Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives
Abstract
1. Introduction
1.1. Overview of Extreme Environments
1.2. Importance of Wear and Corrosion Resistance in Critical Applications
1.3. Scope and Objectives of the Review
2. Types of Extreme Environments and Their Challenges
2.1. High-Temperature Environments
2.2. Corrosive Chemical Environments
2.3. High-Pressure and Abrasive Conditions
2.4. Space and Cryogenic Applications
3. Coating Materials for Extreme Environments
3.1. Ceramic-Based Coatings
3.2. Metallic and Alloy Coatings
3.3. Polymer and Composite Coatings
3.4. Nanostructured and Multilayered Coatings
4. Coating Techniques and Processes
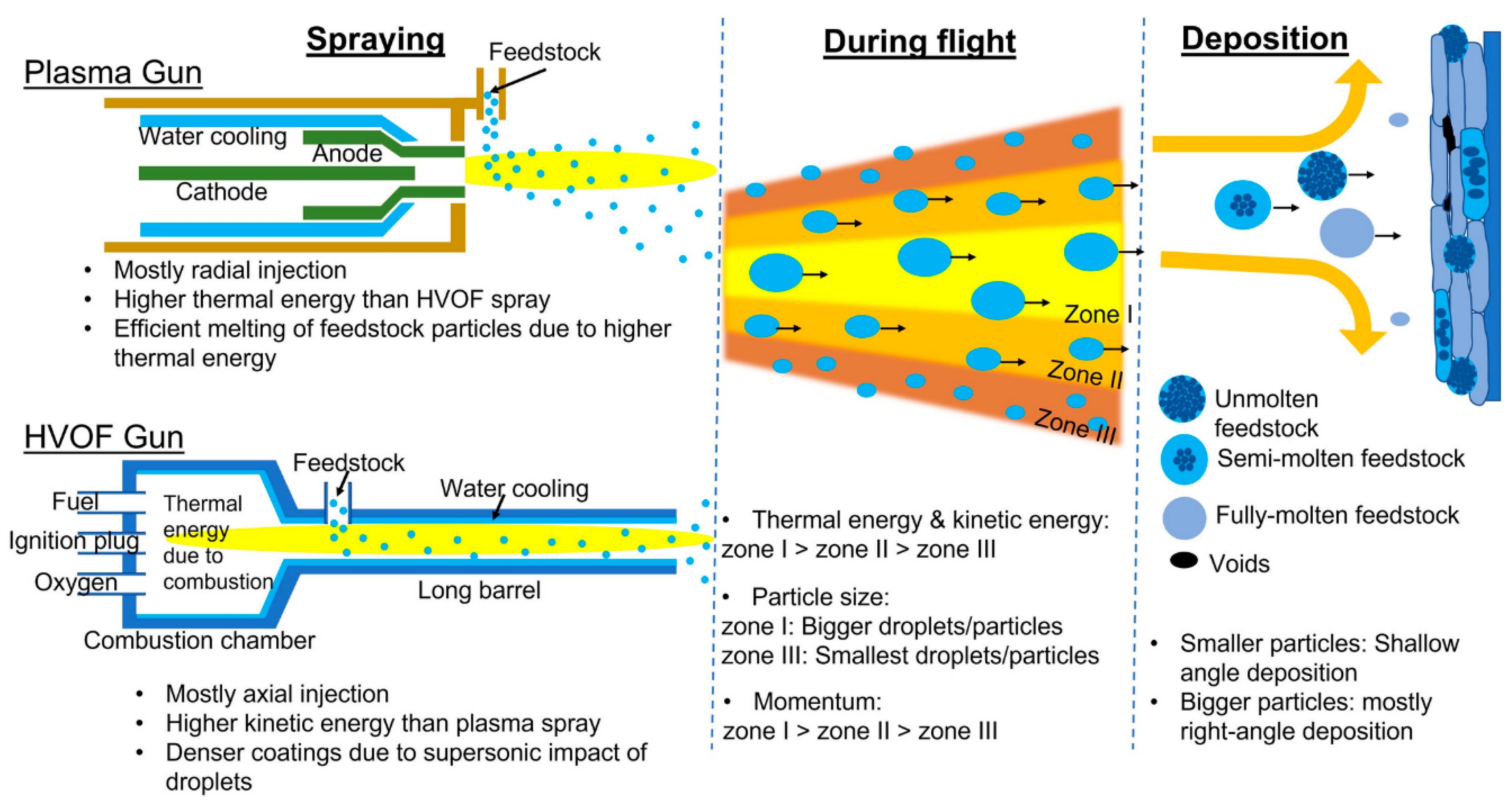
4.1. Thermal Spray Coatings: Plasma Spray and High-Velocity Oxygen Fuel (HVOF)
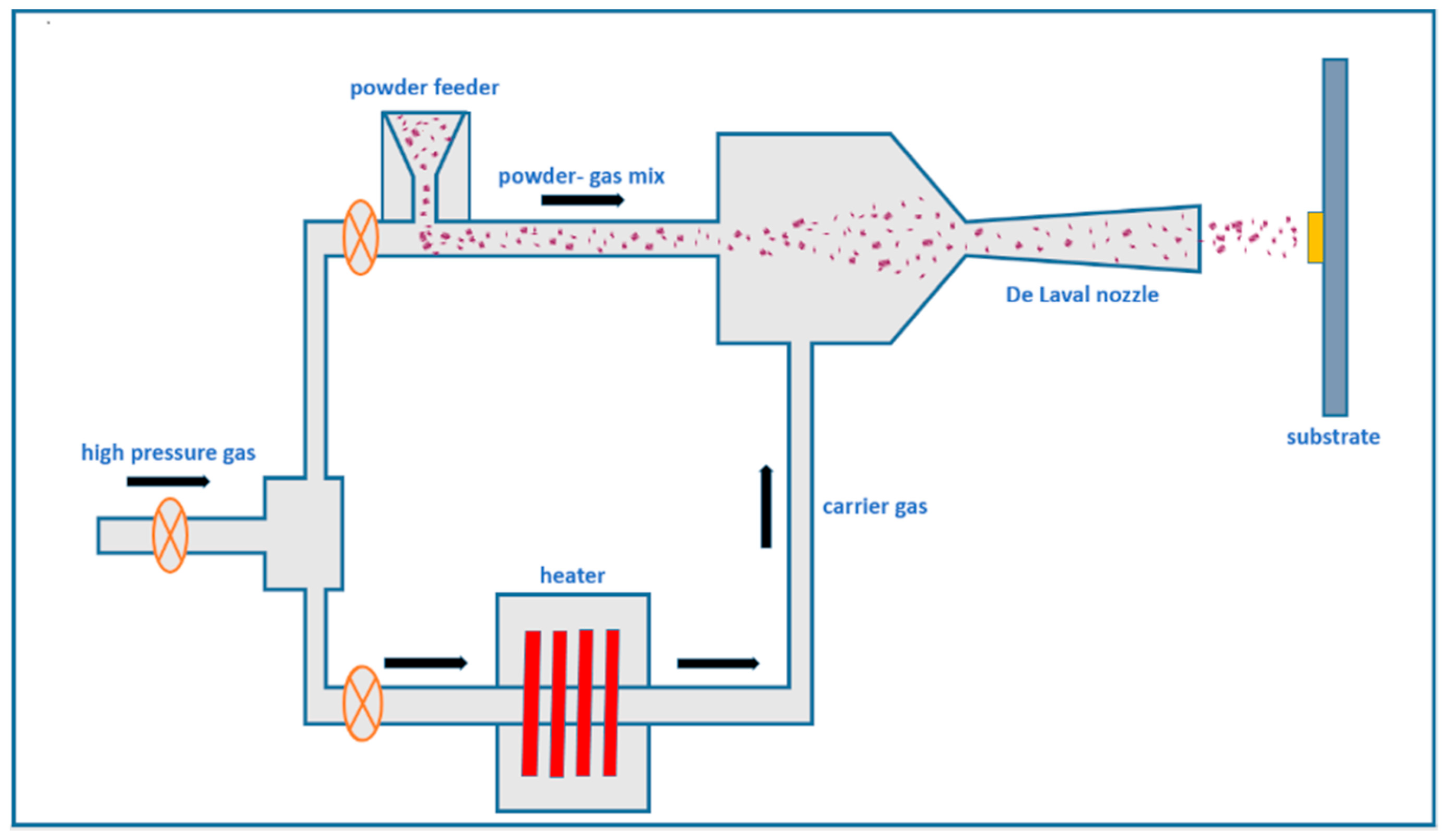
4.2. Cold Spray Technology
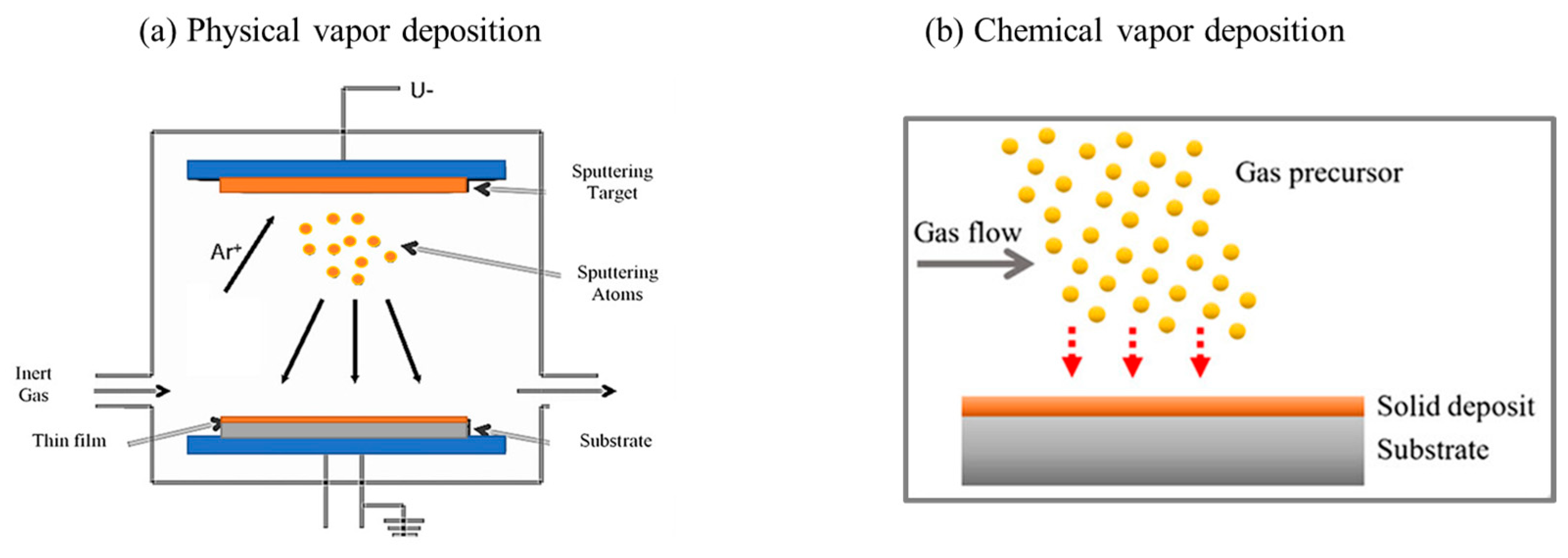
4.3. Chemical and Physical Vapor Deposition (CVD and PVD)
4.4. Electrochemical and Electrodeposition Methods
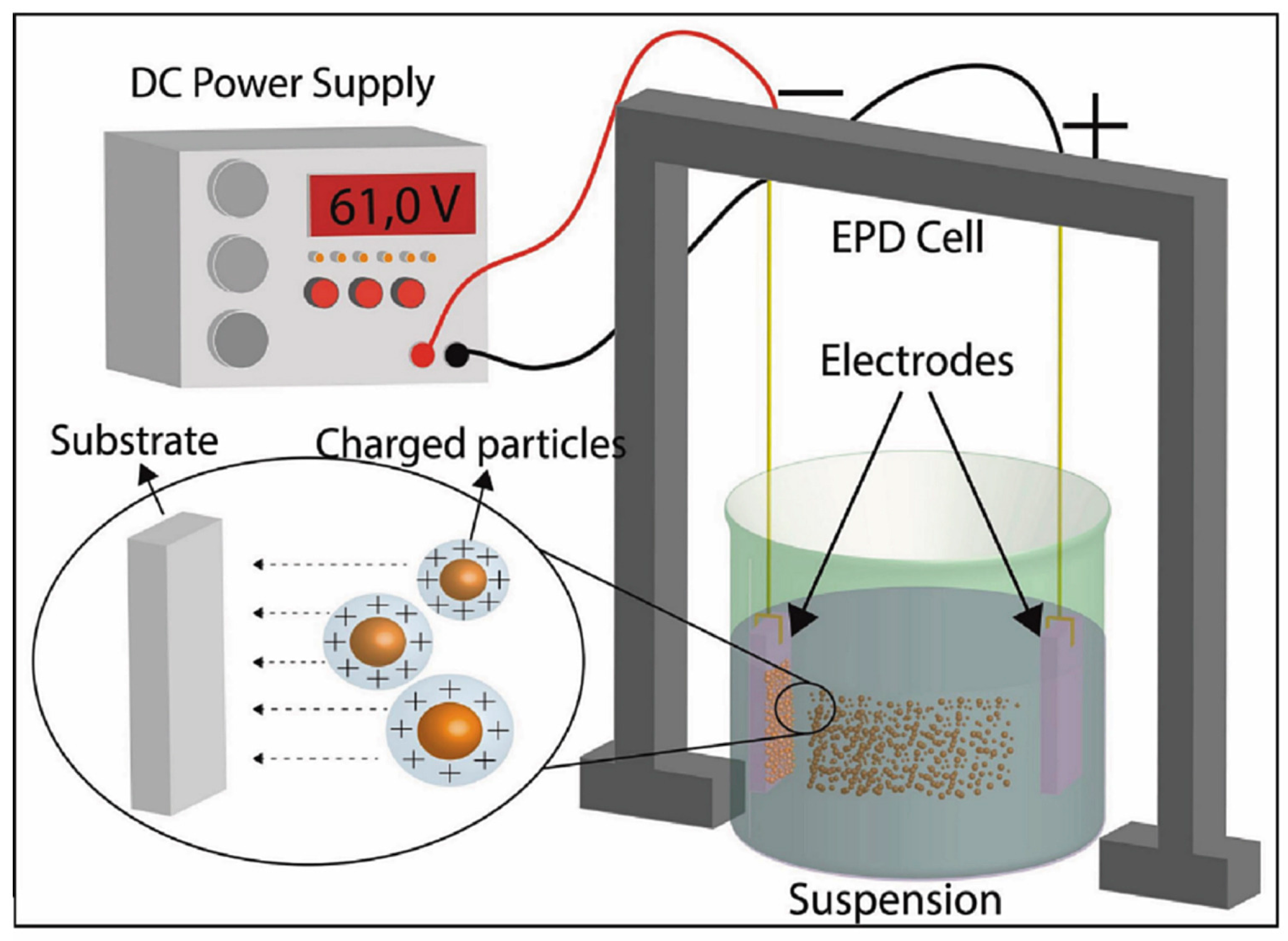
4.5. Electrophoretic Deposition and Aerosol Deposition
4.6. Additive Manufacturing and In Situ Coating Techniques
4.7. Coating Techniques for Polymers
5. Wear and Corrosion Mechanisms in Coatings
5.1. Adhesive and Abrasive Wear
