Abstract
Automotive coatings and the processes used to coat automobile surfaces exemplify the avant-garde of technologies that are capable of producing durable surfaces, exceeding customers’ expectations of appearance, maximizing efficiency, and meeting environmental regulations. These accomplishments are rooted in 100 years of experience, trial-and-error approaches, technique and technology advancements, and theoretical assessments. Because of advancements directed at understanding the how, why, when, and where of automobile coatings, the progress in controlling droplets and their deposition attributes, and the development of new technologies and paint chemistries, a comprehensive and up-to-date review of automobile coatings and coating technologies was considered to be of value to industrial practitioners and researchers. Overall, the critical performance factors driving the development and use of advanced automotive coatings and coating technologies are (a) aesthetic characteristics; (b) corrosion protection; (c) mass production; (d) cost and environmental requirements; and (e) appearance and durability. Although the relative importance of each of these factors is debatable, the perfection of any one at the expense of another would be unacceptable. Hence, new developments in automotive coatings are described and discussed in the following review, and then related to improvements in production technologies and paints. Modern automotive coating procedures are also discussed in detail. Finally, an extrapolation into the future of automotive coating is offered with a view of the developments and technologies needed for an increasingly efficient and more sustainable coatings industry.
1. Introduction
At the beginning of the automotive industry about 100 years ago, cars were painted with a varnish-like product that was brushed onto the vehicle surfaces; this coating was sanded and smoothed, and then the varnish was reapplied and refinished to establish several layers of the coating. After multiple layers of varnish were applied, vehicles were polished to produce shiny surfaces. Some manufacturers, including Ford in the Model T line, employed a combination of brushing, dipping, and even pouring to fully cover and protect various parts of a car [1]. All of these coating steps were implemented manually, and it was not uncommon for the period between the beginning of the coating process to the end, when the coating was dry and the car was ready to sell, to take up to 40 days. Between the 1920s and 1940s, automotive coating technologies transitioned to using spray equipment and “stoving enamels” that were based on alkyd resins; these two advancements decreased application and drying times to a week or less. Because of the newly developed spray coating technologies, the surface finishes were more even and less sanding was needed.
In 1923, E.I. DuPont De Nemours developed nitrocellulose lacquer systems that included many choices of color and offered improved applicability for the use of spray paint guns [2]. These lacquer systems required the application of 3–4 coats to achieve the desired surface properties and, because of their chemical makeup, had relatively poor resistance to chemical solvents like hydrochloric acid. This disadvantage hindered the ability of coatings to endure acidic environments with various chemicals.
Another significant enhancement in paint technology was the development of “alkyd” enamel paints that were introduced on some car models in the early 1930s [3]. These enamels formed a very durable film because of molecular bonding reactions that occurred after the paint was sprayed onto the vehicles and then cured or baked in an oven. Each of the cured paint films was about 0.002 inches (51 µm) thick, and had high resistance to chemicals and solvents; another advantage was that enamel paints had shorter application times that included 2–3 steps instead of the 3–4 steps for lacquers. Furthermore, organic pigments were available in many different colors, the selection of which pleased customers. However, alkyd enamel paints were degraded by oxidization in sunlight, which caused colors to fade slowly or dull.
The durability of enamel finishes was improved considerably by the 1960s with the introduction of acrylic stoving enamels [4]. They were applied using a paint spray gun and then oven-baked, resulting in a resilient, glossy finish. However, the paint spray was applied manually, which could lead to uneven coating thicknesses during application of the multiple coats that were required. Nevertheless, distinct layers were applied for different reasons by this time, including corrosion protection associated with the primers, smoothness and chip resistance associated with the primer surfacers that were often applied at the front ends and exposed areas of the automobiles, color, and weather resistance associated with the final topcoat layer.
In the 1950s, application of the dip coating process was plagued with safety, environmental, and processing issues. The use of solvent-borne or water-borne dip tanks was beset by explosions and fire hazards [5]. These issues drove the introduction of anodic electrodeposition paints, which were introduced by the mid-1960s and were mostly based on maleinized natural oils. However, by the mid-1970s, cathodic deposition coatings replaced anodic electrodeposition because better corrosion protection was offered by the modified epoxy resin backbones and reactive polyurethane-based crosslinkers of these resins; this process also provided an increased throwing power, resulting in higher coating deposition at lower currents, and higher process reliability [6].
To further improve coatings’ appearance and durability, a new type of wet-on-wet finish was developed and introduced in the late 1970s, consisting of a thin basecoat and a thicker clearcoat [7]. The topcoat painting process was split into a pigmented enamel basecoat, followed by a clear enamel finish. A key to the success of this new technology was the development of a clearcoat material with superior durability in all climates. Even though the cost of the basecoat/clearcoat paint process was prohibitive for the less expensive automobile lines, it was used on more expensive, high-end automobiles. Then, refinements in the material and processing technologies reduced costs, and by the late 1980s the use of basecoat/clearcoat processing had become widespread [2], such that only a small fraction of cars manufactured today do not use this painting process. Additionally, the first water-based basecoats were introduced at Opel in Germany in the 1980s, followed by water-based primer surfacers in the 1990s [8]. Hence, in a relatively short period, automobile coating processes had evolved to be compatible with the high throughput needs of the industry with the ability to apply even coatings with thicknesses near 100–140 μm; this thickness implies an average of 9–16 kg of paint used per automobile; paints had also evolved to be very effective and durable. Importantly, it is now estimated that the corrosion protection and durability of color and gloss are about double what was typical 25 years ago [9]. Today, most clear coats in Europe are based on a two-component (2K) formulation. This formulation, incorporates an acrylic resin with OH-functionalities and a reactive polyurethane crosslinker. The rest of the world mostly uses a one-component formulation based on acrylic resins and melamine crosslinkers [10].
Novel developments in paint pigments have been accomplished simultaneously with improved processing and paint chemistries. For example, flake-based pigments based on aluminum and interference pigments that change color depending on the angle at which they are viewed (otherwise known as the “flip flopping” effect) have enhanced the brilliance, color, appearance, and customer satisfaction of automotive coatings [11]. At first, these new pigments were challenging to use with spray gun technology; however, new spray guns and spray gun configurations have been developed to meet these challenges.
Manual spray painting required significant craftsmanship because of the need to apply enough of a coating with an even thickness independent of whether the surfaces were relatively flat or highly curved. Now, with computer-controlled spray guns, the need for spray painting craftsmanship has been dramatically reduced. Furthermore, these automated processes have undergone improvements that ensure worker safety and increase the ratio of deposited paint-to-paint sprayed [12]. Nevertheless, automobile paint shops are still a major energy-consuming area and the most expensive operational aspect of an automobile assembly plant, consuming 30%–50% of the total costs of the manufacturing of automobiles [13]. These costs are wrapped into the energy used for air handling and conditioning (HVAC), as well as for paint drying and treatment of emissions generated by paint droplets that are not deposited on automobile surfaces; the painting booths must be purged to remove evaporated solvent, overspray paint particles, and regulated pollutants (like VOCs). Hence, the energy associated with only booth ventilation is significant [14]. In general, up to 70% of the total energy costs in assembly plants is within the painting operations [15]. Although the energy used to dry a 200-µm film on an automobile surface could be calculated to be (and is) not significant, it must be realized that paint drying includes heating of the paint and underlying automobile body, and the dollies and the carriers on which automobiles are moved through a painting process.
Today, automobile painting processes are more standardized than they have ever been because of the benefits of inorganic pretreatments, cathodic electrodeposition, liquid or powder primer surfacers, liquid base coats, and one or two component solvent-borne clear coats. For example, the development of new and highly reliable powder coatings has attained a point where many car manufacturers have decided to use them; as an added advantage, powder coatings introduce the capacity to aggressively meet environmental regulations [16]. Powder coatings are now used throughout primer surfacer operations in North America at Chrysler in all actual running plants, at GM for their truck plants, and in all new paint shops. In Europe, in some plants at BMW, powder coatings are also used for the clearcoat process [17]. This expansion of powder coating applications has coincided with a dramatic shift in the type of materials used in automobile body construction. Formerly made mostly of steel, today’s automobile bodies consist typically of up to 30% of aluminum and high-strength steel. Other lightweight materials are also finding application, including magnesium and polymer composites made of glass and carbon-fiber-reinforced thermosets and thermoplastics [18].
Automotive coatings continue to evolve as they either satisfy or are anticipated to meet customer expectations and environmental regulations while also lowering manufacturing and ownership costs. One of these evolutions is in the use of smart coatings because they offer the potential to significantly improve surface durability while adding additional functionalities or properties like self-healing, super-hydrophobicity, self-stratifying, self-sensing, sound proofing, and vibration damping. For example, a smart coating could respond to its environment to enhance the coating life; a smart coating with self-healing properties would be useful in response to an abrasive, mechanical trigger or to a corrosive event in which the coating is self-healing as a result of UV, heat, or mechanical activation [19]. Self-healing can also be achieved by employing shape memory polymers that are triggered with temperature and humidity manipulations, or with UV radiation; self-healing associated with the swelling of special clays such as montmorillonite is also possible [20]. Other smart coatings include those with internal sensing capabilities that entail the passive or active triggering of fluorescent molecules or quantum dots [21]. In the former, the sensing system signals and activates changes in or repair of the coating by sending data to an external detector; in the latter, the sensing system itself would be responsible for outputting the response signal.
Another development is the eventual introduction of self-stratifying coatings that are formulated with a compatible combination of liquid and powder coatings and based on partially compatible polymer blends that produce micro-heterogeneous structures [22]. These self-stratifying coatings would enable the basecoat and clearcoat to be applied in a single layer and could streamline the now rather complex automobile painting processes in which one layer at a time is coated and then cured. Of course, it is necessary that these novel coating compositions preserve current automobile coating attributes and characteristics such as appearance and high gloss, and durability to corrosion, abrasion, and UV light exposure [22]. They could also offer other properties like high reflectivity of infrared radiation that helps to keep the car interior as cool as possible. Also, personal identity inscriptions in the form of holograms and color change features could be incorporated that are not possible now [7].
The previous discussion gave a brief history of automotive body coatings and coating processes. It alluded to recent trends in automotive coating processes, and gave a snapshot of potential future developments. In the following, a comprehensive overview of modern automotive coating methods is described, coating assessment tools are discussed, and then alternative and possibly attractive coating processes are presented.
