Abstract
A cyclic olefin copolymer (COC) is a thermoplastic polymer endowed with glass-like optical transparency, unique biocompatibility, low autofluorescence, good chemical stability, and excellent water vapor barriers. COC is a promising new material for optics, medical devices, nanotechnology, and microelectronics. The applicability of the COC depends on its modification through different techniques from plasma treatment to lithography. Presently, pulsed laser deposition (PLD) is employed to deposit an aluminum thin film on selected areas of the COC surface. The study of the wettability, morphology, composition, and optical characteristics of both pristine and modified COC has been evaluated by scanning electron and atomic force microscopies, the sessile drop method, and UV/ViS optical spectroscopy. The prospective recycling of the COC deposited by PLD is proposed.
1. Introduction
Although polymer science is a relatively new discipline, the concept of a polymer started to be recognized as one of the great ideas of the 20th century when the pioneer H. Staudinger received the Nobel Prize in 1953. Since then, the use of polymers has been steadily rising in material science, bioscience, and industry.
Plastic became an essential part of life in consumer products and packaging, fundamental science [1], and technology [2]. The extensive use of polymers includes controlled drug delivery, sensors, and medical implants. Polymers can be used in colloidal drug carrier systems [3], revealing the advantage of optimized drug loading and release. They can be used, as with the latest nano-particulate polymeric systems, to cross the blood-brain barrier [4]. Recently, Tong et al., reported on the replacement of organic liquid electrolytes in Lithium-ion batteries (LIBs) with solid-state electrolytes with ionically conductive polymers [5]. This encouraged the design of the new generation of solid-state lithium metal batteries (SSLMBs) exhibiting more benefits than the traditional LIBs such as the removal of volatile components, higher energy density and storage, longer cycle life, and improved safety promising for the production of high-capacity electrode materials.
The unique properties possessed by polymers, such as their lightweight, low-cost, flexibility, optical properties, and specific strength comparable to metals, make them highly valuable in the packaging sector. However, the dominant type of polymers for packaging, due to their physical and mechanical properties, are non-biodegradable, such as polyethylene (PE), polypropylene (PP), ethylene vinyl alcohol, poly(ethylene terephthalate), poly(vinyl chloride) (PVC) [6]. Some of these polymers (i.e., PVC) can never be recycled, while others, such as multilayer packaging material with aluminum foil as the center layer, can.
However, the plastic waste market increases progressively [7] with a negative impact on public health and the environment.
The fermentations in the development of polymers recurred when Kaminsky and Sinn discovered a new class of catalysts, the so-called “metallocene complex” for ethylene polymerization in 1976. Their catalysts, besides several modifications, led to the synthesis of ethylene-α olefin copolymers with a high degree of uniformity in their composition [8].
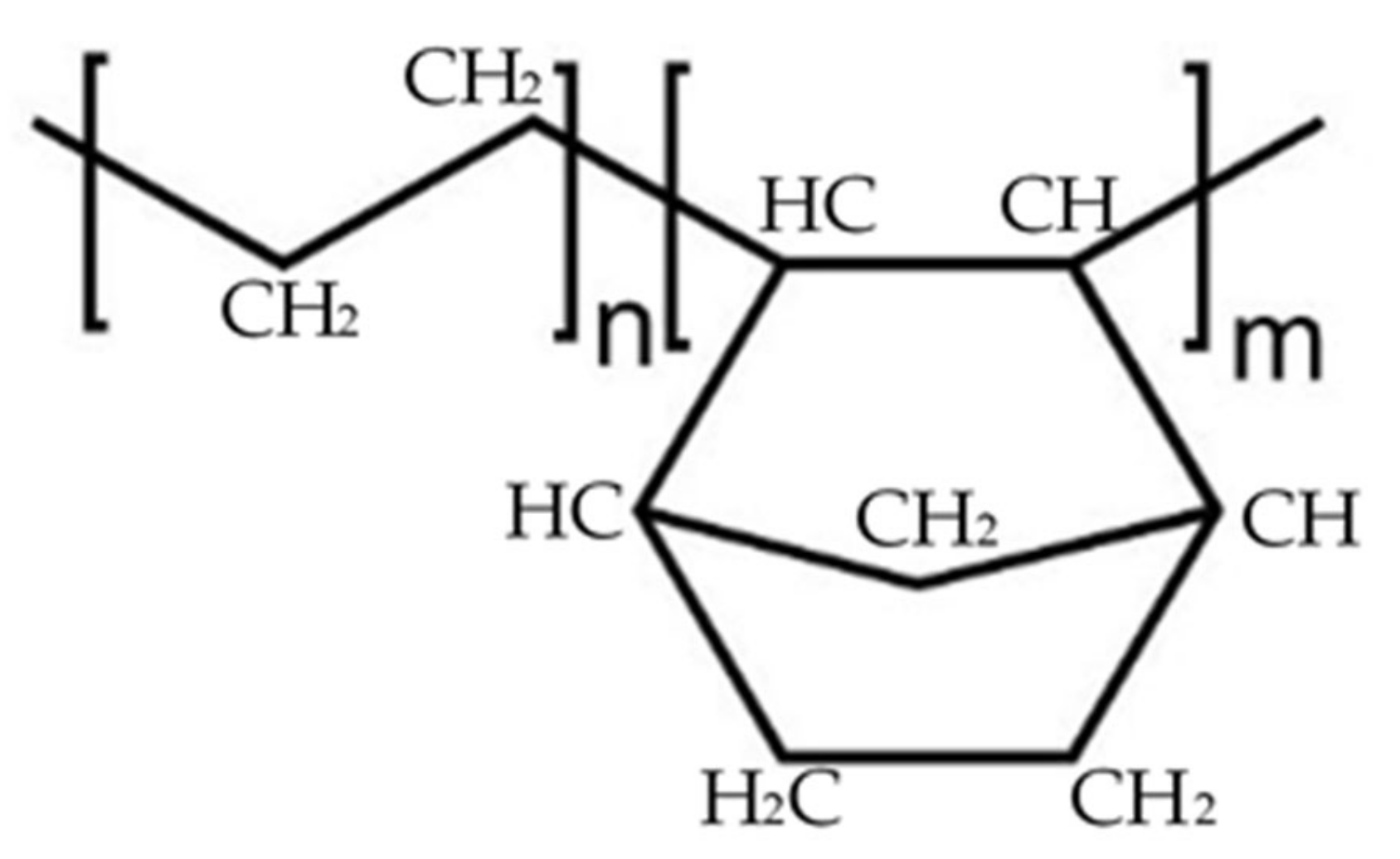
COC is an organic compound containing carbon and hydrogen joined together in straight chains, branched chains, or non-aromatic rings. COC films are prepared by copolymerization of norbornene with ethylene. In Figure 1, the subscripts n and m identify the ethylene and norbornene components, respectively. Plenty of norbornene derivatives exist and by changing the percentage of norbornene, it is possible to adjust the glass transition temperature of the COC.
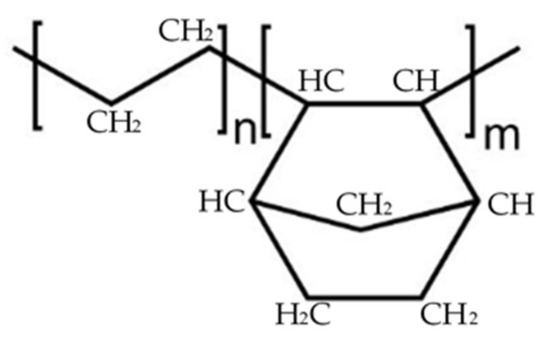
Figure 1.