5.2. Oxidation and Hot Corrosion
5.3. Stress Corrosion Cracking and Pitting
5.4. Tribocorrosion: Synergistic Wear and Corrosion
6. Applications of Wear- and Corrosion-Resistant Coatings
6.1. Aerospace and Aviation Components
6.2. Oil and Gas Drilling Equipment
6.3. Marine and Offshore Structures
6.4. Nuclear and Power Generation Systems
6.5. Biomedical Implants in Aggressive Body Environments
7. Advances and Innovations in Coating Technologies
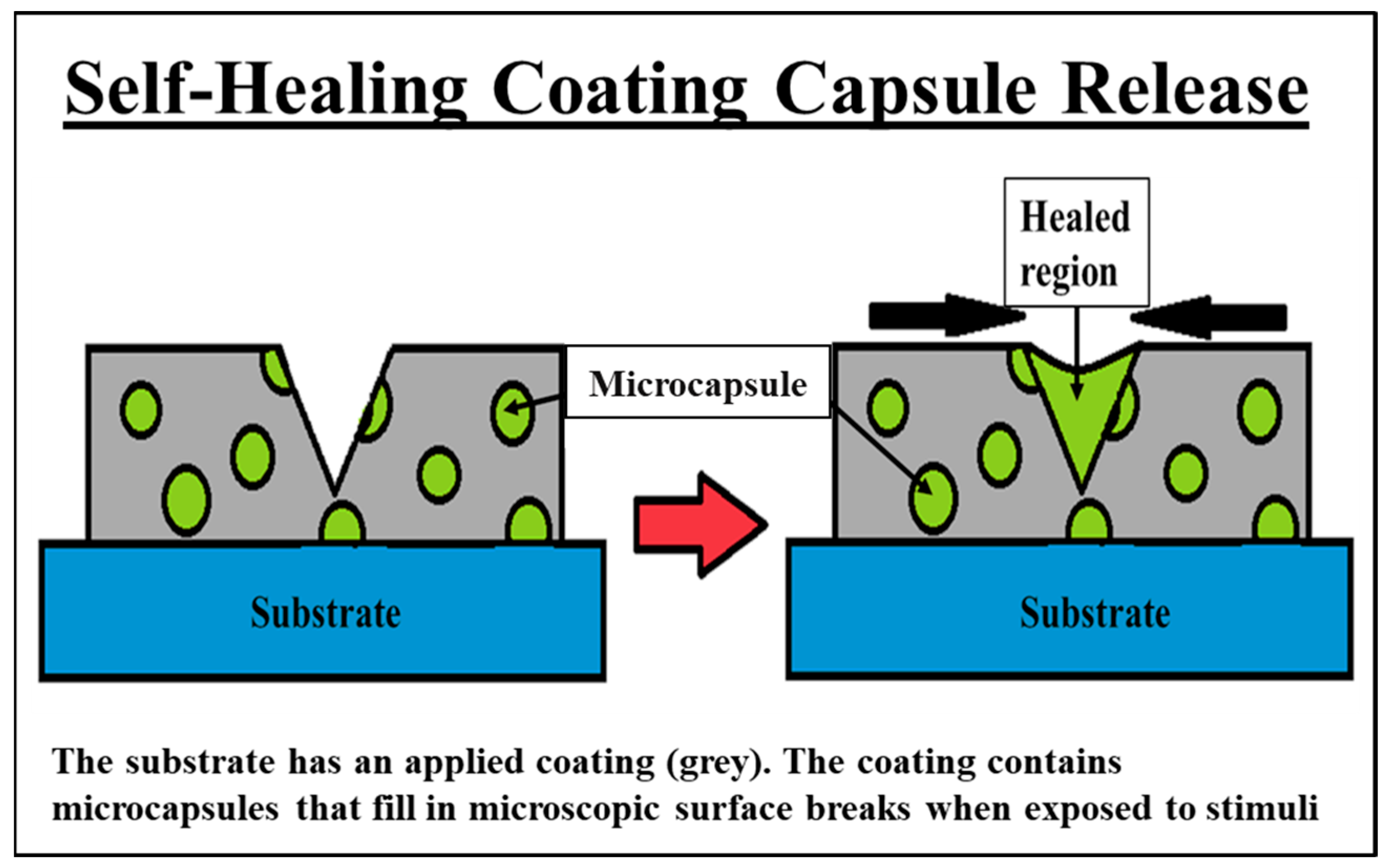
7.1. Self-Healing and Smart Coatings
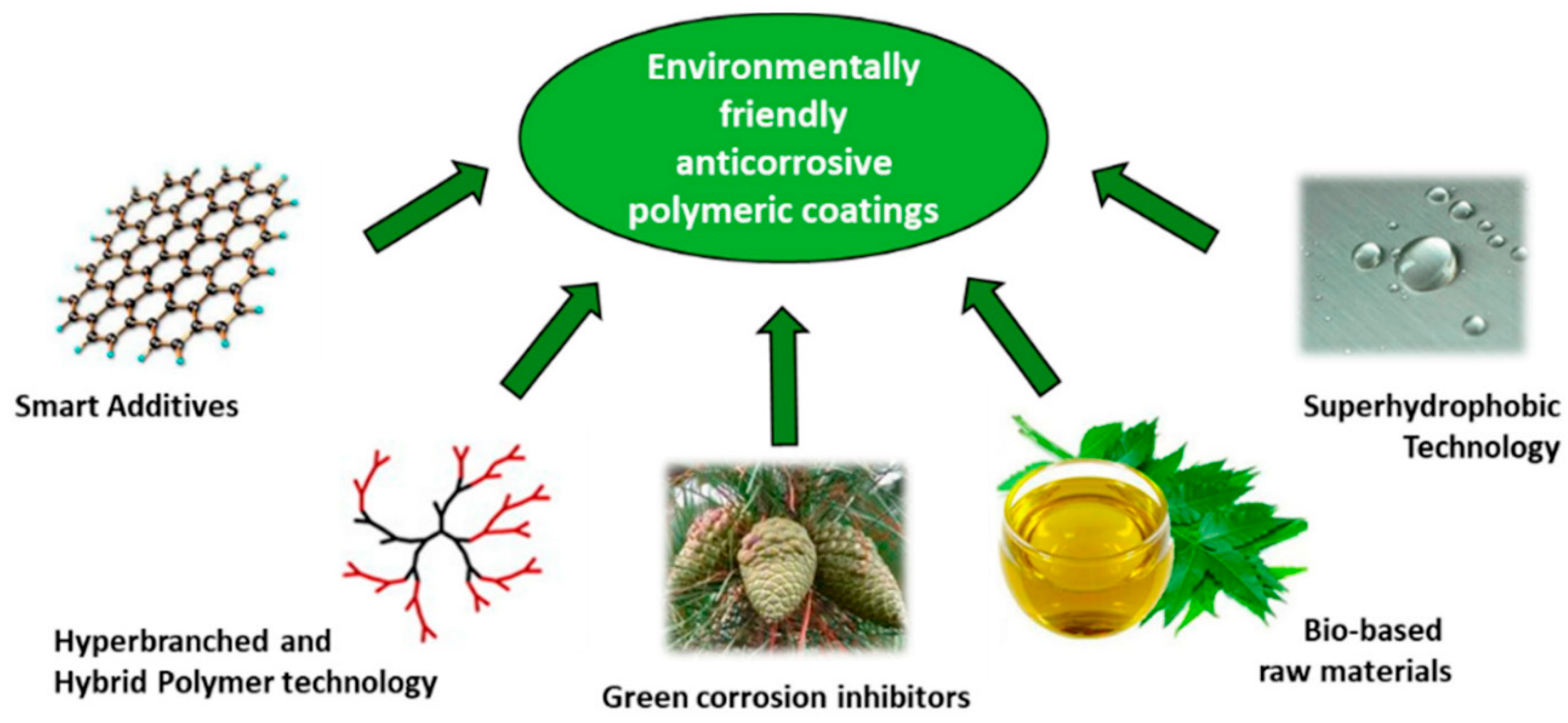
7.2. Environmentally Benign Coating Process
7.3. Nanostructured and Functionally Graded Coatings
7.4. Role of Machine Learning in Coating Design and Optimization
8. Challenges and Limitations
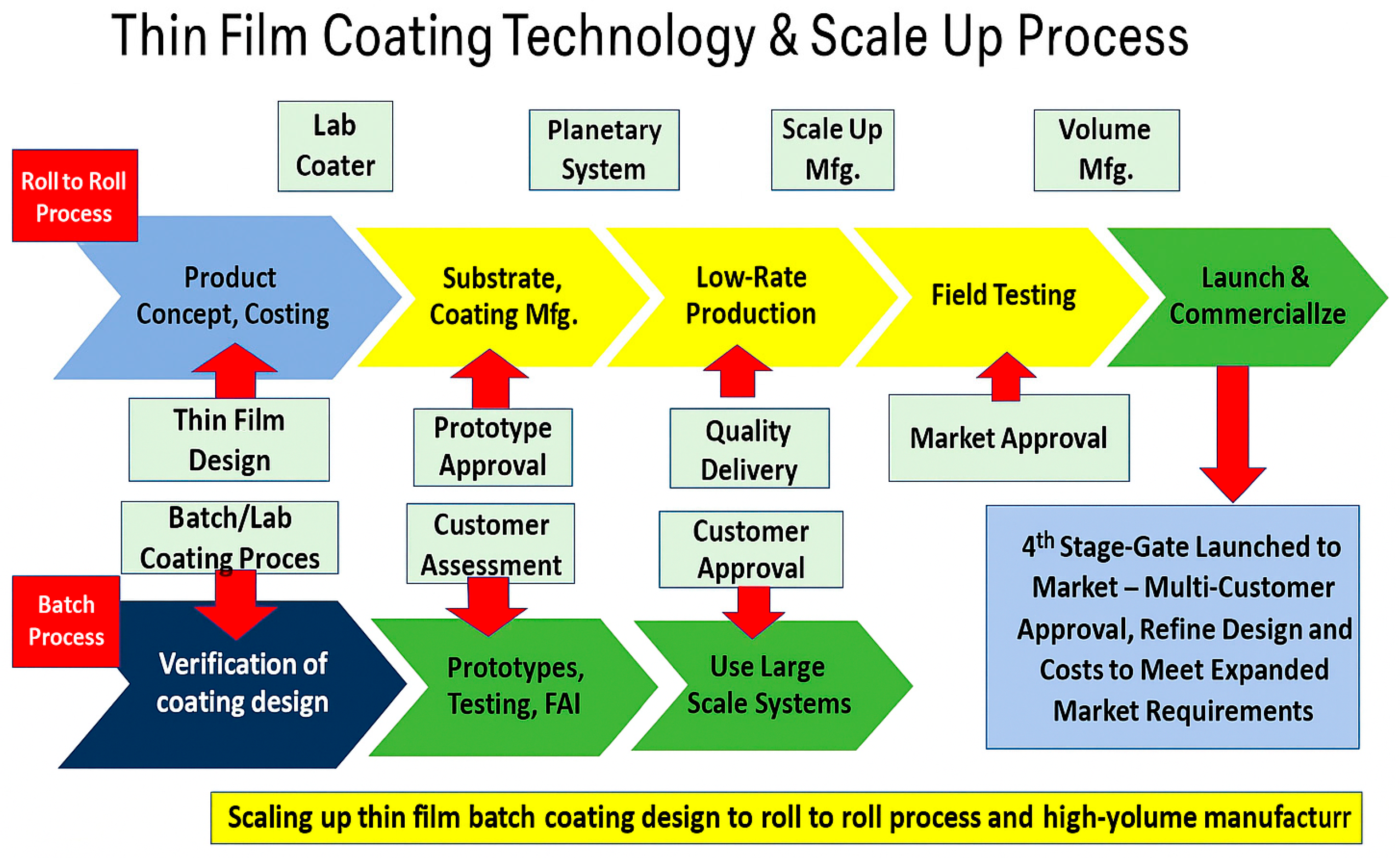
8.1. Scalability and Cost of Coating Techniques
8.2. Long-Term Durability and Maintenance
8.3. Environmental and Regulatory Considerations
9. Future Directions
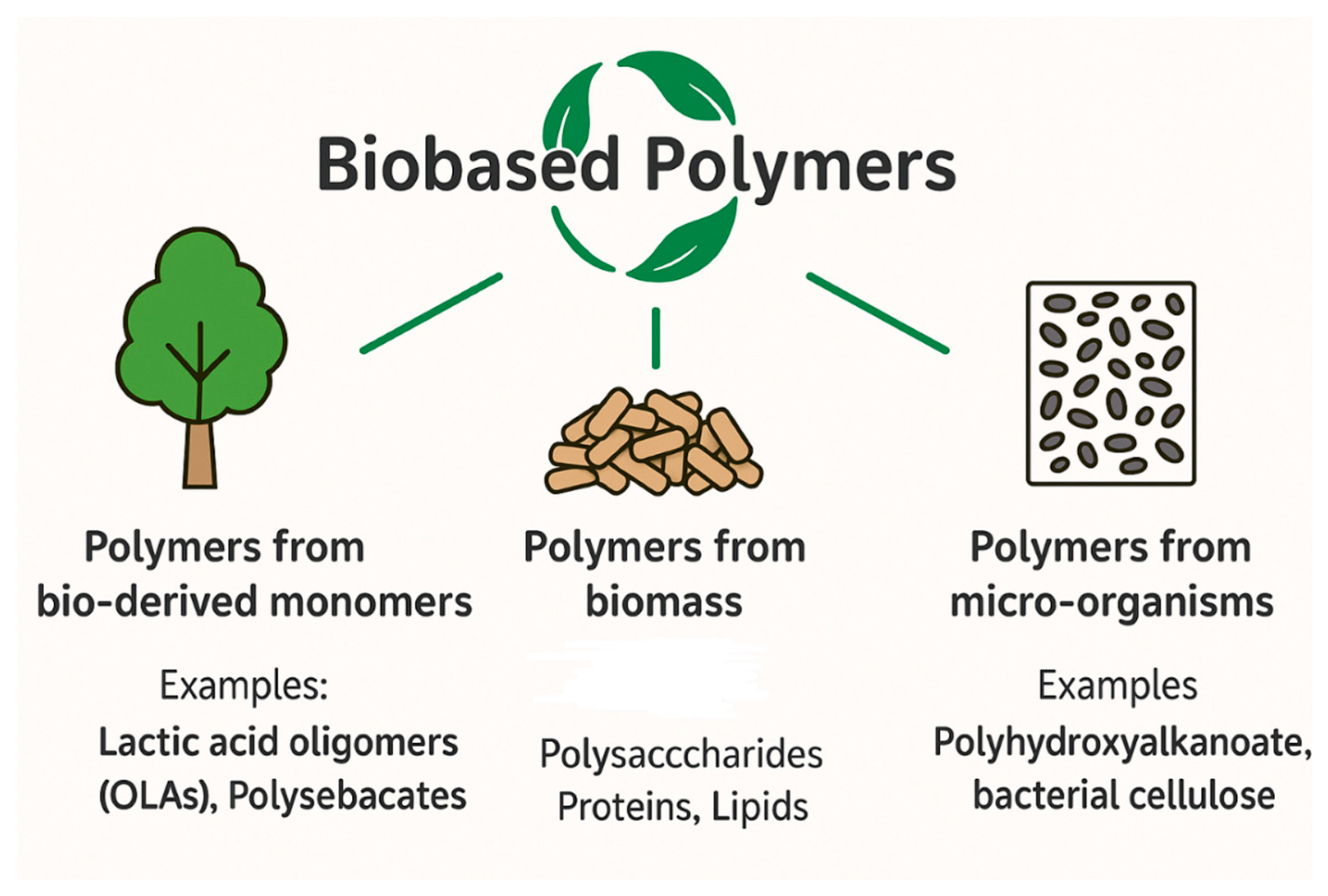
9.1. Development of Sustainable Coating Materials
9.2. Exploration of Coatings for Emerging Applications
10. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Firoozi, A.; Oyejobi, D.; Avudaiappan, S.; Flores, E. Enhanced durability and environmental sustainability in marine infrastructure: Innovations in anti-corrosive coating technologies. Results Eng. 2025, 26, 105144. [Google Scholar] [CrossRef]
