Preparation of Metal Coatings on Steel Balls Using Mechanical Coating Technique and Its Process Analysis
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
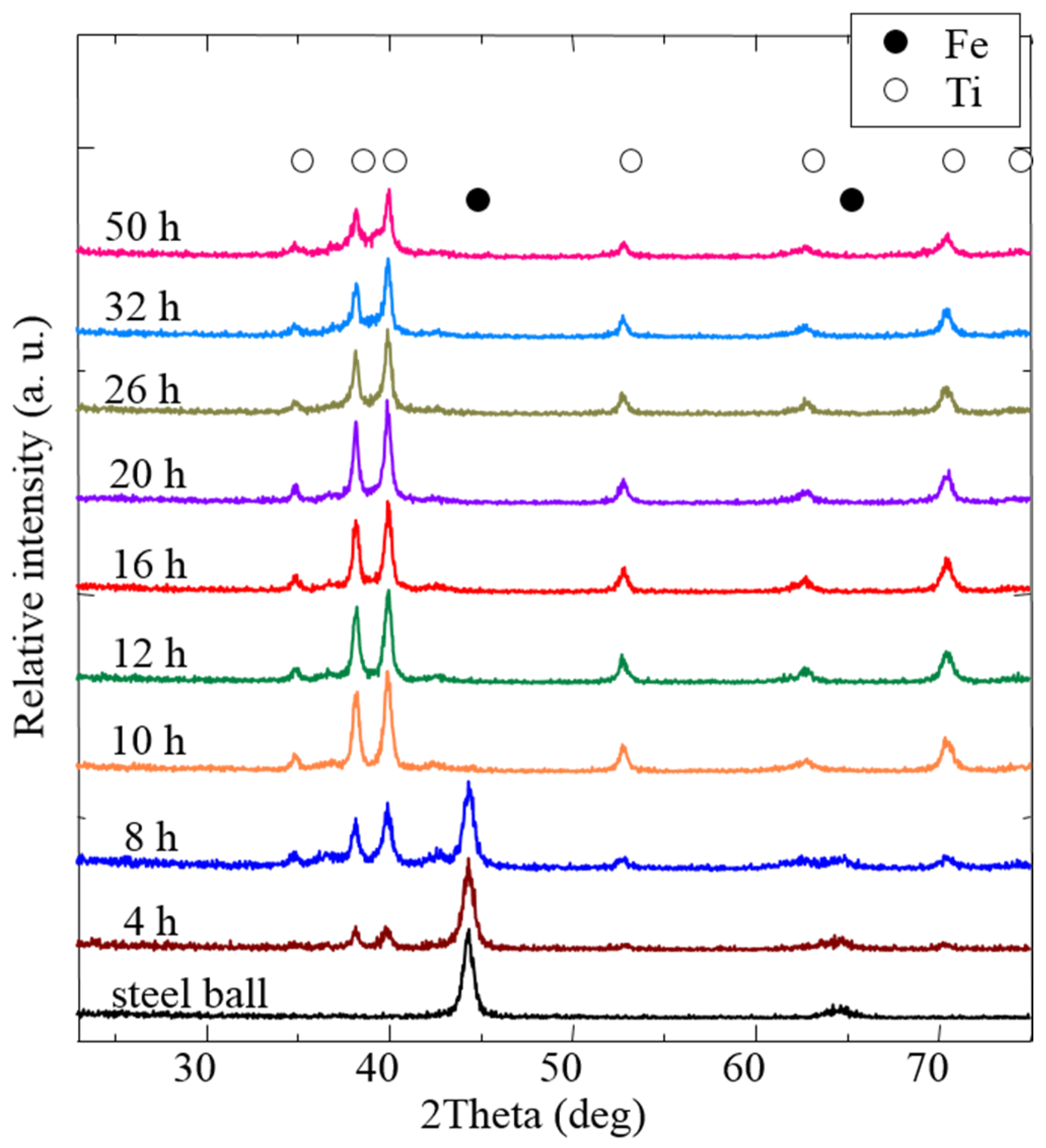
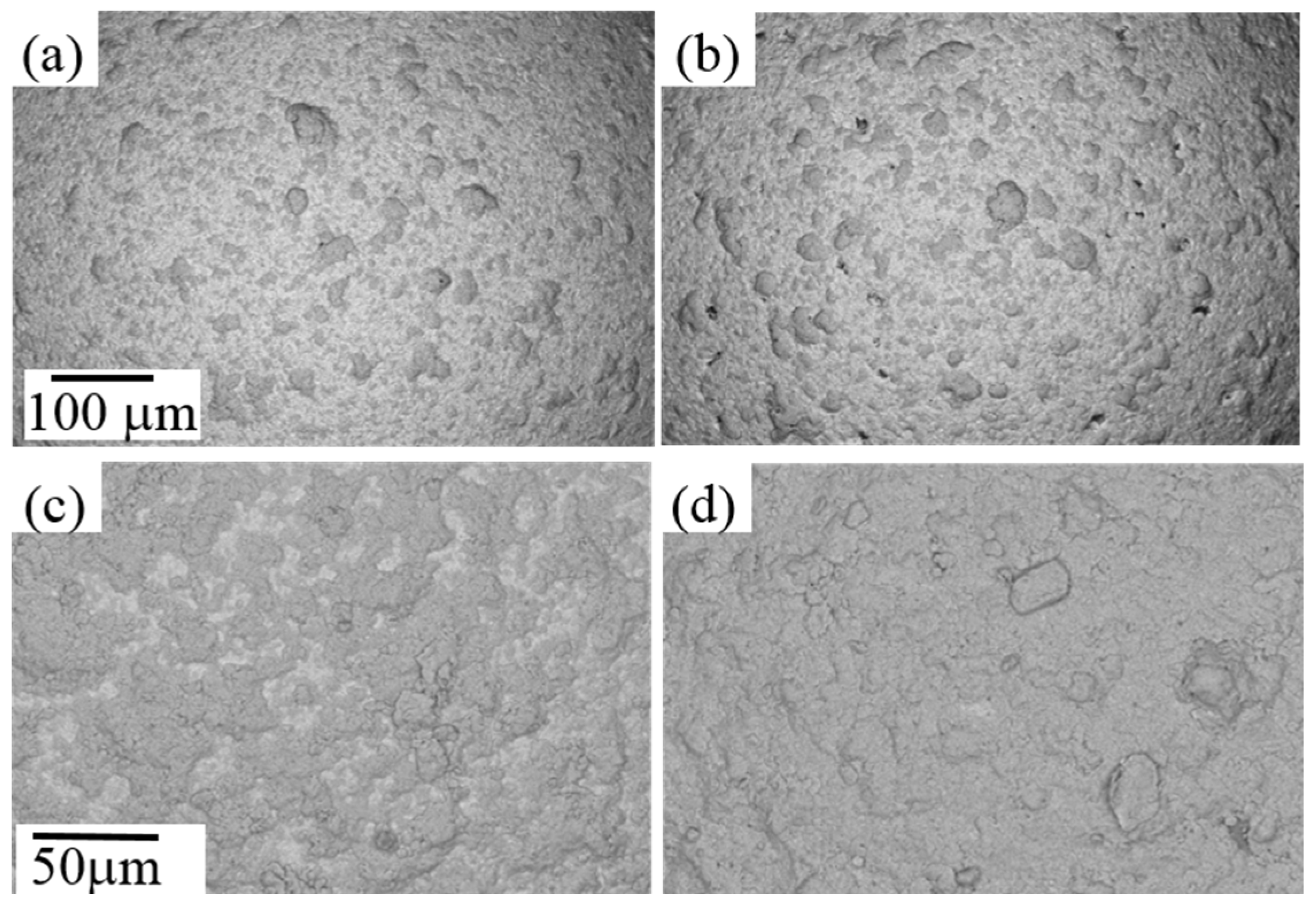
3.1. Preparation of Ti Coatings on Steel Balls
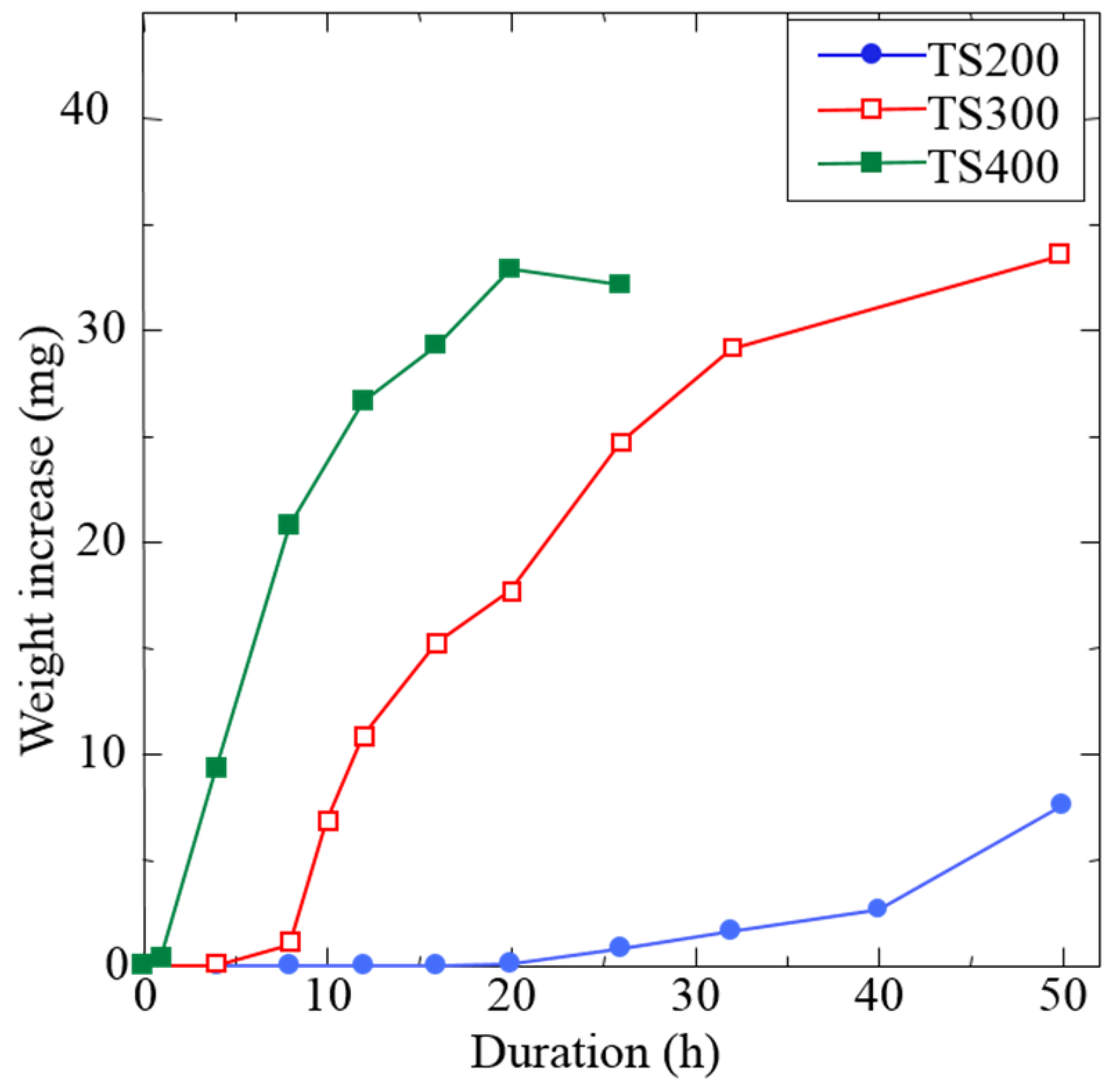
3.2. Influence of Rotational Speed
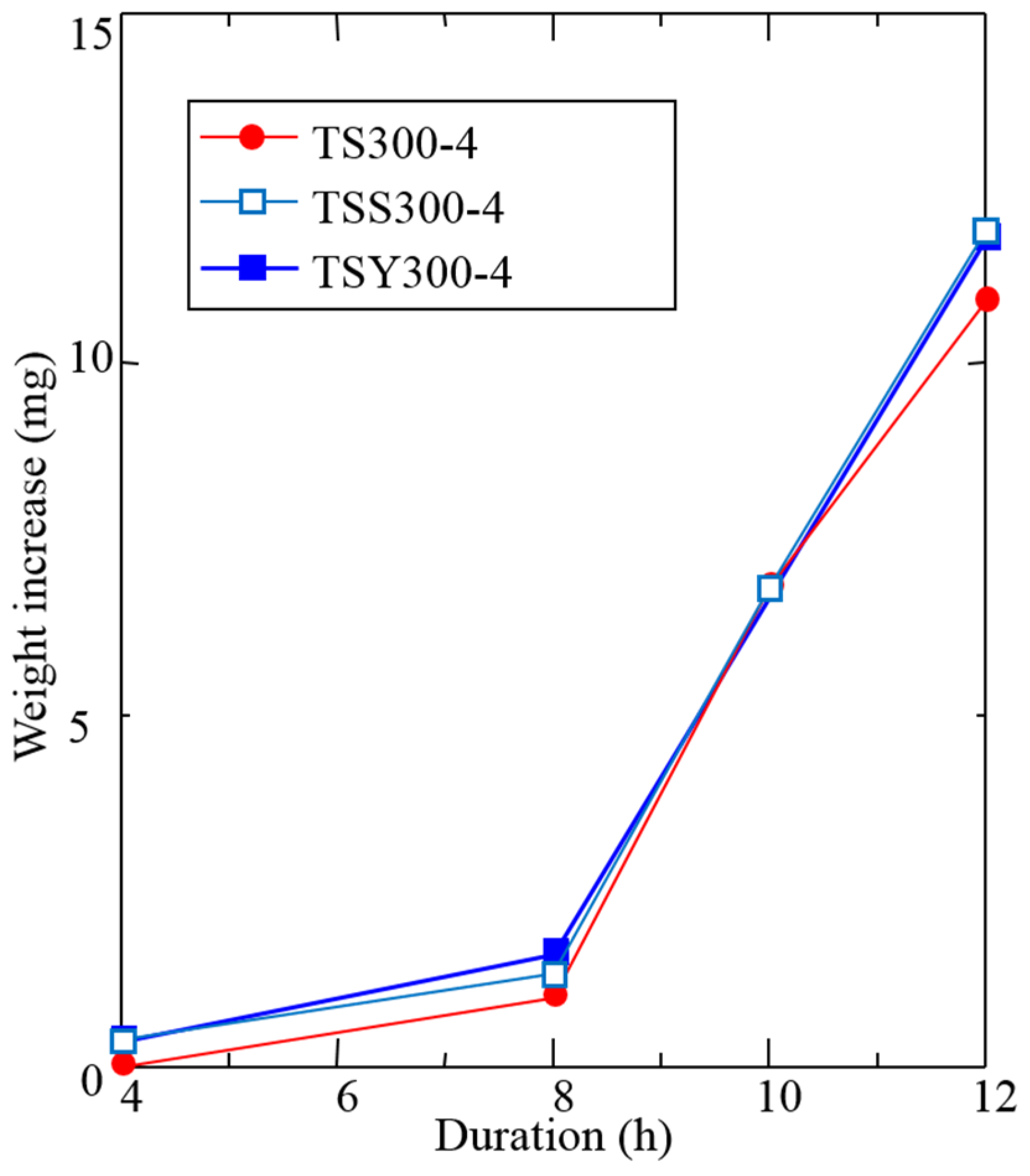
3.3. Influence of Surface Roughness and Hardness
3.4. Influence of Substrate Material
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ramanauskas, R.; Quintana, P.; Maldonado, L.; Pomés, R.; Pech-Canul, M.A. Corrosion resistance and microstructure of electrodeposited Zn and Zn alloy coatings. Surf. Coat. Technol. 1997, 92, 16–21. [Google Scholar] [CrossRef]
- Tang, J.J.; Bai, Y.; Zhang, J.C.; Liu, K.; Liu, X.Y.; Zhang, P.; Wang, Y.; Zhang, L.; Liang, G.Y.; Gao, Y.; et al. Microstructural design and oxidation resistance of CoNiCrAlY alloy coatings in thermal barrier coating system. J. Alloys Compd. 2016, 688, 729–741. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, Q.; Li, Y. Thermal shock behavior of nanostructured and microstructured thermal barrier coatings on a Fe-based alloy. Surf. Coat. Technol. 2013, 217, 70–75. [Google Scholar] [CrossRef]