Structure of the two cycloolefin copolymers.
Norbornene has a rigid structure preventing crystallization and making the COC totally amorphous, improving its stiffness and increasing its glass transition temperature (Tg) proportionally to its inclusion in the chains.
Typically, COC may be mixed with polyethylene (PE) to make monolayer or multilayer films to increase the strength and salability of packaging.
When more than 25% of COC is combined with low-density PE (LDPE) the film stiffness can increase up to ten times with respect to other films. Laminated films of COC exhibit mirror-like properties, reducing the permeability to light and amplifying the moisture barrier property up to twice that of high-density PE and three times that of low-density PE [9].
Further benefits offered by COC with respect to other organic polymers are the comparable transmittance between the visible and the near UV to that of polymethylmethacrylate (PMMA). It is also more accented than PS and PC, while its lower density and less brittle nature are ideal features for replacing glassy optical components [9].
These unique properties heightened the use of COC leading to the development of a research line addressed to the improvement of its original qualities. Recently, also the in vitro biocompatibility of different types of COC [10] has been assessed through an accurate study of their cytotoxicity evaluated according to ISO 10993-5 guidelines [11,12], and of their hemocompatibility exploring the prospective use of COC as implantable biomaterials. The combination of biocompatibility along with the high moisture barrier, clarity, and recyclability are undoubtedly factors promoting the use of COC for packaging.
This work proposes the use of pulsed laser deposition (PLD) as a promising technique for the manufacturing of a thin layer of aluminum on COC in a controlled manner.
The design of aluminum laminated COC film is a requisite to improve the barrier properties of the COC foil to maintain the food quality, medicament inertness, health, and consequently the reduction of food spoiling. Unfortunately, even if the laminated COC films are highly demanding, their recyclability can be difficult.
The laser-based approach is suited for the modification [13] and the transfer/deposition of all types of solid material and compounds, without the large waste of material, providing accurate and precise thickness control and due to the energy of the deposited particles better adherence to the substrate with respect to other deposition techniques.
Although the arrangement for the PLD is simple, its processing is the result of the interplay of several parameters, such as laser wavelength and energy, pulse width, target-substrate distance, background gas composition, and pressure, to name a few. The adjusting of some of these parameters can produce a film with customized thickness, composition, and structure. Presently, an Nd:YAG laser operating in the IR region [14] has been used for PLD processing. Its typical gaussian energy profile enables the intrinsic variation of the fluence; the chosen wavelength can induce thermal effects and support the emission of high density, the large size of emitted particulate, and droplet formation [15].
The COC has been selected also in view of its high thermal conductivity to minimize the damage induced by the droplets phase emitted from the laser plume and deposited on the COC surface. Furthermore, to obtain a more uniformly deposited layer and to minimize the droplet phase in the laser plume, which negatively affects the final properties of the deposited layer, higher linear and angular target-substrate distances have been adopted during the PLD processing and the Al target has been polished to reduce its roughness [16] and obtain a smaller size of deposited particles.
The purpose of this study is to address a slight modification of the COC transparency for a clear identification of the packaged product; the improvement of its reflectivity against external influences; the manufacturing of a deposited layer with an adherence better than obtained with traditional deposition systems; and the characterization of the modified COC foil to be easily recycled in an environmentally friendly manner. A mild deposition of aluminum on the COC surface by PLD has been proven effective for the accomplishment of the mentioned features to upgrade the COC performance in the packaging sector and assist its recycling when it is assembled as laminated.
2. Materials and Methods
2.1. Preparation of COC Film
The formulation mr-IT85-5.0 copolymer supplied by micro resist technology GmbH (Berlin, Germany) [17] has been chosen for the manufacturing of a defined COC film thickness. The concentration of the polymer is claimed to be more appropriate and the solvents are less toxic than the ones usually used.
Silicon wafers have been cleaned using acetone and isopropanol and then dehydrated at 200 °C for 15 min on a hotplate. Polydimethylsiloxane (PDMS) is an elastomer purchased from Dow Corning Corp (Midland, MI, USA). obtained by mixing the monomer and the curing agent in a 10:1 w/w ratio and subsequently degassed for 45–60 min under vacuum in a desiccator. The obtained PDMS solution has been uniformly spread on the previously cleaned wafers by spin coater (SPIN150, SPS-Europe B.V., Hamburg, Germany), according to the indications of the supplier: 400 rpm for 45 s, 800 rpm for 45 s, followed by 1200 rpm for 60 s, 2000 rpm for 60 s and 4000 rpm (ramp 200 rpm/s) for 60 s.
Wafers covered with a PDMS layer have been cured on the hotplate at 95 °C for 30 min and placed inside a plasma etcher (ICP-RIE plasma etcher SI 500, SENTECH Instruments GmbH, Berlin, Germany) for 30 s to activate the PDMS surface with oxygen plasma. The working conditions of the ICP were 50 W of power, 20 cm3/s of oxygen flow, 13 Pa of pressure, and 10 °C of cooling temperature. After this processing, the wafers were ready to be used for thin cyclic olefin copolymer (COC) layer deposition.
The formulation mr-I T85-5.0 copolymer has been spin-coated on PDMS (LabClaster, SÜSS MicroTec, Garching, Germany) at 500 rpm for 10 s and then 3000 rpm (500 rpm/s ramp) and hot baked at 120 °C for 2 min. The final 10 μm COC film has been peeled off and cut into pieces of about 2 cm × 3 cm.