- Fan, S.; Mi, B.; Wang, J.; Liu, P.; Ma, X.; Chen, T.; Li, W. Research progress on the preparation of irradiation-resistant coating based on PVD technology. J. Mater. Res. Technol. 2024, 32, 4069–4091. [Google Scholar] [CrossRef]
- Vakili, M.; Koutník, P.; Kohout, J.; Gholami, Z. Analysis, Assessment, and Mitigation of Stress Corrosion Cracking in Austenitic Stainless Steels in the Oil and Gas Sector: A Review. Surfaces 2024, 7, 589–642. [Google Scholar] [CrossRef]
- Lazorenko, G.; Kasprzhitskii, A.; Nazdracheva, T. Anti-corrosion coatings for protection of steel railway structures exposed to atmospheric environments: A review. Constr. Build. Mater. 2021, 288, 123115. [Google Scholar] [CrossRef]
- Bender, R.; Féron, D.; Mills, D.; Ritter, S.; Bäßler, R.; Bettge, D.; De Graeve, I.; Dugstad, A.; Grassini, S.; Hack, T.; et al. Corrosion challenges towards a sustainable society. Mater. Corros. 2022, 73, 1730–1751. [Google Scholar] [CrossRef]
- The History of Ship Deck Coatings-JMS. Available online: https://jms.uk/advice/the-history-of-ship-deck-coatings/?utm_source=chatgpt.com (accessed on 13 July 2025).