2. Modern Automotive Coating Processes
Modern automotive coating methods consist of five main steps. They include the following:
- Pretreatment: removes and cleans excess metal and forms an appropriate surface structure enabling bonding of a corrosion protection layer.
- The next step is electrodeposition (ED) of the anti-corrosion or rust prevention layer.
- A sealer like Poly Vinyl Chloride (PVC) is applied for anti-corrosion, elimination of water leaks, and minimization of chipping and vibrational noise.
- A primer is then applied to promote adhesion between the surface and the basecoat; it also imparts a smoother surface for subsequent layers and has anti-chipping properties.
- Finally, the topcoats that include a basecoat and clearcoat are applied; they provide surface properties that are sought after, including color, appearance, gloss, smoothness, and weather resistance.
The areas of an automobile depicted in Figure 1 show where these five steps of coating and other additional coatings are used. As can be realized when examining this figure, a significant number of specific coatings and materials are needed in addition to the above-listed steps to manufacture a salable automobile.
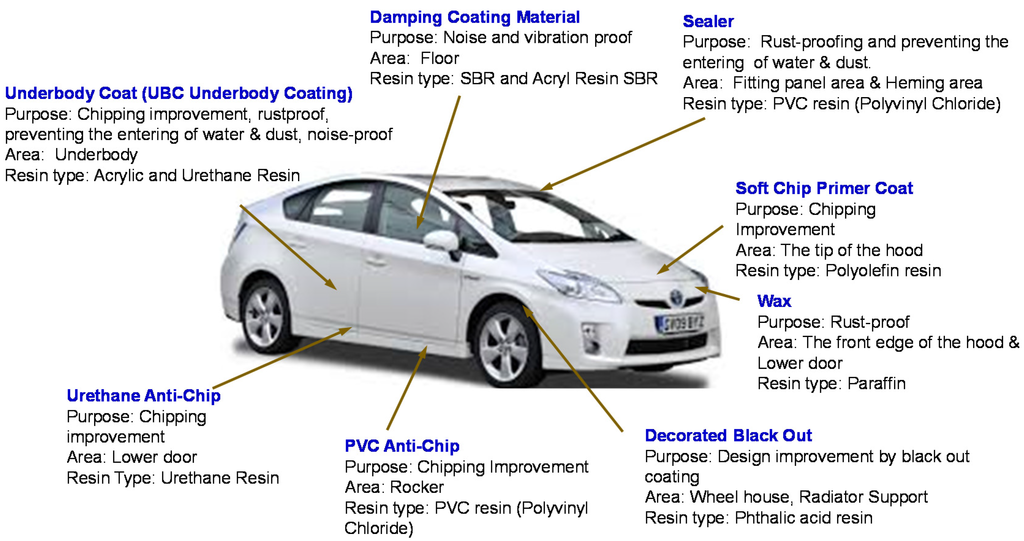
Figure 1.
Summary of coating tasks for specific areas.
For the external surfaces of an automobile to be easily visualized, the five main steps of coating produce a layered structure depicted in Figure 2. These coatings are applied in a particular order and have different material and protection or finishing functionalities; also, each layer relates to the others and imparts a desired balance of properties [23].
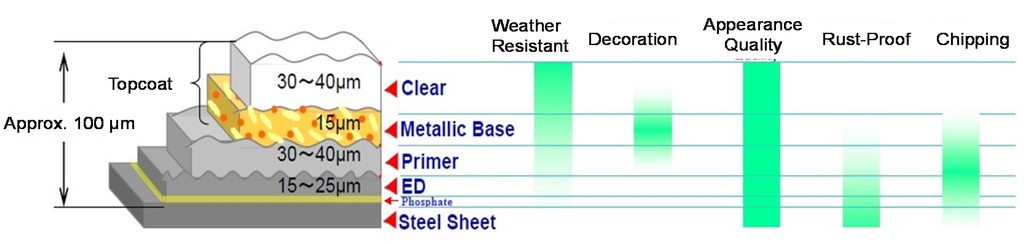
Figure 2.
Automotive coating layers, their thicknesses and purposes for an exterior surface.
2.1. Pretreatment
When the automobile body sheet metal components are welded together, the resulting structure is referred to as the body-in-white (BIW). The first sequence in preparing the BIW for subsequent coating is pretreatment—see Figure 3. Pretreatment consists of cleaning the body surface to remove remaining oils from the stamping process and welding residues through three primary liquid dip processes of degreasing, conditioning, and phosphating [24]. An additional pretreatment cleaning that employs ~pH 9 hot water can also be used. Pretreatment helps the primer to bond onto the metal. A phosphate treatment applies an inert layer of metal phosphate, providing resistance to corrosion spread [25]. The degreasing solution is composed of alkaline salts and surfactants and can include caustic soda, trisodium phosphate, and sodium carbonate. The surfactants are types of detergents for emulsifying oils and lubricants on the BIW. The degreasing zone typically consists of at least two sequences, including a spray stage known as knock-off-degrease (KOD) and a dip sequence. The advantage of using these two is that a significant portion of the oil and dirt will be removed by the high-pressure spray, thereby leaving a relatively lower load for the dip sequence to clean.
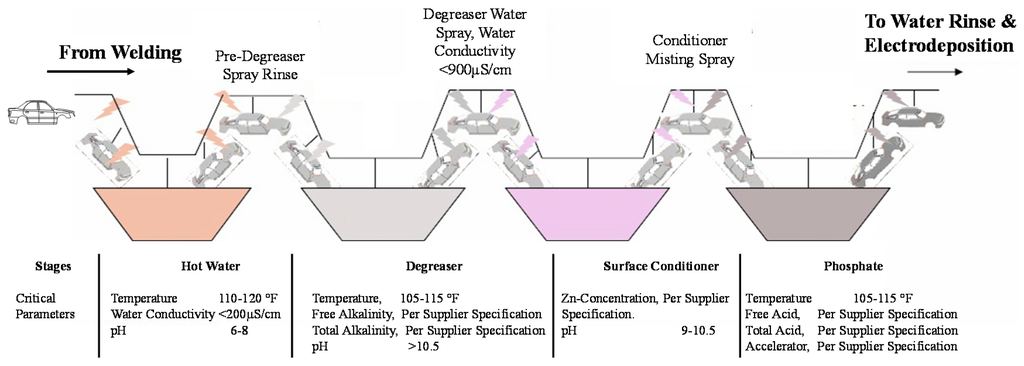
Figure 3.
Pretreatment sequences for the body-in-white.
The next sequence is surface conditioning (also called activation), which creates nucleation sites for phosphate crystal growth. It increases the number of crystallization nuclei on the metal surface that enhance bonding mechanisms for the subsequent phosphate sequence [26]. In conditioning, an aqueous dispersion of titanium orthophosphate with pH between 8 and 10 is typically used. Finally, the liquid dip phosphate solution is composed of phosphoric acid, phosphate ions, nitrate ions, zinc and other divalent metal ions, hydrogen ions, and an accelerating compound. The free acid etches the steel surface, causing hydrogen to be released while metal phosphate ions are precipitated onto the surface in a crystalline form. The primary objective during phosphating is to deposit a thin, dense and uniform conversion layer on the cleaned and prepared metal surface [27].
2.2. Electrodeposition (ED)
The metal underbody and frames of automobiles are coated to prevent corrosion, whereas other areas like the roof are not rust-proofed. When not coated to prevent corrosion, the structural metals (or other materials) are primed before applying additional coatings. Figure 4 displays types of corrosion protection coatings for cold rolled steel.
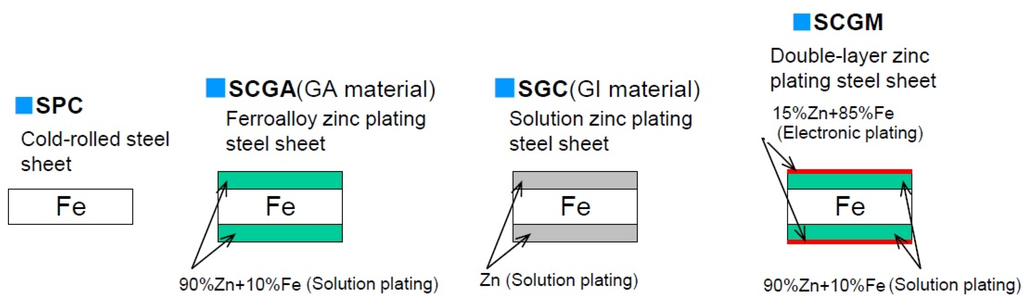
Figure 4.
Various types of steel sheets for car body.
An SCGA electrodeposition solution having a 90% Zn–10% Fe composition (GA material) is the primary coating used in modern automobiles to prevent corrosion. In comparison to the SGC solution, i.e., GI material, with 100% Zn during electrodeposition, the GA material minimizes ED gas pin (paint quality defects) and maximizes spot welding performance; however, the SGC process is still used in Europe. The SCGM corrosion protection method was used in previous decades, and it did enable minimization of ED gas pin. However, its cost was higher than the GA material coating.
The electrodeposition coat or E-coat provides rust and corrosion protection and was first introduced in the 1960s. Since then the use of E-coat has grown at a rapid pace. By 1970, 10% of all cars were electrocoated and by 1990, 90% of all cars were electrocoated. Today, it is the most conventional coating process used in vehicle manufacturing.
The E-coat process has not only grown at a rapid rate but has changed significantly since it was first introduced. From 1964 to 1972 an anodic E-coat process was used, and from 1976 to the present, a cathodic E-coat has been used. The E-coat film thickness has also varied during this time frame. Thicknesses started at 25 µm and remained that way until 1976 when the cathodic process was adopted, and an 18 µm thickness was established; it remained at this value until 1984, when the film thickness was increased to 35 µm. This relatively thick film was used until 1992 when E-coat film thicknesses were decreased to the current value of 20 µm [5].