- Yamamoto, K.; Ito, H.; Kujime, S. Nano-multilayered CrN/BCN coating for anti-wear and low friction applications. Surf. Coat. Technol. 2007, 201, 5244–5248. [Google Scholar] [CrossRef]
- Wei, S.; Pei, X.; Shi, B.; Shao, T.; Li, T.; Li, Y.; Xie, Y. Wear resistance and anti-friction of expansion cone with hard coating. Petrol. Explor. Dev. 2016, 43, 326–331. [Google Scholar] [CrossRef]
- Wang, K.; Wang, C.; Yin, Y.; Chen, K. Modification of Al pigment with graphene for infrared/visual stealth compatible fabric coating. J. Alloys Compd. 2017, 690, 741–748. [Google Scholar] [CrossRef]
- Yoshida, H.; Lu, Y.; Nakayama, H.; Hirohashi, M. Fabrication of TiO2 film by mechanical coating technique and its photocatalytic activity. J. Alloys Compd. 2009, 475, 383–386. [Google Scholar] [CrossRef]
- Khosravani, S.; Dehaghi, S.B.; Askari, M.B.; Khodadadi, M. The effect of various oxidation temperatures on structure of Ag-TiO2 thin film. Microelectron. Eng. 2016, 163, 67–77. [Google Scholar] [CrossRef]
- Sun, T.; Wang, M. Low-temperature biomimetic formation of apatite/TiO2 composite coatings on Ti and NiTi shape memory alloy and their characterization. Appl. Surf. Sci. 2008, 255, 396–400. [Google Scholar]
- Cotolan, N.; Rak, M.; Bele, M.; Cör, A.; Muresan, L.; Milošev, I. Sol-gel synthesis, characterization and properties of TiO2 and Ag-TiO2 coatings on titanium substrate. Surf. Coat. Technol. 2016, 307A, 790–799. [Google Scholar] [CrossRef]