- Guo, L.; He, W.; Chen, W.; Xue, Z.; He, J.; Guo, Y.; Wu, Y.; Gao, L.; Li, D.; Zhang, Z.; et al. Progress on high-temperature protective coatings for aero-engines. Surf. Sci. Technol. 2023, 1, 1–39. [Google Scholar] [CrossRef]
- Haubner, R.; Lessiak, M.; Pitonak, R.; Köpf, A.; Weissenbacher, R. Evolution of conventional hard coatings for its use on cutting tools. Int. J. Refract. Met. Hard Mater. 2017, 62, 210–218. [Google Scholar] [CrossRef]
- Bao, W.; Deng, Z.; Zhang, S.; Ji, Z.; Zhang, H. Next-Generation Composite Coating System: Nanocoating. Front. Mater. 2019, 6, 72. [Google Scholar] [CrossRef]
- Mondal, K.; Nuñez, L.; Downey, C.M.; van Rooyen, I.J. Recent advances in the thermal barrier coatings for extreme environments. Mater. Sci. Energy Technol. 2021, 4, 208–210. [Google Scholar] [CrossRef]
- Wang, W.; Sanchez-Fischer, A.; Hawkins, T. New Ultra-Durable Anti-Corrosion Coating System for Steel Infrastructure in Extreme Environments. In Proceedings of the CONFERENCE 2023, Denver, CO, USA, 19–23 March 2023; pp. 1–7. [Google Scholar] [CrossRef]
- Cui, G.; Bi, Z.; Wang, S.; Liu, J.; Xing, X.; Li, Z.; Wang, B. A comprehensive review on smart anti-corrosive coatings. Prog. Org. Coatings 2020, 148, 105821. [Google Scholar] [CrossRef]
- Feng, X.; Wang, H.; Liu, X.; Wang, C.; Cui, H.; Song, Q.; Huang, K.; Li, N.; Jiang, X. Effect of Al content on wear and corrosion resistance of Ni-based alloy coatings by laser cladding. Surf. Coat. Technol. 2021, 412, 126976. [Google Scholar] [CrossRef]
- Huang, B.; Zhang, C.; Zhang, G.; Liao, H. Wear and corrosion resistant performance of thermal-sprayed Fe-based amorphous coatings: A review. Surf. Coat. Technol. 2019, 377, 124896. [Google Scholar] [CrossRef]
- Fan, Q. High-Temperature Alloys in Aerospace Applications and Future Innovations. Highlights Sci. Eng. Technol. 2025, 125, 296–302. [Google Scholar] [CrossRef]
- Ndumia, J.N.; Kang, M.; Gbenontin, B.V.; Lin, J.; Nyambura, S.M. A Review on the Wear, Corrosion and High-Temperature Resistant Properties of Wire Arc-Sprayed Fe-Based Coatings. Nanomaterials 2021, 11, 2527. [Google Scholar] [CrossRef] [PubMed]
- Hebbale, A.M.; Ramesh, M.; Petru, J.; Chandramouli, T.; Srinath, M.; Shetty, R.K. A microstructural study and high-temperature oxidation behaviour of plasma sprayed NiCrAlY based composite coatings. Results Eng. 2025, 25, 103926. [Google Scholar] [CrossRef]
- Mévrel, R. State of the art on high-temperature corrosion-resistant coatings. Mater. Sci. Eng. A 1989, 120–121, 13–24. [Google Scholar] [CrossRef]
- Mao, D.; Xu, Y.; Dong, L.; Wu, J.; Zhao, M.; Li, D. Optimization of the Oxidation Behavior and Mechanical Properties by Designing the TiB2/ZrO2 Multilayers. Coatings 2019, 9, 600. [Google Scholar] [CrossRef]
- Tong, Y.; Zhang, T.; Zhang, S. Influence of oxides on the formation of self-lubricating layer and anti-wear performance during sliding. Tribol. Int. 2022, 179, 108188. [Google Scholar] [CrossRef]
- Rokosz, K. Surface Engineering of Metals and Alloys. Metals 2022, 12, 542. [Google Scholar] [CrossRef]
- Ijiri, M.; Okada, N.; Kanetou, S.; Yamamoto, M.; Nakagawa, D.; Tanaka, K.; Yoshimura, T. Thermal Stress Relaxation and High-Temperature Corrosion of Cr-Mo Steel Processed Using Multifunction Cavitation. Materials 2018, 11, 2291. [Google Scholar] [CrossRef]
- Sobha Jayakrishnan, D. Electrodeposition: The versatile technique for nanomaterials. In Corrosion Protection and Control Using Nanomaterials; Saji, V.S., Cook, R., Eds.; Woodhead Publishing Limited: Sawston, UK, 2012; pp. 86–125. ISBN 9781845699499. [Google Scholar]
- Mischler, S. Triboelectrochemical techniques and interpretation methods in tribocorrosion: A comparative evaluation. Tribol. Int. 2008, 41, 573–583. [Google Scholar] [CrossRef]
- Zhao, X.; Huang, W.; Li, G.; Feng, Y.; Zhang, J. Effect of CO2/H2S and Applied Stress on Corrosion Behavior of 15Cr Tubing in Oil Field Environment. Metals 2020, 10, 409. [Google Scholar] [CrossRef]
- Yeh, C.-P.; Tsai, K.-C.; Huang, J.-Y. Influence of Chloride Concentration on Stress Corrosion Cracking and Crevice Corrosion of Austenitic Stainless Steel in Saline Environments. Materials 2020, 13, 5640. [Google Scholar] [CrossRef]
- Al Madan, A.; Hussein, A.; Akhtar, S.S. A review on internal corrosion of pipelines in the oil and gas industry due to hydrogen sulfide and the role of coatings as a solution. Corros. Rev. 2025, 43, 189–208. [Google Scholar] [CrossRef]
- Syrek-Gerstenkorn, B.; Paul, S.; Davenport, A.J. Sacrificial Thermally Sprayed Aluminium Coatings for Marine Environments: A Review. Coatings 2020, 10, 267. [Google Scholar] [CrossRef]
- Pourhashem, S.; Saba, F.; Duan, J.; Rashidi, A.; Guan, F.; Nezhad, E.G.; Hou, B. Polymer/Inorganic nanocomposite coatings with superior corrosion protection performance: A review. J. Ind. Eng. Chem. 2020, 88, 29–57. [Google Scholar] [CrossRef]
- Tiwari, A.; Seman, S.; Singh, G.; Jayaganthan, R. Nanocrystalline Cermet Coatings for Erosion–Corrosion Protection. Coatings 2019, 9, 400. [Google Scholar] [CrossRef]
- Jose, S.A.; Kasar, A.K.; Menezes, P.L. Cold spray deposition of cermets: Insights into bonding mechanism and critical parameters. Int. J. Adv. Manuf. Technol. 2024, 133, 1–23. [Google Scholar] [CrossRef]
- Wang, L.; Lin, Y.; Zeng, Z.; Liu, W.; Xue, Q.; Hu, L.; Zhang, J. Electrochemical corrosion behavior of nanocrystalline Co coatings explained by higher grain boundary density. Electrochimica Acta 2007, 52, 4342–4350. [Google Scholar] [CrossRef]
- Ralston, K.D.; Birbilis, N. Effect of Grain Size on Corrosion: A Review. Corrosion 2010, 66, 075005. [Google Scholar] [CrossRef]
- Ralston, K.; Fabijanic, D.; Birbilis, N. Effect of grain size on corrosion of high purity aluminium. Electrochimica Acta 2011, 56, 1729–1736. [Google Scholar] [CrossRef]
- Bertuccioli, C.; Garzoni, A.; Martini, C.; Morri, A.; Rondelli, G. Plasma Electrolytic Oxidation (PEO) Layers from Silicate/Phosphate Baths on Ti-6Al-4V for Biomedical Components: Influence of Deposition Conditions and Surface Finishing on Dry Sliding Behaviour. Coatings 2019, 9, 614. [Google Scholar] [CrossRef]
- Yao, W.; Tian, C.; Teng, Y.; Diao, F.; Du, X.; Gu, P.; Zhou, W. Development of deep-sea mining and its environmental impacts: A review. Front. Mar. Sci. 2025, 12, 1598584. [Google Scholar] [CrossRef]
- Mishakin, V.V.; Verichev, S.N.; Razov, E.N. Investigation of the influence of high hydrostatic pressure on the abrasive wear of hard-alloy materials. J. Frict. Wear 2017, 38, 286–291. [Google Scholar] [CrossRef]
- Qian, H.; Ma, L.; Zhang, D.; Li, Z.; Huang, L.; Lou, Y.; Du, C. Microbiologically influenced corrosion of 304 stainless steel by halophilic archaea Natronorubrum tibetense. J. Mater. Sci. Technol. 2020, 46, 12–20. [Google Scholar] [CrossRef]
- Ding, K.; Guo, W.; Qiu, R.; Hou, J.; Fan, L.; Xu, L. Corrosion Behavior of Q235 Steel Exposed in Deepwater of South China Sea. J. Mater. Eng. Perform. 2018, 27, 4489–4496. [Google Scholar] [CrossRef]
- Wani, I.N.; Aggarwal, K.; Bishnoi, S.; Shukla, P.K.; Harursampath, D.; Garg, A. Materials used in space shuttle: Evolution, challenges, and future prospects—An overview. Compos. Part B Eng. 2025, 303, 112540. [Google Scholar] [CrossRef]
- Giolli, C.; Scrivani, A.; Rizzi, G.; Borgioli, F.; Bolelli, G.; Lusvarghi, L. Failure Mechanism for Thermal Fatigue of Thermal Barrier Coating Systems. J. Therm. Spray Technol. 2009, 18, 223–230. [Google Scholar] [CrossRef]
- Tao, Q.; Wang, Y.; Zheng, Y. Fatigue Behaviour and Life Prediction of YSZ Thermal Barrier Coatings at Elevated Temperature under Cyclic Loads. Coatings 2024, 14, 960. [Google Scholar] [CrossRef]
- Altaf, S.F.; Rahman, A.; Wani, M. Nanostructured thermal barrier coatings: Enhancing performance for high-temperature applications: A comprehensive review. Surfaces Interfaces 2025, 72, 107033. [Google Scholar] [CrossRef]
- Sukumaran, A.K.; Orikasa, K.; Rengifo, S.; Renfro, M.; Scott, W.; Gray, A.; Garino, G.; Hernandez, A.F.; Nisar, A.; Mazurkivich, M.; et al. Wear and neutron shielding resilience of titanium-hexagonal boron nitride coatings against extreme lunar radiation and thermal cycles. Surf. Coat. Technol. 2024, 492, 131185. [Google Scholar] [CrossRef]
- Shi, B.; Wu, Y.; Liu, Y.; Wang, L.; Gao, J.; Hei, H.; Zheng, K.; Yu, S. A review on diamond-like carbon-based films for space tribology. Mater. Sci. Technol. 2022, 38, 1151–1167. [Google Scholar] [CrossRef]
- Zhou, M.; Tong, R.; Zhang, T.; Liu, G. Application of MoS2 in the space environment: A review. Front. Mech. Eng. 2023, 18, 39. [Google Scholar] [CrossRef]