The E-coat solution consists of a mixture of resin, binder, a paste containing the pigments, and a solvent. During anodic electrodeposition, metal parts are positively charged while the paint is negatively charged; because small amounts of metal ions migrate into the paint film, it limits the performance properties of the coating. Hence, anodic coatings are mainly used for interiors and offer excellent color and gloss control. In cathodic electrodeposition, the metal part is negatively charged while the paint is positively charged. In contrast to the anodic process, the cathodic process reduces the amount of iron that can enter into the paint film. Hence, cathodic coatings offer high performance and excellent corrosion resistance and have mostly replaced anodic coatings in North America. The E-coat process is considered to be relatively environmentally friendly; it uses only a 0.5% solvent solution [28].
The pH of the solvent in the ED tank is a measure of the balance of acidic and caustic components in the tank. The balance is dominated by the functional groups in the resin and the neutralizing agents. A decreased pH in the cathodic ED tank lowers film thickness so that higher voltage needs to be applied with the danger of rupture effects [5]. The pH can be influenced by the pretreatment chemicals in a detrimental way. In such a case, conductivity and pH drift to values outside of the specification and corrections have to be made by replenishing ultrafiltrate with deionized water (D.I.) water.
As shown in Figure 5, E-coating involves dipping automobile bodies into the coating solution and passing an electric current through the body and the liquid ED paint solution. Because of the charged nature of this coating process, the ED paint penetrates into places a spray would not reach. The ED paint is promoted to bond with the metal substrate, and a uniform coating thickness is produced. The result is an insoluble, deposited layer with a very high solids content and firm adherence to the pretreated body [25].
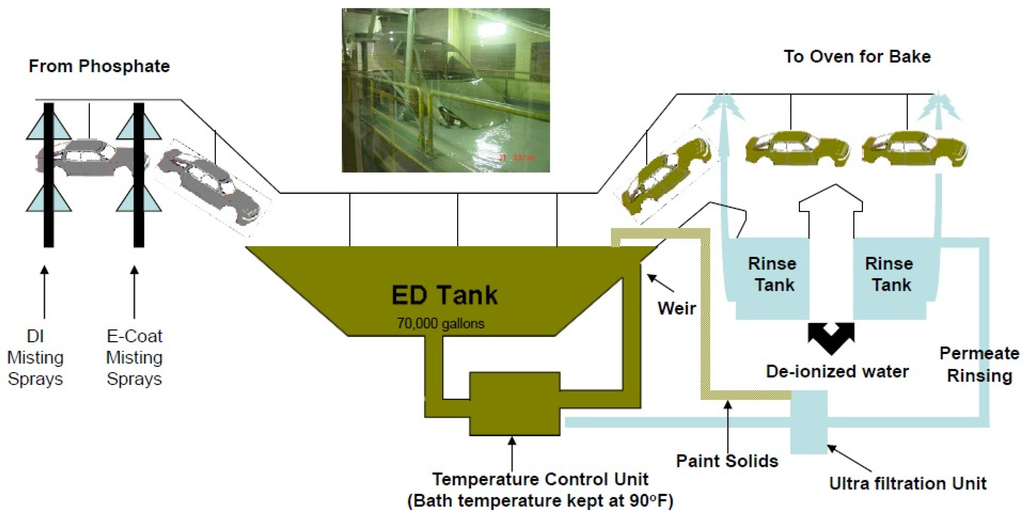
Figure 5.
Electrocoating process.
During operation, a mixture of resin, binder, and a paste containing the pigments is fed into the electrodeposition tank. The automobile body is lowered into the tank, and an electric current applied; the solution in the tank consists of 80%–90% deionized water and 10%–20% paint solids (consisting of resin, binder, and pigment). The deionized water acts as a carrier for the paint solids, which are under constant agitation. The resin is the backbone of the final paint film and provides corrosion protection, durability, and toughness. Pigments are used to provide color and gloss. To control paint performance during cathodic E-coating, purging of the paint and regulating the amount of applied voltage are essential. As the coating grows in thickness, it becomes more and more insulating, thereby causing the deposition process to slow. As an automobile body exits the tank, paint solids adhering to the surface but not bound to the surface are rinsed off to recover the ED solids via an ultrafiltration unit to ensure surface smoothness [28].
After the E-coat, the automobile body enters a bake oven, in which heating and keeping at a temperature of 160 °C for 10 min causes film curing to promote maximum performance properties. The oven temperature and heating time primarily enhance chip resistance and film adhesion to the body; corrosion protection is affected less by these conditions. Because surface roughness influences the smoothness and brilliance of the topcoat, some sanding of the surface is usually performed to remove or eliminate defects before the body enters the next application step.
A common challenge in the current E-coating process is that of water spots that contaminate coated surfaces. These spots can appear on the air-dried surface if water droplets from the conveyor or other sources fall on the body, especially if the water in the tanks is not deionized properly and has high conductivity; the water spots are usually a remnant of unwanted ions in the water and will cause problems in subsequent coating applications and their adherence and appearance. If water spots are present, they have to be removed by sanding. Hence, it is necessary to use well-deionized water and routinely monitor its conductivity. Alternately, operators also add surfactants or ultrafiltrates to the water rinse zone to eliminate or manage water spots.
2.3. Rust-Proof Materials: Sealer/PVC
The third step is underbody coating (UBC) and seam sealing using PVC (Polyvinyl Chloride) and urethane. This sealing has multiple purposes, as shown in Table 1.

Table 1.
Purposes and types of sealants used in automobiles.
The sealant is applied around and inside the doors, hood, trunk, and front dash, and onto the exterior and interior of metal joints and outer area of the back wheel well; application of the sealant is accomplished either manually or with robots and prevents air and water ingress and inhibits rust formation. Recently, PVC and acryl/urethane sealants have also been used in the underbody areas, a process called the Dampening Coat (DC), to impart noise-proofing and vibration-deadening. The noise and vibration are transferred from the engine, drive train, suspension system, road noise of the tires, and flowing air, and the underbody sealants reduce noise transfer into the passenger compartment of the automobile [29]. The underbody coating also provides anti-corrosion and chipping protection; typically, it is accomplished using robots equipped with airless sprayers. Figure 6 displays the lower door sections and rocker panel locations where PVC or urethanes are applied as an anti-chipping protection, and shows the cross section of this layer with respect to the other coatings on the body parts.
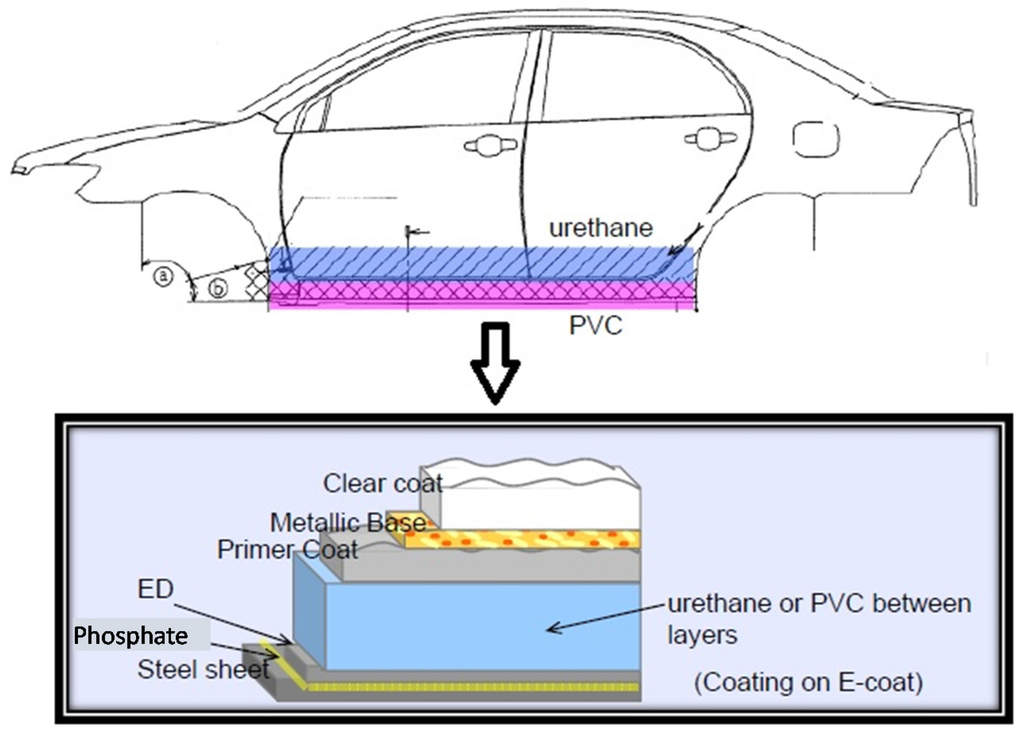
Figure 6.
The anti-chipping material coating on lower doors and rocker panels.
During this third step, it is also typical to apply a soft tip primer coat that improves the chipping resistance (resistance against chipping by flying stones or debris). This layer is applied to the front edge of the hood, which is an area prone to chipping shock, using a high elastic resin that resides between the ED and primer coats. Also, during the third step, the radiator supports, wheel housing, and under rear areas of the body are coated with a relatively dull black pigment called a blackout coating.
2.4. Primer
The fourth coating step is the application of a primer surfacer or simply primer. It can be water-borne, solvent-borne, or a powder. The main reason for primer application until 1990 was to improve weather resistance, appearance, and chipping resistance. Since 1990, primers have been developed in solvent-borne and water-borne materials, and powders (explicitly for reducing the amount of volatile organic compounds (VOC) discharged into the atmosphere or pollutant capture equipment). Today’s primers have to improve adhesion between the E-coat and topcoat, provide chipping protection, enhance paint appearance and, also, be environmentally compatible with emission regulations.
The primer fills and smooths minor imperfections and scratches that may be created during prior steps and by the intermediate sanding or grinding used to remove small imperfections. Additionally, most automobile bodies have surface imperfections that arise from the pressing, stamping, and welding operations, and these defects are typically accentuated during the ED step. Hence, although the main purpose of the primer has been to act as a leveler that produces a smoother finished surface [30], it also provides additional protection against corrosion and maximizes adhesion between it and the E-coat and basecoat; thereby, the primer increases paint durability. However, as pressing and stamping operations become more sophisticated and create fewer imperfections, the smoothing characteristics of primers become less important.
