D.; Ping, H.; Zhang, R. Design and fabrication of UV band-pass filters based on SiO2/Si3N4 dielectric distributed bragg reflector. Appl. Surf. Sci. 2016, 364, 886–891. [Google Scholar] [CrossRef]
- Guo, C.; Sun, T.; Cao, F.; Liu, Q.; Ren, R. Metallic nanostructures for light trapping in energy-harvesting devices. Light Sci. Appl. 2014, 3, 161–173. [Google Scholar]
- Cordier, S.; Kirakci, K.; Méry, D.; Perrin, C.; Astruc, D. Mo6 Nanocluster Cores (X: Br, I): From inorganic solid state compounds to hybrids. Inorg. Chim. Acta 2006, 359, 1705–1709. [Google Scholar] [CrossRef]
- Cordier, S.; Grasset, F.; Molard, Y.; Amela-Cortes, M.; Boukherroub, R.; Ravaine, S.; Mortier, M.; Ohashi, N.; Saito, N.; Haneda, N. Inorganic molybdenum octahedral nanosized cluster units, versatile functional building block for nanoarchitectonics. J. Inorg. Organomet. Polym. 2015, 25, 189–204. [Google Scholar] [CrossRef]
- Renaud, A.; Grasset, F.; Dierre, B.; Uchikoshi, T.; Ohashi, N.; Takei, T.; Planchat, A.; Cario, L.; Jobic, S.; Odobel, F.; et al. Inorganic molybdenum clusters as light-harvester in all inorganic solar cells: A proof of concept. Chem. Sel. 2016, 1, 2284–2289. [Google Scholar] [CrossRef]
- Zhao, Y.; Lunt, R.R. Transparent luminescent solar concentrators for large-area solar windows enabled by massive stokes-shift nanocluster phosphors. Adv. Ener. Mater. 2013, 3, 1143–1148. [Google Scholar] [CrossRef]
- Kuttipillai, P.S.; Zhao, Y.; Traverse, C.J.; Staples, R.J.; Levine, B.G.; Lunt, R.R. Light-emitting diodes: Phosphorescent nanocluster light-emitting diodes. Adv. Mater. 2016, 28, 319. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kumar, S.; Cordier, S.; Paofai, S.; Boukherroub, R.; Jain, S. Photoreduction of CO2 to methanol with hexanuclear molybdenum [Mo6Br14]2− cluster units under visible light irradiation. RSC Adv. 2014, 4, 10420–10423. [Google Scholar] [CrossRef]
- Mitzi, D.B.; Schwartz, R.W.; Narayanan, M. Solution Processing of Inorganic Materials; Mitzi, D.B., Ed.; John Wiley & Sons, Inc.: Etobicoke, Toronto, ON, Canada, 2008; Volume 2, pp. 33–76. [Google Scholar]
- Schneller, T.; Waser, R.; Kosec, M.; Payne, D. Chemical Solution Deposition of Functional Oxide Thin Films; Springer Science & Business Media: Aachen, Germany, 2014; pp. 233–319. [Google Scholar]
- Willmott, P.R.; Huber, J.R. Pulsed laser vaporization and deposition. Rev. Mod. Phys. 2000, 72, 315–328. [Google Scholar] [CrossRef]