- Ferrick, M.; Mulherin, N.; Haehnel, R.; Coutermarsh, B.; Durell, G.; Tantillo, T.; Curtis, L.; Clair, T.S.; Weiser, E.; Cano, R.; et al. Evaluation of ice release coatings at cryogenic temperature for the space shuttle. Cold Reg. Sci. Technol. 2008, 52, 224–243. [Google Scholar] [CrossRef]
- Wyatt, B.C.; Nemani, S.K.; Hilmas, G.E.; Opila, E.J.; Anasori, B. Ultra-high temperature ceramics for extreme environments. Nat. Rev. Mater. 2023, 9, 773–789. [Google Scholar] [CrossRef]
- Ward, T.Z.; Wilkerson, R.P.; Musicó, B.L.; Foley, A.; Brahlek, M.; Weber, W.J.; E Sickafus, K.; Mazza, A.R. High entropy ceramics for applications in extreme environments. J. Physics Mater. 2024, 7, 021001. [Google Scholar] [CrossRef]
- Grilli, M.; Bellezze, T.; Gamsjäger, E.; Rinaldi, A.; Novak, P.; Balos, S.; Piticescu, R.; Ruello, M. Solutions for Critical Raw Materials under Extreme Conditions: A Review. Materials 2017, 10, 285. [Google Scholar] [CrossRef]
- Jithesh, K.; Arivarasu, M. Comparative Studies on the Hot Corrosion Behavior of Air Plasma Spray and High Velocity Oxygen Fuel Coated Co-Based L605 Superalloys in a Gas Turbine Environment. Int. J. Miner. Metall. Mater. 2020, 27, 649–659. [Google Scholar] [CrossRef]
- Dixit, S.; Rodriguez, S.; Jones, M.R.; Buzby, P.; Dixit, R.; Argibay, N.; DelRio, F.W.; Lim, H.H.; Fleming, D. Refractory High-Entropy Alloy Coatings for High-Temperature Aerospace and Energy Applications. J. Therm. Spray Technol. 2022, 31, 1021–1031. [Google Scholar] [CrossRef]
- Kumar, M.; Kant, S.; Kumar, S. Corrosion behavior of wire arc sprayed Ni-based coatings in extreme environment. Mater. Res. Express 2019, 6, 106427. [Google Scholar] [CrossRef]
- Zhang, F.; Ju, P.; Pan, M.; Zhang, D.; Huang, Y.; Li, G.; Li, X. Self-healing mechanisms in smart protective coatings: A review. Corros. Sci. 2018, 144, 74–88. [Google Scholar] [CrossRef]
- Johnson, S.; Gasch, M.J.; Squire, T.; Lawson, J.; Stackpoole, M.M.; Gusman, M.I. Ultra High Temperature Ce-ramics: Issues and Prospects. In Proceedings of the Seventh International Conference on High Temperature Ceramic Materials and Composites, Bayreuth, Germany, 20–22 September 2010; pp. 819–831. [Google Scholar]
- Xiang, H.; Xing, Y.; Dai, F.-Z.; Wang, H.; Su, L.; Miao, L.; Zhang, G.; Wang, Y.; Qi, X.; Yao, L.; et al. High-entropy ceramics: Present status, challenges, and a look forward. J. Adv. Ceram. 2021, 10, 385–441. [Google Scholar] [CrossRef]
- Prameela, S.E.; Pollock, T.M.; Raabe, D.; Meyers, M.A.; Aitkaliyeva, A.; Chintersingh, K.-L.; Cordero, Z.C.; Graham-Brady, L. Materials for extreme environments. Nat. Rev. Mater. 2022, 8, 81–88. [Google Scholar] [CrossRef]
- Schmitt-Thomas, K.; Dietl, U. Thermal barrier coatings with improved oxidation resistance. Surf. Coat. Technol. 1994, 68–69, 113–115. [Google Scholar] [CrossRef]
- Kim, J.; Pyeon, J.; Kim, B.-G.; Khadaa, T.; Choi, H.; Zhe, L.; Dube, T.; Zhang, J.; Yang, B.-I.; Jung, Y.-G.; et al. Oxidation Behavior of NiCoCrAlY Coatings Deposited by Vacuum Plasma Spraying and High-Velocity Oxygen Fuel Processes. Coatings 2023, 13, 319. [Google Scholar] [CrossRef]
- Ju, J.; Yu, H.; Zhao, Y.; Yang, T.; Xiao, B.; Peng, P.; Wang, R.; Wang, H.; Zeng, X.; Wang, J.; et al. Understanding the oxidation behaviors of a Ni-Co-based superalloy at elevated temperatures through multiscale characterization. Corros. Sci. 2023, 227, 111800. [Google Scholar] [CrossRef]
- Xiong, W.; Guo, A.X.; Zhan, S.; Liu, C.-T.; Cao, S.C. Refractory high-entropy alloys: A focused review of preparation methods and properties. J. Mater. Sci. Technol. 2022, 142, 196–215. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, M.; Handa, A. Erosion corrosion behaviour and mechanical properties of wire arc sprayed Ni-Cr and Ni-Al coating on boiler steels in a real boiler environment. Mater. High Temp. 2020, 37, 370–384. [Google Scholar] [CrossRef]
- Mahesh, R.; Jayaganthan, R.; Prakash, S. A study on hot corrosion behaviour of Ni–5Al coatings on Ni- and Fe-based superalloys in an aggressive environment at 900°C. J. Alloys Compd. 2008, 460, 220–231. [Google Scholar] [CrossRef]
- Parit, M.; Saha, P.; Davis, V.A.; Jiang, Z. Transparent and Homogenous Cellulose Nanocrystal/Lignin UV-Protection Films. ACS Omega 2018, 3, 10679–10691. [Google Scholar] [CrossRef]
- Sun, J.; Li, W.; Li, N.; Zhan, Y.; Tian, L.; Wang, Y. Effect of surface modified nano-SiO2 particles on properties of TO@CA/SR self-healing anti-corrosion composite coating. Prog. Org. Coat. 2022, 164, 106689. [Google Scholar] [CrossRef]
- Plis, E.A.; Engelhart, D.P.; Cooper, R.; Johnston, W.R.; Ferguson, D.; Hoffmann, R. Review of Radiation-Induced Effects in Polyimide. Appl. Sci. 2019, 9, 1999. [Google Scholar] [CrossRef]
- Chen, Y.; Ni, H.; Park, J.-J.; Lv, S. A Review of the Preparation, Modification, and Applications of Polyetheretherketone Coating. Coatings 2024, 14, 1451. [Google Scholar] [CrossRef]
- Barra, G.; Guadagno, L.; Raimondo, M.; Santonicola, M.G.; Toto, E.; Ciprioti, S.V. A Comprehensive Review on the Thermal Stability Assessment of Polymers and Composites for Aeronautics and Space Applications. Polymers 2023, 15, 3786. [Google Scholar] [CrossRef] [PubMed]
- Naveen, R.; Kumar, M.; Ramesh, M.; Abinaya, R. Prasath An investigation on effect of ultraviolet (UV) rays on mechanical properties of epoxy laminates. Mater. Today Proc. 2023. [Google Scholar] [CrossRef]
- Ielo, I.; Giacobello, F.; Sfameni, S.; Rando, G.; Galletta, M.; Trovato, V.; Rosace, G.; Plutino, M.R. Nanostructured Surface Finishing and Coatings: Functional Properties and Applications. Materials 2021, 14, 2733. [Google Scholar] [CrossRef] [PubMed]
- Farooq, S.A.; Raina, A.; Mohan, S.; Singh, R.A.; Jayalakshmi, S.; Haq, M.I.U. Nanostructured Coatings: Review on Processing Techniques, Corrosion Behaviour and Tribological Performance. Nanomaterials 2022, 12, 1323. [Google Scholar] [CrossRef]
- Gu, C.; Lian, J.; He, J.; Jiang, Z.; Jiang, Q. High corrosion-resistance nanocrystalline Ni coating on AZ91D magnesium alloy. Surf. Coat. Technol. 2006, 200, 5413–5418. [Google Scholar] [CrossRef]
- El-Hamid, H.K.A.; Gaber, A.A.; Ngida, R.E.A.; Sadek, H.E.H.; Khattab, R.M.; Mandour, H.S. Study of microstructure and corrosion behavior of nano-Al2O3 coating layers on TiO2 substrate via polymeric method and microwave combustion. Sci. Rep. 2024, 14, 18417. [Google Scholar] [CrossRef]
- Veloso, R.C.; Maia, J.; Praça, R.; Souza, A.; Ventura, J.; Ramos, N.M.; Corvacho, H. Impact of SiO2, TiO2 and ZnO nanoparticles incorporation on the thermo-optical properties of dark-coloured façade coatings. J. Build. Eng. 2024, 84, 108517. [Google Scholar] [CrossRef]
- Han, X.; Wei, P.; Zhao, Y.; Wang, Z.; Li, C.; Wu, X.; Zhang, H. Enhanced Pitting Corrosion Resistance of Nanostructured AISI 304 Stainless Steel via Pipe Inner Surface Grinding Treatment. Nanomaterials 2023, 13, 318. [Google Scholar] [CrossRef]
- Lin, J.; Dahan, I. Nanostructured chromium coatings with enhanced mechanical properties and corrosion resistance. Surf. Coat. Technol. 2015, 265, 154–159. [Google Scholar] [CrossRef]
- Kim, J.-H.; Lee, M.-H. A Study on Cavitation Erosion and Corrosion Behavior of Al-, Zn-, Cu-, and Fe-Based Coatings Prepared by Arc Spraying. J. Therm. Spray Technol. 2010, 19, 1224–1230. [Google Scholar] [CrossRef]
- Rezaee, S.; Kadivar, E.; el Moctar, O. Molecular dynamics simulations of a nanobubble’s collapse-induced erosion on nickel boundary and porous nickel foam boundary. J. Mol. Liq. 2024, 397, 124029. [Google Scholar] [CrossRef]
- Kadivar, E.; Rezaee, S.; Löschner, U.; el Moctar, O. Effects of Materials and Riblets on Erosion Mitigation Induced by Multiple Collapses of Cavitation Bubbles. Appl. Sci. 2024, 14, 6452. [Google Scholar] [CrossRef]
- Rezaee, S.; Kadivar, E.; el Moctar, O. The role of sawtooth-shaped nano riblets on nanobubble dynamics and collapse-induced erosion near solid boundary. J. Mol. Liq. 2024, 405, 124947. [Google Scholar] [CrossRef]
- Leonov, A.A.; Denisova, Y.A.; Denisov, V.V.; Syrtanov, M.S.; Shmakov, A.N.; Savostikov, V.M.; Teresov, A.D. Structure and Properties of CrN/TiN Multi-Layer Coatings Obtained by Vacuum-Arc Plasma-Assisted Deposition Method. Coatings 2023, 13, 351. [Google Scholar] [CrossRef]
- Liu, J.; Mei, H.; Hua, J.; Wang, J.; Wang, Y.; Yi, G.; Deng, X. High-Temperature Oxidation and Wear Resistance of TiAlSiN/AlCrN Multilayer Coatings Prepared by Multi-Arc Ion Plating. Nanomaterials 2025, 15, 503. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, L.; Xu, Y. Structure, mechanical, and thermal properties of Ti1−xAlxN/CrAlN (x = 0.48, 0.58, and 0.66) multilayered coatings. J. Am. Ceram. Soc. 2017, 101, 845–855. [Google Scholar] [CrossRef]