The primer layer is important for stone chip resistance. When chipping exposes the bare metal, the perforation will inevitably lead to corrosion. At the same time, good adhesion of the top coat to the primer surfacer is required to ensure minimal detraction from the visual appearance if chipping has occurred. Hence, it is common to incorporate suitable colors in primers that are compatible with the topcoat, masking damage to the topcoat caused by stone chipping.
The type of primer surfacer used also affects operational aspects of this coating step. A few decades ago most paint shops only used solvent-borne paints, but improvements in water-borne and powder technologies have resulted in widespread shifts to the use of water-borne and powder primers [31]. In the future, it is possible that powder technologies will become the preferred choice among most automotive manufacturers. Because each country has different regulations regarding emissions and environmental controls, the primer surfacers used also vary from country to country. Nevertheless, primer surfacers have to satisfy a broad range of requirements, such as environmental compatibility, compatibility with the manufacturing processes and application methods, and associated costs. For these reasons, it is usually the cooperation between automotive manufacturers, paint suppliers, and coating manufacturers that results in the creation of a new product.
Water-borne primers and paints can be thinned and applicators cleaned with water. They produce significantly lower VOC emissions than solvent-borne paints; water-borne paints and primers also have better thermoplastic properties and are more durable than solvent-borne paints. On the other hand, solvent-borne paints and primers produce a harder finish that is more tolerant of adverse conditions during the drying period [32].
Powdered primers are composed almost exclusively of non-volatile materials; importantly, any overspray from powders can be recycled, whereas such recycling is very difficult with water-borne and solvent-borne primers. Hence, powdered primers are more environmentally friendly and, with recycling, can be less expensive than the alternatives.
During the development of primers, they are usually tested for surface hardness and elasticity, as well as for their adhesive properties onto ED coating and base coating materials [33]. It is crucial to test for surface hardness because it plays a significant role in compatibility with defect correction measures such as sanding. Elasticity provides an indication of the potential stone chip protection capacity of the primer surfacer coat. Adhesion to ED coats and base coats is also essential to minimize or prevent corrosion and to obtain an optimal surface finish.
Besides the mechanical and adhesive properties of primers, their chemical reactivity to ultraviolet (UV) radiation and extreme weather conditions such as high humidity and low or high temperatures has to be assessed because automobiles are constantly exposed to these conditions during their life cycle [33]. For example, if a primer surfacer chalks or powders when exposed to UV light, it is evidence of a disintegration of the film-forming agent within the coat and, as a result, a drastic loss of adhesion and delamination of the topcoat is likely to take place [34]. Hence, a high-gloss surface is important throughout the lifetime of an automobile and is enhanced in currently applied primers through the addition of appropriate pigments that not only provide color but also support the mechanical performance of the film. These types of issues are important for owners who, after having an automobile for some time, may see significant paint degradation as a sign of the poor quality of the vehicle. In other words, both long-term impressions and short-term reactions to paint quality are necessary for purchases and ownership of vehicles. Hence, manufacturers of primers test the smoothness of the primer coatings to ensure their applicability and appearance effects on the topcoat. Coating smoothness obtained via a wave scan method generates values in short-wave and long-wave scales.
Rheology of the liquid primer surfacer is necessary to assess its effectiveness. Rheology relates to flow characteristics, the measure of which is the viscosity as determined by shear rates using a rheometer [35]. A rather high viscosity level prevents sediment formation during primer storage, but spray application requires a viscosity as low as is feasible to optimize atomization. However, if the viscosity is too low, film sagging on vertical surfaces will result. Even for ideal viscosity, sagging on vertical surfaces will occur if the primer film is too thick. For powder coatings, the particle size distribution is a critical factor [36]. A high content of fine powder will significantly impact the effect of van der Waals forces of attraction between the particles, the result of which is a tendency for particles to agglomerate and become impossible or difficult to spray.
Primer coating operations in an automobile assembly plant are usually implemented in three stages: interior coating, exterior coating, and then curing in an oven. Manual spray painting is used for the interior coating of the doors, engine compartment, and luggage or trunk space. Some of these areas are not sprayed with basecoat. It is important that the primer color matches the basecoat to be able to impart uniform coating appearances between interior and exterior coats. The film thickness of the interior layer is near 20 μm. Robot primer painting of the exterior is accomplished after the interior priming is completed, typically using rotating bell applicators, to form a film of 25–40 μm in thickness. Subsequently, the primer is cured by passing the body through an oven at 140 °C for 30 min. For water-borne primers, preheating (or flashing) of the body before oven exposure is necessary because of the higher specific heat and longer drying times of water compared to that of solvent-borne primers.
2.5. Topcoat
The final step in the body coating process is to apply the topcoat, which consists of two layers—the basecoat and clearcoat. The basecoat contains the primary coloring pigment, and the clearcoat provides a protective coating against environmental effects, corrosion, and UV light degradation, promotes unmatched color retention, and provides a smooth, unblemished, and even finish [25]. The body is first coated with either a water-borne or solvent-borne basecoat. If the manufacturer is using a wet-on-wet process, then the clearcoat is applied onto the basecoat after a short flash-off, before curing in an oven. In the absence of a wet-on-wet process, the basecoat is cured before the clearcoat is applied. Independent of which process is employed, typical thermal cure times and temperatures are 30 to 40 min at 125 °C, respectively [37].
Application of these final coats occurs within spray booths that include: air handling systems for temperature, humidity, and cleanliness control; and paint and VOC capturing equipment. The air handling system contains filtration media for dust and dirt control. Water and large air flow are used to capture paint particles in wet scrubbers. The controlled air flow within the booth transports the paint particles into the capturing equipment in the under-section of the booth. The force and direction of the air flow, the efficiency of filtration, and the characteristics of the coating equipment determine not only the overall efficiency of the coating operation but also the quality of the final finish.
It must be noted that advancement in dry scrubber technologies is leading the industry in the replacement of wet scrubbers with dry scrubbers. Dry scrubbers offer a significant reduction in water usage and carbon dioxide and particulate emissions.
2.5.1. Basecoat
As part of the topcoat, the basecoat is the third layer added to the car after the primer and before the clearcoat. It imparts the automobile’s color; roughly 40,000 base coat colors are known today, and approximately 1000 new colors are added to this list each year [5].
Value, i.e., lightness or darkness, along with hue and chroma, are standard terms used in the color industry to describe the three-dimensional nature of the color created by using pigments in an automobile’s basecoat. Pigments entail any colored, black, white, or fluorescent particulate solid that is insoluble in the paint in which the pigment is incorporated. Pigments retain their crystal or particulate structure throughout the basecoat coloration process. Effect pigments include aluminum flakes, micas, and other types of light interference agents [11]. With aluminum flake effect pigments, the value or lightness of the coating and its differentiation between a white, gray, or black scale depends on the observation angle; this effect is called lightness flop. With mica effect pigments, the hue, i.e., color classification, depends on the observation angle; this effect is referred to as a color flop. With variation in the concentration of a pigment, the chroma or color saturation, i.e., the color’s level of intensity and richness, can be changed [38]. Hence, with various pigments and effect materials, it is possible to design an unlimited number of automobile finishes having unique and distinct values, hues, and chroma.
Three different types of basecoats are used for automobiles, including solvent-borne medium solids (MS), solvent-borne high solids (HS), and water-borne. The MS basecoats contain 15%–20% solid levels and have a spray viscosity of 100 mPa.s at 1000 rpm. The HS basecoats were developed from MS basecoats in response to the U.S. Environmental Protection Agency (EPA) requesting that the solid levels in solvent-borne spray paints be increased, and the organic solvents decreased to minimize VOC emissions. Waterborne basecoats have become the primary basecoat used throughout the U.S. automobile manufacturing industry since 2000 due to their environmental benefits.
2.5.2. Clearcoat
The final coating on an automobile’s surface is the clearcoat; it provides durability, environmental etch, and scratch resistance to the overall coating. It protects against damage such as fading that is caused by UV rays from the sun and imparts a gloss and depth that otherwise are not as vivid. The clearcoat also makes repairs and maintenance easier.
Environmental etch is an appearance issue associated with the formation of permanent water spots or non-removable marks from bird droppings, tree resin, or other chemicals after contact with an automobile’s surface. The physical damage resulting from etching is associated with a localized loss of material and deformation or pitting of the clearcoat surface.
To impart UV durability, HALS (hindered amine light stabilizer) chemicals are mixed with UVA (ultraviolet light absorbers) into automotive clearcoats during their manufacturing. The primary functions of these chemicals are to prevent UV-induced degradation of the polymeric backbone (by HALS) and to absorb UV radiation within the 290–400 nm wavelength range. The concentration of UVAs in clearcoats has been shown to decrease by about 50% after four years of exposure to the sun; this depletion is a consequence of photochemical degradation and subsequent diffusion from the clearcoat. The HALS complements the activity of the UVAs by scavenging free radicals that are formed on the surface of the coating, where the UVAs cannot effectively shield the polymer, or within the coating at wavelengths at which the UVAs may not absorb efficiently [39].
The scratch resistance of clearcoats is another factor that affects the long-term appearance of an automobile’s surface. Some fine scratches can only be seen when light is scattering from the scratches themselves; for example, fine scratches or marring introduced by car wash machines are a consequence of slight surface deformations that can level over time. Other scratches cause the clearcoat to ablate or break. The flow characteristics and mechanical durability of clearcoats depend on whether they are 1K (does not require a hardener, catalyst, or activator for hardening) or 2K (needs to be mixed with a hardener, catalyst, or activator to harden). 1K clearcoats have shown a higher resistance to plastic flow and 2K clearcoats commonly have shown a higher fracture and harder impact resistance. This scratch resistance has been related to the cross-linking density and elasticity of the polymer network [40].
Liquid clearcoats are the most dominant form worldwide because the application techniques, as well as the chemistry, are well understood and optimized; they also have excellent performance for automotive coating. The chemical composition of the liquid clearcoats varies somewhat according to different market needs or customer specifications, but nearly all systems are based on acrylic resin backbones that are stable during storage and handling at room temperature [41].