- He, J.; Luo, Q.; Cai, Q.Z.; Li, X.W.; Zhang, D.Q. Microstructure and photocatalytic properties of WO3/TiO2 composite films by plasma electrolytic oxidation. Mater. Chem. Phys. 2011, 129, 242–248. [Google Scholar] [CrossRef]
- Stojadinović, S.; Radić, N.; Vasilić, R.; Petković, M.; Stefanov, P.; Zeković, L.; Grbić, B. Photocatalytic properties of TiO2/WO3 coatings formed by plasma electrolytic oxidation of titanium in 12-tungstosilicic acid. Appl. Catal B Environ. 2012, 126, 334–341. [Google Scholar] [CrossRef]
- Ghicov, A.; Macak, J.M.; Tsuchiya, H.; Kunze, J.; Haeublein, V.; Frey, L.; Schmuki, P. Ion implantation and annealing for an efficient N-doping of TiO2 nanotubes. Nano Lett. 2006, 6, 1080–1082. [Google Scholar] [CrossRef]
- Schlott, F.; Ohser-Wiedemann, R.; Jordan, T.; Kreisel, G. Effect of the electrolyte composition on the anatase fraction of photocatalytic active TiO2 coatings prepared by plasma assisted anodic oxidation. Thin Solid Films 2012, 520, 2549–2553. [Google Scholar] [CrossRef]
- Lu, Y.; Matuszaka, K.; Hao, L.; Hirakawa, Y.; Yoshida, H.; Pan, F. Photocatalytic activity of TiO2/Ti composite coatings fabricated by mechanical coating technique and subsequent heat oxidation. Mater. Sci. Semicond. Proc. 2013, 16, 1949–1956. [Google Scholar] [CrossRef]