- Vereschaka, A.; Milovich, F.; Andreev, N.; Seleznev, A.; Alexandrov, I.; Muranov, A.; Mikhailov, M.; Tatarkanov, A. Comparison of properties of ZrHf-(Zr,Hf)N-(Zr,Hf,Cr,Mo,Al)N and Ti-TiN-(Ti,Cr,Al)N nanostructured multilayer coatings and cutting properties of tools with these coatings during turning of nickel alloy. J. Manuf. Process. 2023, 88, 184–201. [Google Scholar] [CrossRef]
- George, S.M. Atomic Layer Deposition: An Overview. Chem. Rev. 2009, 110, 111–131. [Google Scholar] [CrossRef]
- Greene, J.E. Review Article: Tracing the recorded history of thin-film sputter deposition: From the 1800s to 2017. J. Vac. Sci. Technol. A 2017, 35, 05C204. [Google Scholar] [CrossRef]
- Lemons, J.E. Ceramics: Past, present, and future. Bone 1996, 19, S121–S128. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, D.E.; Ryan, C.J.; DeSalle, C.M.; Stepanoff, S.P.; Aronson, B.I.; Boring, Z.M.; Reiss, J.A.; Albert, P.E.; Nicastro, J.K.; Fjeldsted, A.P. A comprehensive review of modern engineered ceramics coatings for optimised resistance to wear and corrosion. Adv. Appl. Ceram. 2023, 122, 81–100. [Google Scholar] [CrossRef]
- Janssen, R.; Scheppokat, S.; Claussen, N. Tailor-made ceramic-based components—Advantages by reactive processing and advanced shaping techniques. J. Eur. Ceram. Soc. 2008, 28, 1369–1379. [Google Scholar] [CrossRef]
- Grilli, M.L.; Valerini, D.; Slobozeanu, A.E.; Postolnyi, B.O.; Balos, S.; Rizzo, A.; Piticescu, R.R. Critical Raw Materials Saving by Protective Coatings under Extreme Conditions: A Review of Last Trends in Alloys and Coatings for Aerospace Engine Applications. Materials 2021, 14, 1656. [Google Scholar] [CrossRef]
- Bird, R.K.; Wallace, T.A.; Sankaran, S.N. Development of Protective Coatings for High-Temperature Metallic Materials. J. Spacecr. Rocket. 2004, 41, 213–220. [Google Scholar] [CrossRef]
- Piticescu, R.R.; Urbina, M.; Rinaldi, A.; Cuesta-Lopez, S.; Sobetkii, A. Development of Novel Material Systems and Coatings for Extreme Environments: A Brief Overview. JOM 2018, 71, 683–690. [Google Scholar] [CrossRef]
- Ekeocha, J.; Ellingford, C.; Pan, M.; Wemyss, A.M.; Bowen, C.; Wan, C. Challenges and Opportunities of Self-Healing Polymers and Devices for Extreme and Hostile Environments. Adv. Mater. 2021, 33, 2008052. [Google Scholar] [CrossRef]
- Orlov, M.V.; O Badeghaish, W. Advanced Non-Metallic Coatings and Composite Materials for O & G Industry. J. Phys. Conf. Ser. 2021, 1990, 012001. [Google Scholar] [CrossRef]
- Kumar, K.; Jain, A.; Siddiquee, A.N. Special Issue: Multi-Functional Nanostructured Sustainable Coatings. Coatings 2022, 12, 1987. [Google Scholar] [CrossRef]
- Birks, N.; Meier, G.H.; Pettit, F.S. Introduction to the High Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: Cambridge, UK, 2006; ISBN 978-0-521-48042-0. [Google Scholar]
- Oksa, M.; Turunen, E.; Suhonen, T.; Varis, T.; Hannula, S.-P. Optimization and Characterization of High Velocity Oxy-fuel Sprayed Coatings: Techniques, Materials, and Applications. Coatings 2011, 1, 17–52. [Google Scholar] [CrossRef]
- Jose, S.A.; John, M.; Menezes, P.L. Cermet Systems: Synthesis, Properties, and Applications. Ceramics 2022, 5, 210–236. [Google Scholar] [CrossRef]
- Mittal, G.; Paul, S. Suspension and Solution Precursor Plasma and HVOF Spray: A Review. J. Therm. Spray Technol. 2022, 31, 1443–1475. [Google Scholar] [CrossRef]
- Liu, Y.; Fischer, T.E.; Dent, A. Comparison of HVOF and plasma-sprayed alumina/titania coatings—Microstructure, mechanical properties and abrasion behavior. Surf. Coat. Technol. 2003, 167, 68–76. [Google Scholar] [CrossRef]
- Anusha, K.; Routara, B.C.; Guha, S. A Review on High-Velocity Oxy-Fuel (HVOF) Coating Technique. J. Inst. Eng. India Ser. D 2023, 104, 831–848. [Google Scholar] [CrossRef]
- Sharma, R.K.; Das, R.K.; Kumar, S.R. Microstructure, mechanical and tribological properties of high velocity oxy fuel thermal spray coating: A review. Mater. Werkst. 2023, 54, 90–97. [Google Scholar] [CrossRef]
- Rana, N.; Mahapatra, M.M.; Jayaganthan, R.; Prakash, S. Deposition of nanocrystalline coatings by modified LVOF thermal spray method. J. Alloys Compd. 2014, 615, 779–783. [Google Scholar] [CrossRef]
- Jose, S.A.; Kasar, A.K.; Stanford, M.; Menezes, P.L. Unlocking Tribological Performance of Silver-Infused Cu-Al2O3 Self-Lubricating Cermet. J. Tribol. 2025, 147, 1–20. [Google Scholar] [CrossRef]
- Chang, J.; Zhai, H.; Hu, Z.; Li, J. Ultra-thin metal composites for electromagnetic interference shielding. Compos. Part B Eng. 2022, 246, 110269. [Google Scholar] [CrossRef]
- Assadi, H.; Kreye, H.; Gärtner, F.; Klassen, T. Cold Spraying–A Materials Perspective. Acta Mater. 2016, 116, 382–407. [Google Scholar] [CrossRef]
- Karthikeyan, J. 4-The Advantages and Disadvantages of the Cold Spray Coating Process. In The Cold Spray Materials Deposition Process; Champagne, V.K., Ed.; Woodhead Publishing Series in Metals and Surface Engineering; Woodhead Publishing: Cambridge, UK, 2007; pp. 62–71. ISBN 978-1-84569-181-3. [Google Scholar]
- Ozdemir, O.C.; Widener, C.A.; Helfritch, D.; Delfanian, F. Estimating the Effect of Helium and Nitrogen Mixing on Deposition Efficiency in Cold Spray. J. Therm. Spray Technol. 2016, 25, 660–671. [Google Scholar] [CrossRef]
- Nagarajan, S.; Carter, M.; Curtis, T.; Crawford, G. Cold spray-friction stir hybrid additive manufacturing of 316L SS: Feasibility, microstructure, and mechanical performance. Mater. Des. 2025, 255, 114164. [Google Scholar] [CrossRef]
- Li, W.; Cao, C.; Wang, G.; Wang, F.; Xu, Y.; Yang, X. ‘Cold spray +’ as a new hybrid additive manufacturing technology: A literature review. Sci. Technol. Weld. Join. 2019, 24, 420–445. [Google Scholar] [CrossRef]
- Schalk, N.; Tkadletz, M.; Mitterer, C. Hard coatings for cutting applications: Physical vs. chemical vapor deposition and future challenges for the coatings community. Surf. Coat. Technol. 2022, 429, 127949. [Google Scholar] [CrossRef]
- Gudmundsson, J.T.; Anders, A.; von Keudell, A. Foundations of physical vapor deposition with plasma assistance. Plasma Sources Sci. Technol. 2022, 31, 083001. [Google Scholar] [CrossRef]
- Liu, F.; Li, P.; An, H.; Peng, P.; McLean, B.; Ding, F. Achievements and Challenges of Graphene Chemical Vapor Deposition Growth. Adv. Funct. Mater. 2022, 32, 2203191. [Google Scholar] [CrossRef]
- Hassanin, H.; Sheikholeslami, G.; Sareh, P.; Ishaq, R.B. Microadditive Manufacturing Technologies of 3D Microelectromechanical Systems. Adv. Eng. Mater. 2021, 23, 2100422. [Google Scholar] [CrossRef]
- Kim, J.; Kim, H.; Han, G.H.; Hong, S.; Park, J.; Bang, J.; Kim, S.Y.; Ahn, S.H. Electrodeposition: An efficient method to fabricate self-supported electrodes for electrochemical energy conversion systems. Exploration 2022, 2, 20210077. [Google Scholar] [CrossRef]
- Miao, M.; Duan, H.; Luo, J.; Wang, X. Recent progress and prospect of electrodeposition-type catalysts in carbon dioxide reduction utilizations. Mater. Adv. 2022, 3, 6968–6987. [Google Scholar] [CrossRef]
- Schwarzacher, W. Electrodeposition: A Technology for the Future. Electrochem. Soc. Interface 2006, 15, 32–33. [Google Scholar] [CrossRef]
- Cheng, X.; Liu, Y.; Liu, O.; Lu, Y.; Liao, Z.; Hadzhieva, Z.; Chen, L.; Leeuwenburgh, S.G.; Boccaccini, A.R.; Yang, F. Electrophoretic deposition of coatings for local delivery of therapeutic agents. Prog. Mater. Sci. 2023, 136, 101111. [Google Scholar] [CrossRef]
- Ali, M.; Elsherif, M.; Salih, A.E.; Ul-Hamid, A.; Hussein, M.; Park, S.; Yetisen, A.K.; Butt, H. Surface modification and cytotoxicity of Mg-based bio-alloys: An overview of recent advances. J. Alloys Compd. 2020, 825, 154140. [Google Scholar] [CrossRef]
- Wang, X.; Camacho, R.A.P.; Xu, X.; Wang, Y.; Qiang, Y.; Kungl, H.; Eichel, R.-A.; Zhang, Y.; Lu, L. Aerosol deposition technology and its applications in batteries. Nano Mater. Sci. 2024, 6, 24–37. [Google Scholar] [CrossRef]
- Cold Spray and Aerosol Deposition Enable High Purity Coating with Unique Properties-ASM International. Available online: https://www.asminternational.org/results/-/journal_content/56/10192/40877484/NEWS/?srsltid=AfmBOoqln33VWcgHbpikqFa9A8237ORiITkLYSEJeTE1q_GbKCmcAkEy (accessed on 15 July 2025).
- Hossain, S.; Taheri, H. In Situ Process Monitoring for Additive Manufacturing Through Acoustic Techniques. J. Mater. Eng. Perform. 2020, 29, 6249–6262. [Google Scholar] [CrossRef]
- In-Situ Monitoring for Additive Manufacturing: Opportunities and Challenges|Quality Magazine. Available online: https://www.qualitymag.com/articles/97540-in-situ-monitoring-for-additive-manufacturing-opportunities-and-challenges (accessed on 1 July 2025).
- Interim, In Situ Additive Manufacturing Inspection|T2 Portal. Available online: https://technology.nasa.gov/patent/MFS-TOPS-70 (accessed on 1 July 2025).