2.2. Laser Processing
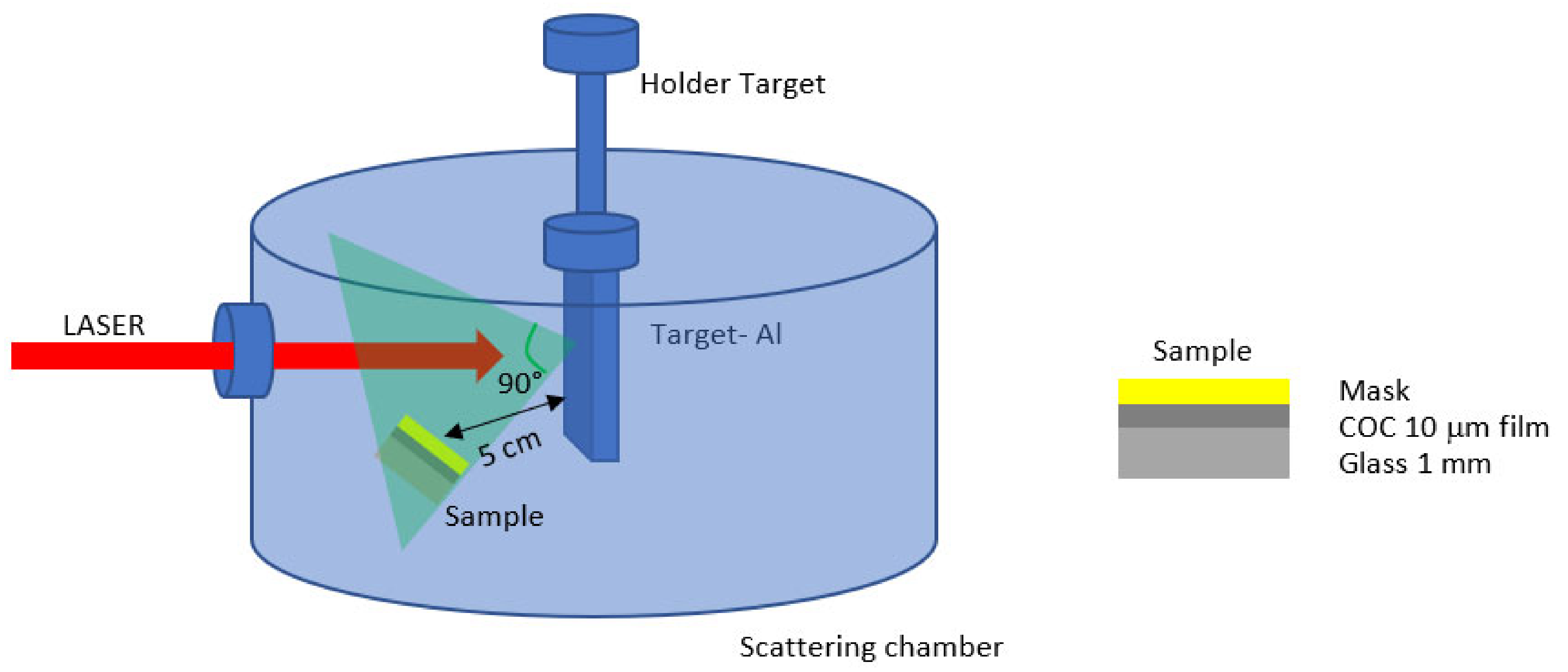
Pulsed laser deposition (PLD) [18] is a well-established technique used for the modification of the surface properties of a material maintaining the bulk attributes. The fast and controlled deposition of a film on a surface material is routinely performed through direct laser irradiation in the repetition rate mode of a solid sample located in a vacuum chamber. At the ablation threshold of the solid sample, a plasma plume with the same stoichiometry as the irradiated sample is generated. The products of the laser-sample interaction, such as electrons, ions, atoms, and clusters, are ejected at high energy and then deposited on the substrates located near the solid sample, as depicted in Figure 2. Nd:YAG laser system supplied by Litron [19], operating at 1064 nm wavelength, 5 ns pulse duration, and 10 Hz repetition rate, generates a beam that has been focused at 1 mm2 spot size on the aluminum (Al) solid sample (99.99% purity) located in a scattering chamber at a vacuum of 3.5 × 10−5 mbar [20]. PLD has been performed at the laser energy of 600 mJ, at a 0° incidence angle from the target surface. The focused laser beam spot on the target was 1 mm in diameter, and 8000 laser shots through a quartz window hit the Al target for about 13 min. The laser fluence was about 60 J/cm2.
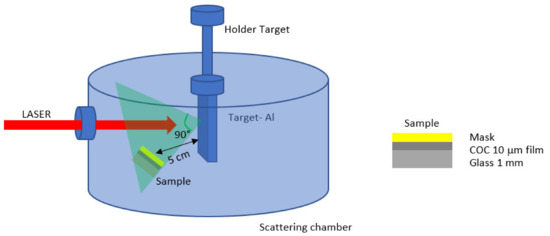
Figure 2.
Schematic illustration of the pulsed laser deposition system.
Substrates consisting of glass covered with 10 μm COC and a superimposed metallic mask 1 mm thick have been placed at 45° with respect to the normal of the target surface and at 5 cm from the main target as shown in Figure 2. This particular configuration has been chosen to obtain a mild deposition layer on the COC foil covered with deposited Al (COC/Al). The energy of aluminum ions generated during the interaction of the laser with the Al target has been detected by a Faraday Cup (FC) [1] fixed at 45° and 1 m distance with respect to the normal of the target surface in the Time-Of-Flight configuration (TOF). This means that the energy of the generated ions has been evaluated knowing the target Faraday Cup distance and recording with fast storage oscilloscope, the time necessary for the emitted ions to reach the detector.
2.3. Optical Characterization (UV-VIS)
The optical properties of the laser-deposited COC foils have been examined by the UV-ViS system based on the use of an AvaSpec-2048 spectrometer (Apeldoorn, The Netherlands) provided with a UB-600 lines/mm grating and working at a wavelength between 310 nm to 650 nm. A dual source, consisting of Halogen/deuterium lights, has been used to illuminate an area of 2 mm2 of the surface in two different configurations. In transmission mode, the lights aim towards the front of the sample at 0°, and a fiber, optically connected to the spectrometer, has been positioned behind the sample. In the reflection configuration, the lights point towards the sample surface at 15° incidence angle from the normal while the optical fiber, connected to the spectrometer, is at –15° incidence angle [21]. Optical analysis has been performed on the pristine and laser-deposited COC. In addition, the deposited foils have been immersed in acetone (C3H6O) for 60 s to test the feasibility of acetone for restoring the native optical property of COC. It is known that COC dissolves in non-polar organic solvents (i.e., hydrocarbons), while it is resistant to polar organic solvents (i.e., acetone), so it is expected that the used solvent removes the deposited layer without affecting the COC substrate.
2.4. Wettability Measurements
The alteration of the wettability of the COC substrates deposited by the PLD has been evaluated by the sessile drop method. A drop of deionized water with a volume of 3 µL has been deposited on pristine and PLD-modified COC foils. The contact angle formed on the horizontal surface of the investigated samples has been displayed by optical microscopy and measured by dedicated software [22]. This angle has been measured in different areas of the sample surface to acquire its average standard deviation value. The wettability on the laser-deposited COC has been estimated also after immersion in acetone (C3H6O) for 60 s.
2.5. Scanning Electron (SEM) and Atomic Force (AFM) Microscopies
The morphological modifications, the produced micro-structure, and roughness over the virgin and the deposited samples have been monitored by scanning electron microscopy (SEM, JSM-5600LV, JEO, Akishima, Japan). An LYRA3 GMU (Tescan, Brno, Czech Republic) at an acceleration voltage of 5 kV and in the secondary-electron mode has been employed to analyze a view field of 15 μm. The energy-dispersive X-ray fluorescence (EDX) coupled to SEM provides the maps collected from the characteristic Kα lines in C (about 277 eV), O (around 525 eV), and Al (1.49 keV) elements on the surface and on the cross-section of the deposited COC/Al sample.
The AFM has been performed using a Dimension ICON (Bruker Corp., Billerica, MA, USA) equipped with the ScanAsyst mode in air. Regions of 10 × 10 μm2 of pristine and deposited COC have been captured at 1 s exposure and processed by NanoScope Analysis software (1.40r1sr3) for the evaluation of the surface average roughness (Ra) and the mean roughness parameter (RMS).