- Guan, S.; Hao, L.; Lu, Y.; Yoshida, H.; Pan, F.; Asanuma, H. Fabrication of oxygen-deficient TiO2 coatings with nano-fiber morphology for visible-light photocatalysis. Mater. Sci. Semicond. Proc. 2016, 41, 358–363. [Google Scholar] [CrossRef]
- Lu, Y.; Hirohashi, M.; Zhang, S. Fabrication of oxide film by mechanical coating technique. In Proceedings of the International Conference on Surface, Coatings and Nanostructured Materials, Aveiro, Portugal, 7–9 September 2005. Paper No. FP117. [Google Scholar]
- Kobayashi, K. Formation of coating film on milling balls for mechanical alloying. Mater. Trans. 1995, 36, 134–137. [Google Scholar] [CrossRef]
- Romankov, S.; Sha, W.; Kaloshkin, S.D.; Kaevitser, K. Formation of Ti-Al coatings by mechanical alloying method. Surf. Coat. Technol. 2006, 201, 3235–3245. [Google Scholar] [CrossRef]
- Gupta, G.; Mondal, K.; Balasubramaniam, R. In situ nanocrystalline Fe-Si coating by mechanical alloy. J. Alloys Compd. 2009, 482, 118–122. [Google Scholar] [CrossRef]
- Farahbakhsh, I.; Zakeri, A.; Manikandan, P.; Hokamoto, K. Evaluation of nanostructured coating layers formed on Ni balls during mechanical alloying of Cu powder. Appl. Surf. Sci. 2011, 257, 2830–2837. [Google Scholar] [CrossRef]