- Gondoni, P.; Ghidelli, M.; Fonzo, D.F.; Bassi, A.L.; Casari, C.S. Fabrication of nano-engineered transparent conducting oxides by pulsed laser deposition. J. Vis. Exp. 2013, 72, 50297. [Google Scholar] [CrossRef] [PubMed]
- Gondoni, P.; Ghidelli, M.; Fonzo, F.D.; Russo, V.; Bruno, P.; Martí-Rujas, J.; Bottani, C.E.; Bassi, A.L.; Casari, C.S. Highly performing Al: ZnO thin films grown by pulsed laser deposition at room temperature. Nanosci. Nanotechnol. Lett. 2013, 5, 484–486. [Google Scholar] [CrossRef]
- Tabellion, J.; Clasen, R. Electrophoretic deposition from aqueous suspensions for near-shape manufacturing of advanced ceramics and glasses-applications. J. Mater. Sci. 2004, 39, 803–811. [Google Scholar] [CrossRef]
- Boccaccini, A.; Peters, C.; Roether, J.; Eifler, D.; Misra, S.; Minay, S. Electrophoretic deposition of polyetheretherketone (PEEK) and PEEK/Bioglass coatings on NiTi shape memory alloy wires. J. Mater. Sci. 2006, 41, 8152–8159. [Google Scholar] [CrossRef]
- Yoshioka, T.; Chávez-Valdez, A.; Roether, J.; Schubert, D.; Boccaccini, A. AC electrophoretic deposition of organic–inorganic composite coatings. J. Colloid Interface Sci. 2013, 392, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Li, X.; Li, H.; Zhang, D.; Lai, C.; Li, W. A Comprehensive investigation on the electrophoretic deposition (EPD) of Nano-Al/Ni energetic composite coatings for the combustion application. Surf. Coat. Technol. 2015, 265, 83–91. [Google Scholar] [CrossRef]
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–61. [Google Scholar] [CrossRef]
- Corni, I.; Ryan, M.; Boccaccini, A.A. Electrophoretic deposition: From traditional ceramics to nanotechnology. J. Eur. Ceram. Soc. 2008, 28, 1353–1367. [Google Scholar] [CrossRef]
- Diba, M.; Fam, D.; Boccaccini, A.; Shaffer, M. Electrophoretic deposition of graphene-related materials: A review of the fundamentals. Prog. Mater. Sci. 2016, 82, 83–117. [Google Scholar] [CrossRef]
- Acevedo-Peña, P.; González, I. TiO2 photoanodes prepared by cathodic electrophoretic deposition in 2-propanol: Effect of the electric field and deposition time. J. Solid State Electrochem. 2013, 17, 519–526. [Google Scholar] [CrossRef]
- Cordero-Arias, L.; Cabanas-Polo, S.; Goudouri, O.; Misra, S.; Gilabert, J.; Valsami-Jones, E.; Sanchez, E.; Virtanen, S.; Boccaccini, A. Electrophoretic deposition of ZnO/alginate and ZnO-bioactive glass/alginate composite coatings for antimicrobial applications. Mater. Sci. Eng. C 2015, 55, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Fiorilli, S.; Baino, F.; Crepaldi, M.; Demarchi, D.; Cauda, V.; Vitale-Brovarone, C.; Onida, B. Electrophoretic deposition of mesoporous bioactive glass on glass–ceramic foam scaffolds for bone tissue engineering. J. Mater. Sci. Mater. Med. 2015, 26, 21. [Google Scholar] [CrossRef] [PubMed]