- Ulaeto, S.B.; Ravi, R.P.; Udoh, I.I.; Mathew, G.M.; Rajan, T.P.D. Polymer-Based Coating for Steel Protection, Highlighting Metal–Organic Framework as Functional Actives: A Review. Corros. Mater. Degrad. 2023, 4, 284–316. [Google Scholar] [CrossRef]
- Martusciello, M.; Hervieu, C.; Di Fonzo, D.; Lanfranchi, A.; Lova, P.; Comoretto, D. Dip-Coating Fabrication of All-Polymer Multilayer Photonic Crystals through 3D Printer Conversion. ACS Appl. Polym. Mater. 2025, 7, 4779–4786. [Google Scholar] [CrossRef] [PubMed]
- Melentiev, R.; Yu, N.; Lubineau, G. Polymer metallization via cold spray additive manufacturing: A review of process control, coating qualities, and prospective applications. Addit. Manuf. 2021, 48, 102459. [Google Scholar] [CrossRef]
- Jiang, Y.; Minett, M.; Hazen, E.; Wang, W.; Alvarez, C.; Griffin, J.; Jiang, N.; Chen, W. New Insights into Spin Coating of Polymer Thin Films in Both Wetting and Nonwetting Regimes. Langmuir 2022, 38, 12702–12710. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Tewari, U.; Bijwe, J. On the abrasive wear of some polyimides and their composites. Tribol. Int. 1991, 24, 247–254. [Google Scholar] [CrossRef]
- Podgornik, B. Adhesive Wear Failures. J. Fail. Anal. Prev. 2022, 22, 113–138. [Google Scholar] [CrossRef]
- Thakare, M.; Wharton, J.; Wood, R.; Menger, C. Effect of abrasive particle size and the influence of microstructure on the wear mechanisms in wear-resistant materials. Wear 2012, 276–277, 16–28. [Google Scholar] [CrossRef]
- Krzyzak, A.; Racinowski, D.; Szczepaniak, R.; Mucha, M.; Kosicka, E. The Impact of Selected Atmospheric Conditions on the Process of Abrasive Wear of CFRP. Materials 2020, 13, 3965. [Google Scholar] [CrossRef] [PubMed]
- Pint, B.A.; Dryepondt, S.; Unocic, K.A. Oxidation of Superalloys in Extreme Environments. In Superalloy 718 and Derivatives; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2010; pp. 859–875. ISBN 978-1-118-49522-3. [Google Scholar]
- Bianco, G.; Nisar, A.; Zhang, C.; Boesl, B.; Agarwal, A. Predicting oxidation damage in ultra high-temperature borides: A machine learning approach. Ceram. Int. 2022, 48, 29763–29769. [Google Scholar] [CrossRef]
- Keddam, M.; Lizee, J.-F.; Pallotta, C.; Takenouti, H. Electrochemical Behavior of Passive Iron in Acid Medium: I. Impedance Approach. J. Electrochem. Soc. 1984, 131, 2016–2024. [Google Scholar] [CrossRef]
- Melchers, R.; Jeffrey, R. The critical involvement of anaerobic bacterial activity in modelling the corrosion behaviour of mild steel in marine environments. Electrochim. Acta 2008, 54, 80–85. [Google Scholar] [CrossRef]
- Ramteke, S.M.; Walczak, M.; De Stefano, M.; Ruggiero, A.; Rosenkranz, A.; Marian, M. 2D materials for Tribo-corrosion and -oxidation protection: A review. Adv. Colloid Interface Sci. 2024, 331, 103243. [Google Scholar] [CrossRef]
- García-León, R.A.; Afanador-García, N.; Guerrero-Gómez, G. A Scientometric Review on Tribocorrosion in Hard Coatings. J. Bio- Tribo-Corros. 2023, 9, 39. [Google Scholar] [CrossRef]
- Masahashi, N.; Mori, Y.; Inoue, H. Biocompatible functional surface of titanium-based implant materials. Int. Mater. Rev. 2025. [Google Scholar] [CrossRef]
- Zhang, Z.; Dandu, R.S.B.; Klu, E.E.; Cai, W. A Review on Tribocorrosion Behavior of Aluminum Alloys: From Fundamental Mechanisms to Alloy Design Strategies. Corros. Mater. Degrad. 2023, 4, 594–622. [Google Scholar] [CrossRef]
- Khalid, M.Y.; Umer, R.; Khan, K.A. Review of recent trends and developments in aluminium 7075 alloy and its metal matrix composites (MMCs) for aircraft applications. Results Eng. 2023, 20, 101372. [Google Scholar] [CrossRef]
- Bonu, V.; Barshilia, H.C. High-Temperature Solid Particle Erosion of Aerospace Components: Its Mitigation Using Advanced Nanostructured Coating Technologies. Coatings 2022, 12, 1979. [Google Scholar] [CrossRef]
- Li, N.; Wang, Q.; Dong, F.; Liu, X.; Han, P.; Han, Y. Research Progress of Coating Preparation on Light Alloys in Aviation Field: A Review. Materials 2022, 15, 8535. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, L.; Fedorov, S.; Ivanova, Y.; Voronina, M.; Fomina, L.; Morozov, A. Improving drill pipe durability by wear-resistant surfacing. Mater. Today Proc. 2020, 30, 398–403. [Google Scholar] [CrossRef]
- Solovyeva, V.A.; Almuhammadi, K.H.; Badeghaish, W.O. Current Downhole Corrosion Control Solutions and Trends in the Oil and Gas Industry: A Review. Materials 2023, 16, 1795. [Google Scholar] [CrossRef] [PubMed]
- Nassar, N.E.A. Corrosion in marine and offshore steel structures: Classification and overview. Int. J. Adv. Eng. Sci. Appl. 2022, 3, 7–11. [Google Scholar] [CrossRef]
- Pei, W.; Pei, X.; Xie, Z.; Wang, J. Research progress of marine anti-corrosion and wear-resistant coating. Tribol. Int. 2024, 198, 109864. [Google Scholar] [CrossRef]
- Pélissier, K.; Thierry, D. Powder and High-Solid Coatings as Anticorrosive Solutions for Marine and Offshore Applications? A Review. Coatings 2020, 10, 916. [Google Scholar] [CrossRef]
- Williamson, E.H.; Gee, M.; Robertson, D.; Watts, J.F.; Whiting, M.J.; Yeomans, J.A. A comparative study of the wear performance of hard coatings for nuclear applications. Wear 2022, 488–489, 204124. [Google Scholar] [CrossRef]
- Fulger, M.; Khumsa-Ang, K.; Šípová, M.; Ducu, C.M.; Sáez-Maderuelo, A. Special Issue: Behavior of Materials (Alloys, Coatings) in Conditions Specific to Gen IV Nuclear Reactors. Coatings 2022, 13, 58. [Google Scholar] [CrossRef]
- Behera, A.; Mallick, P.; Mohapatra, S.S. Chapter 13—Nanocoatings for anticorrosion: An introduction. In Micro and Nano Technologies; Rajendran, S., Nguyen, T.A.N.H., Kakooei, S., Yeganeh, M., Li, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 227–243. ISBN 978-0-12-819359-4. [Google Scholar]
- Mosas, K.K.A.; Chandrasekar, A.R.; Dasan, A.; Pakseresht, A.; Galusek, D. Recent Advancements in Materials and Coatings for Biomedical Implants. Gels 2022, 8, 323. [Google Scholar] [CrossRef]
- Daniyan, A.A.; Akpomejero, T.L.; Ige, O.O.; Olubambi, P.A. Corrosion Prevention of Biomedical Implants: Surface Coating Techniques Perspective. ChemistrySelect 2023, 8, e202300223. [Google Scholar] [CrossRef]
- Thakur, A.; Kumar, A.; Kaya, S.; Marzouki, R.; Zhang, F.; Guo, L. Recent Advancements in Surface Modification, Characterization and Functionalization for Enhancing the Biocompatibility and Corrosion Resistance of Biomedical Implants. Coatings 2022, 12, 1459. [Google Scholar] [CrossRef]
- Udoh, I.I.; Shi, H.; Daniel, E.F.; Li, J.; Gu, S.; Liu, F.; Han, E.-H. Active anticorrosion and self-healing coatings: A review with focus on multi-action smart coating strategies. J. Mater. Sci. Technol. 2022, 116, 224–237. [Google Scholar] [CrossRef]
- Liu, C.; Wu, H.; Qiang, Y.; Zhao, H.; Wang, L. Design of smart protective coatings with autonomous self-healing and early corrosion reporting properties. Corros. Sci. 2021, 184, 109355. [Google Scholar] [CrossRef]
- Sanyal, S.; Park, S.; Chelliah, R.; Yeon, S.-J.; Barathikannan, K.; Vijayalakshmi, S.; Jeong, Y.-J.; Rubab, M.; Oh, D.H. Emerging Trends in Smart Self-Healing Coatings: A Focus on Micro/Nanocontainer Technologies for Enhanced Corrosion Protection. Coatings 2024, 14, 324. [Google Scholar] [CrossRef]
- Koleske, J.V. Paint and Coating Testing Manual: 15th Edition of the Gardner-Sward Handbook, 15th ed.; ASTM International: West Conshohocken, PA, USA, 2012; ISBN 978-0-8031-7017-9. [Google Scholar]
- Joshi, S.; Nylen, P. Advanced Coatings by Thermal Spray Processes. Technologies 2019, 7, 79. [Google Scholar] [CrossRef]
- Noreen, A.; Zia, K.M.; Zuber, M.; Tabasum, S.; Saif, M.J. Recent trends in environmentally friendly water-borne polyurethane coatings: A review. Korean J. Chem. Eng. 2015, 33, 388–400. [Google Scholar] [CrossRef]
- The Global Leader in Nanotechnology-Tesla Nanocoatings. Available online: https://teslanano.com/ (accessed on 1 July 2025).
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef]
- Sheikh, K.A.; Khan, M.M.; Dey, A.; Wani, M.F. Advancements in surface engineering: A comprehensive review of functionally graded coatings for biomaterial implants. Results Surf. Interfaces 2025, 19, 100509. [Google Scholar] [CrossRef]
- Huan, Y.; Wu, K.; Li, C.; Liao, H.; Debliquy, M.; Zhang, C. Micro-nano structured functional coatings deposited by liquid plasma spraying. J. Adv. Ceram. 2020, 9, 517–534. [Google Scholar] [CrossRef]
- Butler, K.T.; Davies, D.W.; Cartwright, H.; Isayev, O.; Walsh, A. Machine learning for molecular and materials science. Nature 2018, 559, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, T.; Ju, W.; Shi, S. Materials discovery and design using machine learning. J. Mater. 2017, 3, 159–177. [Google Scholar] [CrossRef]
- Naveed, M.; Qadir, A. Effect of plasma process parameters on the wear behavior of High-Power Pulsed Magnetron Sputtering deposited Aluminum Titanium Nitride coatings. Open Ceram. 2025, 21, 100746. [Google Scholar] [CrossRef]
- Franz, G. Plasma Enhanced Chemical Vapor Deposition of Organic Polymers. Processes 2021, 9, 980. [Google Scholar] [CrossRef]
- Mehta, A.; Vasudev, H. Advancements in ceramic-coated metals: Enhancing thermal spray coatings for improved performance in aerospace applications using surface treatments. Results Surf. Interfaces 2024, 18, 100387. [Google Scholar] [CrossRef]
- Thin Film Coatings–Scaling New Product Development to Accelerate Commercialization|LinkedIn. Available online: https://www.linkedin.com/pulse/thin-film-coatings-scaling-new-product-development-accelerate-blake/ (accessed on 2 July 2025).