The 1K acrylic melamine clearcoat is the most common paint used in the automotive industry because of its good cost and performance balance [42]. It is typically based on a combination of acrylic polyols (Ac) and amino cross-linking agents (MF, melamine resins). Versions of clearcoat with improved etch and scratch resistance include acrylic melamine silane and carbamate melamine. Both 1K and 2K polyurethane clearcoats also offer an excellent balance of etch and scratch resistance and 1K and 2K epoxy acid clearcoats are among the most commercially successful clearcoats regarding acid etch resistance [43]. However, when formulated as a 1K system, the shelf life of epoxy acid clearcoats is rather limited.
Waterborne clear coat was first introduced in the automotive industry in 1990 and is based on a polyester acrylate cross-linked with isocyanate and melamine resins. Powder clearcoats are becoming more prevalent in the automobile industry because of their environmental benefits, i.e., no emission of VOCs during application [44]. Besides this, powder clearcoats have other advantages, including:
- Direct recycling—overspray powder can be collected and be reused in the original coating process;
- No wastewater or paint sludge produced during their application;
- No need to use organic solvents for cleaning of application equipment or spray booth;
- Reduction of the total energy used during application because the air supplied to the spray booth can be recycled;
- No VOCs produced and very low toxic aspects;
- Uniform film thicknesses and similar appearance on horizontal and vertical surfaces.
Challenges with primer and topcoats:
Some of the challenges or problems, and their causes, associated with primer surfacers and topcoats include:
- Problem: Orange peel—surface looks like the skin of an orange.
- Causes: Nozzle held too far away from the surface; shop temperature is too high (for best results, apply between 65 °F and 75 °F); spray nozzle is clogged.
- Problem: Blistering—bubbling, pitted, or swollen appearance.
- Causes: Trapped solvents; painting over dirt or moisture; rust under the surface.
- Problem: Mottled surface—uneven appearance (generally affects metallic coatings).
- Causes: Paint not mixed thoroughly; painting nozzle held too close to the surface; surface too hot or too cold.
- Problem: Fish eyes—small crater-like blemishes in the finish.
- Causes: Improperly cleaned surface, e.g., silicone on the surface.
- Problem: Lifting or wrinkling—a swelling or wet film.
- Causes: Improper drying of the previous coating; lacquer applied over enamel.
- Problem: Runs or sags—coating does not adhere uniformly to the surface being painted.
- Causes: Paint drying too slowly; paint sprayed over wax, oil, or grease; finishing coats applied too thick; paint or work surface too hot or too cold.
- Problem: Dry spray—dry paint dust settled onto a previously painted area.
- Causes: Paint is sprayed too quickly or too slowly; sprayer tip is too far from the surface.
2.5.3. Spray Coating
The anti-chip coating, primer surfacer, basecoat, and clearcoat are typically applied by spraying, thereby requiring the use of atomizers. The quality of atomization influences the quality of the sprayed coating. Also, other factors such as the chemistry and material composition of the coatings influence the quality of the finished surfaces.
A spray is considered as a system of droplets immersed in a continuous gaseous phase [45]; spray phenomena are common in power and propulsion applications, industrial applications, and nature [46]. Sprays are produced as a result of atomization, which is defined as the disintegration of liquid into droplets [45,47] by diverse mechanisms including aerodynamic, mechanical, ultrasonic, or electrostatic forces. For example, the breakup of liquid into droplets can be achieved by impingement on a liquid with gas in two-fluid atomization, by centrifugal forces in rotary atomization, by rapid vibrations established by a piezoelectric transducer in ultrasonic atomization, or by electrostatic/electromagnetic fields in electrostatic/electromagnetic atomization.
For the automotive industry, spray coating is achieved by using either air spray guns or high-speed rotary bell atomizers, both of which can be either electrostatic or non-electrostatic. Automatic spray coating requires the use of robots and, as such, automatic coating is sometimes referred to as “robotic coating”. Spray coatings require the use of spray booths. These are large, environmentally controlled enclosures that help to increase the transfer efficiency (TE) of the paint onto a target and also direct overspray (paint particles that do not coat the car and float in the air) and solvents into an emission abatement section of the booths.
Automotive spray painting applicators consist of two main types: air spray guns and high-speed rotary bell. However, the most commonly used as well as the most efficient spray applicator currently used in automotive applications is the electrostatic rotary bell (ESRB) [48].
2.5.4. Issues with the Current Automotive Spray Coating Technologies
The paint spray cycle of an automobile can be visualized to start from the paint guns that spray toward a car surface. After the applicator atomizes the paint, only a portion of the paint droplets coat a vehicle surface. Broad and significant issues with automotive painting include: paint transfer efficiency (TE); coating deposition; energy consumption; energy efficiency; inspection of finished surface qualities; and environmental and workplace safety. Issues related to finish quality can be analyzed regarding the atomizer designs, paint formulation and composition, and surface modification.
The TE of paint can be defined as the ratio of the amount of paint that coats a surface relative to the amount that is supplied to an atomizer. Although automotive spray painting is among the most sophisticated and controlled industrial painting operations currently performed, only 50%–60% of the paint supplied to a paint atomizer is reported to reach and remain on a vehicle [49]. This relatively low TE remains a fundamental problem in automobile painting. Finding ways to increase the TE while maintaining a high-quality finish is a challenging task that has attracted the attention of researchers for many years.
In general, poor TE is an economic burden and an environmental and worker’s health concern. A one percent improvement in TE can save millions of dollars per year in the cost of paint-shop bulk material usage. A higher TE reduces the amount of VOC emissions, the amount of paint sludge produced, associated after-treatment costs, and overspray paint capturing hardware. Furthermore, decreased amounts of over-spray allow a reduction in the booth airflow requirements, creating significant energy and cost savings.
The following items are well-documented factors that affect TE [50]:
- Target surface characteristics (composition, geometry, and position);
- Finishing spray technologies;
- Operating parameters;
- Spray booth air conditions (relative humidity and temperature);
- Liquid coating characteristics.
One implication of a 50%–60% TE is that the remaining 40%–50% overspray paint must be captured before it is emitted into the environment. Due to the substantial amount of paint used, water washing or wet scrubbing has been the preferred overspray capturing method. The ratio of the amount of overspray captured to the amount entering the capturing system is defined as the overspray capturing efficiency. Capturing more overspray while using less energy is fundamentally important because of both environmental and operational cost issues. A strong demand exists for efficient overspray capturing technologies [51,52] because of environmental regulations.
3. Automotive Coating Performance
The performance of an automotive coating can be assessed from different views, e.g., the durability of the exterior and interior paint finishes or the aesthetic features [53]. However, limitations exist on the paint properties, process capabilities, and, most importantly, the amount of money that can be spent to improve the finish [2]. Consequently, each automotive company defines its color and appearance standards, which are to meet or exceed the levels reached by competitors and customer expectations. Particularly important is the uniformity—or harmony—among all components. In the case of the exterior finish, it is necessary that add-on parts such as bumpers, spoilers, mirror housings, and other decorative trim parts match the neighboring body panels. Differences in color and appearance are especially obvious on automobile models with panels having very tight fits to each other.
3.1. Coating Quality
Coating qualities can be judged on three main criteria: protection against harsh environments; durability; and appearance quality. A harsh environment refers to damage caused by falling objects (fall out), ultraviolet (UV) rays, hot (above 80 °C) or cold (below −20 °C), scratches, rock hits (chipping), and the rust from salt or road deicers. A measure of durability is to have an automobile with rust proofing for over 20 years and good color/gloss appearance for over a decade. The three parameters for determining appearance quality are color, paint smoothness, and gloss [54].
To better understand elements of the features mentioned above, a closer look at the components of paints or coatings is necessary. The components of paints that become the coating films on an automobile include the pigments, resins and additives, and solvents that impart fluidity. Pigments mainly give color and luster; they also help to establish thickness to the coatings. Resins, including synthetic and cross-linkers, and hardening agents, form the coating film. Finally, additives, which act as anti-settling agents, stabilize paint and make it easy to use. The factors creating appearance quality are visual quality (aesthetics), determined by the spray coating and involving color and brightness and influenced by the presence or absence of metallic flakes; appearance (smoothness), which is related to film unevenness with wavelengths in the range of 1–5 mm; shine, which is relevant to unevenness with a wavelength of 0.01–0.1mm; and color and flopping performance or the ability of the film to show color changing properties depending on the angle viewed.
3.1.1. Gloss and Smoothness of Paint Material
Unevenness in the films formed during spraying is mainly related to atomization performance and liquid properties. The better the atomization of a paint material, i.e., producing smaller Sauter mean diameters of the droplets, the less unevenness is formed in the film when spraying. The more a paint wets a surface and with less viscosity, the more the amplitude of unevenness is decreased (Figure 7); lower viscosities facilitate more film leveling and also sagging. During curing of the coatings, any unevenness in the film transfers to the surface when the flow speed of the wet film decreases below the film shrinkage speed. When the flow speed of the wet film falls below the shrinkage speed, minute unevenness and non-uniform distribution of the pigment transfers to the surface and deteriorates the gloss (Figure 8). The suppression of shrinkage in a complete layered system can be achieved by using optimized solvent contents and additional hardening agents, and by increasing the coating thickness to enable adequate flow.

Figure 7.
Effects of wettability and paint particle size on final coating smoothness.
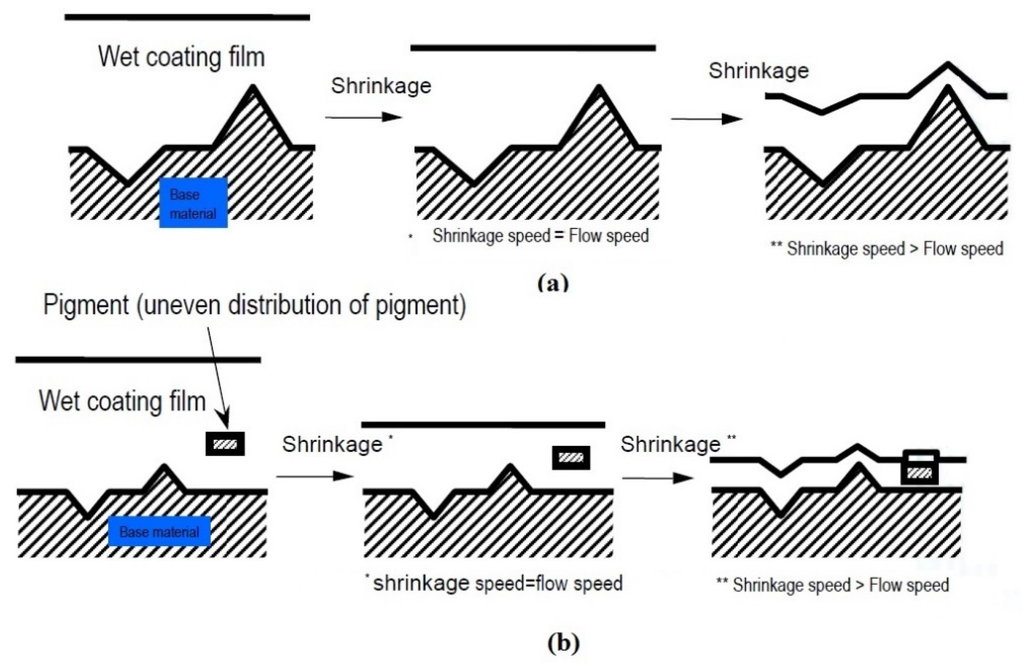
Figure 8.