3. Results
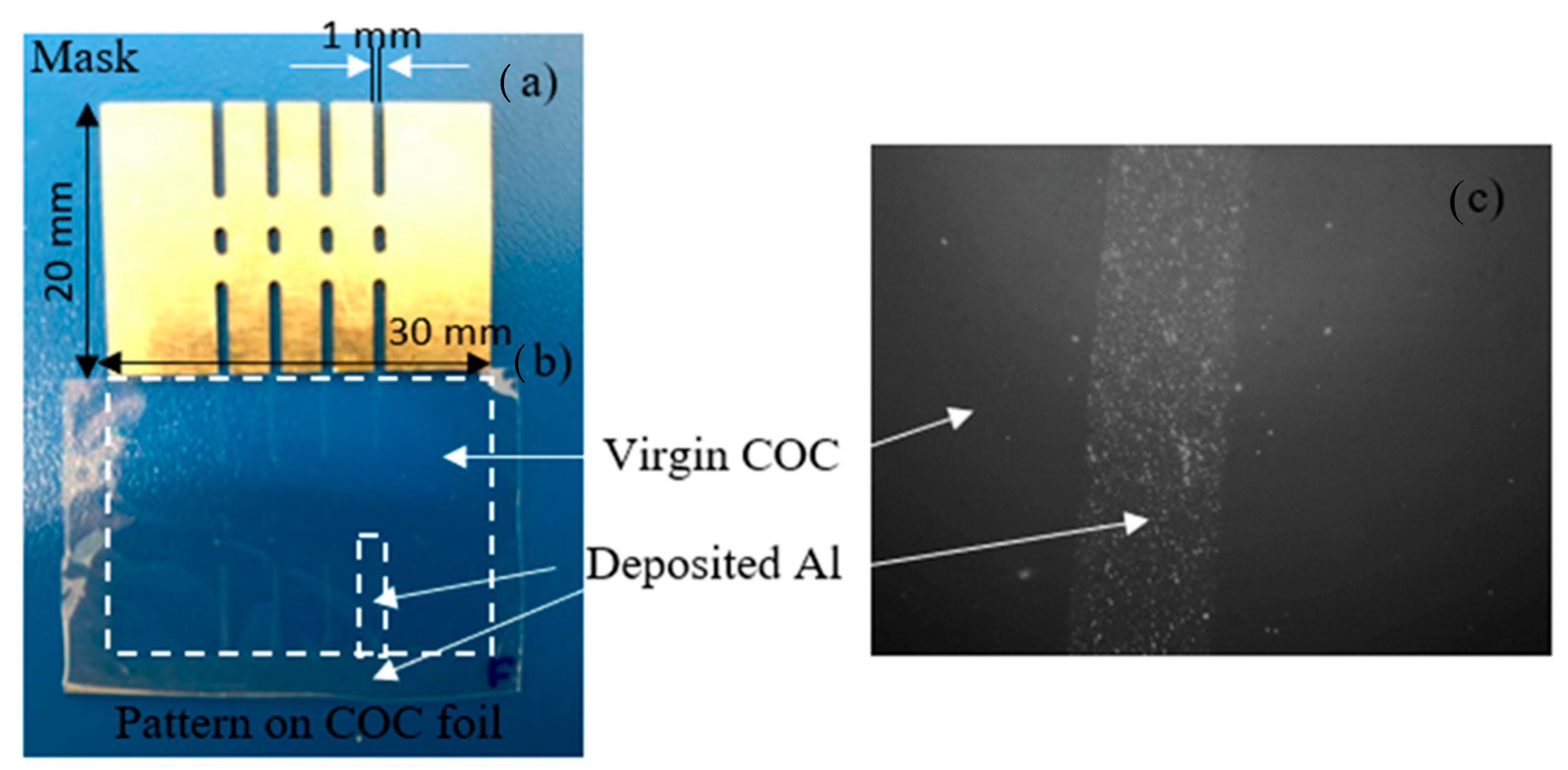
The indirect patterning of Al on COC foil has been obtained using a metallic mask, 1 mm thick, size of 20 mm × 30 mm where four parallel long lines, 1 mm wide, in the design displayed in Figure 3a have been machined. In Figure 3b it is possible to distinguish the unirradiated areas and the deposited Al reproducing the pattern of the mask on the COC foil marked by dashed white lines. Figure 3c displays an optical image 10× magnified pointing out a detail of the pattern realized by laser deposition of Al ions. The reported Al cover film thickness in this SEM image is about 100 nm.
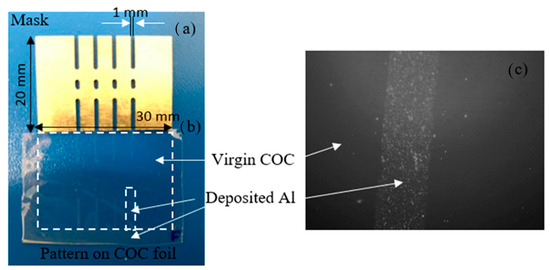
Figure 3.
Picture of the mask (a) used to realize a pattern on COC foil (b) and optical image 4× magnified of a detail of the pattern reproduced on COC (c).
The products of the laser-matter interactions [23] responsible for the deposited pattern formed on COC foil are multi-energy and multi-species ions. Since high-energy ions can be implanted at high depths while low-energy ions can induce deposition on the most superficial layer of the substrate, the ion energy seems to be a key parameter affecting the structural, chemical, and physical modifications in the materials. Typically, a Faraday Cup (FC) is the detector used to monitor the energy of the emitted ions in the Time-Of-Flight (TOF) configuration [1] during the PLD.
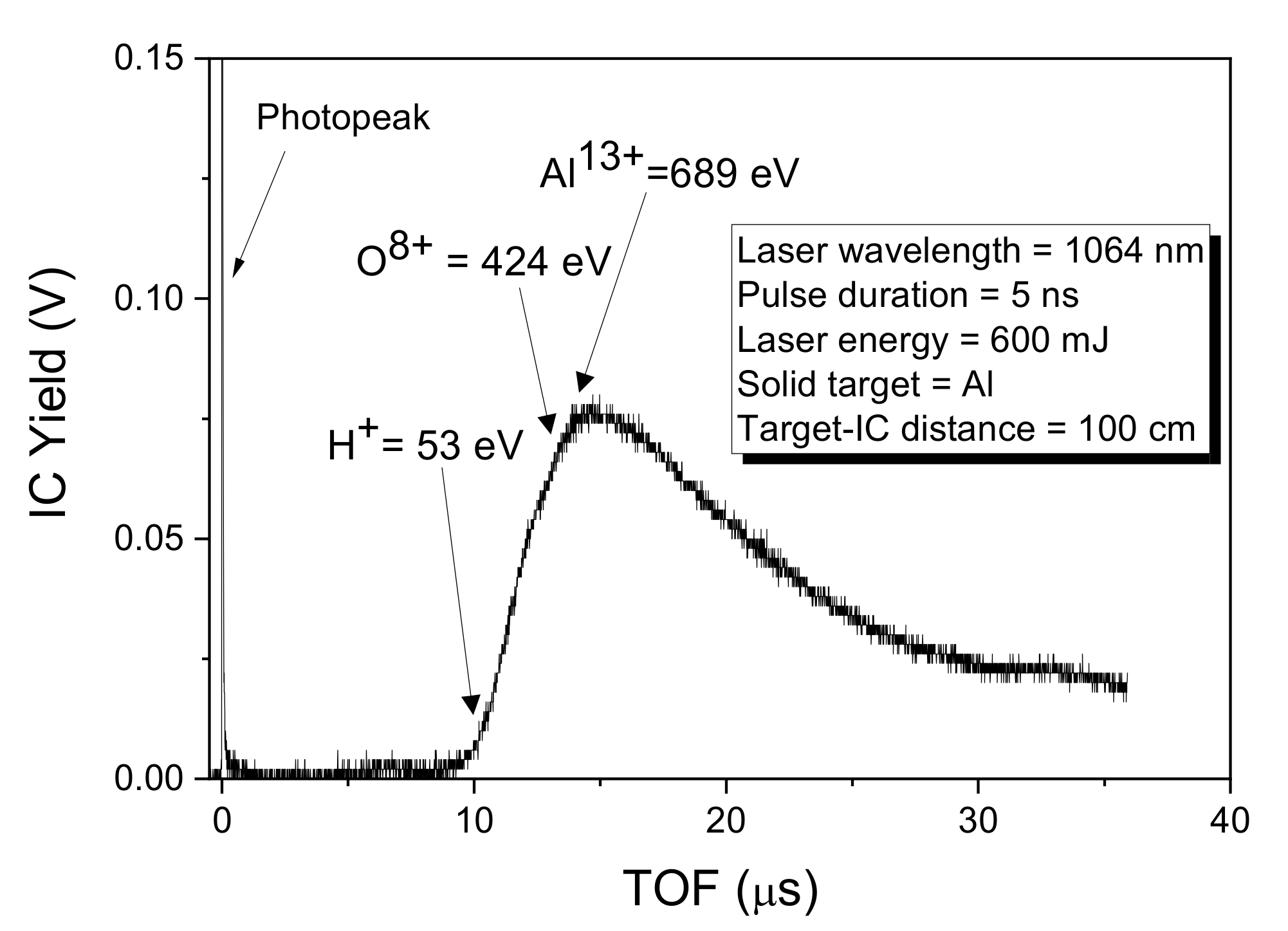
Figure 4 reveals the FC spectrum collected when the solid sample of Al has been irradiated by a laser beam with an energy of 600 m J. The first sharp, fast, and narrow peak labeled photopeak is due to the photoelectric effect. It is assignable to photons coming from the plasma-generating photoelectron on the collector’s metallic surface. Further features which can be observed on the TOF spectrum concern the ion peak with a long tail due to the detection of protons, oxygen, and aluminum ions, in agreement with the literature [23].