- Meng, D.; Francis, L.; Boccaccini, A. Using electrophoretic deposition to identify protein charge in biological medium. J. Appl. Electrochem. 2011, 41, 919–923. [Google Scholar] [CrossRef]
- Clavijo, S.; Membrives, F.; Quiroga, G.; Boccaccini, A.; Santillán, M. Electrophoretic deposition of chitosan/Bioglasss® and chitosan/Bioglasss®/TiO2 compositecoatingsforbioimplants. Ceram. Int. 2016, 42, 14206–14213. [Google Scholar] [CrossRef]
- Truong, G.; Dierre, B.; Grasset, F.; Saito, N.; Saito, N.; Nguyen, N.; Takahashi, K.; Uchikoshi, T.; Amela-Cortes, M.; Molard, Y.; et al. Visible tunable lighting system based on polymer composites embedding ZnO and metallic clusters: From colloids to thin films. Sci. Technol. Adv. Mater. 2016, 17, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Grasset, F.; Dierre, B.; Matsunaga, C.; Cordier, S.; Lemoine, P.; Ohashi, N.; Uchikoshi, T. Fabrication of transparent thin film of octahedral molybdenum metal clusters by electrophoretic deposition. ECS J. Solid State Sci. Technol. 2016, 5, 178–186. [Google Scholar] [CrossRef]
- Nguyen, T.K.N.; Dierre, B.; Grasset, F.; Renaud, A.; Cordier, S.; Lemoine, P.; Ohashi, N.; Uchikoshi, T. Formation mechanism of transparent Mo6 metal atom cluster film prepared by electrophoretic deposition. J. Electrochem. Soc. 2017, 164, 412–418. [Google Scholar] [CrossRef]
- Gray, H.B.; Maverick, A.W. Solar chemistry of metal complexes. Science 1981, 214, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Seethapathy, S.; Górecki, T. Applications of polydimethylsiloxane in analytical chemistry: A review. Anal. Chim. Acta 2012, 750, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Kirakci, K.; Cordier, S.; Perrin, C. Synthesis and characterization of Cs2Mo6X14 (X = Br or I) hexamolybdenum cluster halides: Efficient Mo6 cluster precursors for solution chemistry syntheses. Z. Anorg. Allg. Chem. 2005, 631, 411–416. [Google Scholar] [CrossRef]
- Guggenberger, L.; Sleight, A. Structural and bonding characterizations of molybdenum dibromide, Mo6Br12·2H2O. Inorg. Chem. 1969, 8, 2041–2049. [Google Scholar] [CrossRef]