Formation mechanisms of unevenness with (a) wavelength 1–5 mm and gloss unevenness with (b) wavelength 0.01–0.1mm caused by paint material shrinkage on a base material.
3.1.2. Color
Uniform and consistent color is essential to achieve the impression of a high-quality finish and avoid customer complaints. This issue is important not only at the time of purchase but also throughout the lifetime of a vehicle. Factors that influence color tone are pigment orientation and concentration. Pigment orientation is impacted by atomization, the viscosity of the paint after coating a surface, the interference of dust, and the spray impact speed onto a surface. Pigment concentration on a surface is also affected by the TE of the spray and pigments onto the substrate. An acceptable, good metallic luster surface is achieved if the metallic or mica effect pigments are oriented parallel to the surface; if not, the surface will look dull and has a smaller value (i.e., darker in appearance). In practice, coloring pigments act like fine flakes that move within a wet film. Therefore, it is hard to ensure pigments are oriented with their largest particle area parallel to the surface. For this reason, several additives are used to ensure aluminum flakes orient parallel to the surface and are uniformly distributed; generally, these additives increase paint viscosity immediately after a surface is sprayed and, thereby, prevent pigment movement and disorientation.
Additionally, to ensure a surface-parallel orientation of aluminum flakes, it is necessary that the spray droplet impact speed with a surface is high enough; the speed is affected by viscosity and paint droplet diameter. If the droplets are too small, the concentration of pigments within them is also small, and a low TE will ensue. When the TE is high, the concentration of aluminum flakes in the coating will be high, as will the level of brightness.
Figure 9b shows the distribution of lustrous pigment particles and paint droplets. The density of the lustrous pigment within paint droplets as a function of the paint particle size has a bell-shaped curve (Figure 9a); it is small for small droplets such that the area ratio of the lustrous pigment-to-paint droplet is small. Small diameter paint droplets either do not contain lustrous pigment or contain little lustrous pigment, making the lustrous pigment concentration low. Increasing the size of the paint droplets increases this area ratio, such that medium diameter paint droplets contain significant lustrous pigment, making the lustrous pigment density high. On the other hand, after attaining a maximum value, the area ratio decreases as the size of the paint particles increases. Because large diameter particles contain lustrous pigments of various diameters, it results in a medium pigment density. In contrast, the arrangement or orientation of the lustrous pigment is determined by impact orientation and shrinkage orientation (Figure 10). Impact orientation is caused by the deformation of the paint droplets when they impact the surface or the wet layer, and is hardly affected by viscosity. Shrinkage orientation is caused by shrinkage of the wet film when the viscosity is above a critical value; if viscosity is not above this value, the pigment particles will not orient and flopping performance decreases. Hence, flopping performance of metallic flakes is controlled through changes in their diameter distribution (the main effect), concentration, and arrangement conditions of the lustrous pigment.
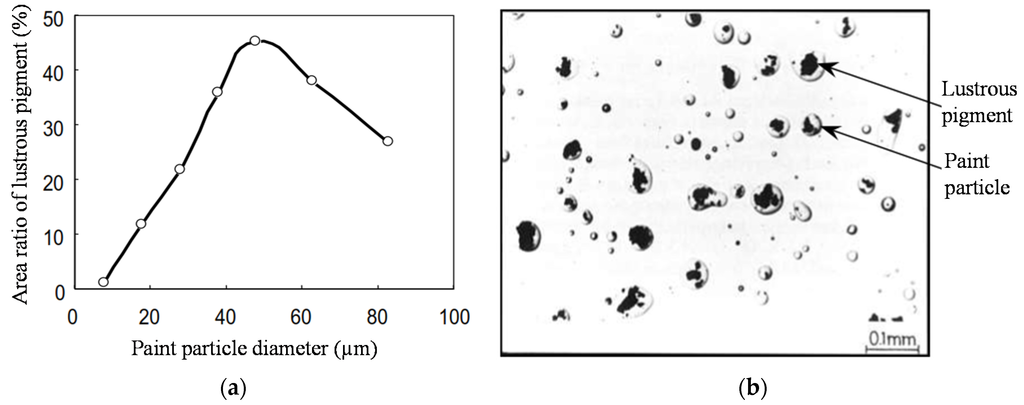
Figure 9.
(a) Lustrous pigment density in paint particles; (b) distribution of lustrous pigment for small, medium, and large paint particles.

Figure 10.
Affective parameters on the arrangement conditions of lustrous pigment.
3.2. Corrosion Protection
Automobile coatings are subjected to a number of different environments and situations during their service life. Degradation of the coating depends on three main factors: coating formula, the intensities of the environment or situation, and their duration. Exposure to UV from the sun, heat, and humidity has a long-term deleterious effect with different degrees of intensity depending on the climate and weather conditions. However, these three factors can be simulated in controlled, laboratory weathering tests to assess the lifespan of the coatings. Often, automobile coatings are also subjected to corrosive gasses and microorganisms; these degradation factors are usually only recreated in laboratory tests for special application situations [55].
To suppress or eliminate corrosion, it is necessary to gain a solid understanding of it. All metals after production or shaping have a tendency to seek their lower energy, a natural state which originally was that in the ore from which it originated. This tendency is caused by the Law of Entropy, i.e., the lower energy state is that of an oxidized, corroded state. In automobiles, corrosion usually results from electrochemical reactions in which the steel is immersed in a suitable electrolyte, like water and road salt, and will create some localized anodes and cathodes. As depicted in Figure 11, anodic oxidation and cathodic reduction are occurring, during which electrons flow from the anodes to cathodes. As a result of the anodic dissolution, a perforation in the metal will happen eventually. Furthermore, corrosion can occur when two different metals are in contact in the presence of an electrolyte. Called bimetallic corrosion or the galvanic effect, the metal with the lower potential will become the anode with the production of electrons and associated metallic ions, and the other metal will act as the cathode.
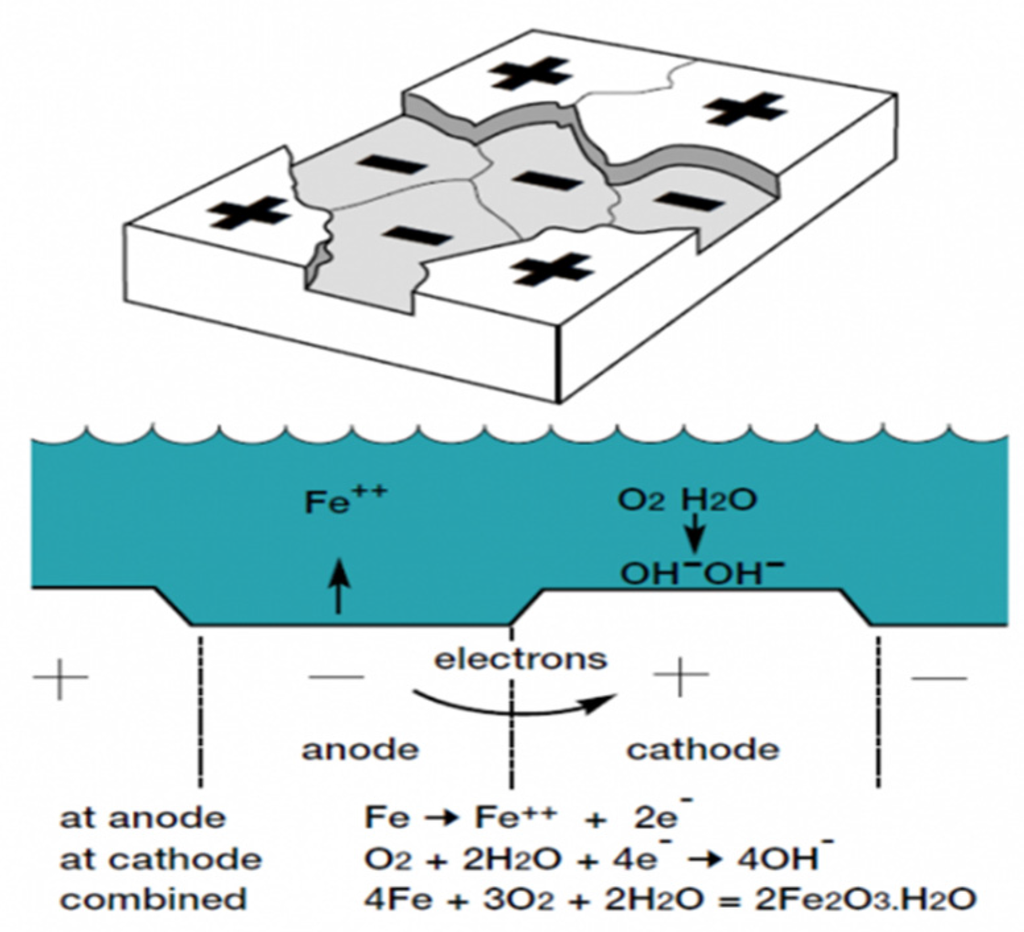
Figure 11.
Corrosion of an automotive body structure.
Factors that affect corrosion include humidity, temperature, a variety of air pollutants such as chlorine, sulfur oxides, and nitrogen oxides, and winter road de-icing salts. These factors can lead to several forms of corrosion.