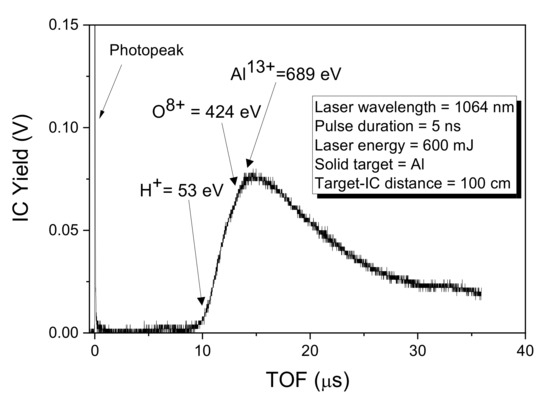
Figure 4.
Spectrum in TOF obtained from a Faraday Cup for a target of Al irradiated by laser.
The intense photopeak is followed by a large, structured peak extending from 9 to about 35 μs.
This structure is due to the convolution of the signal of the fast protons followed by contaminants such as O ions (the aluminum has some oxidized surface layers) and Al ions with different charge states. The maximum energy of the deposited Al ions is about 689 eV while that of the less energetic, present in the peak tail, is about 53 eV, corresponding to the Al13+ and Al1+ ions, respectively.
Within the measured Al ion energy, ranging approximately between 53 eV and 689 eV, PLD induces dominant nuclear-stopping power responsible for the deterioration of the deposited surface, higher roughness, and sputtering effects on the COC surface.
To explore the quality of the deposited pattern obtained by laser deposition of Al ions, AFM and SEM analyses have been performed. Figure 5 reports on both the average surface roughness (Ra) and the mean roughness parameter (RMS) of pristine and deposited COC surfaces. The NanoScope Analysis software provides also the difference between the investigated area of the sample and the rough surface of the same sample, which increases from 5.55% in pristine COC to 6.99% in COC deposited by PLD. This result marks the increase in the surface roughness in agreement with the values of RMS rising from 12.8 nm in pristine COC to 43.7 nm in the deposited COC, respectively. The size of the structures deposited on COC by PLD lower than 21.2 nm is ascribable to the good polishing of the target and the low energy of deposited ions during PLD.

Figure 5.
AFM images of the pristine COC (a) and of the deposited COC (b).
The elemental composition of COC has been investigated before and after the PLD processing by EDX analysis and is reported in Table 1. The pristine COC contains as the main component C at 99.0 atm. % in agreement with its nominal formulation as the EDX cannot detect hydrogen, which should be the second main component. The O concentration of about 11.1% in deposited COC is probably ascribable to the presence of oxygen contaminants on the surface of Al samples used to be deposited on COC.

Table 1.
Atomic composition of pristine and deposited COC.
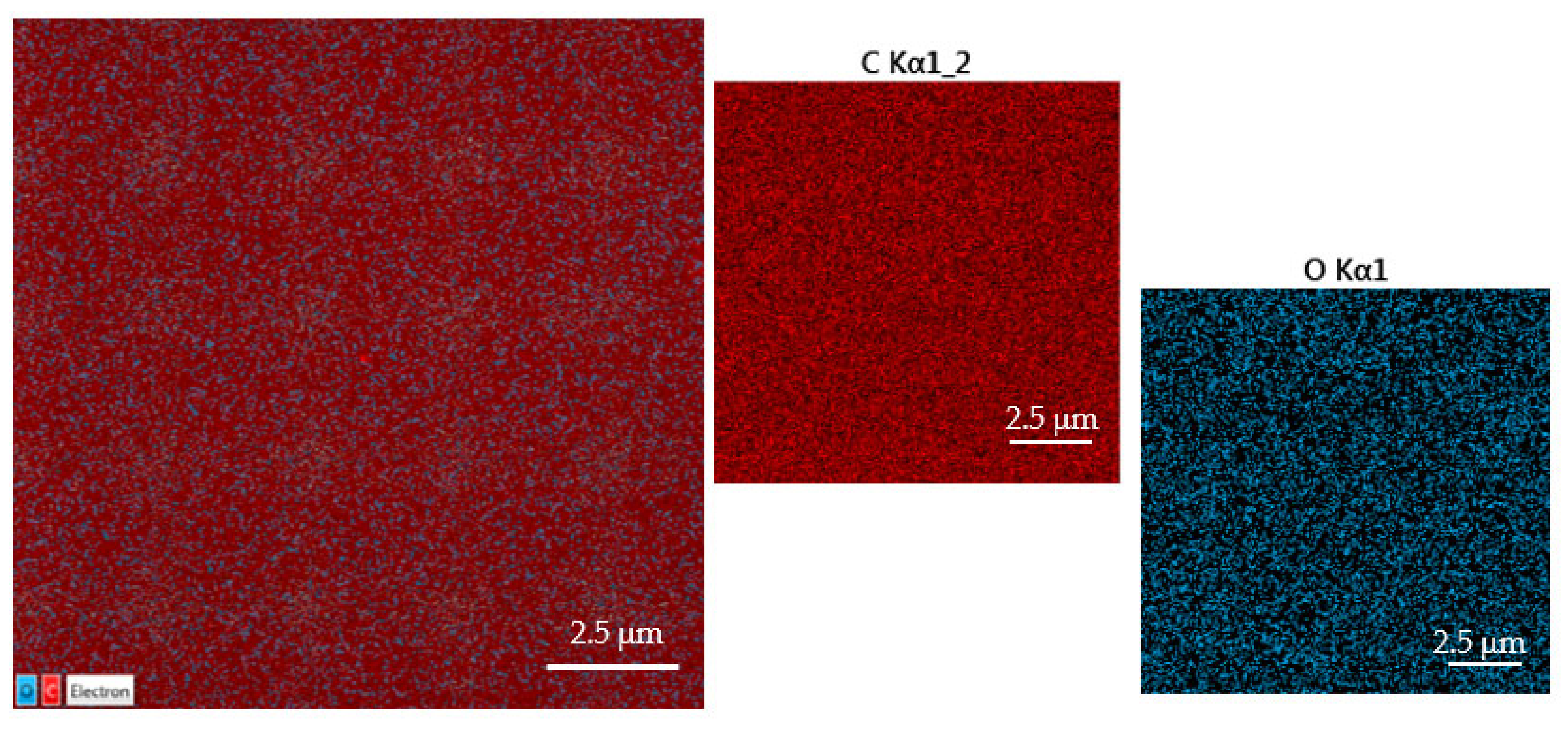
The monitored concentration of Al deposited on COC foil is about 5.6 atm. %. The maps obtained by 5 keV electron X-ray fluorescence illustrate the results listed in Table 1. Figure 6 displays all the maps referring to C and O Kα lines provided by the energy-dispersive X-ray coupled to SEM.