- Saito, N.; Cordier, S.; Lemoine, P.; Ohsawa, T.; Wada, Y.; Grasset, F.; Cross, S.J.; Ohashi, N. Lattice and valence electronic structures of crystalline octahedral molybdenum halide clusters-based compounds, Cs2[Mo6X14] (X = Cl, Br, I), studied by density functional theory calculations. Inorg. Chem. 2017, 56, 6234–6243. [Google Scholar] [CrossRef] [PubMed]
- Engwall, A.M.; Rao, Z.; Chason, E. Origins of residual stress in thin films: Interaction between microstructure and growth kinetics. Mater. Des. 2016, 110, 616–623. [Google Scholar] [CrossRef]
- Ghidelli, M.; Sebastiani, M.; Collet, C.; Guillemet, R. Determination of the elastic moduli and residual stresses of freestanding Au-TiW bilayer thin films by nanoindentation. Mater. Des. 2016, 106, 436–445. [Google Scholar] [CrossRef]
- Cypryk, M.; Apeloig, Y. Mechanism of the Acid-Catalyzed Si–O bond cleavage in siloxanes and siloxanols: A theoretical study. Organometallics 2002, 21, 2165–2175. [Google Scholar] [CrossRef]




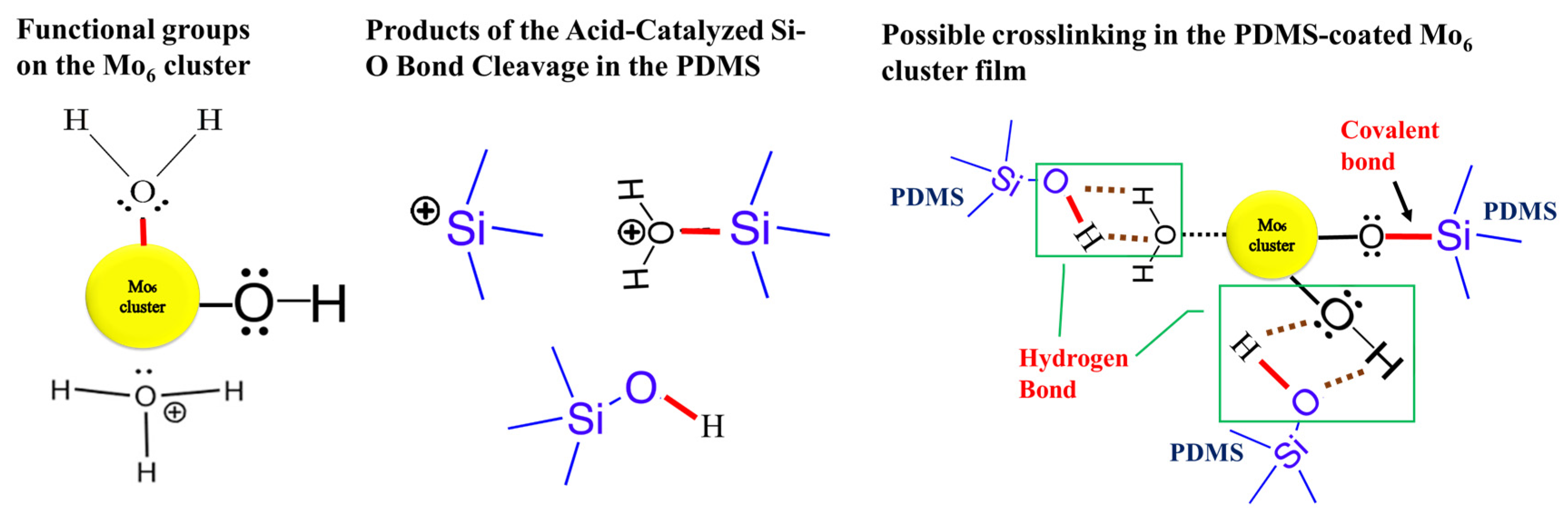
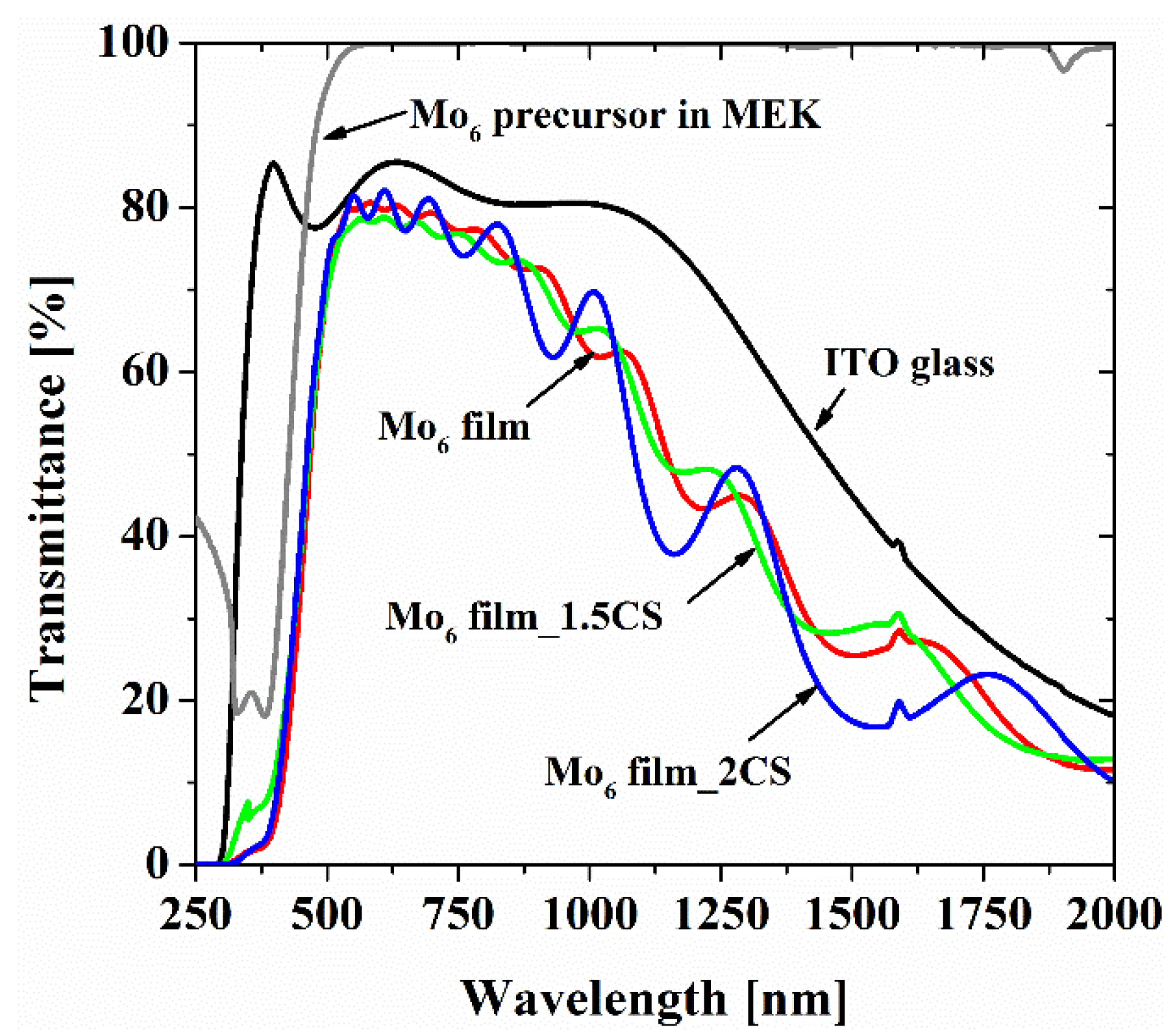
| Samples | Br/Mo Atomic Ratio (at.% Br/at.% Mo) Theoretical Index of 2.33 (14Br/6Mo) | Thickness (μm) |
|---|---|---|
| Mo6 precursor | 2.29 | – |
| Mo6 film | 2.20 | 1.53 ± 0.09 |
| Mo6 film_1.5CS | 2.21 | 1.49 ± 0.07 |
| Mo6 film_2CS | 2.17 | 1.54 ± 0.08 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.K.N.; Dierre, B.; Grasset, F.; Dumait, N.; Cordier, S.; Lemoine, P.; Renaud, A.; Fudouzi, H.; Ohashi, N.; Uchikoshi, T. Electrophoretic Coating of Octahedral Molybdenum Metal Clusters for UV/NIR Light Screening. Coatings 2017, 7, 114. https://doi.org/10.3390/coatings7080114
Nguyen TKN, Dierre B, Grasset F, Dumait N, Cordier S, Lemoine P, Renaud A, Fudouzi H, Ohashi N, Uchikoshi T. Electrophoretic Coating of Octahedral Molybdenum Metal Clusters for UV/NIR Light Screening. Coatings. 2017; 7(8):114. https://doi.org/10.3390/coatings7080114
Chicago/Turabian StyleNguyen, Thi Kim Ngan, Benjamin Dierre, Fabien Grasset, Noée Dumait, Stéphane Cordier, Pierric Lemoine, Adèle Renaud, Hiroshi Fudouzi, Naoki Ohashi, and Tetsuo Uchikoshi. 2017. "Electrophoretic Coating of Octahedral Molybdenum Metal Clusters for UV/NIR Light Screening" Coatings 7, no. 8: 114. https://doi.org/10.3390/coatings7080114
APA StyleNguyen, T. K. N., Dierre, B., Grasset, F., Dumait, N., Cordier, S., Lemoine, P., Renaud, A., Fudouzi, H., Ohashi, N., & Uchikoshi, T. (2017). Electrophoretic Coating of Octahedral Molybdenum Metal Clusters for UV/NIR Light Screening. Coatings, 7(8), 114. https://doi.org/10.3390/coatings7080114