- Hao, L.; Lu, Y.; Asanuma, H.; Guo, J. The influence of the processing parameters on the formation of iron thin films on alumina balls by mechanical coating technique. J. Mater. Process. Technol. 2012, 212, 1169–1176. [Google Scholar] [CrossRef]
- Hao, L.; Lu, Y.; Sato, H.; Asanuma, H. Fabrication of zinc coatings on alumina balls from zinc powder by mechanical coating technique and the process analysis. Powder Technol. 2012, 228, 377–384. [Google Scholar] [CrossRef]
- Lü, L.; Lai, M.; Zhang, S. Modeling of the mechanical-alloy process. J. Mater. Process. Technol. 1995, 52, 539–546. [Google Scholar] [CrossRef]
- Lu, Y.; Guan, S.; Hao, L.; Yoshida, H. Review on the photocatalyst coatings of TiO2: Fabrication by mechanical coating technique and its application. Coatings 2015, 5, 545–556. [Google Scholar] [CrossRef]
- Hao, L.; Lu, Y.; Sato, H.; Asanuma, H.; Guo, J. Influence of metal properties on the formation and evolution of metal coatings during mechanical coating. Metall. Mater. Trans. A 2013, 44, 2717–2724. [Google Scholar] [CrossRef]





| No. | C | Si | Mn | P | S | Ni | Cr | Others |
|---|---|---|---|---|---|---|---|---|
| SUJ-2 | 0.95–1.10 | 0.15–0.35 | ≤0.50 | ≤0.025 | ≤0.025 | – | 1.30–1.60 | Fe |
| SUS-304 | ≤0.08 | ≤1.00 | ≤2.00 | ≤0.045 | ≤0.030 | 8.00–10.50 | 18.00–20.00 | Fe |
| Raw Materials | Weight (g) | Average Diameter (mm) | Purity (%) | |
|---|---|---|---|---|