Figure 6.
SEM-EDX images and spectrum of pristine COC.
In Figure 6, the red color uniformly covers all the pristine C/O atomic seems to confirm the absence of heavy elements in COC which is proof of its claimed purity.
Figure 7 shows all the maps relating to C, O, and Al Kα lines provided by the energy-dispersive X-ray coupled to SEM referring to the surface of the COC deposited with Al.

Figure 7.
SEM-EDX images of Al deposited COC on the surface.
In Figure 7, the violet color distributed on the COC sample confirms the uniform deposition of Al during the pulsed laser deposition process. The contribution of the O content can be ascribed to impurities contained in the surface of the Al solid target from which the Al ions/clusters have been ejected during PLD. The big circular structures visible on the surface of the deposited COC are clusters of Al and micrometric debris as validated from Kα Al spectrum obtained from EDX indicating a concentration of 66.7% of aluminum in these circular structures.
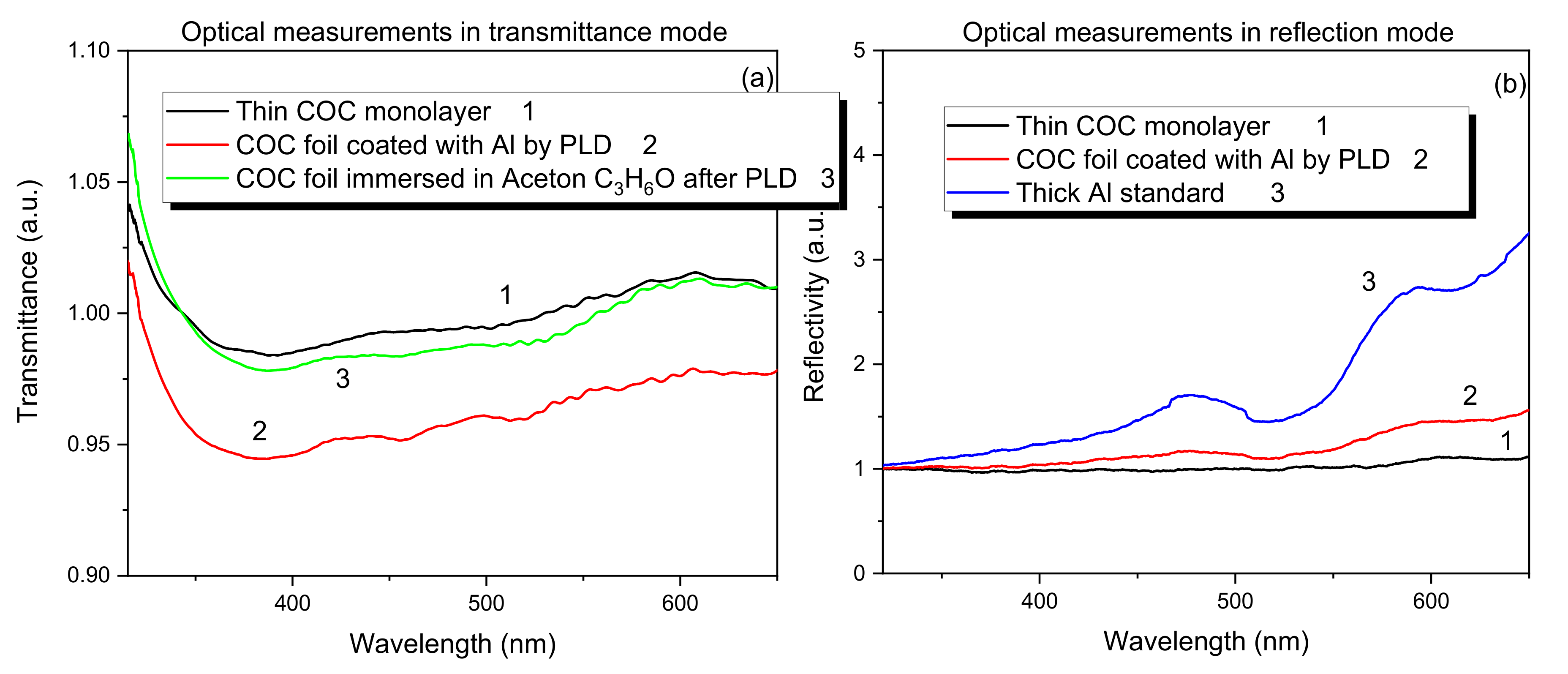
To estimate the clarity of the COC surface after the PLD processing, transmitted and reflective analyses have been performed. The spectra in Figure 8a,b show the optical measurements in transmission (see Figure 8a) and in reflectivity (see Figure 8b) modes, respectively.
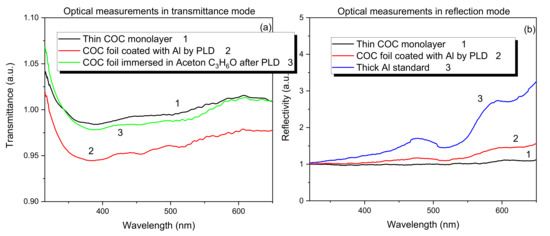
Figure 8.
Transmission (a) and reflectivity (b) for pristine COC and for COC deposited with Al ions.
Figure 8a shows that in the COC film deposited with Al, after the immersion in acetone for 60 s at room temperature, a total removal of Al has been induced as suggested by the optical transmission to the visible light close to the one for the pristine COC.
Figure 8b shows that the optical reflectivity of the visible light increases with the thin Al film coverage of COC, transforming its transparency to opaque and reflecting film in the visible wavelength range.
The alteration of the intensity for both the transmissivity and the reflectivity in the deposited COC is not significantly higher than in the pristine COC due to the low amount of deposited Al on its surface, as confirmed by SEM and AFM analyses.