- Voevodin, A.; Zabinski, J. Nanocomposite and nanostructured tribological materials for space applications. Compos. Sci. Technol. 2005, 65, 741–748. [Google Scholar] [CrossRef]
- Davis, J.R. (Ed.) Surface Engineering for Corrosion and Wear Resistance; ASM International: Materials Park, OH, USA, 2001; ISBN 978-1-62708-315-7. [Google Scholar]
- Zhang, C.; Wang, B.; Wang, Y.; Brown, B.; Weber, J.; Liang, Z.; Browning, C.; Kramer, L. Technology roadmap for composite joining and repair. Compos. Part B Eng. 2025, 293, 112132. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, S.; Shi, Q.; Ge, X.; Wang, W. Multilayer Coatings for Tribology: A Mini Review. Nanomaterials 2022, 12, 1388. [Google Scholar] [CrossRef]
- Impact.Nace.Org/Executive-Summary. Aspx. Available online: http://impact.nace.org/executive-summary.aspx (accessed on 1 July 2025).
- All News-ECHA. Available online: https://echa.europa.eu/-/echa-proposes-restrictions-on-chromium-vi-substances-to-protect-health (accessed on 1 July 2025).
- Kendig, M.W.; Buchheit, R.G. Corrosion Inhibition of Aluminum and Aluminum Alloys by Soluble Chromates, Chro-mate Coatings, and Chromate-Free Coatings. Corrosion 2003, 59, 379–400. [Google Scholar] [CrossRef]
- Al-Amiery, A.; Isahak, W.N.R.W.; Al-Azzawi, W.K. Sustainable corrosion Inhibitors: A key step towards environmentally responsible corrosion control. Ain Shams Eng. J. 2024, 15, 102672. [Google Scholar] [CrossRef]
- Hirsch, J. Recent development in aluminium for automotive applications. Trans. Nonferrous Met. Soc. China 2014, 24, 1995–2002. [Google Scholar] [CrossRef]
- ISO/TR 27921:2020(En); Carbon Dioxide Capture, Transportation, and Geological Storage—Cross Cutting Issues—CO2 Stream Composition. ISO: Geneva, Switzerland, 2020. Available online: https://www.iso.org/obp/ui/en/#iso:std:iso:tr:27921:ed-1:v1:en (accessed on 1 July 2025).
- Faccini, M.; Bautista, L.; Soldi, L.; Escobar, A.M.; Altavilla, M.; Calvet, M.; Domènech, A.; Domínguez, E. Environmentally Friendly Anticorrosive Polymeric Coatings. Appl. Sci. 2021, 11, 3446. [Google Scholar] [CrossRef]
- Mohamed, M.G.; Ahmed, N.M.; Samir, M. Sustainable coatings based on core–shell pigments to enhance antibacterial and other beneficial paper properties. J. Coat. Technol. Res. 2023, 20, 1857–1866. [Google Scholar] [CrossRef]
- OECD. Global Plastics Outlook: Policy Scenarios to 2060; OECD: Paris, France, 2022; ISBN 978-92-64-97364-0. [Google Scholar]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan based nanocomposite films and coatings: Emerging antimicrobial food packaging alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Jose, S.A.; Cowan, N.; Davidson, M.; Godina, G.; Smith, I.; Xin, J.; Menezes, P.L. A Comprehensive Review on Cellulose Nanofibers, Nanomaterials, and Composites: Manufacturing, Properties, and Applications. Nanomaterials 2025, 15, 356. [Google Scholar] [CrossRef]
- Mohammed, M.; Jawad, A.J.M.; Mohammed, A.M.; Oleiwi, J.K.; Adam, T.; Osman, A.F.; Dahham, O.S.; Betar, B.O.; Gopinath, S.C.; Jaafar, M. Challenges and advancement in water absorption of natural fiber-reinforced polymer composites. Polym. Test. 2023, 124, 108083. [Google Scholar] [CrossRef]
- Sutthiwanjampa, C.; Hong, S.; Kim, W.J.; Kang, S.H.; Park, H. Hydrophilic Modification Strategies to Enhance the Surface Biocompatibility of Poly(dimethylsiloxane)-Based Biomaterials for Medical Applications. Adv. Mater. Interfaces 2023, 10, 2202333. [Google Scholar] [CrossRef]
- Pellicori, S.F.; Martinez, C.L.; Hausgen, P.; Wilt, D. Development and testing of coatings for orbital space radiation environments. Appl. Opt. 2014, 53, A339–A350. [Google Scholar] [CrossRef]
- Wetegrove, M.; Duarte, M.J.; Taube, K.; Rohloff, M.; Gopalan, H.; Scheu, C.; Dehm, G.; Kruth, A. Preventing Hydrogen Embrittlement: The Role of Barrier Coatings for the Hydrogen Economy. Hydrogen 2023, 4, 307–322. [Google Scholar] [CrossRef]
- Nazir, A.; Gokcekaya, O.; Billah, K.M.M.; Ertugrul, O.; Jiang, J.; Sun, J.; Hussain, S. Multi-material additive manufacturing: A systematic review of design, properties, applications, challenges, and 3D printing of materials and cellular metamaterials. Mater. Des. 2023, 226, 111661. [Google Scholar] [CrossRef]


| Coating Type | Temperature Rating | Corrosion Resistance/Key Benefit | Reference |
|---|---|---|---|
| Ni-20Cr | 900 °C | High resistance in molten salt environments | [53] |
| RHEAs | >1200 °C | Strong mechanical properties; variants for radiation or corrosive use | [52] |
| TBCs | ~1300 °C | Prevent oxygen ingress; ideal for aerospace engine applications | [50] |
| Self-Healing Coatings | ~1200 °C | Maintain integrity and corrosion resistance over long service durations | [54] |
| Coating Type | Hardness (GPa) | Thermal Stability (°C) | Corrosion/Oxidation Resistance | Advantages | References |
|---|---|---|---|---|---|
| Ultra-High-Temperature Ceramics (UHTCs) | 20–30 | 3000–4200 | Excellent | Extremely high melting points, oxidation resistance at ultra-high temps | [55] |
| High-Entropy Ceramics (HECs) | 15–25 | >1600 | Excellent | Tunable properties, compatibility with SiC electronics | [56] |
| Thermal Barrier Coatings (TBCs) | 10–15 | ~1500 | Good | Thermal insulation, reduced thermal conductivity | [57,58] |
| Superalloy Coatings | 5–10 | ~1100–1200 | Very Good | High creep strength, good aerospace performance | [59,60] |
| Refractory High-Entropy Alloys (RHEAs) | 12–18 | >1200 | Very Good to Excellent | High strength at elevated temperatures, radiation resistance | [61] |
| Ni-Based Alloy Coatings | 6–10 | ~1000–1100 | Good | Strong adhesion, cost-effective, suitable for boiler tubes | [62,63] |
| Feature | Nanostructured Coatings | Multilayered Coatings |
|---|---|---|
| Structure | Nano-scale grains/particles | Stacked layers with distinct functions |
| Thickness | 1–100 nm | Nanometers to micrometers |
| Material Type | Ceramics, metals, hybrid composites | Ceramics, polymers, metals, composites |
| Properties | High hardness, toughness, stability | Combined mechanical, thermal, and chemical benefits |
| Applications | Biomedical, aerospace, electronics | Optical barriers, wear and corrosion resistance |
| Deposition methods | Physical vapor deposition (PVD), chemical vapor deposition (CVD), electrochemical deposition | Magnetron sputtering, pulsed laser deposition (PLD), atomic layer deposition (ALD), sol–gel |
| Material type | Advantages | Limitations | Suitable extreme environments/Applications | References |
|---|---|---|---|---|
| Ceramic-Based Coatings | Excellent thermal and oxidation resistance, high hardness, corrosion protection | Brittle, low fracture toughness, often require high-temperature processing | High-temperature environments, chemically aggressive media, and thermal barrier applications | [87,88,89] |
| Metallic and Alloy Coatings | Good ductility, thermal conductivity, electrical conductivity, oxidation resistance (in some alloys) | Lower hardness than ceramics, susceptible to corrosion if unprotected | High-pressure and thermal cycling environments, marine and structural applications | [90,91] |
| Polymer and Composite Coatings | Lightweight, corrosion-resistant, chemically versatile, easy to apply | Low thermal stability, prone to UV degradation over time | Chemical plants, biomedical devices, electronics, and moderate corrosion and abrasion environments | [92,93,94] |
| Nanostructured and Multilayered Coatings | Enhanced hardness, wear, and corrosion resistance, tunable properties | Complex fabrication, scalability issues, and cost | Wear-prone components, aerospace and microelectronics, dynamic or multifunctional environments | [70,95,96] |
| Attribute | Physical Vapor Deposition (PVD) | Chemical Vapor Deposition (CVD) |
|---|---|---|
| Starting Material | Solid (target material) | Gaseous precursors |
| Deposition Mechanism | Physical (evaporation/sputtering) | Chemical reaction at the substrate surface |
| Coating Properties | High hardness, wear and temperature resistance | Precise thickness and composition |
| Typical Applications | Cutting tools, optics, and electronics | Semiconductors, protective coatings |
| Method | Description | Applications | Advantages | Limitations |
|---|---|---|---|---|
| Electrochemical Techniques | Electron transfer-based coating processes | Batteries, sensors, corrosion testing | Broad material range, scalable | Requires conductive substrates |
| Electrodeposition | Ion reduction and metal deposition | Electronics, corrosion protection | Uniform coating, cost-effective | Limited to conductive surfaces |
| Localized Pulsed Electrodeposition (L-PED) | Pulsed micro-deposition using electrolyte meniscus | 3D microelectronics, nano-sensors | Tailored microstructure, high precision | Complex, requires precise control |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antony Jose, S.; Lapierre, Z.; Williams, T.; Hope, C.; Jardin, T.; Rodriguez, R.; Menezes, P.L. Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives. Coatings 2025, 15, 878. https://doi.org/10.3390/coatings15080878
Antony Jose S, Lapierre Z, Williams T, Hope C, Jardin T, Rodriguez R, Menezes PL. Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives. Coatings. 2025; 15(8):878. https://doi.org/10.3390/coatings15080878
Chicago/Turabian StyleAntony Jose, Subin, Zachary Lapierre, Tyler Williams, Colton Hope, Tryon Jardin, Roberto Rodriguez, and Pradeep L. Menezes. 2025. "Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives" Coatings 15, no. 8: 878. https://doi.org/10.3390/coatings15080878
APA StyleAntony Jose, S., Lapierre, Z., Williams, T., Hope, C., Jardin, T., Rodriguez, R., & Menezes, P. L. (2025). Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives. Coatings, 15(8), 878. https://doi.org/10.3390/coatings15080878