A number of effective corrosion protection systems are available commercially, the most common of which includes the coating of steels with zinc. It provides the substrate with resistance to corrosion by cathodic control because zinc has some characteristics that make it well suited for iron and steel products. Zinc coating, or galvanization, is carried out through different methods including:
- Batch Hot-Dip Galvanizing: is the process of immersing fabricated steel or iron into a bath of molten zinc. The iron in the steel metallurgically reacts with the zinc to form a tightly-bonded alloy coating.
- Metallizing: is done by melting zinc powder or zinc wire in a flame or electric arc and then projecting the liquid zinc droplets via air or gas transport onto the surface to be coated.
- Zinc Rich Paint: Zinc painting, commonly referred to by the misnomer “cold galvanizing”, is the application by brush or spray of zinc dust mixed with organic or inorganic binders.
- Continuous Sheet Galvanizing: is also a hot-dip process, but is only applied to steel sheet, strip, and wire.
- Electroplating: electrogalvanized (electroplated) coatings are created by applying zinc to steel sheet and strip by electrodeposition.
- Mechanical Plating: mechanical zinc plating is accomplished by tumbling small parts in a drum with zinc and proprietary chemicals.
- Zinc Plating: is identical to electrogalvanizing in principle because both are electrodeposition processes. However, zinc plating is used on small parts such as fasteners, crank handles, springs, and other hardware items rather than on sheet metal.
In addition to zinc coatings, effective design principles of automobile bodies, in addition to providing aesthetic and aerodynamic qualities, can decrease the accumulation and ingress of debris and minimize the prominence of features prone to stone chipping. Aspects of design that influence corrosion resistance can be divided into the following.
- Styling features: it is at the external “clay” modeling stage of an automobile body that the major panel split can be decided, i.e., when the actual panel configuration is planned that determines the number of joints and associated seams, all of which are potential sites for crevice corrosion.
- Subassembly: in subassembly design it is important to avoid complexity because complexity inevitably impairs adequate coverage of panels by paint and waxes. The real danger areas are touching surfaces that promote crevice corrosion and cause decreased coating thicknesses.
- Panel and Design: individual panel design features should include drain holes located at the lowest extremity of the section in subsequent parent subassemblies.
4. Trends in Automotive Coating Processes
As described in the above, current trends in automotive coating process are motivated by lowering manufacturing costs, delivering customer satisfaction via aesthetic features and corrosion protection, and mitigating environmental concerns. A tremendous amount of consideration has been put into current automotive coating systems, and they have a level of sophistication that satisfies most customers around the world. Compared to 30 years ago, the problem of corrosion is almost alleviated, and the durability and appearance of the topcoats have become acceptable for the lifetime of a car. In fact, with the worldwide emergence of two-layer topcoats, the color, gloss, and chip resistance of automobile coating remain in relatively excellent condition during the first 7–10 years of an automobile’s use. Aesthetic features are also trending with automotive fashions. Hence, a closer look at recent trends in automotive coatings is presented in the following.
4.1. Powder Coating
One solution to the emission of VOCs has been the replacement of liquid coatings with coatings in the form of dry, particulate solids, commonly called “powder” coatings. Their compositions contain very low concentrations of volatile solvents, on the order of 2%, substantially less than any other paint system [56]. Currently, the automotive industry uses powder coating on wheels, bumpers, hubcaps, door handles, decorative trim and accent parts, truck beds, radiators, filters, and numerous engine parts. A clear powder topcoat has also been developed; BMW and Volvo are using it on their new model cars, and GM, Ford, and Chrysler have formed a consortium to test it on their production lines. Powder coatings are an advanced method to apply a decorative and protective finish to almost any type of metal and can be used both by industry and consumers [57].
The powder used is a mixture of fine particles of pigment and resin that is electrostatically sprayed onto the surface to be coated. The charged powder particles adhere to the electrically grounded surfaces until the powder is heated and fused into a smooth coating in the curing oven. The result is a uniform, durable, high-quality, and attractive finish. Powder coating is the fastest growing finishing technology in North America, representing over 10% of all industrial finishing applications.
Powder coatings emit zero or near-zero VOCs, and can be used to produce much thicker coatings than conventional liquid coatings without running or sagging. Over-spray can be recycled, thereby meaning it may be possible to achieve nearly 100% TE of the coating. Capital equipment and operating costs for a powder line are less than for conventional liquid lines. Items that are powder coated have fewer differences in appearance between horizontally and vertically coated surfaces than liquid coated items. Also, a wide range of specialty effects that are impossible to achieve with other coating processes is easily incorporated into powder coatings. While powder coatings have many advantages over other coating methods, there are some disadvantages to the technology, one of which occurs when powder coatings are used in conjunction with particulate metal particles like aluminum flakes.
4.2. 3-Wet Paint
The automobile industry seeks to eliminate or minimize the extent of spray application processes, space, and baking ovens because of the significant savings in material and energy. One approach has already been successfully established—a wet-on-wet-on-wet-system (3-wet) that mostly eliminates the primer oven in the coating process. An additional step has also been taken—to remove primer surfacer application—with its function preserved by the application of the first basecoat layer.
Waterborne, 3-wet paint systems have been developed to reduce VOCs and CO2 emissions. These systems apply the primer coat, then basecoat and finally the clearcoat, after which curing is accomplished in only one oven. This system has also eliminated the storage/inspection step between the primer and top coat application (Figure 12). Hence, the overall time of coating operations and paint shop size can be reduced to one-half of what is needed without this innovation. More importantly, the amount of VOC emissions is reduced by two thirds. However, some disadvantages are evident in this system, such as a telegraphing of rough surface profiles of the substrate and primer layer and water in the base coat diffusing into the primer coat. These issues may be mitigated by improved steel roughness, ED appearance, and the use of a two-stage bake. Recently, a waterborne 3-wet paint system with appearance qualities equivalent to a conventional three-coat, two-bake (3C2B) paint system was developed by adopting base resins with a low glass transition point to promote leveling, and then by reducing the melamine content of the paint to minimize shrinkage during curing [58].

Figure 12.
Existing and 3-wet processes of the primer coat.
The 3-wet paint technology has the potential to be cost-efficient and environmentally rigorous, and produce durable, high-quality painted vehicles.
5. Summary and Conclusions
The technologies employed in automotive coatings have become much more sophisticated and efficient, and are evolving rapidly. The pace of performance improvement over the past 100 years has been dramatic, and it will continue to accelerate. Coating compositions and application processes are inseparable and mutually dependent. As such, the final coating is an elaborate assembly of multiple layers that perform in unison. Coating composition, together with application procedures, film formation processes, and coated surface characteristics, determines the appearance of a coating film.
Automotive coatings confront an almost limitless variety of environments and environmental assaults. Targeting customers’ expectations, together with maximizing efficiencies and meeting environmental regulations with new processes, has brought automotive coating to a level not imagined 100 years ago. The appearance (color, gloss, and texture) of the surface significantly affects a customer’s perception of product quality. Additionally, customer expectations for the attributes given by the appearance of coatings continue to increase as manufacturers compete to provide surfaces that offer enhanced surface characteristics.
The current paper gave a semi-comprehensive and up-to-date review of these new processes and coating technologies to be of value to industrial practitioners and researchers. New developments in automotive coatings were chronicled and discussed, and then related to improvements in production technologies and paints. Modern automotive coating procedures were also discussed in detail. Finally, the discussion covered recent trends in automotive coating processes and potential future developments.
Acknowledgments
This work was partially supported by development funds of the Institute of Research for Technology Development (IR4TD), College of Engineering, University of Kentucky.
Author Contributions
N.K.A. and K.S. conceived and designed the experiments; N.K.A, G.P, and K.L. performed the experiments; N.K.A., A.S., S.P., and G.P. analyzed the data; N.K.A., S.P., and G.P. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kensium. History Timeline and Types of Automotive Paint. Available online: http://www.eastwood.com/blog/eastwood-chatter/history-timeline-and-types-of-automotive-paint/ (accessed on 7 June 2016).
- Khanna, A.S. High-performance Organic Coatings; Woodhead Publishing Limited: Cambridge, UK, 2008. [Google Scholar]
- Standeven, H. The Development of Decorative Gloss Paints in Britain and the United States C. 1910–1960. J. Am. Inst. Conserv. 2006, 45, 51–65. [Google Scholar] [CrossRef]
- Learner, T. A review of synthetic binding media in twentieth-century paints. Conservator 2000, 24, 96–103. [Google Scholar] [CrossRef]
- Streitberger, H.-J.; Dossel, K.-F. Automotive Paints and Coatings; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008. [Google Scholar]
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–6. [Google Scholar] [CrossRef]
- Fettis, G. Automotive Paints and Coatings; Wiley-VCH: New York, NY, USA, 2008. [Google Scholar]
- Jürgens, U. Implanting change: The role of ‘indigenous transplants’ in transforming the German productive model. In The Transfer and Hybridization of Productive Models in the International Automobile Industry; Boyer, R., Charron, E., Jurgens, U., Tolliday, S., Eds.; Oxford University Press: Oxford, UK, 1998; pp. 319–341. [Google Scholar]
- Prieto, J. Painting the future green. Eur. Coat. J. 2010, 4, 20–25. [Google Scholar]
- Melchiors, M.; Sonntag, M.; Kobusch, C.; Jürgens, E. Recent developments in aqueous two-component polyurethane (2K-PUR) coatings. Prog. Org. Coat. 2000, 40, 99–109. [Google Scholar] [CrossRef]
- Maile, F.J.; Pfaff, G.; Reynders, P. Effect pigments—Past, present and future. Prog. Org. Coat. 2005, 54, 150–163. [Google Scholar] [CrossRef]
- Liu, Y.; Sparer, J.; Woskie, S.R.; Cullen, M.R.; Chung, J.S.; Holm, C.T.; Redlich, C.A. Qualitative assessment of isocyanate skin exposure in auto body shops: A pilot study. Am. J. Ind. Med. 2000, 37, 265–274. [Google Scholar] [CrossRef]
- Bensalah, W.; Loukil, N.; Wery, M.D.-P.; Ayedi, H.F. Assessment of automotive coatings used on different metallic substrates. Int. J. Corros. 2014, 2014, 838054. [Google Scholar] [CrossRef]
- Galitsky, C.; Worrel, E. Energy Efficiency Improvement and Cost Saving Opportunities for the Vehicle Assembly Industry: An Energy Star Guide for Energy and Plant Managers; Lawrence Berkeley National Laboratory, University of California: Berkeley, CA, USA, 2008. Available online: https://www.energystar.gov/ia/business/industry/LBNL-50939.pdf (accessed on 8 June 2016).