The wettability of the pristine and deposited COC has been studied by the evaluation of the contact angle formed at the interface between the liquid/solid and the liquid/air interfaces before and after the PLD process. In Figure 9, can be observed the decrease of the contact angle from 106° in pristine COC (see Figure 9a) to 88° in deposited COC (see Figure 9b) and to 98° in deposited COC after immersion in acetone (see Figure 9c). The reduced wettability affecting the deposited COC may be due to the presence of structures with sizes bigger than the 10.5 nm roughness evaluated in pristine COC as suggested by AFM.

Figure 9.
Contact angle for virgin COC (a), for COC deposited with Al ions (b), and for COC deposited and then immersed in acetone (c).
The reduction in the wettability of the COC covered with an Al thin film is expected because the wetting angle in the pure Al surface is about 79.7° [24], while in the thin Al foil, it is correlated to the COC substrate. Results show that the COC hydrophobicity is slightly decreased with the Al thin film coverage inducing slightly higher spreading of the water, but an important benefit offered by the presence of the Al layer is the reduction of the visible radiation transmitted by the thin COC foil as confirmed by optical analysis. This can support a reduction in the deterioration of foods and other organic materials produced by visible light that could be enveloped in it, and simultaneously, keeping the water repellency of the sheet used almost unchanged.
The preliminary comparison of the stiffness in pristine and laser-deposited COC has been estimated by the hardness test, suggesting a slight enhancement of the PLD-deposited COC with respect to the pristine COC.
4. Discussion
In this study, the modifications induced on cyclic olefin copolymer (COC) foil by the pulsed laser deposition technique (PLD) for application in packaging have been examined.
The PLD processing involves the interaction of a laser beam with a solid target forming a plasma from which the generated particles have been ejected on a substrate, where a thin film is obtained. Presently, adequate choices of laser fluence, number of laser pulses, linear and angular distances between the solid target and the substrate, and the background pressure have been crucial parameters to form a thin layer of Al on the COC surface of the order of 100 nm thick. Typically, in PLD processing, minimum laser energy is required to remove the material from the direct laser-irradiated target. If the laser energy is lower than the ablation threshold of the irradiated material, the plasma plume is not formed, and the material ablation cannot take place; if it is slightly higher than the ablation threshold, this generates a low ablation rate and slow deposition rate and, in the case of multi-elements, also generates a preferential element ablation rate. At high laser energy rates, like those selected in this study, the ablation rate increases logarithmically, and the kinetic energy of the species ejected from the target increases, assisting their adhesion on the substrate. The risks of substrate damage and sputtering effects on the formed deposited film [15] have been mitigated by the high thermal conductivity of the native COC substrate.
SEM-EDX analyses show uniformity and purity in the pristine COC whereas, on the surface of the deposited layer, a few droplets with an average size of about 1 μm can be seen. It is well known that the presence of a droplets phase in a coating obtained by PLD, worsens its quality and properties. According to the literature, a decrease in the formation of a condensed phase of droplets with nano or micro sizes in the material ejected from the plasma can be accomplished [16] by minimizing the surface roughness of the primary target to obtain a reduction of the droplet phase in the laser plume [25] or increasing the target-substrate distance since the decrease of the laser spot ellipticity leads to a reduction in the ablated liquid phase. To this aim, presently, the target-substrate distance was about 5 cm and the primary target was polished prior to laser irradiation.
The concentration of 99% for C in the pristine is consistent with the native composition of the used copolymer, taking into account the limitation of the EDX in the detection of H which is the nominal second component. The presence of the oxygen content detected on the deposited film by SEM-EDX is attributable to the gases (i.e., hydrogen, oxygen) desorbed from the surface of the laser-irradiated target during the PLD processing in a vacuum. Therefore, it appears appropriate for the use of a shutter in front of the substrate to be deployed for the first laser pulses to reduce the oxygen content flowing from the plasma plume and deposited on the substrate. The low concentration of Al of about 5.6 atm% is correlated with the laser geometry adopted during the PLD processing. The uniform layer of Al formed on the deposited COC matches the optical analysis carried out in transmission and reflection modes.
The value of the optical transmission decreases as a consequence of the Al layer on the COC surface. However, the very thin film of Al on the COC film supports the reduction of the visible component of the light and does not prevent the optimal identification of the material which could be wrapped in it. Furthermore, the intensity of the reflectivity value rises slightly, reducing the sunlight exposure and avoiding the degradation of imaginary food, organic material, biological samples, and tissues covered with the laser-modified COC.
The hydrophobicity, as an important property of the native COC, should be preserved in packaging applications, because it inhibits the adhesion of liquids with the film surface. The result of our measurements on deposited COC is that the contact angle decreases slightly, leading to a more favorable spreading of the water on the packaging film, which is an unwanted feature because it can be responsible for the spoiling of the food.
The obtained wettability is ascribable to the presence of the thin layer of Al. Indeed, it is closer to the value of typical aluminum foil than to the COC. The turning from strong hydrophobicity to slight hydrophilicity is associated with the increase of the surface roughness stated by AFM images.
Indeed, the structures on the deposited COC surface display sizes of about 21.2 nm as pointed out by AFM.
In the frame of the investigation, to monitor the preservation of sustainability at a low cost, the COC film deposited with Al by PLD has been immersed in acetone, which is biodegradable and not harmful to the environment and human health. Subsequently, the transparency and the wettability of the deposited COC after a bath in acetone have been monitored and from the outcome emerged that after immersion in the polar solvent, the transparency of the COC deposited by PLD is closer to the pristine COC as well as the wettability.
Moreover, to state the promotion of the stiffness in the modified COC, the hardness test has been carried out preliminarily, suggesting a slight increase in the strength of the deposited COC.
Taking into account the significance of the improvement of the stiffness of the COC/Al film, for its prospective reuse and for the safety of the packed product, work is in progress for a detailed quantification.
The functionality of the packaging quality and properties can result in benefits such as the reduction of food wastage and organic materials, the prolongation of the quality and shelf life of the products, as well as the possibility of sustainable and low-cost recyclable laminated packaging.
5. Conclusions
The aim of this work is to characterize the modification of cyclic olefin copolymer (COC) foil after the pulsed laser deposition process and simultaneously to evaluate if the modification preserved its feasibility for the packaging sector application. The native COC is biocompatible and recyclable and meets many of the features demanded in packaging technology: low-cost production, lightweight, strength, reusable, transparency, thermal and chemical resistance, hydrophobicity, and high moisture barrier.
With this aim, the PLD technique has been used to generate a thin layer of Al on the COC surface of the order of 100 nm thick. Optical measurements indicate a slight reduction in transmissivity, enabling clear identification of the packaged product; a low increase in the reflectivity, attributable to the low deposition time and low Al thickness obtained, can reduce the exposure to sunlight, thereby preventing the degradation of the food.
The reduction of the hydrophobicity after indirect laser processing of the COC is minimal and does not transform its hydrophobicity into hydrophilicity, maintaining all the favorable conditions for food packing.
Typically, the contact angle is correlated with the surface roughness, the particle shape, size, and chemical compositions of the layer formed on the substrates as confirmed by SEM and AFM analyses. The PLD processing slightly modified COC wettability, which is connected to the structures deposited on the COC surface and is responsible for the slightly higher spreading of the water on the packaging film.
However, the monitoring of the wettability and the optical analysis after immersing the deposited COC foil in acetone shows good potential for eco-friendly recycling of the wasted Al/COC film.
The work is in progress for the detailed quantification of the improved strength of COC for repetitive reuse and product safety and for the customization of deposited layers with antimicrobial properties by PLD with the advantage of obtaining functionalized packaging in one step, overcoming costing and time-consuming post-processing.