| Metal powder | Ti powder | 20.0 | 0.03 | 99.1 |
| Substrates | Steel balls | 58.5 | 1.0 | SUJ-2 |
| Stainless steel balls | 59.5 | 1.0 | SUS-304 | |
| Sample Symbol | Substrate | Surface Roughness | Hardness (HV) |
|---|---|---|---|
| TSx-y | SUJ-2 | original | 809 |
| TSSx-y | SUJ-2 | polished | 809 |
| TSYx-y | SUJ-2 | original | 201 |
| TBx-y | SUS-304 | original | 187 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, L.; Yoshida, H.; Itoi, T.; Lu, Y. Preparation of Metal Coatings on Steel Balls Using Mechanical Coating Technique and Its Process Analysis. Coatings 2017, 7, 53. https://doi.org/10.3390/coatings7040053
Hao L, Yoshida H, Itoi T, Lu Y. Preparation of Metal Coatings on Steel Balls Using Mechanical Coating Technique and Its Process Analysis. Coatings. 2017; 7(4):53. https://doi.org/10.3390/coatings7040053
Chicago/Turabian StyleHao, Liang, Hiroyuki Yoshida, Takaomi Itoi, and Yun Lu. 2017. "Preparation of Metal Coatings on Steel Balls Using Mechanical Coating Technique and Its Process Analysis" Coatings 7, no. 4: 53. https://doi.org/10.3390/coatings7040053
APA StyleHao, L., Yoshida, H., Itoi, T., & Lu, Y. (2017). Preparation of Metal Coatings on Steel Balls Using Mechanical Coating Technique and Its Process Analysis. Coatings, 7(4), 53. https://doi.org/10.3390/coatings7040053