- Galitsky, C.; Worrel, E. ENERGY STAR® Guides for Energy Efficiency Opportunities, Featuring the Motor Vehicle Assembly Industry. Available online: http://aceee.org/files/proceedings/2003/data/papers/SS03_Panel4_Paper_11.pdf (accessed on 8 June 2016).
- Geffen, C.A.; Rothenberg, S. Suppliers and environmental innovation: the automotive paint process. Int. J. Oper. Prod. Manag. 2000, 20, 166–186. [Google Scholar] [CrossRef]
- Kwaambwa, H. A review of current and future challenges in paints and coatings chemistry. Prog. Multidiscip. Res. J. 2013, 3, 75–101. [Google Scholar]
- Cheah, L.W. Cars on a Diet: The Material and Energy Impacts of Passenger Vehicle Weight Reduction in the U.S. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, September 2010. [Google Scholar]
- Caruso, M.M.; Delafuente, D.A.; Ho, V.; Sottos, N.R.; Moore, J.S.; White, S.R. Solvent-promoted self-healing epoxy materials. Macromolecules 2007, 40, 8830–8832. [Google Scholar] [CrossRef]
- Yang, Y.; Urban, M.W. Self-healing polymeric materials. Chem. Soc. Rev. 2013, 42, 7446–7467. [Google Scholar] [CrossRef] [PubMed]
- Fischer, H.R.; García, S.J. Active Protective Coatings. In Active Protective Coatings: New-Generation Coatings for Metals; Hughes, A.E., Mol, J.M.C., Zheludkevich, M.L., Buchheit, R.G., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 139–156. [Google Scholar]
- Baghdachi, J.; Hernandez, H.R.P.; Templeman, C.G. Self-stratifying automotive topcoat compositions and processes. U.S. Patent 7,863,375, 4 January 2011. [Google Scholar]
- Ansdell, D. Painting of plastic body components for cars. Ind. Product. Eng. 1980, 3, 30–35. [Google Scholar]
- Chang, L.P. Analysis of the Influence of Car Body Structure on the Pretreatment & Electrophoresis Process. Shanghai Coat. 2011, 8, 27–28. [Google Scholar]
- Akafuah, N.K. Automotive paint spray characterization and visualization. In Automotive Painting Technology; Springer: Dordrecht, The Netherlands, 2013; pp. 121–165. [Google Scholar]
- Maeda, S. Surface chemistry of galvanized steel sheets relevant to adhesion performance. Prog. Org. Coat. 1996, 28, 227–238. [Google Scholar] [CrossRef]
- Narayanan, T.S. Surface pretreatment by phosphate conversion coatings—A review. Rev. Adv. Mater. Sci. 2005, 9, 130–177. [Google Scholar]
- Loop, F.M. Cathodic Automotive Electrodeposition; No. 780189; SAE Technical Paper; SAE International: Warrendale, PA, USA, 1978. [Google Scholar]
- Tomalino, M.; Bianchini, G. Heat-expandable microspheres for car protection production. Prog. Org. Coat. 1997, 32, 17–24. [Google Scholar] [CrossRef]
- Moon, J.I.; Lee, Y.H.; Kim, H.J.; Noh, S.M.; Nam, J.H. Synthesis of elastomeric polyester and physical properties of polyester coating for automotive pre-primed system. Prog. Org. Coat. 2012, 75, 65–71. [Google Scholar] [CrossRef]
- Poth, U. Automotive Coatings Formulation: Chemistry, Physics und Practices. Vincentz Network GmbH & Co KG: Hannover, Germany, 2008. [Google Scholar]
- Noble, K.L. Waterborne polyurethanes. Prog. Org. Coat. 1997, 32, 131–136. [Google Scholar] [CrossRef]
- Misev, T.A.; Van der Linde, R. Powder coatings technology: New developments at the turn of the century. Prog. Org. Coat. 1998, 34, 160–168. [Google Scholar] [CrossRef]
- Razin, A.A.; Ramezanzadeh, B.; Yari, H. Detecting and estimating the extent of automotive coating delamination and damage indexes after stone chipping using electrochemical impedance spectroscopy. Prog. Org. Coat. 2016, 92, 95–109. [Google Scholar] [CrossRef]
- Lenges, C.P.; Niu, Y.; Hsiao, Y.L.; Ding, J.; Barsotti, R.J.; Kelly, R.J.; Kim, Y.H. Rheology control agents for coating compositions. U.S. Patent 7,652,121, 26 January 2010. [Google Scholar]
- Ageev, E.V.; Novikov, E.P.; Altukhov, A.Y.; Tikhomirov, V.P. Size Distribution of Powdered Aluminium Sample Microparticles Produced Using Electroerosion Dispersion. In Proceedings of the 2015 International Conference on Mechanical Engineering, Automation and Control Systems (MEACS), Tomsk, Russian Federation, 1–4 December 2015; pp. 1–4.
- Wu, Y.H.; Surapaneni, S.; Srinivasan, K.; Stibich, P. Automotive Vehicle Body Temperature Prediction in a Paint Oven; No. 2014-01-0644; SAE Technical Paper; SAE International: Warrendale, PA, USA, 2014. [Google Scholar]
- Pfaff, G. Special Effect Pigments: Technical Basics and Applications; Vincentz Network GmbH & Co KG: Hannover, Germany, 2008. [Google Scholar]
- Gerlock, J.L.; Kucherov, A.V.; Smith, C.A. Determination of active HALS in automotive paint systems II: HALS distribution in weathered clearcoat/basecoat paint systems. Polym. Degrad. Stab. 2001, 73, 201–210. [Google Scholar] [CrossRef]
- Betz, P.; Bartelt, A. Scratch resistant clear coats: Development of new testing methods for improved coatings. Prog. Org. Coat. 1993, 22, 27–37. [Google Scholar] [CrossRef]
- Noh, S.M.; Lee, J.W.; Nam, J.H.; Byun, K.H.; Park, J.M.; Jung, H.W. Dual-curing behavior and scratch characteristics of hydroxyl functionalized urethane methacrylate oligomer for automotive clearcoats. Prog. Org. Coat. 2012, 74, 257–269. [Google Scholar] [CrossRef]
- Dutt, W.; King, J.G. Method of producing a polished metal effect finish on a vehicle. U.S. Patent 8,512,802, 20 August 2013. [Google Scholar]
- Collong, W.; Göbel, A.; Kleuser, B.; Lenhard, W.; Sonntag, M. 2K waterborne clearcoat—A competition between crosslinking and side reactions. Prog. Org. Coat. 2002, 45, 205–209. [Google Scholar] [CrossRef]
- Papasavva, S.; Kia, S.; Claya, J.; Gunther, R. Characterization of automotive paints: An environmental impact analysis. Prog. Org. Coat. 2001, 43, 193–206. [Google Scholar] [CrossRef]
- Lefebvre, A. Atomization and Sprays; CRC Press: Boca Raton, FL, USA, 1988; Volume 1040. [Google Scholar]
- Sirignano, W.A. Fluid Dynamics and Transport of Droplets and Sprays; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Liu, L.S. Experimental and Theoretical Investigation on the Characteristics and Two-Phase Spray Flow Field of Effervescent Atomizers. Ph.D. Thesis, Tianjing University, Tianjin, China, 2001. [Google Scholar]
- Corbeels, P.L.; Senser, D.W.; Lefebvre, A.H. Atomization characteristics of a highspeed rotary-bell paint applicator. At. Sprays 1992, 2, 87–99. [Google Scholar] [CrossRef]
- Domnick, J. Effect of bell geometry in high-speed rotary bell atomization. In Proceedings of the 23rd Annual Conference on Liquid Atomization and Spray Systems, Brno, Czech, 6–8 September 2010; p. 69.
- Lee, I.; Kim, D.; Koo, J. Liquid jet breakup structure and transfer efficiency of a two-stage air-blast injector. At. Sprays 2012, 22, 561–579. [Google Scholar] [CrossRef]
- Salazar, A.J.; McDonough, J.M.; Saito, K. Computational Fluid Dynamics Simulation of the Automotive Spray Painting Process. Comput. Model. Simul. Eng. 1997, 2, 13–144. [Google Scholar]
- Salazar, A.J.; Saito, K.; Alloo, R.P.; Tanaka, N. Wet scrubber and paint spray booth including the wet scrubber. U.S. Patent 6,093,250, 25 July 2000. [Google Scholar]
- Wicks, Z.W.; Jones, F.N.; Pappas, S.P.; Wicks, D.A. Color and Appearance. In Organic Coatings: Science and Technology, 3rd ed.; Wiley-Interscience: New York, NY, USA, 1999; pp. 382–416. [Google Scholar]
- Gómez, O.; Perales, E.; Chorro, E.; Burgos, F.J.; Viqueira, V.; Vilaseca, M.; Pujol, J. Visual and instrumental assessments of color differences in automotive coatings. Color Res. Appl. 2016, 41, 384–391. [Google Scholar] [CrossRef]
- Schulz, U.; Trubiroha, P.; Schernau, U.; Baumgart, H. The effects of acid rain on the appearance of automotive paint systems studied outdoors and in a new artificial weathering test. Prog. Org. Coat. 2000, 40, 151–165. [Google Scholar] [CrossRef]
- Craven, J.M. Powder coating composition for automotive topcoat. U.S. Patent 4,346,144, 24 August 1982. [Google Scholar]
- Weiss, K.D. Paint and coatings: A mature industry in transition. Prog. Polym. Sci. 1997, 22, 203–245. [Google Scholar] [CrossRef]
- Hayashi, H.; Ishii, M. Development of 3-Wet Paint System with Improved Appearance-Paint Design Based on Transfer Mechanism of Unevenness from Under Layers; No. 2014-01-1048; SAE Technical Paper; SAE International: Warrendale, PA, USA, 2014. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).