Author Contributions
Conceptualization, M.C.; validation, M.C.; formal analysis, M.C., L.T., P.S. and D.F.; data curation, M.C. and L.T.; writing—original draft preparation, M.C.; writing—review and editing, M.C., L.T., L.S., V.H., A.M., P.M., R.M., J.M., M.S., P.A., D.F. and P.S.; funding acquisition, A.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by GACR, grant number 22-10536S. The research has been realized at the CANAM (Center of Accelerators and Nuclear Analytical Methods) infrastructure LM 2015056. This publication was supported by OP RDE, MEYS, Czech Republic under the project CANAM OP, CZ.02.1.01/0.0/0.0/16_013/0001812. The authors acknowledge the assistance provided by the Research Infrastructure NanoEnviCz (Project No.LM2018124) and the project Pro-NanoEnviCz (Reg. No. CZ.02.1.01/0.0/0.0/16_013/0001821 and CZ.02.1.01/0.0/0.0/18_046/0015586), supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union European Structural and Investments Funds in the frame of the Operational Programme Re-search Development and Education.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data that support the findings of this study are included within the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Torrisi, L.; Cutroneo, M.; Silipigni, L.; Costa, G.; Rosinski, M.; Badziak, J.; Wołowski, J.; Zaraś-Szydłowska, A.; Parys, P. Protons accelerated in the target normal sheath acceleration regime by a femtosecond laser. Phys. Rev. Accel. Beams 2019, 22, 021302. [Google Scholar] [CrossRef]
- Cutroneo, M.; Havranek, V.; Mackova, A.; Malinsky, P.; Miksova, R.; Ceccio, G.; Ando’, L.; Michalcova, A. Overview of Polyethylene Terephthalate Foils Patterned Using 10 MeV Carbon Ions for Realization of Micromembranes. Micromachines 2023, 14, 284. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Yadav, T.; Sharma, S.; Nayak, A.; Kumari, A.A.; Mishra, N. Polymers in Drug Delivery. J. Biosci. Med. 2016, 4, 447–451. [Google Scholar] [CrossRef]
- Zhang, W.; Mehta, A.; Tong, Z.; Esser, L.; Voelcker, N.H. Development of Polymeric Nanoparticles for Blood–Brain Barrier Transfer—Strategies and Challenges. Adv. Sci. 2021, 8, 2003937. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.; Song, Z.; Wu, H.; Wang, X.; Feng, W.; Zhou, Z.; Zhang, H. Ion transport and structural design of lithium-ion conductive solid polymer electrolytes: A perspective. Mater. Futures 2022, 1, 042103. [Google Scholar] [CrossRef]
- Muthuraj, R.; Misra, M.; Mohanty, A.K. Biodegradable compatibilized polymer blends for packaging applications: A literature review. J. Appl. Polym. Sci. 2018, 135, 1–35. [Google Scholar] [CrossRef]
- Plastic Waste Management Global Market Report. 2022. Available online: https://www.reportlinker.com/p06319263/?utm_source=GNW (accessed on 12 September 2022).
- Krentsel, B.A.; Kissin, Y.V.; Kleiner, V.J.; Strotskava, L. Polymers and Co-Polymers of α-Olefins; Hanser/Gardner Pub.: Munich, Gemany, 1997. [Google Scholar]
- Lamonte, R.R.; McNaIIy, D. Cyclic olefin copolymer. Adv. Mater. Process. 2001, 159, 33–36. [Google Scholar]
- Bernard, M.; Jubeli, E.; Bakar, J.; Tortolano, L.; Saunier, J.; Yagoubi, N. Biocompatibility assessment of cyclic olefin copolymers: Impact of two additives on cytotoxicity, oxidative stress, inflammatory reactions and hemocompatibility. J. Biomed. Mater. Res. Part A 2017, 105, 3333–3349. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993; Biological Evaluation of Medical Devices. International Organization for Standardization, Various Dates. Parts 1–20. ISO: Geneva, Switzerland, 2009.
- ISO 10993-5; Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. ISO: Geneva, Switzerland, 2009.
- Visco, A.; Brancato, V.; Campo, N.; Torrisi, L.; Caridi, F.; Cutroneo, M. Modification in polyethylene–iron oxide joints induced by laser irradiation. Appl. Surf. Sci. 2013, 272, 99–103. [Google Scholar] [CrossRef]
- Cutroneo, M.; Torrisi, L.; Calcagno, L.; Torrisi, A. Characterization of thin films for TNSA laser irradiation. J. Phys. Conf. Ser. 2014, 508, 012012. [Google Scholar] [CrossRef]
- Ojeda-G-P, A.; Döbeli, M.; Lippert, T. Influence of Plume Properties on Thin Film Composition in Pulsed Laser Deposition. Adv. Mater. Interfaces 2018, 5, 1701062. [Google Scholar] [CrossRef]
- Lozovan, A.A.; Alexandrova, S.S. DropletPhase Reduction During the Pulsed Laser Deposition of Coatings onto the Inner Surface of Tubes. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2014, 8, 607–611. [Google Scholar] [CrossRef]
- Available online: https://www.microresist.de/ (accessed on 1 January 2022).
- Cutroneo, M.; Havranek, V.; Flaks, J.; Malinsky, P.; Torrisi, L.; Silipigni, L.; Slepicka, P.; Fajstavr, D.; Mackova, A. Pulsed laser deposition and laser-induced backward transfer to modify polydimethylsiloxane. Coatings 2021, 11, 1521. [Google Scholar] [CrossRef]
- Available online: https://litron.co.uk (accessed on 1 January 2022).
- Torrisi, L.; Cutroneo, M.; Silipigni, L.; Fazio, M. Effects of the laser irradiation on graphene oxide foils in vacuum and air. Phys. Solid State 2019, 61, 1327–1331. [Google Scholar] [CrossRef]
- Cutroneo, M.; Havranek, V.; Mackova, A.; Malinsky, P.; Torrisi, L.; Silipigni, L.; Fazio, B.; Torrisi, A.; Szokolova, K.; Sofer, Z.; et al. Effects of the ion bombardment on the structure and composition of GO and rGO foils. Mater. Chem. Phys. 2019, 232, 272–277. [Google Scholar] [CrossRef]
- Cutroneo, M.; Torrisi, A.; Ryukhtin, V.; Dopita, M.; Silipigni, L.; Mackova, A.; Malinsky, P.; Slepicka, P. Polydimethylsiloxane containing gold nanoparticles for optical applications. J. Instrum. 2020, 15, C03044. [Google Scholar] [CrossRef]
- Torrisi, L.; Cutroneo, M. Aluminium plasma production at high laser intensity. J. Appl. Phys. 2014, 115, 083105. [Google Scholar] [CrossRef]
- Kamaraj, A.B.; Shaw, V.; Sundaram, M.M. Novel Fabrication of Un-Coated Super-Hydrophobic Aluminum via Pulsed Electrochemical Surface Modification. Procedia Manuf. 2015, 1, 892–903. [Google Scholar] [CrossRef]
- Ang, L.K.; Lau, Y.Y.; Gilgenbach, R.M.; Spindler, H.L.; Lash, J.S.; Kovaleski, S.D. Surface instability of multipulse laser ablation on a metallic target. J. Appl. Phys. 1998, 83, 4466. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).